Preparation of Gelatin and Gelatin/Hyaluronic Acid Cryogel Scaffolds for the 3D Culture of Mesothelial Cells and Mesothelium Tissue Regeneration
Abstract
1. Introduction
2. Results and Discussion
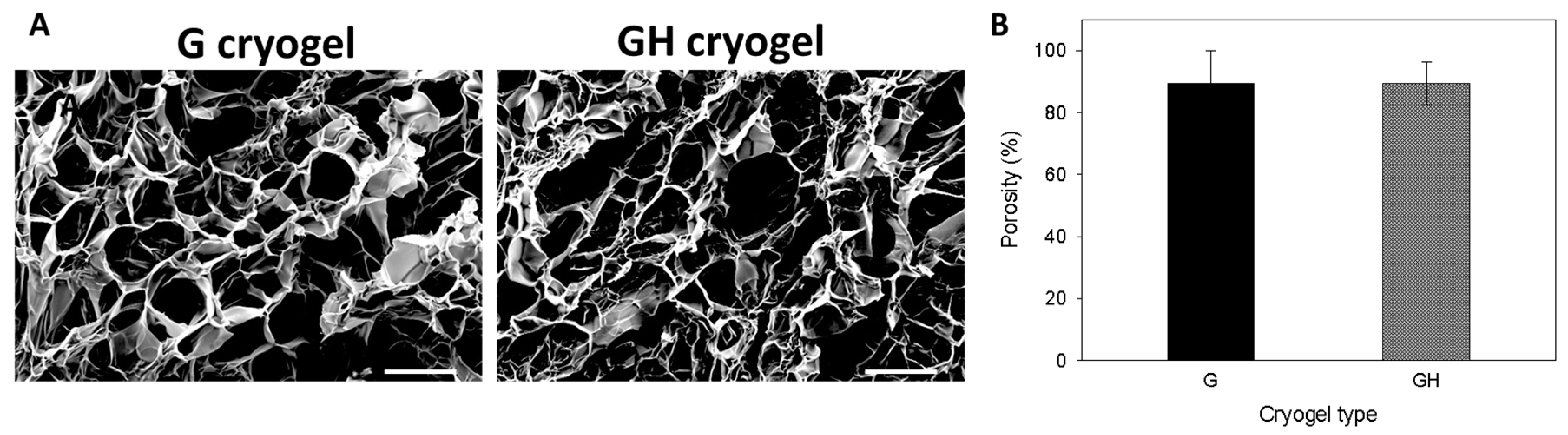
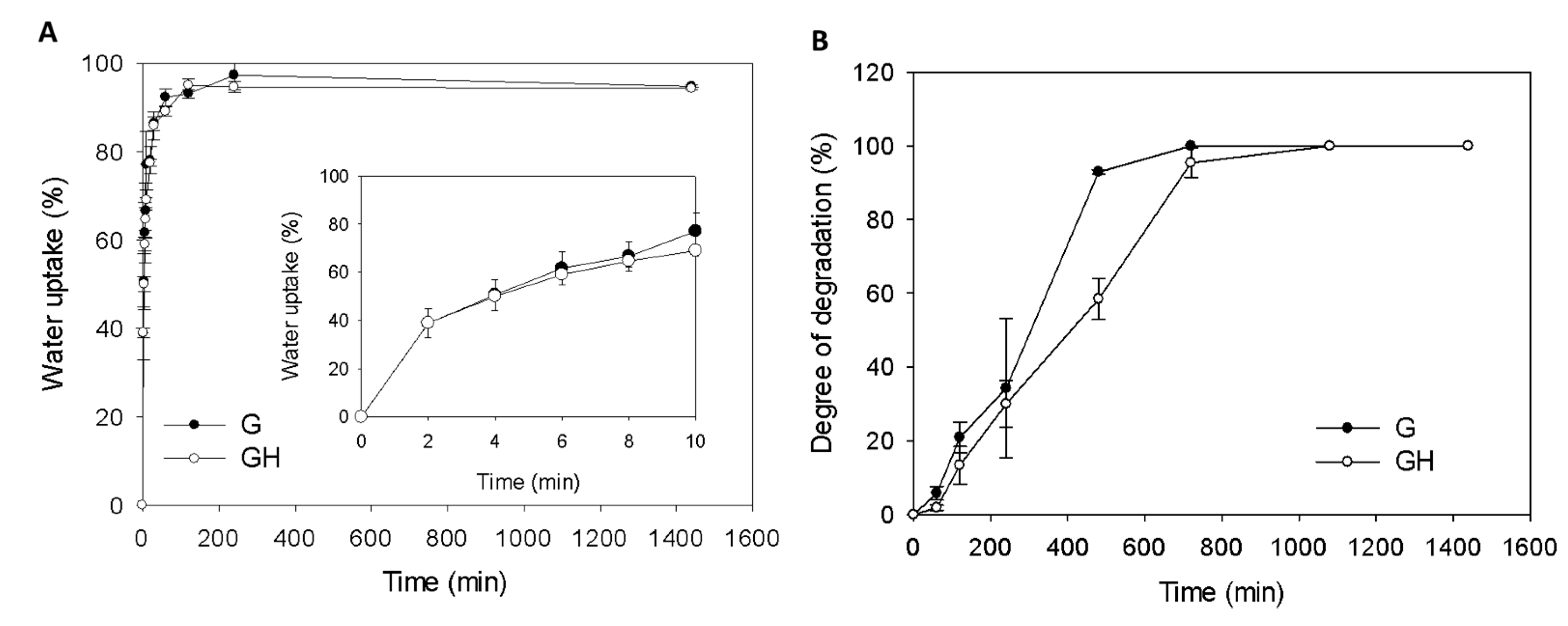
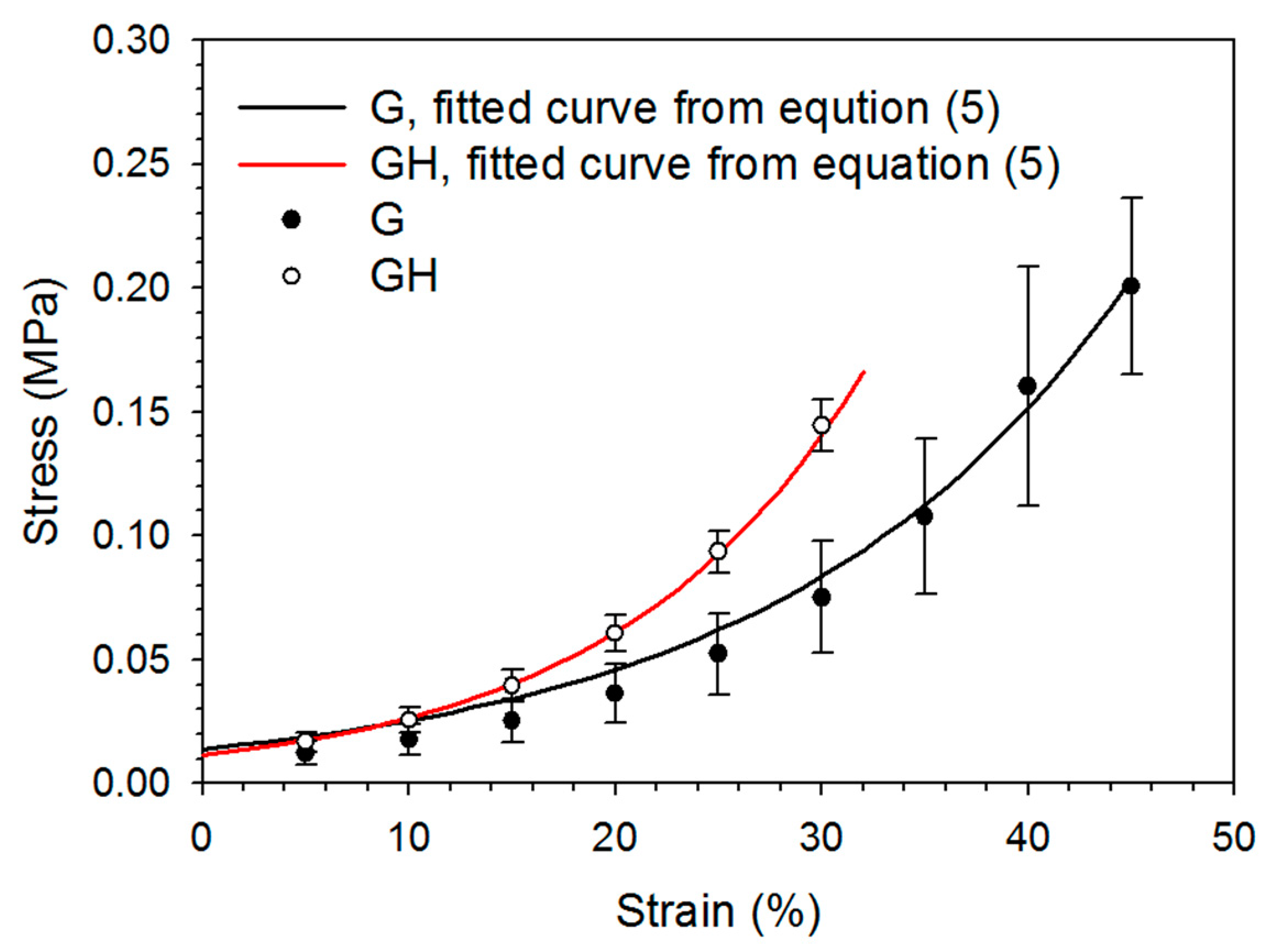
2.1. Synthesis and Characterization of Gelatin (G) and Gelatin/Hyaluronic Acid (HA) Cryogels
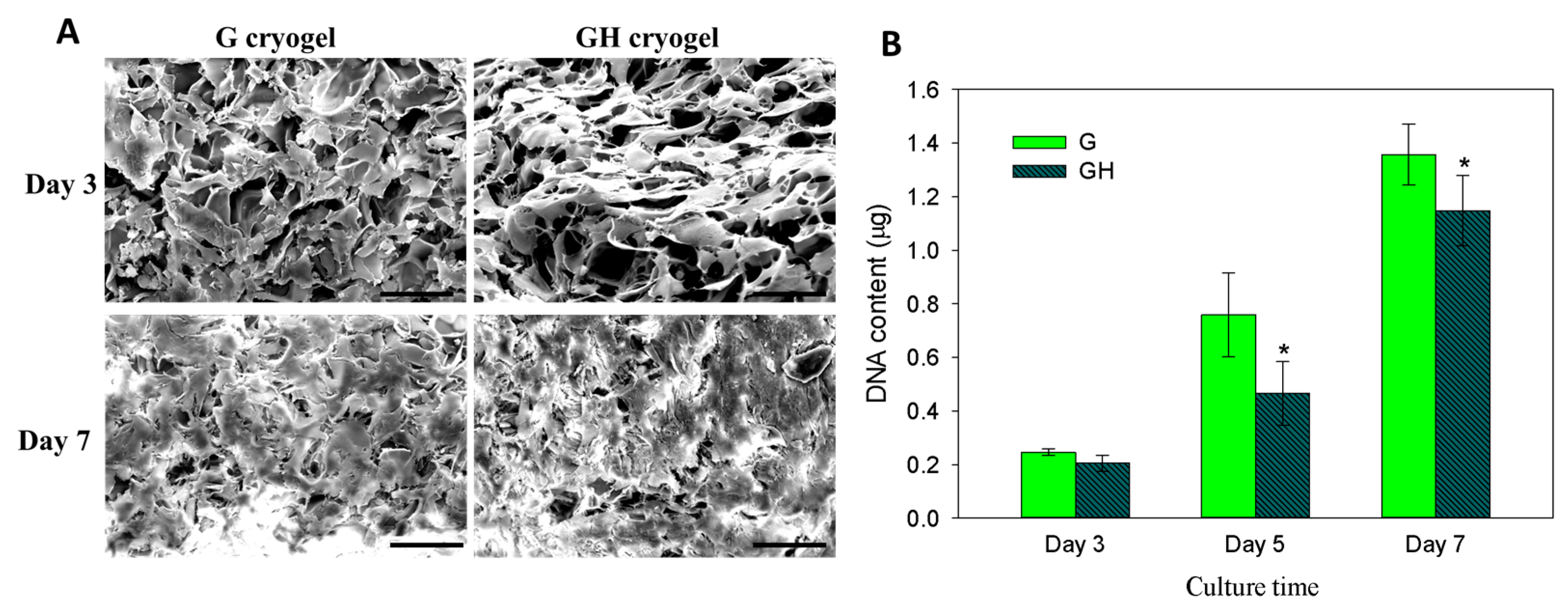
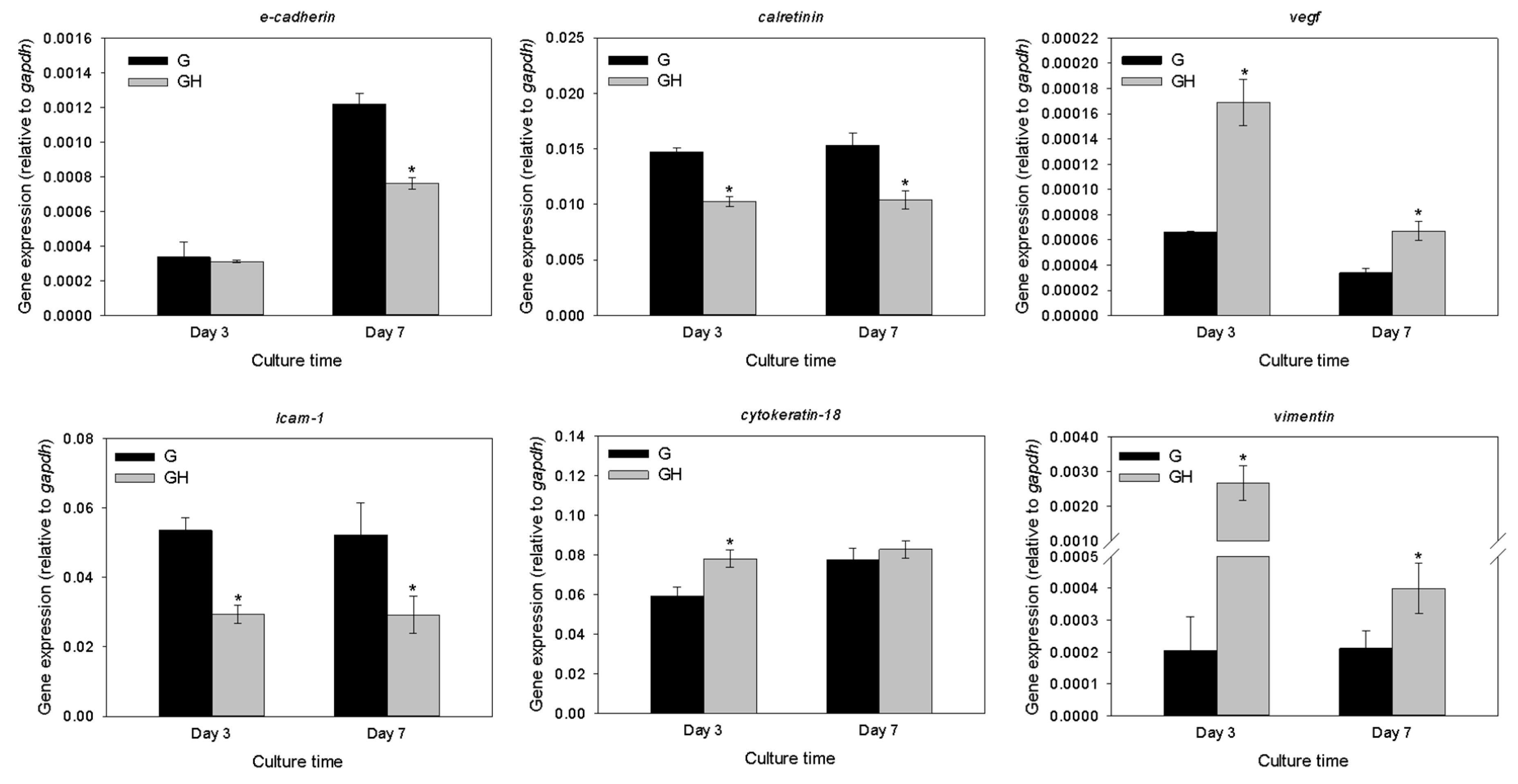
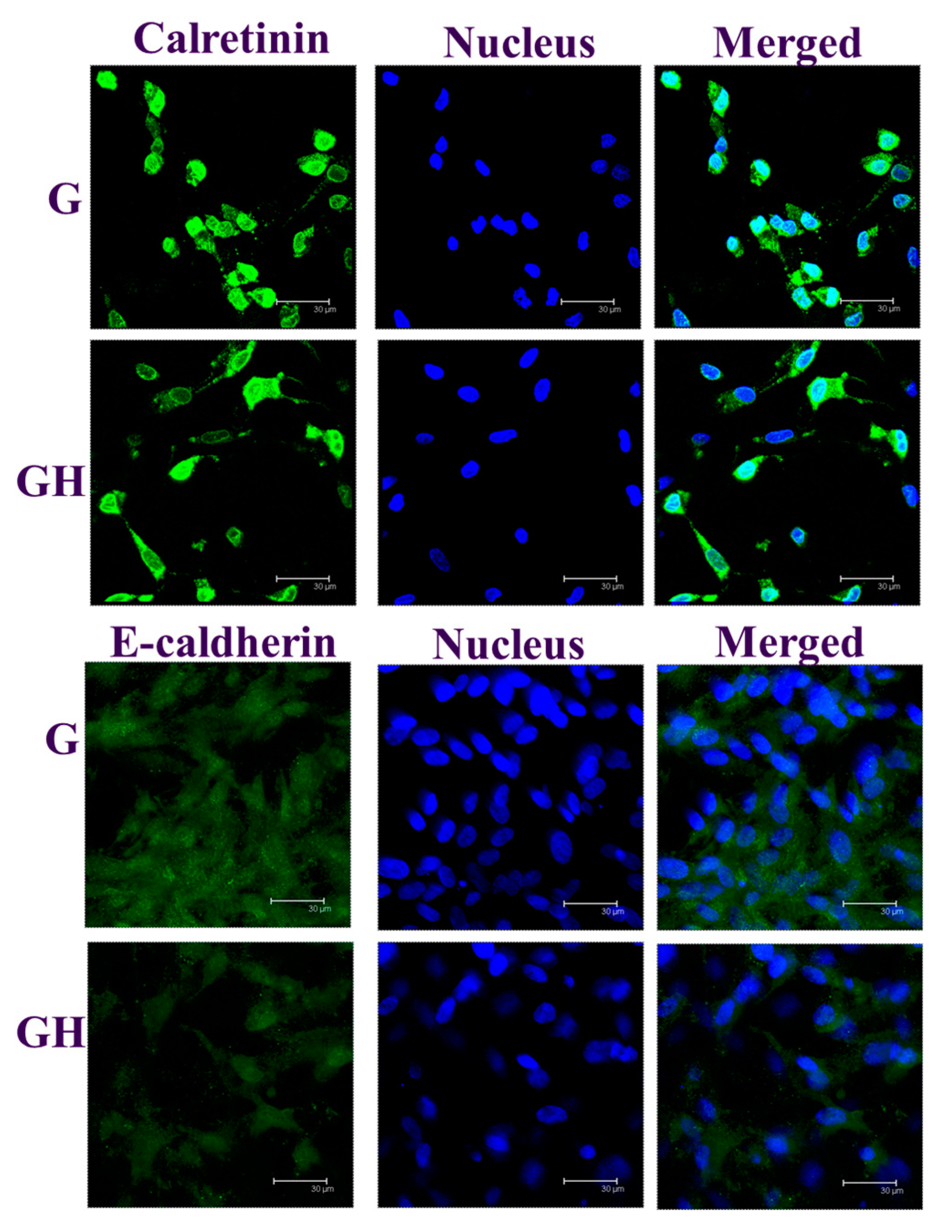
2.2. In Vitro Cell Culture
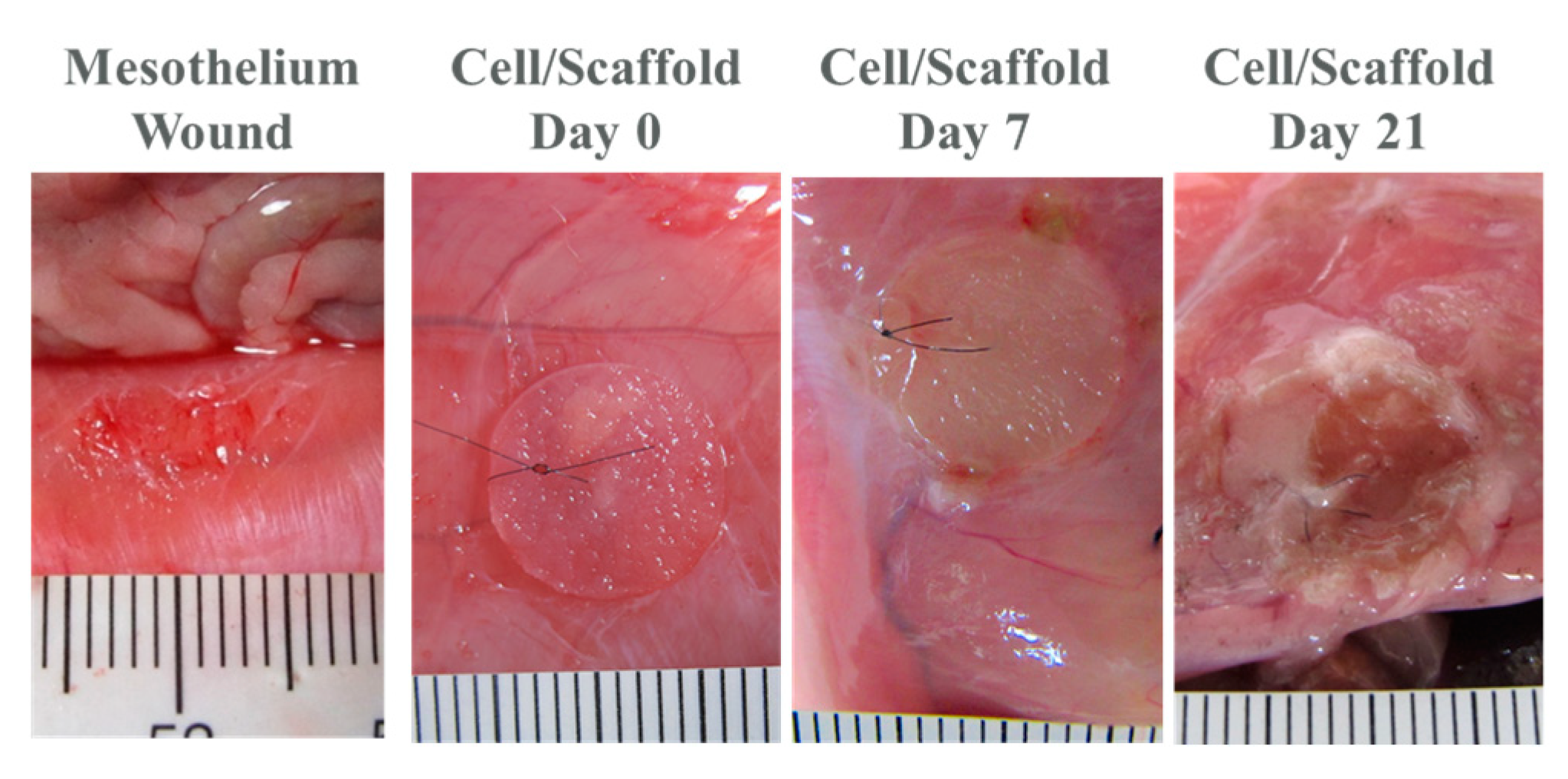
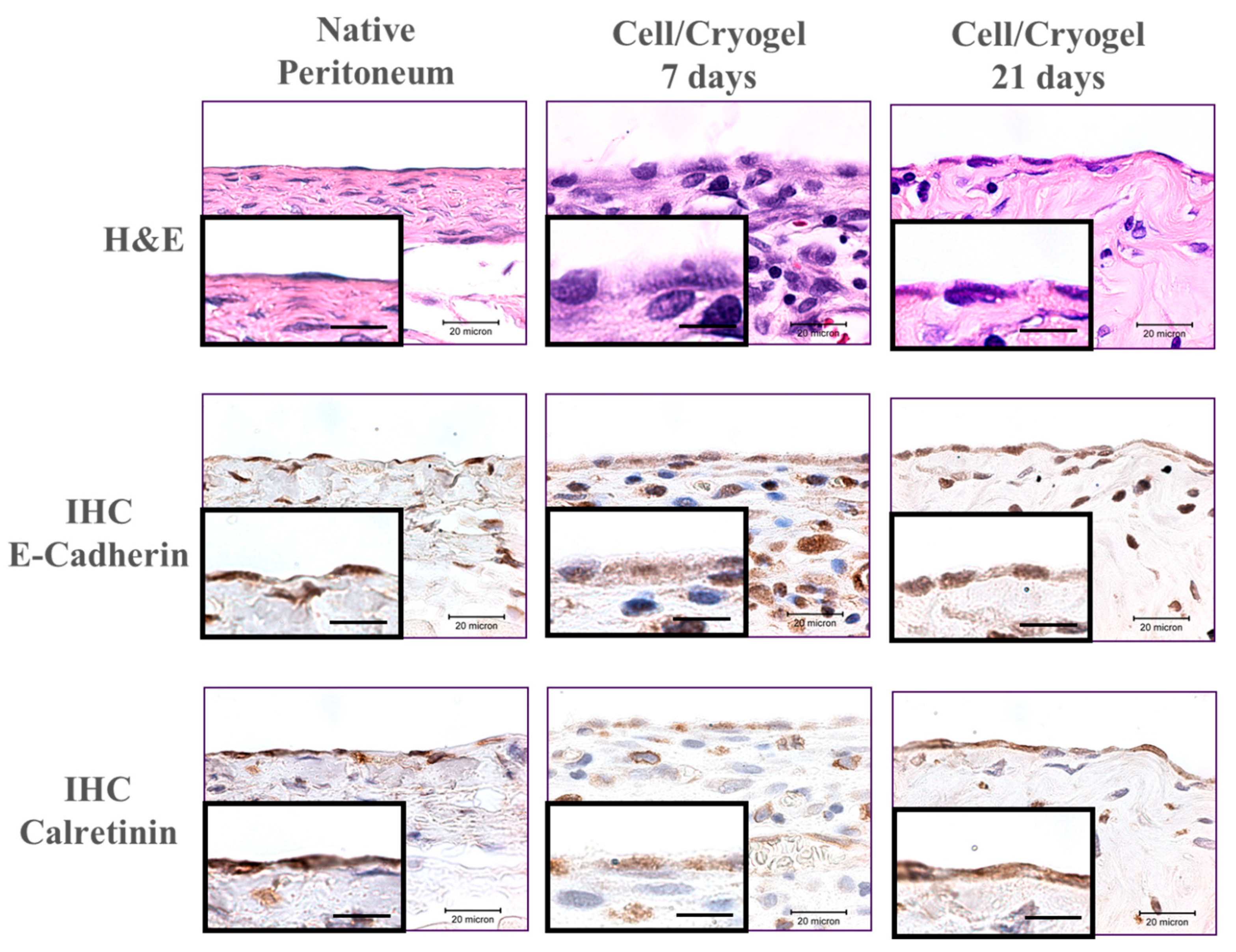
2.3. In Vivo Studies
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Preparation of Gelatin (G) and Gelatin-Hyaluronic Acid (GH) Scaffold by Cryogelation
4.3. Characteristics of Cryogel
4.4. Culture of Mesothelial Cells in Cryogel Scaffolds
4.4.1. Isolation and Harvest of Mesothelial Cells
4.4.2. In Vitro Cell Culture
4.4.3. SEM Analysis
4.4.4. DNA Quantification
4.4.5. Live/Dead Staining
4.4.6. Cell Cytoskeleton Staining
4.4.7. Quantitative Real-Time Polymerase Chain Reaction (qPCR)
4.4.8. Immunofluorescence (IF) Staining
4.5. In Vivo Studies
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mutsaers, S.E. The mesothelial cell. Int. J. Biochem. Cell Biol. 2004, 36, 9–16. [Google Scholar] [CrossRef]
- Yung, S.; Chan, T.M. Pathophysiological changes to the peritoneal membrane during PD-related peritonitis: The role of mesothelial cells. Mediat. Inflamm. 2012, 2012, 484167. [Google Scholar] [CrossRef] [PubMed]
- Margetts, P.J.; Churchill, D.N. Acquired ultrafiltration dysfunction in peritoneal dialysis patients. J. Am. Soc. Nephrol. 2002, 13, 2787–2794. [Google Scholar]
- O’Brien, F.J. Biomaterials & scaffolds for tissue engineering. Mater. Today 2011, 14, 88–95. [Google Scholar]
- Bencherif, S.A.; Sands, R.W.; Bhatta, D.; Arany, P.; Verbeke, C.S.; Edwards, D.A.; Mooney, D.J. Injectable preformed scaffolds with shape-memory properties. Proc. Natl. Acad. Sci. USA 2012, 109, 19590–19595. [Google Scholar] [CrossRef] [PubMed]
- Dainiak, M.B.; Kumar, A.; Galaev, I.Y.; Mattiasson, B. Detachment of affinity-captured bioparticles by elastic deformation of a macroporous hydrogel. Proc. Natl. Acad. Sci. USA 2006, 103, 849–854. [Google Scholar] [CrossRef]
