Insights into the Role of MicroRNAs in the Onset and Development of Diabetic Neuropathy
Abstract
1. Types of Diabetes
2. Diabetes Complications
3. MicroRNAs and Diabetic Neuropathy
3.1. Diabetic Neuropathy
3.2. Functions of MicroRNAs
3.3. Role of MiRNAs in the Nervous System
3.4. MiRNAs Involved in the Pathogenesis of DN
3.5. MiRNAs Contribute in Animal Models of DN
3.6. Human Studies of MicroRNAs in DN
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| miRNAs | microRNAs |
| DM | Diabetes mellitus |
| T1D | Type 1 diabetes |
| HLA | Human leukocyte antigen |
| T2D | Type 2 diabetes |
| IR | Insulin resistance |
| ADA | American Diabetes Association |
| ED | Endothelial dysfunction |
| CVD | Cardiovascular diseases |
| PN | Peripheral neuropathy |
| NO | Nitric oxide |
| TGF | Transforming growth factor |
| DN | Diabetic neuropathy |
| DSPN | Distal symmetric polyneuropathy |
| DPN | Diabetic peripheral neuropathy |
| STZ | Streptozotocin |
| miRs | microRNAs |
| RNAi | RNA interference |
| AD | Alzheimer’s disease |
| PD | Parkinson’s disease |
| NF-κB | Nuclear factor κB |
| MCP | Monocyte chemoattractant protein |
| PTEN | Phosphatase and tensin homologue |
| VEGF | Vascular endothelial growth factor |
| IL | Interleukin 2 |
| IKAK 1 | Interleukin-1 receptor-associated kinase 1 |
| TRAF6 | Tumor necrosis factor receptor-associated kinase 1 |
| ADAMTS3 | A disintegrin and metalloproteinase with thrombospondin motifs 3 |
| 12/15-LOX | 12/15 lipoxygenase |
| DRG | Dorsal root ganglia |
| DH | Dorsal horn |
| CALHM1 | Calcium homeostasis modulator 1 |
| P2X7 | Purinergic receptor P2X ligand gated ion channel 7 |
| Podxl | Podocalyxin-like |
| Hoxa1 | Homeobox A1 |
| SLC17A6 | Solute carrier family 17A6 |
| VGLUT2 | Vesicular-glutamate transporter 2 |
| rev | Reviewed |
| EVs | Extracellular vesicles |
| NDNMT3A | DNA target proteins methyltransferase-3a |
| NUMB | Endocytic adaptor protein |
| SNAP25 | Synaptosome associated protein 25 |
| GAP43 | Growth-associated protein-43 |
| HG | High glucose |
| PRKCI | Protein kinase C iota |
| SMAD | Small mother against decapentaplegic |
| CNS | Central nervous system |
| SNPs | Single nucleotide polymorphisms |
| CAN | Cardiovascular autonomic neuropathy |
| SERPINE2 | Serine protease inhibitor E2 |
| DEGs | Differentially expressed genes |
| PPAR | Peroxisome proliferator-activated receptor |
| AMP | Adenosine monophosphate |
| AMPK | AMP-activated protein kinase |
| PPARG | PPAR gamma |
| SCD | Stearoyl-CoA desaturase |
| CD36 | Cluster of differentiation 36 |
| PCK1 | Phosphoenolpyruvate carboxykinase 1 |
| ANXA9 | Annexin A9 |
| ANGPTL4 | Angiopoietin like 4 |
| CHI3L2 | Chitinase 3 Like 2 |
| ADRBK2 | Adrenoceptor Beta 2 |
| CSN1S1 | Casein Alpha S1 |
| GALM | Galactose mutarotase |
| EN1 | Homeobox protein engrailed-1 |
| FAM89A | Family with sequence similarity 89 member A |
References
- Atkinson, M.A.; Eisenbarth, G.S.; Michels, A.W. Type 1 diabetes. Lancet 2014, 383, 69–82. [Google Scholar] [CrossRef]
- In’t Veld, P. Insulitis in human type 1 diabetes: The quest for an elusive lesion. Islets 2011, 3, 131–138. [Google Scholar] [CrossRef]
