Current Knowledge of the Potential Links between Inflammation and Prostate Cancer
Abstract
1. Introduction
2. Acquisition of Evidence
3. Findings
3.1. What Is the Prevalence of Prostate Inflammation in the General Population and PCa Patients?
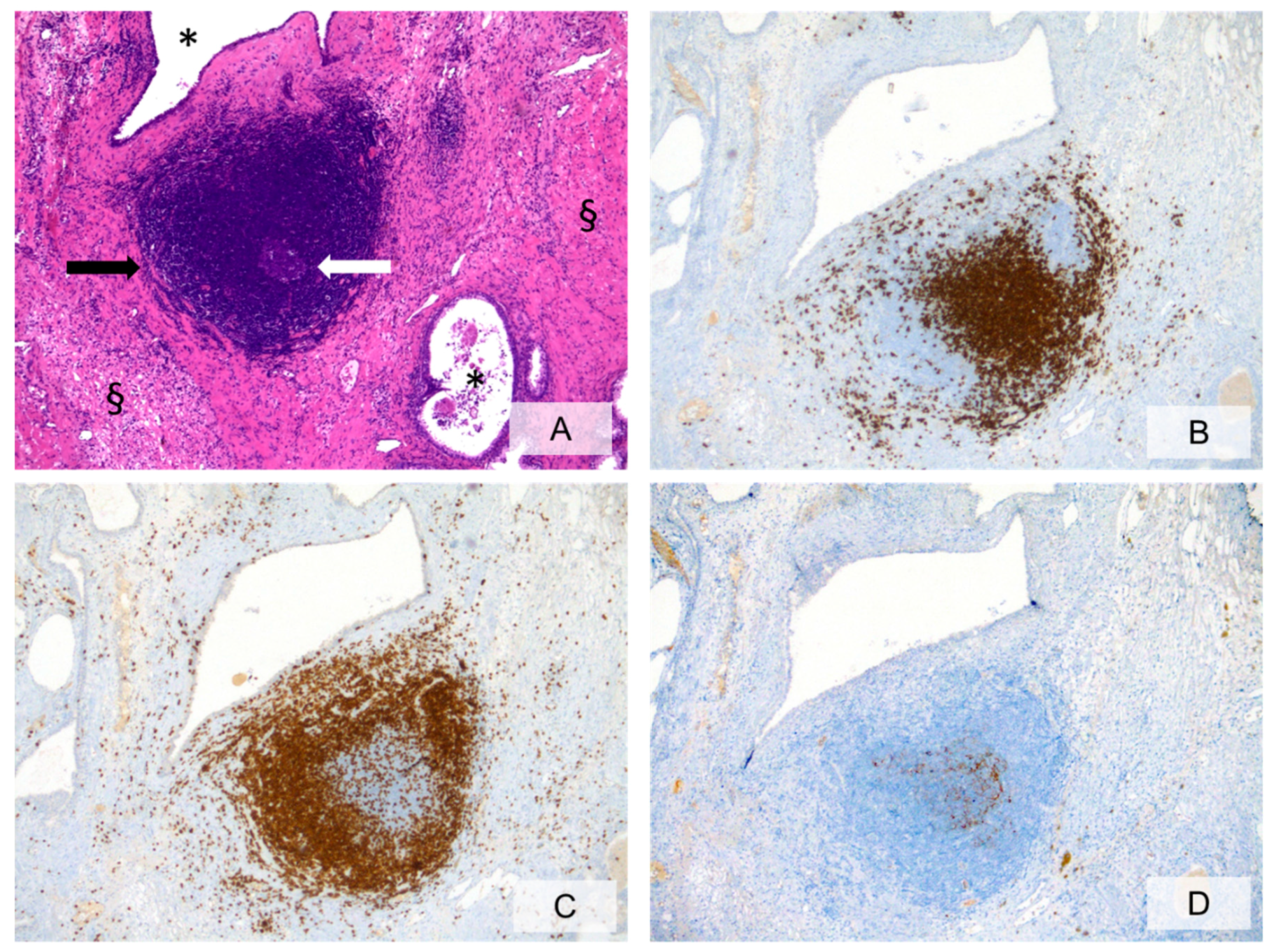
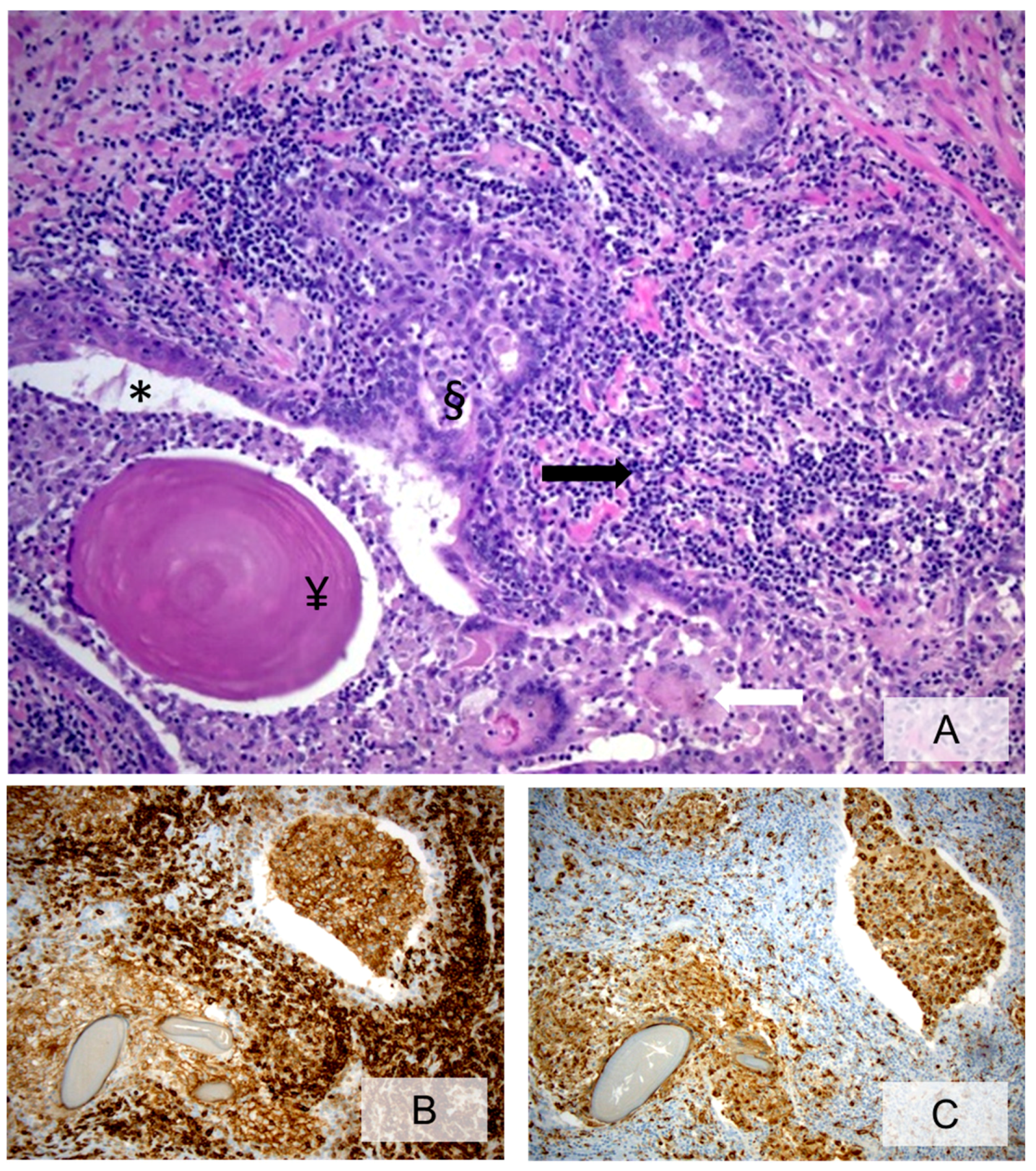
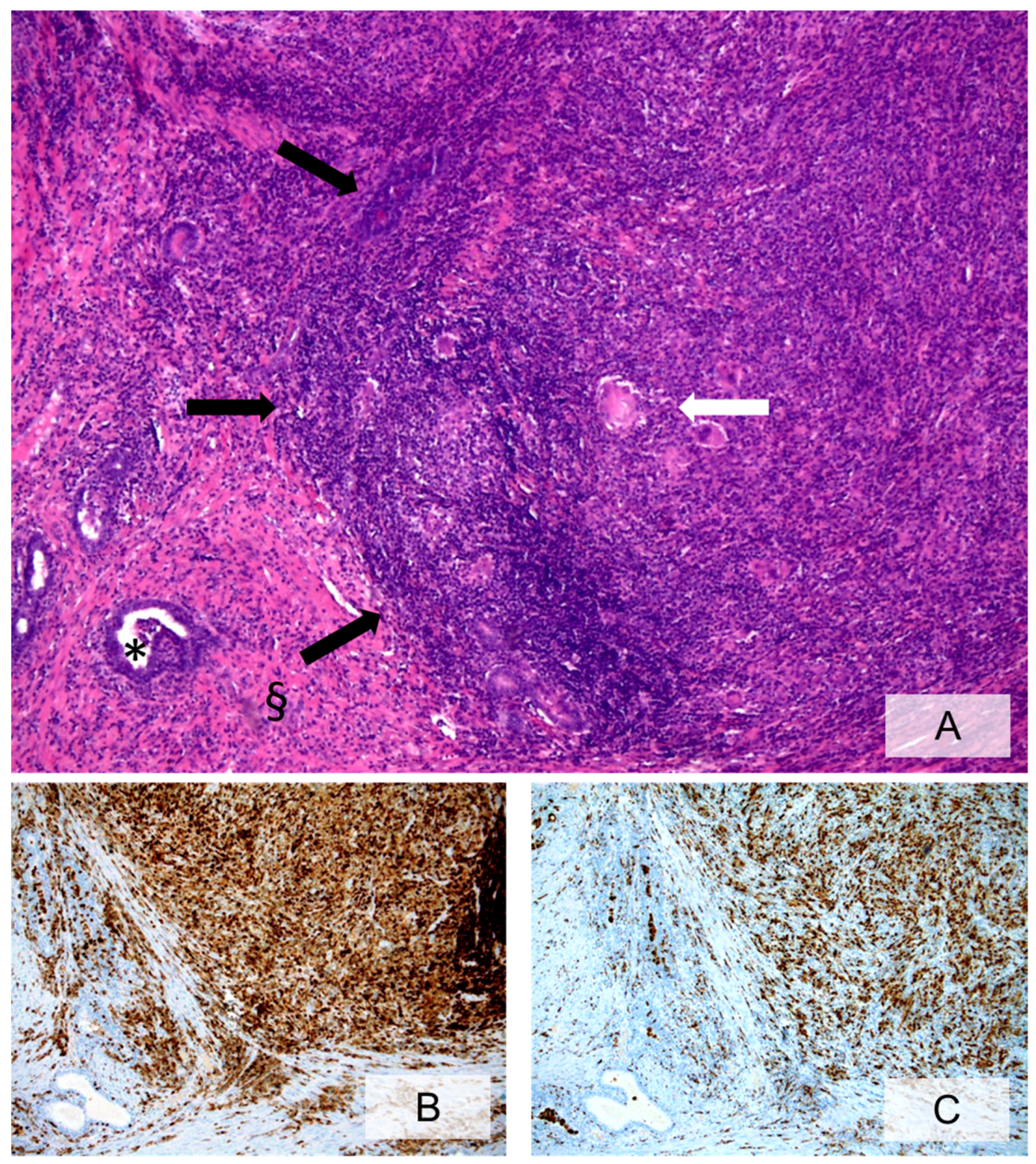
3.2. Cellular Components of Prostate Inflammation
3.3. Histopathologic Characteristics of Prostate Inflammation
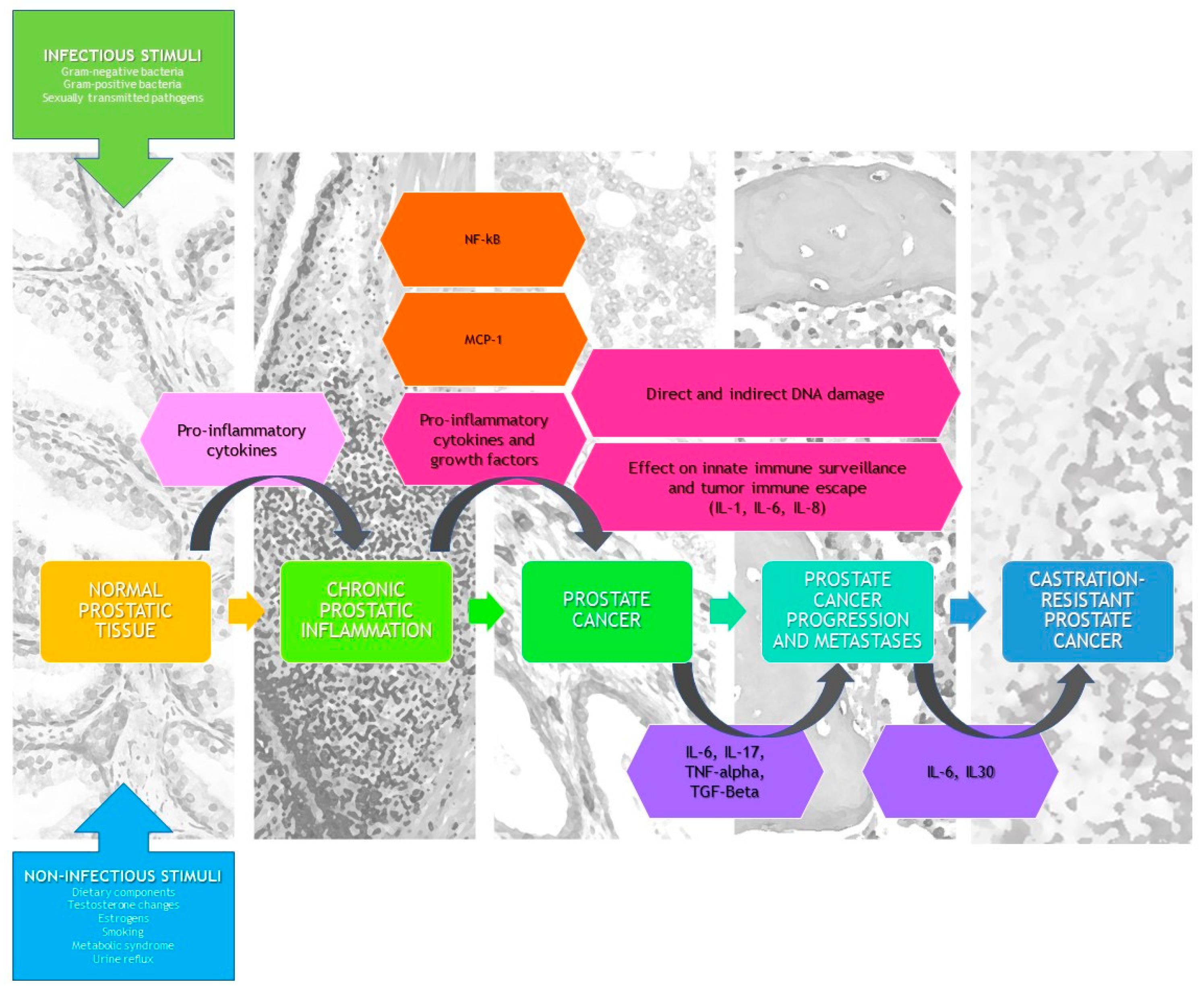
3.4. What Is the Role of Histologically Demonstrated Inflammation in PCa Development and Progression?
3.5. What Are the Causes of Prostate Inflammation?
3.5.1. Infectious Etiology
3.5.2. Noninfectious Etiology
3.6. How Can Prostate Inflammation Be Detected in Everyday Clinical Practice?
4. Clinical Remarks
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Rebbeck, T.R. Prostate cancer genetics: Variation by race, ethnicity, and geography. Semin. Radiat. Oncol. 2017, 27, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Modena, A.; Iacovelli, R.; Scarpa, A.; Brunelli, M.; Ciccarese, C.; Fantinel, E.; Bimbatti, D.; Massari, F.; Martignoni, G.; Tortora, G. Investigating BRCA mutations: A breakthrough in precision medicine of castration-resistant prostate cancer. Target. Oncol. 2016, 11, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Perletti, G.; Monti, E.; Magri, V.; Cai, T.; Cleves, A.; Trinchieri, A.; Montanari, E. The association between prostatitis and prostate cancer. Systematic review and meta-analysis. Arch. Ital. Urol. Androl. 2017, 89, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Davidsson, S.; Fiorentino, M.; Andrén, O.; Fang, F.; Mucci, L.A.; Varenhorst, E.; Fall, K.; Rider, J.R. Inflammation, focal atrophic lesions, and prostatic intraepithelial neoplasia with respect to risk of lethal prostate cancer. Cancer Epidemiol. Prev. Biomark. 2011, 20, 2280–2287. [Google Scholar] [CrossRef] [PubMed]
- Boehm, K.; Valdivieso, R.; Meskawi, M.; Larcher, A.; Schiffmann, J.; Sun, M.; Graefen, M.; Saad, F.; Parent, M.É.; Karakiewicz, P.I. Prostatitis, other genitourinary infections and prostate cancer: Results from a population based case-control study. World J. Urol. 2016, 34, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Dennis, L.K.; Lynch, C.F.; Torner, J.C. Epidemiologic association between prostatitis and prostate cancer. Urology 2002, 60, 78–83. [Google Scholar] [CrossRef]
