A Hydroquinone-Based Derivative Elicits Apoptosis and Autophagy via Activating a ROS-Dependent Unfolded Protein Response in Human Glioblastoma
Abstract
1. Introduction
2. Results
2.1. Effects of EA-100C Red on the Cell Proliferation of GBM Cells
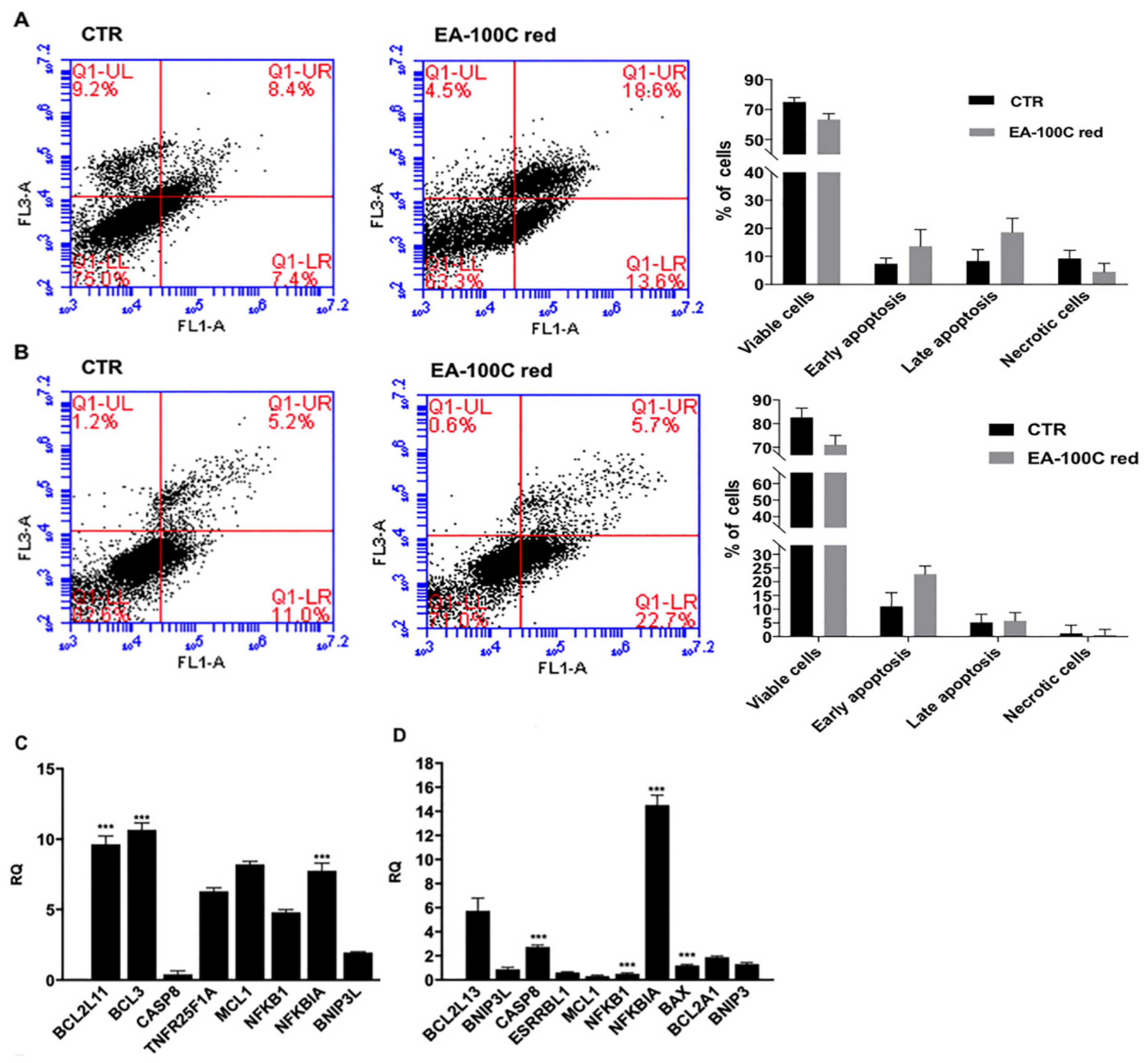
2.2. Effects of EA-100C Red on Apoptosis
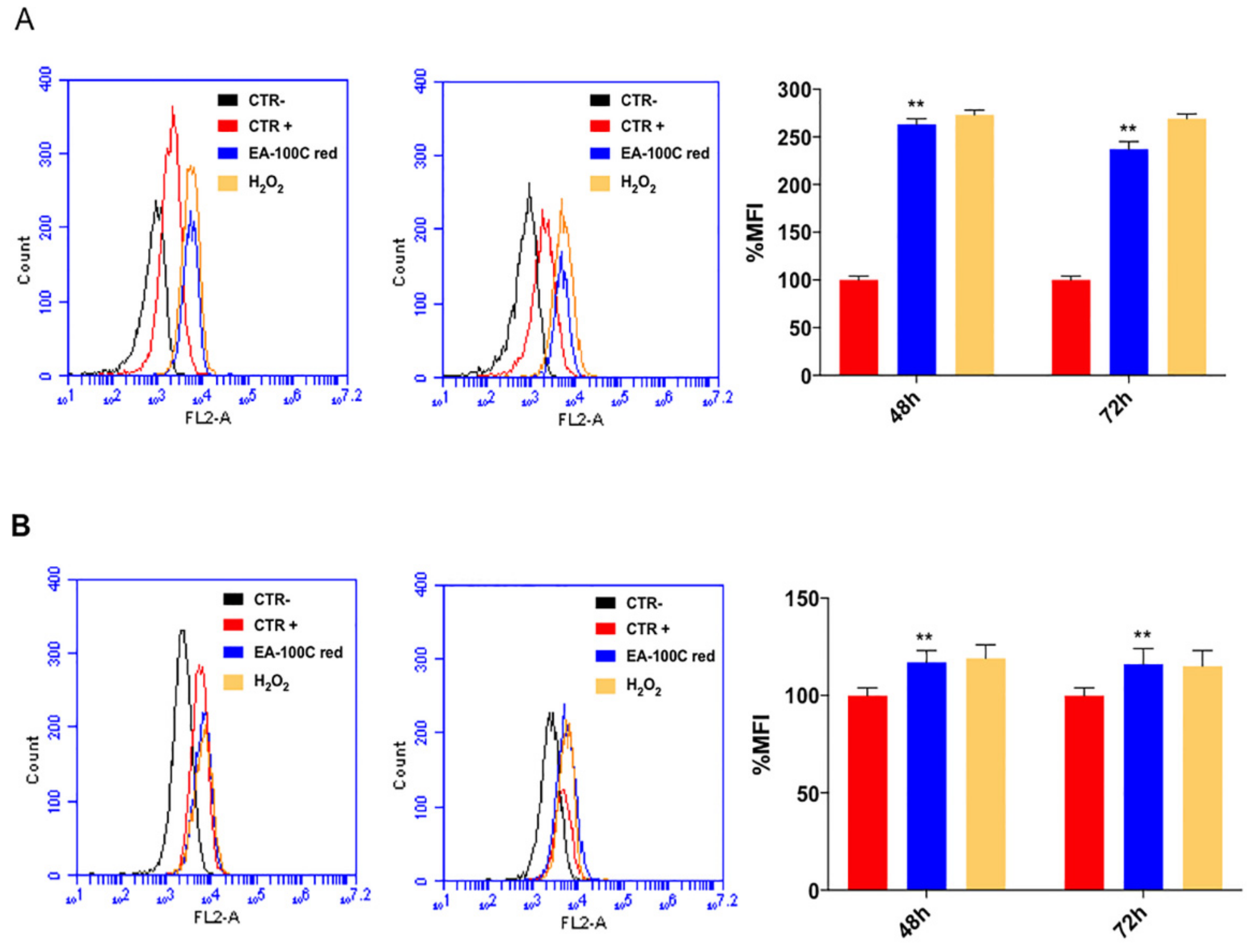
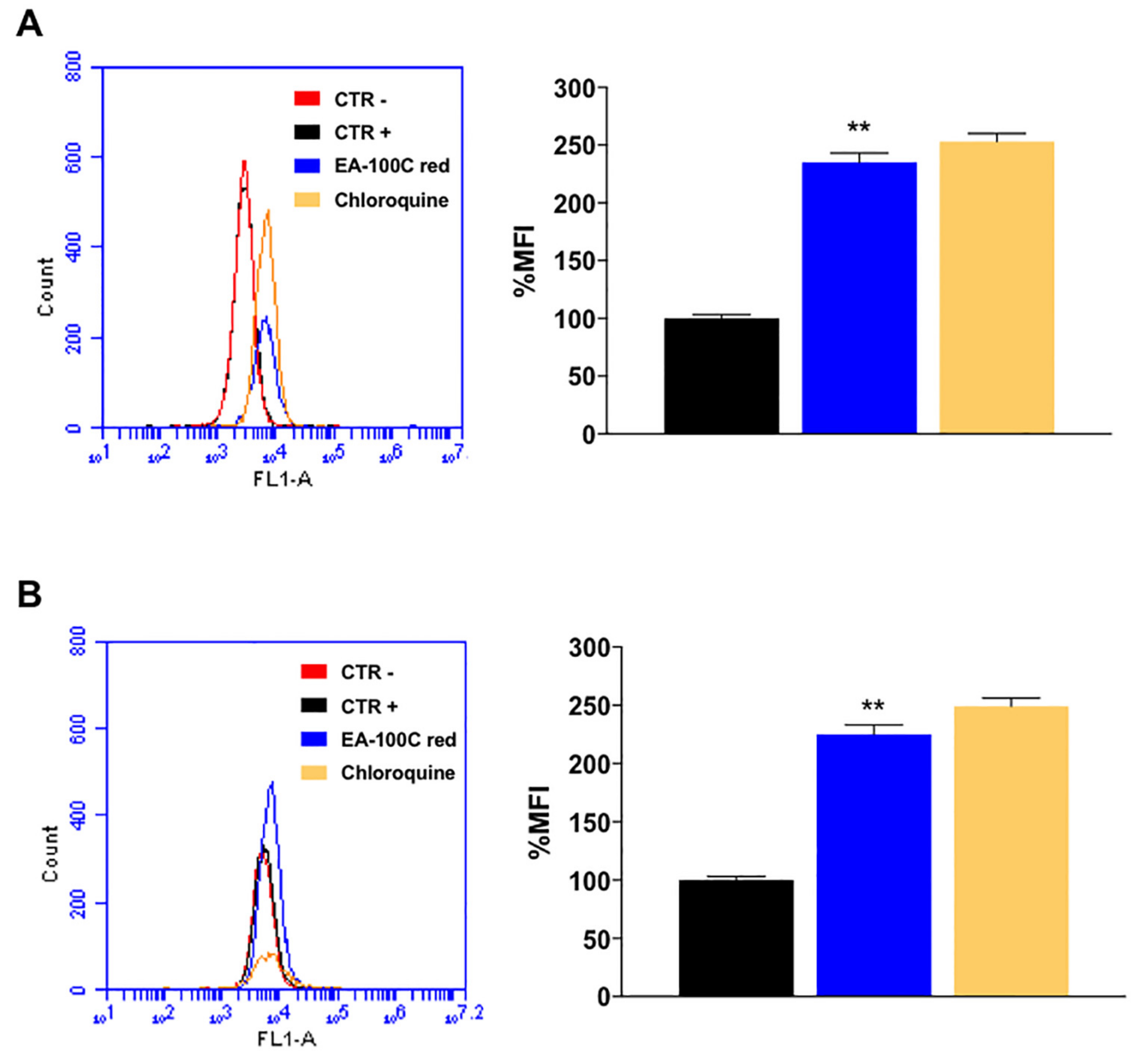
2.3. Effects of EA-100C Red on Mitochondrial Membrane Potential and Autophagy
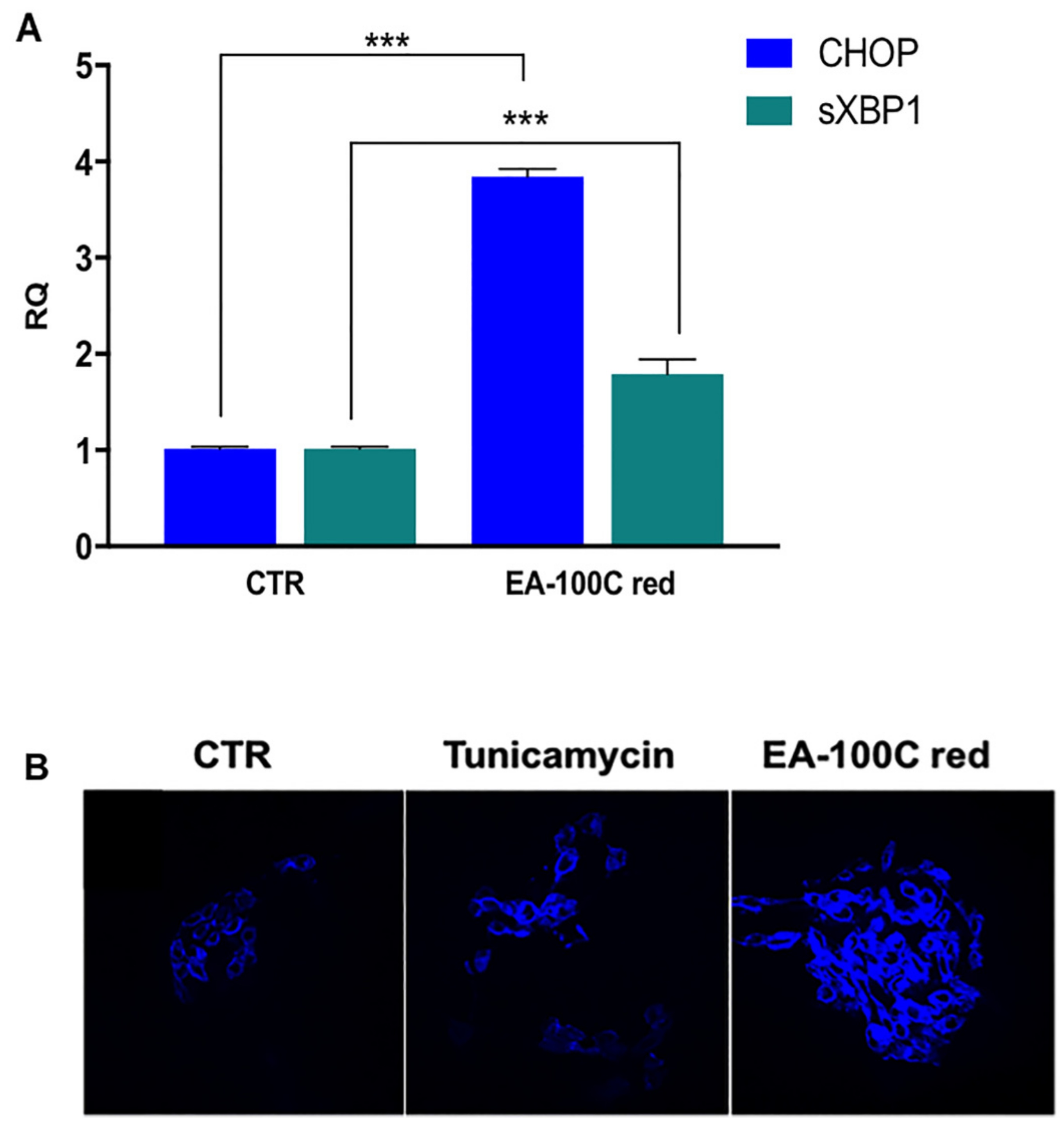
2.4. Effect of EA-100C Red on ER-Stress
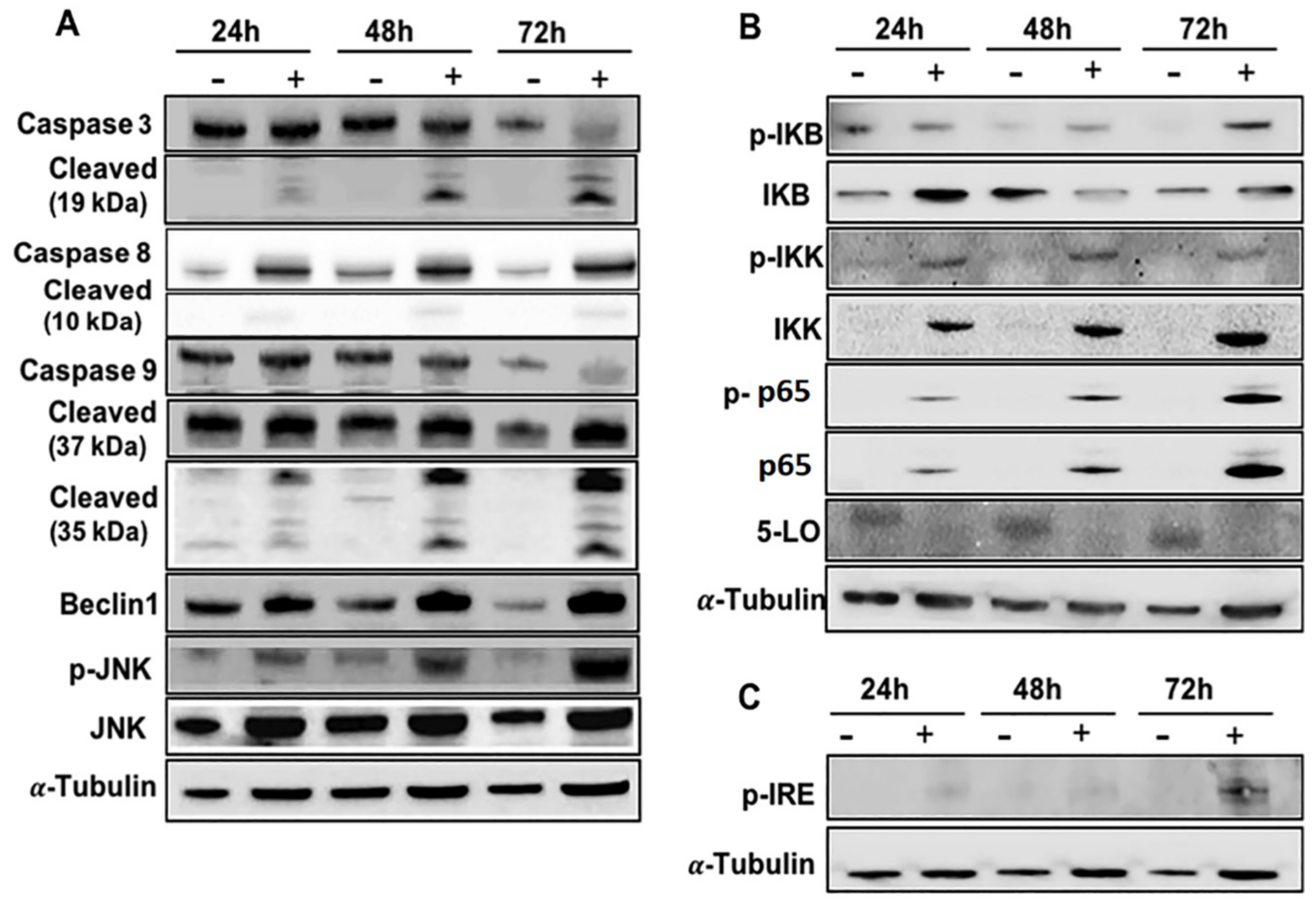
2.5. Effects of EA-100C Red on Signal Transduction Pathways
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Chemistry
4.3. Cell Culture
4.4. Cell Viability Assay
4.5. Analysis of Apoptosis by FACS
4.6. FACS Analysis of Mitochondrial Potential
4.7. Flow Cytometric Analysis of Autophagy
4.8. Taqman Human Apoptosis Array-Real-Time-PCR
4.9. Immunofluorescence with ER Tracker
4.10. Western Blot Analysis
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| GBM | Glioblastoma |
| TMZ | Temozolomide |
| FTM | Fotemustine |
| CNS | Central nervous system |
| BBB | Blood–brain barrier |
| 5-LO | 5-Lipoxygenase |
| MMP | Mitocondrial membrane potential |
| ER | Endoplasmic reticulum |
| UPR | Unfolded protein response |
References
- Huncharek, S.; Muscat, J. Treatment of recurrent high grade astrocytoma: Results of a systematic review of 1415 patients. Anticancer Res. 1998, 18, 1303–1311. [Google Scholar] [PubMed]
- Stupp, R.; Mason, W.P.; van denBent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Nieder, C.; Adam, M.; Molls, M.; Grosu, A.L. Therapeutic options for recurrent high-grade glioma in adult patients: Recent advances. Crit. Rev. Oncol. Hematol. 2006, 60, 181–193. [Google Scholar] [CrossRef] [PubMed]
