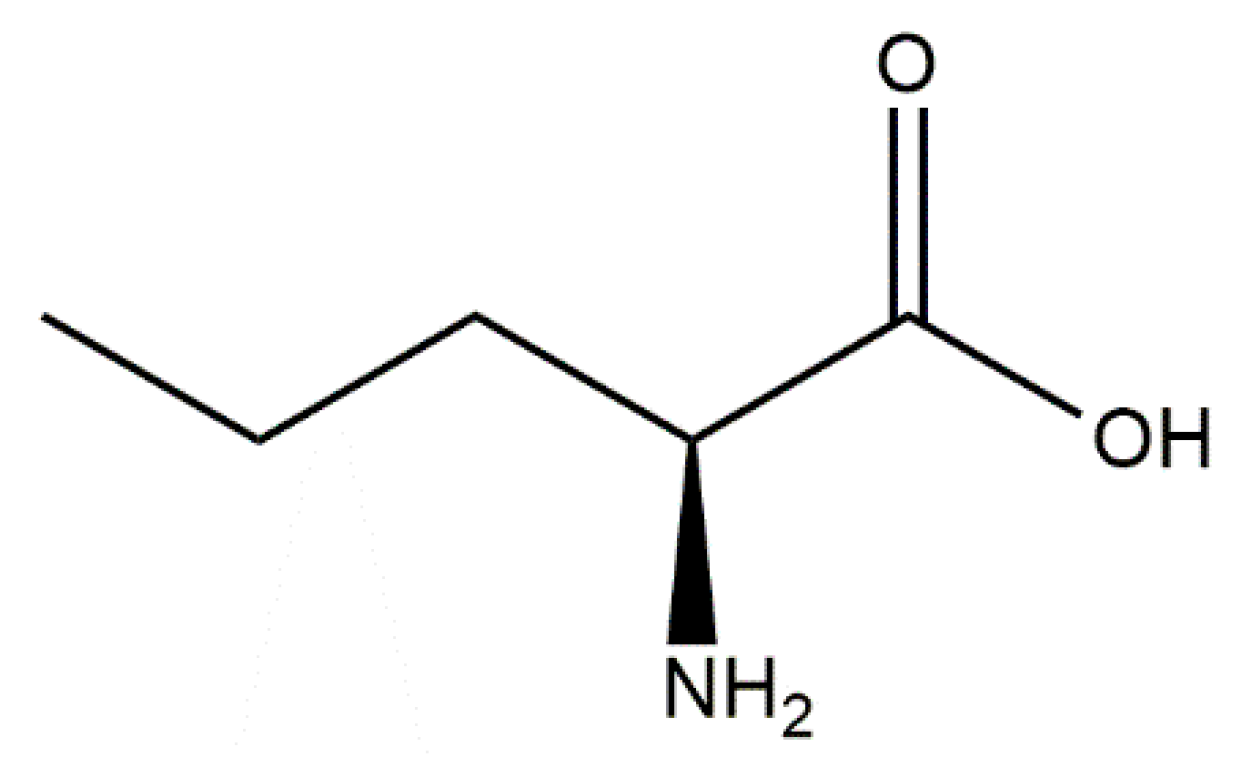
Norvaline Restores the BBB Integrity in a Mouse Model of Alzheimer’s Disease
Abstract
1. Introduction
2. Results
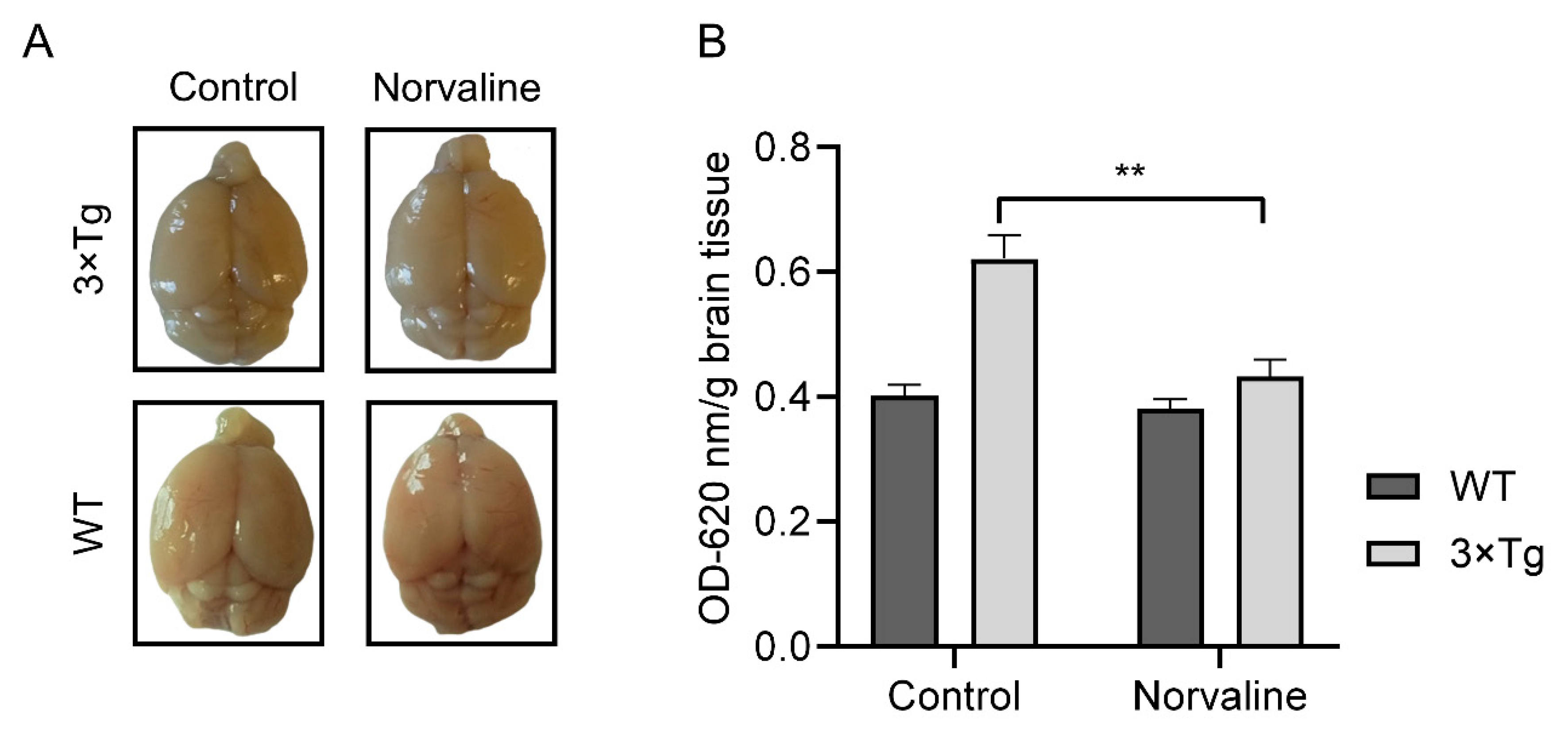