- Karakiewicz, P.I.; Benayoun, S.; Begin, L.R.; Duclos, A.; Valiquette, L.; McCormack, M.; Bénard, F.; Saad, F.; Perrotte, P. Chronic inflammation is negatively associated with prostate cancer and high-grade prostatic intraepithelial neoplasia on needle biopsy. Int. J. Clin. Pract. 2007, 61, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Yli-Hemminki, T.H.; Laurila, M.; Auvinen, A.; Määttänen, L.; Huhtala, H.; Tammela, T.L.; Kujala, P.M. Histological inflammation and risk of subsequent prostate cancer among men with initially elevated serum prostate-specific antigen (PSA) concentration in the Finnish prostate cancer screening trial. BJU Int. 2013, 112, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Li, J.; Zhang, Y.; Zhu, H.; Liu, J.; Pumill, C. The role of prostatitis in prostate cancer: Meta-analysis. PLoS ONE 2013, 8, e85179. [Google Scholar] [CrossRef] [PubMed]
- Nickel, J.C.; Roehrborn, C.G.; O’Leary, M.P.; Bostwick, D.G.; Somerville, M.C.; Rittmaster, R.S. The relationship between prostate inflammation and lower urinary tract symptoms: Examination of baseline data from the REDUCE trial. Eur. Urol. 2008, 54, 1379–1384. [Google Scholar] [CrossRef] [PubMed]
- Schatteman, P.H.; Hoekx, L.; Wyndaele, J.J.; Jeuris, W.; Van Marck, E. Inflammation in prostate biopsies of men without prostatic malignancy or clinical prostatitis: Correlation with total serum PSA and PSA density. Eur. Urol. 2000, 37, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Sfanos, K.S.; De Marzo, A.M. Prostate cancer and inflammation: The evidence. Histopathology 2012, 60, 199–215. [Google Scholar] [CrossRef] [PubMed]
- Murtola, T.J.; Gurel, B.; Umbehr, M.; Lucia, M.S.; Thompson, I.M., Jr.; Goodman, P.J.; Kristal, A.R.; Parnes, H.L.; Lippman, S.M.; Sutcliffe, S.; et al. Inflammation in benign prostate tissue and prostate cancer in the finasteride arm of the prostate cancer prevention trial. Cancer Epidemiol. Prev. Biomark. 2016, 25, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.M.; Lundberg, K.; Ozenci, V.; Banham, A.H.; Hellström, M.; Egevad, L.; Pisa, P. CD4 + CD25high T cells are enriched in the tumor and peripheral blood of prostate cancer patients. J. Immunol. 2006, 177, 7398–7405. [Google Scholar] [CrossRef] [PubMed]
- Burdova, A.; Rulisek, P.; Bouchal, J.; Král, M.; Student, V.; Kolar, Z. Infiltration of prostate cancer by CD204+ and CD3+ cells correlates with ERG expression and TMPRSS2-ERG gene fusion. Klin. Onkol. 2018, 31, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.P.; Li, J.; Yadav, S.S.; Tewari, A.K. Recent insights into NF-kappaB signalling pathways and the link between inflammation and prostate cancer. BJU Int. 2014, 114, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Karan, D.; Dubey, S. From inflammation to prostate cancer: The role of inflammasomes. Adv. Urol. 2016, 2016, 3140372. [Google Scholar] [CrossRef]
