Conflicting Roles of 20-HETE in Hypertension and Stroke
Abstract
1. Introduction
2. 20-HETE in Hypertension
2.1. 20-HETE
2.2. 20-HETE in Hypertension
2.2.1. 20-HETE and the Kidney
2.2.2. 20-HETE and the Vasculature
3. 20-HETE in Stroke
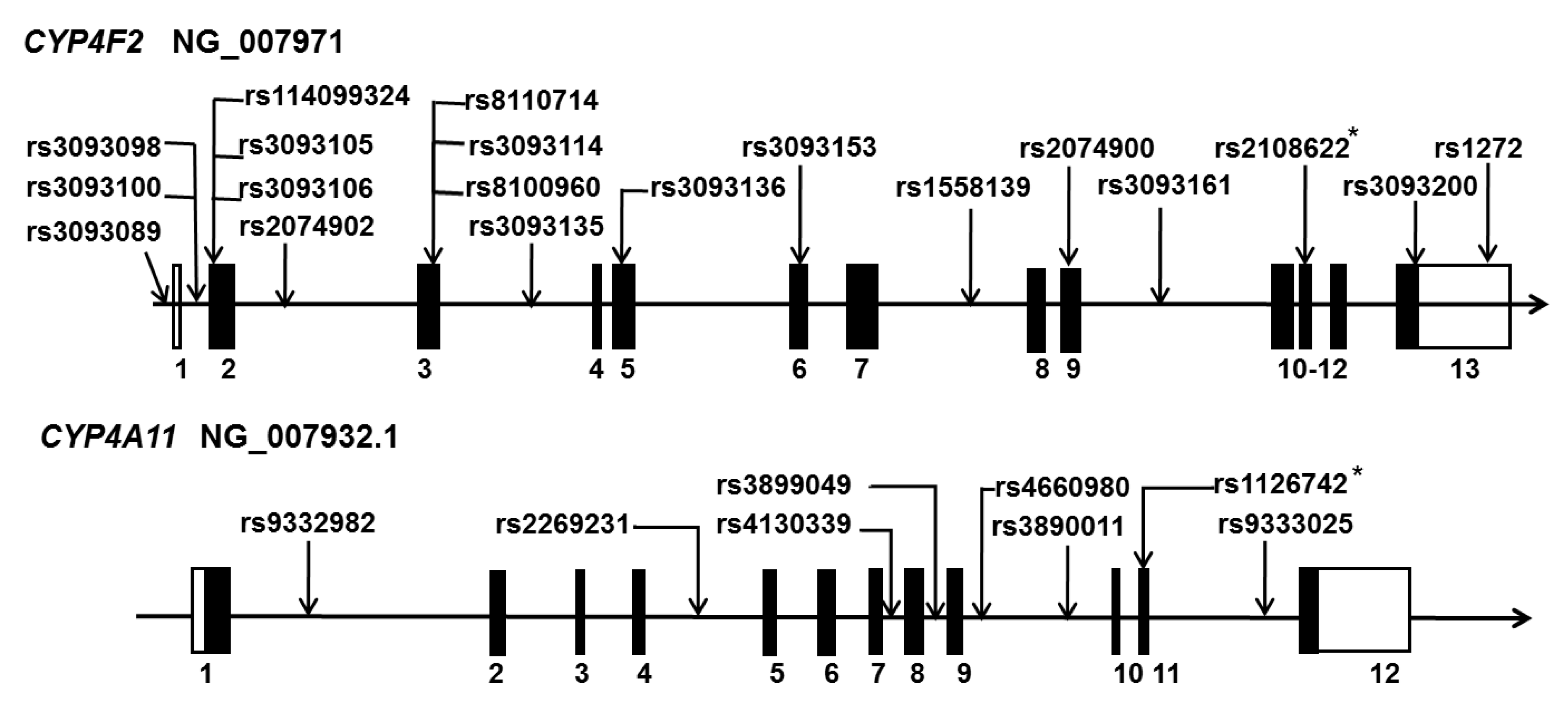
3.1. 20-HETE in the Susceptibility of the Onset of Stroke
3.2. 20-HETE in the Progression of Stroke
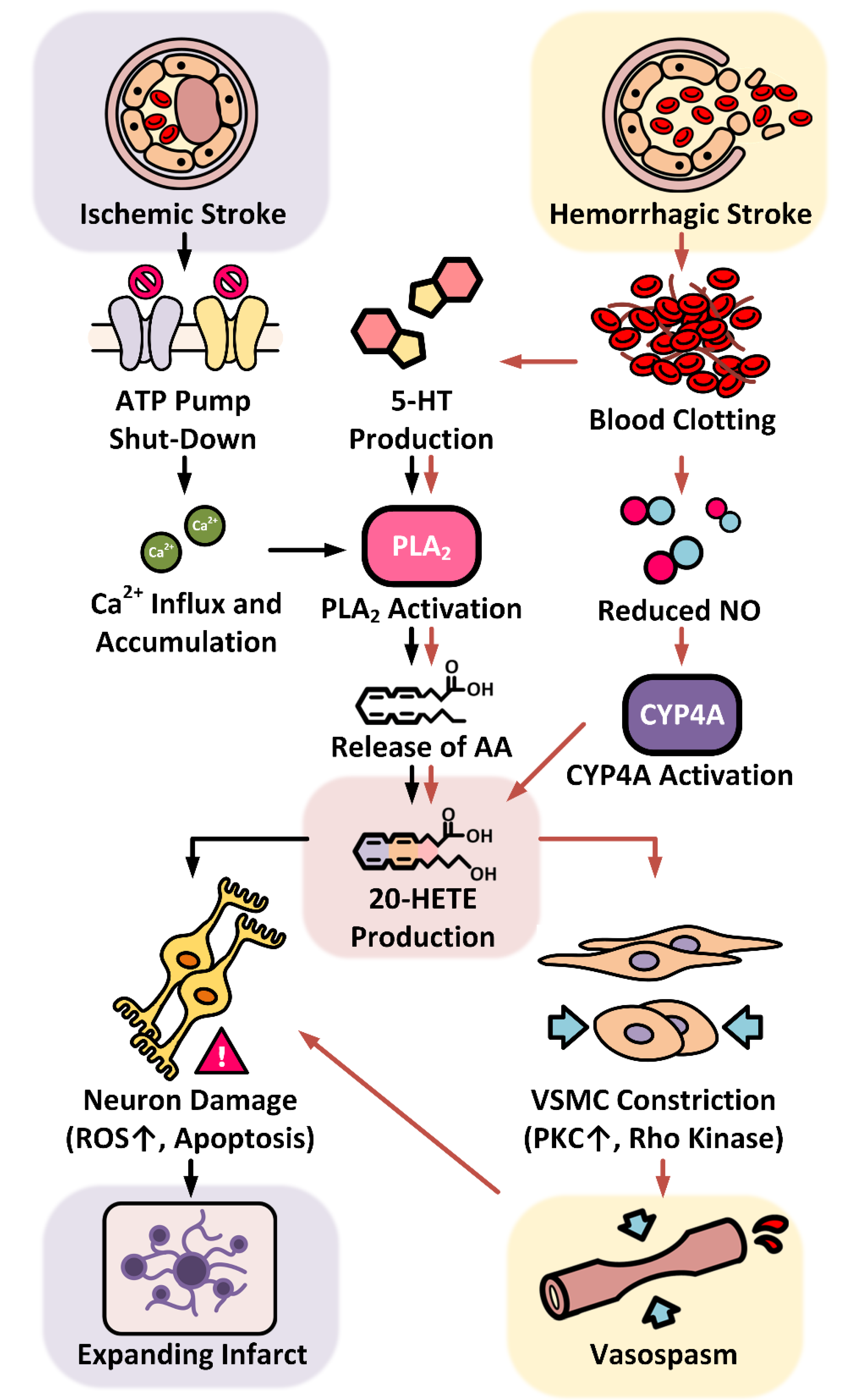
3.2.1. 20-HETE in the Progression of Ischemic Stroke
3.2.2. 20-HETE in the Progression of Hemorrhagic Stroke
3.3. 20-HETE in the Prognosis of Stroke
4. Conclusion and Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 20-COOH-HETE | 20-carboxy-eicosatetraenoic acid |
| 20-HETE | 20-hydroxyeicosatetraenoic acid |
| 5-HT | serotonin |
| GWAS | Genome-wide association study |
| AA | arachidonic acid |
| ACE | angiotensin-converting enzyme |
| ACI | acute cerebral ischemia |
| ADH | alcohol dehydrogenase |
| Af-art | afferent arteriole |
| ANG II | angiotensin II |
| AT1 | angiotensin II receptor 1 |
| BBB | blood-brain barrier |
| BK | blocking calcium-activated potassium channel |
| CADASIL | cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy |
| CARASIL | cerebral autosomal-recessive arteriopathy with subcortical infarcts and leukoencephalopathy |
| COX | cyclooxygenase |
| COX2 | cyclooxygenase-2 |
| CSD | cortical spreading depolarization |
| CSF | cerebrospinal fluid |
| CSI | cortical spreading ischemia |
| DCI | delayed cerebral ischemia |
| EETs | epoxyeicosatrienoic acids |
| EPC | endothelial progenitor cells |
| FAA | free fatty acids |
| FGF2 | fibroblast growth factor 2 |
| KO | knock out |
| LOX | lipoxygenase |
| MAPKs | mitogen-activated protein kinases |
| MCAo | middle cerebral artery occlusion |
| ND | neurological dysfunction |
| NHE3 | sodium-hydrogen exchanger 3 |
| NKCC2 | Na-K-Cl cotransporter |
| NO | nitric oxide |
| PKC | protein kinase C |
| PLA2 | phospholipase A2 |
| PT | proximal tubule |
| RAAS | renin-angiotensin-aldosterone system |
| ROMK2 | renal outer medullary potassium channel 2 |
| SAH | subarachnoid hemorrhage |
| SHR | spontaneously hypertensive rat |
| SNP | single nucleotide polymorphisms |
| SS | salt sensitive |
| TALH | thick ascending loop of Henle |
| TBI | traumatic brain injury |
| TGF | tubuloglomerular feedback responses |
| TRPC6 | transient receptor potential cation channel 6 |
| TRPV1 | transient receptor potential vanilloid 1 |
| UGT | uridine 5’-diphosphoglucuronosyltransferase |
| VEGF | vascular endothelial growth factor |
| VSMC | vascular smooth muscle cell |
| WT | wild type |
References
- Nwankwo, T.; Yoon, S.S.; Burt, V.; Gu, Q. Hypertension among adults in the United States: National Health and Nutrition Examination Survey, 2011-2012. NCHS Data Brief. 2013, 1–8. [Google Scholar]
- Murphy, S.L.; Xu, J.; Kochanek, K.D.; Curtin, S.C.; Arias, E. Deaths: Final Data for 2015. Natl. Vital Stat Rep. 2017, 66, 1–75. [Google Scholar] [PubMed]
- Shekhar, S.; Liu, R.; Travis, O.K.; Roman, R.J.; Fan, F. Cerebral Autoregulation in Hypertension and Ischemic Stroke: A Mini Review. J. Pharm. Sci. Exp. Pharmacol. 2017, 2017, 21–27. [Google Scholar] [PubMed]