- Gregg, B.E.; Moore, P.C.; Demozay, D.; Hall, B.A.; Li, M.; Husain, A.; Wright, A.J.; Atkinson, M.A.; Rhodes, C.J. Formation of a human beta-cell population within pancreatic islets is set early in life. J. Clin. Endocrinol. Metab. 2012, 97, 3197–3206. [Google Scholar] [CrossRef]
- Keenan, H.A.; Sun, J.K.; Levine, J.; Doria, A.; Aiello, L.P.; Eisenbarth, G.; Bonner-Weir, S.; King, G.L. Residual insulin production and pancreatic ss-cell turnover after 50 years of diabetes: Joslin Medalist Study. Diabetes 2010, 59, 2846–2853. [Google Scholar] [CrossRef]
- Cohen, A.; Horton, E.S. Progress in the treatment of type 2 diabetes: New pharmacologic approaches to improve glycemic control. Curr. Med. Res. Opin. 2007, 23, 905–917. [Google Scholar] [CrossRef]
- Yaribeygi, H.; Farrokhi, F.R.; Butler, A.E.; Sahebkar, A. Insulin resistance: Review of the underlying molecular mechanisms. J. Cell Physiol. 2019, 234, 8152–8161. [Google Scholar] [CrossRef]
- Nathan, D.M. Long-term complications of diabetes mellitus. N. Engl. J. Med. 1993, 328, 1676–1685. [Google Scholar] [CrossRef]
- De Mattia, G.; Bravi, M.C.; Laurenti, O.; Moretti, A.; Cipriani, R.; Gatti, A.; Mandosi, E.; Morano, S. Endothelial dysfunction and oxidative stress in type 1 and type 2 diabetic patients without clinical macrovascular complications. Diabetes Res. Clin. Pract. 2008, 79, 337–342. [Google Scholar] [CrossRef]
- He, Z.; Rask-Madsen, C.; King, G.L. Managing heart disease and potential new pharmacological therapies. Eur. Heart J. Suppl. 2003, 5, B51–B57. [Google Scholar] [CrossRef]
- Lim, A. Diabetic nephropathy-complications and treatment. Int. J. Nephrol. Renovasc. Dis. 2014, 7, 361–381. [Google Scholar] [CrossRef]
- Deckert, T.; Poulsen, J.E.; Larsen, M. Prognosis of diabetics with diabetes onset before the age of thirtyone. Diabetologia 1978, 14, 363–370. [Google Scholar] [CrossRef]
- Ballard, D.J.; Humphrey, L.L.; Melton, L.J., III; Frohnert, P.P.; Chu, P.C.; O’Fallon, W.M.; Palumbo, P.J. Epidemiology of persistent proteinuria in type II diabetes mellitus: Population-based study in Rochester, Minnesota. Diabetes 1988, 37, 405–412. [Google Scholar] [CrossRef]
- Schena, F.P.; Gesualdo, L. Pathogenetic mechanisms of diabetic nephropathy. J. Am. Soc. Nephrol. 2005, 16, S30–S33. [Google Scholar] [CrossRef]
- Fong, D.S.; Aiello, L.; Gardner, T.W.; King, G.L.; Blankenship, G.; Cavallerano, J.D.; Ferris, F.L., III; Klein, R. Retinopathy in diabetes. Diabetes Care 2004, 27, S84–S87. [Google Scholar] [CrossRef]
- Keenan, H.A.; Costacou, T.; Sun, J.K.; Doria, A.; Cavellerano, J.; Coney, J.; Orchard, T.J.; Aiello, L.P.; King, G.L. Clinical factors associated with resistance to microvascular complications in diabetic patients of extreme disease duration: The 50-year medalist study. Diabetes Care 2007, 30, 1995–1997. [Google Scholar] [CrossRef]
- Fowler, M.J. Microvascular and Macrovascular Complications of Diabetes. Clin. Diabetes 2008, 26, 77–82. [Google Scholar] [CrossRef]
- Vinik, A.; Casellini, C.; Nevoret, M.L.; Feingold, K.R.; Anawalt, B.; Boyce, A.; Chrousos, G.; Dungan, K.; Grossman, A.; Hershman, J.M.; et al. Endotext [Internet]. Available online: MDText.com (accessed on 12 September 2019).