- De Rossi, A.; Rossi, L.; Laudisi, A.; Sini, V.; Toppo, L.; Marchesi, F.; Tortorelli, G.; Leti, M.; Turriziani, M.; Aquino, A.; et al. Focus on Fotemustine. J. Exp. Clin. Cancer Res. 2006, 25, 461–468. [Google Scholar] [PubMed]
- Malhaire, J.P.; Lucas, B.; Simon, H.; Person, H.; Dam-Hieu, P.; Labat, J.P. Fotemustine (Muphoran) in 22 patients with relapses of high-grade cerebral gliomas. Bull Cancer 1999, 86, 289–294. [Google Scholar]
- Meulemans, A.; Giroux, B.; Hannoun, P.; Robine, D.; Henzel, D. Comparative diffusion study of two nitrosoureas: Carmustine and fotemustine in normal rat brain, human and rat brain biopsies. Chemotherapy 1991, 37, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Levin, V.A. Relationship of octanol/water partition coefficient and molecular weight to rat brain capillary permeability. J. Med. Chem. 1980, 23, 682–684. [Google Scholar] [CrossRef]
- Fischel, J.L.; Formento, P.; Etienne, M.C.; Gioanni, J.; Frenay, M.; Deloffre, P.; Bizzari, J.P.; Milano, G. In vitro chemosensitivity testing of Fotemustine (S 10036), a new antitumor nitrosourea. Cancer Chemother. Pharmacol. 1990, 25, 337–341. [Google Scholar] [CrossRef]
- Boado, R.J.; Pardridge, W.M.; Vinters, H.V.; Black, K.L. Differential expression of arachidonate 5-lipoxygenase transcripts in human brain tumors: Evidence for the expression of a multitranscript family. Proc. Natl. Acad. Sci. USA 1992, 89, 9044–9048. [Google Scholar] [CrossRef]
- Werz, O.; Steinhilber, D. Therapeutic options for 5-lipoxygenase inhibitors. Pharmacol. Ther. 2006, 112, 701–718. [Google Scholar] [CrossRef]
- Pergola, C.; Werz, O. 5-Lipoxygenase inhibitors: A review of recent developments and patents. Expert. Opin. Ther. Pat. 2010, 20, 355–375. [Google Scholar] [CrossRef] [PubMed]