- Shekhar, S.; Cunningham, M.W.; Pabbidi, M.R.; Wang, S.; Booz, G.W.; Fan, F. Targeting vascular inflammation in ischemic stroke: Recent developments on novel immunomodulatory approaches. Eur. J. Pharmacol. 2018, 833, 531–544. [Google Scholar] [CrossRef] [PubMed]
- Francis, J.; Raghunathan, S.; Khanna, P. The role of genetics in stroke. Postgrad. Med. J. 2007, 83, 590–595. [Google Scholar] [CrossRef]
- Chauhan, G.; Debette, S. Genetic Risk Factors for Ischemic and Hemorrhagic Stroke. Curr. Cardiol. Rep. 2016, 18, 124. [Google Scholar] [CrossRef] [PubMed]
- Fan, F.; Ge, Y.; Lv, W.; Elliott, M.R.; Muroya, Y.; Hirata, T.; Booz, G.W.; Roman, R.J. Molecular mechanisms and cell signaling of 20-hydroxyeicosatetraenoic acid in vascular pathophysiology. Front. Biosci. (Landmark Ed.) 2016, 21, 1427–1463. [Google Scholar] [PubMed]
- Fan, F.; Roman, R.J. Effect of Cytochrome P450 Metabolites of Arachidonic Acid in Nephrology. J. Am. Soc. Nephrol. 2017, 28, 2845–2855. [Google Scholar] [CrossRef] [PubMed]
- Fava, C.; Ricci, M.; Melander, O.; Minuz, P. Hypertension, cardiovascular risk and polymorphisms in genes controlling the cytochrome P450 pathway of arachidonic acid: A sex-specific relation? Prostaglandins Other Lipid Mediat. 2012, 98, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Fava, C.; Bonafini, S. Eicosanoids via CYP450 and cardiovascular disease: Hints from genetic and nutrition studies. Prostaglandins Other Lipid Mediat. 2018, 139, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Fan, F.; Simino, J.; Auchus, A.; Knopman, D.; Boerwinkle, E.; Fornage, M.; Mosley, T.; Roman, R. Functional variants in CYP4A11 and CYP4F2 are associated with cognitive impairment and related dementia endophenotypes in the elderly. In Proceedings of the 16th International Winter Eicosanoid Conference, Baltimore, MD, USA, 13 March 2016; p. CV5. [Google Scholar]
- Zhang, C.; Booz, G.W.; Yu, Q.; He, X.; Wang, S.; Fan, F. Conflicting roles of 20-HETE in hypertension and renal end organ damage. Eur. J. Pharmacol. 2018, 833, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Nakayama, T.; Sato, N.; Izumi, Y.; Kasamaki, Y.; Shindo, A.; Ohta, M.; Soma, M.; Aoi, N.; Sato, M.; et al. A haplotype of the CYP4F2 gene is associated with cerebral infarction in Japanese men. Am. J. Hypertens. 2008, 21, 1216–1223. [Google Scholar] [CrossRef] [PubMed]
- Ward, N.C.; Tsai, I.J.; Barden, A.; van Bockxmeer, F.M.; Puddey, I.B.; Hodgson, J.M.; Croft, K.D. A single nucleotide polymorphism in the CYP4F2 but not CYP4A11 gene is associated with increased 20-HETE excretion and blood pressure. Hypertension 2008, 51, 1393–1398. [Google Scholar] [CrossRef] [PubMed]
- Fava, C.; Montagnana, M.; Almgren, P.; Rosberg, L.; Lippi, G.; Hedblad, B.; Engstrom, G.; Berglund, G.; Minuz, P.; Melander, O. The V433M variant of the CYP4F2 is associated with ischemic stroke in male Swedes beyond its effect on blood pressure. Hypertension. 2008, 52, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Munshi, A.; Sharma, V.; Kaul, S.; Al-Hazzani, A.; Alshatwi, A.A.; Shafi, G.; Koppula, R.; Mallemoggala, S.B.; Jyothy, A. Association of 1347 G/A cytochrome P450 4F2 (CYP4F2) gene variant with hypertension and stroke. Mol. Biol. Rep. 2012, 39, 1677–1682. