Omega-3 Fatty Acids and Neurodegenerative Diseases: New Evidence in Clinical Trials
Abstract
1. Introduction
2. Parkinson’s Disease
2.1. The Role of Omega-3 Polyunsaturated Fatty Acids in PD: Observational Studies
2.2. The Role of Omega-3 Polyunsaturated Fatty Acids in PD: Randomized, Double-Blind, Placebo-Controlled Clinical Trials
3. Alzheimer’s Disease
3.1. The Role of Omega-3 Polyunsaturated Fatty Acids in AD: Observational Studies
3.2. The Role of Omega-3 Polyunsaturated Fatty Acids in AD: Randomized, Double-Blind, Placebo-Controlled Clinical Trials
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 5-LOX | 5-lipoxygenase |
| AD | Alzheimer’s disease |
| ADAS-Cog | Alzheimer’s disease assessment scale–cognitive subscale |
| ADCS | Activities of daily living scales |
| ALA | α linolenic acid |
| ApoE | apolipoprotein E |
| APP | amyloid precursor protein |
| ARA | arachidonic acid |
| Aβ | amyloid beta peptide |
| BADLS | Bristol’s activities of daily living scale |
| BASDEC | brief assessment schedule depression cards |
| BBB | blood–brain barrier |
| BDI | Beck depression inventory scale |
| CDR | clinical dementia rating scale |
| CGI | clinical global impression scale |
| CIBIC-Plus | clinician interview-based impression of change, plus carer interview |
| COX | cyclooxygenase |
| cPLA2 | cytosolic calcium-dependent phospholipase A2 |
| CSF | cerebrospinal fluid |
| DHA | docosahexaenoic acid |
| DHA-FFA | free nonesterified form docosahexaenoic acid |
| DSM-IV | Diagnostic and Statistical Manual of Mental Disorders, fourth edition |
| EPA | eicosapentaenoic acid |
| FABPs | fatty acid binding proteins |
| HDRS | Hamilton depression rating scale |
| hs-CRP | C-reactive protein |
| HVLT-R | Hopkins verbal learning test–revised |
| iPLA2 | cytosolic calcium-independent phospholipase A2 |
| LA | linoleic acid |
| LXA4 | lipoxin A4 |
| MADRS | Montgomery–Åsberg depression rating scale |
| MMSE | mini-mental state examination |
| MUFAs | monounsaturated fatty acids |
| NPI | Neuropsychiatric Inventory |
| NTB | neuropsychological test battery |
| PD | Parkinson’s disease |
| PKC | protein kinase C |
| PLA2 | phospholipase A2 |
| PUFAs | omega-3 polyunsaturated fatty acids |
| sPLA2 | secretory phospholipase A2 |
| RvD1 | resolvin D1 |
| TAC | total antioxidant capacity |
| UPDRS | unified Parkinson’s disease rating scale |
References
- Cansev, M.; Wurtman, R.J.; Sakamoto, T.; Ulus, I.H. Oral Administration of Circulating Precursors for Membrane Phosphatides Can Promote the Synthesis of New Brain Synapses. Alzheimers Dement. J. Alzheimers Assoc. 2008, 4, S153–S168. [Google Scholar] [CrossRef] [PubMed]
- Beltz, B.S.; Tlusty, M.F.; Benton, J.L.; Sandeman, D.C. Omega-3 Fatty Acids Upregulate Adult Neurogenesis. Neurosci. Lett. 2007, 415, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Kawakita, E.; Hashimoto, M.; Shido, O. Docosahexaenoic Acid Promotes Neurogenesis in Vitro and in Vivo. Neuroscience 2006, 139, 991–997. [Google Scholar] [CrossRef] [PubMed]
- Cutuli, D. Functional and Structural Benefits Induced by Omega-3 Polyunsaturated Fatty Acids During Aging. Curr. Neuropharmacol. 2017, 15, 534–542. [Google Scholar] [CrossRef] [PubMed]
- Aryal, S.; Hussain, S.; Drevon, C.A.; Nagelhus, E.; Hvalby, Ø.; Jensen, V.; Walaas, S.I.; Davanger, S. Omega-3 Fatty Acids Regulate Plasticity in Distinct Hippocampal Glutamatergic Synapses. Eur. J. Neurosci. 2019, 49, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Castro-Gómez, P.; Garcia-Serrano, A.; Visioli, F.; Fontecha, J. Relevance of Dietary Glycerophospholipids and Sphingolipids to Human Health. Prostaglandins Leukot. Essent. Fat. Acids 2015, 101, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, C.; Afonso, C.; Bandarra, N.M. Dietary DHA and Health: Cognitive Function Ageing. Nutr. Res. Rev. 2016, 29, 281–294. [Google Scholar] [CrossRef]
- Calviello, G.; Su, H.-M.; Weylandt, K.H.; Fasano, E.; Serini, S.; Cittadini, A. Experimental Evidence of ω-3 Polyunsaturated Fatty Acid Modulation of Inflammatory Cytokines and Bioactive Lipid Mediators: Their Potential Role in Inflammatory, Neurodegenerative, and Neoplastic Diseases. Biomed Res. Int. 2013, 2013. [Google Scholar] [CrossRef]
- Moore, K.; Hughes, C.F.; Ward, M.; Hoey, L.; McNulty, H. Diet, Nutrition and the Ageing Brain: Current Evidence and New Directions. Proc. Nutr. Soc. 2018, 77, 152–163. [Google Scholar] [CrossRef]
- Yassine, H.N.; Braskie, M.N.; Mack, W.J.; Castor, K.J.; Fonteh, A.N.; Schneider, L.S.; Harrington, M.G.; Chui, H.C. Association of Docosahexaenoic Acid Supplementation with Alzheimer Disease Stage in Apolipoprotein E Ε4 Carriers: A Review. JAMA Neurol. 2017, 74, 339–347. [Google Scholar] [CrossRef]
- Araya-Quintanilla, F.; Gutiérrez-Espinoza, H.; Sánchez-Montoya, U.; Muñoz-Yañez, M.J.; Baeza-Vergara, A.; Petersen-Yanjarí, M.; Fernández-Lecaros, L. Effectiveness of Omega-3 Fatty Acid Supplementation in Patients with Alzheimer Disease: A Systematic Review and Meta-Analysis. Neurol. Barc. Spain 2017. [Google Scholar] [CrossRef]
- Butler, M.; Nelson, V.A.; Davila, H.; Ratner, E.; Fink, H.A.; Hemmy, L.S.; McCarten, J.R.; Barclay, T.R.; Brasure, M.; Kane, R.L. Over-the-Counter Supplement Interventions to Prevent Cognitive Decline, Mild Cognitive Impairment, and Clinical Alzheimer-Type Dementia: A Systematic Review. Ann. Intern. Med. 2018, 168, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Burckhardt, M.; Herke, M.; Wustmann, T.; Watzke, S.; Langer, G.; Fink, A. Omega-3 Fatty Acids for the Treatment of Dementia. Cochrane Database Syst. Rev. 2016, 4. [Google Scholar] [CrossRef] [PubMed]
- Weiser, M.J.; Butt, C.M.; Mohajeri, M.H. Docosahexaenoic Acid and Cognition throughout the Lifespan. Nutrients 2016, 8, 99. [Google Scholar] [CrossRef] [PubMed]
- Chappus-McCendie, H.; Chevalier, L.; Roberge, C.; Plourde, M. Omega-3 PUFA Metabolism and Brain Modifications during Aging. Prog. Neuropsychopharmacol. Biol. Psychiatry 2019, 94. [Google Scholar] [CrossRef] [PubMed]
