Author Contributions
H.C. performed the investigation, analyzed the data, and wrote the paper. W.G., B.Z., G.L., T.L., Y.Y. (Yanyan Yuan), N.Z., Y.Y. (Yuwei Yang), W.F., F.C., and T.L. performed the investigation. S.W., B.X., P.W., and H.L. designed the study and amended the paper. All the other authors read, improved, and approved the manuscript.
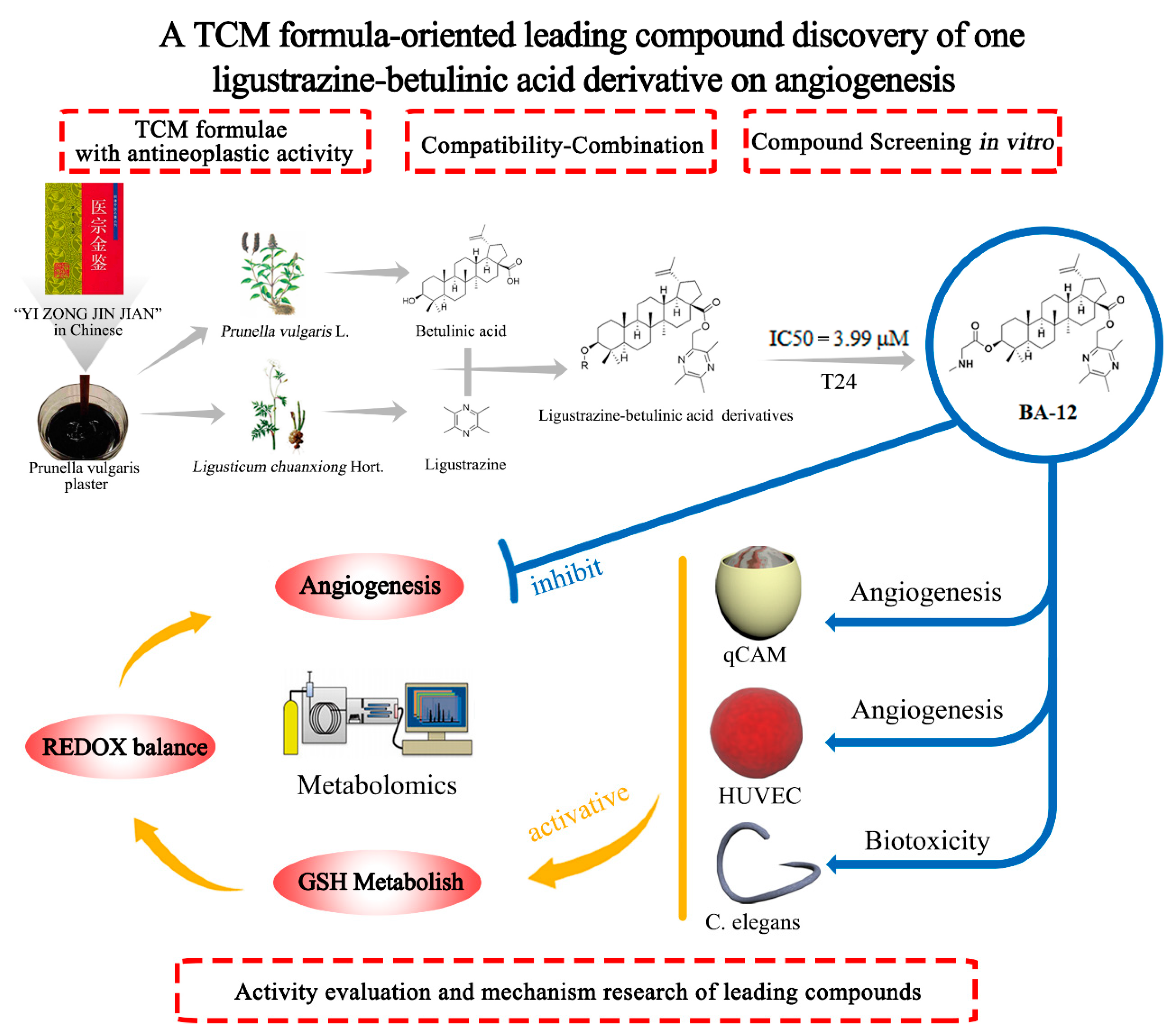
Figure 1.
The leading compound discovery strategy based on the compatibility theory of traditional Chinese medicine and the combination principle of pharmacochemistry.
Figure 1.
The leading compound discovery strategy based on the compatibility theory of traditional Chinese medicine and the combination principle of pharmacochemistry.
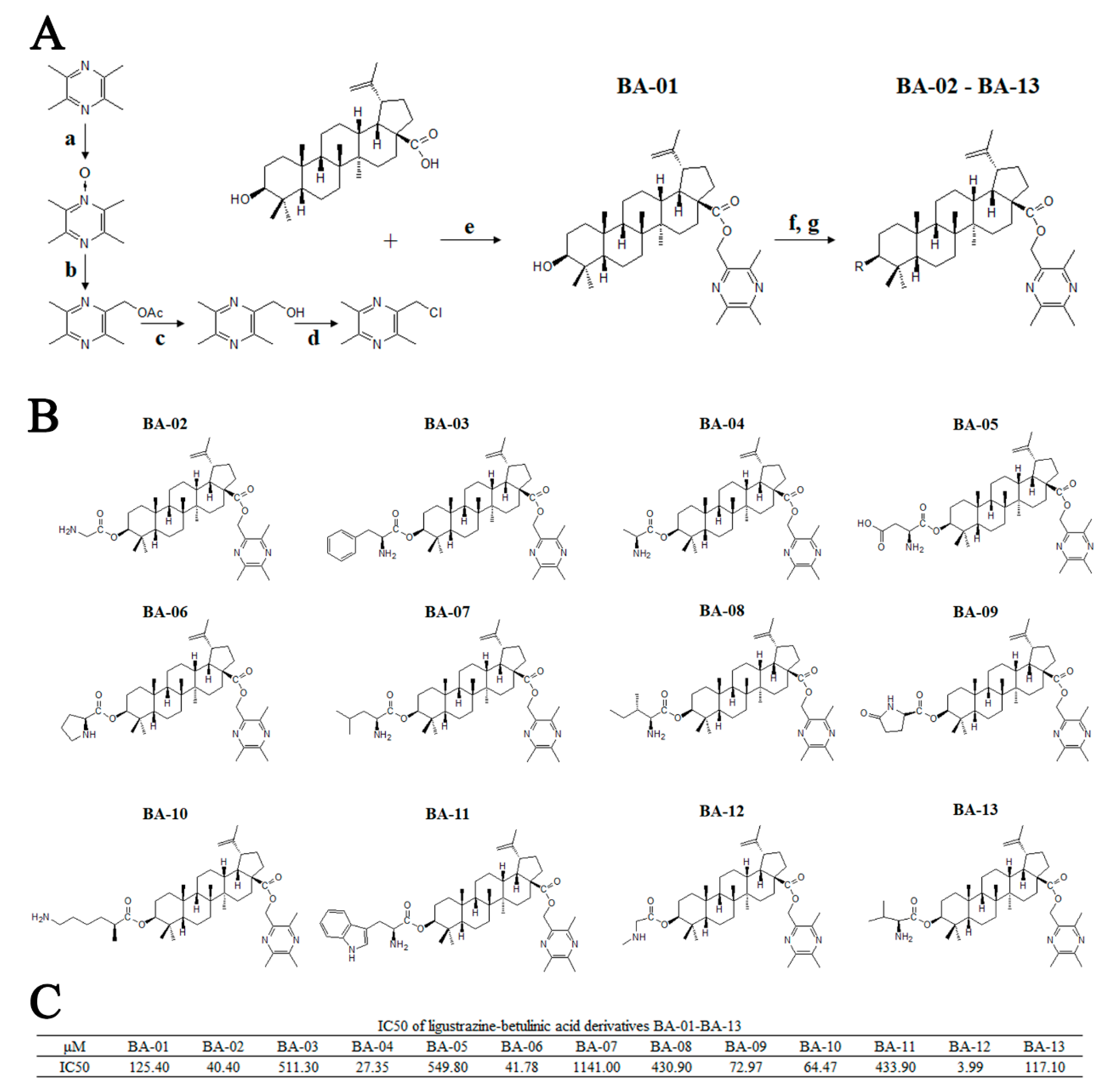
Figure 2.
