Derailed Ceramide Metabolism in Atopic Dermatitis (AD): A Causal Starting Point for a Personalized (Basic) Therapy
Abstract
1. Introduction
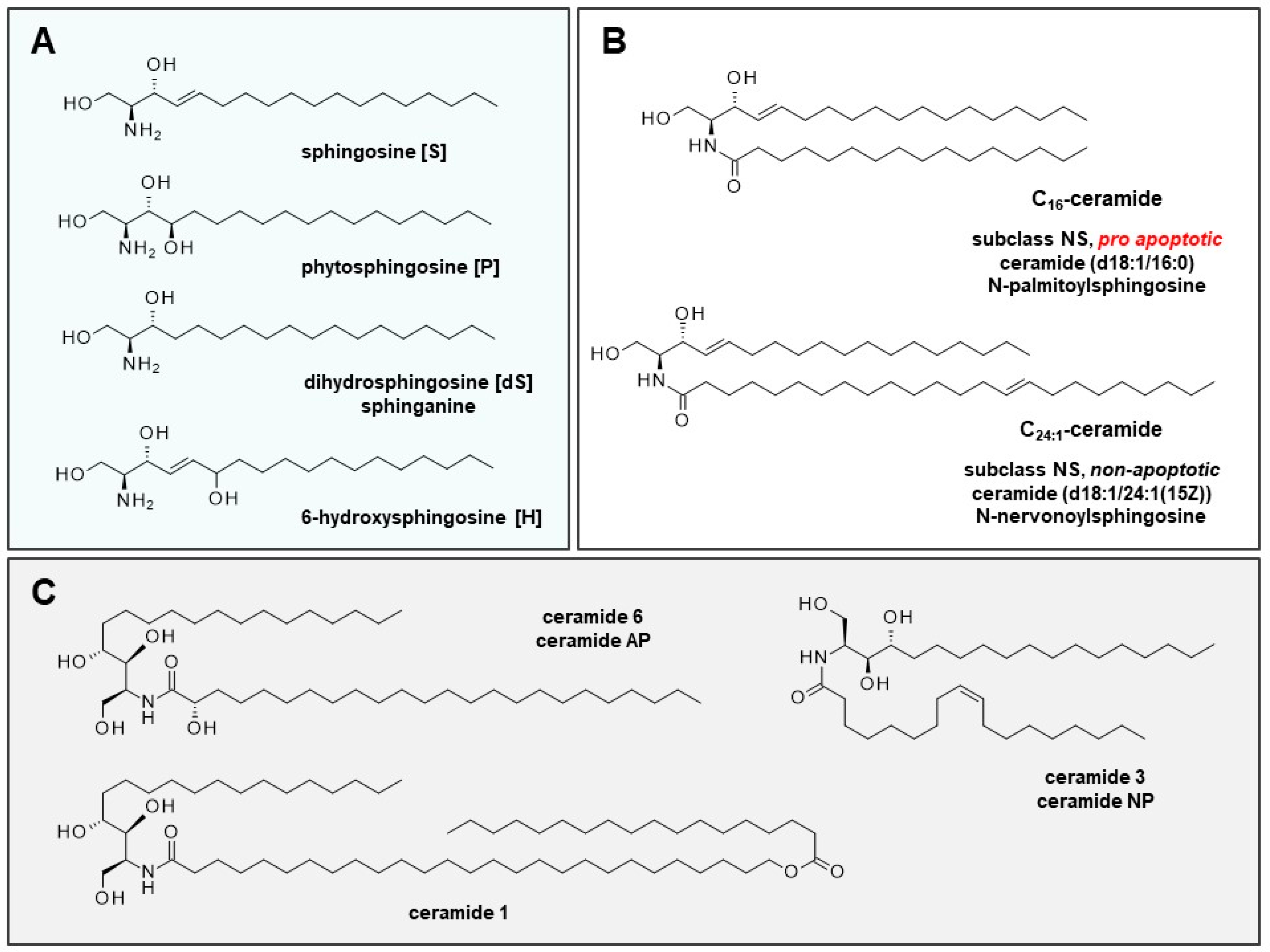
2. Ceramides and Altered Sphingolipid Profile in AD
2.1. Role of Ceramides in Cells and Skin Development
2.2. (Sphingo)Lipid Profile of Sensitive and Inflammatory Skin and Lesions of Patients with AD
2.3. Impaired Maturation and Premature Apoptosis of Keratinocytes
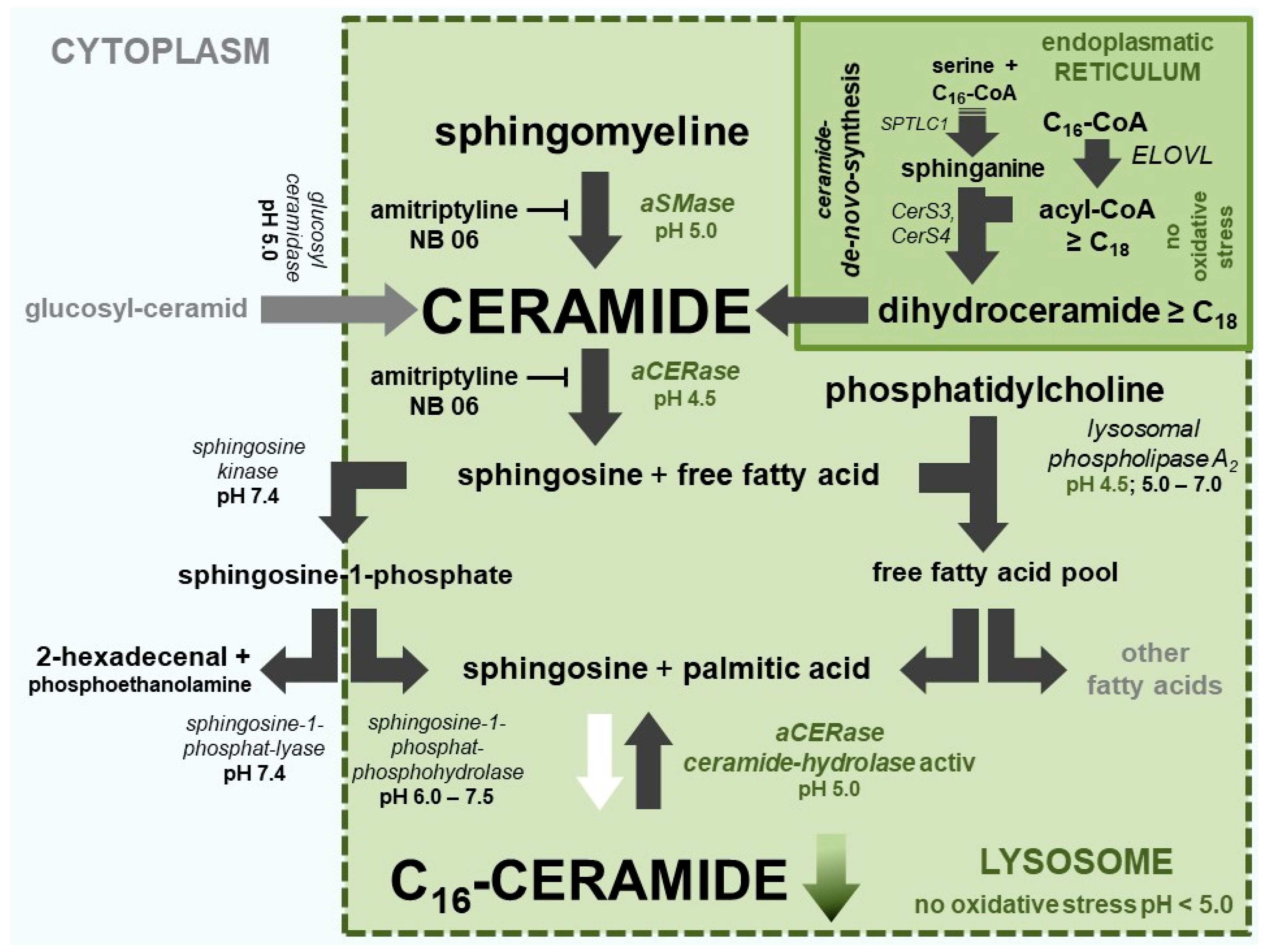
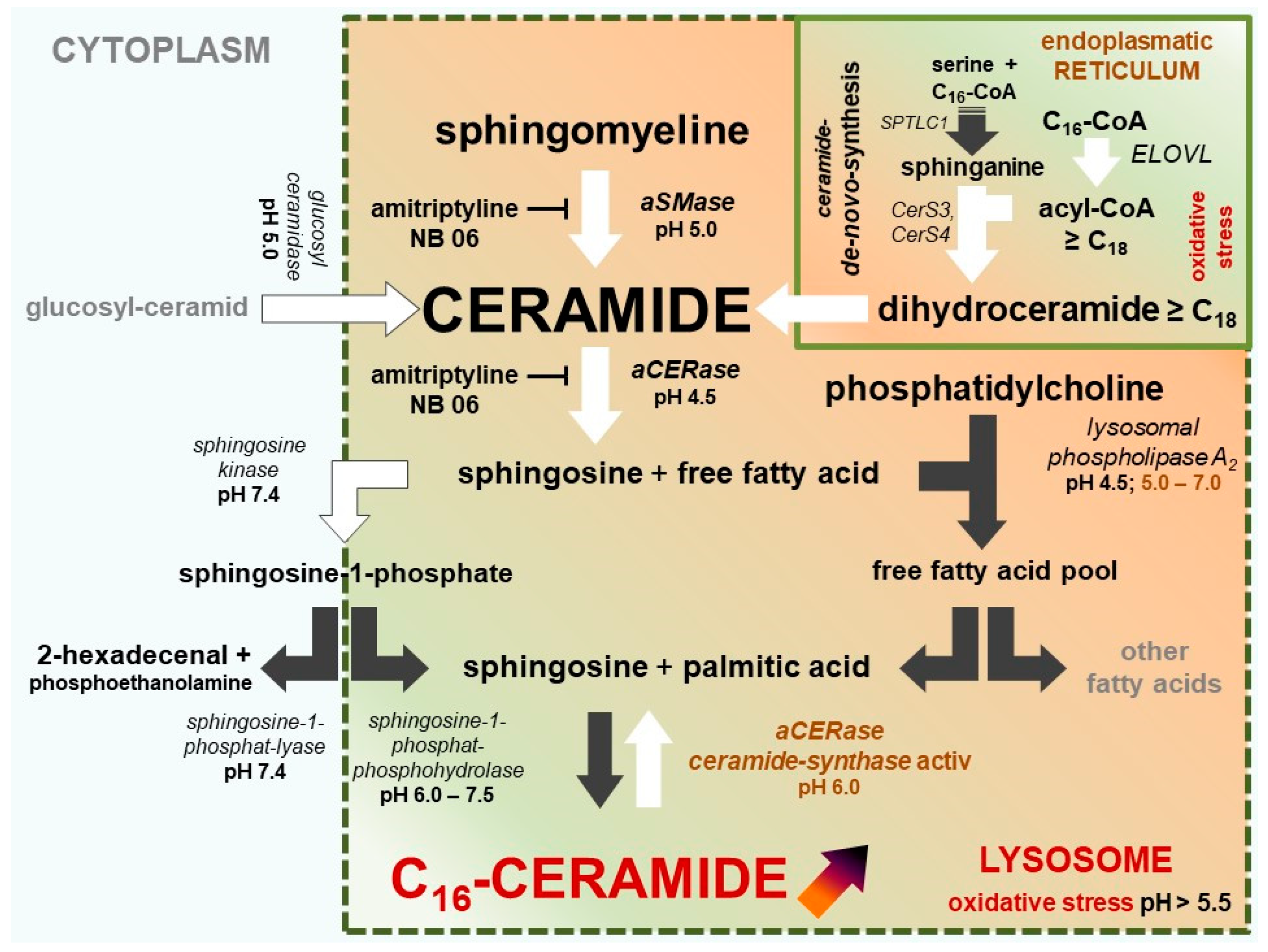
2.4. The Two Different pH-Dependent Enzymatic Activities of aCERase
2.5. aCERase: Shift from Ceramidase to revaCERase Activity in Lysosomes
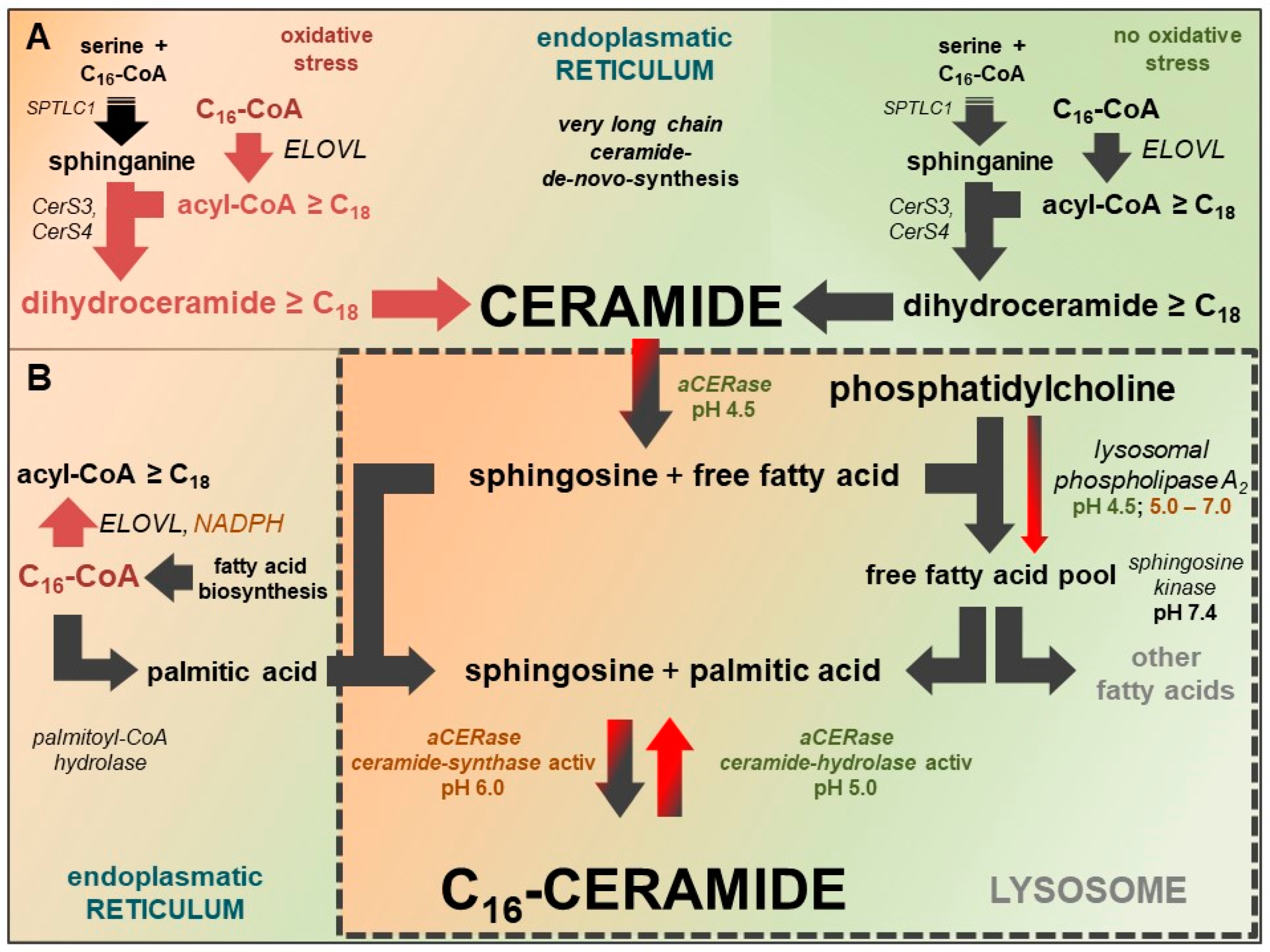
3. New Therapy Concept: Active Adjustment of Ceramide Metabolism
4. Addressed Therapeutic Targets in Keratinocytes
5. Therapy Objectives for Treatment and Prophylaxis
- Inhibition of forming C16-ceramide in lysosomes.
- Stabilization of very long-chain fatty acid-CoA synthesis (≥C18) for very long-chain ceramide de novo synthesis.
- Protecting lysosomal V-ATPase from inactivation (formation of the disulfide bond between Cys 254 and Cys 532) and stabilizing the ceramidase activity of aCERase.
- Modulating expression of AD-relevant gene.
- Enrichment of very long-chain ceramides of de novo synthesis.
6. Implementation of Therapy Objectives
- 2.
- Stabilization of very long-chain fatty acid-CoA synthesis (≥C18) for very long-chain ceramide de novo synthesis and
- 3.
- Protecting lysosomal V-ATPase from inactivation and stabilizing the ceramidase activity of aCERase
7. Therapeutic Concept
