Mutagenic Study of Benzimidazole Derivatives with (+S9) and without (−S9) Metabolic Activation
Abstract
1. Introduction
2. Results
2.1. Genetic Analysis
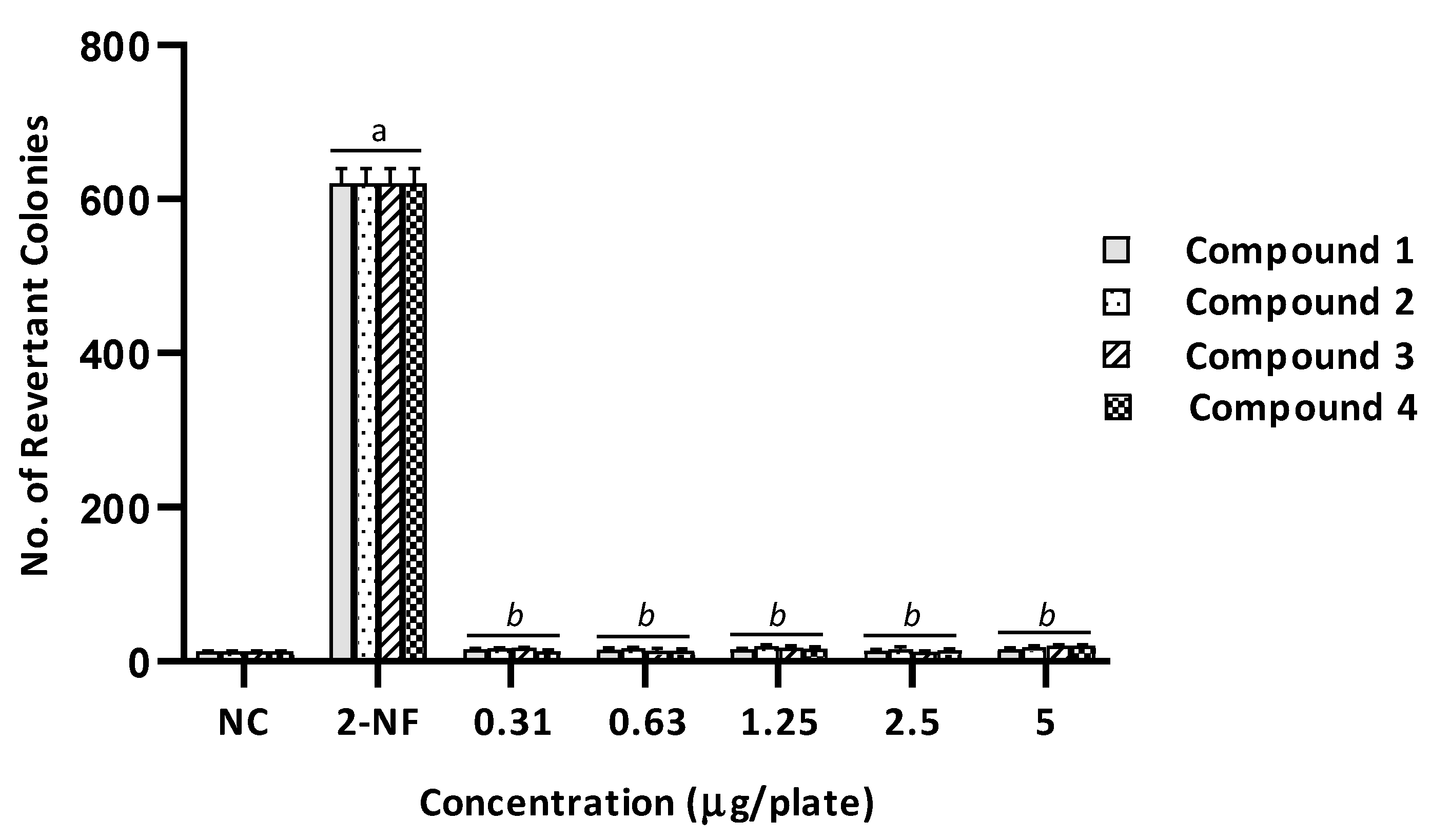
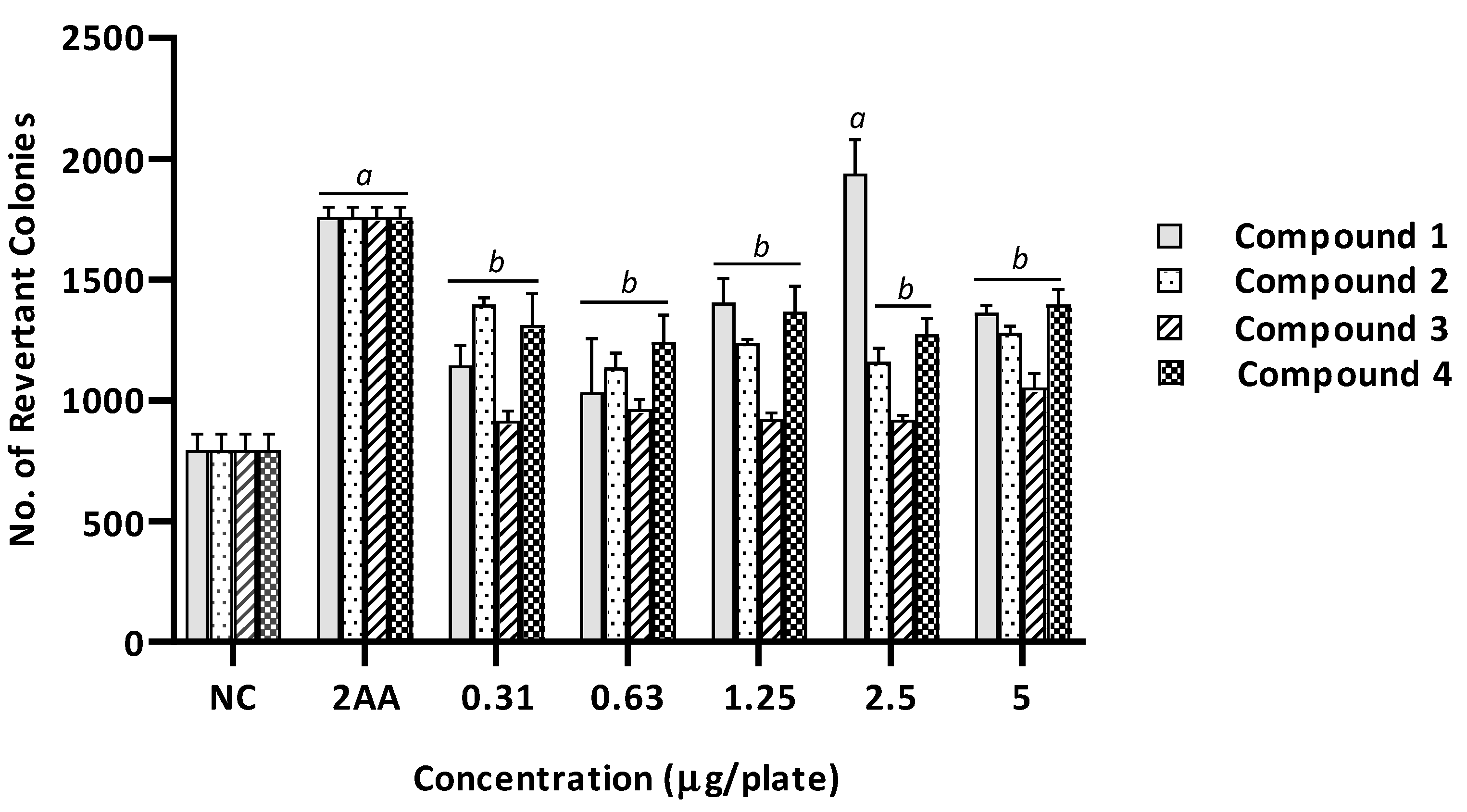
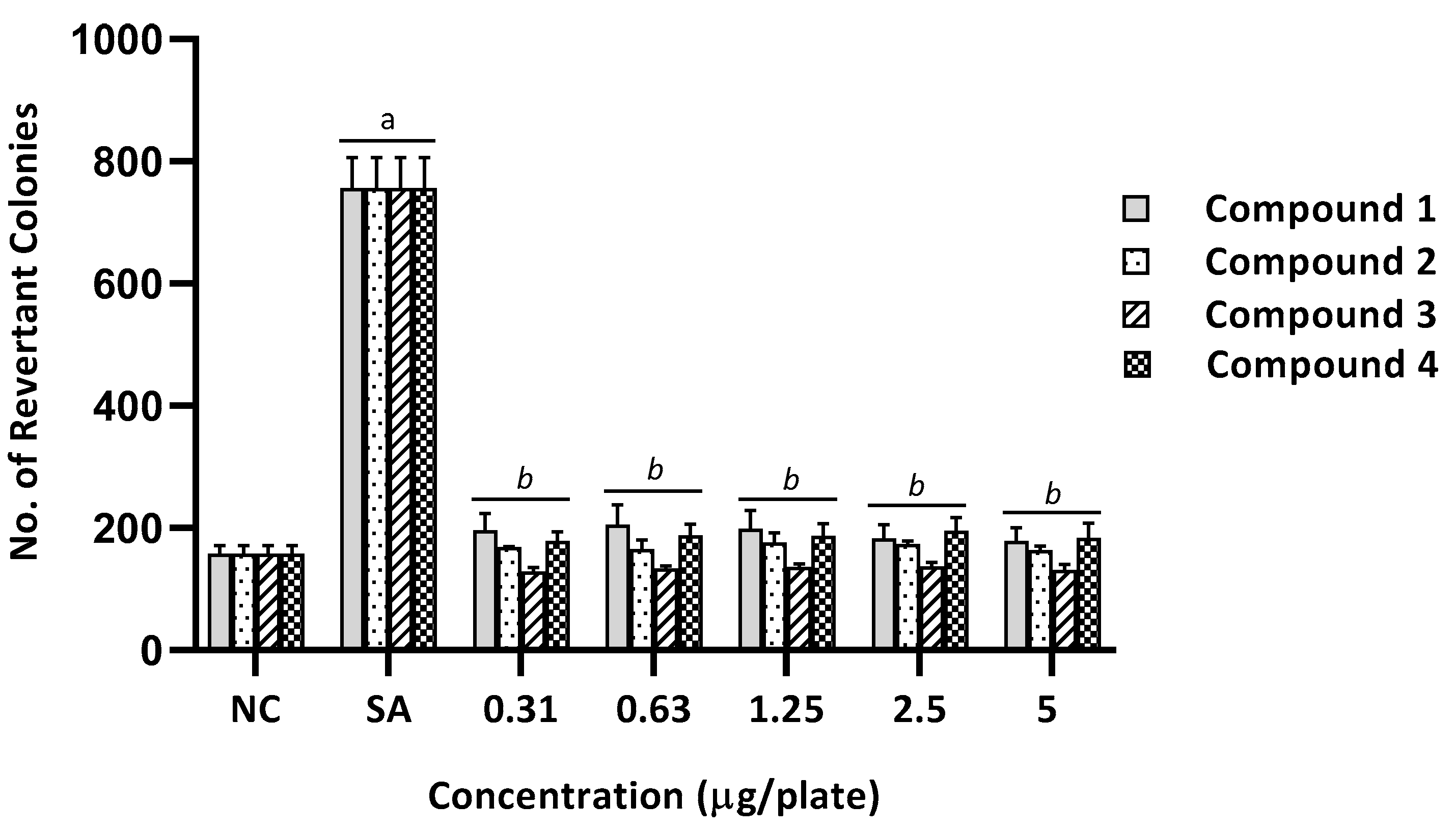
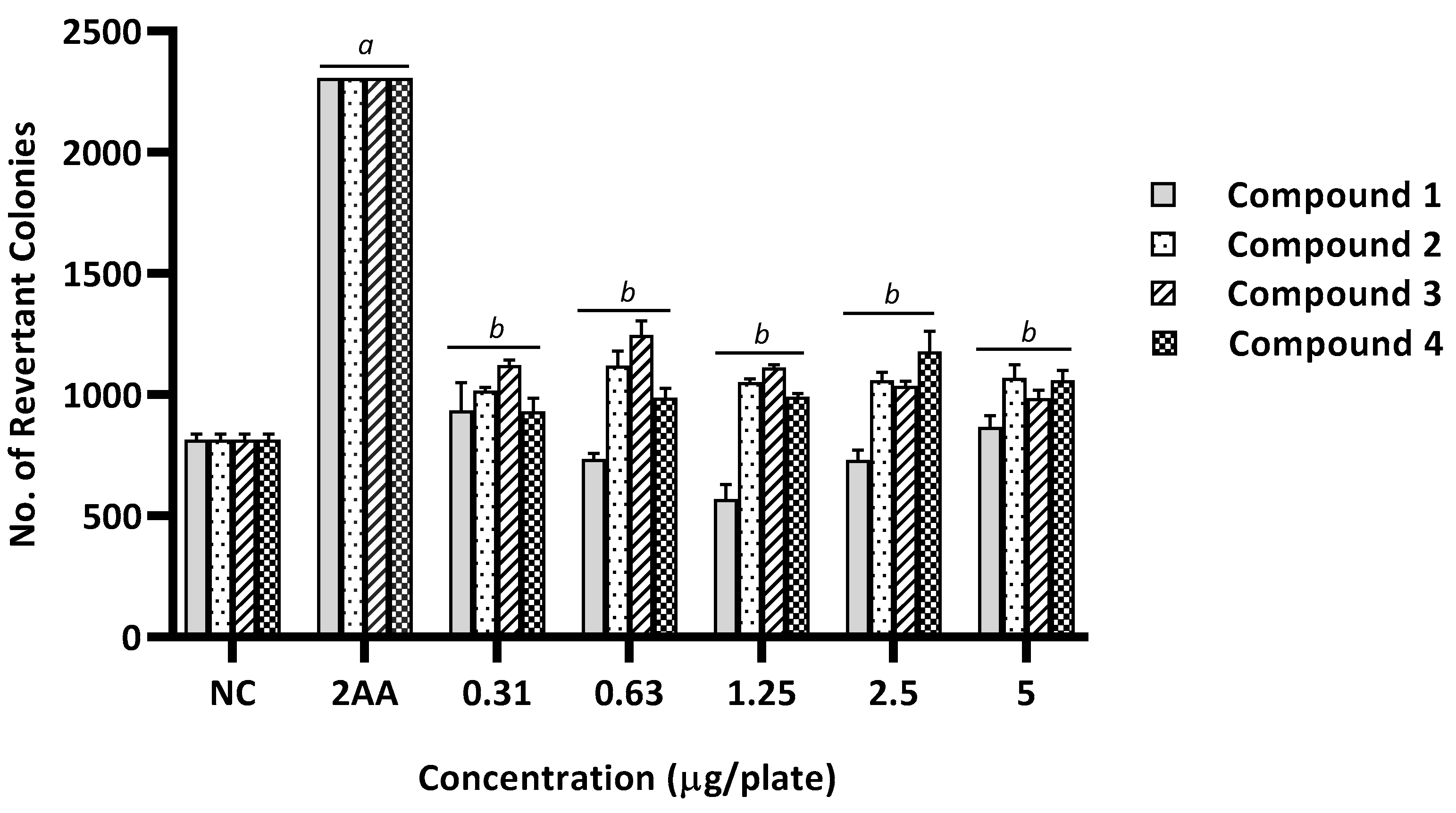
2.2. Mutagenicity Assay
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Bacterial Strains
4.3. Genetic Analysis
4.4. S9 Metabolic Activation System
4.5. Mutagenicity Assay
4.6. Data Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mortelmans, K.; Zeiger, E. The Ames Salmonella/microsome mutagenicity assay. Mutat. Res. 2000, 455, 29–60. [Google Scholar] [CrossRef]
- Loeb, K.R.; Loeb, L.A. Significance of multiple mutations in cancer. Carcinogenesis 2000, 21, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Słoczyńska, K.; Powroźnik, B.; Pękala, E.; Waszkielewicz, A.M. Antimutagenic compounds and their possible mechanisms of action. J. Appl. Genet. 2014, 55, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Verheyen, G.R.; Deun, K.V.; Miert, S.V. Testing the mutagenicity potential of chemicals. J. Genet. Genome Res. 2017, 4. [Google Scholar] [CrossRef]
