Mesenchymal Stem Cells Extract (MSCsE)-Based Therapy Alleviates Xerostomia and Keratoconjunctivitis Sicca in Sjogren’s Syndrome-Like Disease
Abstract
1. Introduction
2. Results
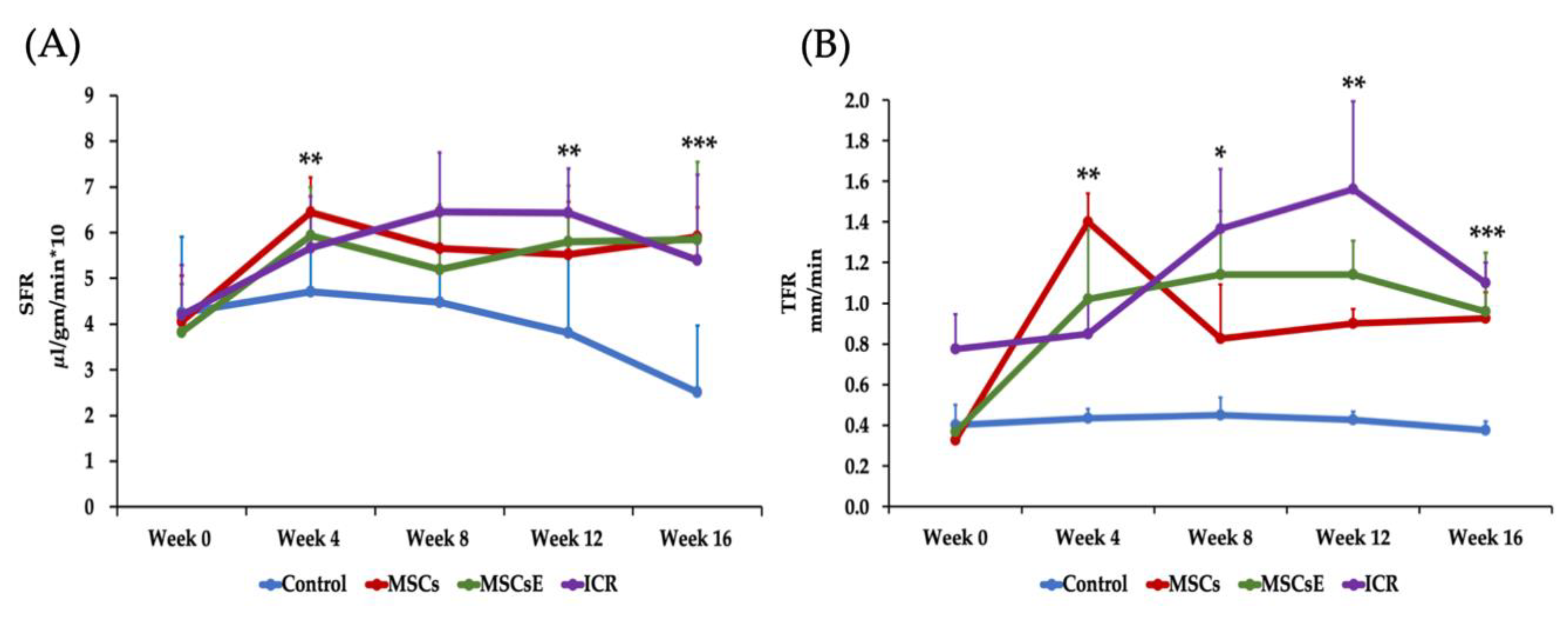
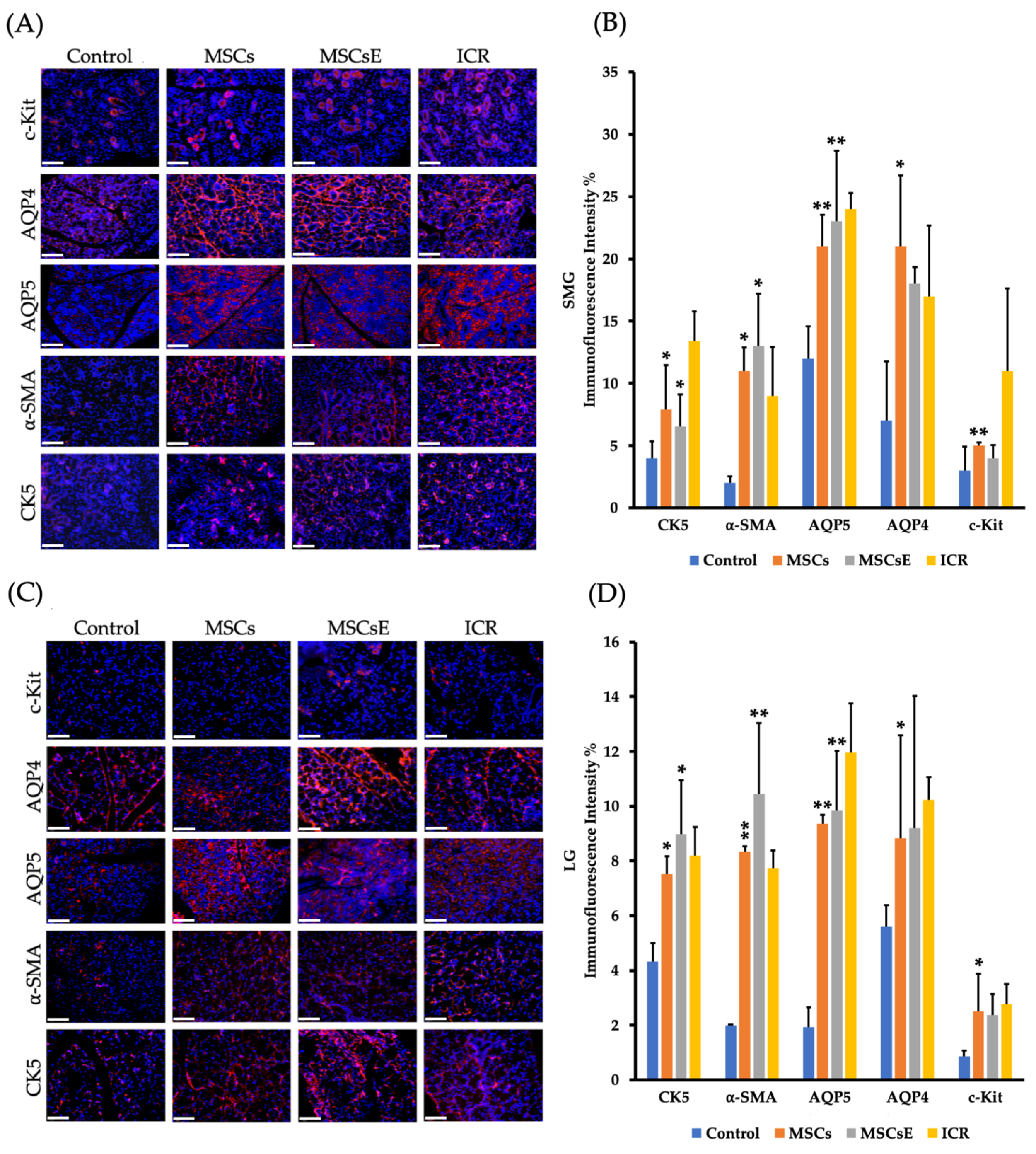
2.1. MSCs and MSCsE both Preserved Salivary and Lacrimal Gland Functions, Preserved Specialized Cells, and Upregulated Key Genes in the Gland Restoration
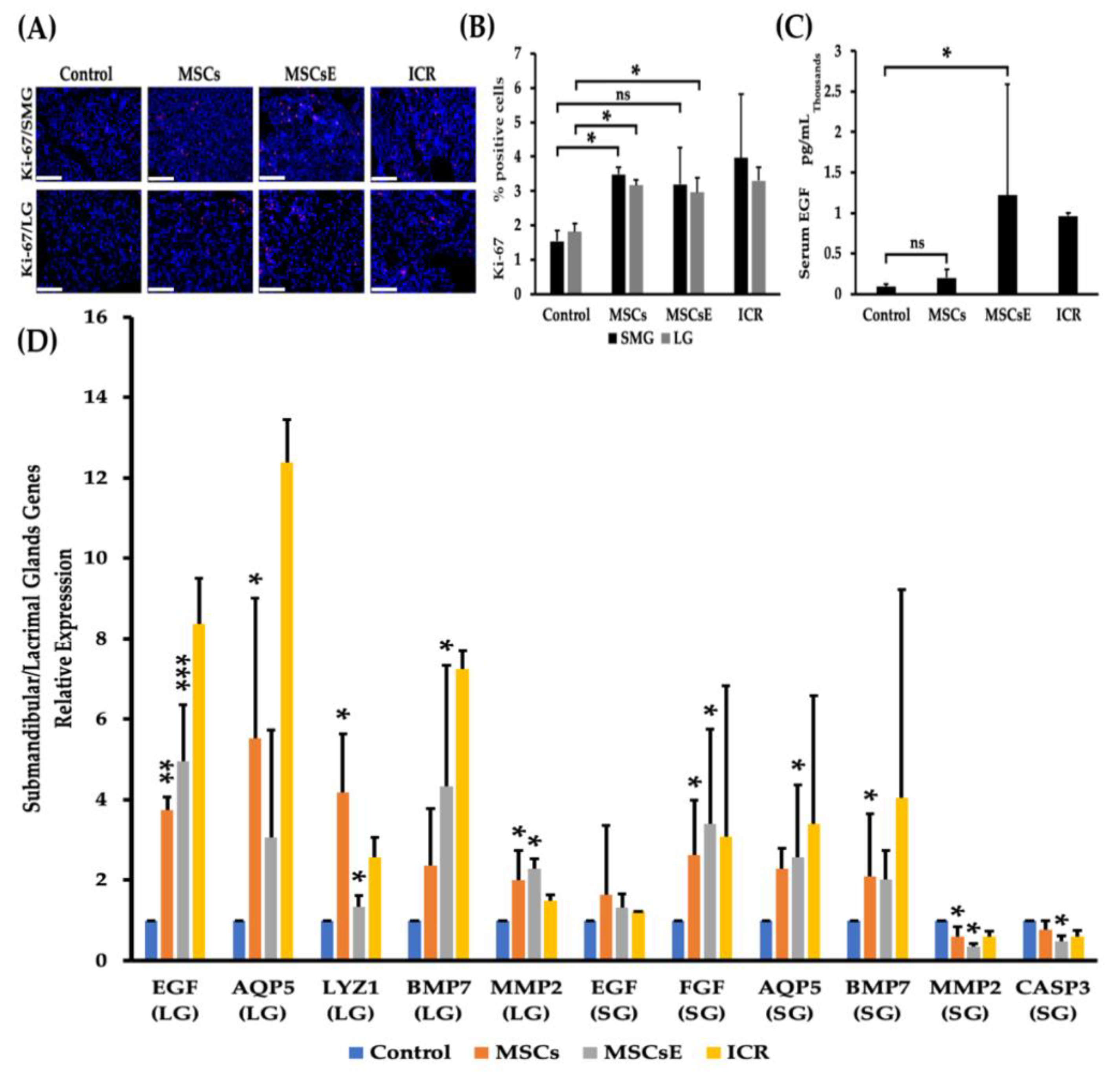
2.2. MSCs/MSCsE Treatments Promoted Proliferation, Elevated Systemic EGF Levels, and Modified Specific Key Genes in Glands Function, Proliferation, Regeneration, and Apoptosis
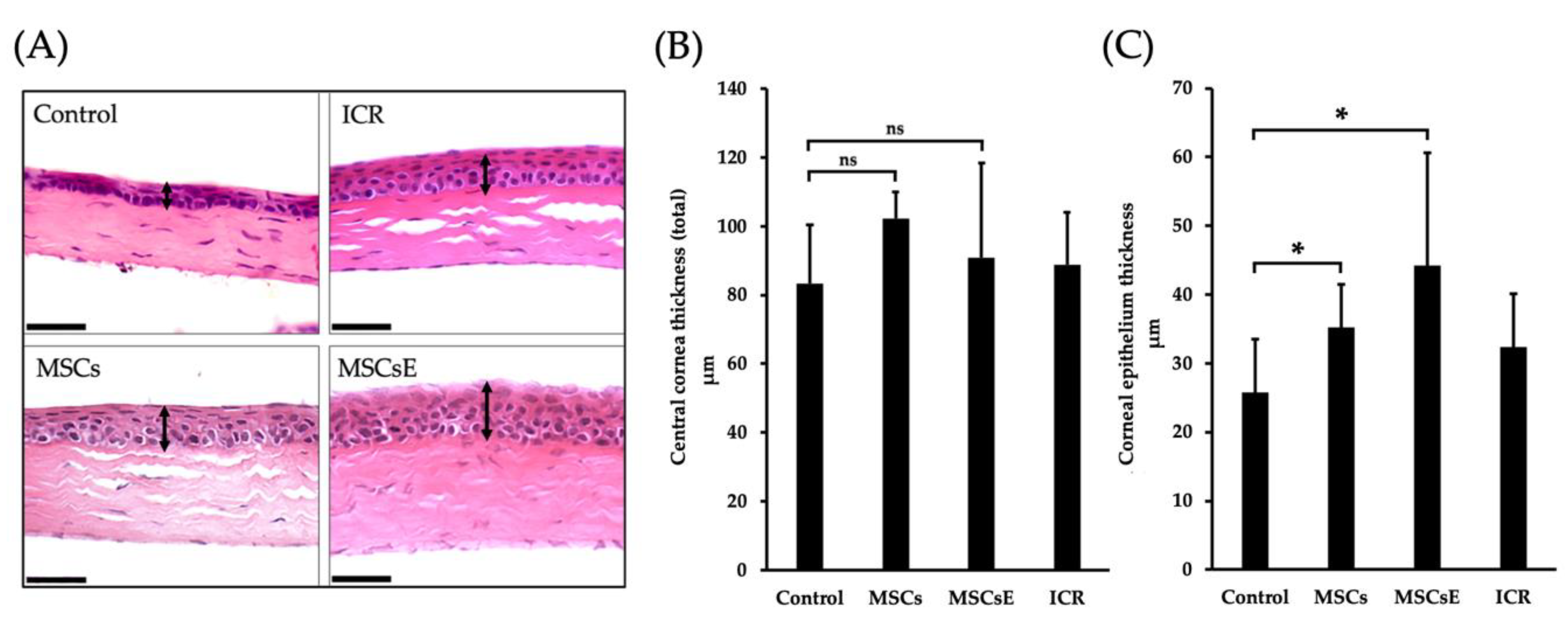
2.3. MSCs and MSCsE Protected the Cornea Integrity by Preserving its Epithelial Thickness
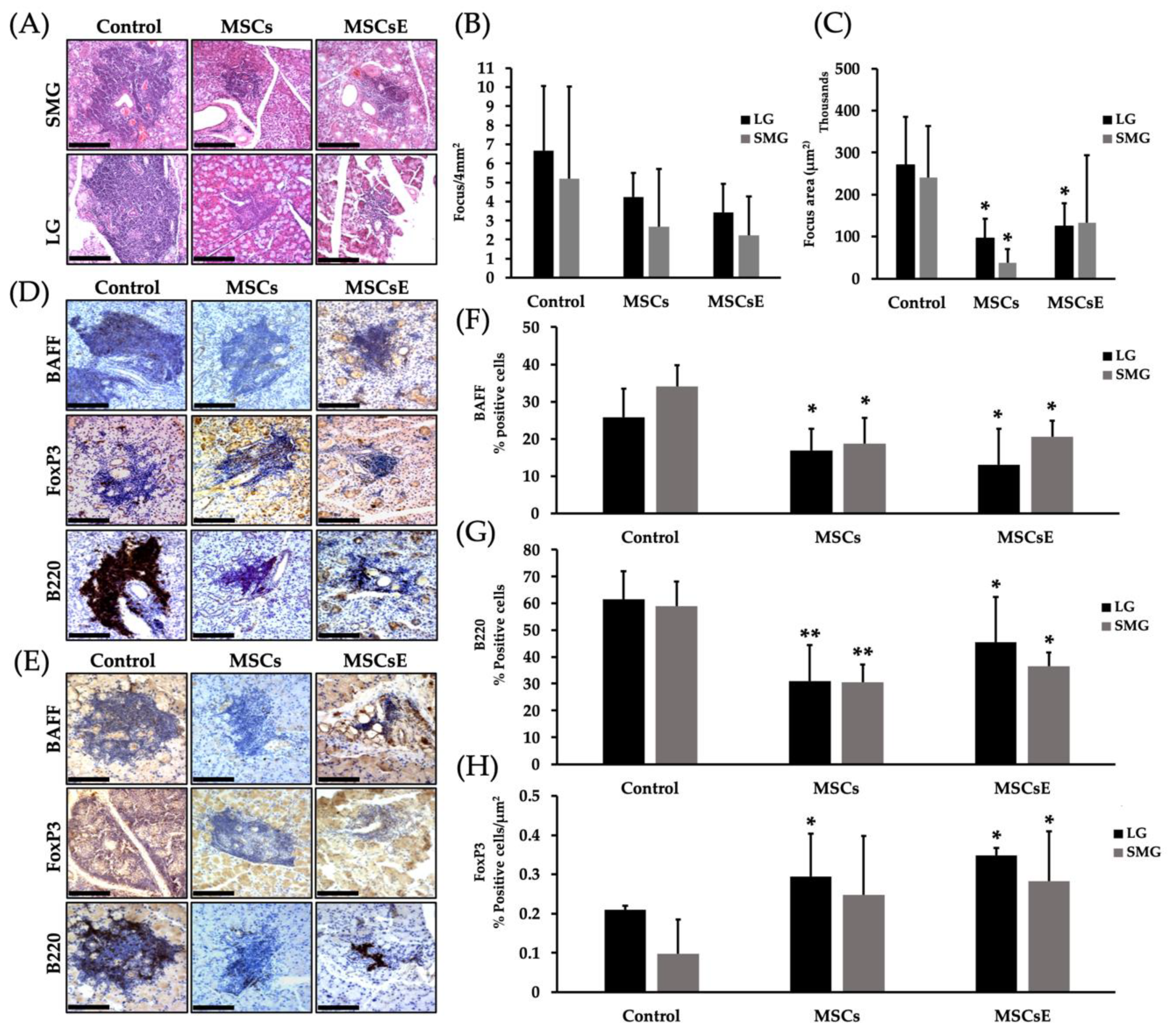
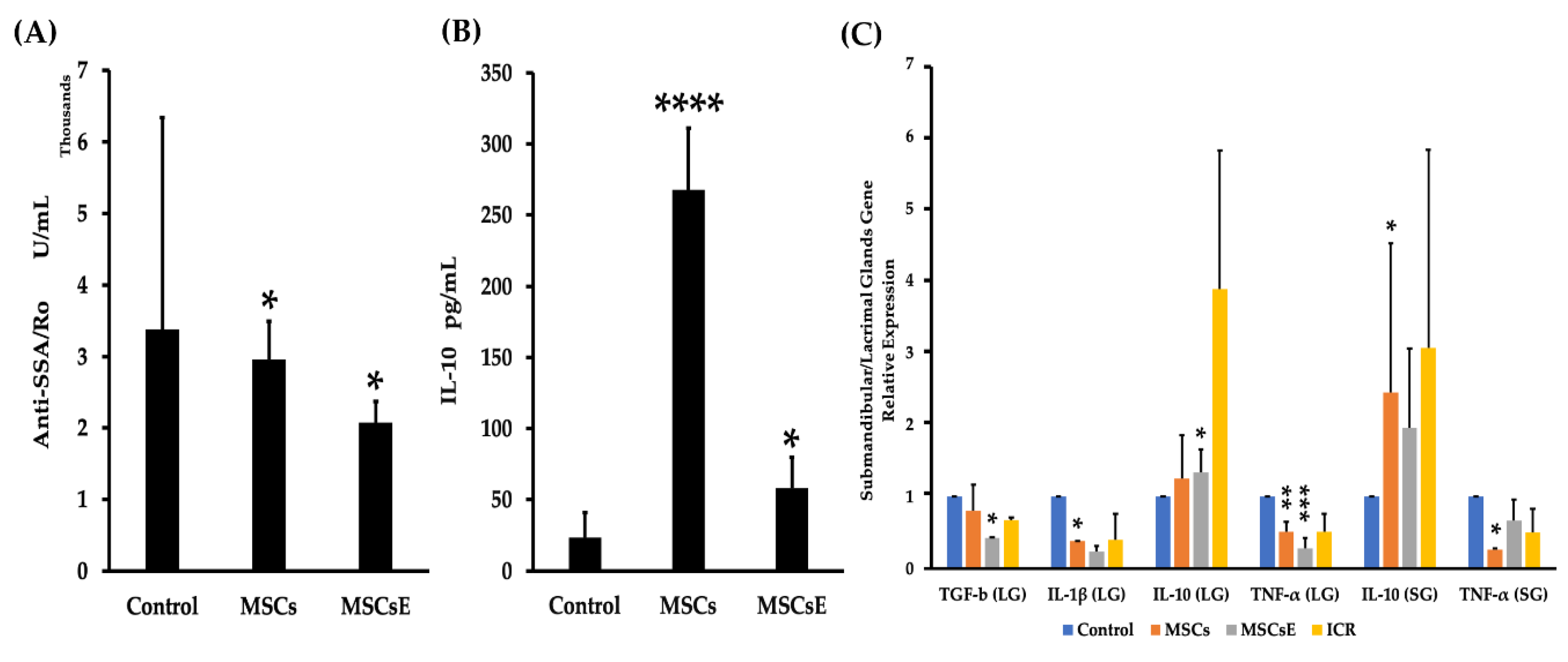
2.4. MSCs/MSCsE Immunomodulatory and Immunosuppressive Functions Were Evidenced by A Decrease in Lymphocytic Influx, A Selective Suppression of B Cells, An Upregulation of IL-10 Secretion and Its mRNA, A Down-Regulation of Gene Expression of Inflammatory Cytokines, and A Lower Levels of Anti-SSA/Ro Autoantibodies
3. Discussion
4. Materials and Methods
4.1. Animal Models
4.1.1. Recipient
4.1.2. Donors
4.1.3. Wild Type Control
4.2. Blood Glucose Monitoring
4.3. Mesenchymal Stem Cells (MSCs) Culture, Preparation of the Extract (MSCsE), and Their Transplantation
4.4. Secretory Function of the Salivary and Lacrimal Glands (Salivary Flow Rate: SFR and Tear Flow Rate: TFR)
4.5. Submandibular and Lacrimal Gland Tissue and Serum Analysis
4.5.1. Serum Preparation and Analysis
4.5.2. Focus Score and Focus Area
4.5.3. Immunohistochemistry
4.5.4. Immunofluorescence
4.5.5. Quantitative Real-Time PCR
4.5.6. Central Cornea Thickness Analysis
4.5.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fox, R.I.; Howell, F.V.; Bone, R.C.; Michelson, P. Primary Sjogren syndrome: Clinical and immunopathologic features. Semin. Arthritis Rheum. 1984, 14, 77–105. [Google Scholar] [CrossRef]
- Garcia-Carrasco, M.; Fuentes-Alexandro, S.; Escarcega, R.O.; Salgado, G.; Riebeling, C.; Cervera, R. Pathophysiology of Sjogren’s syndrome. Arch. Med. Res. 2006, 37, 921–932. [Google Scholar] [CrossRef] [PubMed]
- Wahren-Herlenius, M.; Dörner, T. Immunopathogenic mechanisms of systemic autoimmune disease. Lancet 2013, 382, 819–831. [Google Scholar] [CrossRef]
- Trontzas, P.I.; Andrianakos, A.A. Sjogren’s syndrome: A population based study of prevalence in Greece. The ESORDIG study. Ann. Rheum. Dis. 2005, 64, 1240–1241. [Google Scholar] [CrossRef] [PubMed]
- Alamanos, Y.; Tsifetaki, N.; Voulgari, P.V.; Venetsanopoulou, A.I.; Siozos, C.; Drosos, A.A. Epidemiology of primary Sjogren’s syndrome in north-west Greece, 1982–2003. Rheumatology 2006, 45, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Kabasakal, Y.; Kitapcioglu, G.; Turk, T.; Oder, G.; Durusoy, R.; Mete, N.; Egrilmez, S.; Akalin, T. The prevalence of Sjogren’s syndrome in adult women. Scand. J. Rheumatol. 2006, 35, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Birlik, M.; Akar, S.; Gurler, O.; Sari, I.; Birlik, B.; Sarioglu, S.; Oktem, M.A.; Saglam, F.; Can, G.; Kayahan, H.; et al. Prevalence of primary Sjogren’s syndrome in Turkey: A population-based epidemiological study. Int. J. Clin. Pract. 2009, 63, 954–961. [Google Scholar] [CrossRef]
- Anagnostopoulos, I.; Zinzaras, E.; Alexiou, I.; Papathanasiou, A.A.; Davas, E.; Koutroumpas, A.; Barouta, G.; Sakkas, L.I. The prevalence of rheumatic diseases in central Greece: A population survey. BMC Musculoskelet. Disord. 2010, 11, 98. [Google Scholar] [CrossRef]
- Goransson, L.G.; Haldorsen, K.; Brun, J.G.; Harboe, E.; Jonsson, M.V.; Skarstein, K.; Time, K.; Omdal, R. The point prevalence of clinically relevant primary Sjogren’s syndrome in two Norwegian counties. Scand. J. Rheumatol. 2011, 40, 221–224. [Google Scholar] [CrossRef]