- Kumar, A.; Srivastava, A. Cell separation using cryogel-based affinity chromatography. Nat. Protoc. 2010, 5, 1737–1747. [Google Scholar] [CrossRef]
- Lozinsky, V.I.; Plieva, F.M.; Galaev, I.Y.; Mattiasson, B. The potential of polymeric cryogels in bioseparation. Bioseparation 2001, 10, 163–188. [Google Scholar] [CrossRef] [PubMed]
- Lozinsky, V.I.; Galaev, I.Y.; Plieva, F.M.; Savina, I.N.; Jungvid, H.; Mattiasson, B. Polymeric cryogels as promising materials of biotechnological interest. Trends Biotechnol. 2003, 21, 445–451. [Google Scholar] [CrossRef]
- Hwang, Y.; Sangaj, N.; Varghese, S. Interconnected macroporous poly(ethylene glycol) cryogels as a cell scaffold for cartilage tissue engineering. Tissue Eng. Part. A 2010, 16, 3033–3041. [Google Scholar] [CrossRef]
- Chang, K.H.; Liao, H.T.; Chen, J.P. Preparation and characterization of gelatin/hyaluronic acid cryogels for adipose tissue engineering: In vitro and in vivo studies. Acta Biomater. 2013, 9, 9012–9026. [Google Scholar] [CrossRef]
- Sharma, A.; Bhat, S.; Nayak, V.; Kumar, A. Efficacy of supermacroporous poly(ethylene glycol)-gelatin cryogel matrix for soft tissue engineering applications. Mater. Sci. Eng. CMater. Biol. Appl. 2015, 47, 298–312. [Google Scholar] [CrossRef]
- Liao, H.-T.; Shalumon, K.T.; Chang, K.-H.; Sheu, C.; Chen, J.-P. Investigation of synergistic effects of inductive and conductive factors in gelatin-based cryogels for bone tissue engineering. J. Mater. Chem. B 2016, 4, 1827–1841. [Google Scholar] [CrossRef]
- Chang, C.H.; Liu, H.C.; Lin, C.C.; Chou, C.H.; Lin, F.H. Gelatin-chondroitin-hyaluronan tri-copolymer scaffold for cartilage tissue engineering. Biomaterials 2003, 24, 4853–4858. [Google Scholar] [CrossRef]
- Yin, B.; Ma, P.; Chen, J.; Wang, H.; Wu, G.; Li, B.; Li, Q.; Huang, Z.; Qiu, G.; Wu, Z. Hybrid macro-porous titanium ornamented by degradable 3d gel/nha micro-scaffolds for bone tissue regeneration. Int. J. Mol. Sci. 2016, 17, 575. [Google Scholar] [CrossRef]
- Echave, M.C.; Saenz del Burgo, L.; Pedraz, J.L.; Orive, G. Gelatin as biomaterial for tissue engineering. Curr. Pharm. Des. 2017, 23, 3567–3584. [Google Scholar] [CrossRef]
- Borzacchiello, A.; Mayol, L.; Ramires, P.A.; Pastorello, A.; Di Bartolo, C.; Ambrosio, L.; Milella, E. Structural and rheological characterization of hyaluronic acid-based scaffolds for adipose tissue engineering. Biomaterials 2007, 28, 4399–4408. [Google Scholar] [CrossRef]