- Brenna, J.T.; Diau, G.-Y. The Influence of Dietary Docosahexaenoic Acid and Arachidonic Acid on Central Nervous System Polyunsaturated Fatty Acid Composition. Prostaglandins Leukot. Essent. Fat. Acids 2007, 77, 247–250. [Google Scholar] [CrossRef] [PubMed]
- Wassall, S.R.; Leng, X.; Canner, S.W.; Pennington, E.R.; Kinnun, J.J.; Cavazos, A.T.; Dadoo, S.; Johnson, D.; Heberle, F.A.; Katsaras, J.; et al. Docosahexaenoic Acid Regulates the Formation of Lipid Rafts: A Unified View from Experiment and Simulation. Biochim. Biophys. Acta Biomembr. 2018, 1860, 1985–1993. [Google Scholar] [CrossRef]
- Uauy, R.; Dangour, A.D. Nutrition in Brain Development and Aging: Role of Essential Fatty Acids. Nutr. Rev. 2006, 64, S24–S33. [Google Scholar] [CrossRef]
- De Bus, I.; Witkamp, R.; Zuilhof, H.; Albada, B.; Balvers, M. The Role of N-3 PUFA-Derived Fatty Acid Derivatives and Their Oxygenated Metabolites in the Modulation of Inflammation. Prostaglandins Other Lipid Mediat. 2019, 144. [Google Scholar] [CrossRef]
- Janssen, C.I.F.; Kiliaan, A.J. Long-Chain Polyunsaturated Fatty Acids (LCPUFA) from Genesis to Senescence: The Influence of LCPUFA on Neural Development, Aging, and Neurodegeneration. Prog. Lipid Res. 2014, 53, 1–17. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The Omega-6/Omega-3 Fatty Acid Ratio, Genetic Variation, and Cardiovascular Disease. Asia Pac. J. Clin. Nutr. 2008, 17, 131–134. [Google Scholar] [PubMed]
- Schmitz, G.; Ecker, J. The Opposing Effects of N-3 and n-6 Fatty Acids. Prog. Lipid Res. 2008, 47, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Subbaiah, P.V. Regioisomers of Phosphatidylcholine Containing DHA and Their Potential to Deliver DHA to the Brain: Role of Phospholipase Specificities. Lipids 2013, 48, 675–686. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.-Z.; Mita, R.; Beaulieu, M.; Gao, Z.; Godbout, R. Fatty Acid Binding Proteins in Brain Development and Disease. Int. J. Dev. Biol. 2010, 54, 1229–1239. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Scanlon, M.J.; Owada, Y.; Yamamoto, Y.; Porter, C.J.H.; Nicolazzo, J.A. Fatty Acid-Binding Protein 5 Facilitates the Blood-Brain Barrier Transport of Docosahexaenoic Acid. Mol. Pharm. 2015, 12, 4375–4385. [Google Scholar] [CrossRef] [PubMed]
- Vandal, M.; Alata, W.; Tremblay, C.; Rioux-Perreault, C.; Salem, N.; Calon, F.; Plourde, M. Reduction in DHA Transport to the Brain of Mice Expressing Human APOE4 Compared to APOE2. J. Neurochem. 2014, 129, 516–526. [Google Scholar] [CrossRef] [PubMed]
- Ouellet, M.; Emond, V.; Chen, C.T.; Julien, C.; Bourasset, F.; Oddo, S.; LaFerla, F.; Bazinet, R.P.; Calon, F. Diffusion of Docosahexaenoic and Eicosapentaenoic Acids through the Blood-Brain Barrier: An in Situ Cerebral Perfusion Study. Neurochem. Int. 2009, 55, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.T.; Green, J.T.; Orr, S.K.; Bazinet, R.P. Regulation of Brain Polyunsaturated Fatty Acid Uptake and Turnover. Prostaglandins Leukot. Essent. Fat. Acids 2008, 79, 85–91. [Google Scholar] [CrossRef]
- Zárate, R.; El Jaber-Vazdekis, N.; Tejera, N.; Pérez, J.A.; Rodríguez, C. Significance of Long Chain Polyunsaturated Fatty Acids in Human Health. Clin. Transl. Med. 2017, 6, 25. [Google Scholar] [CrossRef]
- Murakami, M. Lipoquality Control by Phospholipase A2 Enzymes. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2017, 93, 677–702. [Google Scholar] [CrossRef]
- Innes, J.K.; Calder, P.C. Omega-6 Fatty Acids and Inflammation. Prostaglandins Leukot. Essent. Fat. Acids 2018, 132, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Tassoni, D.; Kaur, G.; Weisinger, R.S.; Sinclair, A.J. The Role of Eicosanoids in the Brain. Asia Pac. J. Clin. Nutr. 2008, 17 (Suppl. 1), 220–228. [Google Scholar] [PubMed]
- Tapiero, H.; Ba, G.N.; Couvreur, P.; Tew, K.D. Polyunsaturated Fatty Acids (PUFA) and Eicosanoids in Human Health and Pathologies. Biomed. Pharm. 2002, 56, 215–222. [Google Scholar] [CrossRef]
- Dyall, S.C.; Michael-Titus, A.T. Neurological Benefits of Omega-3 Fatty Acids. Neuromol. Med. 2008, 10, 219–235. [Google Scholar] [CrossRef] [PubMed]
- Pawlosky, R.J.; Hibbeln, J.R.; Novotny, J.A.; Salem, N. Physiological Compartmental Analysis of Alpha-Linolenic Acid Metabolism in Adult Humans. J. Lipid Res. 2001, 42, 1257–1265. [Google Scholar] [PubMed]
- Brenna, J.T. Efficiency of Conversion of Alpha-Linolenic Acid to Long Chain n-3 Fatty Acids in Man. Curr. Opin. Clin. Nutr. Metab. Care 2002, 5, 127–132. [Google Scholar] [CrossRef]
- Burdge, G.C.; Wootton, S.A. Conversion of Alpha-Linolenic Acid to Eicosapentaenoic, Docosapentaenoic and Docosahexaenoic Acids in Young Women. Br. J. Nutr. 2002, 88, 411–420. [Google Scholar] [CrossRef]
- Burdge, G.C. Metabolism of Alpha-Linolenic Acid in Humans. Prostaglandins Leukot. Essent. Fat. Acids 2006, 75, 161–168. [Google Scholar] [CrossRef]
- Thompson, M.; Hein, N.; Hanson, C.; Smith, L.M.; Anderson-Berry, A.; Richter, C.K.; Stessy Bisselou, K.; Kusi Appiah, A.; Kris-Etherton, P.; Skulas-Ray, A.C.; et al. Omega-3 Fatty Acid Intake by Age, Gender, and Pregnancy Status in the United States: National Health and Nutrition Examination Survey 2003–2014. Nutrients 2019, 11, 177. [Google Scholar] [CrossRef]
- Scientific Opinion on the Tolerable Upper Intake Level of Eicosapentaenoic Acid (EPA), Docosahexaenoic Acid (DHA) and Docosapentaenoic Acid (DPA). EFSA J. 2012, 10. [CrossRef]
- Ghasemi Fard, S.; Wang, F.; Sinclair, A.J.; Elliott, G.; Turchini, G.M. How Does High DHA Fish Oil Affect Health? A Systematic Review of Evidence. Crit. Rev. Food Sci. Nutr. 2019, 59, 1684–1727. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, Q.V.; Malau-Aduli, B.S.; Cavalieri, J.; Nichols, P.D.; Malau-Aduli, A.E.O. Enhancing Omega-3 Long-Chain Polyunsaturated Fatty Acid Content of Dairy-Derived Foods for Human Consumption. Nutrients 2019, 11, 743. [Google Scholar] [CrossRef] [PubMed]
- Office of Dietary Supplements—Omega-3 Fatty Acids. Available online: https://ods.od.nih.gov/factsheets/Omega3FattyAcids-HealthProfessional/ (accessed on 7 June 2019).