- Petronzi, C.; Filosa, R.; Peduto, A.; Monti, M.C.; Margarucci, L.; Massa, A.; Ercolino, S.F.; Bizzarro, V.; Parente, L.; Riccio, R.; et al. Structure-based design, synthesis and preliminary anti-inflammatory activity of bolinaquinone analogues. Eur. J. Med. Chem. 2011, 46, 488–496. [Google Scholar] [CrossRef]
- Filosa, R.; Peduto, A.; Aparoy, P.; Schaible, A.M.; Luderer, S.; Krauth, V.; Petronzi, C.; Massa, A.; de Rosa, M.; Reddanna, P.; et al. Discovery and biologicalevaluation of novel 1,4-benzoquinone and relatedresorcinolderivativesthatinhibit 5-lipoxygenase. Eur. J. Med. Chem. 2013, 67, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Petronzi, C.; Festa, M.; Peduto, A.; Castellano, M.; Marinello, J.; Massa, A.; Capasso, A.; Capranico, G.; La Gatta, A.; De Rosa, M.; et al. Cyclohexa-2,5-diene-1,4-dione-based antiproliferative agents: Design, synthesis, and cytotoxic evaluation. J. Exp. Clin. Cancer Res. 2013, 32, 24. [Google Scholar] [CrossRef] [PubMed]
- Peduto, A.; Krauth, V.; Collarile, S.; Dehm, F.; Ambruosi, M.; Belardo, C.; Guida, F.; Massa, A.; Esposito, V.; Maione, S.; et al. Exploring the role of chloro and methyl substitutions in 2-phenylthiomethyl-benzoindole derivatives for 5-LOX enzyme inhibition. Eur. J. Med. Chem. 2016, 108, 466–475. [Google Scholar] [CrossRef] [PubMed]
- Schaible, A.M.; Filosa, R.; Temml, V.; Krauth, V.; Matteis, M.; Peduto, A.; Bruno, F.; Luderer, S.; Roviezzo, F.; Di Mola, A.; et al. Elucidation of the molecular mechanism and the efficacy in vivo of a novel 1,4-benzoquinone that inhibits 5-lipoxygenase. Br. J. Pharmacol. 2014, 171, 2399–2412. [Google Scholar] [CrossRef] [PubMed]