- Maldini, C.; Seror, R.; Fain, O.; Dhote, R.; Amoura, Z.; De Bandt, M.; Delassus, J.L.; Falgarone, G.; Guillevin, L.; Le Guern, V.; et al. Epidemiology of primary Sjogren’s syndrome in a French multiracial/multiethnic area. Arthritis Care Res. 2014, 66, 454–463. [Google Scholar] [CrossRef]
- Bolstad, A.I.; Skarstein, K. Epidemiology of Sjogren’s Syndrome-from an Oral Perspective. Curr. Oral Health Rep. 2016, 3, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Kassan, S.S.; Moutsopoulos, H.M. Clinical manifestations and early diagnosis of Sjogren syndrome. Arch. Intern. Med. 2004, 164, 1275–1284. [Google Scholar] [CrossRef] [PubMed]
- Mitsias, D.I.; Kapsogeorgou, E.K.; Moutsopoulos, H.M. Sjogren’s syndrome: Why autoimmune epithelitis? Oral Dis. 2006, 12, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Kyriakidis, N.C.; Kapsogeorgou, E.K.; Tzioufas, A.G. A comprehensive review of autoantibodies in primary Sjogren’s syndrome: Clinical phenotypes and regulatory mechanisms. J. Autoimmun. 2014, 51, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.C.; Huynh, K.; Grubbs, J., Jr.; Davis, R.M. Autoimmunity in the pathogenesis and treatment of keratoconjunctivitis sicca. Curr. Allergy Asthma Rep. 2014, 14, 403. [Google Scholar] [CrossRef] [PubMed]
- Fox, R.I.; Stern, M. Sjogren’s syndrome: Mechanisms of pathogenesis involve interaction of immune and neurosecretory systems. Scand. J. Rheumatol. 2002, 116, 3–13. [Google Scholar] [CrossRef]
- Maslinska, M.; Przygodzka, M.; Kwiatkowska, B.; Sikorska-Siudek, K. Sjogren’s syndrome: Still not fully understood disease. Rheumatol. Int. 2015, 35, 233–241. [Google Scholar] [CrossRef]
- Scully, C. Sjogren’s syndrome: Clinical and laboratory features, immunopathogenesis, and management. Oral Surg. Oral Med. Oral Pathol. 1986, 62, 510–523. [Google Scholar] [CrossRef]
- Delaleu, N.; Jonsson, R.; Koller, M.M. Sjogren’s syndrome. Eur. J. Oral Sci. 2005, 113, 101–113. [Google Scholar] [CrossRef]
- Manthorpe, R.; Jacobsson, L.T. Sjogren’s syndrome. Bailliere’s Clin. Rheumatol. 1995, 9, 483–496. [Google Scholar] [CrossRef]
- Saraux, A.; Pers, J.O.; Devauchelle-Pensec, V. Treatment of primary Sjogren syndrome. Nat. Rev. Rheumatol. 2016, 12, 456–471. [Google Scholar] [CrossRef]
- Cartee, D.L.; Maker, S.; Dalonges, D.; Manski, M.C. Sjogren’s Syndrome: Oral Manifestations and Treatment, a Dental Perspective. J. Dent. Hyg. 2015, 89, 365–371. [Google Scholar] [PubMed]
- Carsons, S.E.; Vivino, F.B.; Parke, A.; Carteron, N.; Sankar, V.; Brasington, R.; Brennan, M.T.; Ehlers, W.; Fox, R.; Scofield, H.; et al. Treatment Guidelines for Rheumatologic Manifestations of Sjogren’s Syndrome: Use of Biologic Agents, Management of Fatigue, and Inflammatory Musculoskeletal Pain. Arthritis Care Res. 2017, 69, 517–527. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.J.; Carsons, S.E. Management of extraglandular manifestations of primary Sjogren’s syndrome. Oral Maxillofac. Surg. Clin. N. Am. 2014, 26, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Pinto, A. Management of xerostomia and other complications of Sjogren’s syndrome. Oral Maxillofac. Surg. Clin. N. Am. 2014, 26, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Martinez, G.; Zamora-Legoff, V.; Hernandez Molina, G. Oral health-related quality of life in primary Sjogren’s syndrome. Reumatol. Clin. 2018. [Google Scholar] [CrossRef]
- Ngo, D.Y.J.; Thomson, W.M.; Nolan, A.; Ferguson, S. The lived experience of Sjogren’s Syndrome. BMC Oral Health 2016, 16, 7. [Google Scholar] [CrossRef]
- Hammitt, K.M.; Naegeli, A.N.; van den Broek, R.W.M.; Birt, J.A. Patient burden of Sjogren’s: A comprehensive literature review revealing the range and heterogeneity of measures used in assessments of severity. RMD Open 2017, 3, e000443. [Google Scholar] [CrossRef]
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage potential of adult human mesenchymal stem cells. Science 1999, 284, 143–147. [Google Scholar] [CrossRef]