- Da Silva, T.M.; Munhoz, R.P.; Alvarez, C.; Naliwaiko, K.; Kiss, Á.; Andreatini, R.; Ferraz, A.C. Depression in Parkinson’s Disease: A Double-Blind, Randomized, Placebo-Controlled Pilot Study of Omega-3 Fatty-Acid Supplementation. J. Affect. Disord. 2008, 111, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Kamel, F.; Goldman, S.M.; Umbach, D.M.; Chen, H.; Richardson, G.; Barber, M.R.; Meng, C.; Marras, C.; Korell, M.; Kasten, M.; et al. Dietary Fat Intake, Pesticide Use, and Parkinson’s Disease. Parkinsonism Relat. Disord. 2014, 20, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Elfawy, H.A.; Das, B. Crosstalk between Mitochondrial Dysfunction, Oxidative Stress, and Age Related Neurodegenerative Disease: Etiologies and Therapeutic Strategies. Life Sci. 2019, 218, 165–184. [Google Scholar] [CrossRef] [PubMed]
- Canhada, S.; Castro, K.; Perry, I.S.; Luft, V.C. Omega-3 Fatty Acids’ Supplementation in Alzheimer’s Disease: A Systematic Review. Nutr. Neurosci. 2018, 21, 529–538. [Google Scholar] [CrossRef] [PubMed]
- Abbott, R.D.; Webster Ross, G.; White, L.R.; Sanderson, W.T.; Burchfiel, C.M.; Kashon, M.; Sharp, D.S.; Masaki, K.H.; Curb, J.D.; Petrovitch, H. Environmental, Life-Style, Andphysical Precursors of Clinical Parkinson’s Disease: Recentfindings from the Honolulu-Asia Aging Study. J. Neurol. 2003, 250. [Google Scholar] [CrossRef]
- De Lau, L.M.L.; Bornebroek, M.; Witteman, J.C.M.; Hofman, A.; Koudstaal, P.J.; Breteler, M.M.B. Dietary Fatty Acids and the Risk of Parkinson Disease: The Rotterdam Study. Neurology 2005, 64, 2040–2045. [Google Scholar] [CrossRef]
- Gao, X.; Chen, H.; Fung, T.T.; Logroscino, G.; Schwarzschild, M.A.; Hu, F.B.; Ascherio, A. Prospective Study of Dietary Pattern and Risk of Parkinson Disease. Am. J. Clin. Nutr. 2007, 86, 1486–1494. [Google Scholar] [CrossRef]
- Miyake, Y.; Sasaki, S.; Tanaka, K.; Fukushima, W.; Kiyohara, C.; Tsuboi, Y.; Yamada, T.; Oeda, T.; Miki, T.; Kawamura, N.; et al. Dietary Fat Intake and Risk of Parkinson’s Disease: A Case-Control Study in Japan. J. Neurol. Sci. 2010, 288, 117–122. [Google Scholar] [CrossRef]
- Pomponi, M.; Loria, G.; Salvati, S.; Di Biase, A.; Conte, G.; Villella, C.; Righino, E.; Ciciarelli, C.; Bria, P.; La Torre, G.; et al. DHA Effects in Parkinson Disease Depression. Basal Ganglia 2014, 4, 61–66. [Google Scholar] [CrossRef]
- Taghizadeh, M.; Tamtaji, O.R.; Dadgostar, E.; Daneshvar Kakhaki, R.; Bahmani, F.; Abolhassani, J.; Aarabi, M.H.; Kouchaki, E.; Memarzadeh, M.R.; Asemi, Z. The Effects of Omega-3 Fatty Acids and Vitamin E Co-Supplementation on Clinical and Metabolic Status in Patients with Parkinson’s Disease: A Randomized, Double-Blind, Placebo-Controlled Trial. Neurochem. Int. 2017, 108, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Sanz, P.; Ruiz-Gabarre, D.; García-Escudero, V. Modulating Effect of Diet on Alzheimer’s Disease. Dis. Basel Switz. 2019, 7, 12. [Google Scholar] [CrossRef]
- Grimm, M.O.W.; Michaelson, D.M.; Hartmann, T. Omega-3 Fatty Acids, Lipids, and ApoE Lipidation in Alzheimer’s Disease: A Rationale for Multi-Nutrient Dementia Prevention. J. Lipid Res. 2017, 58, 2083–2101. [Google Scholar] [CrossRef] [PubMed]
- Cunnane, S.C.; Plourde, M.; Pifferi, F.; Bégin, M.; Féart, C.; Barberger-Gateau, P. Fish, Docosahexaenoic Acid and Alzheimer’s Disease. Prog. Lipid Res. 2009, 48, 239–256. [Google Scholar] [CrossRef]
- Kalmijn, S.; Launer, L.J.; Ott, A.; Witteman, J.C.; Hofman, A.; Breteler, M.M. Dietary Fat Intake and the Risk of Incident Dementia in the Rotterdam Study. Ann. Neurol. 1997, 42, 776–782. [Google Scholar] [CrossRef]
- Morris, M.C.; Evans, D.A.; Bienias, J.L.; Tangney, C.C.; Bennett, D.A.; Wilson, R.S.; Aggarwal, N.; Schneider, J. Consumption of Fish and N-3 Fatty Acids and Risk of Incident Alzheimer Disease. Arch. Neurol. 2003, 60, 940–946. [Google Scholar] [CrossRef]
- Huang, T.L.; Zandi, P.P.; Tucker, K.L.; Fitzpatrick, A.L.; Kuller, L.H.; Fried, L.P.; Burke, G.L.; Carlson, M.C. Benefits of Fatty Fish on Dementia Risk Are Stronger for Those without APOE Epsilon4. Neurology 2005, 65, 1409–1414. [Google Scholar] [CrossRef]
- Schaefer, E.J.; Bongard, V.; Beiser, A.S.; Lamon-Fava, S.; Robins, S.J.; Au, R.; Tucker, K.L.; Kyle, D.J.; Wilson, P.W.F.; Wolf, P.A. Plasma Phosphatidylcholine Docosahexaenoic Acid Content and Risk of Dementia and Alzheimer Disease: The Framingham Heart Study. Arch. Neurol. 2006, 63, 1545–1550. [Google Scholar] [CrossRef]
