Mast Cell Responses to Viruses and Pathogen Products
Abstract
1. Introduction
2. Historical Studies of MC–Virus and MC–Pathogen Product Interactions
3. Mast Cells and Mosquito-Borne Viruses
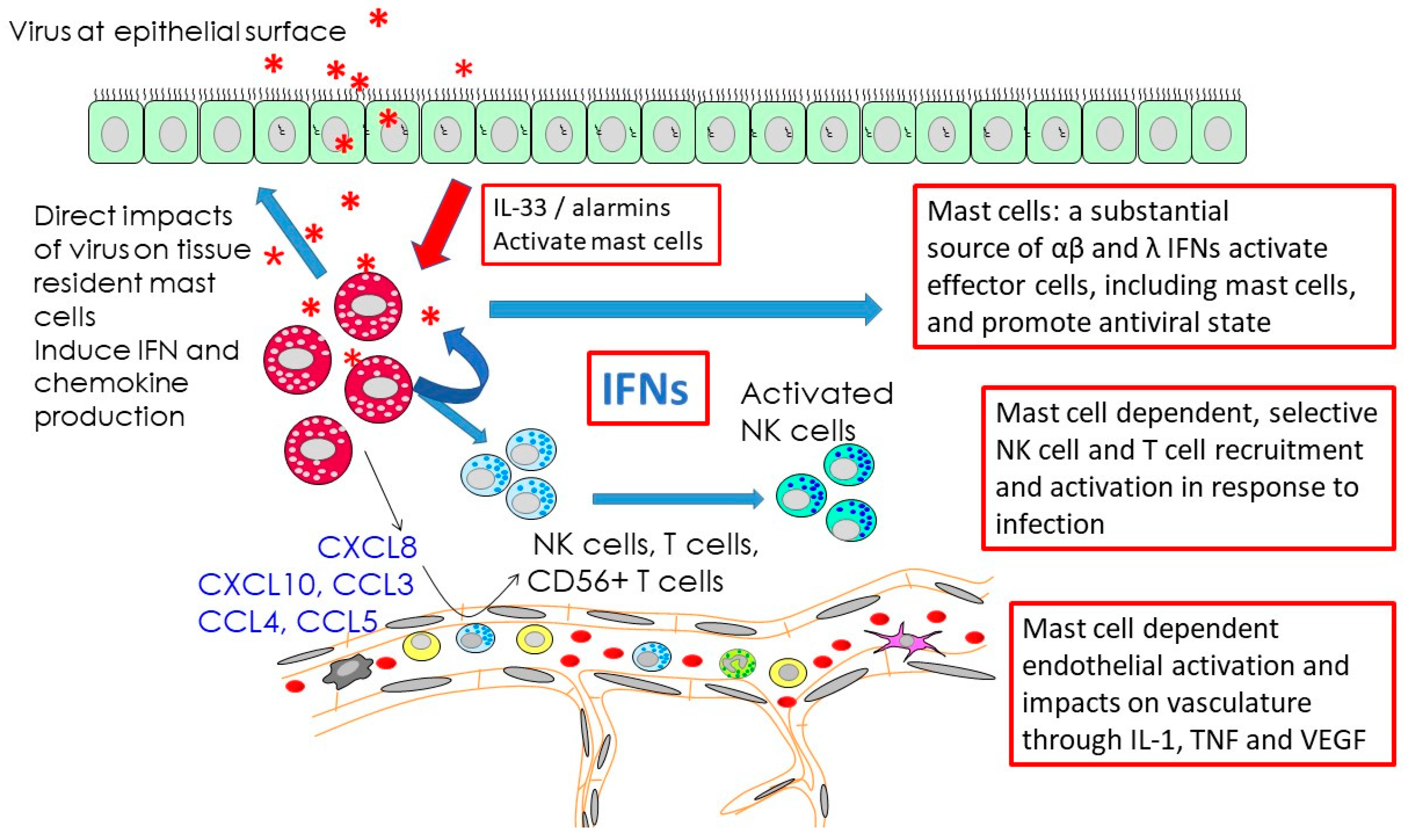
4. Responses to Respiratory Viruses
5. Mast Cell Responses to HIV
6. Mast Cell Responses to Hepatitis Viruses
7. Mast Cell Promotion of Effective Immunity and Response to Oncolytic Viruses
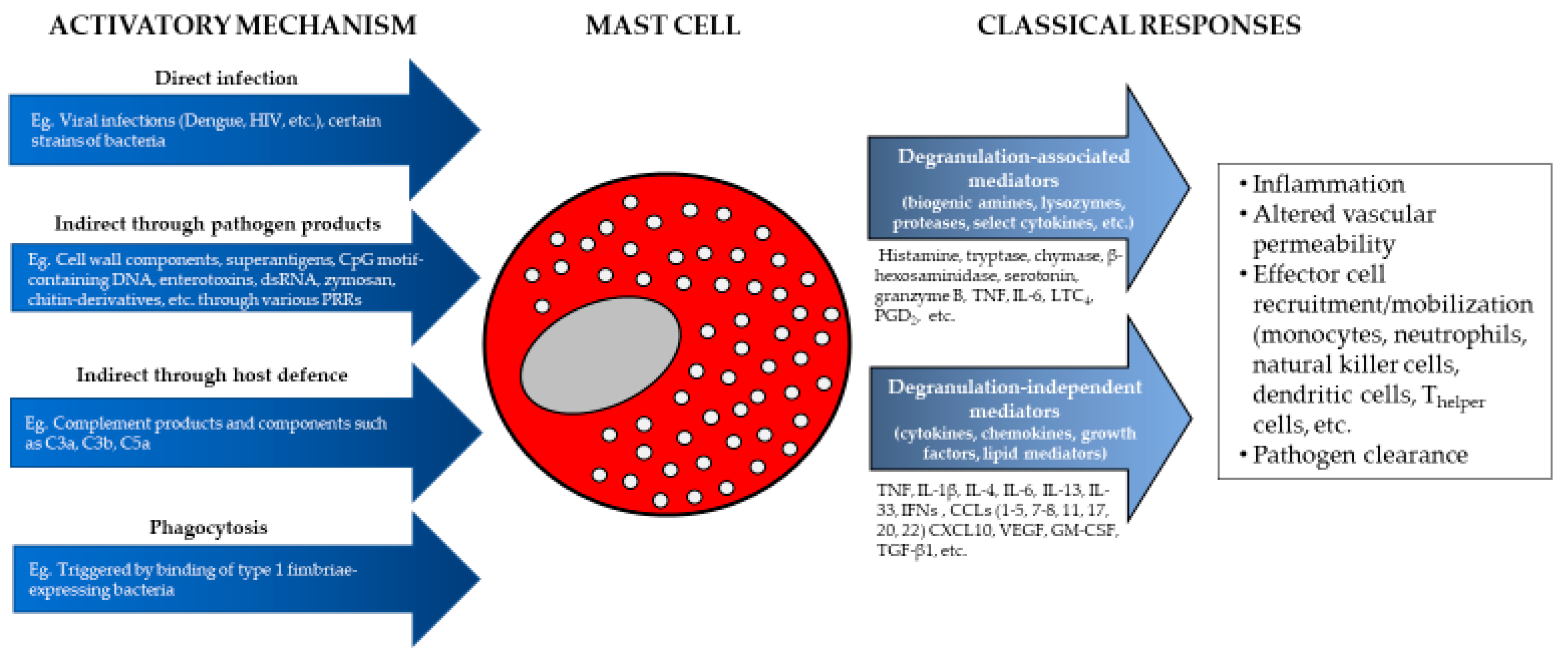
8. Pathogen Products that Activate Mast Cells
9. Bacterial Pathogens and Products
10. Viral Pathogen Products
11. Fungal Pathogens and Products
12. Conclusions
Funding
Conflicts of Interest
References
- Leal-Berumen, I.; Conlon, P.; Marshall, J.S. IL-6 production by rat peritoneal mast cells is not necessarily preceded by histamine release and can be induced by bacterial lipopolysaccharide. J. Immunol. 1994, 152, 5468–5476. [Google Scholar] [PubMed]
- Leal-Berumen, I.; O’Byrne, P.; Gupta, A.; Richards, C.D.; Marshall, J.S. Prostanoid enhancement of interleukin-6 production by rat peritoneal mast cells. J. Immunol. 1995, 154, 4759–4767. [Google Scholar] [PubMed]
- Sugiyama, K. Histamine release from rat mast cells induced by Sendai virus. Nature 1977, 270, 614–615. [Google Scholar] [CrossRef] [PubMed]
- Gomperts, B.D.; Baldwin, J.M.; Micklem, K.J. Rat mast cells permeabilized with Sendai virus secrete histamine in response to Ca2+ buffered in the micromolar range. Biochem. J. 1983, 210, 737–745. [Google Scholar] [CrossRef] [PubMed]
- Rein, A.; Keller, J.; Schultz, A.M.; Holmes, K.L.; Medicus, R.; Ihle, J.N. Infection of immune mast cells by Harvey sarcoma virus: Immortalization without loss of requirement for interleukin-3. Mol. Cell. Biol. 1985, 5, 2257–2264. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.W.; Wong, P.M.; Shen-Ong, G.; Ruscetti, S.; Ishizaka, T.; Eaves, C.J. Production of granulocyte-macrophage colony-stimulating factor by Abelson virus-induced tumorigenic mast cell lines. Blood 1986, 68, 1074–1081. [Google Scholar]
- Reynolds, D.S.; Serafin, W.E.; Faller, D.V.; Wall, D.A.; Abbas, A.K.; Dvorak, A.M.; Austen, K.F.; Stevens, R.L. Immortalization of murine connective tissue-type mast cells at multiple stages of their differentiation by coculture of splenocytes with fibroblasts that produce Kirsten sarcoma virus. J. Biol. Chem. 1988, 263, 12783–12791. [Google Scholar]
- Mokhtarian, F.; Griffin, D.E. The role of mast cells in virus-induced inflammation in the murine central nervous system. Cell. Immunol. 1984, 86, 491–500. [Google Scholar] [CrossRef]
- Ogunbiyi, P.O.; Black, W.D.; Eyre, P. Parainfluenza-3 virus-induced enhancement of histamine release from calf lung mast cells--effect of levamisole. J. Vet. Pharmacol. Ther. 1988, 11, 338–344. [Google Scholar] [CrossRef]
- Liu, H.; Tan, J.; Liu, J.; Feng, H.; Pan, D. Altered mast cell activity in response to rhinovirus infection provides novel insight into asthma. J. Asthma 2019, 18, 1–9. [Google Scholar] [CrossRef]
- Akoto, C.; Davies, D.E.; Swindle, E.J. Mast cells are permissive for rhinovirus replication: Potential implications for asthma exacerbations. Clin. Exp. Allergy 2017, 47, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Hosoda, M.; Yamaya, M.; Suzuki, T.; Yamada, N.; Kamanaka, M.; Sekizawa, K.; Butterfield, J.H.; Watanabe, T.; Nishimura, H.; Sasaki, H. Effects of rhinovirus infection on histamine and cytokine production by cell lines from human mast cells and basophils. J. Immunol. 2002, 169, 1482–1491. [Google Scholar] [CrossRef] [PubMed]
- Troupin, A.; Shirley, D.; Londono-Renteria, B.; Watson, A.M.; McHale, C.; Hall, A.; Hartstone-Rose, A.; Klimstra, W.B.; Gomez, G.; Colpitts, T.M. A Role for Human Skin Mast Cells in Dengue Virus Infection and Systemic Spread. J. Immunol. 2016, 197, 4382–4391. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.T.; Wan, S.W.; Lu, Y.T.; Yao, J.H.; Lin, C.F.; Hsu, L.J.; Brown, M.G.; Marshall, J.S.; Anderson, R.; Lin, Y.S. Autophagy facilitates antibody-enhanced dengue virus infection in human pre-basophil/mast cells. PLoS ONE 2014, 9, e110655. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.G.; McAlpine, S.M.; Huang, Y.Y.; Haidl, I.D.; Al-Afif, A.; Marshall, J.S.; Anderson, R. RNA sensors enable human mast cell anti-viral chemokine production and IFN-mediated protection in response to antibody-enhanced dengue virus infection. PLoS ONE 2012, 7, e34055. [Google Scholar] [CrossRef] [PubMed]
- Al-Afif, A.; Alyazidi, R.; Oldford, S.A.; Huang, Y.Y.; King, C.A.; Marr, N.; Haidl, I.D.; Anderson, R.; Marshall, J.S. Respiratory syncytial virus infection of primary human mast cells induces the selective production of type I interferons, CXCL10, and CCL4. J. Allergy Clin. Immunol. 2015, 136, 1346–1354. [Google Scholar] [CrossRef]