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, M.K.; Crago, E.A.; Conley, Y.P.; Balzer, J.R.; Ren, D.; Ducruet, A.F.; Kochanek, P.M.; Sherwood, P.R.; Poloyac, S.M. 20-HETE is associated with unfavorable outcomes in subarachnoid hemorrhage patients. J. Cereb. Blood Flow Metab. 2015, 35, 1515–1522. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Zhu, G.; Liu, F.; Zhang, H.; Qin, X.; Li, L.; Zhiyi, H. CYP4F2 gene V433M polymorphism is associated with ischemic stroke in the male Northern Chinese Han population. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2010, 34, 664–668. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.; Cui, G.; Zhang, L.; Xu, Y.; Bao, X.; Tu, Y.; Wu, B.; Wang, Q.; Hui, R.; Wang, W.; et al. Association of common variants of CYP4A11 and CYP4F2 with stroke in the Han Chinese population. Pharm. Genom. 2010, 20, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Jin, L.; Mu, T.; Fan, Y.; Zhang, H.; Zhu, Y.; Mao, X.; Li, R.; Tang, S. Associations of CYP4A11 gene-gene and gene-smoking interactions with essential hypertension in the male eastern Chinese Han population. Clin. Exp. Hypertens. 2017, 39, 448–453. [Google Scholar] [CrossRef]
- Fu, Z.; Nakayama, T.; Sato, N.; Izumi, Y.; Kasamaki, Y.; Shindo, A.; Ohta, M.; Soma, M.; Aoi, N.; Sato, M.; et al. Haplotype-based case study of human CYP4A11 gene and cerebral infarction in Japanese subject. Endocrine 2008, 33, 215–222. [Google Scholar] [CrossRef]
- Fu, Z.; Nakayama, T.; Sato, N.; Izumi, Y.; Kasamaki, Y.; Shindo, A.; Ohta, M.; Soma, M.; Aoi, N.; Sato, M.; et al. A haplotype of the CYP4A11 gene associated with essential hypertension in Japanese men. J. Hypertens. 2008, 26, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Gainer, J.V.; Lipkowitz, M.S.; Yu, C.; Waterman, M.R.; Dawson, E.P.; Capdevila, J.H.; Brown, N.J.; Group, A.S. Association of a CYP4A11 variant and blood pressure in black men. J. Am. Soc. Nephrol. 2008, 19, 1606–1612. [Google Scholar] [CrossRef] [PubMed]
- Laffer, C.L.; Gainer, J.V.; Waterman, M.R.; Capdevila, J.H.; Laniado-Schwartzman, M.; Nasjletti, A.; Brown, N.J.; Elijovich, F. The T8590C polymorphism of CYP4A11 and 20-hydroxyeicosatetraenoic acid in essential hypertension. Hypertension 2008, 51, 767–772. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.Y.; Lee, S.J.; Min, J.; Oh, K.S.; Kim, D.H.; Kim, H.S.; Shin, J.G. Identification of novel CYP4F2 genetic variants exhibiting decreased catalytic activity in the conversion of arachidonic acid to 20-hydroxyeicosatetraenoic acid (20-HETE). Prostaglandins Leukot. Essent. Fatty Acids. 2018, 131, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Stec, D.E.; Roman, R.J.; Flasch, A.; Rieder, M.J. Functional polymorphism in human CYP4F2 decreases 20-HETE production. Physiol. Genom. 2007, 30, 74–81. [Google Scholar] [CrossRef]
- Gainer, J.V.; Bellamine, A.; Dawson, E.P.; Womble, K.E.; Grant, S.W.; Wang, Y.; Cupples, L.A.; Guo, C.Y.; Demissie, S.; O’Donnell, C.J.; et al. Functional variant of CYP4A11 20-hydroxyeicosatetraenoic acid synthase is associated with essential hypertension. Circulation 2005, 111, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Honda, M.; Takahashi, M.; Tsukada, C.; Ito, M.; Katono, Y.; Hosono, H.; Saigusa, D.; Suzuki, N.; Tomioka, Y.; et al. Functional characterization of 10 CYP4A11 allelic variants to evaluate the effect of genotype on arachidonic acid omega-hydroxylation. Drug Metab. Pharmacokinet. 2015, 30, 119–122. [Google Scholar] [CrossRef]