- McCarren, P.; Springer, C.; Whitehead, L. An investigation into pharmaceutically relevant mutagenicity data and the influence on Ames predictive potential. J. Cheminformatics 2011, 3, 51. [Google Scholar] [CrossRef] [PubMed]
- Anjum, R.; Krakat, N.; Reza, M.T.; Klocke, M. Assessment of mutagenic potential of pyrolysis biochars by Ames Salmonella/mammalian-microsomal mutagenicity test. Ecotoxicol. Environ. Saf. 2014, 107, 306–312. [Google Scholar] [CrossRef] [PubMed]
- Bansal, Y.; Silakari, O. The therapeutic journey of benzimidazoles: A review. Bioorganic Med. Chem. 2012, 20, 6208–6236. [Google Scholar] [CrossRef] [PubMed]
- El Rashedy, A.A.; Aboul-Enein, H.Y. Benzimidazole derivatives as potential anticancer agents. Mini Rev. Med. Chem. 2013, 13, 399–407. [Google Scholar] [PubMed]
- Pullagura, M.K.P.; Kanvinde, A.; Raja, S. Potent Biological Agent Benzimidazole-A Review. Int. J. Pharm. Pharm. Sci. 2016, 8, 22–33. [Google Scholar]
- Rao, G.E.; Babu, P.S.; Koushik, O.S.; Sharmila, R.; Vijayabharathi, M.; Maruthikumar, S.; Pavankumar, P. A review on chemistry of benzimidazole nucleus and its biological significance. Int. J. Pharm. Chem. Biol. Sci. 2016, 6, 227–232. [Google Scholar]
- Zawawi, N.K.N.A.; Taha, M.; Ahmat, N.; Wadood, A.; Ismail, N.H.; Rahim, F.; Abdullah, N. Benzimidazole derivatives as new α-glucosidase inhibitors and in silico studies. Bioorganic Chem. 2016, 64, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Refaat, H.M. Synthesis and anticancer activity of some novel 2-substituted benzimidazole derivatives. Eur. J. Med. Chem. 2010, 45, 2949–2956. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.T.; Jiang, Z.; Shen, J.J.; Yi, H.; Zhan, Y.C.; Sha, M.Q.; Li, Z.R. Design, synthesis and biological evaluation of novel benzimidazole-2-substituted phenyl or pyridine propyl ketene derivatives as antitumour agents. Eur. J. Med. Chem. 2016, 114, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Gümüş, F.; Demirci, A.B.; Özden, T.; Eroğlu, H.; Diril, N. Synthesis, characterization and mutagenicity of new cis-[Pt (2-substituted-benzimidazole) 2Cl2] complexes. Pharmazie 2003, 58, 303–307. [Google Scholar] [PubMed]