- Mammana, S.; Gugliandolo, A.; Cavalli, E.; Diomede, F.; Iori, R.; Zappacosta, R.; Bramanti, P.; Conti, P.; Fontana, A.; Pizzicannella, J.; et al. Human gingival mesenchymal stem cells pretreated with vesicular moringin nanostructures as a new therapeutic approach in a mouse model of spinal cord injury. J. Tissue Eng. Regen. Med. 2019. [Google Scholar] [CrossRef]
- Gebler, A.; Zabel, O.; Seliger, B. The immunomodulatory capacity of mesenchymal stem cells. Trends Mol. Med. 2012, 18, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Volkman, R.; Offen, D. Concise Review: Mesenchymal Stem Cells in Neurodegenerative Diseases. Stem Cells 2017, 35, 1867–1880. [Google Scholar] [CrossRef]
- Manimaran, K.; Sankaranarayanan, S.; Ravi, V.R.; Elangovan, S.; Chandramohan, M.; Perumal, S.M. Treatment of osteoradionecrosis of mandible with bone marrow concentrate and with dental pulp stem cells. Ann. Maxillofac. Surg. 2014, 4, 189–192. [Google Scholar] [CrossRef] [PubMed]
- Hass, R.; Kasper, C.; Böhm, S.; Jacobs, R. Different populations and sources of human mesenchymal stem cells (MSC): A comparison of adult and neonatal tissue-derived MSC. Cell Commun. Signal. 2011, 9, 12. [Google Scholar] [CrossRef] [PubMed]
- Freitag, J.; Wickham, J.; Shah, K.; Tenen, A. Effect of autologous adipose-derived mesenchymal stem cell therapy in the treatment of acromioclavicular joint osteoarthritis. BMJ Case Rep. 2019, 12. [Google Scholar] [CrossRef] [PubMed]
- Cuiffo, B.G.; Karnoub, A.E. Mesenchymal stem cells in tumor development: Emerging roles and concepts. Cell Adhes. Migr. 2012, 6, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, S.A.; Roobrouck, V.D.; Verfaillie, C.M.; Van Gool, S.W. Immunological characteristics of human mesenchymal stem cells and multipotent adult progenitor cells. Immunol. Cell Biol. 2013, 91, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Ryan, J.M.; Barry, F.P.; Murphy, J.M.; Mahon, B.P. Mesenchymal stem cells avoid allogeneic rejection. J. Inflamm. 2005, 2, 8. [Google Scholar] [CrossRef] [PubMed]
- Rota, C.; Morigi, M.; Imberti, B. Stem Cell Therapies in Kidney Diseases: Progress and Challenges. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef]
- Wang, J.; Sun, M.; Liu, W.; Li, Y.; Li, M. Stem Cell-Based Therapies for Liver Diseases: An Overview and Update. Tissue Eng. Regen. Med. 2019, 16, 107–118. [Google Scholar] [CrossRef]
- Barbado, J.; Tabera, S.; Sanchez, A.; Garcia-Sancho, J. Therapeutic potential of allogeneic mesenchymal stromal cells transplantation for lupus nephritis. Lupus 2018. [Google Scholar] [CrossRef] [PubMed]
- Canibano-Hernandez, A.; Saenz Del Burgo, L.; Espona-Noguera, A.; Ciriza, J.; Pedraz, J.L. Current advanced therapy cell-based medicinal products for type-1-diabetes treatment. Int. J. Pharm. 2018, 543, 107–120. [Google Scholar] [CrossRef] [PubMed]
- de Aguiar, C.F.; Castoldi, A.; Andrade-Oliveira, V.; Ignacio, A.; da Cunha, F.F.; Felizardo, R.J.F.; Bassi, E.J.; Camara, N.O.S.; de Almeida, D.C. Mesenchymal stromal cells modulate gut inflammation in experimental colitis. Inflammopharmacology 2018, 26, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Genc, B.; Bozan, H.R.; Genc, S.; Genc, K. Stem Cell Therapy for Multiple Sclerosis. Adv. Exp. Med. Biol. 2018. [Google Scholar] [CrossRef]
- Lu, Y.L.; Zhang, X.M. Experimental research on mesenchymal stem cells (MSCs) therapy for autoimmune uveitis. Chin. J. Ophthalmol. 2018, 54, 712–715. [Google Scholar] [CrossRef]
- Nasri, F.; Mohtasebi, M.S.; Hashemi, E.; Zarrabi, M.; Gholijani, N.; Sarvestani, E.K. Therapeutic Efficacy of Mesenchymal Stem Cells and Mesenchymal Stem Cells-derived Neural Progenitors in Experimental Autoimmune Encephalomyelitis. Int. J. Stem Cells 2018, 11, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Sevim, H.; Kocaefe, Y.C.; Onur, M.A.; Uckan-Cetinkaya, D.; Gurpinar, O.A. Bone marrow derived mesenchymal stem cells ameliorate inflammatory response in an in vitro model of familial hemophagocytic lymphohistiocytosis 2. Stem Cell Res. Ther. 2018, 9, 198. [Google Scholar] [CrossRef]