- Fan, F.; Geurts, A.M.; Murphy, S.R.; Pabbidi, M.R.; Jacob, H.J.; Roman, R.J. Impaired myogenic response and autoregulation of cerebral blood flow is rescued in CYP4A1 transgenic Dahl salt-sensitive rat. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 308, R379–R390. [Google Scholar] [CrossRef]
- Williams, J.M.; Fan, F.; Murphy, S.; Schreck, C.; Lazar, J.; Jacob, H.J.; Roman, R.J. Role of 20-HETE in the antihypertensive effect of transfer of chromosome 5 from Brown Norway to Dahl salt-sensitive rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2012, 302, R1209–R1218. [Google Scholar] [CrossRef]
- Lu, L.; Wang, M.; Wei, X.; Li, W. 20-HETE Inhibition by HET0016 Decreases the Blood-Brain Barrier Permeability and Brain Edema After Traumatic Brain Injury. Front. Aging Neurosci. 2018, 10, 207. [Google Scholar] [CrossRef]
- Lu, L.; Wang, M.; Yuan, F.; Wei, X.; Li, W. Roles of elevated 20HETE in the breakdown of blood brain barrier and the severity of brain edema in experimental traumatic brain injury. Mol. Med. Rep. 2018, 17, 7339–7345. [Google Scholar] [CrossRef] [PubMed]
- Roman, R.J.; Fan, F. 20-HETE: Hypertension and Beyond. Hypertension 2018, 72, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Liao, D.; Yi, X.; Zhang, B.; Zhou, Q.; Lin, J. Interaction Between CYP4F2 rs2108622 and CPY4A11 rs9333025 Variants Is Significantly Correlated with Susceptibility to Ischemic Stroke and 20-Hydroxyeicosatetraenoic Acid Level. Genet. Test Mol. Biomark. 2016, 20, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Fan, F.; Muroya, Y.; Roman, R.J. Cytochrome P450 eicosanoids in hypertension and renal disease. Curr. Opin. Nephrol. Hypertens. 2015, 24, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Orozco, L.D.; Liu, H.; Perkins, E.; Johnson, D.A.; Chen, B.B.; Fan, F.; Baker, R.C.; Roman, R.J. 20-Hydroxyeicosatetraenoic acid inhibition attenuates balloon injury-induced neointima formation and vascular remodeling in rat carotid arteries. J. Pharmacol. Exp. Ther. 2013, 346, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Hill, E.; Fitzpatrick, F.; Murphy, R.C. Biological activity and metabolism of 20-hydroxyeicosatetraenoic acid in the human platelet. Br. J. Pharmacol. 1992, 106, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Fan, F.; Sun, C.W.; Maier, K.G.; Williams, J.M.; Pabbidi, M.R.; Didion, S.P.; Falck, J.R.; Zhuo, J.; Roman, R.J. 20-Hydroxyeicosatetraenoic acid contributes to the inhibition of K+ channel activity and vasoconstrictor response to angiotensin II in rat renal microvessels. PLoS ONE 2013, 8, e82482. [Google Scholar] [CrossRef] [PubMed]
- Garcia, V.; Gilani, A.; Shkolnik, B.; Pandey, V.; Zhang, F.F.; Dakarapu, R.; Gandham, S.K.; Reddy, N.R.; Graves, J.P.; Gruzdev, A.; et al. 20-HETE Signals Through G Protein-Coupled Receptor GPR75 (Gq) to Affect Vascular Function and Trigger Hypertension. Circ. Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Fan, F.; He, X.; Cornelius, C.C.; Roman, R.J. The potential neuroprotective role of 20-HETE in association with its receptor GPR75 expression in hypertension. In Proceedings of the 17th International Winter Eicosanoid Conference, Baltimore, MA, USA, 11 March 2018. [Google Scholar]