- Alanyalı, F.S.; Arıcı, M.; Artagan, Ö.; Işıkdağ, İ.; Özkay, Y. Mutagenicity of Bisbenzimidazole Derivatives. Z. Naturforsch C. 2010, 65, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Maron, D.M.; Ames, B.N. Revised methods for the Salmonella mutagenicity test. Mutat. Res. 1983, 113, 173–215. [Google Scholar] [CrossRef]
- Creager, A.N.H.; Boudia, S.; Jas, N. The Political Life of Mutagens: A History of the Ames Test. In Powerless Science? Science and Politics in a Toxic World; Boudia, S., Jas, N., Eds.; Berghahn Books: New York, NY, USA, 2014; pp. 285–306. [Google Scholar]
- Santos, J.L.; Bosquesi, P.L.; Almeida, A.E.; Chin, C.M.; Varanda, E.A. Mutagenic and genotoxic effect of hydroxyurea. Int. J. Biomed. Sci.: IJBS 2011, 7, 263–267. [Google Scholar] [PubMed]
- Vijayan, V.; Pathak, U.; Meshram, G.P. Mutagenicity and antimutagenicity studies of DRDE-07 and its analogs against sulfur mustard in the in vitro Ames Salmonella/microsome assay. Mutat. Res. 2014, 773, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, C.C.; Véras, J.H.; Góes, B.R.; Pérez, C.N.; Chen-Chen, L. Mutagenicity and antimutagenicity of Salacia crassifolia (mart. Ex. Schult.) G. Don. evaluated by Ames test. Braz. J. Biol. 2018, 78, 345–350. [Google Scholar] [CrossRef] [PubMed]
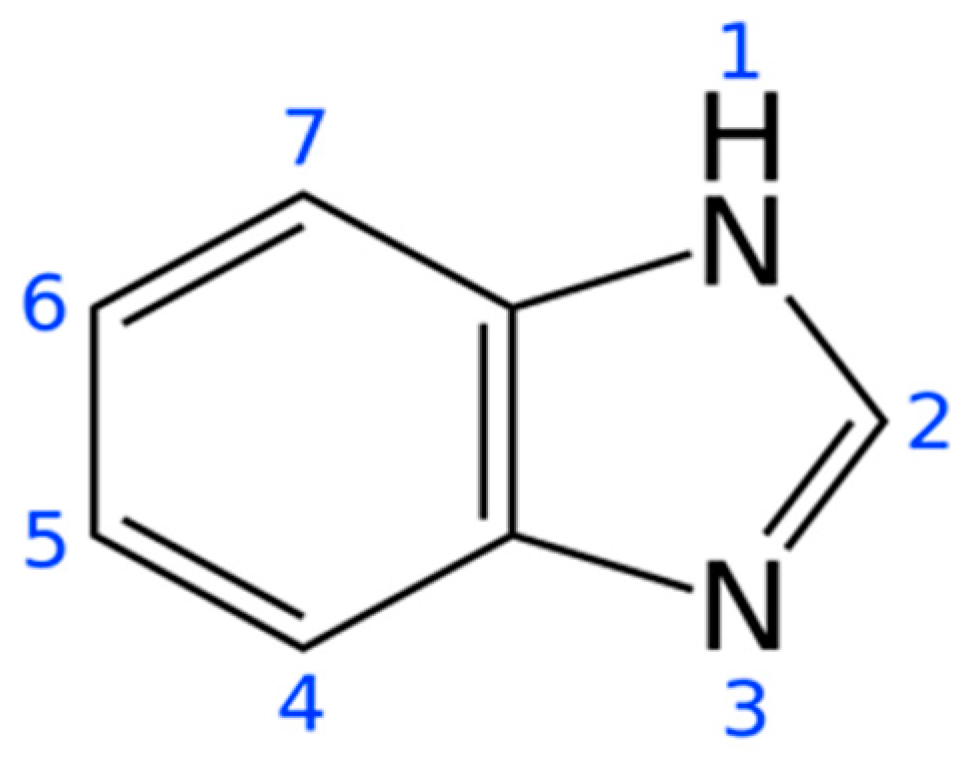
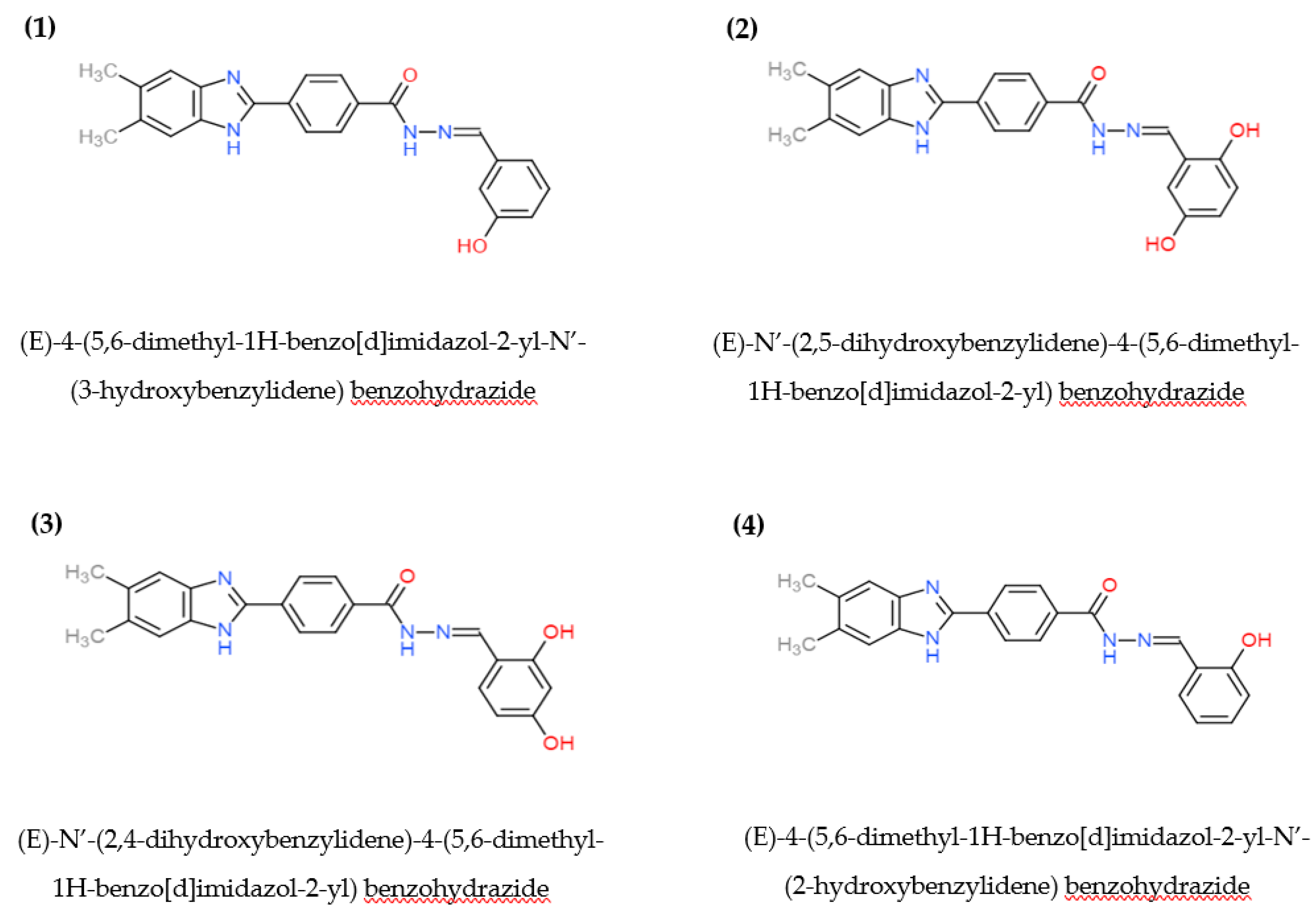
| Genetic Properties | Presence of Bacterial Growth on Plate | |
|---|---|---|
| TA98 | TA100 | |
| 1. Histidine dependence | x | x |
| 2. Biotin dependence | / | / |
| 3. Presence of rfa mutation | x | x |
| 4. Presence of uvrB deletion | x | x |
| 5. Presence of plasmid pKM101 | / | / |
| Compounds | Concentration (µg/plate) | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NC | PC | 0.31 | 0.63 | 1.25 | 2.5 | 5 | |||||||||
| −S9 | +S9 | −S9 | +S9 | −S9 | +S9 | −S9 | +S9 | −S9 | +S9 | −S9 | +S9 | −S9 | +S9 | ||
| 1 | 14 ± 2 (1.0) | 876 ± 83 (1.0) | 644 ± 6 (46) a | 1787 ± 82 (2.0) a | 15 ± 2 (1.1) | 1144 ± 85 (1.3) | 15 ± 3 (1.0) | 1033 ± 223 (1.2) | 15 ± 2 (1.1) | 1405 ± 99 (1.6) | 14 ± 3 (1.0) | 1939 ± 142 (2.2) a | 15 ± 3 (1.1) | 1363 ± 31 (1.6) | |