8. Personalized Therapy
9. Does the Concept Work?
10. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| aCERase | lysosomal acidic ceramidase |
| acyl-CoA | fatty acid-coenzyme A |
| AD | atopic dermatitis |
| EASI score | eczema area and severity index score |
| ELOVL | very longchain-3-oxoacyl-CoA synthases |
| CerS | ceramide synthases |
| GSH | glutathione |
| revaCERase | reverse ceramide synthase activity of aCERase |
| V-ATPase | vacuolar H+-ATPase |
References
- Williams, H.C. Atopic Dermatitis. N. Engl. J. Med. 2005, 352, 2314–2324. [Google Scholar] [CrossRef] [PubMed]
- Paller, A.S.; Kabashima, K.; Bieber, T. Therapeutic pipeline for atopic dermatitis: End of the drought? J. Allergy Clin. Immunol. 2017, 140, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Werfel, T.; Aberer, W.; Ahrens, F.; Augustin, M.; Biedermann, T.; Diepgen, T.; Fölster-Holst, R.; Gieler, U.; Heratizadeh, A.; Kahle, J.; et al. Leitlinie Neurodermitis [atopisch es Ekzem; atopische Dermatitis] Entwicklungsstufe: S2k. J. Dtsch. Dermatol. Ges. 2016, 14, e1–e75. [Google Scholar] [CrossRef] [PubMed]
- Wollenberg, A.; Barbarot, S.; Bieber, T.; Christen-Zaech, S.; Deleuran, M.; Fink-Wagner, A.; Gieler, U.; Girolomoni, G.; Lau, S.; Muraro, A.; et al. Consensus-based European guidelines for treatment of atopic eczema (atopic dermatitis) in adults and children: Part I. J. Eur. Acad. Dermatol. Venereol. 2018, 32, 657–682. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.U.; D’Ambra, V.; Feldman, S.R. Increasing Adherence with Topical Agents for Atopic Dermatitis. Am. J. Clin. Dermatol. 2017, 18, 323–332. [Google Scholar] [CrossRef] [PubMed]