- Peduto, A.; Bruno, F.; Dehm, F.; Krauth, V.; de Caprariis, P.; Weinigel, C.; Barz, D.; Massa, A.; De Rosa, M.; Werz, O.; et al. Further studies on ethyl 5-hydroxy-indole-3-carboxylate scaffold: Design, synthesis and evaluation of 2-phenylthiomethyl-indole derivatives as efficient inhibitors of human 5-lipoxygenase. Eur. J. Med. Chem. 2014, 81, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Filosa, R.; Peduto, A.; Schaible, A.M.; Krauth, V.; Weinigel, C.; Barz, D.; Petronzi, C.; Bruno, F.; Roviezzo, F.; Spaziano, G.; et al. Novelseries of benzoquinones with high potencyagainst 5-lipoxygenase in human polymorphonuclearleukocytes. Eur. J. Med. Chem. 2015, 94, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Schaible, A.M.; Filosa, R.; Krauth, V.; Temml, V.; Pace, S.; Garscha, U.; Liening, S.; Weinigel, C.; Rummler, S.; Schieferdecker, S.; et al. The 5-lipoxygenase inhibitor RF-22c potently suppresses leukotriene biosynthesis in cellulo and blocks bronchoconstriction and inflammation in vivo. Biochem. Pharmacol. 2016, 112, 60–71. [Google Scholar] [CrossRef]
- Bruno, F.; Errico, S.; Pace, S.; Nawrozkij, M.B.; Mkrtchyan, A.S.; Guida, F.; Maisto, R.; Olgaç, A.; D’Amico, M.; Maione, S.; et al. Structural insight into the optimization of ethyl 5-hydroxybenzo[g]indol-3-carboxylates and their bioisosteric analogues as 5-LO/m-PGES-1 dual inhibitors able to suppress inflammation. Eur. J. Med. Chem. 2018, 155, 946–960. [Google Scholar] [CrossRef] [PubMed]
- Peduto, A.; Pagano, B.; Petronzi, C.; Massa, A.; Esposito, V.; Virgilio, A.; Paduano, F.; Trapasso, F.; Fiorito, F.; Florio, S.; et al. Design, synthesis, biophysical and biological studies of trisubstituted naphthalimides as G-quadruplexligands. Bioorg. Med. Chem. 2011, 19, 6419–6429. [Google Scholar] [CrossRef] [PubMed]