| 2 | 12 ± 2 (1.0) | 816 ± 79 (1.0) | 561 ± 7 (45.5) a | 1787 ± 82 (2.2) a | 17 ± 1 (1.4) | 1396 ± 28 (1.7) | 17 ± 2 (1.4) | 1137 ± 60 (1.4) | 20 ± 2 (1.6) | 1239 ± 13 (1.5) | 15 ± 3 (1.2) | 1160 ± 56 (1.4) | 18 ± 3 (1.5) | 1280 ± 28 (1.6) | |
| 3 | 13 ± 3 (1.0) | 675 ± 18 (1.0) | 631 ± 12 (48.5) a | 1680 ± 106 (2.5) a | 17.± 1 (1.3) | 916 ± 39 (1.4) | 14 ± 3 (1.1) | 965 ± 39 (1.4) | 18 ± 2 (1.4) | 923 ± 25 (1.4) | 12 ± 2 (1.0) | 920 ± 19 (1.4) | 20 ± 2 (1.5) | 1053 ± 57 (1.6) | |
| 4 | 14 ± 2 (1.0) | 816 ± 79 (1.0) | 644 ± 6 (46) a | 1787 ± 82 (2.2) a | 13 ± 2 (0.9) | 1311 ± 132 (1.6) | 14 ± 3 (1.0) | 1243 ± 111 (1.5) | 16 ± 2 (1.2) | 1368 ± 104 (1.7) | 14 ± 2 (1.0) | 1273 ± 66 (1.6) | 20 ± 2 (1.4) | 1396 ± 64 (1.7) | |
| Compounds | Concentration (µg/plate) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NC | PC | 0.31 | 0.63 | 1.25 | 2.5 | 5 | ||||||||
| −S9 | +S9 | −S9 | +S9 | −S9 | +S9 | −S9 | +S9 | −S9 | +S9 | −S9 | +S9 | −S9 | +S9 | |