- van Smeden, J.; Janssens, M.; Gooris, G.S.; Bouwstra, J.A. The important role of stratum corneum lipids for the cutaneous barrier function. Biochim. Biophys. Acta 2014, 1841, 295–313. [Google Scholar] [CrossRef] [PubMed]
- Pettus, B.J.; Chalfant, C.E.; Hannun, Y.A. Ceramide in apoptosis: An overview and current perspectives. Biochim. Biophys. Acta BBA-Mol. Cell Biol. Lipids 2002, 1585, 114–125. [Google Scholar] [CrossRef]
- Gómez-Muñoz, A. Ceramide 1-phosphate/ceramide, a switch between life and death. Biochim. Biophys. Acta BBA-Biomembr. 2006, 1758, 2049–2056. [Google Scholar] [CrossRef]
- Lee, J.Y.; Leonhardt, L.G.; Obeid, L.M. Cell-cycle-dependent changes in ceramide levels preceding retinoblastoma protein dephosphorylation in G2/M. Biochem. J. 1998, 334, 457–461. [Google Scholar] [CrossRef]
- Van Brocklyn, J.R.; Williams, J.B. The control of the balance between ceramide and sphingosine-1-phosphate by sphingosine kinase: Oxidative stress and the seesaw of cell survival and death. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2012, 163, 26–36. [Google Scholar] [CrossRef]
- Thomas, R.L.; Matsko, C.M.; Lotze, M.T.; Amoscato, A.A. Mass Spectrometric Identification of Increased C16 Ceramide Levels During Apoptosis. J. Biol. Chem. 1999, 274, 30580–30588. [Google Scholar] [CrossRef] [PubMed]
- Hoopes, M.I.; Noro, M.G.; Longo, M.L.; Faller, R. Bilayer Structure and Lipid Dynamics in a Model Stratum Corneum with Oleic Acid. J. Phys. Chem. B 2011, 115, 3164–3171. [Google Scholar] [CrossRef] [PubMed]