- Filosa, R.; Peduto, A.; Micco, S.D.; Caprariis, P.; Festa, M.; Petrella, A.; Capranico, G.; Bifulco, G. Molecular modelling studies, synthesis and biological activity of a series of novel bisnaphthalimides and their development as new DNA topoisomerase II inhibitors. Bioorg. Med. Chem. 2009, 17, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Peduto, A.; More, V.; de Caprariis, P.; Festa, M.; Capasso, A.; Piacente, S.; De Martino, L.; De Feo, V.; Filosa, R. Synthesis and cytotoxic activity of new beta-carboline derivatives. Mini Rev. Med. Chem. 2011, 11, 486–491. [Google Scholar] [CrossRef] [PubMed]
- Schaible, A.M.; Traber, H.; Temml, V.; Noha, S.M.; Filosa, R.; Peduto, A.; Weinigel, C.; Barz, D.; Schuster, D.; Werz, O. Potent inhibition of human 5-lipoxygenase and microsomal prostaglandin E(2) synthase-1 by the anti-carcinogenic and anti-inflammatory agent embelin. Biochem. Pharmacol. 2013, 86, 476–486. [Google Scholar] [CrossRef] [PubMed]
- Zappavigna, S.; Scuotto, M.; Cossu, A.M.; Ingrosso, D.; De Rosa, M.; Schiraldi, C.; Filosa, R.; Caraglia, M. The 1,4 benzoquinone-featured 5-lipoxygenase inhibitor RF-Id induces apoptotic death through downregulation of IAPs in human glioblastoma cells. J. Exp. Clin. Cancer Res. 2016, 35, 167. [Google Scholar] [CrossRef] [PubMed]
- Peduto, A.; Scuotto, M.; Krauth, V.; Roviezzo, F.; Rossi, A.; Temml, V.; Esposito, V.; Stuppner, H.; Schuster, D.; D’Agostino, B.; et al. Optimization of benzoquinone and hydroquinone derivatives as potent inhibitors of human 5-lipoxygenase. Eur. J. Med. Chem. 2017, 127, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Keegstra, E.M.D.; Huisman, B.H.; Paardekooper, E.M.; Hoogesteger, F.J.; Zwikker, J.W.; Jenneskens, L.W.; Kooijman, H.; Schouten, A.; Veldman, N.; Spek, A.L. 2,3,5,6-tetraalkoxy-1,4 benzoquinones and structurally related tetraalkoxy-benzene derivatives: Synthesis, properties and solid state packing motifs. J. Chem. Soc. Perk. Trans. 2 1996. [Google Scholar] [CrossRef]