- Fan, F.; Roman, R. GPR75 Identified as the First 20-HETE Receptor: A Chemokine Receptor Adopted by a New Family. Circ. Res. 2017, 12, 1696–1698. [Google Scholar] [CrossRef] [PubMed]
- Tripathy, D.; Thirumangalakudi, L.; Grammas, P. RANTES upregulation in the Alzheimer’s disease brain: a possible neuroprotective role. Neurobiol. Aging 2010, 31, 8–16. [Google Scholar] [CrossRef]
- Dedoni, S.; Campbell, L.A.; Harvey, B.K.; Avdoshina, V.; Mocchetti, I. The orphan G-protein-coupled receptor 75 signaling is activated by the chemokine CCL5. J. Neurochem. 2018, 146, 526–539. [Google Scholar] [CrossRef]
- Tunaru, S.; Bonnavion, R.; Brandenburger, I.; Preussner, J.; Thomas, D.; Scholich, K.; Offermanns, S. 20-HETE promotes glucose-stimulated insulin secretion in an autocrine manner through FFAR1. Nat. Commun. 2018, 9, 177. [Google Scholar] [CrossRef]
- Park, S.K.; Herrnreiter, A.; Pfister, S.L.; Gauthier, K.M.; Falck, B.A.; Falck, J.R.; Campbell, W.B. GPR40 is a low-affinity epoxyeicosatrienoic acid receptor in vascular cells. J. Biol. Chem. 2018, 293, 10675–10691. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Lopez, B.; Dos Santos, E.A.; Falck, J.R.; Roman, R.J. Effects of 20-HETE on Na+ transport and Na+-K+-ATPase activity in the thick ascending loop of Henle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 292, R2400–R2405. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Murphy, S.R.; Fan, F.; Williams, J.M.; Falck, J.R.; Liu, R.; Roman, R.J. Role of 20-HETE in the impaired myogenic and TGF responses of the Af-Art of Dahl salt-sensitive rats. Am. J. Physiol. Renal. Physiol. 2014, 307, F509–F515. [Google Scholar] [CrossRef] [PubMed]
- Stec, D.E.; Deng, A.Y.; Rapp, J.P.; Roman, R.J. Cytochrome P4504A genotype cosegregates with hypertension in Dahl S rats. Hypertension 1996, 27, 564–568. [Google Scholar] [CrossRef]
- Roman, R.J. P-450 metabolites of arachidonic acid in the control of cardiovascular function. Physiol. Rev. 2002, 82, 131–185. [Google Scholar] [CrossRef]
- Carmichael, S.T. Rodent models of focal stroke: size, mechanism, and purpose. NeuroRx 2005, 2, 396–409. [Google Scholar] [CrossRef]
- Feigin, V.; Parag, V.; Lawes, C.M.; Rodgers, A.; Suh, I.; Woodward, M.; Jamrozik, K.; Ueshima, H. Asia Pacific Cohort Studies Collaboration. Smoking and elevated blood pressure are the most important risk factors for subarachnoid hemorrhage in the Asia-Pacific region: an overview of 26 cohorts involving 306,620 participants. Stroke 2005, 36, 1360–1365. [Google Scholar] [CrossRef]
- Vlak, M.H.; Rinkel, G.J.; Greebe, P.; Greving, J.P.; Algra, A. Lifetime risks for aneurysmal subarachnoid haemorrhage: multivariable risk stratification. J. Neurol. Neurosurg. Psychiatry 2013, 84, 619–623. [Google Scholar] [CrossRef]
- Cossu, G.; Messerer, M.; Oddo, M.; Daniel, R.T. To look beyond vasospasm in aneurysmal subarachnoid haemorrhage. Biomed. Res. Int. 2014, 2014, 628597. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, R.L. Delayed neurological deterioration after subarachnoid haemorrhage. Nat. Rev. Neurol. 2014, 10, 44–58. [Google Scholar] [CrossRef] [PubMed]