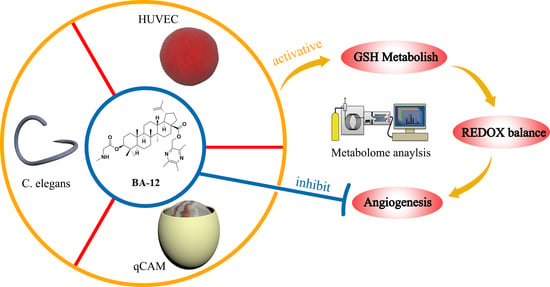
Synthesis, chemical information, and antitumor activities of ligustrazine–betulinic acid derivatives (BA-01–BA-13) on human bladder carcinoma (T24) cells. (A) Synthesis of ligustrazine–betulinic acid derivatives (BA-01–BA-13). Conditions and reagents: (a) acetic acid and 30% H2O2, reflux at 90 °C for 6 h; (b) acetic anhydride, reflux at 105 °C for 2 h; (c) methanol (MeOH):tetrahydrofuran (THF):H2O ¼ 1:3:1 with NaOH for 1 h; (d) tosyl chloride (TsCl), THF, 4-dimethylaminopyridine (DMAP), trimethylamine (TEA) for 12 h; (e) dry K2CO3, dry N,N-dimethylformamide (DMF) at 25 °C for 12 h; (f) N-benzyloxycarbonyl (N-Cbz) or N-butyloxy carbonyl (N-Boc) amino acids, DMAP, 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDCI), dichloromethane (DCM) at 25 °C for 12 h; (g) Pd/C (10%), MeOH at 25°C for 12 h, or trifluoroacetic acid (TFA) in dry DCM at 0 °C for 2 h. (B) Structures of ligustrazine–betulinic acid derivatives (BA-01–BA-13). (C) Half maximal inhibitory concentration (IC50) values of ligustrazine–betulinic acid derivatives (BA-01–BA-13) on T24 cells, evaluated using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assays.
Figure 2.
Synthesis, chemical information, and antitumor activities of ligustrazine–betulinic acid derivatives (BA-01–BA-13) on human bladder carcinoma (T24) cells. (A) Synthesis of ligustrazine–betulinic acid derivatives (BA-01–BA-13). Conditions and reagents: (a) acetic acid and 30% H2O2, reflux at 90 °C for 6 h; (b) acetic anhydride, reflux at 105 °C for 2 h; (c) methanol (MeOH):tetrahydrofuran (THF):H2O ¼ 1:3:1 with NaOH for 1 h; (d) tosyl chloride (TsCl), THF, 4-dimethylaminopyridine (DMAP), trimethylamine (TEA) for 12 h; (e) dry K2CO3, dry N,N-dimethylformamide (DMF) at 25 °C for 12 h; (f) N-benzyloxycarbonyl (N-Cbz) or N-butyloxy carbonyl (N-Boc) amino acids, DMAP, 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDCI), dichloromethane (DCM) at 25 °C for 12 h; (g) Pd/C (10%), MeOH at 25°C for 12 h, or trifluoroacetic acid (TFA) in dry DCM at 0 °C for 2 h. (B) Structures of ligustrazine–betulinic acid derivatives (BA-01–BA-13). (C) Half maximal inhibitory concentration (IC50) values of ligustrazine–betulinic acid derivatives (BA-01–BA-13) on T24 cells, evaluated using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assays.