- Khare, A.R.; Peppas, N.A. Swelling/deswelling of anionic copolymer gels. Biomaterials 1995, 16, 559–567. [Google Scholar] [CrossRef]
- Brulez, H.F.; Verbrugh, H.A. First-line defense mechanisms in the peritoneal cavity during peritoneal dialysis. Perit. Dial. Int. 1995, 15, S24–S33. [Google Scholar]
- Stylianou, E.; Jenner, L.A.; Davies, M.; Coles, G.A.; Williams, J.D. Isolation, culture and characterization of human peritoneal mesothelial cells. Kidney Int. 1990, 37, 1563–1570. [Google Scholar] [CrossRef]
- Kuga, H.; Morisaki, T.; Nakamura, K.; Onishi, H.; Matsuda, T.; Sueishi, K.; Tanaka, M.; Katano, M. Construction of a transplantable tissue-engineered artificial peritoneum. Eur. Surg. Res. Eur. Chir. Forsch. Rech. Chir. Eur. 2004, 36, 323–330. [Google Scholar] [CrossRef]
- Takazawa, R.; Yamato, M.; Kageyama, Y.; Okano, T.; Kihara, K. Mesothelial cell sheets cultured on fibrin gel prevent adhesion formation in an intestinal hernia model. Tissue Eng. 2005, 11, 618–625. [Google Scholar] [CrossRef]
- Inagaki, N.F.; Inagaki, F.F.; Kokudo, N.; Miyajima, A. Cell-based therapy for preventing postoperative adhesion and promoting regeneration after hepatectomy. J. Hepato-Biliary-Pancreat. Sci. 2015, 22, 524–530. [Google Scholar] [CrossRef]
- Kawanishi, K.; Yamato, M.; Sakiyama, R.; Okano, T.; Nitta, K. Peritoneal cell sheets composed of mesothelial cells and fibroblasts prevent intra-abdominal adhesion formation in a rat model. Jounal Tissue Eng. Regen. Med. 2016, 10, 855–866. [Google Scholar] [CrossRef]
- Tripathi, A.; Kumar, A. Multi-featured macroporous agarose-alginate cryogel: Synthesis and characterization for bioengineering applications. Macromol. Biosci. 2011, 11, 22–35. [Google Scholar] [CrossRef]
- Zeltinger, J.; Sherwood, J.K.; Graham, D.A.; Mueller, R.; Griffith, L.G. Effect of pore size and void fraction on cellular adhesion, proliferation, and matrix deposition. Tissue Eng. 2001, 7, 557–572. [Google Scholar] [CrossRef]
- Jain, E.; Srivastava, A.; Kumar, A. Macroporous interpenetrating cryogel network of poly(acrylonitrile) and gelatin for biomedical applications. J. Mater. Sci. Mater. Med. 2009, 20, S173–S179. [Google Scholar] [CrossRef]
- Jayakumar, R.; Ramachandran, R.; Sudheesh Kumar, P.T.; Divyarani, V.V.; Srinivasan, S.; Chennazhi, K.P.; Tamura, H.; Nair, S.V. Fabrication of chitin–chitosan/nano ZrO2 composite scaffolds for tissue engineering applications. Int. J. Biol. Macromol. 2011, 49, 274–280. [Google Scholar] [CrossRef]