- Kozubek, A.; Zarnowski, R.; Stasiuk, M.; Gubernator, J. Natural amphiphilic phenols as bioactive compounds. Cell. Mol. Biol. Lett. 2001, 6, 351–355. [Google Scholar]
- Chaturvedula, V.S.; Schilling, J.K.; Kingston, D.G. New cytotoxic coumarins and prenylated benzophenone derivatives from the bark of Ochrocarpos punctatus from the Madagascar rainforest. J. Nat. Prod. 2002, 65, 965–972. [Google Scholar] [CrossRef]
- Kozubek, A.; Tyman, J.H. Resorcinolic Lipids, the Natural Non-isoprenoid Phenolic Amphiphiles and Their Biological Activity. Chem. Rev. 1999, 99, 1–26. [Google Scholar] [CrossRef]
- Arisawa, M.; Ohmura, K.; Kobayashi, A.; Morita, N. A cytotoxic constituent of Lysimachiaja ponica THUNB. (Primulaceae) and the structure-activity relationships of related compounds. Chem. Pharm. Bull. (Tokyo) 1989, 37, 2431–2434. [Google Scholar] [CrossRef] [PubMed]
- Lopez, P.; Ruffa, M.J.; Cavallaro, L.; Campos, R.; Martino, V.; Ferraro, G. 1,3-dihydroxy-5-(tridec-4’,7’-dienyl)benzene: A new cytotoxic compound from Lithraea molleoides. Phytomedicine 2005, 12, 108–111. [Google Scholar] [CrossRef] [PubMed]
- Loo, G. Redox-sensitive mechanisms of phytochemical-mediated inhibition of cancer cell proliferation (review). J. Nutr. Biochem. 2003, 14, 64–73. [Google Scholar] [CrossRef]
- Calvisi, D.F.; Pinna, F.; Pellegrino, R.; Sanna, V.; Sini, M.; Daino, L.; Simile, M.M.; De Miglio, M.R.; Frau, M.; Tomasi, M.L.; et al. Ras-driven proliferation and apoptosis signaling during ratl iver carcinogenesisis under genetic control. Int. J. Cancer 2008, 123, 2057–2064. [Google Scholar] [CrossRef] [PubMed]