- Shirato, K.; Taguchi, F. Mast cell degranulation is induced by A549 airway epithelial cell infected with respiratory syncytial virus. Virology 2009, 386, 88–93. [Google Scholar] [CrossRef] [PubMed]
- McAlpine, S.M.; Issekutz, T.B.; Marshall, J.S. Virus stimulation of human mast cells results in the recruitment of CD56(+) T cells by a mechanism dependent on CCR5 ligands. FASEB J. 2012, 26, 1280–1289. [Google Scholar] [CrossRef]
- Portales-Cervantes, L.; Haidl, I.D.; Lee, P.W.; Marshall, J.S. Virus-Infected Human Mast Cells Enhance Natural Killer Cell Functions. J. Innate Immun. 2017, 9, 94–108. [Google Scholar] [CrossRef]
- Jiang, A.P.; Jiang, J.F.; Wei, J.F.; Guo, M.G.; Qin, Y.; Guo, Q.Q.; Ma, L.; Liu, B.C.; Wang, X.; Veazey, R.S.; et al. Human Mucosal Mast Cells Capture HIV-1 and Mediate Viral trans-Infection of CD4+ T Cells. J. Virol. 2015, 90, 2928–2937. [Google Scholar] [CrossRef]
- Qi, J.C.; Stevens, R.L.; Wadley, R.; Collins, A.; Cooley, M.; Naif, H.M.; Nasr, N.; Cunningham, A.; Katsoulotos, G.; Wanigasek, Y.; et al. IL-16 regulation of human mast cells/basophils and their susceptibility to HIV-1. J. Immunol. 2002, 168, 4127–4134. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Meng, D.; Wei, T.; Zhang, S.; Hu, Y.; Wang, M. Apoptosis and pro-inflammatory cytokine response of mast cells induced by influenza A viruses. PLoS ONE 2014, 9, e100109. [Google Scholar] [CrossRef] [PubMed]
- Graham, A.C.; Hilmer, K.M.; Zickovich, J.M.; Obar, J.J. Inflammatory response of mast cells during influenza A virus infection is mediated by active infection and RIG-I signaling. J. Immunol. 2013, 190, 4676–4684. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Jin, Y.; Han, D.; Zhang, G.; Cao, S.; Xie, J.; Xue, J.; Li, Y.; Meng, D.; Fan, X.; et al. Mast cell-induced lung injury in mice infected with H5N1 influenza virus. J. Virol. 2012, 86, 3347–3356. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.H.; Kim, H.S.; Seo, S.H. Porcine mast cells infected with H1N1 influenza virus release histamine and inflammatory cytokines and chemokines. Arch. Virol. 2017, 162, 1067–1071. [Google Scholar] [CrossRef] [PubMed]
- Zarnegar, B.; Mendez-Enriquez, E.; Westin, A.; Soderberg, C.; Dahlin, J.S.; Gronvik, K.O.; Hallgren, J. Influenza Infection in Mice Induces Accumulation of Lung Mast Cells through the Recruitment and Maturation of Mast Cell Progenitors. Front. Immunol. 2017, 8, 310. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Bao, L.; Li, F.; Lv, Q.; Yuan, J.; Xu, Y.; Deng, W.; Yao, Y.; Yu, P.; Qin, C. Intranasal immunization of mice with inactivated virus and mast cell activator C48/80 elicits protective immunity against influenza H1 but not H5. Immunol. Invest. 2014, 43, 224–235. [Google Scholar] [CrossRef] [PubMed]
- Meng, S.; Liu, Z.; Xu, L.; Li, L.; Mei, S.; Bao, L.; Deng, W.; Li, L.; Lei, R.; Xie, L.; et al. Intranasal immunization with recombinant HA and mast cell activator C48/80 elicits protective immunity against 2009 pandemic H1N1 influenza in mice. PLoS ONE 2011, 6, e19863. [Google Scholar] [CrossRef] [PubMed]
- Morrison, J.; Rathore, A.P.S.; Mantri, C.K.; Aman, S.A.B.; Nishida, A.; St John, A.L. Transcriptional Profiling Confirms the Therapeutic Effects of Mast Cell Stabilization in a Dengue Disease Model. J. Virol. 2017, 91. [Google Scholar] [CrossRef]
- Chu, Y.T.; Wan, S.W.; Chang, Y.C.; Lee, C.K.; Wu-Hsieh, B.A.; Anderson, R.; Lin, Y.S. Antibodies against nonstructural protein 1 protect mice from dengue virus-induced mast cell activation. Lab. Invest. 2017. [Google Scholar] [CrossRef]
- Chu, Y.T.; Wan, S.W.; Anderson, R.; Lin, Y.S. Mast cell-macrophage dynamics in modulation of dengue virus infection in skin. Immunology 2015, 146, 163–172. [Google Scholar] [CrossRef] [PubMed]
- St John, A.L.; Rathore, A.P.; Raghavan, B.; Ng, M.L.; Abraham, S.N. Contributions of mast cells and vasoactive products, leukotrienes and chymase, to dengue virus-induced vascular leakage. Elife 2013, 2, e00481. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; MacLeod, D.T.; Di Nardo, A. Commensal bacteria lipoteichoic acid increases skin mast cell antimicrobial activity against vaccinia viruses. J. Immunol. 2012, 189, 1551–1558. [Google Scholar] [CrossRef] [PubMed]
- Depinay, N.; Hacini, F.; Beghdadi, W.; Peronet, R.; Mecheri, S. Mast cell-dependent down-regulation of antigen-specific immune responses by mosquito bites. J. Immunol. 2006, 176, 4141–4146. [Google Scholar] [CrossRef] [PubMed]
- Demeure, C.E.; Brahimi, K.; Hacini, F.; Marchand, F.; Peronet, R.; Huerre, M.; St-Mezard, P.; Nicolas, J.F.; Brey, P.; Delespesse, G.; et al. Anopheles mosquito bites activate cutaneous mast cells leading to a local inflammatory response and lymph node hyperplasia. J. Immunol. 2005, 174, 3932–3940. [Google Scholar] [CrossRef] [PubMed]