- Yang, Z.A.; Xia, B.Q.; Lv, H.X.; Zhen, E.M.; Cui, L.J.; Zhang, L. The curative effects of bone mesenchymal stem cells transplanted into rats with Sjogren’s syndrome. Shanghai J. Stomatol. 2011, 20, 152–158. [Google Scholar]
- Khalili, S.; Liu, Y.; Kornete, M.; Roescher, N.; Kodama, S.; Peterson, A.; Piccirillo, C.A.; Tran, S.D. Mesenchymal stromal cells improve salivary function and reduce lymphocytic infiltrates in mice with Sjogren’s-like disease. PLoS ONE 2012, 7, e38615. [Google Scholar] [CrossRef]
- Xu, J.; Wang, D.; Liu, D.; Fan, Z.; Zhang, H.; Liu, O.; Ding, G.; Gao, R.; Zhang, C.; Ding, Y.; et al. Allogeneic mesenchymal stem cell treatment alleviates experimental and clinical Sjogren syndrome. Blood 2012, 120, 3142–3151. [Google Scholar] [CrossRef]
- Aluri, H.S.; Samizadeh, M.; Edman, M.C.; Hawley, D.R.; Armaos, H.L.; Janga, S.R.; Meng, Z.; Sendra, V.G.; Hamrah, P.; Kublin, C.L.; et al. Delivery of Bone Marrow-Derived Mesenchymal Stem Cells Improves Tear Production in a Mouse Model of Sjogren’s Syndrome. Stem Cells Int. 2017, 2017, 3134543. [Google Scholar] [CrossRef] [PubMed]
- Elghanam, G.A.; Liu, Y.; Khalili, S.; Fang, D.; Tran, S.D. Compact Bone-Derived Multipotent Mesenchymal Stromal Cells (MSCs) for the Treatment of Sjogren’s-like Disease in NOD Mice. Methods Mol. Biol. 2017, 1553, 25–39. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Wang, X.; Nian, H.; Yang, D.; Wei, R. Mesenchymal stem cells for treating autoimmune dacryoadenitis. Stem Cell Res. Ther. 2017, 8, 126. [Google Scholar] [CrossRef] [PubMed]
- Ruan, G.F.; Zheng, L.; Huang, J.S.; Huang, W.X.; Gong, B.D.; Fang, X.X.; Zhang, X.Y.; Tang, J.P. Effect of mesenchymal stem cells on Sjogren-like mice and the microRNA expression profiles of splenic CD4+ T cells. Exp. Ther. Med. 2017, 13, 2828–2838. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Q.; Wang, Y.X.; Hua, H. Characteristics of Labial Gland Mesenchymal Stem Cells of Healthy Individuals and Patients with Sjogren’s Syndrome: A Preliminary Study. Stem Cells Dev. 2017, 26, 1171–1185. [Google Scholar] [CrossRef]
- Sun, L.; Akiyama, K.; Zhang, H.; Yamaza, T.; Hou, Y.; Zhao, S.; Xu, T.; Le, A.; Shi, S. Mesenchymal Stem Cell Transplantation Reverses Multi-Organ Dysfunction in Systemic Lupus Erythematosus Mice and Humans. Stem Cells 2009, 27, 1421–1432. [Google Scholar] [CrossRef] [PubMed]
- Pizzicannella, J.; Gugliandolo, A.; Orsini, T.; Fontana, A.; Ventrella, A.; Mazzon, E.; Bramanti, P.; Diomede, F.; Trubiani, O. Engineered Extracellular Vesicles From Human Periodontal-Ligament Stem Cells Increase VEGF/VEGFR2 Expression During Bone Regeneration. Front. Physiol. 2019, 10, 512. [Google Scholar] [CrossRef]
- Ghiroldi, A.; Piccoli, M.; Cirillo, F.; Monasky, M.M.; Ciconte, G.; Pappone, C.; Anastasia, L. Cell-Based Therapies for Cardiac Regeneration: A Comprehensive Review of Past and Ongoing Strategies. Int. J. Mol. Sci. 2018, 19. [Google Scholar] [CrossRef]
- Le Blanc, K.; Davies, L.C. MSCs—Cells with many sides. Cytotherapy 2018, 20, 273–278. [Google Scholar] [CrossRef]
- Uccelli, A.; Prockop, D.J. Why should mesenchymal stem cells (MSCs) cure autoimmune diseases? Curr. Opin. Immunol. 2010, 22, 768–774. [Google Scholar] [CrossRef]
- Cagliani, J.; Grande, D.; Molmenti, E.P.; Miller, E.J.; Rilo, H.L.R. Immunomodulation by Mesenchymal Stromal Cells and Their Clinical Applications. J. Stem Cell Regen. Biol. 2017, 3. [Google Scholar] [CrossRef]
- Yi, T.; Song, S.U. Immunomodulatory properties of mesenchymal stem cells and their therapeutic applications. Arch. Pharmacal Res. 2012, 35, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Meisel, R.; Zibert, A.; Laryea, M.; Gobel, U.; Daubener, W.; Dilloo, D. Human bone marrow stromal cells inhibit allogeneic T-cell responses by indoleamine 2,3-dioxygenase-mediated tryptophan degradation. Blood 2004, 103, 4619–4621. [Google Scholar] [CrossRef] [PubMed]