- Barberger-Gateau, P.; Raffaitin, C.; Letenneur, L.; Berr, C.; Tzourio, C.; Dartigues, J.F.; Alpérovitch, A. Dietary Patterns and Risk of Dementia: The Three-City Cohort Study. Neurology 2007, 69, 1921–1930. [Google Scholar] [CrossRef]
- Devore, E.E.; Grodstein, F.; van Rooij, F.J.A.; Hofman, A.; Rosner, B.; Stampfer, M.J.; Witteman, J.C.M.; Breteler, M.M.B. Dietary Intake of Fish and Omega-3 Fatty Acids in Relation to Long-Term Dementia Risk. Am. J. Clin. Nutr. 2009, 90, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.C.; Tangney, C.C.; Wang, Y.; Sacks, F.M.; Bennett, D.A.; Aggarwal, N.T. MIND Diet Associated with Reduced Incidence of Alzheimer’s Disease. Alzheimers Dement. J. Alzheimers Assoc. 2015, 11, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- Freund-Levi, Y.; Eriksdotter-Jönhagen, M.; Cederholm, T.; Basun, H.; Faxén-Irving, G.; Garlind, A.; Vedin, I.; Vessby, B.; Wahlund, L.-O.; Palmblad, J. Omega-3 Fatty Acid Treatment in 174 Patients with Mild to Moderate Alzheimer Disease: OmegAD Study: A Randomized Double-Blind Trial. Arch. Neurol. 2006, 63, 1402–1408. [Google Scholar] [CrossRef] [PubMed]
- Freund-Levi, Y.; Basun, H.; Cederholm, T.; Faxén-Irving, G.; Garlind, A.; Grut, M.; Vedin, I.; Palmblad, J.; Wahlund, L.-O.; Eriksdotter-Jönhagen, M. Omega-3 Supplementation in Mild to Moderate Alzheimer’s Disease: Effects on Neuropsychiatric Symptoms. Int. J. Geriatr. Psychiatry 2008, 23, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.-C.; Su, K.-P.; Cheng, T.-C.; Liu, H.-C.; Chang, C.-J.; Dewey, M.E.; Stewart, R.; Huang, S.-Y. The Effects of Omega-3 Fatty Acids Monotherapy in Alzheimer’s Disease and Mild Cognitive Impairment: A Preliminary Randomized Double-Blind Placebo-Controlled Study. Prog. Neuropsychopharmacol. Biol. Psychiatry 2008, 32, 1538–1544. [Google Scholar] [CrossRef] [PubMed]
- Quinn, J.F.; Raman, R.; Thomas, R.G.; Yurko-Mauro, K.; Nelson, E.B.; Van Dyck, C.; Galvin, J.E.; Emond, J.; Jack, C.R.; Weiner, M.; et al. Docosahexaenoic Acid Supplementation and Cognitive Decline in Alzheimer Disease: A Randomized Trial. JAMA 2010, 304, 1903–1911. [Google Scholar] [CrossRef] [PubMed]
- Scheltens, P.; Kamphuis, P.J.G.H.; Verhey, F.R.J.; Olde Rikkert, M.G.M.; Wurtman, R.J.; Wilkinson, D.; Twisk, J.W.R.; Kurz, A. Efficacy of a Medical Food in Mild Alzheimer’s Disease: A Randomized, Controlled Trial. Alzheimers Dement. J. Alzheimers Assoc. 2010, 6. [Google Scholar] [CrossRef] [PubMed]
- Kamphuis, P.J.G.H.; Verhey, F.R.J.; Olde Rikkert, M.G.M.; Twisk, J.W.R.; Swinkels, S.H.N.; Scheltens, P. Efficacy of a Medical Food on Cognition in Alzheimer’s Disease: Results from Secondary Analyses of a Randomized, Controlled Trial. J. Nutr. Health Aging 2011, 15, 720–724. [Google Scholar] [CrossRef]
- Scheltens, P.; Twisk, J.W.R.; Blesa, R.; Scarpini, E.; von Arnim, C.A.F.; Bongers, A.; Harrison, J.; Swinkels, S.H.N.; Stam, C.J.; de Waal, H.; et al. Efficacy of Souvenaid in Mild Alzheimer’s Disease: Results from a Randomized, Controlled Trial. J. Alzheimers Dis. 2012, 31, 225–236. [Google Scholar] [CrossRef]
- Shah, R.C.; Kamphuis, P.J.; Leurgans, S.; Swinkels, S.H.; Sadowsky, C.H.; Bongers, A.; Rappaport, S.A.; Quinn, J.F.; Wieggers, R.L.; Scheltens, P.; et al. The S-Connect Study: Results from a Randomized, Controlled Trial of Souvenaid in Mild-to-Moderate Alzheimer’s Disease. Alzheimers Res. 2013, 5. [Google Scholar] [CrossRef]
- Faxén-Irving, G.; Freund-Levi, Y.; Eriksdotter-Jönhagen, M.; Basun, H.; Hjorth, E.; Palmblad, J.; Vedin, I.; Cederholm, T.; Wahlund, L.-O. Effects on Transthyretin in Plasma and Cerebrospinal Fluid by DHA-Rich n − 3 Fatty Acid Supplementation in Patients with Alzheimer’s Disease: The OmegAD Study. J. Alzheimers Dis. 2013, 36, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Shinto, L.; Quinn, J.; Montine, T.; Dodge, H.H.; Woodward, W.; Baldauf-Wagner, S.; Waichunas, D.; Bumgarner, L.; Bourdette, D.; Silbert, L.; et al. A Randomized Placebo-Controlled Pilot Trial of Omega-3 Fatty Acids and Alpha Lipoic Acid in Alzheimer’s Disease. J. Alzheimers Dis. 2014, 38, 111–120. [Google Scholar] [CrossRef] [PubMed]
- De Waal, H.; Stam, C.J.; Lansbergen, M.M.; Wieggers, R.L.; Kamphuis, P.J.G.H.; Scheltens, P.; Maestú, F.; van Straaten, E.C.W. The Effect of Souvenaid on Functional Brain Network Organisation in Patients with Mild Alzheimer’s Disease: A Randomised Controlled Study. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Phillips, M.A.; Childs, C.E.; Calder, P.C.; Rogers, P.J. No Effect of Omega-3 Fatty Acid Supplementation on Cognition and Mood in Individuals with Cognitive Impairment and Probable Alzheimer’s Disease: A Randomised Controlled Trial. Int. J. Mol. Sci. 2015, 16, 24600–24613. [Google Scholar] [CrossRef] [PubMed]