- Bissonnette, E.Y.; Rossignol, P.A.; Befus, A.D. Extracts of mosquito salivary gland inhibit tumour necrosis factor alpha release from mast cells. Parasite Immunol. 1993, 15, 27–33. [Google Scholar] [CrossRef] [PubMed]
- King, C.A.; Marshall, J.S.; Alshurafa, H.; Anderson, R. Release of vasoactive cytokines by antibody-enhanced dengue virus infection of a human mast cell/basophil line. J. Virol. 2000, 74, 7146–7150. [Google Scholar] [CrossRef] [PubMed]
- King, C.A.; Anderson, R.; Marshall, J.S. Dengue virus selectively induces human mast cell chemokine production. J. Virol. 2002, 76, 8408–8419. [Google Scholar] [CrossRef]
- Brown, M.G.; Huang, Y.Y.; Marshall, J.S.; King, C.A.; Hoskin, D.W.; Anderson, R. Dramatic caspase-dependent apoptosis in antibody-enhanced dengue virus infection of human mast cells. J. Leukoc. Biol. 2009, 85, 71–80. [Google Scholar] [CrossRef]
- Brown, M.G.; King, C.A.; Sherren, C.; Marshall, J.S.; Anderson, R. A dominant role for FcgammaRII in antibody-enhanced dengue virus infection of human mast cells and associated CCL5 release. J. Leukoc. Biol. 2006, 80, 1242–1250. [Google Scholar] [CrossRef]
- Furuta, T.; Murao, L.A.; Lan, N.T.; Huy, N.T.; Huong, V.T.; Thuy, T.T.; Tham, V.D.; Nga, C.T.; Ha, T.T.; Ohmoto, Y.; et al. Association of mast cell-derived VEGF and proteases in Dengue shock syndrome. PLoS Negl. Trop. Dis. 2012, 6, e1505. [Google Scholar] [CrossRef] [PubMed]
- Syenina, A.; Jagaraj, C.J.; Aman, S.A.; Sridharan, A.; St John, A.L. Dengue vascular leakage is augmented by mast cell degranulation mediated by immunoglobulin Fcgamma receptors. Elife 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Tissera, H.; Rathore, A.P.S.; Leong, W.Y.; Pike, B.L.; Warkentien, T.E.; Farouk, F.S.; Syenina, A.; Eong Ooi, E.; Gubler, D.J.; Wilder-Smith, A.; et al. Chymase Level Is a Predictive Biomarker of Dengue Hemorrhagic Fever in Pediatric and Adult Patients. J. Infect. Dis. 2017, 216, 1112–1121. [Google Scholar] [CrossRef] [PubMed]
- Rathore, A.P.; Mantri, C.K.; Aman, S.A.; Syenina, A.; Ooi, J.; Jagaraj, C.J.; Goh, C.C.; Tissera, H.; Wilder-Smith, A.; Ng, L.G.; et al. Dengue virus-elicited tryptase induces endothelial permeability and shock. J. Clin. Invest. 2019, 130. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.G.; Hermann, L.L.; Issekutz, A.C.; Marshall, J.S.; Rowter, D.; Al-Afif, A.; Anderson, R. Dengue virus infection of mast cells triggers endothelial cell activation. J. Virol. 2011, 85, 1145–1150. [Google Scholar] [CrossRef] [PubMed]
- St John, A.L. Influence of mast cells on dengue protective immunity and immune pathology. PLoS Pathog. 2013, 9, e1003783. [Google Scholar] [CrossRef] [PubMed]
- Wan, S.W.; Wu-Hsieh, B.A.; Lin, Y.S.; Chen, W.Y.; Huang, Y.; Anderson, R. The monocyte-macrophage-mast cell axis in dengue pathogenesis. J. Biomed. Sci. 2018, 25, 77. [Google Scholar] [CrossRef]
- Guhl, S.; Franke, R.; Schielke, A.; Johne, R.; Kruger, D.H.; Babina, M.; Rang, A. Infection of in vivo differentiated human mast cells with hantaviruses. J. Gen. Virol. 2010, 91, 1256–1261. [Google Scholar] [CrossRef]
- Sager, G.; Gabaglio, S.; Sztul, E.; Belov, G.A. Role of Host Cell Secretory Machinery in Zika Virus Life Cycle. Viruses 2018, 10. [Google Scholar] [CrossRef]
- Kimman, T.G.; Terpstra, G.K.; Daha, M.R.; Westenbrink, F. Pathogenesis of naturally acquired bovine respiratory syncytial virus infection in calves: Evidence for the involvement of complement and mast cell mediators. Am. J. Vet. Res. 1989, 50, 694–700. [Google Scholar]
- Jolly, S.; Detilleux, J.; Desmecht, D. Extensive mast cell degranulation in bovine respiratory syncytial virus-associated paroxystic respiratory distress syndrome. Vet. Immunol. Immunopathol. 2004, 97, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Oymar, K.; Halvorsen, T.; Aksnes, L. Mast cell activation and leukotriene secretion in wheezing infants. Relation to respiratory syncytial virus and outcome. Pediatr. Allergy Immunol. 2006, 17, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Dakhama, A.; Lee, Y.M.; Ohnishi, H.; Jing, X.; Balhorn, A.; Takeda, K.; Gelfand, E.W. Virus-specific IgE enhances airway responsiveness on reinfection with respiratory syncytial virus in newborn mice. J. Allergy Clin. Immunol. 2009, 123, 138–145. [Google Scholar] [CrossRef]
- Graziano, F.M.; Tilton, R.; Hirth, T.; Segaloff, D.; Mullins, T.; Dick, E.; Buckner, C.K.; Busse, W.W. The effect of parainfluenza 3 infection on guinea pig basophil and lung mast cell histamine release. Am. Rev. Respir. Dis. 1989, 139, 715–720. [Google Scholar] [CrossRef] [PubMed]