| 1 | 172 ± 23 (1.0) | 821 ± 32 (1.0) | 676 ± 5 (3.9) a | 2307 ± 202 (2.8) a | 196 ± 28 (1.1) | 934 ± 116 (1.1) | 205 ± 33 (1.2) | 735 ± 23 (0.9) | 199 ± 30 (1.2) | 570 ± 59 (0.7) | 183 ± 23 (1.1) | 731 ± 40 (0.9) | 178 ± 22 (1.0) | 867 ± 47 (1.1) |
| 2 | 169 ± 4 (1.0) | 768 ± 94 (1.0) | 788 ± 28 (4.7) a | 2307 ± 202 (3.0) a | 168 ± 2 (0.1) | 1017 ± 13 (1.3) | 166 ± 15 (1.0) | 1120 ± 61 (1.5) | 176 ± 16 (1.1) | 1051 ± 15 (1.4) | 174 ± 5 (1.0) | 1060 ± 32 (1.4) | 164 ± 6 (1.0) | 1069 ± 54 (1.4) |
| 3 | 119 ± 5 (1.0) | 897 ± 5 (1.0) | 885 ± 27 (7.5) a | 2307 ± 202 (2.6) a | 129 ± 7 (1.1) | 1121 ± 21 (1.2) | 133 ± 4 (1.1) | 1246 ± 58 (1.4) | 136 ± 6 (1.2) | 1112 ± 12 (1.2) | 137 ± 6 (1.2) | 1036 ± 19 (1.2) | 131 ± 10 (1.1) | 985 ± 34 (1.1) |
| 4 | 172 ± 23 (1.0) | 768 ± 94 (1.0) | 676 ± 5 (3.9) a | 2307 ± 202 (3.0) a | 178 ± 15 (1.0) | 931 ± 55 (1.2) | 188 ± 19 (1.1) | 987 ± 40 (1.3) | 187 ± 20 (1.1) | 991 ± 15 (1.3) | 195 ± 23 (1.1) | 1179 ± 83 (1.5) | 183 ± 25 (1.1) | 1060 ± 40 (1.4) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azahar, N.H.; Ab dullah, S.S.; Abdullah, R.; Ahmat, N.; Md Akim, A.; Ab