2.1. Norvaline Improves the BBB Integrity in the 3×Tg-AD Mice
2.2. Norvaline Attenuates Cerebral Amyloid Angiopathy
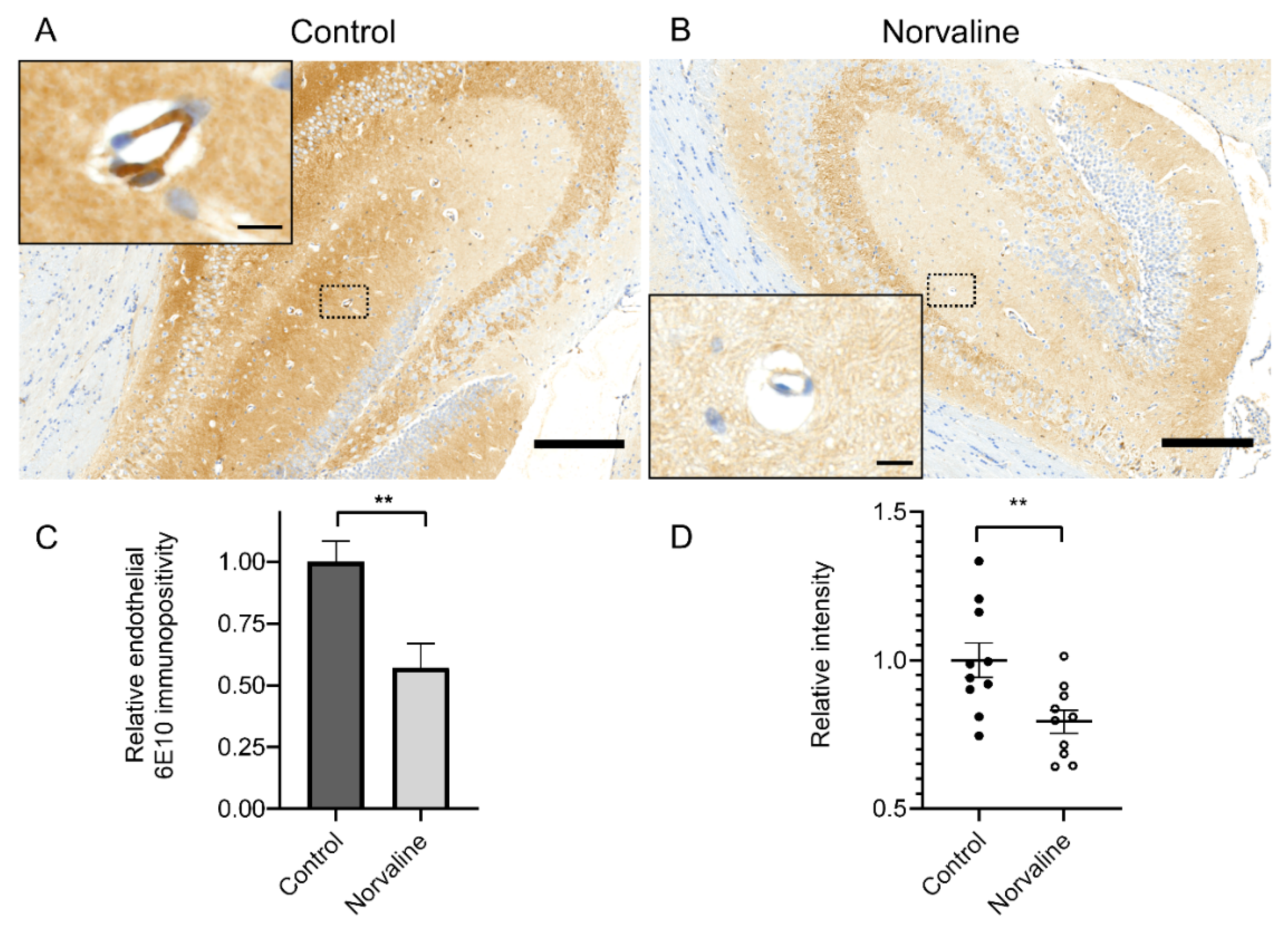
2.2.1. Norvaline Reduces Endothelial Amyloidosis
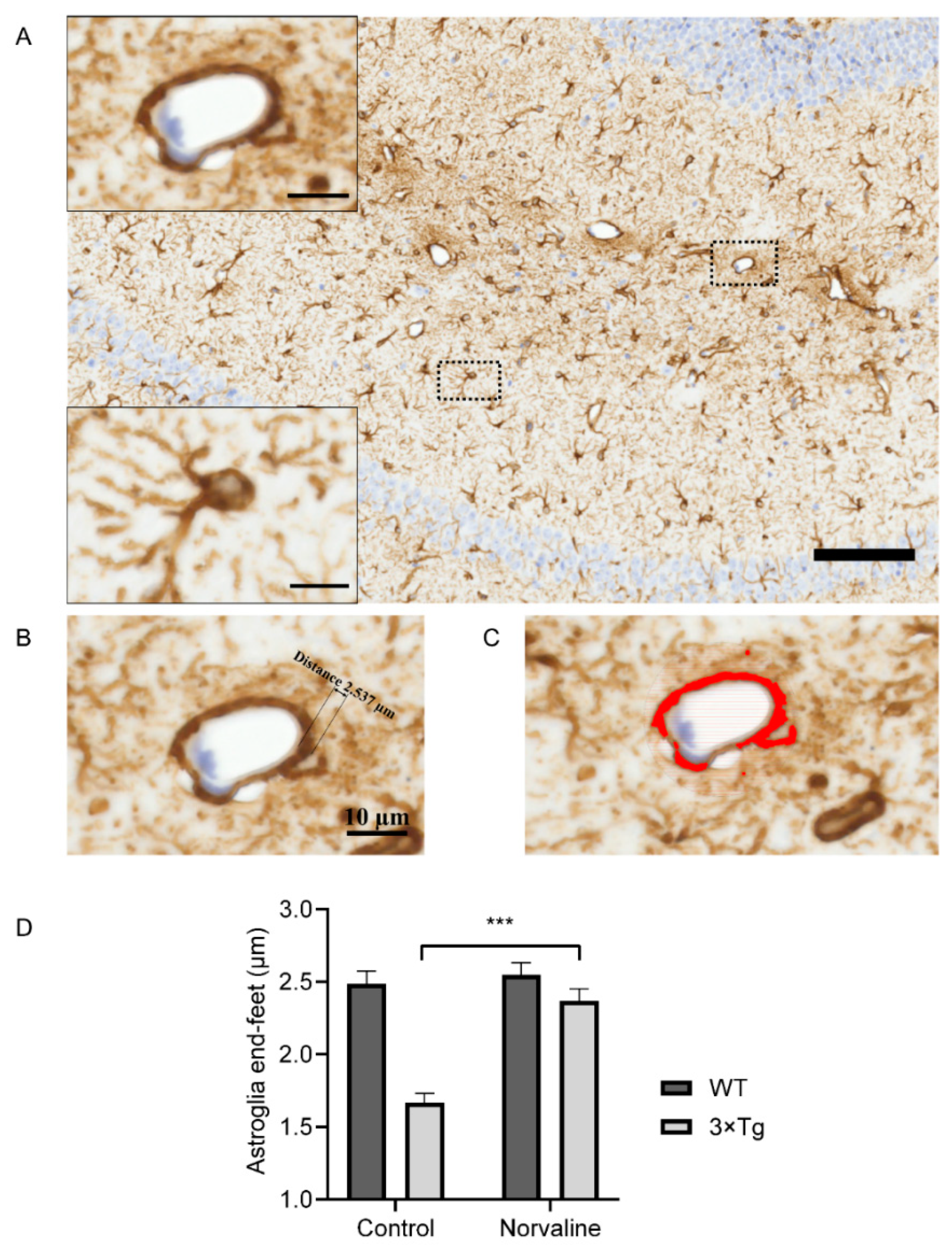