- Vergouwen, M.D.; Vermeulen, M.; van Gijn, J.; Rinkel, G.J.; Wijdicks, E.F.; Muizelaar, J.P.; Mendelow, A.D.; Juvela, S.; Yonas, H.; Terbrugge, K.G.; et al. Definition of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage as an outcome event in clinical trials and observational studies: proposal of a multidisciplinary research group. Stroke 2010, 41, 2391–2395. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Yi, X.; Wang, C.; Liao, D.; Lin, J.; Chi, L. Cytochrome 4A11 Genetic Polymorphisms Increase Susceptibility to Ischemic Stroke and Associate with Atherothrombotic Events After Stroke in Chinese. Genet. Test Mol. Biomark. 2015, 19, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Yi, X.; Han, Z.; Zhou, Q.; Lin, J.; Liu, P. 20-Hydroxyeicosatetraenoic Acid as a Predictor of Neurological Deterioration in Acute Minor Ischemic Stroke. Stroke 2016, 47, 3045–3047. [Google Scholar] [CrossRef] [PubMed]
- Ward, N.C.; Croft, K.D.; Blacker, D.; Hankey, G.J.; Barden, A.; Mori, T.A.; Puddey, I.B.; Beer, C.D. Cytochrome P450 metabolites of arachidonic acid are elevated in stroke patients compared with healthy controls. Clin. Sci. (Lond.) 2011, 121, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Dunn, K.M.; Renic, M.; Flasch, A.K.; Harder, D.R.; Falck, J.; Roman, R.J. Elevated production of 20-HETE in the cerebral vasculature contributes to severity of ischemic stroke and oxidative stress in spontaneously hypertensive rats. Am. J. Physiol. Heart Circ. Physiol. 2008, 295, H2455–H2465. [Google Scholar] [CrossRef]
- Marumo, T.; Eto, K.; Wake, H.; Omura, T.; Nabekura, J. The inhibitor of 20-HETE synthesis, TS-011, improves cerebral microcirculatory autoregulation impaired by middle cerebral artery occlusion in mice. Br. J. Pharmacol. 2010, 161, 1391–1402. [Google Scholar] [CrossRef]
- Omura, T.; Tanaka, Y.; Miyata, N.; Koizumi, C.; Sakurai, T.; Fukasawa, M.; Hachiuma, K.; Minagawa, T.; Susumu, T.; Yoshida, S.; et al. Effect of a new inhibitor of the synthesis of 20-HETE on cerebral ischemia reperfusion injury. Stroke 2006, 37, 1307–1313. [Google Scholar] [CrossRef]
- Kawasaki, T.; Marumo, T.; Shirakami, K.; Mori, T.; Doi, H.; Suzuki, M.; Watanabe, Y.; Chaki, S.; Nakazato, A.; Ago, Y.; et al. Increase of 20-HETE synthase after brain ischemia in rats revealed by PET study with 11C-labeled 20-HETE synthase-specific inhibitor. J. Cereb. Blood Flow Metab. 2012, 32, 1737–1746. [Google Scholar] [CrossRef]
- Auguet, T.; Aragones, G.; Colom, M.; Aguilar, C.; Martin-Paredero, V.; Canela, N.; Ruyra, X.; Richart, C. Targeted metabolomic approach in men with carotid plaque. PLoS ONE 2018, 13, e0200547. [Google Scholar] [CrossRef] [PubMed]
- Yi, X.Y.; Liao, D.X.; Wang, C.; Cheng, W.; Fu, X.Q.; Zhang, B. Cytochrome P450 Genetic Variants and Their Metabolite Levels Associated with Plaque Stability in Patients with Ischemic Stroke. J. Atheroscler. Thromb. 2016, 23, 330–338. [Google Scholar] [CrossRef]
- Fujii, M.; Yan, J.; Rolland, W.B.; Soejima, Y.; Caner, B.; Zhang, J.H. Early brain injury, an evolving frontier in subarachnoid hemorrhage research. Transl. Stroke Res. 2013, 4, 432–446. [Google Scholar] [CrossRef] [PubMed]