![Ijms 20 04062 g002 Ijms 20 04062 g002]()
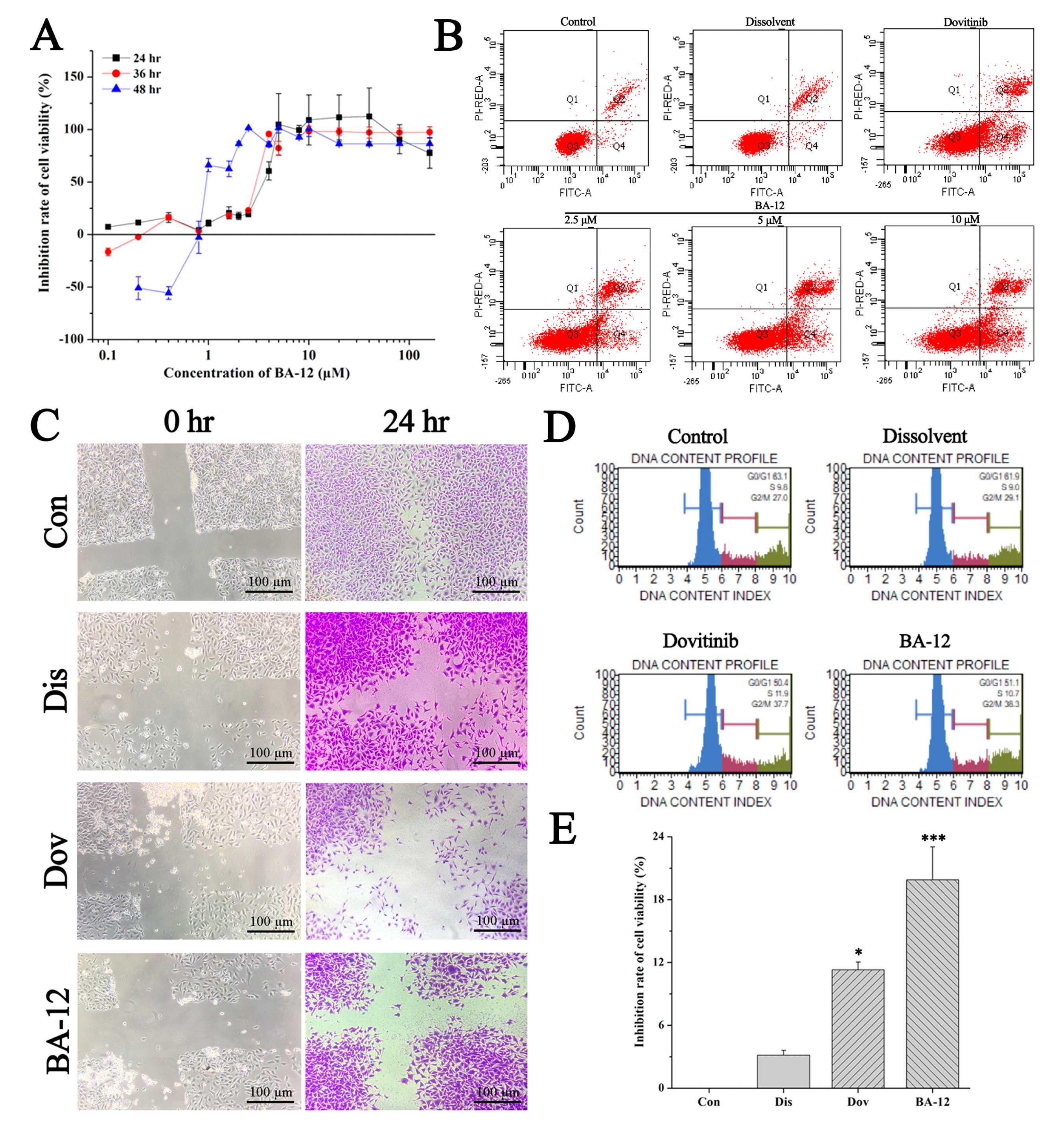
Figure 3.
The in vitro antitumor activity of BA-12 on T24 cells. (A) Inhibition rate of cell viability of T24 cells for MTT assays (cells treated with BA-12 at doses of 0.25–160 μM) for 24, 48, and 72 h. (B) Apoptosis analysis of T24 cells induced by agents using Annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) staining. (C) Results for wound scratch assay (cells treated with BA-12 at doses of 2.5 μM) after 24 h under the microscope (100×). The most representative fields are shown. (D) Cell cycle analysis using PI staining (cells treated with BA-12 at doses of 2.5 μM). (E) Inhibition rate of cell viability of T24 cells for MTT assays (cells treated with BA-12 at doses of 2.5 μM) for 24 h. ANOVA with a post hoc test was used to calculate the significance of the differences; * p < 0.05, *** p < 0.001 compared with the dissolvent group. Experiments were executed three times. Results are displayed as means ± SD.
Figure 3.
The in vitro antitumor activity of BA-12 on T24 cells. (A) Inhibition rate of cell viability of T24 cells for MTT assays (cells treated with BA-12 at doses of 0.25–160 μM) for 24, 48, and 72 h. (B) Apoptosis analysis of T24 cells induced by agents using Annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) staining. (C) Results for wound scratch assay (cells treated with BA-12 at doses of 2.5 μM) after 24 h under the microscope (100×). The most representative fields are shown. (D) Cell cycle analysis using PI staining (cells treated with BA-12 at doses of 2.5 μM). (E) Inhibition rate of cell viability of T24 cells for MTT assays (cells treated with BA-12 at doses of 2.5 μM) for 24 h. ANOVA with a post hoc test was used to calculate the significance of the differences; * p < 0.05, *** p < 0.001 compared with the dissolvent group. Experiments were executed three times. Results are displayed as means ± SD.
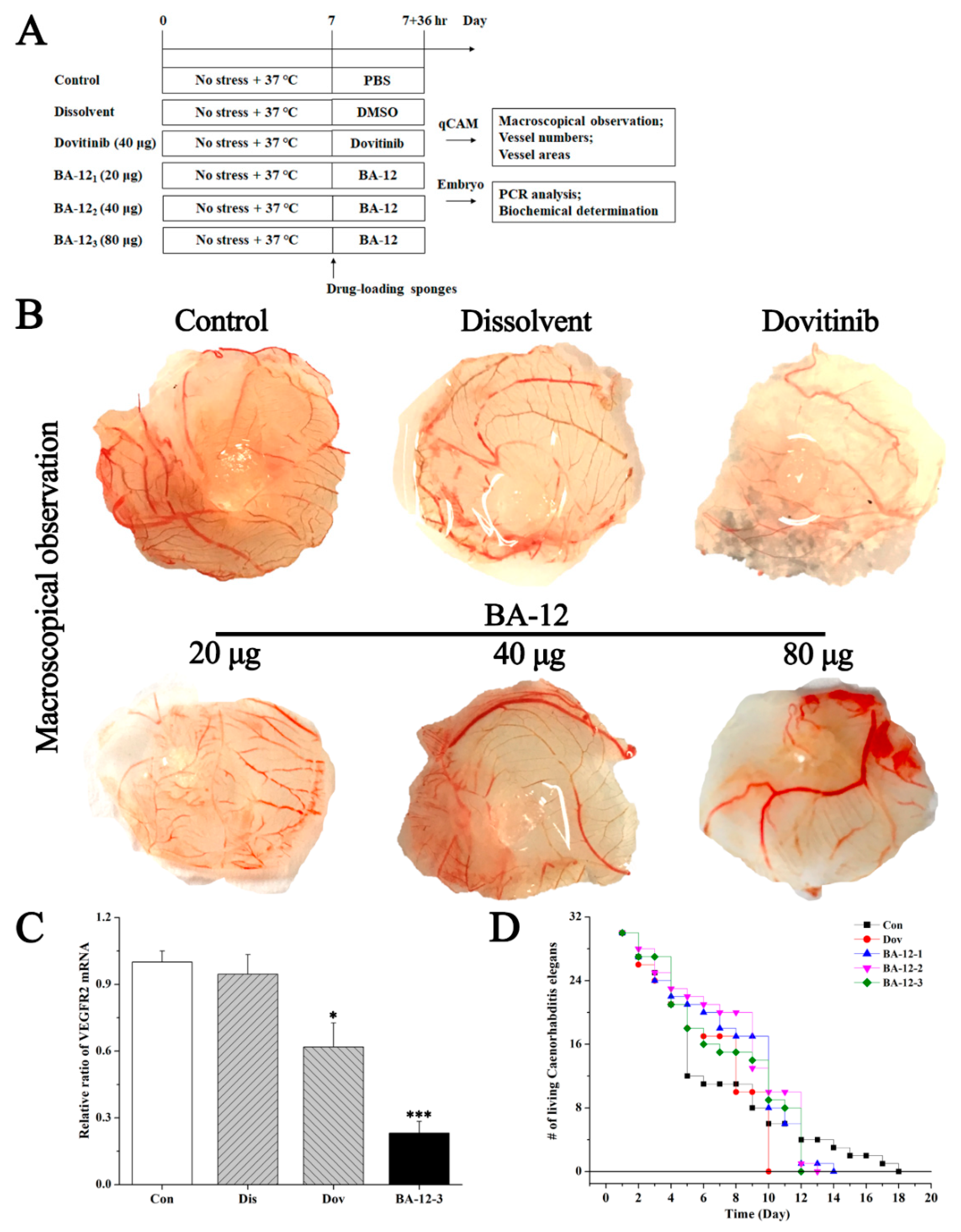
Figure 4.