- Sorden, S.D.; Castleman, W.L. Brown Norway rats are high responders to bronchiolitis, pneumonia, and bronchiolar mastocytosis induced by parainfluenza virus. Exp. Lung Res. 1991, 17, 1025–1045. [Google Scholar] [CrossRef] [PubMed]
- Uhl, E.W.; Castleman, W.L.; Sorkness, R.L.; Busse, W.W.; Lemanske, R.F., Jr.; McAllister, P.K. Parainfluenza virus-induced persistence of airway inflammation, fibrosis, and dysfunction associated with TGF-beta 1 expression in brown Norway rats. Am. J. Respir. Crit. Care. Med. 1996, 154, 1834–1842. [Google Scholar] [CrossRef] [PubMed]
- Marcet, C.W.; St Laurent, C.D.; Moon, T.C.; Singh, N.; Befus, A.D. Limited replication of influenza A virus in human mast cells. Immunol. Res. 2013, 56, 32–43. [Google Scholar] [CrossRef]
- Naclerio, R.M.; Proud, D.; Kagey-Sobotka, A.; Lichtenstein, L.M.; Hendley, J.O.; Gwaltney, J.M., Jr. Is histamine responsible for the symptoms of rhinovirus colds? A look at the inflammatory mediators following infection. Pediatr. Infect. Dis. J. 1988, 7, 218–222. [Google Scholar] [CrossRef]
- Bannert, N.; Farzan, M.; Friend, D.S.; Ochi, H.; Price, K.S.; Sodroski, J.; Boyce, J.A. Human Mast cell progenitors can be infected by macrophagetropic human immunodeficiency virus type 1 and retain virus with maturation in vitro. J. Virol. 2001, 75, 10808–10814. [Google Scholar] [CrossRef]
- Li, Y.; Li, L.; Wadley, R.; Reddel, S.W.; Qi, J.C.; Archis, C.; Collins, A.; Clark, E.; Cooley, M.; Kouts, S.; et al. Mast cells/basophils in the peripheral blood of allergic individuals who are HIV-1 susceptible due to their surface expression of CD4 and the chemokine receptors CCR3, CCR5, and CXCR4. Blood 2001, 97, 3484–3490. [Google Scholar] [CrossRef]
- Sundstrom, J.B.; Little, D.M.; Villinger, F.; Ellis, J.E.; Ansari, A.A. Signaling through Toll-like receptors triggers HIV-1 replication in latently infected mast cells. J. Immunol. 2004, 172, 4391–4401. [Google Scholar] [CrossRef] [PubMed]
- Sundstrom, J.B.; Ellis, J.E.; Hair, G.A.; Kirshenbaum, A.S.; Metcalfe, D.D.; Yi, H.; Cardona, A.C.; Lindsay, M.K.; Ansari, A.A. Human tissue mast cells are an inducible reservoir of persistent HIV infection. Blood 2007, 109, 5293–5300. [Google Scholar] [CrossRef] [PubMed]
- Nelson, A.M.; Auerbach, A.; Man, Y.G. Failure to detect active virus replication in mast cells at various tissue sites of HIV patients by immunohistochemistry. Int. J. Biol. Sci. 2009, 5, 603–610. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rossi, F.W.; Prevete, N.; Rivellese, F.; Lobasso, A.; Napolitano, F.; Granata, F.; Selleri, C.; de Paulis, A. HIV-1 Nef promotes migration and chemokine synthesis of human basophils and mast cells through the interaction with CXCR4. Clin. Mol. Allergy 2016, 14, 15. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Franceschini, B.; Russo, C.; Dioguardi, N.; Grizzi, F. Increased liver mast cell recruitment in patients with chronic C virus-related hepatitis and histologically documented steatosis. J. Viral Hepat. 2007, 14, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.; Zhen, S.; Yu, Q.; Gong, Z. HCV-E2 inhibits hepatocellular carcinoma metastasis by stimulating mast cells to secrete exosomal shuttle microRNAs. Oncol. Lett. 2017, 14, 2141–2146. [Google Scholar] [CrossRef] [PubMed]
- Patella, V.; Bouvet, J.P.; Marone, G. Protein Fv produced during vital hepatitis is a novel activator of human basophils and mast cells. J. Immunol. 1993, 151, 5685–5698. [Google Scholar] [PubMed]
- Genovese, A.; Borgia, G.; Bouvet, J.P.; Detoraki, A.; de Paulis, A.; Piazza, M.; Marone, G. Protein Fv produced during viral hepatitis is an endogenous immunoglobulin superantigen activating human heart mast cells. Int. Arch. Allergy Immunol. 2003, 132, 336–345. [Google Scholar] [CrossRef]
- Aoki, R.; Kawamura, T.; Goshima, F.; Ogawa, Y.; Nakae, S.; Nakao, A.; Moriishi, K.; Nishiyama, Y.; Shimada, S. Mast cells play a key role in host defense against herpes simplex virus infection through TNF-alpha and IL-6 production. J. Invest. Derm. 2013, 133, 2170–2179. [Google Scholar] [CrossRef]
- Royer, D.J.; Zheng, M.; Conrady, C.D.; Carr, D.J. Granulocytes in Ocular HSV-1 Infection: Opposing Roles of Mast Cells and Neutrophils. Invest. Ophthalmol. Vis. Sci. 2015, 56, 3763–3775. [Google Scholar] [CrossRef]
- Domenico, J.; Lucas, J.J.; Fujita, M.; Gelfand, E.W. Susceptibility to vaccinia virus infection and spread in mice is determined by age at infection, allergen sensitization and mast cell status. Int. Arch. Allergy Immunol. 2012, 158, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lai, Y.; Bernard, J.J.; Macleod, D.T.; Cogen, A.L.; Moss, B.; Di Nardo, A. Skin mast cells protect mice against vaccinia virus by triggering mast cell receptor S1PR2 and releasing antimicrobial peptides. J. Immunol. 2012, 188, 345–357. [Google Scholar] [CrossRef] [PubMed]