- Faturechi, R.; Karimi, A.; Hashemi, S.A.; Navidbakhsh, M. Mechanical characterization of peritoneum/fascia under uniaxial loading. J. Biomater. Tissue Eng. 2014, 4, 189–193. [Google Scholar] [CrossRef]
- White, E.J.; Cunnane, E.M.; McMahon, M.; Walsh, M.T.; Coffey, J.C.; O’Sullivan, L. Mechanical characterisation of porcine non-intestinal colorectal tissues for innovation in surgical instrument design. Proc. Inst. Mech. Eng. Part. H J. Eng. Med. 2018, 232, 796–806. [Google Scholar] [CrossRef]
- McKenzie, A.J.; Hicks, S.R.; Svec, K.V.; Naughton, H.; Edmunds, Z.L.; Howe, A.K. The mechanical microenvironment regulates ovarian cancer cell morphology, migration, and spheroid disaggregation. Sci. Rep. 2018, 8, 7228. [Google Scholar] [CrossRef]
- Sarac, T.P.; Carnevale, K.; Smedira, N.; Tanquilut, E.; Augustinos, P.; Patel, A.; Naska, T.; Clair, D.; Ouriel, K. In vivo and mechanical properties of peritoneum/fascia as a novel arterial substitute. J. Vasc. Surg. 2005, 41, 490–497. [Google Scholar] [CrossRef][Green Version]
- Mutsaers, S.E. Mesothelial cells: Their structure, function and role in serosal repair. Respirology 2002, 7, 171–191. [Google Scholar] [CrossRef]
- Mutsaers, S.E.; Wilkosz, S. Structure and function of mesothelial cells. Cancer Treat. Res. 2007, 134, 1–19. [Google Scholar]
- Thiery, J.P.; Acloque, H.; Huang, R.Y.; Nieto, M.A. Epithelial-mesenchymal transitions in development and disease. Cell 2009, 139, 871–890. [Google Scholar] [CrossRef]
- Asahina, K.; Zhou, B.; Pu, W.T.; Tsukamoto, H. Septum transversum-derived mesothelium gives rise to hepatic stellate cells and perivascular mesenchymal cells in developing mouse liver. Hepatology 2011, 53, 983–995. [Google Scholar] [CrossRef]
- Que, J.; Wilm, B.; Hasegawa, H.; Wang, F.; Bader, D.; Hogan, B.L. Mesothelium contributes to vascular smooth muscle and mesenchyme during lung development. Proc. Natl. Acad. Sci. USA 2008, 105, 16626–16630. [Google Scholar] [CrossRef]
- Rinkevich, Y.; Mori, T.; Sahoo, D.; Xu, P.X.; Bermingham, J.R., Jr.; Weissman, I.L. Identification and prospective isolation of a mesothelial precursor lineage giving rise to smooth muscle cells and fibroblasts for mammalian internal organs, and their vasculature. Nat. Cell Biol. 2012, 14, 1251–1260. [Google Scholar] [CrossRef]