The effect of BA-12 on angiogenesis using a quail chick chorioallantoic membrane (qCAM) and human umbilical vein endothelial cells (HUVECs), and its biotoxicity based on a survival essay with Caenorhabditis elegans. (A) Schematic diagram of the experimental design. Con, control group treated with phosphate-buffered saline (PBS); Dis, dissolvent group treated with dissolvent containing 0.5% dimethyl sulfoxide (DMSO); Pos, positive control group treated with dovitinib (40 μg); BA-12 1–3, groups treated with BA-12 at doses of 20, 40, and 80 μg, respectively. (B) Inhibition of angiogenesis by BA-12 for qCAM assays in a dose-dependent manner. (C) Messenger RNA (mRNA) levels of vascular endothelial growth factor receptor 2 (VEGFR2) in qCAM samples. The most representative fields are shown. (D) Survival assay using Caenorhabditis elegans for biotoxicity detection. ANOVA with a post hoc test was used to calculate the significance of the differences; * p < 0.05, *** p < 0.001 compared with the dissolvent group. Experiments were executed three times. Results are displayed as means ± SD.
Figure 4.
The effect of BA-12 on angiogenesis using a quail chick chorioallantoic membrane (qCAM) and human umbilical vein endothelial cells (HUVECs), and its biotoxicity based on a survival essay with Caenorhabditis elegans. (A) Schematic diagram of the experimental design. Con, control group treated with phosphate-buffered saline (PBS); Dis, dissolvent group treated with dissolvent containing 0.5% dimethyl sulfoxide (DMSO); Pos, positive control group treated with dovitinib (40 μg); BA-12 1–3, groups treated with BA-12 at doses of 20, 40, and 80 μg, respectively. (B) Inhibition of angiogenesis by BA-12 for qCAM assays in a dose-dependent manner. (C) Messenger RNA (mRNA) levels of vascular endothelial growth factor receptor 2 (VEGFR2) in qCAM samples. The most representative fields are shown. (D) Survival assay using Caenorhabditis elegans for biotoxicity detection. ANOVA with a post hoc test was used to calculate the significance of the differences; * p < 0.05, *** p < 0.001 compared with the dissolvent group. Experiments were executed three times. Results are displayed as means ± SD.
![Ijms 20 04062 g004 Ijms 20 04062 g004]()
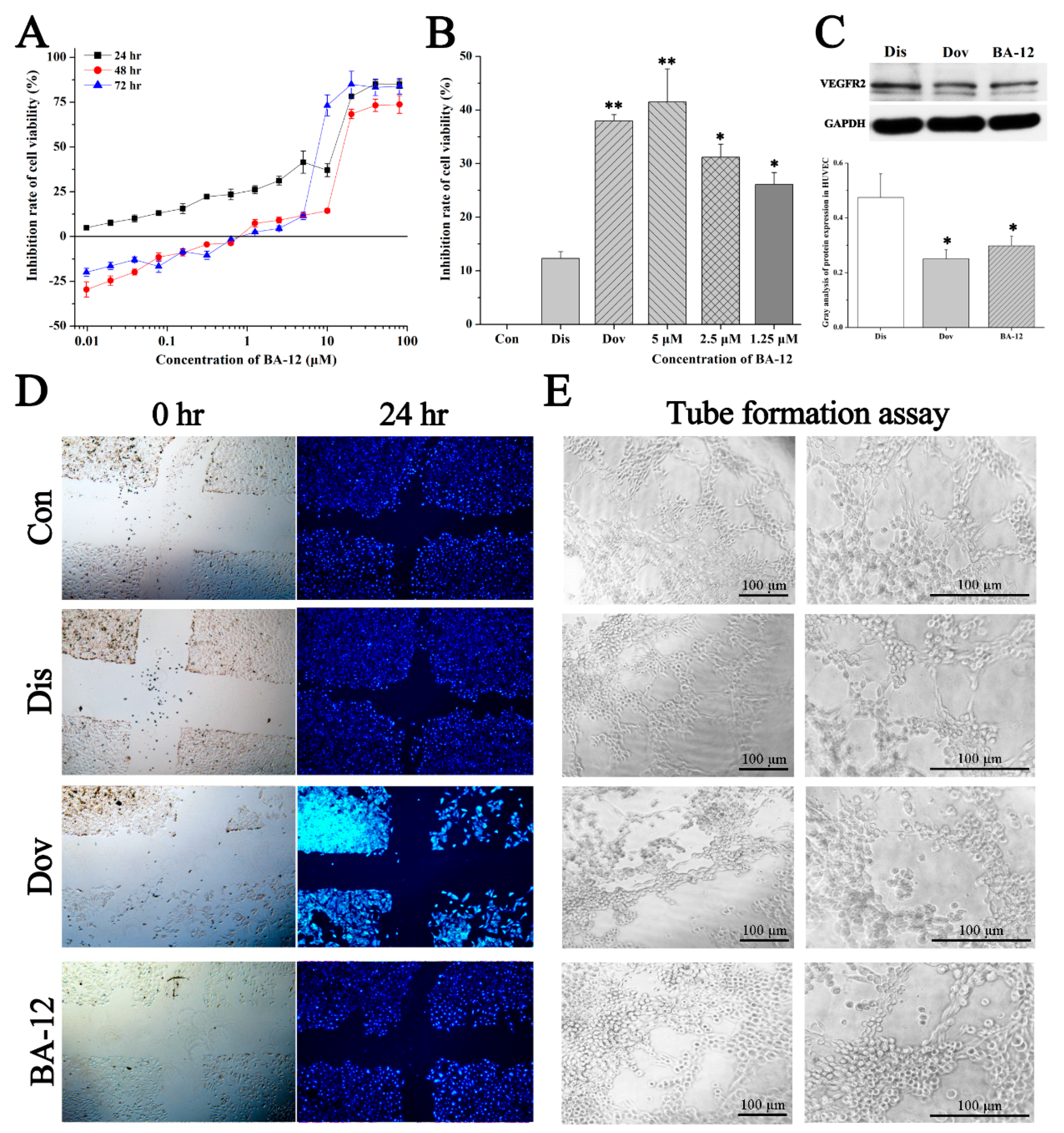
Figure 5.