- Burke, S.M.; Issekutz, T.B.; Mohan, K.; Lee, P.W.; Shmulevitz, M.; Marshall, J.S. Human mast cell activation with virus-associated stimuli leads to the selective chemotaxis of natural killer cells by a CXCL8-dependent mechanism. Blood 2008, 111, 5467–5476. [Google Scholar] [CrossRef] [PubMed]
- Oldford, S.A.; Salsman, S.P.; Portales-Cervantes, L.; Alyazidi, R.; Anderson, R.; Haidl, I.D.; Marshall, J.S. Interferon alpha2 and interferon gamma induce the degranulation independent production of VEGF-A and IL-1 receptor antagonist and other mediators from human mast cells. Immun. Inflamm. Dis. 2018, 6, 176–189. [Google Scholar] [CrossRef] [PubMed]
- Enoksson, M.; Lyberg, K.; Moller-Westerberg, C.; Fallon, P.G.; Nilsson, G.; Lunderius-Andersson, C. Mast cells as sensors of cell injury through IL-33 recognition. J. Immunol. 2011, 186, 2523–2528. [Google Scholar] [CrossRef] [PubMed]
- Supajatura, V.; Ushio, H.; Nakao, A.; Akira, S.; Okumura, K.; Ra, C.; Ogawa, H. Differential responses of mast cell Toll-like receptors 2 and 4 in allergy and innate immunity. J. Clin. Invest. 2002, 109, 1351–1359. [Google Scholar] [CrossRef] [PubMed]
- McCurdy, J.D.; Lin, T.J.; Marshall, J.S. Toll-like receptor 4-mediated activation of murine mast cells. J. Leukoc. Biol. 2001, 70, 977–984. [Google Scholar]
- Matsushima, H.; Yamada, N.; Matsue, H.; Shimada, S. TLR3-, TLR7-, and TLR9-mediated production of proinflammatory cytokines and chemokines from murine connective tissue type skin-derived mast cells but not from bone marrow-derived mast cells. J. Immunol. 2004, 173, 531–541. [Google Scholar] [CrossRef]
- Zhu, F.G.; Marshall, J.S. CpG-containing oligodeoxynucleotides induce TNF-alpha and IL-6 production but not degranulation from murine bone marrow-derived mast cells. J. Leukoc. Biol. 2001, 69, 253–262. [Google Scholar]
- Malaviya, R.; Gao, Z.; Thankavel, K.; van der Merwe, P.A.; Abraham, S.N. The mast cell tumor necrosis factor alpha response to FimH-expressing Escherichia coli is mediated by the glycosylphosphatidylinositol-anchored molecule CD48. Proc. Natl. Acad. Sci. USA 1999, 96, 8110–8115. [Google Scholar] [CrossRef]
- Genovese, A.; Bouvet, J.P.; Florio, G.; Lamparter-Schummert, B.; Bjorck, L.; Marone, G. Bacterial immunoglobulin superantigen proteins A and L activate human heart mast cells by interacting with immunoglobulin E. Infect. Immun. 2000, 68, 5517–5524. [Google Scholar] [CrossRef] [PubMed]
- Komisar, J.; Rivera, J.; Vega, A.; Tseng, J. Effects of staphylococcal enterotoxin B on rodent mast cells. Infect. Immun. 1992, 60, 2969–2975. [Google Scholar] [PubMed]
- Ono, H.K.; Nishizawa, M.; Yamamoto, Y.; Hu, D.L.; Nakane, A.; Shinagawa, K.; Omoe, K. Submucosal mast cells in the gastrointestinal tract are a target of staphylococcal enterotoxin type A. FEMS Immunol. Med. Microbiol. 2012, 64, 392–402. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Kitano, T.; Nishiyama, S.; Sanjo, H.; Onozaki, K.; Taki, S.; Itoh, S.; Hida, S. Staphylococcal superantigen-like 12 activates murine bone marrow derived mast cells. Biochem. Biophys. Res. Commun. 2019, 511, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Arcidiacono, D.; Odom, S.; Frossi, B.; Rivera, J.; Paccani, S.R.; Baldari, C.T.; Pucillo, C.; Montecucco, C.; de Bernard, M. The Vibrio cholerae cytolysin promotes activation of mast cell (T helper 2) cytokine production. Cell. Microbiol. 2008, 10, 899–907. [Google Scholar] [CrossRef] [PubMed]
- Mielcarek, N.; Hornquist, E.H.; Johansson, B.R.; Locht, C.; Abraham, S.N.; Holmgren, J. Interaction of Bordetella pertussis with mast cells, modulation of cytokine secretion by pertussis toxin. Cell. Microbiol. 2001, 3, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Wershil, B.K.; Castagliuolo, I.; Pothoulakis, C. Direct evidence of mast cell involvement in Clostridium difficile toxin A-induced enteritis in mice. Gastroenterology 1998, 114, 956–964. [Google Scholar] [CrossRef]
- Garcia-Rodriguez, K.M.; Goenka, A.; Alonso-Rasgado, M.T.; Hernandez-Pando, R.; Bulfone-Paus, S. The Role of Mast Cells in Tuberculosis: Orchestrating Innate Immune Crosstalk? Front. Immunol. 2017, 8, 1290. [Google Scholar] [CrossRef] [PubMed]
- Munoz, S.; Hernandez-Pando, R.; Abraham, S.N.; Enciso, J.A. Mast cell activation by Mycobacterium tuberculosis: Mediator release and role of CD48. J. Immunol. 2003, 170, 5590–5596. [Google Scholar] [CrossRef] [PubMed]
- Kulka, M.; Alexopoulou, L.; Flavell, R.A.; Metcalfe, D.D. Activation of mast cells by double-stranded RNA: Evidence for activation through Toll-like receptor 3. J. Allergy Clin. Immunol. 2004, 114, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Lund, J.M.; Alexopoulou, L.; Sato, A.; Karow, M.; Adams, N.C.; Gale, N.W.; Iwasaki, A.; Flavell, R.A. Recognition of single-stranded RNA viruses by Toll-like receptor 7. Proc. Natl. Acad Sci. USA 2004, 101, 5598–5603. [Google Scholar] [CrossRef] [PubMed]