- Schroder, K.J.; Tschopp, J. The inflammasomes. Cell 2010, 140, 821–832. [Google Scholar] [CrossRef]
- Chen, G.; Nunez, G. Inflammasomes in intestinal inflammation and cancer. Gastroenterology 2011, 141, 1986–1999. [Google Scholar] [CrossRef]
- Fabbi, M.; Carbotti, G.; Ferrini, S. Context-dependent role of IL-18 in cancer biology and counter-regulation by IL-18BP. J. Leukoc. Biol. 2015, 97, 665–675. [Google Scholar] [CrossRef]
- Chow, M.T.; Sceneay, J.; Paget, C.; Wong, C.S.; Duret, H.; Tschopp, J.; Möller, A.; Smyth, M.J. NLRP3 suppresses NK cell-mediated responses to carcinogen-induced tumors and metastases. Cancer Res. 2012, 72, 5721–5732. [Google Scholar] [CrossRef] [PubMed]
- Sorrentino, C.; Ciummo, S.L.; Cipollone, G.; Caputo, S.; Bellone, M.; Di Carlo, E. Interleukin-30/IL27p28 shapes prostate cancer stem-like cell behavior and is critical for tumor onset and metastasization. Cancer Res. 2018, 78, 2654–2668. [Google Scholar] [CrossRef] [PubMed]
- Garbers, C.; Spudy, B.; Aparicio-Siegmund, S.; Waetzig, G.H.; Sommer, J.; Hölscher, C.; Rose-John, S.; Grötzinger, J.; Lorenzen, I.; Scheller, J. An interleukin-6 receptor-dependent molecular switch mediates signal transduction of the IL-27 cytokine subunit p28 (IL-30) via a gp130 protein receptor homodimer. J. Biol. Chem. 2013, 288, 4346–4354. [Google Scholar] [CrossRef] [PubMed]
- Nickel, J.C.; True, L.D.; Krieger, J.N.; Berger, R.E.; Boag, A.H.; Young, I.D. Consensus development of a histopathological classification system for chronic prostatic inflammation. BJU Int. 2001, 87, 797–805. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.D.; Patterson, M.C. Needle biopsy study of chronic prostatitis. J. Urol. 1966, 96, 519–533. [Google Scholar] [CrossRef]
- Nielsen, M.L.; Asnaes, S.; Hattel, T. Inflammatory changes in the non-infected prostate gland. A clinical, microbiological and histological investigation. J. Urol. 1973, 110, 423–426. [Google Scholar] [CrossRef]
- Kohnen, P.W.; Drach, G.W. Patterns of inflammation in prostatic hyperplasia: A histologic and bacteriologic study. J. Urol. 1979, 121, 755–760. [Google Scholar] [CrossRef]
- Doble, A.; Thomas, B.J.; Furr, P.M.; Walker, M.M.; Harris, J.R.; Witherow, R.O.; Taylor-Robinson, D. A search for infectious agents in chronic abacterial prostatitis using ultrasound guided biopsy. Br. J. Urol. 1989, 64, 297–301. [Google Scholar] [CrossRef]
- Brawn, P.N.; Speights, V.O.; Kuhl, D.; Riggs, M.; Spiekerman, A.M.; McCord, R.G.; Coffield, K.S.; Stewart, D.T.; Lind, M.L. Prostate-specific antigen levels from completely sectioned, clinically benign, whole prostates. Cancer 1991, 68, 1592–1599. [Google Scholar] [CrossRef]