The effect of BA-12 on angiogenesis in HUVECs. (A) Inhibition rate of cell viability of HUVECs by MTT assays (cells treated with BA-12 at doses of 0.25–160 μM) for 24, 48, and 72 h. (B) Inhibition rate of cell viability of HUVECs for MTT assays for 24 h. (C) Cellular VEGFR levels and grayscale analysis. (D,E) Results for wound scratch assay and tube formation assay after 24 h under the microscope (100×). Con, control group treated with PBS; Dis, dissolvent group treated with dissolvent containing 0.5% DMSO; Pos, positive control group treated with dovitinib (2.5 μM); BA-12, groups treated with BA-12 at doses of 2.5 μM. Representative fields are shown. ANOVA with a post hoc test was applied for the significance of the differences; * p < 0.05, ** p < 0.01 compared with the dissolvent group. Experiments were executed three times. Results are displayed as means ± SD.
Figure 5.
The effect of BA-12 on angiogenesis in HUVECs. (A) Inhibition rate of cell viability of HUVECs by MTT assays (cells treated with BA-12 at doses of 0.25–160 μM) for 24, 48, and 72 h. (B) Inhibition rate of cell viability of HUVECs for MTT assays for 24 h. (C) Cellular VEGFR levels and grayscale analysis. (D,E) Results for wound scratch assay and tube formation assay after 24 h under the microscope (100×). Con, control group treated with PBS; Dis, dissolvent group treated with dissolvent containing 0.5% DMSO; Pos, positive control group treated with dovitinib (2.5 μM); BA-12, groups treated with BA-12 at doses of 2.5 μM. Representative fields are shown. ANOVA with a post hoc test was applied for the significance of the differences; * p < 0.05, ** p < 0.01 compared with the dissolvent group. Experiments were executed three times. Results are displayed as means ± SD.
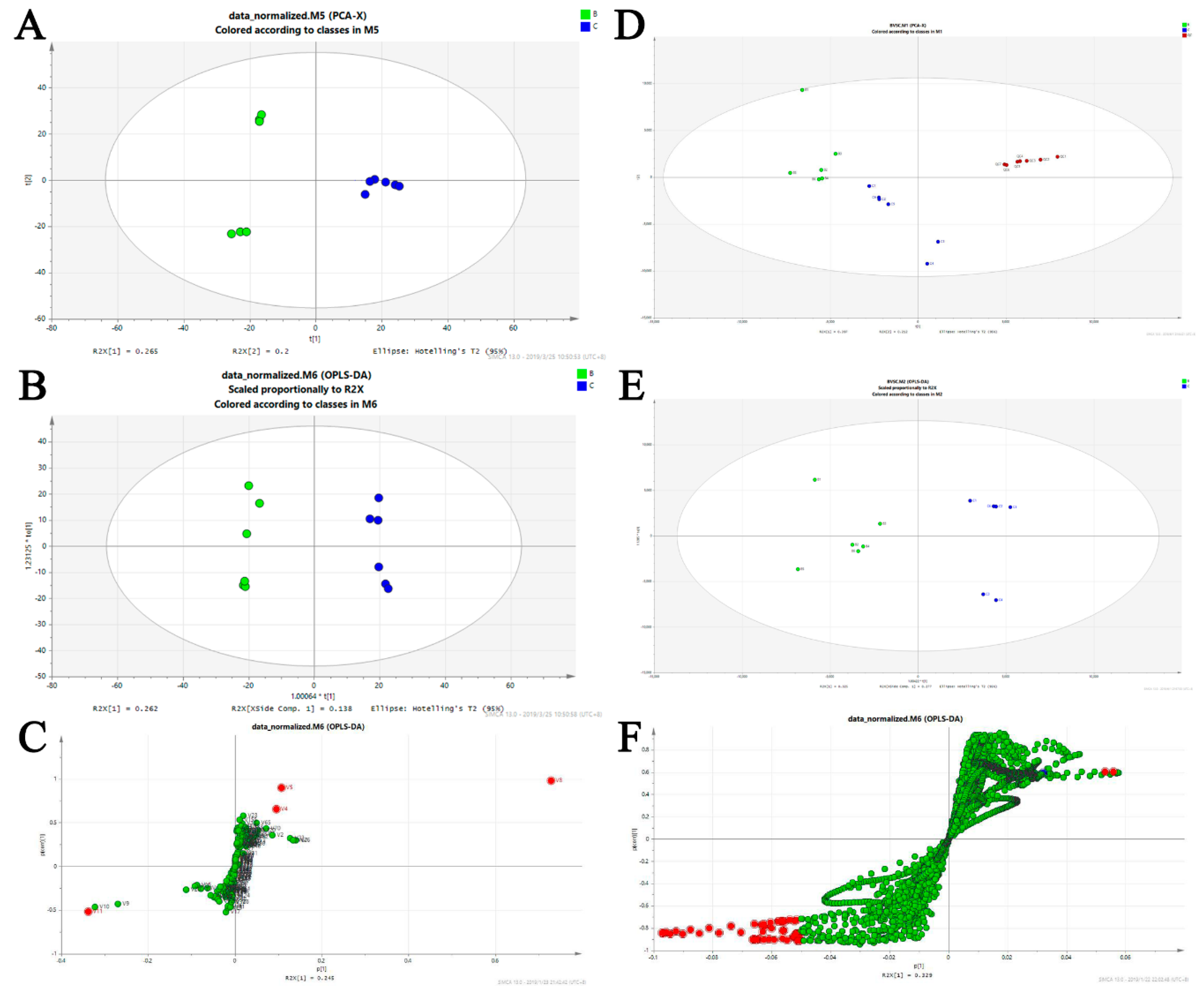
Figure 6.