- Megyesi, J.F.; Vollrath, B.; Cook, D.A.; Findlay, J.M. In vivo animal models of cerebral vasospasm: a review. Neurosurgery 2000, 46, 448–461. [Google Scholar] [CrossRef] [PubMed]
- Harder, D.R.; Dernbach, P.; Waters, A. Possible cellular mechanism for cerebral vasospasm after experimental subarachnoid hemorrhage in the dog. J. Clin. Investig. 1987, 80, 875–880. [Google Scholar] [CrossRef] [PubMed]
- Cambj-Sapunar, L.; Yu, M.; Harder, D.R.; Roman, R.J. Contribution of 5-hydroxytryptamine1B receptors and 20-hydroxyeiscosatetraenoic acid to fall in cerebral blood flow after subarachnoid hemorrhage. Stroke 2003, 34, 1269–1275. [Google Scholar] [CrossRef] [PubMed]
- Hacein-Bey, L.; Harder, D.R.; Meier, H.T.; Varelas, P.N.; Miyata, N.; Lauer, K.K.; Cusick, J.F.; Roman, R.J. Reversal of delayed vasospasm by TS-011 in the dual hemorrhage dog model of subarachnoid hemorrhage. AJNR Am. J. Neuroradiol. 2006, 27, 1350–1354. [Google Scholar] [PubMed]
- Roman, R.J.; Renic, M.; Dunn, K.M.; Takeuchi, K.; Hacein-Bey, L. Evidence that 20-HETE contributes to the development of acute and delayed cerebral vasospasm. Neurol. Res. 2006, 28, 738–749. [Google Scholar] [CrossRef] [PubMed]
- Fordsmann, J.C.; Ko, R.W.; Choi, H.B.; Thomsen, K.; Witgen, B.M.; Mathiesen, C.; Lonstrup, M.; Piilgaard, H.; MacVicar, B.A.; Lauritzen, M. Increased 20-HETE synthesis explains reduced cerebral blood flow but not impaired neurovascular coupling after cortical spreading depression in rat cerebral cortex. J. Neurosci. 2013, 33, 2562–2570. [Google Scholar] [CrossRef] [PubMed]
- Shaik, J.S.; Poloyac, S.M.; Kochanek, P.M.; Alexander, H.; Tudorascu, D.L.; Clark, R.S.; Manole, M.D. 20-Hydroxyeicosatetraenoic Acid Inhibition by HET0016 Offers Neuroprotection, Decreases Edema, and Increases Cortical Cerebral Blood Flow in a Pediatric Asphyxial Cardiac Arrest Model in Rats. J. Cereb. Blood Flow Metab. 2015, 35, 1757–1763. [Google Scholar] [CrossRef] [PubMed]
- Miyata, N.; Seki, T.; Tanaka, Y.; Omura, T.; Taniguchi, K.; Doi, M.; Bandou, K.; Kametani, S.; Sato, M.; Okuyama, S.; et al. Beneficial effects of a new 20-hydroxyeicosatetraenoic acid synthesis inhibitor, TS-011 [N-(3-chloro-4-morpholin-4-yl) phenyl-N’-hydroxyimido formamide], on hemorrhagic and ischemic stroke. J. Pharmacol. Exp. Ther. 2005, 314, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Zhao, X.; Lan, X.; Li, Q.; Gao, Y.; Liu, X.; Wan, J.; Yang, Z.; Chen, X.; Zang, W.; et al. 20-HETE synthesis inhibition promotes cerebral protection after intracerebral hemorrhage without inhibiting angiogenesis. J. Cereb. Blood Flow Metab. 2019, 39, 1531–1543. [Google Scholar] [CrossRef] [PubMed]
- Gebremedhin, D.; Zhang, D.X.; Carver, K.A.; Rau, N.; Rarick, K.R.; Roman, R.J.; Harder, D.R. Expression of CYP 4A omega-hydroxylase and formation of 20-hydroxyeicosatetreanoic acid (20-HETE) in cultured rat brain astrocytes. Prostaglandins Other Lipid Mediat. 2016, 124, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Haley, M.J.; Lawrence, C.B. The blood-brain barrier after stroke: Structural studies and the role of transcytotic vesicles. J. Cereb. Blood Flow Metab. 2017, 37, 456–470. [Google Scholar] [CrossRef] [PubMed]