- Calvisi, D.F.; Ladu, S.; Gorden, A.; Farina, M.; Conner, E.A.; Lee, J.S.; Factor, V.M.; Thorgeirsson, S.S. Ubiquitous activation of Ras and Jak/Stat pathways in human HCC. Gastroenterology 2006, 130, 1117–1128. [Google Scholar] [CrossRef] [PubMed]
- Laurent-Puig, P.; Legoix, P.; Bluteau, O.; Belghiti, J.; Franco, D.; Binot, F.; Monges, G.; Thomas, G.; Bioulac-Sage, P.; Zucman-Rossi, J. Genetic alterations associated with hepatocellular carcinomas define distinct pathways of hepatocarcinogenesis. Gastroenterology 2001, 120, 1763–1773. [Google Scholar] [CrossRef] [PubMed]
- Szegezdi, E.; Logue, S.E.; Gorman, A.M.; Samali, A. Mediators of endoplasmic reticulum stress-induced apoptosis. EMBO Rep. 2006, 7, 880–885. [Google Scholar] [CrossRef]
- Patil, V.; Pal, J.; Somasundaram, K. Elucidating the cancer-specific genetic alteration spectrum of glioblastoma derived cell lines from whole exome and RNA sequencing. Oncotarget 2015, 6, 43452–43471. [Google Scholar] [CrossRef]
- Zhu, Z.; Du, S.; Du, Y.; Ren, J.; Ying, G.; Yan, Z. Glutathione reductase mediates drug resistance in glioblastoma cells by regulating redox homeostasis. J. Neurochem. 2018, 144, 93–104. [Google Scholar] [CrossRef]
- Chaudhary, P.M.; Eby, M.T.; Jasmin, A.; Kumar, A.; Liu, L.; Hood, L. Activation of the NF-kappaB pathway by caspase 8 and its homologs. Oncogene 2000, 19, 4451–4460. [Google Scholar] [CrossRef]
- Chong, W.C.; Shastri, M.D.; Eri, R. Endoplasmic Reticulum Stress and Oxidative Stress: A Vicious Nexus Implicated in Bowel Disease Pathophysiology. Int. J. Mol. Sci. 2017, 18, 771. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Xu-Monette, Z.Y.; Ok, C.Y.; Tzankov, A.; Manyam, G.C.; Sun, R.; Visco, C.; Zhang, M.; Montes-Moreno, S.; Dybkaer, K.; et al. Prognostic impact of c-Rel nuclear expression and REL amplification and crosstalk between c-Rel and the p53 pathway in diffuse large B-cell lymphoma. Oncotarget 2015, 6, 23157–23180. [Google Scholar] [CrossRef] [PubMed]