Biomarker identification and altered metabolic pathways regulated by BA-12. For GC–MS metabolomics: (A) score plots of control (C group, PBS) and BA-12 (B group, 2.5 μM) from principal component analysis (PCA) for principal component 1 (PC1) versus PC2; (B) score plots of control (C group, PBS) and BA-12 (B group, 2.5 μM) from orthogonal projections to latent structures discriminant analysis (OPLS-DA) model for the pairwise comparisons; (C) S-plot of the OPLS-DA model for the B and C groups. The points in red indicate the identified biomarkers. For UPLC–TOF-MS metabolomics: (D) score plots of control (C group, PBS) and BA-12 (B group, 2.5 μM) from PCA in the positive electrospray ionization (ESI+) mode for PC1 versus PC2; (E) score plots of control (C group, PBS) and BA-12 (B group, 2.5 μM) from OPLS-DA model for the pairwise comparisons; (F) S-plot of the OPLS-DA model for the B and C groups. The points in red indicate the identified biomarkers.
Figure 6.
Biomarker identification and altered metabolic pathways regulated by BA-12. For GC–MS metabolomics: (A) score plots of control (C group, PBS) and BA-12 (B group, 2.5 μM) from principal component analysis (PCA) for principal component 1 (PC1) versus PC2; (B) score plots of control (C group, PBS) and BA-12 (B group, 2.5 μM) from orthogonal projections to latent structures discriminant analysis (OPLS-DA) model for the pairwise comparisons; (C) S-plot of the OPLS-DA model for the B and C groups. The points in red indicate the identified biomarkers. For UPLC–TOF-MS metabolomics: (D) score plots of control (C group, PBS) and BA-12 (B group, 2.5 μM) from PCA in the positive electrospray ionization (ESI+) mode for PC1 versus PC2; (E) score plots of control (C group, PBS) and BA-12 (B group, 2.5 μM) from OPLS-DA model for the pairwise comparisons; (F) S-plot of the OPLS-DA model for the B and C groups. The points in red indicate the identified biomarkers.
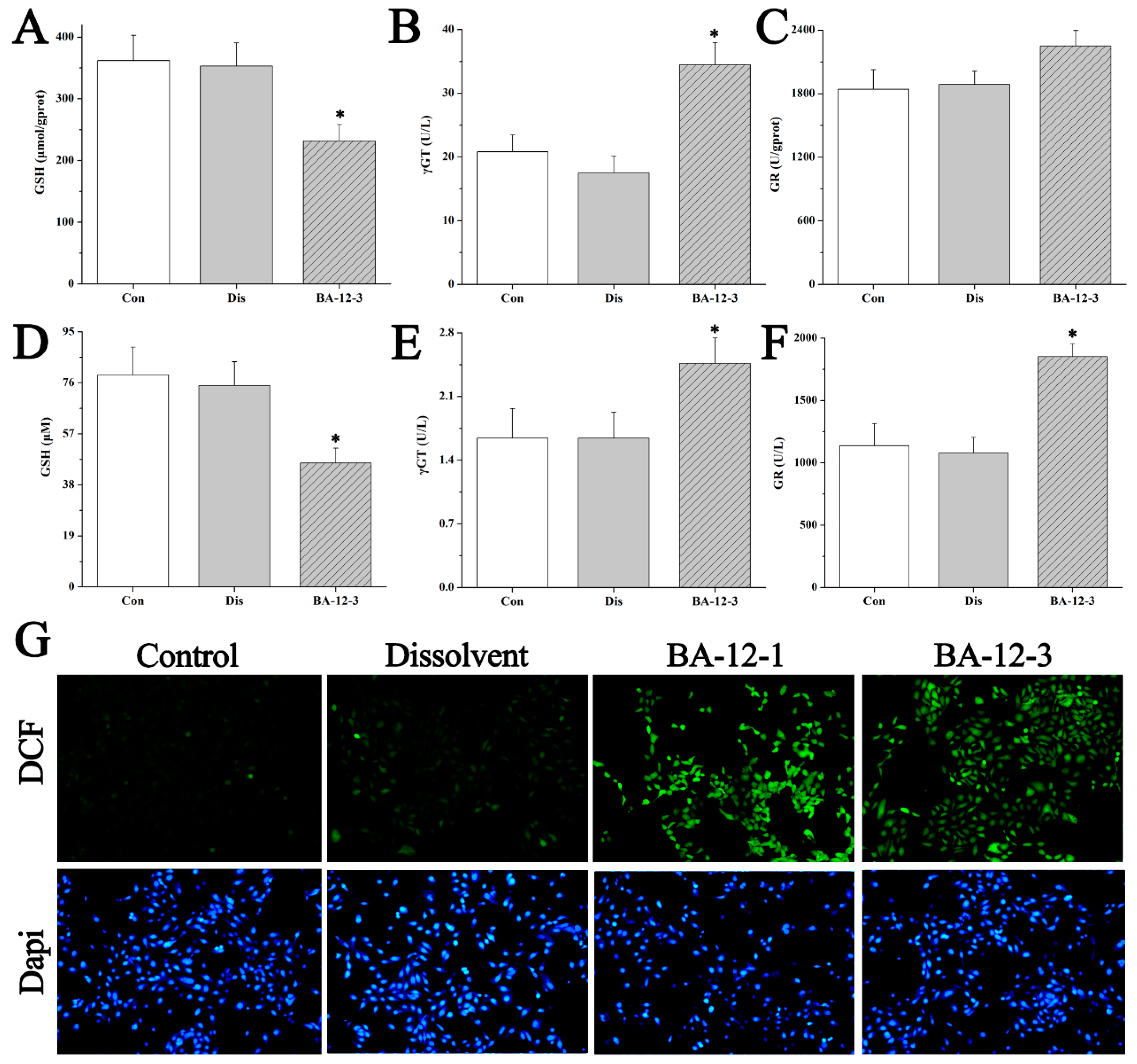
Figure 7.