- Said, G.; Goulon-Goeau, C.; Slama, G.; Tchobroutsky, G. Severe early-onset polyneuropathy in insulin-dependent diabetes mellitus: A clinical and pathological study. N. Engl. J. Med. 1992, 326, 1257–1263. [Google Scholar] [CrossRef]
- Reliability and validity of a diabetes quality-of-life measure for the diabetes control and complications trial (DCCT). Diabetes Care 1988, 11, 725–732. [CrossRef]
- Yagihashi, S.; Yamagishi, S.; Wada, R. Pathology and pathogenetic mechanisms of diabetic neuropathy: Correlation with clinical signs and symptoms. Diabetes Res. Clin. Pract. 2007, 77, S184–S189. [Google Scholar] [CrossRef]
- Litzelman, D.K.; Slemenda, C.W.; Langefeld, C.D.; Hays, L.M.; Welch, M.A.; Bild, D.E.; Ford, E.S.; Vinicor, F. Reduction of lower extremity clinical abnormalities in patients with non-insulin-dependent diabetes mellitus: A randomized, controlled trial. Ann. Intern. Med. 1993, 119, 36–41. [Google Scholar] [CrossRef]
- Boulton, A.J.; Vinik, A.I.; Arezzo, J.C.; Bril, V.; Feldman, E.L.; Freeman, R.; Malik, R.A.; Maser, R.E.; Sosenko, J.M.; Ziegler, D. Diabetic neuropathies: A statement by the American Diabetes Association. Diabetes Care 2005, 28, 956–962. [Google Scholar] [CrossRef]
- Iqbal, Z.; Azmi, S.; Yadav, R.; Ferdousi, M.; Kumar, M.; Cuthbertson, D.J.; Lim, J.; Malik, R.A.; Alam, U. Diabetic peripheral neuropathy: Epidemiology, diagnosis, and pharmacotherapy. Clin. Ther. 2018, 40, 828–849. [Google Scholar] [CrossRef]
- Esteller, M. Non-coding RNAs in human disease. Nat. Rev. Genet. 2011, 12, 861–874. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Niederberger, E.; Kynast, K.; Lotsch, J.; Geisslinger, G. MicroRNAs as new players in the pain game. Pain 2011, 152, 1455–1458. [Google Scholar] [CrossRef]
- Friedman, R.C.; Farh, K.K.H.; Burge, C.B.; Bartel, D.P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009, 19, 92–105. [Google Scholar] [CrossRef]
- Krek, A.; Grun, D.; Poy, M.N.; Wolf, R.; Rosenberg, L.; Epstein, E.J.; MacMenamin, P.; da Piedade, I.; Gunsalus, K.C.; Stoffel, M.; et al. Combinatorial microRNA target predictions. Nat. Genet. 2005, 37, 495–500. [Google Scholar] [CrossRef]
- Chen, X.; Ba, Y.; Ma, L.; Cai, X.; Yin, Y.; Wang, K.; Guo, J.; Zhang, Y.; Chen, J.; Guo, X.; et al. Characterization of microRNAs in serum: A novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res. 2008, 18, 997–1006. [Google Scholar] [CrossRef]
- ElSharawy, A.; Keller, A.; Flachsbart, F.; Wendschlag, A.; Jacobs, G.; Kefer, N.; Brefort, T.; Leidinger, P.; Backes, C.; Meese, E.; et al. Genome-wide miRNA signatures of human longevity. Aging Cell 2012, 11, 607–616. [Google Scholar] [CrossRef]
- Grasso, M.; Piscopo, P.; Confaloni, A.; Denti, M.A. Circulating miRNAs as biomarkers for neurodegenerative disorders. Molecules 2014, 19, 6891–6910. [Google Scholar] [CrossRef]
- Harris, V.K.; Tuddenham, J.F.; Sadiq, S.A. Biomarkers of multiple sclerosis: Current findings. Degener. Neurol. Neuromuscul. Dis. 2017, 7, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Hebert, S.S.; De Strooper, B. Alterations of the microRNA network cause neurodegenerative disease. Trends Neurosci. 2009, 32, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Tan, L.; Tan, L.; Tian, Y.; Ma, J.; Tan, C.C.; Wang, H.F.; Liu, Y.; Tan, M.S.; Jiang, T.; et al. Circulating microRNAs are promising novel biomarkers for drug-resistant epilepsy. Sci. Rep. 2015, 5, 10201. [Google Scholar] [CrossRef] [PubMed]