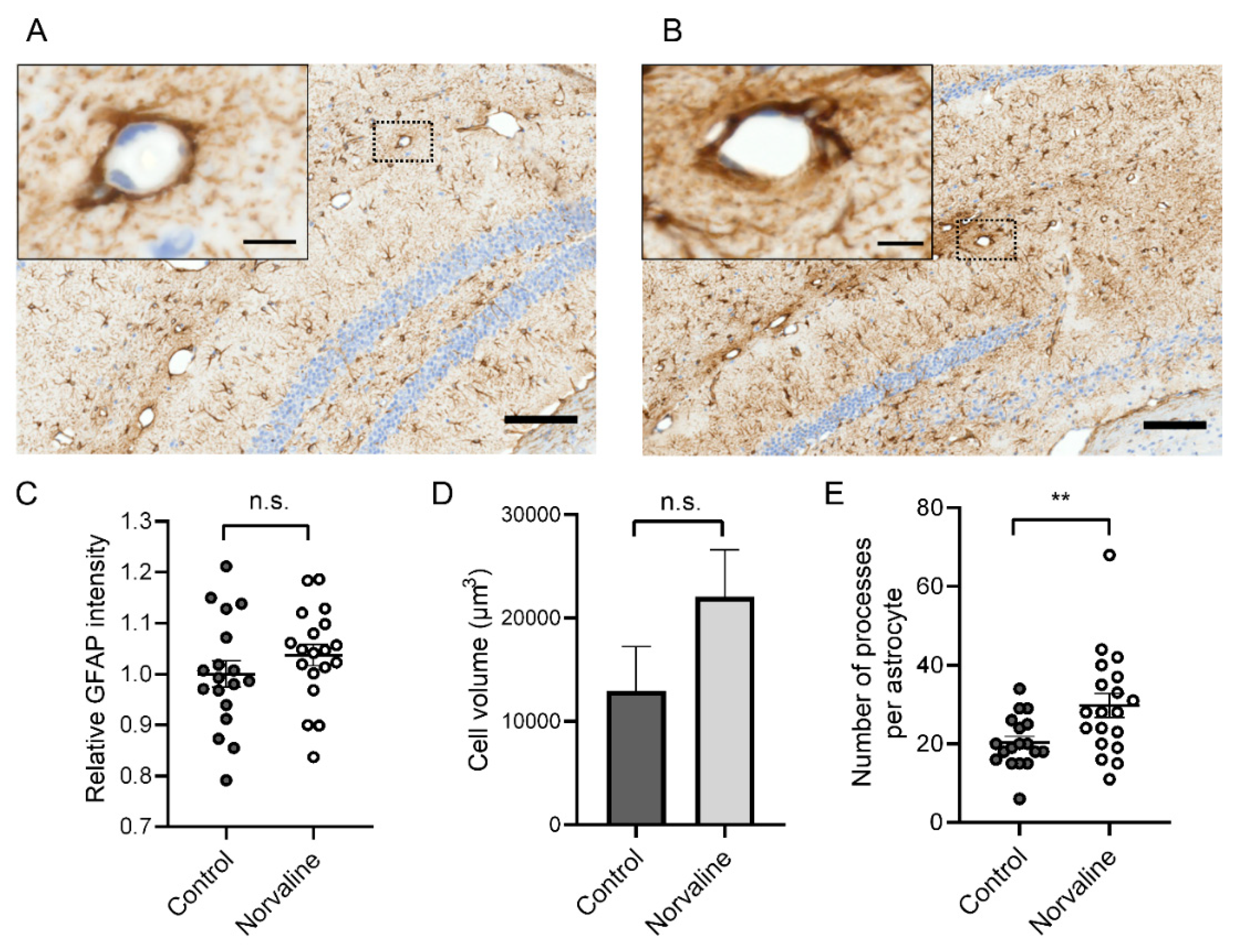
2.2.2. Norvaline Diminishes the Rate of Astrodegeneration in the 3×Tg-AD Mice
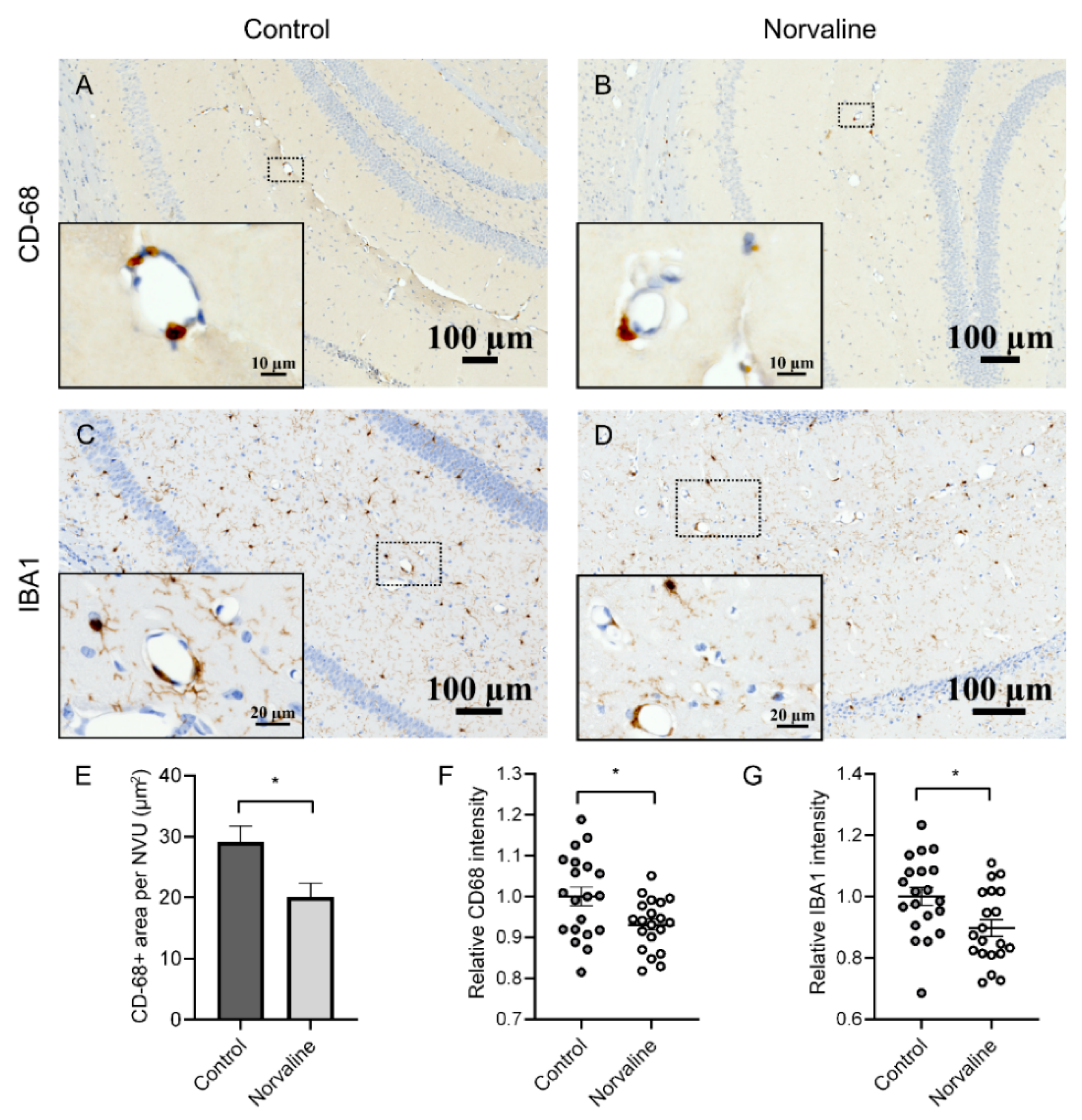
2.2.3. Norvaline Moderates the Activity of the Brain Perivascular Macrophages