- Bare, R.; Hart, L.; McCullough, D.L. Correlation of prostate-specific antigen and prostate-specific antigen density with outcome of prostate biopsy. Urology 1994, 43, 191–196. [Google Scholar] [CrossRef]
- Nadler, R.B.; Humphrey, P.A.; Smith, D.S.; Catalona, W.J.; Ratliff, T.L. Effect of inflammation and benign prostatic hyperplasia on elevated serum prostate specific antigen levels. J. Urol. 1995, 154, 407–413. [Google Scholar] [CrossRef]
- Irani, J.; Levillain, P.; Goujon, J.M.; Bon, D.; Doré, B.; Aubert, J. Inflammation in benign prostatic hyperplasia: Correlation with prostate specific antigen value. J. Urol. 1997, 157, 1301–1303. [Google Scholar] [CrossRef]
- True, L.D.; Berger, R.E.; Rothman, I.; Ross, S.O.; Krieger, J.N. Prostate histopathology and the chronic prostatitis/chronic pelvic pain syndrome: A prospective biopsy study. J. Urol. 1999, 162, 2014–2018. [Google Scholar] [CrossRef]
- Nickel, J.C.; Downey, J.; Young, I.; Boag, S. Asymptomatic inflammation and/or infection in benign prostatic hyperplasia. BJU Int. 1999, 84, 976–981. [Google Scholar] [CrossRef] [PubMed]
- Irani, J.; Goujon, J.M.; Ragni, E.; Peyrat, L.; Hubert, J.; Saint, F.; Mottet, N. High-grade inflammation in prostate cancer as a prognostic factor for biochemical recurrence after radical prostatectomy. Pathologist Multi Center Study Group. Urology 1999, 54, 467–472. [Google Scholar] [CrossRef]
- Anim, J.T.; Udo, C.; John, B. Characterisation of inflammatory cells in benign prostatic hyperplasia. Acta Histochem. 1998, 100, 439–449. [Google Scholar] [CrossRef]
- Blumenfeld, W.; Tucci, S.; Narayan, P. Incidental lymphocytic prostatitis. Selective involvement with nonmalignant glands. Am. J. Surg. Pathol. 1992, 16, 975. [Google Scholar] [CrossRef]
- Attah, E. Nonspecific inflammatory lesions of the prostate. Spectrum and patterns. Int. Surg. 1975, 60, 158–162. [Google Scholar]
- McNeal, J.E. The zonal anatomy of the prostate. Prostate 1981, 2, 35–49. [Google Scholar] [CrossRef]
- McClinton, S.; Eremin, O.; Miller, I.D. Inflammatory infiltrate in prostatic hyperplasia–Evidence of a host response to intraprostatic spermatozoa. Br. J. Urol. 1990, 65, 606–610. [Google Scholar] [CrossRef]
- McClinton, S.; Miller, I.D.; Eremin, O. An immunohistochemical characterisation of the inflammatory cell infiltrate in benign and malignant prostatic disease. Br. J. Cancer 1990, 61, 400–403. [Google Scholar] [CrossRef] [PubMed]