- La Marca, V.; Fierabracci, A. Insights into the diagnostic potential of extracellular vesicles and their miRNA signature from liquid biopsy as early biomarkers of diabetic micro/macrovascular complications. Int. J. Mol. Sci. 2017, 18, 1974. [Google Scholar] [CrossRef] [PubMed]
- McClelland, A.D.; Kantharidis, P. microRNA in the development of diabetic complications. Clin. Sci. 2014, 126, 95–110. [Google Scholar] [CrossRef] [PubMed]
- Kosik, K.S. The neuronal microRNA system. Nat. Rev. Neurosci. 2006, 7, 911–920. [Google Scholar] [CrossRef]
- Davis, T.H.; Cuellar, T.L.; Koch, S.M.; Barker, A.J.; Harfe, B.D.; McManus, M.T.; Ullian, E.M. Conditional loss of Dicer disrupts cellular and tissue morphogenesis in the cortex and hippocampus. J. Neurosci. 2008, 28, 4322–4330. [Google Scholar] [CrossRef]
- Schaefer, A.; O’Carroll, D.; Tan, C.L.; Hillman, D.; Sugimori, M.; Llinas, R.; Greengard, P. Cerebellar neurodegeneration in the absence of microRNAs. J. Exp. Med. 2007, 204, 1553–1558. [Google Scholar] [CrossRef]
- Smalheiser, N.R.; Lugli, G. MicroRNA regulation of synaptic plasticity. Neuromol. Med. 2009, 11, 133–140. [Google Scholar] [CrossRef]
- Konopka, W.; Kiryk, A.; Novak, M.; Herwerth, M.; Parkitna, J.R.; Wawrzyniak, M.; Kowarsch, A.; Michaluk, P.; Dzwonek, J.; Arnsperger, T.; et al. MicroRNA loss enhances learning and memory in mice. J. Neurosci. 2010, 30, 14835–14842. [Google Scholar] [CrossRef]
- Kovacs, B.; Lumayag, S.; Cowan, C.; Xu, S. MicroRNAs in early diabetic retinopathy in streptozotocin-induced diabetic rats. Invest. Ophthalmol. Vis. Sci. 2011, 52, 4402–4409. [Google Scholar] [CrossRef] [PubMed]
- Wilson, N.; Wright, D. Inflammatory mediators in diabetic neuropathy. J. Diabetes Metab. 2011, S5, 4. [Google Scholar] [CrossRef]
- Denby, L.; Ramdas, V.; McBride, M.W.; Wang, J.; Robinson, H.; McClure, J.; Crawford, W.; Lu, R.; Hillyard, D.Z.; Khanin, R.; et al. miR-21 and miR-214 are consistently modulated during renal injury in rodent models. Am. J. Pathol. 2011, 179, 661–672. [Google Scholar] [CrossRef] [PubMed]
- Aiello, L.P.; Pierce, E.A.; Foley, E.D.; Takagi, H.; Chen, H.; Riddle, L.; Ferrara, N.; King, G.L.; Smith, L.E. Suppression of retinal neovascularization in vivo by inhibition of vascular endothelial growth factor (VEGF) using soluble VEGF-receptor chimeric proteins. Proc. Natl. Acad. Sci. USA 1995, 92, 10457–10461. [Google Scholar] [CrossRef] [PubMed]
- Ropper, A.H.; Gorson, K.C.; Gooch, C.L.; Weinberg, D.H.; Pieczek, A.; Ware, J.H.; Kershen, J.; Rogers, A.; Simovic, D.; Schratzberger, P.; et al. Vascular endothelial growth factor gene transfer for diabetic polyneuropathy: A randomized, double-blinded trial. Ann. Neurol. 2009, 65, 386–393. [Google Scholar] [CrossRef] [PubMed]
- Kantharidis, P.; Wang, B.; Carew, R.M.; Lan, H.Y. Diabetes complications: The microRNA perspective. Diabetes 2011, 60, 1832–1837. [Google Scholar] [CrossRef] [PubMed]