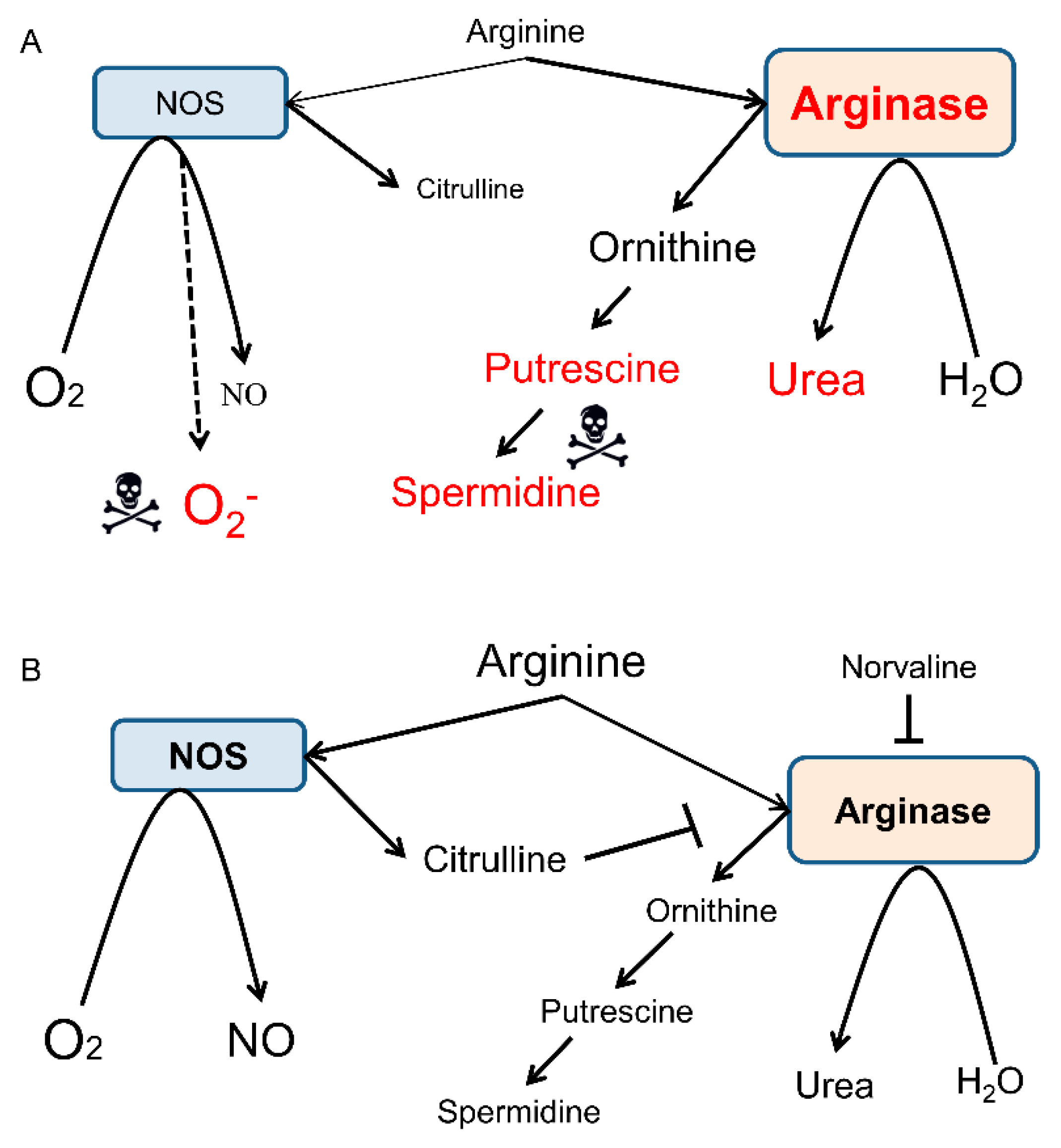
2.3. Neuronal Nitric Oxide Synthase (nNOS) and Inducible Nitric Oxide Synthase (iNOS) mRNA Levels Are Significantly Reduced in the Brains of 3×Tg-AD Mice Compared to the WT Mice.