- Lachaud, C.C.; Pezzolla, D.; Dominguez-Rodriguez, A.; Smani, T.; Soria, B.; Hmadcha, A. Functional vascular smooth muscle-like cells derived from adult mouse uterine mesothelial cells. PLoS ONE 2013, 8, e55181. [Google Scholar] [CrossRef]
- Lansley, S.M.; Searles, R.G.; Hoi, A.; Thomas, C.; Moneta, H.; Herrick, S.E.; Thompson, P.J.; Newman, M.; Sterrett, G.F.; Prele, C.M.; et al. Mesothelial cell differentiation into osteoblast- and adipocyte-like cells. J. Cell. Mol. Med. 2011, 15, 2095–2105. [Google Scholar] [CrossRef]
- Foley-Comer, A.J.; Herrick, S.E.; Al-Mishlab, T.; Prele, C.M.; Laurent, G.J.; Mutsaers, S.E. Evidence for incorporation of free-floating mesothelial cells as a mechanism of serosal healing. J. Cell Sci. 2002, 115, 1383–1389. [Google Scholar]
- Mutsaers, S.E.; Whitaker, D.; Papadimitriou, J.M. Stimulation of mesothelial cell proliferation by exudate macrophages enhances serosal wound healing in a murine model. Am. J. Pathol. 2002, 160, 681–692. [Google Scholar] [CrossRef]
- Reijnen, M.M.; Falk, P.; van Goor, H.; Holmdahl, L. The antiadhesive agent sodium hyaluronate increases the proliferation rate of human peritoneal mesothelial cells. Fertil. Steril. 2000, 74, 146–151. [Google Scholar] [CrossRef]
- Brecht, M.; Mayer, U.; Schlosser, E.; Prehm, P. Increased hyaluronate synthesis is required for fibroblast detachment and mitosis. Biochem. J. 1986, 239, 445–450. [Google Scholar] [CrossRef]
- West, D.C.; Kumar, S. Hyaluronan and angiogenesis. Ciba Found. Symp. 1989, 143, 187–201. [Google Scholar]
- Entwistle, J.; Hall, C.L.; Turley, E.A. HA receptors: Regulators of signalling to the cytoskeleton. J. Cell. Biochem. 1996, 61, 569–577. [Google Scholar] [CrossRef]
- Ferrandez-lzquierdo, A.; Navarro-Fos, S.; Gonzalez-Devesa, M.; Gil-Benso, R.; Llombart-Bosch, A. Immunocytochemical typification of mesothelial cells in effusions: In vivo and in vitro models. Diagn. Cytopathol. 1994, 10, 256–262. [Google Scholar] [CrossRef]
- Herrmann, H.; Bar, H.; Kreplak, L.; Strelkov, S.V.; Aebi, U. Intermediate filaments: From cell architecture to nanomechanics. Nat. Rev. Mol. Cell Biol. 2007, 8, 562–573. [Google Scholar] [CrossRef]
- Goldman, R.D.; Khuon, S.; Chou, Y.H.; Opal, P.; Steinert, P.M. The function of intermediate filaments in cell shape and cytoskeletal integrity. J. Cell Biol. 1996, 134, 971–983. [Google Scholar] [CrossRef]