- Ristoiu, V.; Shibasaki, K.; Uchida, K.; Zhou, Y.; Ton, B.H.; Flonta, M.L.; Tominaga, M. Hypoxia-induced sensitization of transient receptor potential vanilloid 1 involves activation of hypoxia-inducible factor-1 alpha and PKC. Pain 2011, 152, 936–945. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Chen, L.; Luo, Q.; Wu, M.; Chen, Y.; Shi, X. Involvement of microRNA-146a in diabetic peripheral neuropathy through the regulation of inflammation. Drug Des. Devel. Ther. 2018, 12, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Taganov, K.D.; Boldin, M.P.; Chang, K.J.; Baltimore, D. NF-kappaB-dependent induction of microRNA miR-146, an inhibitor targeted to signaling proteins of innate immune responses. Proc. Natl. Acad. Sci. USA 2006, 103, 12481–12486. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Wang, P.; Lin, L.; Liu, X.; Ma, F.; An, H.; Wang, Z.; Cao, X. MicroRNA-146a feedback inhibits RIG-I-dependent Type I IFN production in macrophages by targeting TRAF6, IRAK1, and IRAK2. J. Immunol. 2009, 183, 2150–2158. [Google Scholar] [CrossRef]
- Liu, X.S.; Fan, B.; Szalad, A.; Jia, L.; Wang, L.; Wang, X.; Pan, W.; Zhang, L.; Zhang, R.; Hu, J.; et al. MicroRNA-146a mimics reduce the peripheral neuropathy in type 2 diabetic mice. Diabetes 2017, 66, 3111–3121. [Google Scholar] [CrossRef] [PubMed]
- Lemarchant, S.; Pruvost, M.; Montaner, J.; Emery, E.; Vivien, D.; Kanninen, K.; Koistinaho, J. ADAMTS proteoglycanases in the physiological and pathological central nervous system. J. Neuroinflammation 2013, 10, 133. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Chopp, M.; Szalad, A.; Zhang, Y.; Wang, X.; Zhang, R.L.; Liu, X.S.; Jia, L.; Zhang, Z.G. The role of miR-146a in dorsal root ganglia neurons of experimental diabetic peripheral neuropathy. Neuroscience 2014, 259, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Yousefzadeh, N.; Alipour, M.R.; Soufi, F.G. Deregulation of NF-small ka, CyrillicB-miR-146a negative feedback loop may be involved in the pathogenesis of diabetic neuropathy. J. Physiol. Biochem. 2015, 71, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Xu, D.; Zhu, X.; Yang, G.; Ren, M. MiR-106a Associated with diabetic peripheral neuropathy through the regulation of 12/15-LOX-mediated oxidative/nitrative stress. Curr. Neurovasc. Res. 2017, 14, 117–124. [Google Scholar] [CrossRef] [PubMed]
- El-Lithy, G.M.; El-Bakly, W.M.; Matboli, M.; Abd-Alkhalek, H.A.; Masoud, S.I.; Hamza, M. Prophylactic L-arginine and ibuprofen delay the development of tactile allodynia and suppress spinal miR-155 in a rat model of diabetic neuropathy. Transl. Res. 2016, 177, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Ao, Q.; Guo, Q.; He, W.; Peng, L.; Jiang, J.; Hu, X. miR-9 mediates CALHM1-activated ATP-P2X7R signal in painful diabetic neuropathy rats. Mol. Neurobiol. 2017, 54, 922–929. [Google Scholar] [CrossRef]
- Gong, Q.; Lu, Z.; Huang, Q.; Ruan, L.; Chen, J.; Liang, Y.; Wang, H.; Yue, Y.; Feng, S. Altered microRNAs expression profiling in mice with diabetic neuropathic pain. Biochem. Biophys. Res. Commun. 2015, 456, 615–620. [Google Scholar] [CrossRef]
- Guo, G.; Liu, Y.; Ren, S.; Kang, Y.; Duscher, D.; Machens, H.G.; Chen, Z. Comprehensive analysis of differentially expressed microRNAs and mRNAs in dorsal root ganglia from streptozotocin-induced diabetic rats. PLoS ONE 2018, 13, e0202696. [Google Scholar] [CrossRef]
- Yang, D.; Yang, Q.; Wei, X.; Liu, Y.; Ma, D.; Li, J.; Wan, Y.; Luo, Y. The role of miR-190a-5p contributes to diabetic neuropathic pain via targeting SLC17A6. J. Pain Res. 2017, 10, 2395–2403. [Google Scholar] [CrossRef]