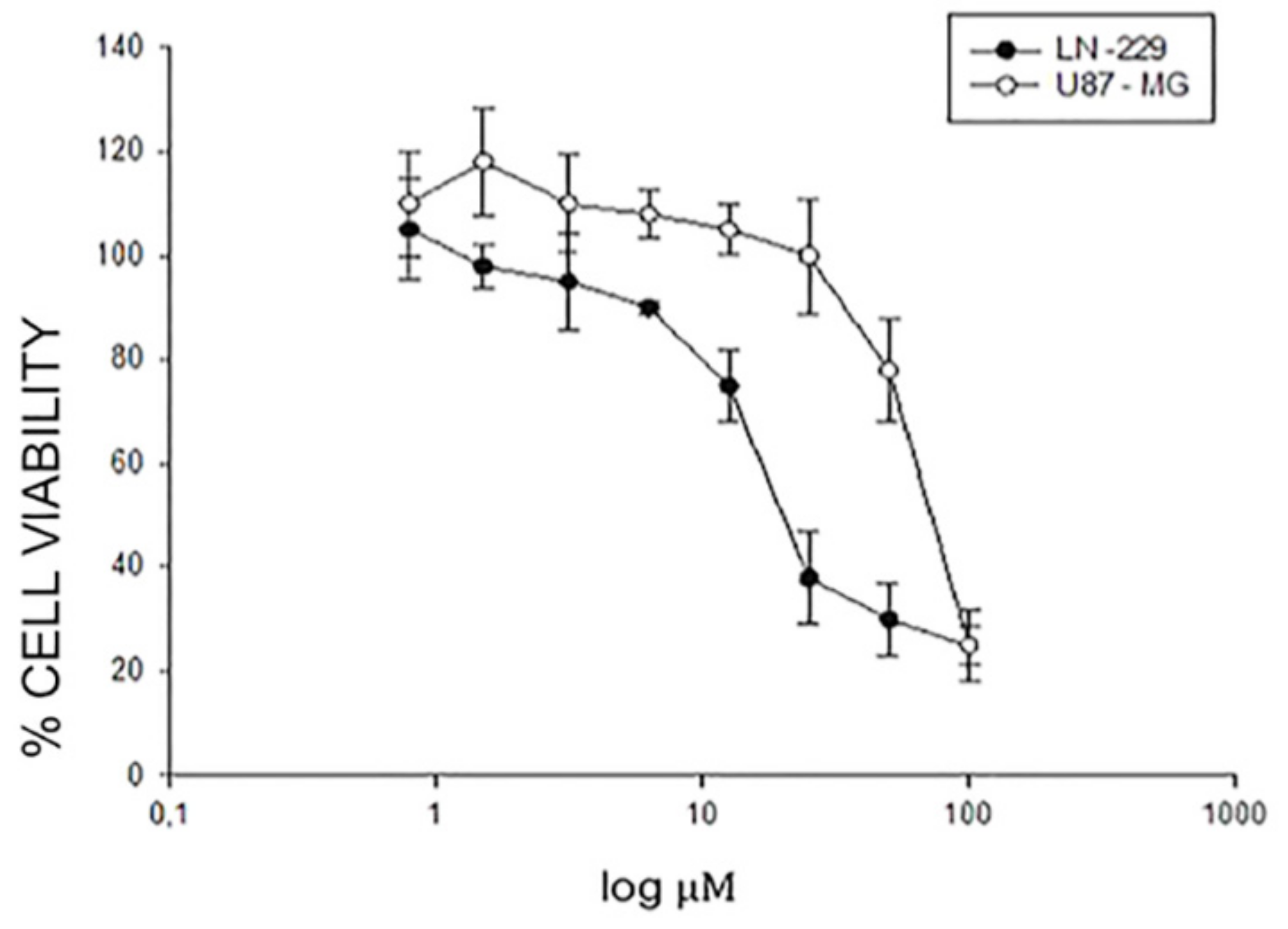
| Name | Structure | U87MG | LN229 | P Value |
|---|---|---|---|---|
| EA-100C Red | 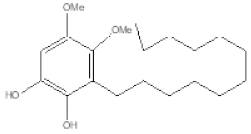 | IC50 [µM] 75.5 ± 0.04 | IC50 [µM] 21 ± 0.05 | ** p ≤ 0.01 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zappavigna, S.; Cossu, A.M.; Abate, M.; Misso, G.; Lombardi, A.; Caraglia, M.; Filosa, R. A Hydroquinone-Based Derivative Elicits Apoptosis and Autophagy via Activating a ROS-Dependent Unfolded Protein Response in Human Glioblastoma. Int. J. Mol. Sci. 2019, 20, 3836. https://doi.org/10.3390/ijms20153836
Zappavigna S, Cossu AM, Abate M, Misso G, Lombardi A, Caraglia M, Filosa R. A Hydroquinone-Based Derivative Elicits Apoptosis and Autophagy via Activating a ROS-Dependent Unfolded Protein Response in Human Glioblastoma. International Journal of Molecular Sciences. 2019; 20(15):3836. https://doi.org/10.3390/ijms20153836
Chicago/Turabian StyleZappavigna, Silvia, Alessia Maria Cossu, Marianna Abate, Gabriella Misso, Angela Lombardi, Michele Caraglia, and Rosanna Filosa. 2019. "A Hydroquinone-Based Derivative Elicits Apoptosis and Autophagy via Activating a ROS-Dependent Unfolded Protein Response in Human Glioblastoma" International Journal of Molecular Sciences 20, no. 15: 3836. https://doi.org/10.3390/ijms20153836
APA StyleZappavigna, S., Cossu, A. M., Abate, M., Misso, G., Lombardi, A., Caraglia, M., & Filosa, R. (2019). A Hydroquinone-Based Derivative Elicits Apoptosis and Autophagy via Activating a ROS-Dependent Unfolded Protein Response in Human Glioblastoma. International Journal of Molecular Sciences, 20(15), 3836. https://doi.org/10.3390/ijms20153836