Hamid, H. Mutagenic Study of Benzimidazole Derivatives with (+S9) and without (−S9) Metabolic Activation. Int. J. Mol. Sci. 2019, 20, 4324. https://doi.org/10.3390/ijms20184324
Azahar NH, Ab dullah SS, Abdullah R, Ahmat N, Md Akim A, Ab Hamid H. Mutagenic Study of Benzimidazole Derivatives with (+S9) and without (−S9) Metabolic Activation. International Journal of Molecular Sciences. 2019; 20(18):4324. https://doi.org/10.3390/ijms20184324
Chicago/Turabian StyleAzahar, Nurul Hafizan, Siti Soleha Ab dullah, Rozaini Abdullah, Norizan Ahmat, Abdah Md Akim, and Hasiah Ab Hamid. 2019. "Mutagenic Study of Benzimidazole Derivatives with (+S9) and without (−S9) Metabolic Activation" International Journal of Molecular Sciences 20, no. 18: 4324. https://doi.org/10.3390/ijms20184324
APA StyleAzahar, N. H., Ab dullah, S. S., Abdullah, R., Ahmat, N., Md Akim, A., & Ab Hamid, H. (2019). Mutagenic Study of Benzimidazole Derivatives with (+S9) and without (−S9) Metabolic Activation. International Journal of Molecular Sciences, 20(18), 4324. https://doi.org/10.3390/ijms20184324