- Wang, H.S.; Yu, G.; Wang, Z.T.; Yi, S.P.; Su, R.B.; Gong, Z.H. Changes in VGLUT1 and VGLUT2 expression in rat dorsal root ganglia and spinal cord following spared nerve injury. Neurochem. Int. 2016, 99, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Simeoli, R.; Montague, K.; Jones, H.R.; Castaldi, L.; Chambers, D.; Kelleher, J.H.; Vacca, V.; Pitcher, T.; Grist, J.; Al-Ahdal, H.; et al. Exosomal cargo including microRNA regulates sensory neuron to macrophage communication after nerve trauma. Nat. Commun. 2017, 8, 1778. [Google Scholar] [CrossRef] [PubMed]
- Fierabracci, A.; Del Fattore, A.; Luciano, R.; Muraca, M.; Teti, A.; Muraca, M. Recent advances in mesenchymal stem cell immunomodulation: The role of microvesicles. Cell Transplant. 2015, 24, 133–149. [Google Scholar] [CrossRef] [PubMed]
- Del Fattore, A.; Luciano, R.; Pascucci, L.; Goffredo, B.M.; Giorda, E.; Scapaticci, M.; Fierabracci, A.; Muraca, M. Immunoregulatory effects of mesenchymal stem cell-derived extracellular vesicles on T Lymphocytes. Cell Transplant. 2015, 24, 2615–2627. [Google Scholar] [CrossRef] [PubMed]
- Jia, L.; Chopp, M.; Wang, L.; Lu, X.; Szalad, A.; Zhang, Z.G. Exosomes derived from high-glucose-stimulated schwann cells promote development of diabetic peripheral neuropathy. FASEB J. 2018, 32, 6911–6922. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, K.; Kani, S.; Shimizu, T.; Bae, Y.K.; Abe, T.; Hibi, M. Atypical protein kinase C regulates primary dendrite specification of cerebellar purkinje cells by localizing Golgi apparatus. J. Neurosci. 2010, 30, 16983–16992. [Google Scholar] [CrossRef] [PubMed]
- Kole, A.J.; Swahari, V.; Hammond, S.M.; Deshmukh, M. miR-29b is activated during neuronal maturation and targets BH3-only genes to restrict apoptosis. Genes Dev. 2011, 25, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Gong, X.; Han, S.; Zhang, Y. MiR-29b protects dorsal root ganglia neurons from diabetic rat. Cell Biochem. Biophys. 2014, 70, 1105–1111. [Google Scholar] [CrossRef]
- Lynch, M.P.; Stein, J.L.; Stein, G.S.; Lian, J.B. The influence of type I collagen on the development and maintenance of the osteoblast phenotype in primary and passaged rat calvarial osteoblasts: Modification of expression of genes supporting cell growth, adhesion, and extracellular matrix mineralization. Exp. Cell Res. 1995, 216, 35–45. [Google Scholar] [CrossRef]
- Cheng, C.; Kobayashi, M.; Martinez, J.A.; Ng, H.; Moser, J.J.; Wang, X.; Singh, V.; Fritzler, M.J.; Zochodne, D.W. Evidence for epigenetic regulation of gene expression and function in chronic experimental diabetic Neuropathy. J. Neuropathol. Exp. Neurol. 2015, 74, 804–817. [Google Scholar] [CrossRef]
- Ciccacci, C.; Morganti, R.; Di Fusco, D.; D’Amato, C.; Cacciotti, L.; Greco, C.; Rufini, S.; Novelli, G.; Sangiuolo, F.; Marfia, G.A.; et al. Common polymorphisms in MIR146a, MIR128a and MIR27a genes contribute to neuropathy susceptibility in type 2 diabetes. Acta Diabetol. 2014, 51, 663–671. [Google Scholar] [CrossRef] [PubMed]
- Ciccacci, C.; Latini, A.; Greco, C.; Politi, C.; D’Amato, C.; Lauro, D.; Novelli, G.; Borgiani, P.; Spallone, V. Association between a MIR499A polymorphism and diabetic neuropathy in type 2 diabetes. J. Diabetes Complicat. 2018, 32, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.B.; Wu, Q.; Liu, J.; Fan, Y.Z.; Yu, K.F.; Cai, Y. miR-199a-3p is involved in the pathogenesis and progression of diabetic neuropathy through downregulation of SerpinE2. Mol. Med. Rep. 2017, 16, 2417–2424. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.B.; Ma, W.; Xie, C.; Zhang, M.; Yin, X.; Wang, F.; Xu, J.; Shi, B. Identification of genes and signaling pathways associated with diabetic neuropathy using a weighted correlation network analysis: A consort study. Medicine 2016, 95, e5443. [Google Scholar] [CrossRef] [PubMed]