2.4. Norvaline Increases the Transcription Levels of nNOS Gene but Not iNOS Gene.
3. Discussion
4. Materials and Methods
4.1. Animals and Treatment
4.2. Evans Blue In Vivo Assay
4.3. Tissue Sampling
4.4. Fixation and Tissue Processing
4.5. Immunohistochemistry
4.6. Imaging and Quantification Analysis
4.6.1. 6E10 Staining and Analysis
4.6.2. GFAP Staining and Analysis
4.6.3. Iba1 and CD-68 Staining and Analysis
4.7. RNA Isolation and Reverse Transcription
4.8. Polymerase Chain Reaction Amplification
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s disease |
| BBB | blood-brain barrier |
| 3×Tg-AD | triple-transgenic mouse model of Alzheimer’s disease |
| NOS | nitric oxide synthase |
| Aβ | amyloid-beta |
| NFT | neurofibrillary tangles |
| CAA | cerebral amyloid angiopathy |
| APP | amyloid precursor protein |
| CNS | central nervous system |
| CSF | cerebrospinal fluid |
| ECs | endothelial cells |
| CNS | central nervous system |
| CSF | cerebrospinal fluid |
| NVU | neurovascular unit |
| BACE1 | β-site APP cleaving enzyme 1 |
| WT | wild-type |
| OD | optical density |
| ANOVA | analysis of variance |
| CA | cornus Ammonis |
| SEM | standard error of the mean |
| CD-68 | cluster of differentiation 68 |
| Iba1 | ionized calcium-binding adapter molecule 1 |
| mRNA | messenger ribonucleic acid |
| PSD-95 | postsynaptic density protein 95 |
| GFAP | glial fibrillary acidic protein |
References
- Alzheimer’s Association. 2016 Alzheimer’s Disease Facts and Figures. Alzheimer’s Dement. 2016, 12, 459–509. [Google Scholar] [CrossRef]
- Goedert, M.; Spillantini, M.G. A century of Alzheimer’s disease. Science 2006. [Google Scholar] [CrossRef] [PubMed]
- Thal, D.R.; Griffin, W.S.T.; de Vos, R.A.I.; Ghebremedhin, E. Cerebral amyloid angiopathy and its relationship to Alzheimer’s disease. Acta Neuropathol. 2008. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Jeong, Y. In vivo Image of Cerebral Amyloid Angiopathy in an Alzheimer’s Disease Mouse Model. J. Stroke 2015, 17, 87. [Google Scholar] [CrossRef] [PubMed]
- Van Dorpe, J.A.; Smeijers, L.; Dewachter, I.; Kurt, S.; Van Den Haute, C.; Tesseur, I.; Sciot, R.; Van Leuven, F. Prominent cerebral amyloid angiopathy in transgenic mice overexpressing the London mutant of human APP in neurons. Neurobiol. Aging 2003. [Google Scholar] [CrossRef][Green Version]
- Daneman, R. The blood-brain barrier in health and disease. Ann. Neurol. 2012. [Google Scholar] [CrossRef] [PubMed]
- Roher, A.E.; Tyas, S.L.; Maarouf, C.L.; Daugs, I.D.; Kokjohn, T.A.; Emmerling, M.R.; Garami, Z.; Belohlavek, M.; Sabbagh, M.N.; Sue, L.I.; et al. Intracranial atherosclerosis as a contributing factor to Alzheimer’s disease dementia. Alzheimer’s Dement. 2011. [Google Scholar] [CrossRef]
- Ryu, J.K.; McLarnon, J.G. A leaky blood-brain barrier, fibrinogen infiltration and microglial reactivity in inflamed Alzheimer’s disease brain. J. Cell. Mol. Med. 2009. [Google Scholar] [CrossRef]
- Erickson, M.A.; Banks, W.A. Blood-brain barrier dysfunction as a cause and consequence of Alzheimer’s disease. J. Cereb. Blood Flow Metab. 2013. [Google Scholar] [CrossRef]
- Yamazaki, Y.; Kanekiyo, T. Blood-brain barrier dysfunction and the pathogenesis of alzheimer’s disease. Int. J. Mol. Sci. 2017, 18, 1965. [Google Scholar] [CrossRef]
- Arvanitakis, Z.; Leurgans, S.E.; Wang, Z.; Wilson, R.S.; Bennett, D.A.; Schneider, J.A. Cerebral amyloid angiopathy pathology and cognitive domains in older persons. Ann. Neurol. 2011. [Google Scholar] [CrossRef] [PubMed]
- Hartz, A.M.S.; Bauer, B.; Soldner, E.L.B.; Wolf, A.; Boy, S.; Backhaus, R.; Mihaljevic, I.; Bogdahn, U.; Klünemann, H.H.; Schuierer, G.; et al. Amyloid-β contributes to blood-brain barrier leakage in transgenic human amyloid precursor protein mice and in humans with cerebral amyloid angiopathy. Stroke 2012. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, S.M.; Gurol, M.E.; Rosand, J.; Smith, E.E. Amyloid angiopathy-related vascular cognitive impairment. Stroke 2004, 35, 2616–2619. [Google Scholar] [CrossRef]
- Pfeifer, L.A.; White, L.R.; Ross, G.W.; Petrovitch, H.; Launer, L.J. Cerebral amyloid angiopathy and cognitive function: The HAAS autopsy study. Neurology 2002. [Google Scholar] [CrossRef]
- Daneman, R.; Prat, A. The Blood–Brain Barrier. Cold Spring Harb. Perspect. Biol. 2015, 7, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Abbott, N.J.; Patabendige, A.A.K.; Dolman, D.E.M.; Yusof, S.R.; Begley, D.J. Structure and function of the blood-brain barrier. Neurobiol. Dis. 2010. [Google Scholar] [CrossRef]
- Attwell, D.; Buchan, A.M.; Charpak, S.; Lauritzen, M.; Macvicar, B.A.; Newman, E.A. Glial and neuronal control of brain blood flow. Nature 2010. [Google Scholar] [CrossRef] [PubMed]
- Muoio, V.; Persson, P.B.; Sendeski, M.M. The neurovascular unit—Concept review. Acta Physiol. 2014. [Google Scholar] [CrossRef]
- Liu, C.Y.; Yang, Y.; Ju, W.N.; Wang, X.; Zhang, H.L. Emerging Roles of Astrocytes in Neuro-Vascular Unit and the Tripartite Synapse With Emphasis on Reactive Gliosis in the Context of Alzheimer’s Disease. Front. Cell. Neurosci. 2018. [Google Scholar] [CrossRef] [PubMed]
- Frost, G.R.; Li, Y.M. The role of astrocytes in amyloid production and Alzheimer’s disease. Open Biol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.C.; Hu, J.; Zhao, N.; Wang, J.; Wang, N.; Cirrito, J.R.; Kanekiyo, T.; Holtzman, D.M.; Bu, G. Astrocytic LRP1 Mediates Brain Aβ Clearance and Impacts Amyloid Deposition. J. Neurosci. 2017. [Google Scholar] [CrossRef]
- Verkhratsky, A.; Olabarria, M.; Noristani, H.N.; Yeh, C.Y.; Rodriguez, J.J. Astrocytes in Alzheimer’s Disease. Neurotherapeutics 2010. [Google Scholar] [CrossRef] [PubMed]