- Alunno, A.; Montanucci, P.; Bistoni, O.; Basta, G.; Caterbi, S.; Pescara, T.; Pennoni, I.; Bini, V.; Bartoloni, E.; Gerli, R.; et al. In vitro immunomodulatory effects of microencapsulated umbilical cord Wharton jelly-derived mesenchymal stem cells in primary Sjogren’s syndrome. Rheumatology 2015, 54, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.X.; Wang, J.; Wen, Y.B.; Fei, Y.Y.; Jiang, M.D.; Zhou, M.Y.; Zhang, W.; Li, H.; Li, X.M.; Zhang, F.C.; et al. Renal involvement in primary Sjogren’s syndrome: A retrospective study of 103 biopsy-proven cases from a single center in China. Int. J. Rheum. Dis. 2018, 21, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Di Nicola, M.; Carlo-Stella, C.; Magni, M.; Milanesi, M.; Longoni, P.D.; Matteucci, P.; Grisanti, S.; Gianni, A.M. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood 2002, 99, 3838–3843. [Google Scholar] [CrossRef]
- Zhou, K.; Zhang, H.; Jin, O.; Feng, X.; Yao, G.; Hou, Y.; Sun, L. Transplantation of human bone marrow mesenchymal stem cell ameliorates the autoimmune pathogenesis in MRL/lpr mice. Cell. Mol. Immunol. 2008, 5, 417–424. [Google Scholar] [CrossRef]
- Norozi, F.; Ahmadzadeh, A.; Shahrabi, S.; Vosoughi, T.; Saki, N. Mesenchymal stem cells as a double-edged sword in suppression or progression of solid tumor cells. Tumour Biol. 2016, 37, 11679–11689. [Google Scholar] [CrossRef]
- Clément, F.; Grockowiak, E.; Zylbersztejn, F.; Fossard, G.; Gobert, S.; Maguer-Satta, V. Stem cell manipulation, gene therapy and the risk of cancer stem cell emergence. Stem Cell Investig. 2017, 4, 67. [Google Scholar] [CrossRef]
- Fang, D.; Hu, S.; Liu, Y.; Quan, V.H.; Seuntjens, J.; Tran, S.D. Identification of the active components in Bone Marrow Soup: A mitigator against irradiation-injury to salivary glands. Sci. Rep. 2015, 5, 16017. [Google Scholar] [CrossRef]
- Arellano-Garcia, M.E.; Misuno, K.; Tran, S.D.; Hu, S. Interferon-gamma induces immunoproteasomes and the presentation of MHC I-associated peptides on human salivary gland cells. PLoS ONE 2014, 9, e102878. [Google Scholar] [CrossRef] [PubMed]
- Yeghiazarians, Y.; Zhang, Y.; Prasad, M.; Shih, H.; Saini, S.A.; Takagawa, J.; Sievers, R.E.; Wong, M.L.; Kapasi, N.K.; Mirsky, R.; et al. Injection of bone marrow cell extract into infarcted hearts results in functional improvement comparable to intact cell therapy. Mol. Ther. 2009, 17, 1250–1256. [Google Scholar] [CrossRef] [PubMed]
- Bullwinkel, J.; Baron-Luhr, B.; Ludemann, A.; Wohlenberg, C.; Gerdes, J.; Scholzen, T. Ki-67 protein is associated with ribosomal RNA transcription in quiescent and proliferating cells. J. Cell. Physiol. 2006, 206, 624–635. [Google Scholar] [CrossRef] [PubMed]
- Villani, E.; Galimberti, D.; Viola, F.; Mapelli, C.; Ratiglia, R. The cornea in Sjogren’s syndrome: An in vivo confocal study. Investig. Ophthalmol. Vis. Sci. 2007, 48, 2017–2022. [Google Scholar] [CrossRef] [PubMed]
- Rosenblum, M.D.; Gratz, I.K.; Paw, J.S.; Abbas, A.K. Treating human autoimmunity: Current practice and future prospects. Sci. Transl. Med. 2012, 4, 125sr121. [Google Scholar] [CrossRef]
- Khalili, S.; Faustman, D.L.; Liu, Y.; Sumita, Y.; Blank, D.; Peterson, A.; Kodama, S.; Tran, S.D. Treatment for salivary gland hypofunction at both initial and advanced stages of Sjogren-like disease: A comparative study of bone marrow therapy versus spleen cell therapy with a 1-year monitoring period. Cytotherapy 2014, 16, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Misuno, K.; Tran, S.D.; Khalili, S.; Huang, J.; Liu, Y.; Hu, S. Quantitative analysis of protein and gene expression in salivary glands of Sjogren’s-like disease NOD mice treated by bone marrow soup. PLoS ONE 2014, 9, e87158. [Google Scholar] [CrossRef]