- Vignozzi, L.; Cellai, I.; Santi, R.; Lombardelli, L.; Morelli, A.; Comeglio, P.; Filippi, S.; Logiodice, F.; Carini, M.; Nesi, G.; et al. Antiinflammatory effect of androgen receptor activation in human benign prostatic hyperplasia cells. J. Endocrinol. 2012, 214, 31–43. [Google Scholar] [CrossRef] [PubMed]
- Vignozzi, L.; Gacci, M.; Cellai, I.; Santi, R.; Corona, G.; Morelli, A.; Rastrelli, G.; Comeglio, P.; Sebastanelli, A.; Maneschi, E.; et al. Fat boosts, while androgen receptor activation counteracts, BPH-associated prostate inflammation. Prostate 2013, 73, 789–800. [Google Scholar] [CrossRef] [PubMed]
- Gandaglia, G.; Zaffuto, E.; Fossati, N.; Cucchiara, V.; Mirone, V.; Montorsi, F.; Briganti, A. The role of prostatic inflammation in the development and progression of benign and malignant diseases. Curr. Opin. Urol. 2017, 27, 99–106. [Google Scholar] [CrossRef] [PubMed]
- De Nunzio, C.; Kramer, G.; Marberger, M.; Montironi, R.; Nelson, W.; Schröder, F.; Sciarra, A.; Tubaro, A. The controversial relationship between benign prostatic hyperplasia and prostate cancer: The role of inflammation. Eur. Urol. 2011, 60, 106–117. [Google Scholar] [CrossRef] [PubMed]
- Sutcliffe, S.; Giovannucci, E.; De Marzo, A.M.; Leitzmann, M.F.; Willett, W.C.; Platz, E.A. Gonorrhea, syphilis, clinical prostatitis, and the risk of prostate cancer. Cancer Epidemiol. Prev. Biomark. 2006, 15, 2160–2166. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Moreira, D.M.; Nickel, J.C.; Gerber, L.; Muller, R.L.; Andriole, G.L.; Castro-Santamaria, R.; Freedland, S.J. Baseline prostate inflammation is associated with a reduced risk of prostate cancer in men undergoing repeat prostate biopsy: Results from the REDUCE study. Cancer 2014, 120, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Cheng, I.; Witte, J.S.; Jacobsen, S.J.; Haque, R.; Quinn, V.P.; Quesenberry, C.P.; Caan, B.J.; Van Den Eeden, S.K. Prostatitis, sexually transmitted diseases, and prostate cancer: The California Men’s Health Study. PLoS ONE 2010, 5, e8736. [Google Scholar] [CrossRef] [PubMed]
- Aykan, S.; Temiz, M.Z.; Yilmaz, M.; Ulus, I.; Yuruk, E.; Muslumanoglu, A.Y. Association between male accesory gland infections and prostate cancer in Turkish men: A case-control study. Andrologia 2018, 8, e13038. [Google Scholar] [CrossRef]
- Kuang, A.G.; Nickel, J.C.; Andriole, G.L.; Castro-Santamaria, R.; Freedland, S.J.; Moreira, D.M. Both acute and chronic inflammation are associated with less perineural invasion in men with prostate cancer on repeat biopsy. BJU Int. 2019, 123, 91–97. [Google Scholar] [CrossRef]