- Heneka, M.T.; Sastre, M.; Dumitrescu-Ozimek, L.; Dewachter, I.; Walter, J.; Klockgether, T.; Van Leuven, F. Focal glial activation coincides with increased BACE1 activation and precedes amyloid plaque deposition in APP[V717I] transgenic mice. J. Neuroinflamm. 2005, 2, 22. [Google Scholar] [CrossRef]
- Price, B.R.; Norris, C.M.; Sompol, P.; Wilcock, D.M. An emerging role of astrocytes in vascular contributions to cognitive impairment and dementia. J. Neurochem. 2018. [Google Scholar] [CrossRef] [PubMed]
- Kulijewicz-Nawrot, M.; Verkhratsky, A.; Chvátal, A.; Syková, E.; Rodríguez, J.J. Astrocytic cytoskeletal atrophy in the medial prefrontal cortex of a triple transgenic mouse model of Alzheimer’s disease. J. Anat. 2012, 221. [Google Scholar] [CrossRef]
- Olabarria, M.; Noristani, H.N.; Verkhratsky, A.; Rodríguez, J.J. Concomitant astroglial atrophy and astrogliosis in a triple transgenic animal model of Alzheimer’s disease. Glia 2010, 58, 831–838. [Google Scholar] [CrossRef]
- Polis, B.; Srikanth, K.D.; Elliott, E.; Gil-Henn, H.; Samson, A.O. L-Norvaline Reverses Cognitive Decline and Synaptic Loss in a Murine Model of Alzheimer’s Disease. Neurotherapeutics 2018. [Google Scholar] [CrossRef] [PubMed]
- Stollg, G.; Jander, S. The role of microglia and macrophages in the pathophysiology of the CNS. Prog. Neurobiol. 1999. [Google Scholar] [CrossRef]
- Zaghi, J.; Goldenson, B.; Inayathullah, M.; Lossinsky, A.S.; Masoumi, A.; Avagyan, H.; Mahanian, M.; Bernas, M.; Weinand, M.; Rosenthal, M.J.; et al. Alzheimer disease macrophages shuttle amyloid-beta from neurons to vessels, contributing to amyloid angiopathy. Acta Neuropathol. 2009. [Google Scholar] [CrossRef][Green Version]
- Hawkes, C.A.; McLaurin, J. Selective targeting of perivascular macrophages for clearance of amyloid in cerebral amyloid angiopathy. Proc. Natl. Acad. Sci. USA 2009. [Google Scholar] [CrossRef]
- Park, L.; Uekawa, K.; Garcia-Bonilla, L.; Koizumi, K.; Murphy, M.; Pistik, R.; Younkin, L.; Younkin, S.; Zhou, P.; Carlson, G.; et al. Brain Perivascular Macrophages Initiate the Neurovascular Dysfunction of Alzheimer Aβ Peptides. Circ. Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Kisumi, M.; Sugiura, M.; Takagi, T.; Chibata, I. Norvaline accumulation by regulatory mutants of Serratia marcescens. J. Antibiot. 1977. [Google Scholar] [CrossRef] [PubMed]
- Polis, B.; Samson, A.O. Arginase as a Potential Target in the Treatment of Alzheimer’s Disease. Adv. Alzheimer’s Dis. 2018, 7, 119–140. [Google Scholar] [CrossRef]
- Ming, X.F.; Rajapakse, A.G.; Carvas, J.M.; Ruffieux, J.; Yang, Z. Inhibition of S6K1 accounts partially for the anti-inflammatory effects of the arginase inhibitor L-norvaline. BMC Cardiovasc. Disord. 2009. [Google Scholar] [CrossRef] [PubMed]
- Pernow, J.; Jung, C. Arginase as a potential target in the treatment of cardiovascular disease: Reversal of arginine steal? Cardiovasc. Res. 2013. [Google Scholar] [CrossRef] [PubMed]
- Montagne, A.; Barnes, S.R.; Sweeney, M.D.; Halliday, M.R.; Sagare, A.P.; Zhao, Z.; Toga, A.W.; Jacobs, R.E.; Liu, C.Y.; Amezcua, L.; et al. Blood-Brain barrier breakdown in the aging human hippocampus. Neuron 2015. [Google Scholar] [CrossRef] [PubMed]
- van de Haar, H.J.; Jansen, J.F.A.; van Osch, M.J.P.; van Buchem, M.A.; Muller, M.; Wong, S.M.; Hofman, P.A.M.; Burgmans, S.; Verhey, F.R.J.; Backes, W.H. Neurovascular unit impairment in early Alzheimer’s disease measured with magnetic resonance imaging. Neurobiol. Aging 2016. [Google Scholar] [CrossRef]
- Montagne, A.; Zhao, Z.; Zlokovic, B.V. Alzheimer’ s disease: A matter of blood—Brain barrier dysfunction? J. Exp. Med. 2017, 214, 3151–3169. [Google Scholar] [CrossRef]
- Tibbling, G.; Link, H.; Öhman, S. Principles of albumin and igg analyses in neurological disorders. I. Establishment of reference values. Scand. J. Clin. Lab. Investig. 1977. [Google Scholar] [CrossRef]
- Elovaara, I.; Icen, A.; Palo, J.; Erkinjuntti, T. CSF in Alzheimer’s disease. Studies on blood-brain barrier function and intrathecal protein synthesis. J. Neurol. Sci. 1985. [Google Scholar] [CrossRef]
- Alafuzoff, I.; Adolfsson, R.; Bucht, G.; Winblad, B. Albumin and immunoglobulin in plasma and cerebrospinal fluid, and blood-cerebrospinal fluid barrier function in patients with dementia of alzheimer type and multi-infarct dementia. J. Neurol. Sci. 1983. [Google Scholar] [CrossRef]
- Ezra, A.; Rabinovich-Nikitin, I.; Rabinovich-Toidman, P.; Solomon, B. Multifunctional Effect of Human Serum Albumin Reduces Alzheimer’s Disease Related Pathologies in the 3xTg Mouse Model. J. Alzheimer’s Dis. 2015. [Google Scholar] [CrossRef] [PubMed]
- d’Uscio, L.V.; He, T.; Katusic, Z.S. Expression and Processing of Amyloid Precursor Protein in Vascular Endothelium. Physiology 2017. [Google Scholar] [CrossRef] [PubMed]
- Kitazume, S.; Tachida, Y.; Kato, M.; Yamaguchi, Y.; Honda, T.; Hashimoto, Y.; Wada, Y.; Saito, T.; Iwata, N.; Saido, T.; et al. Brain endothelial cells produce amyloid β from amyloid precursor protein 770 and preferentially secrete the O-glycosylated form. J. Biol. Chem. 2010. [Google Scholar] [CrossRef] [PubMed]
- Polis, B.; Srikanth, K.; Gurevich, V.; Gil-Henn, H.; Samson, A. L-Norvaline, a new therapeutic agent against Alzheimer’s disease. Neural Regen. Res. 2019, 14, 1562. [Google Scholar] [CrossRef] [PubMed]
- Prat, A.; Biernacki, K.; Wosik, K.; Antel, J.P. Glial cell influence on the human blood-brain barrier. Glia 2001. [Google Scholar] [CrossRef] [PubMed]
- Nedergaard, M.; Ransom, B.; Goldman, S.A. New roles for astrocytes: Redefining the functional architecture of the brain. Trends Neurosci. 2003. [Google Scholar] [CrossRef] [PubMed]