- Ivaska, J.; Pallari, H.M.; Nevo, J.; Eriksson, J.E. Novel functions of vimentin in cell adhesion, migration, and signaling. Exp. Cell Res. 2007, 313, 2050–2062. [Google Scholar] [CrossRef]
- Gong, L.; Zhao, H.; Wang, L.; Sun, B.; Yu, L.; Qi, X.; Zhou, X.; Zhang, L.; Guan, H.; Shao, S. Upregulation of annexin A5 affects the biological behaviors of lung squamous carcinoma cells in vitro. Chin. Sci. Bull. 2014, 59, 3610–3620. [Google Scholar] [CrossRef]
- Graf, T.; Beug, H. Role of the v-erbA and v-erbB oncogenes of avian erythroblastosis virus in erythroid cell transformation. Cell 1983, 34, 7–9. [Google Scholar] [CrossRef]
- Yanez-Mo, M.; Lara-Pezzi, E.; Selgas, R.; Ramirez-Huesca, M.; Dominguez-Jimenez, C.; Jimenez-Heffernan, J.A.; Aguilera, A.; Sanchez-Tomero, J.A.; Bajo, M.A.; Alvarez, V.; et al. Peritoneal dialysis and epithelial-to-mesenchymal transition of mesothelial cells. N. Engl. J. Med. 2003, 348, 403–413. [Google Scholar] [CrossRef]
- Perez-Pomares, J.M.; Munoz-Chapuli, R. Epithelial-mesenchymal transitions: A mesodermal cell strategy for evolutive innovation in Metazoans. Anat. Rec. 2002, 268, 343–351. [Google Scholar] [CrossRef]
- Gotzos, V.; Vogt, P.; Celio, M.R. The calcium binding protein calretinin is a selective marker for malignant pleural mesotheliomas of the epithelial type. Pathol. Res. Pract. 1996, 192, 137–147. [Google Scholar] [CrossRef]
- Nagel, H.; Hemmerlein, B.; Ruschenburg, I.; Huppe, K.; Droese, M. The value of anti-calretinin antibody in the differential diagnosis of normal and reactive mesothelia versus metastatic tumors in effusion cytology. Pathol. Res. Pract. 1998, 194, 759–764. [Google Scholar] [CrossRef]
- Doglioni, C.; Dei Tos, A.P.; Laurino, L.; Iuzzolino, P.; Chiarelli, C.; Celio, M.R.; Viale, G. Calretinin: A novel immunocytochemical marker for mesothelioma. Am. J. Surg. Pathol. 1996, 20, 1037–1046. [Google Scholar] [CrossRef]
- Kitazume, H.; Kitamura, K.; Mukai, K.; Inayama, Y.; Kawano, N.; Nakamura, N.; Sano, J.; Mitsui, K.; Yoshida, S.; Nakatani, Y. Cytologic differential diagnosis among reactive mesothelial cells, malignant mesothelioma, and adenocarcinoma: Utility of combined E-cadherin and calretinin immunostaining. Cancer 2000, 90, 55–60. [Google Scholar] [CrossRef]
- Perl, A.K.; Wilgenbus, P.; Dahl, U.; Semb, H.; Christofori, G. A causal role for E-cadherin in the transition from adenoma to carcinoma. Nature 1998, 392, 190–193. [Google Scholar] [CrossRef]