- Cai, T.; Mazzoli, S.; Meacci, F.; Boddi, V.; Mondaini, N.; Malossini, G.; Bartoletti, R. Epidemiological features and resistance pattern in uropathogens isolated from chronic bacterial prostatitis. J. Microbiol. 2011, 49, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Cai, T.; Pisano, F.; Magri, V.; Verze, P.; Mondaini, N.; D’Elia, C.; Malossini, G.; Mazzoli, S.; Perletti, G.; Gontero, P.; et al. Chlamydia trachomatis infection is related to premature ejaculation in chronic prostatitis patients: Results from a cross-sectional study. J. Sex. Med. 2014, 11, 3085–3092. [Google Scholar] [CrossRef] [PubMed]
- Van Elsland, D.; Neefjes, J. Bacterial infections and cancer. EMBO Rep. 2018, 19. [Google Scholar] [CrossRef] [PubMed]
- Rosadi, F.; Fiorentini, C.; Fabbri, A. Bacterial protein toxins in human cancers. Pathog. Dis. 2016, 74. [Google Scholar] [CrossRef] [PubMed]
- Moreira, D.M.; Nickel, J.C.; Gerber, L.; Muller, R.L.; Andriole, G.L.; Castro-Santamaria, R.; Freedland, S.J. Smoking is associated with acute and chronic prostatic inflammation: Results from the REDUCE study. Cancer Prev. Res. 2015, 8, 312–317. [Google Scholar] [CrossRef]
- Shankar, E.; Bhaskaran, N.; MacLennan, G.T.; Liu, G.; Daneshgari, F.; Gupta, S. Inflammatory signaling involved in high-fat diet induced prostate diseases. J. Urol. Res. 2015, 2, 1018. [Google Scholar]
- De Marzo, A.M.; Nelson, W.G.; Isaacs, W.B.; Epstein, J.I. Pathological and molecular aspects of prostate cancer. Lancet 2003, 361, 955–964. [Google Scholar] [CrossRef]
- Voronov, E.; Shouval, D.S.; Krelin, Y.; Cagnano, E.; Benharroch, D.; Iwakura, Y.; Dinarello, C.A.; Apte, R.N. IL-1 is required for tumor invasiveness and angiogenesis. Proc. Natl. Acad. Sci. USA 2003, 100, 2645–2650. [Google Scholar] [CrossRef]
- Nguyen, D.P.; Li, J.; Tewari, A.K. Inflammation and prostate cancer: The role of interleukin 6 (IL-6). BJU Int. 2014, 113, 986–992. [Google Scholar] [CrossRef]
- Steiner, G.E.; Newman, M.E.; Paikl, D.; Stix, U.; Memaran-Dagda, N.; Lee, C.; Marberger, M.J. Expression and function of pro-inflammatory interleukin IL-17 and IL-17 receptor in normal, benign hyperplastic, and malignant prostate. Prostate 2003, 56, 171–182. [Google Scholar] [CrossRef]
- Michalaki, V.; Syrigos, K.; Charles, P.; Waxman, J. Serum levels of IL-6 and TNF-alpha correlate with clinicopathological features and patient survival in patients with prostate cancer. Br. J. Cancer 2004, 90, 2312–2316. [Google Scholar] [CrossRef] [PubMed]