- Kanoh, H.; Ishitsuka, A.; Fujine, E.; Matsuhaba, S.; Nakamura, M.; Ito, H.; Inagaki, N.; Banno, Y.; Seishima, M. IFN- γ Reduces Epidermal Barrier Function by Affecting Fatty Acid Composition of Ceramide in a Mouse Atopic Dermatitis Model. J. Immunol. Res. 2019, 2019, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Janssens, M.; van Smeden, J.; Gooris, G.S.; Bras, W.; Portale, G.; Caspers, P.J.; Vreeken, R.J.; Hankemeier, T.; Kezic, S.; Wolterbeek, R.; et al. Increase in short-chain ceramides correlates with an altered lipid organization and decreased barrier function in atopic eczema patients. J. Lipid Res. 2012, 53, 2755–2766. [Google Scholar] [CrossRef] [PubMed]
- van Smeden, J.; Janssens, M.; Kaye, E.C.J.; Caspers, P.J.; Lavrijsen, A.P.; Vreeken, R.J.; Bouwstra, J.A. The importance of free fatty acid chain length for the skin barrier function in atopic eczema patients. Exp. Dermatol. 2014, 23, 45–52. [Google Scholar] [CrossRef]
- Ishikawa, J.; Narita, H.; Kondo, N.; Hotta, M.; Takagi, Y.; Masukawa, Y.; Kitahara, T.; Takema, Y.; Koyano, S.; Yamazaki, S.; et al. Changes in the Ceramide Profile of Atopic Dermatitis Patients. J. Investig. Dermatol. 2010, 130, 2511–2514. [Google Scholar] [CrossRef]
- Franco, J.; Ferreira, C.; Paschoal Sobreira, T.J.; Sundberg, J.P.; HogenEsch, H. Profiling of epidermal lipids in a mouse model of dermatitis: Identification of potential biomarkers. PLOS ONE 2018, 13, e0196595. [Google Scholar] [CrossRef]
- Huppertz, B.; Frank, H.-G.; Kaufmann, P. The apoptosis cascade-morphological and immunohistochemical methods for its visualization. Anat. Embryol. (Berl.) 1999, 200, 1–18. [Google Scholar] [CrossRef]