- Takeichi, M. Morphogenetic roles of classic cadherins. Curr. Opin. Cell Biol. 1995, 7, 619–627. [Google Scholar] [CrossRef]
- Suassuna, J.H.; Das Neves, F.C.; Hartley, R.B.; Ogg, C.S.; Cameron, J.S. Immunohistochemical studies of the peritoneal membrane and infiltrating cells in normal subjects and in patients on CAPD. Kidney Int. 1994, 46, 443–454. [Google Scholar] [CrossRef][Green Version]
- Ferrara, N.; Gerber, H.P.; LeCouter, J. The biology of VEGF and its receptors. Nat. Med. 2003, 9, 669–676. [Google Scholar] [CrossRef]
- Romano, M.; Catalano, A.; Nutini, M.; D’Urbano, E.; Crescenzi, C.; Claria, J.; Libner, R.; Davi, G.; Procopio, A. 5-lipoxygenase regulates malignant mesothelial cell survival: Involvement of vascular endothelial growth factor. FASEB J. 2001, 15, 2326–2336. [Google Scholar] [CrossRef]
- Aguilera, A.; Yanez-Mo, M.; Selgas, R.; Sanchez-Madrid, F.; Lopez-Cabrera, M. Epithelial to mesenchymal transition as a triggering factor of peritoneal membrane fibrosis and angiogenesis in peritoneal dialysis patients. Curr. Opin. Investig. Drugs 2005, 6, 262–268. [Google Scholar]
- Selgas, R.; Bajo, A.; Jimenez-Heffernan, J.A.; Sanchez-Tomero, J.A.; Del Peso, G.; Aguilera, A.; Lopez-Cabrera, M. Epithelial-to-mesenchymal transition of the mesothelial cell--its role in the response of the peritoneum to dialysis. Nephrol. Dial. Transplant. 2006, 21, ii2–ii7. [Google Scholar] [CrossRef]
- Yung, S.; Thomas, G.J.; Davies, M. Induction of hyaluronan metabolism after mechanical injury of human peritoneal mesothelial cells in vitro. Kidney Int. 2000, 58, 1953–1962. [Google Scholar] [CrossRef][Green Version]
- Yung, S.; Chan, T.M. Intrinsic cells: Mesothelial cells--central players in regulating inflammation and resolution. Perit. Dial. Int. 2009, 29, S21–S27. [Google Scholar]
- Chen, C.H.; Kuo, C.Y.; Wang, Y.J.; Chen, J.P. Dual function of glucosamine in gelatin/hyaluronic acid cryogel to modulate scaffold mechanical properties and to maintain chondrogenic phenotype for cartilage tissue engineering. Int. J. Mol. Sci. 2016, 17, 1957. [Google Scholar] [CrossRef]
- Xue, W.; Champ, S.; Huglin, M.B.; Jones, T.G.J. Rapid swelling and deswelling in cryogels of crosslinked poly(N-isopropylacrylamide-co-acrylic acid). Eur. Polym. J. 2004, 40, 467–476. [Google Scholar] [CrossRef]