Mechanisms of BA-12 for glutathione (GSH) metabolism activation. (A–C) GSH, γ-glutamyl transferase (γGT), and glutathione reductase (GR) levels in quail samples, respectively. (D–F) GSH, γGT, and GR levels in cell samples, respectively. (G) Morphological observation and relative fluorescence intensity of HUVECs induced by BA-12 (10, 2.5 μM) for 24 h by reactive oxygen species (ROS) assay and 4′,6-diamidino-2-phenylindole (DAPI) staining under the fluorescence microscope (100×). Dichlorofluorescein (DCF), compounds with fluorescence for the reflection of ROS levels. The most representative fields are shown. Con, control group treated with PBS; Dis, dissolvent group treated with dissolvent containing 0.5% DMSO; BA-12-1 and BA-12-3, groups treated with BA-12 at doses of 5 and 1.625 μg, respectively. ANOVA with a post hoc test was applied for the significance of the differences; * p < 0.05 compared with the dissolvent group. Experiments were executed three times. Results are displayed as means ± SD.
Figure 7.
Mechanisms of BA-12 for glutathione (GSH) metabolism activation. (A–C) GSH, γ-glutamyl transferase (γGT), and glutathione reductase (GR) levels in quail samples, respectively. (D–F) GSH, γGT, and GR levels in cell samples, respectively. (G) Morphological observation and relative fluorescence intensity of HUVECs induced by BA-12 (10, 2.5 μM) for 24 h by reactive oxygen species (ROS) assay and 4′,6-diamidino-2-phenylindole (DAPI) staining under the fluorescence microscope (100×). Dichlorofluorescein (DCF), compounds with fluorescence for the reflection of ROS levels. The most representative fields are shown. Con, control group treated with PBS; Dis, dissolvent group treated with dissolvent containing 0.5% DMSO; BA-12-1 and BA-12-3, groups treated with BA-12 at doses of 5 and 1.625 μg, respectively. ANOVA with a post hoc test was applied for the significance of the differences; * p < 0.05 compared with the dissolvent group. Experiments were executed three times. Results are displayed as means ± SD.
![Ijms 20 04062 g007 Ijms 20 04062 g007]()
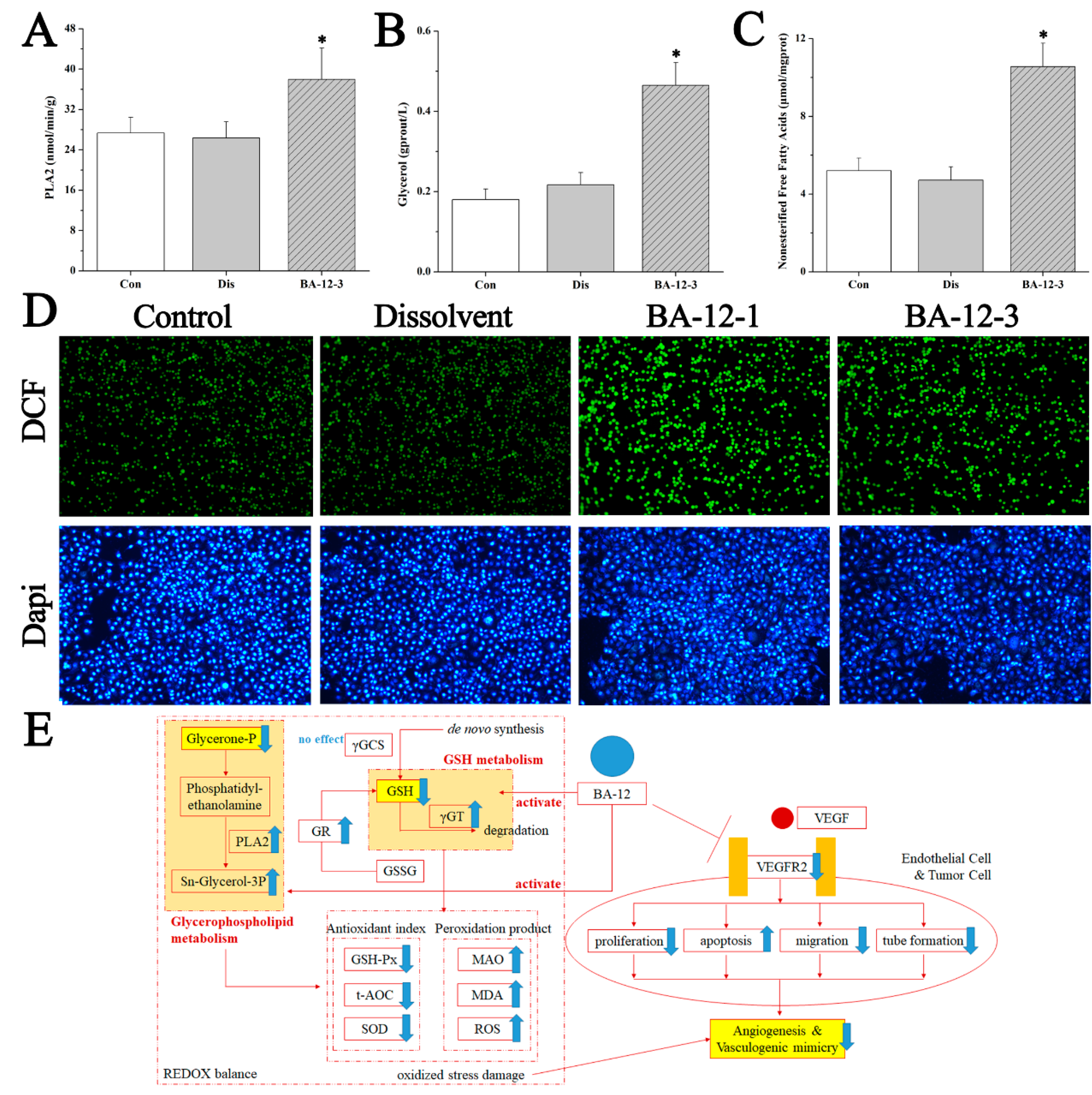
Figure 8.
Mechanisms of BA-12 for glycerophospholipid metabolism activation. (A–C) Phospholipase A2 (PLA2), glycerol, and non-esterified free fatty acid (NEFA) levels in quail samples, respectively. (D) Morphological observation and relative fluorescence intensity of T24 cells induced by BA-12 for 24 h by ROS assay and DAPI staining under the fluorescence microscope (100×). DCF, compounds with fluorescence for the reflection of ROS levels. The most representative fields are shown. (E) Schematic diagram of the efficacy and mechanism of BA-12 for angiogenesis. Con, control group treated with PBS; Dis, dissolvent group treated with dissolvent containing 0.5% DMSO; BA-12-1 and BA-12-3, groups treated with BA-12 at doses of 5 and 1.625 μg, respectively. ANOVA with a post hoc test was applied for the significance of the differences; * p < 0.05 compared with the dissolvent group. Experiments were executed three times. Results are displayed as means ± SD.