- Park, J.I.; Lee, M.G.; Cho, K.; Park, B.J.; Chae, K.S.; Byun, D.S.; Ryu, B.K.; Park, Y.K.; Chi, S.G. Transforming growth factor-beta1 activates interleukin-6 expression in prostate cancer cells through the synergistic collaboration of the Smad2, p38-NF-kappaB, JNK, and Ras signaling pathways. Oncogene 2003, 22, 4314–4332. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Li, Q.; Han, P.; Li, X.; Zeng, H.; Zhu, Y.; Wei, Q. Evaluation of interleukin-8 in expressed prostatic secretion as a reliable biomarker of inflammation in benign prostatic hyperplasia. Urology 2009, 74, 340–344. [Google Scholar] [CrossRef] [PubMed]
- Fujita, K.; Ewing, C.M.; Getzenberg, R.H.; Parsons, J.K.; Isaacs, W.B.; Pavlovich, C.P. Monocyte chemotactic protein-1 (MCP-1/CCL2) is associated with prostatic growth dysregulation and benign prostatic hyperplasia. Prostate 2010, 70, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Ficarra, V.; Sekulovic, S.; Zattoni, F.; Zazzera, M.; Novara, G. Why and how to evaluate chronic prostatic inflammation. Eur. Urol. Suppl. 2013, 12, 110–115. [Google Scholar] [CrossRef]
- Klimas, R.; Bennett, B.; Gardner, W.A., Jr. Prostatic calculi: A review. Prostate 1985, 7, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Geramoutsos, I.; Gyftopoulos, K.; Perimenis, P.; Thanou, V.; Liagka, D.; Siamblis, D.; Barbalias, G. Clinical correlation of prostatic lithiasis with chronic pelvic pain syndromes in young adults. Eur. Urol. 2004, 45, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Cai, T.; Tessarolo, F.; Caola, I.; Piccoli, F.; Nollo, G.; Caciagli, P.; Mazzoli, S.; Palmieri, A.; Verze, P.; Malossini, G.; et al. Prostate calcifications: A case series supporting the microbial biofilm theory. Investig. Clin. Urol. 2018, 59, 187–193. [Google Scholar] [CrossRef]
- Yang, H.J.; Huang, K.H.; Wang, C.W.; Chang, H.C.; Yang, T.K. Prostate calcification worsen lower urinary tract symptoms in middle-aged men. Urology 2013, 81, 1320–1324. [Google Scholar] [CrossRef]
- Shoskes, D.A.; Lee, C.T.; Murphy, D.; Kefer, J.; Wood, H.M. Incidence and significance of prostatic stones in men with chronic prostatitis/chronic pelvic pain syndrome. Urology 2007, 70, 235–238. [Google Scholar] [CrossRef]
- Kwon, Y.K.; Choe, M.S.; Seo, K.W.; Park, C.H.; Chang, H.S.; Kim, B.H.; Kim, C.I. The effect of intraprostatic chronic inflammation on benign prostatic hyperplasia treatment. Korean J. Urol. 2010, 51, 266–270. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nesi, G.; Nobili, S.; Cai, T.; Caini, S.; Santi, R. Chronic inflammation in urothelial bladder cancer. Virchows Arch. 2015, 467, 623–633. [Google Scholar] [CrossRef] [PubMed]
- Nakai, Y.; Nonomura, N. Inflammation and prostate carcinogenesis. Int. J. Urol. 2013, 20, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Fujita, K.; Hayashi, T.; Matsushita, M.; Uemura, M.; Nonomura, N. Obesity, inflammation, and prostate cancer. J. Clin. Med. 2019, 8, 201. [Google Scholar] [CrossRef] [PubMed]