- Eriksdotter, M.; Vedin, I.; Falahati, F.; Freund-Levi, Y.; Hjorth, E.; Faxen-Irving, G.; Wahlund, L.-O.; Schultzberg, M.; Basun, H.; Cederholm, T.; et al. Plasma Fatty Acid Profiles in Relation to Cognition and Gender in Alzheimer’s Disease Patients During Oral Omega-3 Fatty Acid Supplementation: The OmegAD Study. J. Alzheimers Dis. 2015, 48, 805–812. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Hjorth, E.; Vedin, I.; Eriksdotter, M.; Freund-Levi, Y.; Wahlund, L.-O.; Cederholm, T.; Palmblad, J.; Schultzberg, M. Effects of N-3 FA Supplementation on the Release of Proresolving Lipid Mediators by Blood Mononuclear Cells: The OmegAD Study. J. Lipid Res. 2015, 56, 674–681. [Google Scholar] [CrossRef] [PubMed]
- Olde Rikkert, M.G.M.; Verhey, F.R.; Blesa, R.; von Arnim, C.A.F.; Bongers, A.; Harrison, J.; Sijben, J.; Scarpini, E.; Vandewoude, M.F.J.; Vellas, B.; et al. Tolerability and Safety of Souvenaid in Patients with Mild Alzheimer’s Disease: Results of Multi-Center, 24-Week, Open-Label Extension Study. J. Alzheimers Dis. 2015, 44, 471–480. [Google Scholar] [CrossRef]
- Jernerén, F.; Cederholm, T.; Refsum, H.; Smith, A.D.; Turner, C.; Palmblad, J.; Eriksdotter, M.; Hjorth, E.; Faxen-Irving, G.; Wahlund, L.-O.; et al. Homocysteine Status Modifies the Treatment Effect of Omega-3 Fatty Acids on Cognition in a Randomized Clinical Trial in Mild to Moderate Alzheimer’s Disease: The OmegAD Study. J. Alzheimers Dis. 2019, 69, 189–197. [Google Scholar] [CrossRef]
- Freund Levi, Y.; Vedin, I.; Cederholm, T.; Basun, H.; Faxén Irving, G.; Eriksdotter, M.; Hjorth, E.; Schultzberg, M.; Vessby, B.; Wahlund, L.-O.; et al. Transfer of Omega-3 Fatty Acids across the Blood-Brain Barrier after Dietary Supplementation with a Docosahexaenoic Acid-Rich Omega-3 Fatty Acid Preparation in Patients with Alzheimer’s Disease: The OmegAD Study. J. Intern. Med. 2014, 275, 428–436. [Google Scholar] [CrossRef]
- Cummings, J.; Scheltens, P.; McKeith, I.; Blesa, R.; Harrison, J.E.; Bertolucci, P.H.F.; Rockwood, K.; Wilkinson, D.; Wijker, W.; Bennett, D.A.; et al. Effect Size Analyses of Souvenaid in Patients with Alzheimer’s Disease. J. Alzheimers Dis. 2017, 55, 1131–1139. [Google Scholar] [CrossRef]
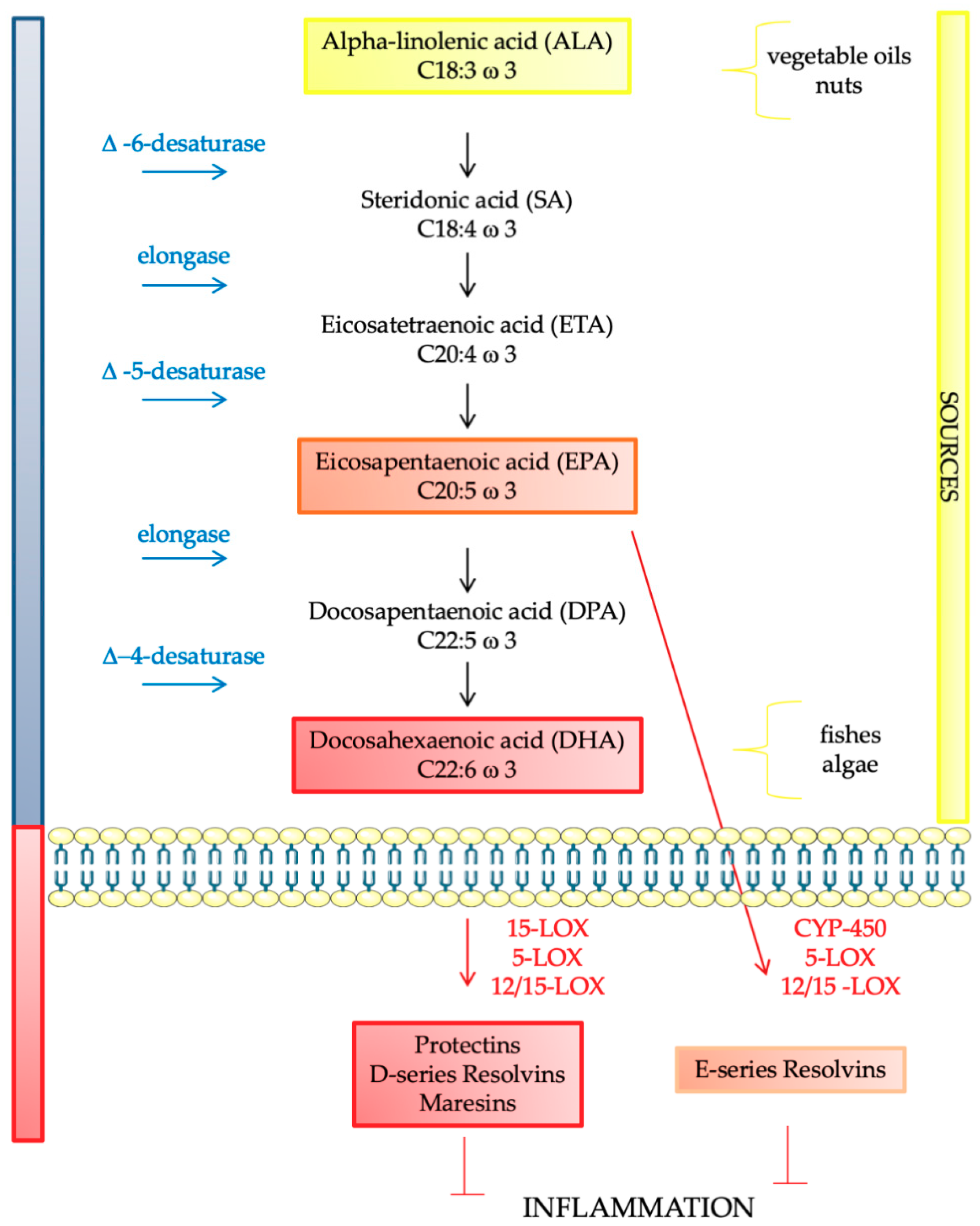
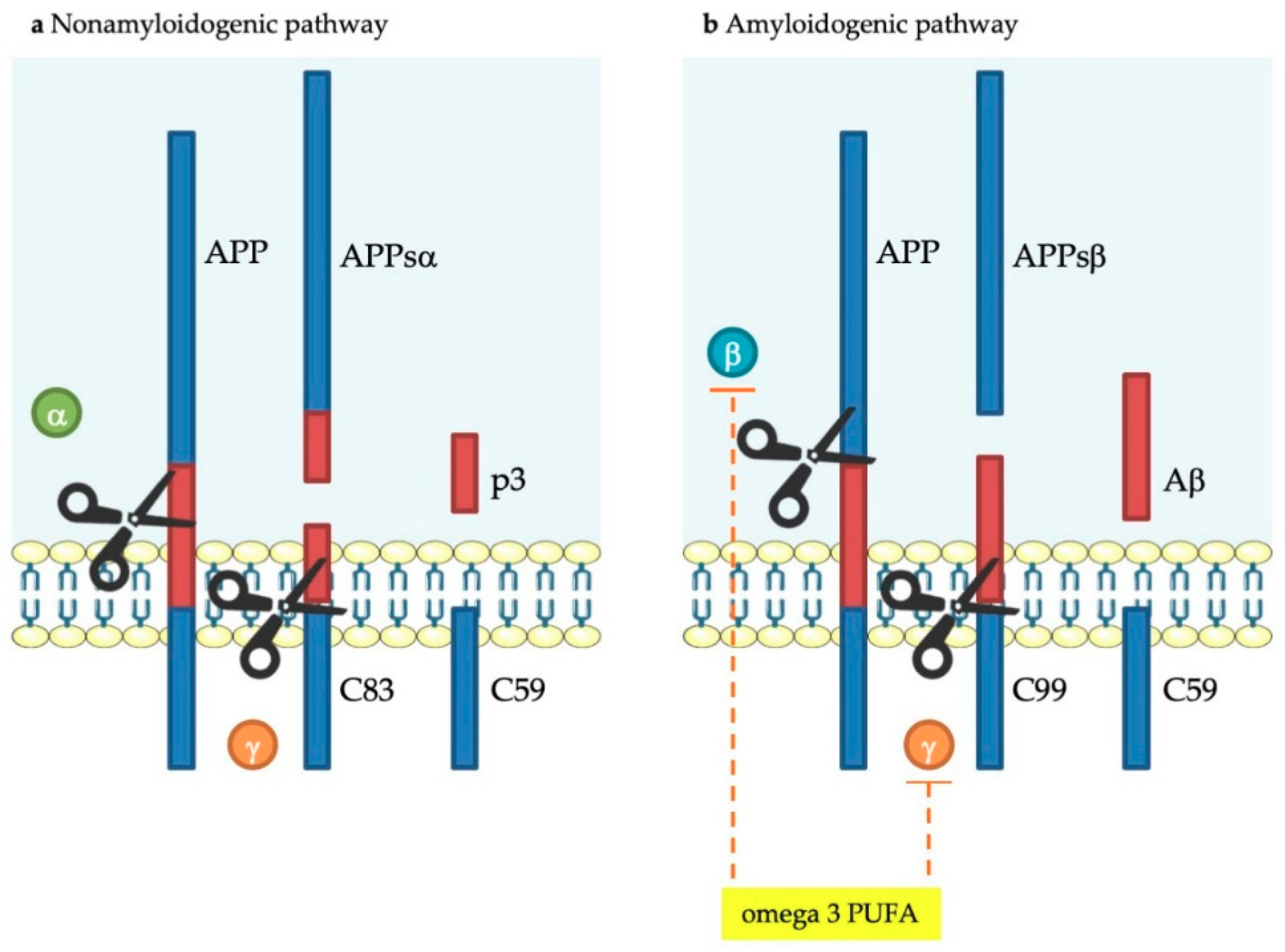
| Fatty Acids | |||
|---|---|---|---|
| Saturated | Unsaturated | ||
| MUFA | PUFA | ||
| omega-9 | omega-3 | omega-6 | |
| stearic acid 18:0 | oleic acid 18:1 | ALA 18:3 EPA 20:5 DHA 22:6 | LA 18:2 ARA 20:4 |
| Food | ALA g/Portion | EPA g/Portion | DHA g/Portion |
|---|---|---|---|
| Baked beans, canned, vegetarian | 0.07 | ||
| Black walnuts | 0.76 | ||
| Bread, whole wheat | 0.04 | ||
| Canola oil | 1.28 | ||
| Chia seeds | 5.06 | ||
| Chicken, breast, roasted, | 0.01 | 0.02 | |
| Cod, Pacific, cooked * | 0.04 | 0.10 | |
| Edamame, frozen, prepared | 0.28 | ||
| Egg, cooked | 0.03 | ||
| English walnuts | 2.57 | ||
| Flaxseed oil | 7.26 | ||
| Flaxseed, whole | 2.35 | ||
| Ground beef, 85% lean, cooked ** | 0.04 | ||
| Herring, Atlantic, cooked * | 0.77 | 0.94 | |
| Kidney beans, canned | 0.10 | ||
| Lobster, cooked * | 0.04 | 0.10 | 0.07 |
| Mackerel, Atlantic, cooked * | 0.43 | 0.59 | |
| Mayonnaise | 0.74 | ||
| Milk, low-fat (1%) | 0.01 | ||
| Oysters, eastern, wild, cooked | 0.14 | 0.30 | 0.23 |
| Refried beans, canned, vegetarian | 0.21 | ||
| Salmon, Atlantic, farmed, cooked | 0.59 | 1.24 | |
| Salmon, Atlantic, wild, cooked | 0.35 | 1.22 | |
| Salmon, pink, canned, drained * | 0.04 | 0.28 | 0.63 |
| Sardines, canned in tomato sauce, drained * | 0.45 | 0.74 | |
| Scallops, cooked * | 0.06 | 0.09 | |
| Sea bass, cooked * | 0.18 | 0.47 | |
| Shrimp, cooked * | 0.12 | 0.12 | |
| Soybean oil | 0.92 | ||
| Tilapia, cooked * | 0.04 | 0.11 | |
| Trout, rainbow, wild, cooked | 0.40 | 0.44 | |
| Tuna, light, canned in water, drained * | 0.02 | 0.17 | |
| Tuna, yellowfin, cooked * | 0.01 | 0.09 |
| Main Scales | Description |
|---|---|
| Activities of Daily Living ADCS-ADL, ADCS-IADL | It measures the functional ability to perform activities of daily life. ADL assess basic living skills such as bathing and eating, whereas Instrumental activities of daily living (IADL) measures more complex tasks such as using the telephone or preparing meals. A higher ADL or IADL score indicates a worsening functionality. |