Figure 8.
Mechanisms of BA-12 for glycerophospholipid metabolism activation. (A–C) Phospholipase A2 (PLA2), glycerol, and non-esterified free fatty acid (NEFA) levels in quail samples, respectively. (D) Morphological observation and relative fluorescence intensity of T24 cells induced by BA-12 for 24 h by ROS assay and DAPI staining under the fluorescence microscope (100×). DCF, compounds with fluorescence for the reflection of ROS levels. The most representative fields are shown. (E) Schematic diagram of the efficacy and mechanism of BA-12 for angiogenesis. Con, control group treated with PBS; Dis, dissolvent group treated with dissolvent containing 0.5% DMSO; BA-12-1 and BA-12-3, groups treated with BA-12 at doses of 5 and 1.625 μg, respectively. ANOVA with a post hoc test was applied for the significance of the differences; * p < 0.05 compared with the dissolvent group. Experiments were executed three times. Results are displayed as means ± SD.
Table 1.
Effects of a ligustrazine–betulinic acid derivative (BA-12) on apoptosis and cell cycle of human bladder carcinoma (T24) cells. G—gap phase; M—mitosis phase; S—synthesis phase.
Table 1.
Effects of a ligustrazine–betulinic acid derivative (BA-12) on apoptosis and cell cycle of human bladder carcinoma (T24) cells. G—gap phase; M—mitosis phase; S—synthesis phase.
| Group | Apoptosis Ratio (%) | G0/G1 (%) | S (%) | G2/M (%) |
|---|
| Control | 5.7 ± 1.2 | 62.5 ± 6.7 | 9.4 ± 1.8 | 27.6 ± 3.4 |
| Dissolvent | 7.2 ± 1.5 | 62.1 ± 5.1 | 9.2 ± 1.3 | 29.0 ± 3.8 |
| Dovitinib (2.5 μM) | 25.5 ± 5.1 ** | 50.8 ± 4.2 * | 11.1 ± 1.5 | 37.5 ± 4.4 * |
| BA-12 (2.5 μM) | 20.3 ± 4.2 * | 51.7 ± 4.4 * | 10.3 ± 1.6 | 38.7 ± 4.0 * |
Table 2.
Differential identified metabolites for discrimination among control and BA-12 groups based on GC–MS metabolomics. RT—retention time; VIP—variable importance for projection; MEOX—methoxime; TMS—trimethylsilyl.
Table 2.
Differential identified metabolites for discrimination among control and BA-12 groups based on GC–MS metabolomics. RT—retention time; VIP—variable importance for projection; MEOX—methoxime; TMS—trimethylsilyl.
| RT (min) | Actual m/z | Metabolite | m/z | Formula | Derivative Type | Derivative Weight/Formula | Predicted m/z | VIP | p (corr) |
|---|
| 3.38 | 236.5340 | Chalcone (HMDB0003066) | 208.2552 | C15H12O | 1 MEOX | C16H15NO | 237.1154 | 1.38 | 0.90 |
| 5.53 | 234.4430 | Melatonin (HMDB0001389) | 232.2783 | C13H16N2O2 | Underivatized | | | 1.16 | 0.65 |
| 16.70 | 340.5060 | 5-Methylcytosine (HMDB0002894) | 125.1286 | C5H7N3O | 3 TMS | C14H31N3OSi3 | 341.6720 | 9.19 | 0.98 |
| 19.80 | 86.1770 | Putrescine (HMDB0001414) | 88.1515 | C4H12N2 | Underivatized | | | 4.42 | −0.52 |
Table 3.
Altered metabolic pathways regulated by BA-12 based on GC–MS metabolomics.
Table 3.
Altered metabolic pathways regulated by BA-12 based on GC–MS metabolomics.
| Pathway | Raw p | Total | Hits |
|---|
| Glutathione metabolism | 0.0617 | 38 | 1 |
| Pyrimidine metabolism | 0.0961 | 60 | 1 |
| Arginine and proline metabolism | 0.1220 | 77 | 1 |
| Tryptophan metabolism | 0.1250 | 79 | 1 |
Table 4.
Differential identified metabolites for discrimination among control and BA-12 groups based on UPLC–TOF-MS metabolomics. KEGG ID—Kyoto Encyclopedia of Genes and Genomes identifier; PC—phosphatidylcholine.
Table 4.
Differential identified metabolites for discrimination among control and BA-12 groups based on UPLC–TOF-MS metabolomics. KEGG ID—Kyoto Encyclopedia of Genes and Genomes identifier; PC—phosphatidylcholine.
| RT (min) | m/z | Metabolite | KEGG ID | Formula | VIP | p (corr) |
|---|
| 3.10 | 154.0373 | Octadecanamide | C13846 | C4H12N2 | 3.76 | −0.83 |
| 15.32 | 496.3440 | 8-Azaxanthine | C04598 | C15H12O | 3.84 | −0.90 |
| 17.18 | 524.3722 | PC (0:0/16:0) (HMDB0010382) | C04230 | C24H50NO7P | 2.31 | −0.89 |
| 20.07 | 284.2902 | LysoPC (18:0) | C04230 | C26H54NO7P | 3.21 | −0.73 |
Table 5.
Altered metabolic pathways regulated by BA-12 based on UPLC–TOF-MS metabolomics. FDR—false discovery rate.
Table 5.
Altered metabolic pathways regulated by BA-12 based on UPLC–TOF-MS metabolomics. FDR—false discovery rate.
| Pathway | Raw p | FDR | Impact |
|---|
| Glycerophospholipid metabolism | 0.000143 | 0.011 | 0.27873 |
Table 6.
The mass data of glutathione (GSH) detected in quail samples by UPLC–QTOF-MS.
Table 6.
The mass data of glutathione (GSH) detected in quail samples by UPLC–QTOF-MS.
| Index | m/z | Chemical Formula | Predicted m/z (M + H)+ | Actual m/z | RT (min) | Integral Area (Mean ± SD) |
|---|
| Control Group | BA-12 (80 μg) Group |
|---|
| GSH | 307.3230 | C10H17N3O6S | 308.1000 | 308.2884 | 19.10 | 58,713 ± 5928 | 33,773 ± 3356 |
| Glycerophospholipid | 170.0578 | C3H7O6P | 171.0578 | 171.0649 | 0.78 | 15,994 ± 1191 | 12,452 ± 1332 |