- Faraco, G.; Iadecola, C. Hypertension: a harbinger of stroke and dementia. Hypertension 2013, 62, 810–817. [Google Scholar] [CrossRef] [PubMed]
- Ergul, A.; Alhusban, A.; Fagan, S.C. Angiogenesis: a harmonized target for recovery after stroke. Stroke 2012, 43, 2270–2274. [Google Scholar] [CrossRef] [PubMed]
- Sa, G.; Murugesan, G.; Jaye, M.; Ivashchenko, Y.; Fox, P.L. Activation of cytosolic phospholipase A2 by basic fibroblast growth factor via a p42 mitogen-activated protein kinase-dependent phosphorylation pathway in endothelial cells. J. Biol. Chem. 1995, 270, 2360–2366. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Ackerman, R.; Saleh, M.; Gotlinger, K.H.; Kessler, M.; Mendelowitz, L.G.; Falck, J.R.; Arbab, A.S.; Scicli, A.G.; Schwartzman, M.L.; et al. 20-HETE regulates the angiogenic functions of human endothelial progenitor cells and contributes to angiogenesis in vivo. J. Pharmacol. Exp. Ther. 2014, 348, 442–451. [Google Scholar] [CrossRef]
- Chen, P.; Guo, M.; Wygle, D.; Edwards, P.A.; Falck, J.R.; Roman, R.J.; Scicli, A.G. Inhibitors of cytochrome P450 4A suppress angiogenic responses. Am. J. Pathol. 2005, 166, 615–624. [Google Scholar] [CrossRef]
- Guo, A.M.; Roman, R.J.; Falck, J.R.; Jafari, K.; Edwards, P.A.; Scicli, A.G. Overexpression of CYP4A1-20-HETE in U251 Glioma Cell Induces Hyperproliferative Phenotypes in vitro and in vivo. Proc. FASEB J. 2008, 22, 1136-13. [Google Scholar]
- Guo, A.M.; Sheng, J.; Scicli, G.M.; Arbab, A.S.; Lehman, N.L.; Edwards, P.A.; Falck, J.R.; Roman, R.J.; Scicli, A.G. Expression of CYP4A1 in U251 human glioma cell induces hyperproliferative phenotype in vitro and rapidly growing tumors in vivo. J. Pharmacol. Exp. Ther. 2008, 327, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Alexanian, A.; Sorokin, A. Targeting 20-HETE producing enzymes in cancer - rationale, pharmacology, and clinical potential. Onco Targets Ther. 2013, 6, 243–255. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, Y.; Zhan, M.; Liu, Y.; Li, Z.; Li, J.; Cheng, G.; Teng, G.; Lu, L. Astrocytic cytochrome P450 4A/20-hydroxyeicosatetraenoic acid contributes to angiogenesis in the experimental ischemic stroke. Brain Res. 2019, 1708, 160–170. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shekhar, S.; Varghese, K.; Li, M.; Fan, L.; Booz, G.W.; Roman, R.J.; Fan, F. Conflicting Roles of 20-HETE in Hypertension and Stroke. Int. J. Mol. Sci. 2019, 20, 4500. https://doi.org/10.3390/ijms20184500
Shekhar S, Varghese K, Li M, Fan L, Booz GW, Roman RJ, Fan F. Conflicting Roles of 20-HETE in Hypertension and Stroke. International Journal of Molecular Sciences. 2019; 20(18):4500. https://doi.org/10.3390/ijms20184500
Chicago/Turabian StyleShekhar, Shashank, Kevin Varghese, Man Li, Letao Fan, George W. Booz, Richard J. Roman, and Fan Fan. 2019. "Conflicting Roles of 20-HETE in Hypertension and Stroke" International Journal of Molecular Sciences 20, no. 18: 4500. https://doi.org/10.3390/ijms20184500
APA StyleShekhar, S., Varghese, K., Li, M., Fan, L., Booz, G. W., Roman, R. J., & Fan, F. (2019). Conflicting Roles of 20-HETE in Hypertension and Stroke. International Journal of Molecular Sciences, 20(18), 4500. https://doi.org/10.3390/ijms20184500