- He, X.; Okino, N.; Dhami, R.; Dagan, A.; Gatt, S.; Schulze, H.; Sandhoff, K.; Schuchman, E.H. Purification and Characterization of Recombinant, Human Acid Ceramidase. Catalytic reactions and interactions with acid sphingomyelinase. J. Biol. Chem. 2003, 278, 32978–32986. [Google Scholar] [CrossRef]
- Okino, N.; He, X.; Gatt, S.; Sandhoff, K.; Ito, M.; Schuchman, E.H. The Reverse Activity of Human Acid Ceramidase. J. Biol. Chem. 2003, 278, 29948–29953. [Google Scholar] [CrossRef]
- Berdyshev, E.; Goleva, E.; Bronova, I.; Dyjack, N.; Rios, C.; Jung, J.; Taylor, P.; Jeong, M.; Hall, C.F.; Richers, B.N.; et al. Lipid abnormalities in atopic skin are driven by type 2 cytokines. JCI Insight 2018, 3, e98006. [Google Scholar] [CrossRef] [PubMed]
- Leidl, K.; Liebisch, G.; Richter, D.; Schmitz, G. Mass spectrometric analysis of lipid species of human circulating blood cells. Biochim. Biophys. Acta BBA-Mol. Cell Biol. Lipids 2008, 1781, 655–664. [Google Scholar]
- Pillay, C.S.; Elliott, E.; Dennison, C. Endolysosomal proteolysis and its regulation. Biochem. J. 2002, 363, 417–429. [Google Scholar] [CrossRef] [PubMed]
- Gille, L.; Nohl, H. The Existence of a Lysosomal Redox Chain and the Role of Ubiquinone. Arch. Biochem. Biophys. 2000, 375, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Nishi, T.; Forgac, M. The vacuolar (H+)-ATPases‒nature’s most versatile proton pumps. Nat. Rev. Mol. Cell Biol. 2002, 3, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Grösch, S.; Schiffmann, S.; Geisslinger, G. Chain length-specific properties of ceramides. Prog. Lipid Res. 2012, 51, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Laviad, E.L.; Albee, L.; Pankova-Kholmyansky, I.; Epstein, S.; Park, H.; Merrill, A.H.; Futerman, A.H. Characterization of Ceramide Synthase 2: tissue distribution, substrate specificity, and inhibition by sphingosine 1-phosphate. J. Biol. Chem. 2008, 283, 5677–5684. [Google Scholar] [CrossRef]
- Tidhar, R.; Zelnik, I.D.; Volpert, G.; Ben-Dor, S.; Kelly, S.; Merrill, A.H.; Futerman, A.H. Eleven residues determine the acyl chain specificity of ceramide synthases. J. Biol. Chem. 2018, 293, 9912–9921. [Google Scholar] [CrossRef]
- Mizutani, Y.; Kihara, A.; Igarashi, Y. Identification of the human sphingolipid C4-hydroxylase, hDES2, and its up-regulation during keratinocyte differentiation. FEBS Lett. 2004, 563, 93–97. [Google Scholar] [CrossRef]
- Naganuma, T.; Kihara, A. Two Modes of Regulation of the Fatty Acid Elongase ELOVL6 by the 3-Ketoacyl-CoA Reductase KAR in the Fatty Acid Elongation Cycle. PLoS ONE 2014, 9, e101823. [Google Scholar] [CrossRef]
- Hassa, P.O.; Haenni, S.S.; Elser, M.; Hottiger, M.O. Nuclear ADP-Ribosylation Reactions in Mammalian Cells: Where Are We Today and Where Are We Going? Microbiol. Mol. Biol. Rev. 2006, 70, 789–829. [Google Scholar] [CrossRef] [PubMed]
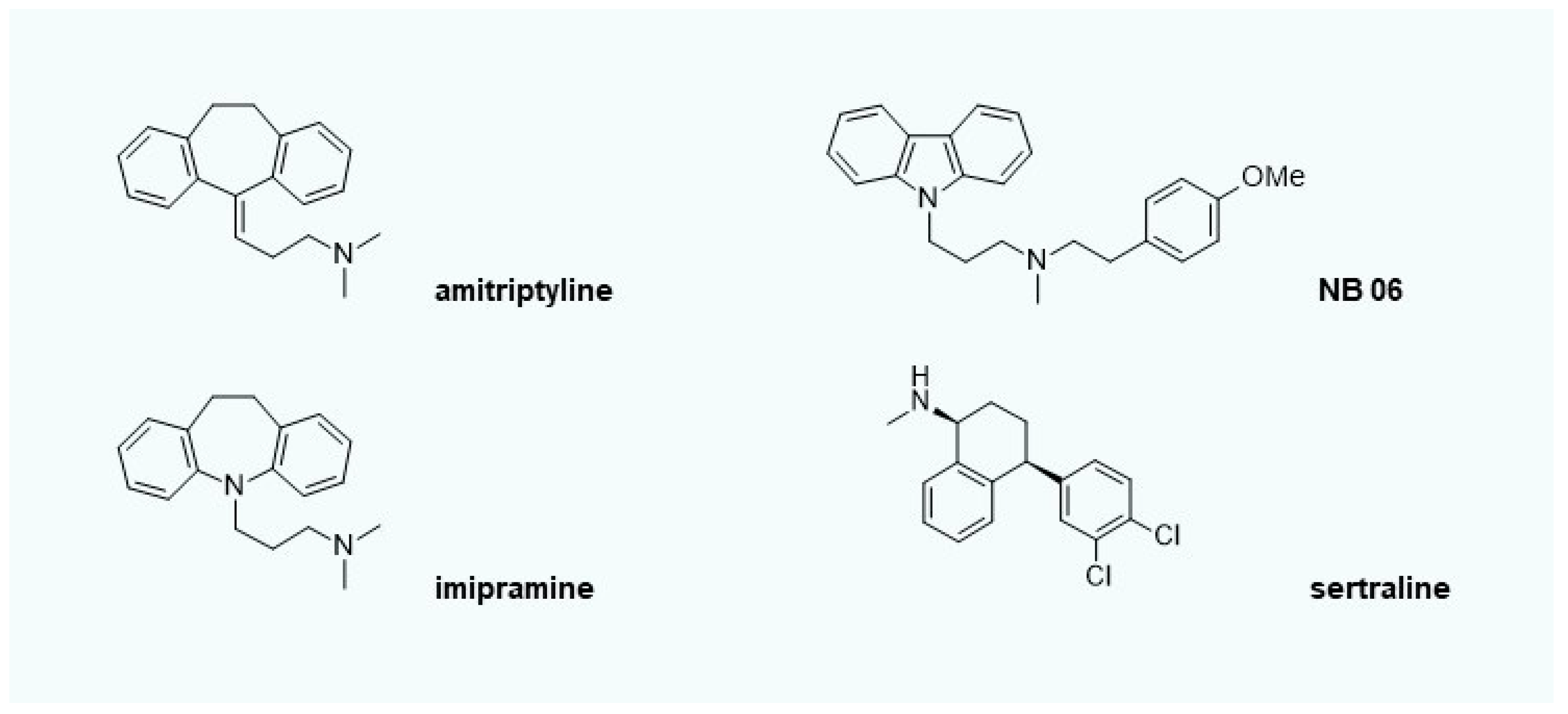
- Blaess, M.; Bibak, N.; Claus, R.A.; Kohl, M.; Bonaterra, G.A.; Kinscherf, R.; Laufer, S.; Deigner, H.-P. NB 06: From a simple lysosomotropic aSMase inhibitor to tools for elucidating the role of lysosomes in signaling apoptosis and LPS-induced inflammation. Eur. J. Med. Chem. 2018, 153, 73–104. [Google Scholar] [CrossRef] [PubMed]