| Alzheimer’s Disease Assessment Scale-Cognitive Subscale (ADAS-Cog) | It is a sensitive and reliable method for the assessment of cognitive function in dementia. It consists of a psychometric scale of 11 items, and scores range from 0 (no impairment) to 70 (very severe impairment). |
| Beck Depression Inventory (BDI) | It is a 21-question multiple-choice self-report inventory, one of the most widely used psychometric tests for measuring the severity of depression. |
| Brief Assessment Schedule Depression Cards (BASDEC) | It is a brief test for screening depression, requiring minimal training to administer. |
| Bristol’s Activities of Daily Living Scale (BADLS) | It is specifically designed for individuals with mild dementia living in the community for completion by caregivers. |
| Clinical Dementia Rating (CDR) | It is a global measure that assesses memory, orientation, judgment, and other features. Is based on caregiver interview. Classifies dementia into questionable, mild, moderate, and severe. |
| Clinical Global Impression Scale (CGI) | It measures symptom severity, treatment response, and the efficacy of treatments in treatment studies of patients with mental disorders. |
| Clinician Interview-Based Impression of Change, plus carer interview (CIBIC-Plus) | It is a global measure capable of detecting changes in cognition, functionality, and behavior, thus assessing dementia’s severity and progression. Requires separate interviews with patients and caregivers. |
| Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV) | It is the handbook used by health care professionals in the United States and much of the world as the authoritative guide to the diagnosis of mental disorders. |
| Hamilton Depression Rating Scale (HDRS) | Is the most widely used clinician-administered depression assessment scale. The original version contains 17 items pertaining to symptoms of depression experienced over the past week. |
| Hoehn and Yahr scale | It is a commonly used system for describing how the symptoms of PD progress. |
| Hopkins Verbal Learning Test–Revised (HVLT-R) | It is a brief verbal learning and memory test ideal in situations calling for repeated neuropsychological examinations. |
| Mini-Mental State Examination (MMSE) | It evaluates cognitive function in the areas of orientation, memory, attention, calculation, language, and visual construction. It is widely translated and used in clinical practice. Patients score between 0 and 30 points, and cutoffs of 23/24 have typically been used to show significant cognitive impairment. |
| Montgomery–Åsberg Depression Rating Scale (MADRS) | It is a ten-item diagnostic questionnaire which psychiatrists use to measure the severity of depressive episodes in patients with mood disorders. |
| Neuropsychiatric Inventory (NPI) | It assesses dementia-related behavioral symptoms. The NPI originally examined 10 sub-domains of behavioral functioning: Delusions, hallucinations, agitation/aggression, dysphoria, anxiety, euphoria, apathy, disinhibition, irritability/lability, and aberrant motor activity. |
| Neuropsychological test battery (NTB) | This scale assesses changes in cognitive function and is seen as a promising method for mild AD. NTB has shown to be able to detect changes in memory performance. |
| Unified Parkinson’s Disease Rating Scale (UPDRS) | It evaluates motor impairment and disability of patients with PD. |
| N° Patients | Population Characteristic | Type and Dose Supplementation | Exposure Period | Results | References |
|---|---|---|---|---|---|
| 8006 | PD Honolulu-Asia Aging Study | Food frequency questionnaire | 30 years | Omega-3 PUFAs appeared protective. | [48] |
| 5289 | PD Rotterdam Study The Netherlands | Semiquantitative food frequency questionnaire | 6 years | Intakes of omega-3 PUFAs were significantly associated with a lower risk of PD. | [49] |
| 131,368 | PD Health Professionals Follow-Up Study and the Nurses’ Health Study USA | Semiquantitative food frequency questionnaire | 16 years | High intakes of fruit, vegetables, whole grains, legumes, poultry, and fish were associated with a lower risk of PD. | [50] |
| 249 | PD Japan | Self-administered diet history questionnaire | 6 years | Consumption of omega-3 PUFA, ALA, EPA, DHA was not associated with PD. | [51] |