- Alvarez-Viejo, M.; Menendez-Menendez, Y.; Blanco-Gelaz, M.A.; Ferrero-Gutierrez, A.; Fernandez-Rodriguez, M.A.; Gala, J.; Otero-Hernandez, J. Quantifying mesenchymal stem cells in the mononuclear cell fraction of bone marrow samples obtained for cell therapy. Transplant. Proc. 2013, 45, 434–439. [Google Scholar] [CrossRef]
- Vasil’ev, V.I.; Simonova, M.V.; Safonova, T.N.; Mach, E.S. Comparative evaluation of the treatment of Sjogren’s syndrome with anti-rheumatic preparations. Ter. Arkhiv 1988, 60, 67–72. [Google Scholar]
- English, K.; Wood, K.J. Mesenchymal stromal cells in transplantation rejection and tolerance. Cold Spring Harb. Perspect. Med. 2013, 3, a015560. [Google Scholar] [CrossRef]
- Su, X.; Fang, D.; Liu, Y.; Ruan, G.; Seuntjens, J.; Kinsella, J.M.; Tran, S.D. Lyophilized bone marrow cell extract functionally restores irradiation-injured salivary glands. Oral Dis. 2018, 24, 202–206. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.H.; Tudares, M.A.; Nguyen, C.Q. Sjogren’s syndrome: An old tale with a new twist. Arch. Immunol. Ther. Exp. 2009, 57, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Toma, C.; Pittenger, M.F.; Cahill, K.S.; Byrne, B.J.; Kessler, P.D. Human mesenchymal stem cells differentiate to a cardiomyocyte phenotype in the adult murine heart. Circulation 2002, 105, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Rani, S.; Ryan, A.E.; Griffin, M.D.; Ritter, T. Mesenchymal Stem Cell-derived Extracellular Vesicles: Toward Cell-free Therapeutic Applications. Mol. Ther. 2015, 23, 812–823. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, G.I.; Augello, A.; De Bari, C. Role of mesenchymal stem cells in reestablishing immunologic tolerance in autoimmune rheumatic diseases. Arthritis Rheum. 2011, 63, 2547–2557. [Google Scholar] [CrossRef] [PubMed]
- Soyfoo, M.S.; Chivasso, C.; Perret, J.; Delporte, C. Involvement of Aquaporins in the Pathogenesis, Diagnosis and Treatment of Sjogren’s Syndrome. Int. J. Mol. Sci. 2018, 19. [Google Scholar] [CrossRef]
- Delporte, C. Aquaporins in secretory glands and their role in Sjogren’s syndrome. Handb. Exp. Pharmacol. 2009, 185–201. [Google Scholar] [CrossRef]
- Delporte, C. Aquaporins and Gland Secretion. Adv. Exp. Med. Biol. 2017, 969, 63–79. [Google Scholar] [CrossRef]
- Ohashi, Y.; Tsuzaka, K.; Takeuchi, T.; Sasaki, Y.; Tsubota, K. Altered distribution of aquaporin 5 and its C-terminal binding protein in the lacrimal glands of a mouse model for Sjogren’s syndrome. Curr. Eye Res. 2008, 33, 621–629. [Google Scholar] [CrossRef]
- Hirai, S.; Ishida, N.; Watanabe, K.; Mita, S. Leakage of aquaporin 5 in the tear of dacryoadenitis mice. Investig. Ophthalmol. Vis. Sci. 2000, 41, 2432–2437. [Google Scholar]
- Ohashi, Y.; Ishida, R.; Kojima, T.; Goto, E.; Matsumoto, Y.; Watanabe, K.; Ishida, N.; Nakata, K.; Takeuchi, T.; Tsubota, K. Abnormal protein profiles in tears with dry eye syndrome. Am. J. Ophthalmol. 2003, 136, 291–299. [Google Scholar] [CrossRef]
- Lombaert, I.M.; Knox, S.M.; Hoffman, M.P. Salivary gland progenitor cell biology provides a rationale for therapeutic salivary gland regeneration. Oral Dis. 2011, 17, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Nanduri, L.S.; Lombaert, I.M.F.; van der Zwaag, M.; Faber, H.; Brunsting, J.F.; van Os, R.P.; Coppes, R.P. Salisphere derived c-Kit+ cell transplantation restores tissue homeostasis in irradiated salivary gland. Radiother. Oncol. 2013, 108, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Miettinen, M.; Lasota, J. KIT (CD117): A review on expression in normal and neoplastic tissues, and mutations and their clinicopathologic correlation. Appl. Immunohistochem. Mol. Morphol. 2005, 13, 205–220. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, S.; Nair, R.M.; Vamadevan, P.; Ali, M.J.; Naik, M.N.; Honavar, S.G.; Vemuganti, G.K. Establishing and characterizing lacrispheres from human lacrimal gland for potential clinical application. Graefe’s Arch. Clin. Exp. Ophthalmol. 2018, 256, 717–727. [Google Scholar] [CrossRef] [PubMed]