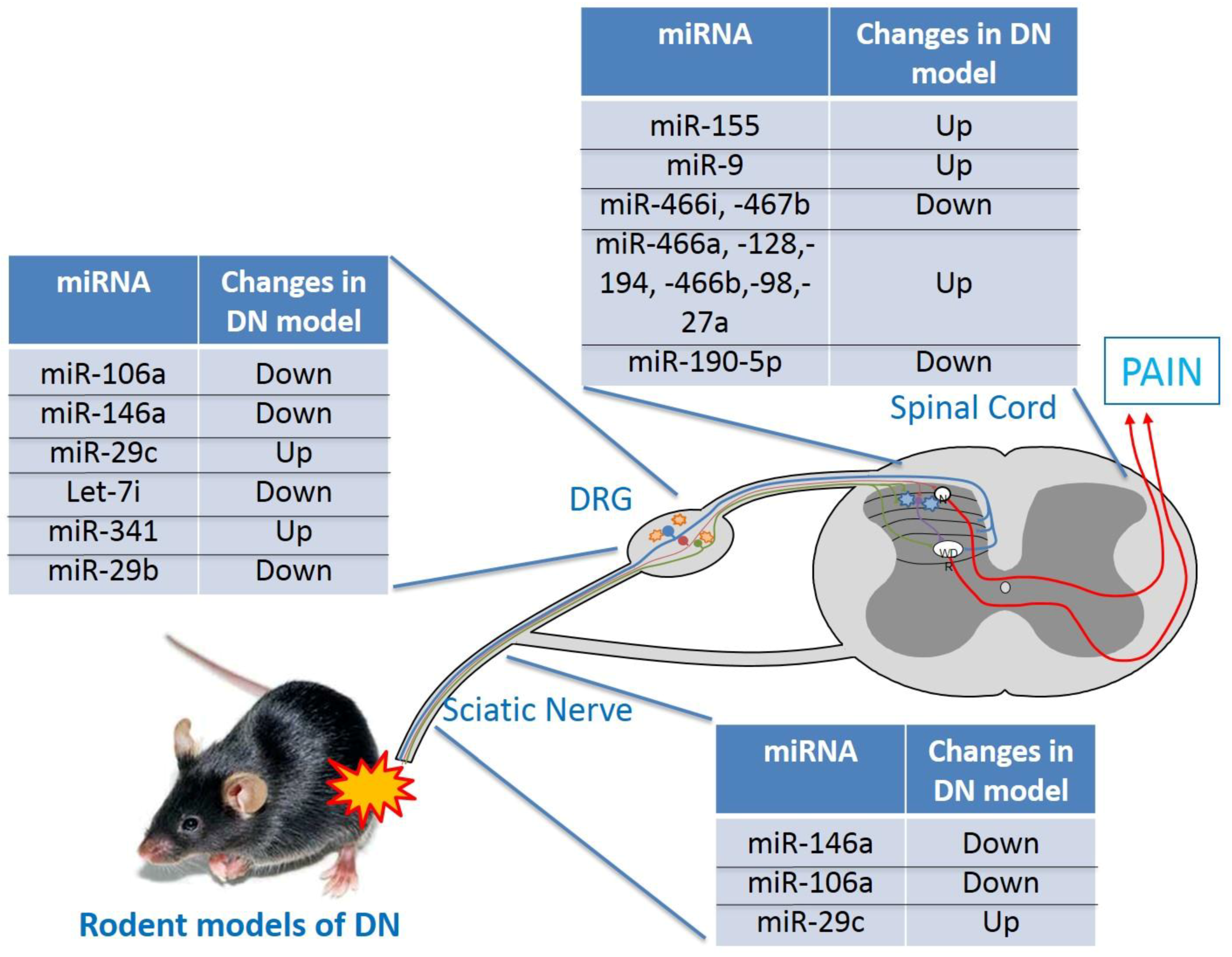
| miRNA | DN Model | Changes in DN | Target (s) | Reference (s) |
|---|---|---|---|---|
| miR-146a | DPN induced by intraperitoneal injection of STZ in HFD-fed rats | Down | IRAK1; TRAF6 | Feng Y et al., 2018 [49] |
| miR-146a | Db/db mice as model of T2D and DN | Down | IRAK1; TRAF6; ADAMTS3 | Liu XS et al., 2017 [52] |
| miR-146a | Db/db mice as model of T2D and DN | Down | IRAK1; TRAF6 | Wang L et al., 2014 [54] |
| miR-146a | Diabetes was induced in rats by a single-dose injection of nicotinammide followed by STZ | Up | IRAK1; TRAF6 | Yousefzadeh N et al., 2015 [55] |
| miR-106a | DPN model was established in mice following single injection of STZ | Down | 12/15-Lipoxygenase (12/15-LOX) | Wu Y et al., 2017 [56] |
| miR-155 | DN was established in rats following injection of STZ | Up | ND | El Lithy GM et al., 2016 [57] |
| miR-9 | DN was established in rats following injection of STZ | Up | CALHM1 is indirect target of miR-9 | Liu W et al., 2016 [58] |
| miR-466i | DPN was induced in mice following injection of STZ | Down | IL-1β; TNF-α; IL-6 | Gong Q et al., 2015 [59] |
| miR-467b | Down | |||
| miR-466a | Up | IL-1β | ||
| miR-128; miR-194; miR-466b; miR-98 | Up | IL-1β | ||
| miR-27a | Up | IL-10 | ||
| miR-194 | Up | IL-13 | ||
| 37 miRNAs were differently expressed in DRG | DN was established in rats following injection of STZ | 15 miRNAs were upregulated whereas 22 were downregulated | Podx1 and Hoxa1 were the most common targets | Guo G et al., 2018 [60] |
| miR-190-5p | DPN model was established in mice following single injection of STZ | Down | SLC17A6 | Yang D et al., 2017 [61] |
| miR-28, miR-31a and miR-130a | In vitro exosomes isolated from high glucose stimulated Swann Cells contained high levels of miRs-28, -31a, -130 | Up | NDNMT3A NUMB, SNAP25 and GAP43 | Jia L et al., 2018 [66] |
| miR-29b | DN was established in rats following injection of STZ | Down | TGF-β/Smad3 | Zhang X et al., 2014 [69] |
| Let-7i and miR-341 | DPN model was established in mice following single injection of STZ | Down | NF-kB | Cheng C et al., 2015 [71] |
| MiRNA | Target (s) | Changes in DN Patients | Clinical Manifestations | Reference |
|---|---|---|---|---|
| miR-499a; -128a; -146a; -27a | ND | Polymorphisms in the miRNA gene sequence | High incidence of developing CAN and DPN in T2D | Ciccacci C et al. [72,73] |
| miR-199a-3p | SerpinE2 | Up in skin biopsies | Promote coagulation in peripheral skin circulation in T2D | Li YB et al., 2017 [74] |
| miR-216a | ANXA9 | Up in nerve tissue samples | Positive association with progressing DN | Li YB et al., 2016 [75] |
| ANGPTL4 | ||||
| CHI3L2 | ||||
| miR-217 | ADRBK2 | |||
| CSN1S1 | ||||
| GALM | ||||
| miR-377 | EN1 | |||
| GAP43 | ||||
| FAM89A |
| MiRNA | Target Genes | Most Regulated Pathways | KEGG Pathway |
|---|---|---|---|
| miR-146a | IRAK1 | NF-kB signaling pathway | mmu04064 |
| Toll-like receptor signaling pathway | mmu04620 | ||