| N° Patients | Population Characteristics | Type and Dose Supplementation | Exposure Period | Results | References |
|---|---|---|---|---|---|
| 24 | PD Italy | 800 mg/die DHA + 290 mg/die EPA from fish oil Placebo: Corn oil | 6 months | Treatment had no statistically significant effect on the rate of change on either UPDRS or Hoehn-Yahr Scale score. In DHA-treated patients, the HDRS score was reduced by at least 50%. | [52] |
| 31 | PD and Major Depression (DSM-IV) Brazil | 480 mg/die DHA + 720 mg/die EPA from fish oil + tocopherol Placebo: Mineral oil | 3 months | Treatment had no statistically significant effect on the rate of change on Hoehn-Yahr Scale score, but there was a significant decrease in MADRS and CGI scores. | [44] |
| 60 | PD Iran | 1000 mg omega-3 fatty acids from flaxseed oil + 400 IU vitamin E Placebo: Not specified | 3 months | Treatment had favorable effects on UPDRS score. | [53] |
| N° Patients | Population Characteristic | Type and Dose Supplementation | Exposure Period | Results | References |
|---|---|---|---|---|---|
| 5386 | AD 37 Rotterdam Study The Netherlands | Semiquantitative food frequency questionnaire | 2.1 years | Fish consumption, an important source of omega-3 PUFA, was inversely related to incident dementia, in particular to Alzheimer’s disease. | [57] |
| 815 | AD 131 Chicago Health and Aging Project USA | Food frequency questionnaire | 3.9 years | Dietary intake of omega-3 PUFA and weakly consumption of fish may reduce the risk of Alzheimer’s disease. | [58] |
| 2233 | AD 190 Cardiovascular Health Cognition Study (CHCS) USA | Food frequency questionnaire | 5.4 years | Consumption of fatty fish more than twice per week was associated with a reduction in the risk of Alzheimer’s disease by 41%. | [59] |
| 488 | AD not reported The Framingham Heart Study USA | Semiquantitative food frequency questionnaire | 9.1 years | Plasma DHA level was associated with a significant 47% reduction in the risk of developing all-cause dementia. | [60] |
| 8085 | AD 183 Three-City cohort study France | Food frequency questionnaire | 3.48 years | Frequent consumption of fruits and vegetables, fish, and omega-3 rich oils may decrease the risk of dementia and Alzheimer’s disease, especially among ApoE ε4 noncarriers. | [61] |
| 5395 | Rotterdam Study The Netherlands | Semiquantitative food frequency questionnaire | 9.6 years | In the cohort with moderate consumption of fish and omega-3 PUFAs these dietary factors did not appear to be associated with long-term dementia risk | [62] |
| 923 | AD Rush Memory and Aging Project USA | Semiquantitative food frequency questionnaire | 4.5 years | High adherence to all three diets may reduce AD risk. | [63] |
| N° Patients | Population Characteristic | Type and Dose Supplementation | Exposure Period | Results | References |
|---|---|---|---|---|---|
| 204 | AD (DSM-IV) MMSE 15–30 OmegAD Study | 1720 mg/die DHA+ 600 mg/die EPA Placebo: 4000 mg corn oil Both groups: + 16 mg/die vitamin E | 12 months | There was no significant statistical difference after 6- or 12-month treatment between groups in MMSE, ADAS-cog, CDR. A subgroup with very mild cognitive dysfunction showed a reduction in decline rate. | [64] |
| 204 | AD (DSM-IV) MMSE 15–30 OmegAD Study | 1720 mg/die DHA+ 600 mg/die EPA Placebo: 4000 mg corn oil Both groups: + 16 mg vitamin E | 12 months | Supplementation with omega-3 did not result in marked effects on neuropsychiatric symptoms except for possible positive effects on depressive symptoms (assessed by MADRS) in non-APOEε4 carriers and agitation symptoms (assessed by NPI) in APOEε4 carriers. | [65] |
| 46 | AD AD (DSM-IV) MMSE 10–26 CDR-score 1–2 | 720 mg/die DHA+ 1080 mg/die EPA Placebo: Olive oil Both groups: + 1.2 mg hydroquinone + 12 mg tocopherols | 6 months | The treated group did not show an improvement in cognitive symptoms measured by MMSE, ADAS-cog, HDR but a relative improvement in CIBIC-plus score. In a subgroup with subjects with mild cognitive impairment (MMSE >27 e CDR 0.5–1) there was an improvement in ADAS-cog. | [66] |
| 402 | AD MMSE 14–26 Alzheimer’s Disease Cooperative Study (ADCS) DHA Supplementation Trial USA | 2000 mg/die DHA from seaweed Placebo: Corn or soy oil | 18 months | Supplementation with DHA compared with placebo did not slow the rate of cognitive and functional decline in patients with mild to moderate Alzheimer’s disease assessed by MMSE, ADAS-cog, CDR, ADS-ADL, NPI. | [67] |