- Muskhelishvili, L.; Latendresse, J.R.; Kodell, R.L.; Henderson, E.B. Evaluation of Cell Proliferation in Rat Tissues with BrdU, PCNA, Ki-67 (MIB-5) Immunohistochemistry and In Situ Hybridization for Histone mRNA. J. Histochem. Cytochem. 2003, 51, 1681–1688. [Google Scholar] [CrossRef] [PubMed]
- Aho, H.J.; Saari, K.M.; Kallajoki, M.; Nevalainen, T.J. Synthesis of group II phospholipase A2 and lysozyme in lacrimal glands. Investig. Ophthalmol. Vis. Sci. 1996, 37, 1826–1832. [Google Scholar]
- Edman, M.; Marchelletta, R.; Hamm-Alvarez, S. Lacrimal gland overview. In Ocular Periphery and Disorders; Dartt, D., Bex, P., D’amore, P., Dana, R., Mcloon, L., Niederkorn, J., Eds.; Elsevier/Academic Press: Amsterdam, The Netherlands, 2011; pp. 68–73. [Google Scholar]
- Vivino, F.B.; Minerva, P.; Huang, C.H.; Orlin, S.E. Corneal melt as the initial presentation of primary Sjogren’s syndrome. J. Rheumatol. 2001, 28, 379–382. [Google Scholar]
- Vivino, F.B.; Carsons, S.E.; Foulks, G.; Daniels, T.E.; Parke, A.; Brennan, M.T.; Forstot, S.L.; Scofield, R.H.; Hammitt, K.M. New Treatment Guidelines for Sjogren’s Disease. Rheum. Dis. Clin. N. Am. 2016, 42, 531–551. [Google Scholar] [CrossRef]
- Ali, N.M.; Hamied, F.M.; Farhood, Q.K. Corneal thickness in dry eyes in an Iraqi population. Clin. Ophthalmol. 2017, 11, 435–440. [Google Scholar] [CrossRef]
- Costa, S.; Schutz, S.; Cornec, D.; Uguen, A.; Quintin-Roue, I.; Lesourd, A.; Berthelot, J.M.; Hachulla, E.; Hatron, P.Y.; Goeb, V.; et al. B-cell and T-cell quantification in minor salivary glands in primary Sjogren’s syndrome: Development and validation of a pixel-based digital procedure. Arthritis Res. Ther. 2016, 18, 21. [Google Scholar] [CrossRef] [PubMed]
- Rosado, M.M.; Bernardo, M.E.; Scarsella, M.; Conforti, A.; Giorda, E.; Biagini, S.; Cascioli, S.; Rossi, F.; Guzzo, I.; Vivarelli, M.; et al. Inhibition of B-cell proliferation and antibody production by mesenchymal stromal cells is mediated by T cells. Stem Cells Dev. 2015, 24, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Szodoray, P.; Jonsson, R. The BAFF/APRIL system in systemic autoimmune diseases with a special emphasis on Sjogren’s syndrome. Scand. J. Immunol. 2005, 62, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Mariette, X.; Roux, S.; Zhang, J.; Bengoufa, D.; Lavie, F.; Zhou, T.; Kimberly, R. The level of BLyS (BAFF) correlates with the titre of autoantibodies in human Sjogren’s syndrome. Ann. Rheum. Dis. 2003, 62, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Hompson, N.; Isenberg, D.A.; Jury, E.C.; Ciurtin, C. Exploring BAFF: Its expression, receptors and contribution to the immunopathogenesis of Sjogren’s syndrome. Rheumatology 2016, 55, 1548–1555. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, S.; Nakano, S.; Watanabe, T.; Tamayama, Y.; Mitsuo, A.; Nakiri, Y.; Suzuki, J.; Nozawa, K.; Amano, H.; Tokano, Y.; et al. Expression of B-cell activating factor of the tumour necrosis factor family (BAFF) in T cells in active systemic lupus erythematosus: The role of BAFF in T cell-dependent B cell pathogenic autoantibody production. Rheumatology 2007, 46, 1083–1086. [Google Scholar] [CrossRef] [PubMed]
- Fox, R.I.; Kang, H.I.; Ando, D.; Abrams, J.; Pisa, E. Cytokine mRNA expression in salivary gland biopsies of Sjogren’s syndrome. J. Immunol. 1994, 152, 5532–5539. [Google Scholar] [PubMed]
- Yamano, S.; Atkinson, J.C.; Baum, B.J.; Fox, P.C. Salivary gland cytokine expression in NOD and normal BALB/c mice. Clin. Immunol. 1999, 92, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Spees, J.L.; Lee, R.H.; Gregory, C.A. Mechanisms of mesenchymal stem/stromal cell function. Stem Cell Res. Ther. 2016, 7, 125. [Google Scholar] [CrossRef]