- Doat, S.; Cénée, S.; Trétarre, B.; Rebillard, X.; Lamy, P.J.; Bringer, J.P.; Iborra, F.; Murez, T.; Sanchez, M.; Menegaux, F. Nonsteroidal anti-inflammatory drugs (NSAIDs) and prostate cancer risk: Results from the EPICAP study. Cancer Med. 2017, 6, 2461–2470. [Google Scholar] [CrossRef] [PubMed]
| Description | Role | Reference | |
|---|---|---|---|
| IL-1 | Proinflammatory cytokine | Promotes cancer growth and progression | Voronov (2003) [58] |
| IL-6 | Proinflammatory cytokine | Promotes PCa growth and progression; has a role in CRPC | Nguyen (2014) [59] |
| IL-8 | Proinflammatory cytokine | Proliferation of prostate stromal cells; to regulates the expression of matrix metalloproteinases and PCa progression | Liu (2009) [63] |
| IL-17 | Proinflammatory cytokine | Promotes PCa growth, angiogenesis and metastasis | Steiner (2003) [60] |
| IL-30 | Proinflammatory cytokine | Promotes PCa progression and metastasis | Sorrentino (2018) [22] |
| MCP-1 | Chemotactic cytokine | Promotes prostatic inflammation | Fujita (2010) [64] |
| TNF-α | Proinflammatory cytokine | Promotes PCa progression and metastasis | Michalaki (2004) [61] |
| TGF-β | Multifunctional cytokine | Promotes PCa progression and metastasis | Park (2003) [62] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, T.; Santi, R.; Tamanini, I.; Galli, I.C.; Perletti, G.; Bjerklund Johansen, T.E.; Nesi, G. Current Knowledge of the Potential Links between Inflammation and Prostate Cancer. Int. J. Mol. Sci. 2019, 20, 3833. https://doi.org/10.3390/ijms20153833
Cai T, Santi R, Tamanini I, Galli IC, Perletti G, Bjerklund Johansen TE, Nesi G. Current Knowledge of the Potential Links between Inflammation and Prostate Cancer. International Journal of Molecular Sciences. 2019; 20(15):3833. https://doi.org/10.3390/ijms20153833
Chicago/Turabian StyleCai, Tommaso, Raffaella Santi, Irene Tamanini, Ilaria Camilla Galli, Gianpaolo Perletti, Truls E. Bjerklund Johansen, and Gabriella Nesi. 2019. "Current Knowledge of the Potential Links between Inflammation and Prostate Cancer" International Journal of Molecular Sciences 20, no. 15: 3833. https://doi.org/10.3390/ijms20153833
APA StyleCai, T., Santi, R., Tamanini, I., Galli, I. C., Perletti, G., Bjerklund Johansen, T. E., & Nesi, G. (2019). Current Knowledge of the Potential Links between Inflammation and Prostate Cancer. International Journal of Molecular Sciences, 20(15), 3833. https://doi.org/10.3390/ijms20153833