| 225 | AD Souvenir I Study | 1700 mg/die DHA+ 600 mg/die EPA (Souvenaid) Placebo: Control drink | 6 months | Supplementation with omega -3 improved delayed verbal recall. However, ADAS-cog, CIBIC-plus, NPI, ADCS-ADL, ADSC-ADL were unchanged. | [68] |
| 225 | AD Souvenir I Study | 1700 mg/die DHA+ 600 mg/die EPA (Souvenaid) Placebo: Control drink | 6 months | Souvenaid had a positive result on ADAS-cog outcome. A higher intake of Souvenaid was also associated with greater cognitive benefit. | [69] |
| 238 | AD Souvenir II Study | 1200 mg/die DHA+ 300 mg/die EPA (Souvenaid) Placebo: Control products | 6 months | In the active group, the NTB memory domain increased. | [70] |
| 527 | AD MMSE 14 – 24 Connect Study | 1200 mg/die DHA+ 300 mg/die EPA (Souvenaid) Placebo: Control products | 6 months | Cognitive performance, as assessed by ADAS-cog, showed a decline over time in both control and active study groups, with no significant difference between study groups. Add-on intake of Souvenaid did not slow cognitive decline in persons treated for mild-to-moderate AD. | [71] |
| 174 | AD mild to moderate OmegAD Study | 1720 mg/die DHA+ 600 mg /die EPA Placebo: 4000 mg corn oil | 12 months | Plasma transthyretin positively correlated with MMSE and inversely with ADAS-Cog, suggesting a potential mechanism for probable positive effects of omega-3 on cognition. | [72] |
| 39 | AD MMSE 15–26 CDR 0.5–1.0 Not depressed (CESD <4.0) | 675 mg/die DHA+ 975 mg /die EPA Group omega-3 plus alpha lipoic acid (LA): 675 mg/die DHA+ 975 mg/die EPA+ 600 mg/die LA Placebo: Soy oil | 12 months | Active groups were no different from the placebo group in ADAS-cog, ADL. Omega-3 + LA group showed less decline assessed by MMSE. IADL differences between placebo e omega-3 and between placebo e omega-3 + LA groups were observed. | [73] |
| 179 | AD mild Souvenir II Study | 1700 mg/die DHA+ 6 mg/die EPA (Souvenaid) Placebo: Control drink | 6 months | The administration contributed to the maintenance of the organization of brain networks in mild AD patients. | [74] |
| 19 | AD MMSE 16–30 | 625 mg/die DHA+ 600 mg/die EPA Placebo: Olive oil Both groups: + 20 mg mixed tocopherols | 4 months | The daily supplementation was associated with none or only negligible benefits on mood and cognition, assessed by MMSE, HVLT-R, BASDEC, BADLS. | [75] |
| 204 | AD OmegAD Study | 1720 mg/die DHA+ 600 mg/die EPA Placebo: 4000 mg corn oil Both groups: + 16 mg vitamin E | 6 months | The daily supplementation stabilized the cognitive performance of AD subjects, assessed by ADAS-cog and MMSE scores. | [76] |
| 204 | AD OmegAD Study | 1720 mg/die DHA+ 600 mg/die EPA Placebo: 4000 mg corn oil Both groups: + 16 mg vitamin E | 6 months | A decrease was observed in RvD1 and LXA4 production from peripheral blood mononuclear cells of AD patients who did not receive omega-3 but not in cells of AD subjects under omega-3 intake. | [77] |
| 201 | AD Open label extension study (OLE) Souvenir II MMSE ≥ 20 | 1200 mg/die DHA+ 300 mg/die EPA (Souvenaid) Placebo: Control drink | 6 months | The intake of Souvenaid was well tolerated with a favorable safety profile. The adherence to Souvenaid was very high reflecting its good tolerability and ease of use. | [78] |
| 171 | AD OmegAD Study | 1720 mg/die DHA+ 600 mg/die EPA Placebo: 1 g corn oil Both groups: + 16 mg vitamin E | 6 months | The effect of omega-3 supplementation on MMSE and CDR appeared to be influenced by homocysteine plasma levels. | [79] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Avallone, R.; Vitale, G.; Bertolotti, M. Omega-3 Fatty Acids and Neurodegenerative Diseases: New Evidence in Clinical Trials. Int. J. Mol. Sci. 2019, 20, 4256. https://doi.org/10.3390/ijms20174256
Avallone R, Vitale G, Bertolotti M. Omega-3 Fatty Acids and Neurodegenerative Diseases: New Evidence in Clinical Trials. International Journal of Molecular Sciences. 2019; 20(17):4256. https://doi.org/10.3390/ijms20174256
Chicago/Turabian StyleAvallone, Rossella, Giovanni Vitale, and Marco Bertolotti. 2019. "Omega-3 Fatty Acids and Neurodegenerative Diseases: New Evidence in Clinical Trials" International Journal of Molecular Sciences 20, no. 17: 4256. https://doi.org/10.3390/ijms20174256
APA StyleAvallone, R., Vitale, G., & Bertolotti, M. (2019). Omega-3 Fatty Acids and Neurodegenerative Diseases: New Evidence in Clinical Trials. International Journal of Molecular Sciences, 20(17), 4256. https://doi.org/10.3390/ijms20174256