- Esensten, J.H.; Muller, Y.D.; Bluestone, J.A.; Tang, Q. Regulatory T-cell therapy for autoimmune and autoinflammatory diseases: The next frontier. J. Allergy Clin. Immunol. 2018, 142, 1710–1718. [Google Scholar] [CrossRef]
- Rubtsov, Y.P.; Rasmussen, J.P.; Chi, E.Y.; Fontenot, J.; Castelli, L.; Ye, X.; Treuting, P.; Siewe, L.; Roers, A.; Henderson, W.R., Jr.; et al. Regulatory T cell-derived interleukin-10 limits inflammation at environmental interfaces. Immunity 2008, 28, 546–558. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, S.; Yamaguchi, T.; Nomura, T.; Ono, M. Regulatory T cells and immune tolerance. Cell 2008, 133, 775–787. [Google Scholar] [CrossRef] [PubMed]
- Battaglia, M.; Gianfrani, C.; Gregori, S.; Roncarolo, M.G. IL-10-producing T regulatory type 1 cells and oral tolerance. Ann. N. Y. Acad Sci. 2004, 1029, 142–153. [Google Scholar] [CrossRef] [PubMed]
- Saxena, A.; Khosraviani, S.; Noel, S.; Mohan, D.; Donner, T.; Hamad, A.R. Interleukin-10 paradox: A potent immunoregulatory cytokine that has been difficult to harness for immunotherapy. Cytokine 2015, 74, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Walter, M.R. The molecular basis of IL-10 function: From receptor structure to the onset of signaling. Curr. Top. Microbiol. Immunol. 2014, 380, 191–212. [Google Scholar] [CrossRef] [PubMed]
- Kyurkchiev, D.; Bochev, I.; Ivanova-Todorova, E.; Mourdjeva, M.; Oreshkova, T.; Belemezova, K.; Kyurkchiev, S. Secretion of immunoregulatory cytokines by mesenchymal stem cells. World J. Stem Cells 2014, 6, 552–570. [Google Scholar] [CrossRef]
- Aggarwal, S.; Pittenger, M.F. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 2005, 105, 1815–1822. [Google Scholar] [CrossRef]
- Hall, B.E.; Zheng, C.; Swaim, W.D.; Cho, A.; Nagineni, C.N.; Eckhaus, M.A.; Flanders, K.C.; Ambudkar, I.S.; Baum, B.J.; Kulkarni, A.B. Conditional overexpression of TGF-beta1 disrupts mouse salivary gland development and function. Lab. Investig. 2010, 90, 543–555. [Google Scholar] [CrossRef] [PubMed]
- Biernacka, A.; Dobaczewski, M.; Frangogiannis, N.G. TGF-β signaling in fibrosis. Growth Factors 2011, 29, 196–202. [Google Scholar] [CrossRef]
- Park, J.H.; Hwang, I.; Hwang, S.H.; Han, H.; Ha, H. Human umbilical cord blood-derived mesenchymal stem cells prevent diabetic renal injury through paracrine action. Diabetes Res. Clin. Pract. 2012, 98, 465–473. [Google Scholar] [CrossRef]
- Kim, J.E.; Nam, J.H.; Cho, J.Y.; Kim, K.S.; Hwang, D.Y. Annual tendency of research papers used ICR mice as experimental animals in biomedical research fields. Lab. Anim. Res. 2017, 33, 171–178. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abughanam, G.; Elkashty, O.A.; Liu, Y.; Bakkar, M.O.; Tran, S.D. Mesenchymal Stem Cells Extract (MSCsE)-Based Therapy Alleviates Xerostomia and Keratoconjunctivitis Sicca in Sjogren’s Syndrome-Like Disease. Int. J. Mol. Sci. 2019, 20, 4750. https://doi.org/10.3390/ijms20194750
Abughanam G, Elkashty OA, Liu Y, Bakkar MO, Tran SD. Mesenchymal Stem Cells Extract (MSCsE)-Based Therapy Alleviates Xerostomia and Keratoconjunctivitis Sicca in Sjogren’s Syndrome-Like Disease. International Journal of Molecular Sciences. 2019; 20(19):4750. https://doi.org/10.3390/ijms20194750
Chicago/Turabian StyleAbughanam, Ghada, Osama A. Elkashty, Younan Liu, Mohammed O. Bakkar, and Simon D. Tran. 2019. "Mesenchymal Stem Cells Extract (MSCsE)-Based Therapy Alleviates Xerostomia and Keratoconjunctivitis Sicca in Sjogren’s Syndrome-Like Disease" International Journal of Molecular Sciences 20, no. 19: 4750. https://doi.org/10.3390/ijms20194750
APA StyleAbughanam, G., Elkashty, O. A., Liu, Y., Bakkar, M. O., & Tran, S. D. (2019). Mesenchymal Stem Cells Extract (MSCsE)-Based Therapy Alleviates Xerostomia and Keratoconjunctivitis Sicca in Sjogren’s Syndrome-Like Disease. International Journal of Molecular Sciences, 20(19), 4750. https://doi.org/10.3390/ijms20194750