- Delale, T.; Paquin, A.; Asselin-Paturel, C.; Dalod, M.; Brizard, G.; Bates, E.E.; Kastner, P.; Chan, S.; Akira, S.; Vicari, A.; et al. MyD88-dependent and -independent murine cytomegalovirus sensing for IFN-alpha release and initiation of immune responses in vivo. J. Immunol. 2005, 175, 6723–6732. [Google Scholar] [CrossRef] [PubMed]
- Tabeta, K.; Georgel, P.; Janssen, E.; Du, X.; Hoebe, K.; Crozat, K.; Mudd, S.; Shamel, L.; Sovath, S.; Goode, J.; et al. Toll-like receptors 9 and 3 as essential components of innate immune defense against mouse cytomegalovirus infection. Proc. Natl. Acad. Sci. USA 2004, 101, 3516–3521. [Google Scholar] [CrossRef] [PubMed]
- Sato, A.; Linehan, M.M.; Iwasaki, A. Dual recognition of herpes simplex viruses by TLR2 and TLR9 in dendritic cells. Proc. Natl. Acad. Sci. USA 2006, 103, 17343–17348. [Google Scholar] [CrossRef] [PubMed]
- St John, A.L.; Rathore, A.P.; Yap, H.; Ng, M.L.; Metcalfe, D.D.; Vasudevan, S.G.; Abraham, S.N. Immune surveillance by mast cells during dengue infection promotes natural killer (NK) and NKT-cell recruitment and viral clearance. Proc. Natl. Acad. Sci. USA 2011, 108, 9190–9195. [Google Scholar] [CrossRef]
- Imlach, W.; McCaughan, C.A.; Mercer, A.A.; Haig, D.; Fleming, S.B. Orf virus-encoded interleukin-10 stimulates the proliferation of murine mast cells and inhibits cytokine synthesis in murine peritoneal macrophages. J. Gen. Virol. 2002, 83, 1049–1058. [Google Scholar] [CrossRef] [PubMed]
- Patella, V.; Florio, G.; Petraroli, A.; Marone, G. HIV-1 gp120 induces IL-4 and IL-13 release from human Fc epsilon RI+ cells through interaction with the VH3 region of IgE. J. Immunol. 2000, 164, 589–595. [Google Scholar] [CrossRef]
- McCurdy, J.D.; Olynych, T.J.; Maher, L.H.; Marshall, J.S. Cutting edge: Distinct Toll-like receptor 2 activators selectively induce different classes of mediator production from human mast cells. J. Immunol. 2003, 170, 1625–1629. [Google Scholar] [CrossRef]
- Elieh Ali Komi, D.; Sharma, L.; Dela Cruz, C.S. Chitin and Its Effects on Inflammatory and Immune Responses. Clin. Rev. Allergy Immunol. 2018, 54, 213–223. [Google Scholar] [CrossRef]
- Lenardon, M.D.; Munro, C.A.; Gow, N.A. Chitin synthesis and fungal pathogenesis. Curr. Opin. Microbiol. 2010, 13, 416–423. [Google Scholar] [CrossRef]
- Upadhya, R.; Baker, L.G.; Lam, W.C.; Specht, C.A.; Donlin, M.J.; Lodge, J.K. Cryptococcus neoformans Cda1 and Its Chitin Deacetylase Activity Are Required for Fungal Pathogenesis. MBio 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, C.A.; Hartl, D.; Liu, W.; Lee, C.G.; Elias, J.A. TLR-2 and IL-17A in chitin-induced macrophage activation and acute inflammation. J. Immunol. 2008, 181, 4279–4286. [Google Scholar] [CrossRef] [PubMed]
- De Zuani, M.; Paolicelli, G.; Zelante, T.; Renga, G.; Romani, L.; Arzese, A.; Pucillo, C.E.M.; Frossi, B. Mast Cells Respond to Candida albicans Infections and Modulate Macrophages Phagocytosis of the Fungus. Front. Immunol. 2018, 9, 2829. [Google Scholar] [CrossRef] [PubMed]
- Nieto-Patlan, A.; Campillo-Navarro, M.; Rodriguez-Cortes, O.; Munoz-Cruz, S.; Wong-Baeza, I.; Estrada-Parra, S.; Estrada-Garcia, I.; Serafin-Lopez, J.; Chacon-Salinas, R. Recognition of Candida albicans by Dectin-1 induces mast cell activation. Immunobiology 2015, 220, 1093–1100. [Google Scholar] [CrossRef] [PubMed]
- Olynych, T.J.; Jakeman, D.L.; Marshall, J.S. Fungal zymosan induces leukotriene production by human mast cells through a dectin-1-dependent mechanism. J. Allergy Clin. Immunol. 2006, 118, 837–843. [Google Scholar] [CrossRef]
- Pinke, K.H.; Lima, H.G.; Cunha, F.Q.; Lara, V.S. Mast cells phagocyte Candida albicans and produce nitric oxide by mechanisms involving TLR2 and Dectin-1. Immunobiology 2016, 221, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Marshall, J.S. Zymosan treatment of mouse mast cells enhances dectin-1 expression and induces dectin-1-dependent reactive oxygen species (ROS) generation. Immunobiology 2009, 214, 321–330. [Google Scholar] [CrossRef]
- Urb, M.; Pouliot, P.; Gravelat, F.N.; Olivier, M.; Sheppard, D.C. Aspergillus fumigatus induces immunoglobulin E-independent mast cell degranulation. J. Infect. Dis. 2009, 200, 464–472. [Google Scholar] [CrossRef]
- Lowy, F.D. Staphylococcus aureus infections. N. Engl. J. Med. 1998, 339, 520–532. [Google Scholar] [CrossRef]
- Dziarski, R.; Gupta, D. Staphylococcus aureus peptidoglycan is a toll-like receptor 2 activator: A reevaluation. Infect. Immun. 2005, 73, 5212–5216. [Google Scholar] [CrossRef]