- Blaess, M.; Le, H.; Claus, R.; Kohl, M.; Deigner, H.-P. Stereospecific induction of apoptosis in tumor cells via endogenous C16-ceramide and distinct transcripts. Cell Death Discov. 2015, 1, 15013. [Google Scholar] [CrossRef] [PubMed]
- Deigner, H.-P.; Claus, R.; Bonaterra, G.A.; Gehrke, C.; Bibak, N.; Blaess, M.; Cantz, M.; Metz, J.; Kinscherf, R. Ceramide induces aSMase expression: Implications for oxLDL-induced apoptosis. FASEB J. 2001, 15, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Kornhuber, J.; Tripal, P.; Reichel, M.; Terfloth, L.; Bleich, S.; Wiltfang, J.; Gulbins, E. Identification of New Functional Inhibitors of Acid Sphingomyelinase Using a Structure−Property−Activity Relation Model. J. Med. Chem. 2008, 51, 219–237. [Google Scholar] [CrossRef] [PubMed]
- Kornhuber, J.; Tripal, P.; Reichel, M.; Mühle, C.; Rhein, C.; Muehlbacher, M.; Groemer, T.W.; Gulbins, E. Functional Inhibitors of Acid Sphingomyelinase (FIASMAs): A Novel Pharmacological Group of Drugs with Broad Clinical Applications. Cell. Physiol. Biochem. 2010, 26, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Thédenat, B.; Loche, F.; Albes, B.; Marguery, M.C.; Bazex, J. Acute Generalized Exanthematous Pustulosis with Photodistribution Pattern Induced by Sertraline. Dermatology 2001, 203, 87–88. [Google Scholar] [CrossRef] [PubMed]
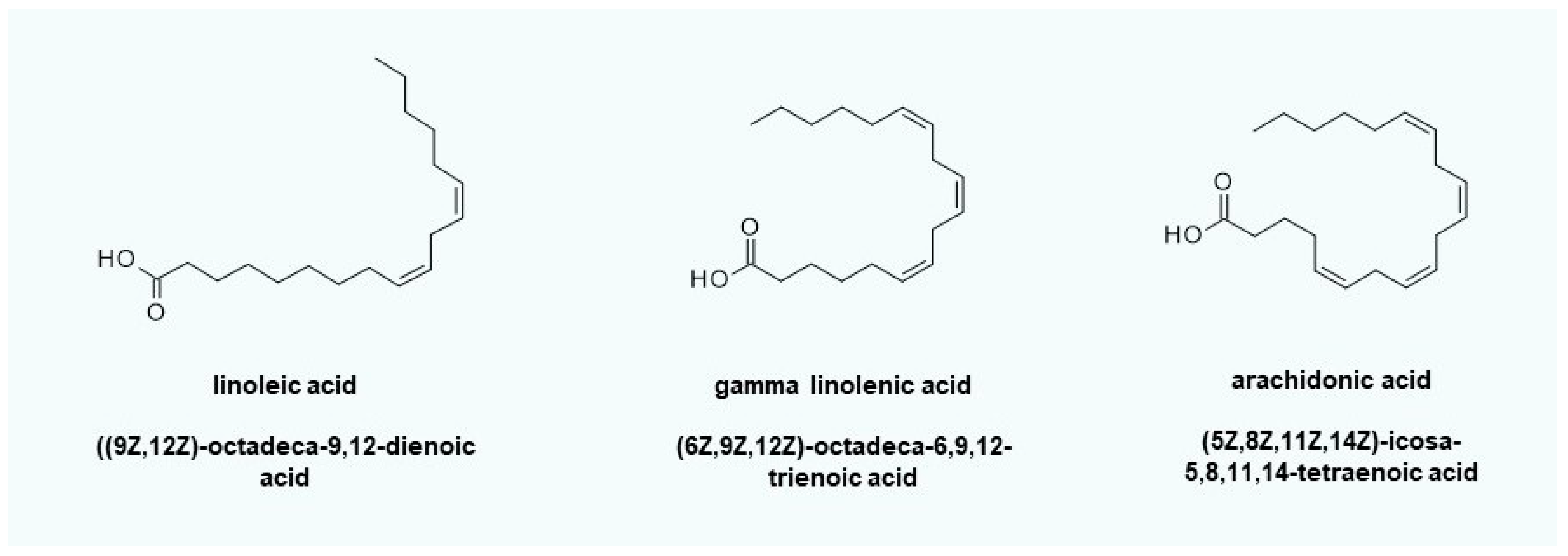
- Blaess, M.; Wenzel, F.; Csuk, R.; Deigner, H.-P. Topical off-label use of amitriptyline and linoleic acid to restore ceramide rheostat in atopic dermatitis lesions-a case report. Pharm. 2019, 74. [Google Scholar] [CrossRef]