- Verkhratsky, A.; Parpura, V. Astrogliopathology in neurological, neurodevelopmental and psychiatric disorders. Neurobiol. Dis. 2016. [Google Scholar] [CrossRef] [PubMed]
- Fiala, M.; Liu, Q.N.; Sayre, J.; Pop, V.; Brahmandam, V.; Graves, M.C.; Vinters, H.V. Cyclooxygenase-2-positive macrophages infiltrate the Alzheimer’s disease brain and damage the blood-brain barrier. Eur. J. Clin. Investig. 2002. [Google Scholar] [CrossRef]
- Mittelbronn, M.; Dietz, K.; Schluesener, H.J.; Meyermann, R. Local distribution of microglia in the normal adult human central nervous system differs by up to one order of magnitude. Acta Neuropathol. 2001. [Google Scholar] [CrossRef]
- Ito, D.; Imai, Y.; Ohsawa, K.; Nakajima, K.; Fukuuchi, Y.; Kohsaka, S. Microglia-specific localisation of a novel calcium binding protein, Iba1. Mol. Brain Res. 1998. [Google Scholar] [CrossRef]
- Lan, X.; Han, X.; Li, Q.; Yang, Q.W.; Wang, J. Modulators of microglial activation and polarization after intracerebral haemorrhage. Nat. Rev. Neurol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Austin, S.A.; Santhanam, A.V.; Hinton, D.J.; Choi, D.S.; Katusic, Z.S. Endothelial nitric oxide deficiency promotes Alzheimer’s disease pathology. J. Neurochem. 2013, 127, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Freudenberg, F.; Alttoa, A.; Reif, A. Neuronal nitric oxide synthase (NOS1) and its adaptor, NOS1AP, as a genetic risk factors for psychiatric disorders. Genes Brain Behav. 2015. [Google Scholar] [CrossRef] [PubMed]
- Blum-Degen, D.; Heinemann, T.; Lan, J.; Pedersen, V.; Leblhuber, F.; Paulus, W.; Riederer, P.; Gerlach, M. Characterization and regional distribution of nitric oxide synthase in the human brain during normal ageing. Brain Res. 1999. [Google Scholar] [CrossRef]
- Bachetti, T.; Comini, L.; Curello, S.; Bastianon, D.; Palmieri, M.; Bresciani, G.; Callea, F.; Ferrari, R. Co-expression and modulation of neuronal and endothelial nitric oxide synthase in human endothelial cells. J. Mol. Cell. Cardiol. 2004. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, S.; Chan, C.K.; Jiang, Y.; Davidge, S.T. Neuronal nitric oxide synthase regulates endothelial inflammation. J. Leukoc. Biol. 2012. [Google Scholar] [CrossRef]
- Finkel, J.; Guptill, V.; Khaibullina, A.; Spornick, N.; Vasconcelos, O.; Liewehr, D.J.; Steinberg, S.M.; Quezado, Z.M.N. The three isoforms of nitric oxide synthase distinctively affect mouse nocifensive behavior. Nitric Oxide Biol. Chem. 2012. [Google Scholar] [CrossRef]
- Colton, C.A.; Wilcock, D.M.; Wink, D.A.; Davis, J.; Van Nostrand, W.E.; Vitek, M.P. The effects of NOS2 gene deletion on mice expressing mutated human AβPP. J. Alzheimer’s Dis. 2008. [Google Scholar] [CrossRef]
- Colton, C.A.; Vitek, M.P.; Wink, D.A.; Xu, Q.; Cantillana, V.; Previti, M.L.; Van Nostrand, W.E.; Weinberg, J.B.; Dawson, H. NO synthase 2 (NOS2) deletion promotes multiple pathologies in a mouse model of Alzheimer’s disease. Proc. Natl. Acad. Sci. USA 2006. [Google Scholar] [CrossRef]
- Austin, S.A.; Santhanam, A.V.; Katusic, Z.S. Endothelial nitric oxide modulates expression and processing of amyloid precursor protein. Circ. Res. 2010. [Google Scholar] [CrossRef]
- Austin, S.A.; D’uscio, L.V.; Katusic, Z.S. Supplementation of Nitric Oxide Attenuates AβPP and BACE1 Protein in Cerebral Microcirculation of eNOS-Deficient Mice. J. Alzheimer’s Dis. 2013. [Google Scholar] [CrossRef] [PubMed]
- Kwak, Y.D.; Wang, R.; Li, J.J.; Zhang, Y.W.; Xu, H.; Liao, F.F. Differential regulation of BACE1 expression by oxidative and nitrosative signals. Mol. Neurodegener. 2011. [Google Scholar] [CrossRef] [PubMed]
- Wink, D.A.; Miranda, K.M.; Espey, M.G.; Pluta, R.M.; Hewett, S.J.; Colton, C.; Vitek, M.; Feelisch, M.; Grisham, M.B. Mechanisms of the Antioxidant Effects of Nitric Oxide. Antioxid. Redox Signal. 2002. [Google Scholar] [CrossRef] [PubMed]
- Chiueh, C.C. Neuroprotective properties of nitric oxide. Ann. N. Y. Acad. Sci. 1999, 890, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Hiramoto, K.; Ohkawa, T.; Oikawa, N.; Kikugawa, K. Is Nitric Oxide (NO) an Antioxidant or a Prooxidant for Lipid Peroxidation? Chem. Pharm. Bull. 2003. [Google Scholar] [CrossRef][Green Version]
- Chakroborty, S.; Kim, J.; Schneider, C.; West, A.R.; Stutzmann, G.E. Nitric Oxide Signaling Is Recruited As a Compensatory Mechanism for Sustaining Synaptic Plasticity in Alzheimer’s Disease Mice. J. Neurosci. 2015. [Google Scholar] [CrossRef] [PubMed]
- De Caterina, R.; Libby, P.; Peng, H.B.; Thannickal, V.J.; Rajavashisth, T.B.; Gimbrone, M.A.; Shin, W.S.; Liao, J.K. Nitric oxide decreases cytokine-induced endothelial activation: Nitric oxide selectively reduces endothelial expression of adhesion molecules and proinflammatory cytokines. J. Clin. Investig. 1995. [Google Scholar] [CrossRef]
- Fonar, G.; Polis, B.; Meirson, T.; Maltsev, A.; Elliott, E.; Samson, A.O. Intracerebroventricular administration of L-arginine improves spatial memory acquisition in triple transgenic mice via reduction of oxidative stress and apoptosis. Transl. Neurosci. 2018. [Google Scholar] [CrossRef]
- Ohtsuka, Y.; Nakaya, J. Effect of oral administration of L-arginine on senile dementia. Am. J. Med. 2000, 108, 2000. [Google Scholar] [CrossRef]
- Tachikawa, M.; Hosoya, K. Transport characteristics of guanidino compounds at the blood-brain barrier and blood-cerebrospinal fluid barrier: relevance to neural disorders. Fluids Barriers CNS 2011, 8, 13. [Google Scholar] [CrossRef] [PubMed]
- de Vera, N.; Serratosa, J.; Artigas, F.; Martínez, E. Toxic effects of putrescine in rat brain: Polyamines can be involved in the action of excitotoxins. Amino Acids 1992. [Google Scholar] [CrossRef]