| MAPK signaling pathway | mmu04010 | ||
| Neurotrophin signaling pathway | mmu04722 | ||
| miR-146a | TRAF6 | NF-kB signaling pathway | mmu04064 |
| Toll-like receptor signaling pathway | mmu04620 | ||
| Neurotrophin signaling pathway | mmu04722 | ||
| IL-17 signaling pathway | mmu04657 | ||
| NOD-like receptor signaling pathway | mmu04621 | ||
| MAPK signaling pathway | mmu04010 | ||
| Ubiquitin mediated proteolysis | mmu04120 | ||
| RIG-I-like receptor signaling | mmu04622 | ||
| Endocytosis | mmu04144 | ||
| miR-106a | 12/15-Lipoxygenase (12/15-LOX) | Arachidonic acid metabolism | mmu00590 |
| miR-190-5p | SLC17A6 | Synaptic vesicle cycle | mmu04721 |
| Glutamatergic synapse | mmu04724 | ||
| miR-31a; miR-130a | NUMB; SNAP25 | Notch signaling pathway | mmu04330 |
| Synaptic vesicle cycle | mmu04721 | ||
| Insulin secretion | mmu04911 | ||
| miR-29b | Smad3 | TGF-beta signaling pathway | mmu04350 |
| FoxO signaling pathway | mmu04068 | ||
| Wnt signaling pathway | mmu04310 | ||
| Th17 cell differentiation | mmu04659 | ||
| Cell cycle | mmu04110 | ||
| miR-29c | PRKCI | Tight junction | mmu04530 |
| Rap1 signaling pathway | mmu04015 | ||
| Endocytosis | mmu04144 | ||
| Insulin signaling pathway | mmu04910 | ||
| miR-466i; miR-467b | TNF-α; Il-6 | NF-kB signaling pathway | mmu04064 |
| Apoptosis | mmu04210 | ||
| TNF signaling pathway | mmu04668 | ||
| HIF-1 signaling pathway | mmu04066 | ||
| PI3K-Akt signaling pathway | mmu04151 | ||
| Insulin resistance | mmu04931 | ||
| Toll-like receptor signaling pathway | mmu04620 | ||
| NOD-like receptor signaling pathway | mmu04621 | ||
| miR-466a; miR-128; -194; -466b; -98 | IL-1β | NOD-like receptor signaling pathway | mmu04621 |
| NF-kB signaling pathway | mmu04064 | ||
| IL-17 signaling pathway | mmu04657 | ||
| Th17 cell differentiation | mmu04659 | ||
| Toll-like receptor signaling pathway | mmu04620 | ||
| Inflammatory mediator regulation of TRP channels | mmu04750 | ||
| miR-27; miR-194 | IL-10; IL-13 | FoxO signaling pathway | mmu04068 |
| TCR signaling pathway | mmu04660 | ||
| IL-17 signaling pathway | mmu04657 | ||
| Th1 and Th2 cell differentiation | mmu04658 | ||
| miR-216a | ANGPTL4 | PPAR signaling pathway | hsa03320 |
| Cholesterol metabolism | hsa04979 | ||
| miR-217 | ADRBK2 GALM | Glutamatergic synapse | hsa04724 |
| Glycolysis and gluconeogenesis | hsa00010 | ||
| Galactose metabolism | hsa00052 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Simeoli, R.; Fierabracci, A. Insights into the Role of MicroRNAs in the Onset and Development of Diabetic Neuropathy. Int. J. Mol. Sci. 2019, 20, 4627. https://doi.org/10.3390/ijms20184627
Simeoli R, Fierabracci A. Insights into the Role of MicroRNAs in the Onset and Development of Diabetic Neuropathy. International Journal of Molecular Sciences. 2019; 20(18):4627. https://doi.org/10.3390/ijms20184627
Chicago/Turabian StyleSimeoli, Raffaele, and Alessandra Fierabracci. 2019. "Insights into the Role of MicroRNAs in the Onset and Development of Diabetic Neuropathy" International Journal of Molecular Sciences 20, no. 18: 4627. https://doi.org/10.3390/ijms20184627
APA StyleSimeoli, R., & Fierabracci, A. (2019). Insights into the Role of MicroRNAs in the Onset and Development of Diabetic Neuropathy. International Journal of Molecular Sciences, 20(18), 4627. https://doi.org/10.3390/ijms20184627