- Guttman-Yassky, E.; Krueger, J.G.; Lebwohl, M.G. Systemic immune mechanisms in atopic dermatitis and psoriasis with implications for treatment. Exp. Dermatol. 2018, 27, 409–417. [Google Scholar] [CrossRef] [PubMed]

| Ceramides | Ceramides + Free Fatty Acids | Free Fatty Acids |
|---|---|---|
| Short chain ceramides, in particular C16-ceramide [NS], are elevated in inflammatory skin and lesions | Free fatty acids and ceramides with very long chains (≥C24) are significantly reduced. | Short-chain free fatty acids palmitic acid (C16:0) and stearic acid (C18: 0) are elevated. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blaess, M.; Deigner, H.-P. Derailed Ceramide Metabolism in Atopic Dermatitis (AD): A Causal Starting Point for a Personalized (Basic) Therapy. Int. J. Mol. Sci. 2019, 20, 3967. https://doi.org/10.3390/ijms20163967
Blaess M, Deigner H-P. Derailed Ceramide Metabolism in Atopic Dermatitis (AD): A Causal Starting Point for a Personalized (Basic) Therapy. International Journal of Molecular Sciences. 2019; 20(16):3967. https://doi.org/10.3390/ijms20163967
Chicago/Turabian StyleBlaess, Markus, and Hans-Peter Deigner. 2019. "Derailed Ceramide Metabolism in Atopic Dermatitis (AD): A Causal Starting Point for a Personalized (Basic) Therapy" International Journal of Molecular Sciences 20, no. 16: 3967. https://doi.org/10.3390/ijms20163967
APA StyleBlaess, M., & Deigner, H.-P. (2019). Derailed Ceramide Metabolism in Atopic Dermatitis (AD): A Causal Starting Point for a Personalized (Basic) Therapy. International Journal of Molecular Sciences, 20(16), 3967. https://doi.org/10.3390/ijms20163967