- Sparapani, M.; Dall’Olio, R.; Gandolfi, O.; Ciani, E.; Contestabile, A. Neurotoxicity of Polyamines and Pharmacological Neuroprotection in Cultures of Rat Cerebellar Granule Cells. Exp. Neurol. 1997. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Fleete, M.S.; Jing, Y.; Collie, N.D.; Curtis, M.A.; Waldvogel, H.J.; Faull, R.L.M.; Abraham, W.C.; Zhang, H. Altered arginine metabolism in Alzheimer’s disease brains. Neurobiol. Aging 2014, 35, 1992–2003. [Google Scholar] [CrossRef] [PubMed]
- Nedvetsky, P.I.; Sessa, W.C.; Schmidt, H.H.H.W. There’s NO binding like NOS binding: Protein-protein interactions in NO/cGMP signaling. Proc. Natl. Acad. Sci. USA 2002, 99, 16510–16512. [Google Scholar] [CrossRef] [PubMed]
- Fryer, J.D. Human Apolipoprotein E4 Alters the Amyloid 40:42 Ratio and Promotes the Formation of Cerebral Amyloid Angiopathy in an Amyloid Precursor Protein Transgenic Model. J. Neurosci. 2005. [Google Scholar] [CrossRef]
- Van Nostrand, W.E.; Xu, F.; Rozemuller, A.J.M.; Colton, C.A. Enhanced capillary amyloid angiopathy-associated pathology in Tg-SwDI mice with deleted nitric oxide synthase 2. Stroke 2010, 41, S135–S138. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Serrano-Pozo, A.; Frosch, M.P.; Masliah, E.; Hyman, B.T. Neuropathological alterations in Alzheimer disease. Cold Spring Harb. Perspect. Med. 2011. [Google Scholar] [CrossRef]
- Wada, H. Blood-Brain Barrier Permeability of the Demented Elderly as Studied by Cerebrospinal Fluid-Serum Albumin Ratio. Intern. Med. 2008, 37, 509–513. [Google Scholar] [CrossRef][Green Version]
- Yan, P.; Zhu, A.; Liao, F.; Xiao, Q.; Kraft, A.W.; Gonzales, E.; Perez, R.; Greenberg, S.M.; Holtzman, D.M.; Lee, J.M. Minocycline Reduces Spontaneous Hemorrhage in Mouse Models of Cerebral Amyloid Angiopathy. Stroke 2015. [Google Scholar] [CrossRef]
- Merlini, M.; Meyer, E.P.; Ulmann-Schuler, A.; Nitsch, R.M. Vascular β-amyloid and early astrocyte alterations impair cerebrovascular function and cerebral metabolism in transgenic arcAβ mice. Acta Neuropathol. 2011. [Google Scholar] [CrossRef] [PubMed]
- Hopperton, K.E.; Mohammad, D.; Trépanier, M.O.; Giuliano, V.; Bazinet, R.P. Markers of microglia in post-mortem brain samples from patients with Alzheimer’s disease: A systematic review. Mol. Psychiatry 2018. [Google Scholar] [CrossRef] [PubMed]
- Minett, T.; Classey, J.; Matthews, F.E.; Fahrenhold, M.; Taga, M.; Brayne, C.; Ince, P.G.; Nicoll, J.A.R.; Boche, D. Microglial immunophenotype in dementia with Alzheimer’s pathology. J. Neuroinflamm. 2016. [Google Scholar] [CrossRef] [PubMed]
- Oddo, S.; Caccamo, A.; Shepherd, J.D.; Murphy, M.P.; Golde, T.E.; Kayed, R.; Metherate, R.; Mattson, M.P.; Akbari, Y.; LaFerla, F.M. Triple-transgenic model of Alzheimer’s Disease with plaques and tangles: Intracellular Aβ and synaptic dysfunction. Neuron 2003, 39, 409–421. [Google Scholar] [CrossRef]
- Wolman, M.; Klatzo, I.; Chui, E.; Wilmes, F.; Nishimoto, K.; Fujiwara, K.; Spatz, M. Evaluation of the dye-protein tracers in pathophysiology of the blood-brain barrier. Acta Neuropathol. 1981, 54, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Uyama, O.; Okamura, N.; Yanase, M.; Narita, M.; Kawabata, K.; Sugita, M. Quantitative evaluation of vascular permeability in the gerbil brain after transient ischemia using Evans blue fluorescence. J. Cereb. Blood Flow Metab. 1988. [Google Scholar] [CrossRef]
- Yen, L.F.; Wei, V.C.; Kuo, E.Y.; Lai, T.W. Distinct Patterns of Cerebral Extravasation by Evans Blue and Sodium Fluorescein in Rats. PLoS ONE 2013. [Google Scholar] [CrossRef]
- Ujiie, M.; Dickstein, D.L.; Carlow, D.A.; Jefferies, W.A. Blood-brain barrier permeability precedes senile plaque formation in an Alzheimer disease model. Microcirculation 2003. [Google Scholar] [CrossRef]
- Yao, L.; Xue, X.; Yu, P.; Ni, Y.; Chen, F. Evans Blue Dye: A Revisit of Its Applications in Biomedicine. Contrast Media Mol. Imaging 2018. [Google Scholar] [CrossRef]
- Sultan, F. Dissection of Different Areas from Mouse Hippocampus. BIO-PROTOCOL 2016. [Google Scholar] [CrossRef]
- Duelli, R.; Kuschinsky, W. Changes in brain capillary diameter during hypocapnia and hypercapnia. J. Cereb. Blood Flow Metab. 1993. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Liao, Y.; Morgan, S.; Mathur, R.; Feustel, P.; Mazurkiewicz, J.; Qian, J.; Chang, J.; Mathern, G.W.; Adamo, M.A.; et al. Noninflammatory Changes of Microglia Are Sufficient to Cause Epilepsy. Cell Rep. 2018. [Google Scholar] [CrossRef] [PubMed]
- Gilinsky, M.A.; Polityko, Y.K.; Markel, A.L.; Latysheva, T.V.; Samson, A.O.; Polis, B.; Naumenko, S.E. Norvaline reduces blood pressure and induces diuresis in rats with inherited stress-induced arterial hypertension. bioRxiv 2019, 678839. [Google Scholar] [CrossRef]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polis, B.; Gurevich, V.; Assa, M.; Samson, A.O. Norvaline Restores the BBB Integrity in a Mouse Model of Alzheimer’s Disease. Int. J. Mol. Sci. 2019, 20, 4616. https://doi.org/10.3390/ijms20184616
Polis B, Gurevich V, Assa M, Samson AO. Norvaline Restores the BBB Integrity in a Mouse Model of Alzheimer’s Disease. International Journal of Molecular Sciences. 2019; 20(18):4616. https://doi.org/10.3390/ijms20184616
Chicago/Turabian StylePolis, Baruh, Vyacheslav Gurevich, Michael Assa, and Abraham O. Samson. 2019. "Norvaline Restores the BBB Integrity in a Mouse Model of Alzheimer’s Disease" International Journal of Molecular Sciences 20, no. 18: 4616. https://doi.org/10.3390/ijms20184616
APA StylePolis, B., Gurevich, V., Assa, M., & Samson, A. O. (2019). Norvaline Restores the BBB Integrity in a Mouse Model of Alzheimer’s Disease. International Journal of Molecular Sciences, 20(18), 4616. https://doi.org/10.3390/ijms20184616