- Vosskuhl, K.; Greten, T.F.; Manns, M.P.; Korangy, F.; Wedemeyer, J. Lipopolysaccharide-mediated mast cell activation induces IFN-gamma secretion by NK cells. J. Immunol. 2010, 185, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Varricchi, G.; Loffredo, S.; Borriello, F.; Pecoraro, A.; Rivellese, F.; Genovese, A.; Spadaro, G.; Marone, G. Superantigenic Activation of Human Cardiac Mast Cells. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- Leal-Berumen, I.; Snider, D.P.; Barajas-Lopez, C.; Marshall, J.S. Cholera toxin increases IL-6 synthesis and decreases TNF-alpha production by rat peritoneal mast cells. J. Immunol. 1996, 156, 316–321. [Google Scholar] [PubMed]
- Brown, G.D.; Herre, J.; Williams, D.L.; Willment, J.A.; Marshall, A.S.; Gordon, S. Dectin-1 mediates the biological effects of beta-glucans. J. Exp. Med. 2003, 197, 1119–1124. [Google Scholar] [CrossRef] [PubMed]
- Dennehy, K.M.; Brown, G.D. The role of the beta-glucan receptor Dectin-1 in control of fungal infection. J. Leukoc. Biol. 2007, 82, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Jesus, S.; Soares, E.; Borchard, G.; Borges, O. Adjuvant Activity of Poly-epsilon-caprolactone/Chitosan Nanoparticles Characterized by Mast Cell Activation and IFN-gamma and IL-17 Production. Mol. Pharm. 2018, 15, 72–82. [Google Scholar] [CrossRef] [PubMed]
| Human MC Source | Virus/Virus Replication | Degranulation | Lipid Mediator | Cytokine Synthesis | Additional Biological Responses | Citation |
|---|---|---|---|---|---|---|
| +ssRNA | ||||||
| HMC-1 cell line | RV16/yes | n.d. | n.d. | IL-6, IL-8, TNF-α, IFN-α | ↑ICAM ↓ Cell viability | [10] |
| LAD cell line and CBMC | RV1B and RV16/yes | NO | n.d. | IFN-β and -λ; CXCL10 and CCL5 | [11] | |
| HMC-1 and KU812 cell lines | RV14/yes | Enhanced following cross-linking of FcεRI | n.d. | Enhanced production of IL-8 and GM-CSF following cross-linking of the FcεRI | ↑ ICAM | [12] |
| Skin MCs and human skin tissue | DENV type 2 (NGC and K0048)/yes | Yes | n.d. | CCL5, IL-6, IL-8, VEGF | -MC mediators releases in response to infection with DENV induce activation and proliferation of endothelial cells -DENV localized in MC cytoplasmic granules was shown to be infectious | [13] |
| HMC-1 and KU812 cell lines | DENV type 2 strain 16681/yes | n.d. | n.d. | n.d. | Anti-DENV neutralizing antibodies enhanced DENV infection in KU812 and HMC-1 cells in a mechanism involving autophagy | [14] |
| CBMC, HMC-1, and KU812 | DENV type 2 strain 16681/n.d. | n.d. | n.d. | CCL2, CCL4, CXCL10, type I IFNs, | MC-derived type I IFNs prevented infection of KU812 with DENV | [15] |
| -ssRNA | ||||||
| RSV long strain/limited | n.d. | No | CCL4, CCL5, CXCL10, IFN-α | CBMC [16] | ||
| HMC-1 | RSV long strain/inefficient | Only in co-culture of MCs with RSV-infected A546 epithelial cells | n.d. | TNF-α only in co-culture of MCs with RSV-infected A546 epithelial cells | [17] | |
| dsRNA | ||||||
| CBMC | Reovirus/yes | No | No | CXCL8, Type I IFNs, IL-10, TNF | Reovirus-infected MC induce the recruitment and activation of NK cells to sites of infection Recruitment of NKT [18] cells was also observed | [15,19] |
| Blood derived MC precursors | HIV-1 (M-tropic)/ yes | n.d | n.d. | n.d. | [20,21] | |
| Non-human MCs | ||||||
| P815 murine cell line | Influenza H1N1 (A/WSN/33), H5N1 (A/Chicken/Henan/1/04), H7N2 (A/Chicken/Hebei/2/02)/yes, dependent on MC apoptosis | n.d. | n.d. | IL-6, IL-18, TNF-α, and CCL2 | MC apoptosis | [22] |
| Murine bone marrow MCs | Influenza Influenza H1N1 (A/WSN/33) virus/inefficient | Yes | Yes | CCL2, CCL3, CCL4, CCL5, CXCL2, CXCL9, CXCL10, IL-6, and TNF-α | [23] | |
| P815 murine cell line | Influenza H5N1 (A/Chicken/Henan/1/04) | Yes | n.d. | IFN-γ | [24] | |
| Porcine primary MCs | Influenza H1N1 (A/Ca/04/2009) virus/inefficient | Yes | n.d. | IL1A, IL6, CXCL9, CXCL10, CXCL11 | [25] |
| Murine Model | Virus | Biological Responses Observed * | Implication(s) | Citation |
|---|---|---|---|---|
| Balb/c | Influenza H1N1 (A/PR/8/34) | Following infection, -MCs progenitors recruited to lungs -MCs associated with inflammatory cells surrounding bronchioles | Increased number of MCs in the lungs in response to influenza may be associated with virus-induced asthma exacerbations | [26] |
| Balb/c immunized with both the HA influenza protein and the MC activator C48/80 | Influenza H1N1 (A/Ca/04/2009) | -Enhanced levels of serum IgG and mucosal IgA against HA protein. -Reduced levels of virus titers in lungs -Predominant Th1 over Th2 cellular responses | The vaccine approach combining HA and mucosal adjuvant C4/80 elicits protective immunity specifically [27] against H1N1 virus | [28] |
| C57BL/6 and B6.Cg-KitW-sh | Influenza H1N1 (A/WSN/33) | MC-deficient mice -Less susceptible to lose weight -Showed reduced numbers of inflammatory cells in lungs | MCs are crucial effectors in the pathological innate immune responses | [23] |