- Woo, S.L.Y.; Gomez, M.A.; Akeson, W.H. The Time and history-dependent viscoelastic properties of the canine medial collateral ligament. J. Biomech. Eng. 1981, 103, 293–298. [Google Scholar] [CrossRef]
- Kim, Y.J.; Sah, R.L.; Doong, J.Y.; Grodzinsky, A.J. Fluorometric assay of DNA in cartilage explants using Hoechst 33258. Anal. Biochem. 1988, 174, 168–176. [Google Scholar] [CrossRef]

| Mechanical Property | G | GH |
|---|---|---|
| Compressive elastic modulus at 10% strain (MPa) | 0.15 ± 0.05 | 0.22 ± 0.03 * |
| Compressive elastic modulus at 20% strain (MPa) | 0.31 ± 0.11 | 0.52 ± 0.04 * |
| Compressive elastic modulus at 30% strain (MPa) | 0.64 ± 0.23 | 1.26 ± 0.16 * |
| Compressive strain to failure (%) | 45.0 ± 4.9 | 32.8 ± 2.2 * |
| Compressive stress to failure (MPa) | 0.21 ± 0.04 | 0.17 ± 0.02 * |
| Toughness (kJ/m3) | 35.0 ± 2.6 | 22.5 ± 5.4 * |
| Compressive stiffness at 0.2 mm displacement (kN/m) | 4.45 ± 1.55 | 6.51 ± 0.67 * |
| Compressive stiffness at 0.4 mm displacement (kN/m) | 8.99 ± 3.08 | 14.54 ± 0.84 * |
| Compressive stiffness at 0.6 mm displacement (kN/m) | 18.20 ± 6.19 | 32.63 ± 3.68 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kao, H.-H.; Kuo, C.-Y.; Chen, K.-S.; Chen, J.-P. Preparation of Gelatin and Gelatin/Hyaluronic Acid Cryogel Scaffolds for the 3D Culture of Mesothelial Cells and Mesothelium Tissue Regeneration. Int. J. Mol. Sci. 2019, 20, 4527. https://doi.org/10.3390/ijms20184527
Kao H-H, Kuo C-Y, Chen K-S, Chen J-P. Preparation of Gelatin and Gelatin/Hyaluronic Acid Cryogel Scaffolds for the 3D Culture of Mesothelial Cells and Mesothelium Tissue Regeneration. International Journal of Molecular Sciences. 2019; 20(18):4527. https://doi.org/10.3390/ijms20184527
Chicago/Turabian StyleKao, Hao-Hsi, Chang-Yi Kuo, Kuo-Su Chen, and Jyh-Ping Chen. 2019. "Preparation of Gelatin and Gelatin/Hyaluronic Acid Cryogel Scaffolds for the 3D Culture of Mesothelial Cells and Mesothelium Tissue Regeneration" International Journal of Molecular Sciences 20, no. 18: 4527. https://doi.org/10.3390/ijms20184527
APA StyleKao, H.-H., Kuo, C.-Y., Chen, K.-S., & Chen, J.-P. (2019). Preparation of Gelatin and Gelatin/Hyaluronic Acid Cryogel Scaffolds for the 3D Culture of Mesothelial Cells and Mesothelium Tissue Regeneration. International Journal of Molecular Sciences, 20(18), 4527. https://doi.org/10.3390/ijms20184527