| Balb/c | Influenza H5N1 (A/Chicken/Henan/1/04) | Severe bronchiolitis and infiltration of inflammatory cells to lungs were reduced in mice treated with ketotifen previous and during infection with H5N1 virus | MC activities, specifically degranulation, promote lung lesions during viral infection | [24] |
| C57BL/6NTac mice | DENV strain EDEN2 | Many of the pathological changes derived from infection with dengue virus, including metabolic dysregulation and inflammation, were reversed by treatment of infected mice with ketotifen | Therapy for dengue virus infection may include the use of MC stabilizer drugs | [29] |
| C3H/HeN | DENV type 2 strain 16681 | MC degranulation and production of CCL-2, CCL5, and CXCL10 in response to dengue virus infection were reduced in mice treated with antibodies targeting the NS1 dengue protein. | Dengue-associated pathological effects can be reduced using anti-NS1 antibodies by mechanisms involving inhibition of MC activities | [30] |
| C57BL/6 and MC-deficient KitW-sh/ HNuhrJaeBsmJ | DENV type 2 strain 16681 | KitW-sh mice: -Were more susceptible to infection with DENV -Showed prolonged bleeding and enhanced production of macrophage-derived CCL2 and macrophage infiltration at inoculation sites | MCs and macrophages coordinately may restrict DENV infection in the skin | [31] |
| C57BL/6 | DENV type 2, strain Eden 2 | -MCs infected with DENV promote increased vascular permeability via chymase and leukotriene production -Usage of MC-stabilizing drugs restore vascular permeability in mice infected with DENV | -DENV-associated vascular leakage might be prevented by therapeutically targeting MC activities -Translation of these data to human settings showed chymase as a predictive biomarker distinguishing dengue fever from dengue hemorrhagic fever | [32] |
| C57BL/6 | Vaccinia virus strain Western Reserve | -LAT-activated MCs showed improved antiviral activities against VV -MCs produce cathelicidin via TLR2 in response to LTA expressed by commensal bacteria | MCs primed via TLR2 fight more efficiently vaccinia virus | [33] |
| Major Pathogen Products | Associated MC Receptor | Example of Pathogens | Citation |
|---|---|---|---|
| Bacterial Pathogens and Products | |||
| Peptidoglycan | TLR2 | S. aureus | [76] |
| Lipopolysaccharide | TLR4 | E. coli | [77] |
| CpG motif-containing bacterial DNA | TLR9 † | Multiple strains | [78,79] |
| Fimbriated adhesion molecule H | CD48 | Fimbriated E. coli | [80] |
| Protein A | Fc receptors | S. aureus | [81] |
| Staphylococcal enterotoxins | Undefined | S. aureus | [82,83] |
| Staphylococcal superantigen-like proteins | TLR2 | S. aureus | [84] |
| Cytolysin | Substance P receptor | V. cholerae | [85] |
| Pertussis toxin | CD48 | B. pertussis | [86] |
| Clostridium toxin | C. difficile | [87] | |
| Mycobacterial antigens | M. tuberculosis | [88,89] | |
| Viral Pathogens and Products | |||
| dsRNA | TLR3 | RSV, Reovirus | [73,90] |
| ssRNA | TLR7 † | Influenza A, VSV, Sendai | [78,91] |
| CpG motif-containing viral DNA | TLR9 † | mCMV, HSV | [78,92,93,94] |
| dsRNA, uncapped viral RNA | RIG-I | Influenza A, Dengue | [15,23,95] |
| Orf virus-encoded IL-10 | IL-10 receptor | Epstein Barr virus | [96] |
| Superantigens (Protein Fv, envelope glycoprotein gp120) | Fc receptors | Viral hepatitis, HIV-1 | [67,68,97] |
| Fungal Pathogens and Products | |||
| Yeast zymosan, chitin and derivatives † | TLR2 | C. albicans S. cerevisiae, C. neoformans | [98,99,100,101,102] |
| β-glucans, zymosan, chitin and derivatives † | Dectin-1 | C. albicans, S. cerevisiae, C. neoformans | [99,100,101,103,104,105,106,107] |
| Mature fungal hyphae | IgE-independent; StuA and MedA transcription factor-mediated | Aspergillus fumigatus | [108] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marshall, J.S.; Portales-Cervantes, L.; Leong, E. Mast Cell Responses to Viruses and Pathogen Products. Int. J. Mol. Sci. 2019, 20, 4241. https://doi.org/10.3390/ijms20174241
Marshall JS, Portales-Cervantes L, Leong E. Mast Cell Responses to Viruses and Pathogen Products. International Journal of Molecular Sciences. 2019; 20(17):4241. https://doi.org/10.3390/ijms20174241
Chicago/Turabian StyleMarshall, Jean S., Liliana Portales-Cervantes, and Edwin Leong. 2019. "Mast Cell Responses to Viruses and Pathogen Products" International Journal of Molecular Sciences 20, no. 17: 4241. https://doi.org/10.3390/ijms20174241
APA StyleMarshall, J. S., Portales-Cervantes, L., & Leong, E. (2019). Mast Cell Responses to Viruses and Pathogen Products. International Journal of Molecular Sciences, 20(17), 4241. https://doi.org/10.3390/ijms20174241





