Periodontal Therapy for Improving Lipid Profiles in Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Results
2.1. Selection of Studies
2.2. Characteristics of Studies
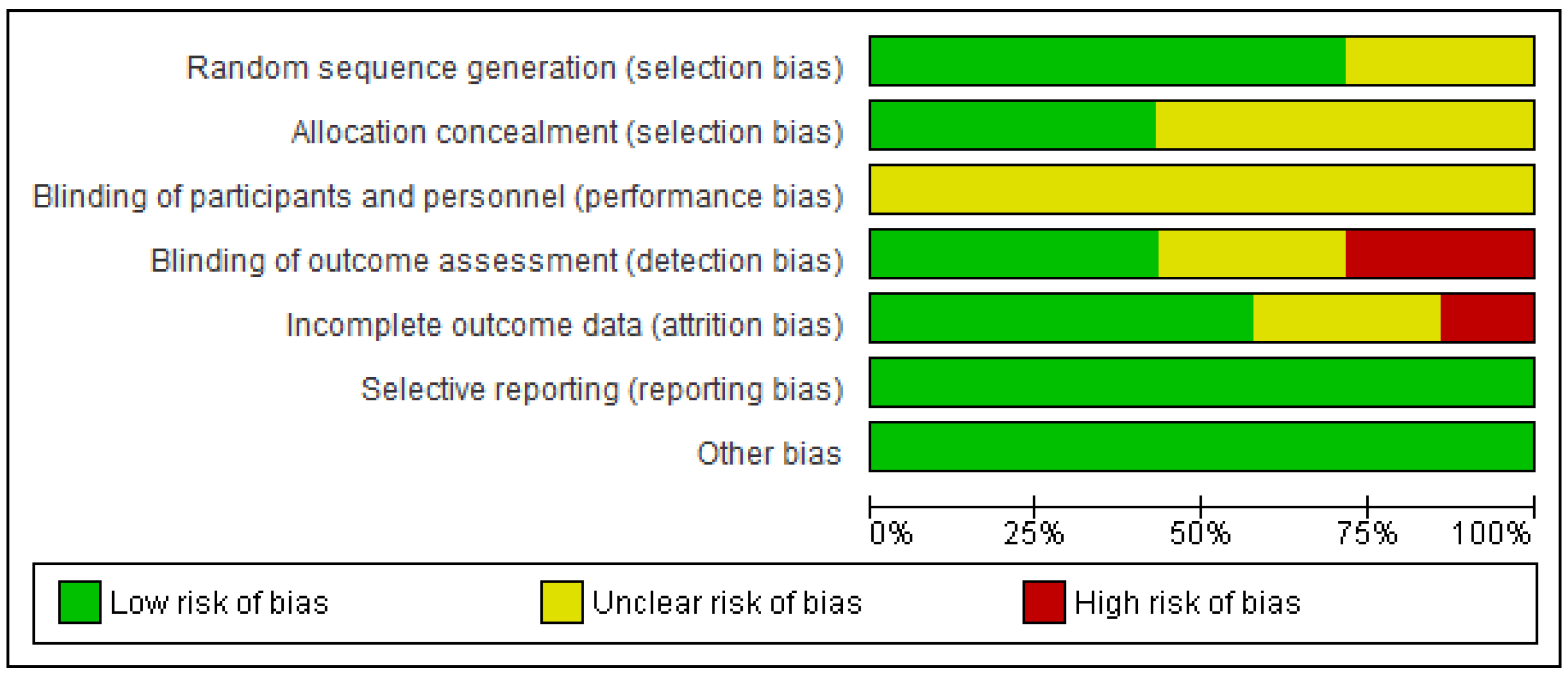
2.3. Risk of Bias within Studies
2.4. Lipid Profiles
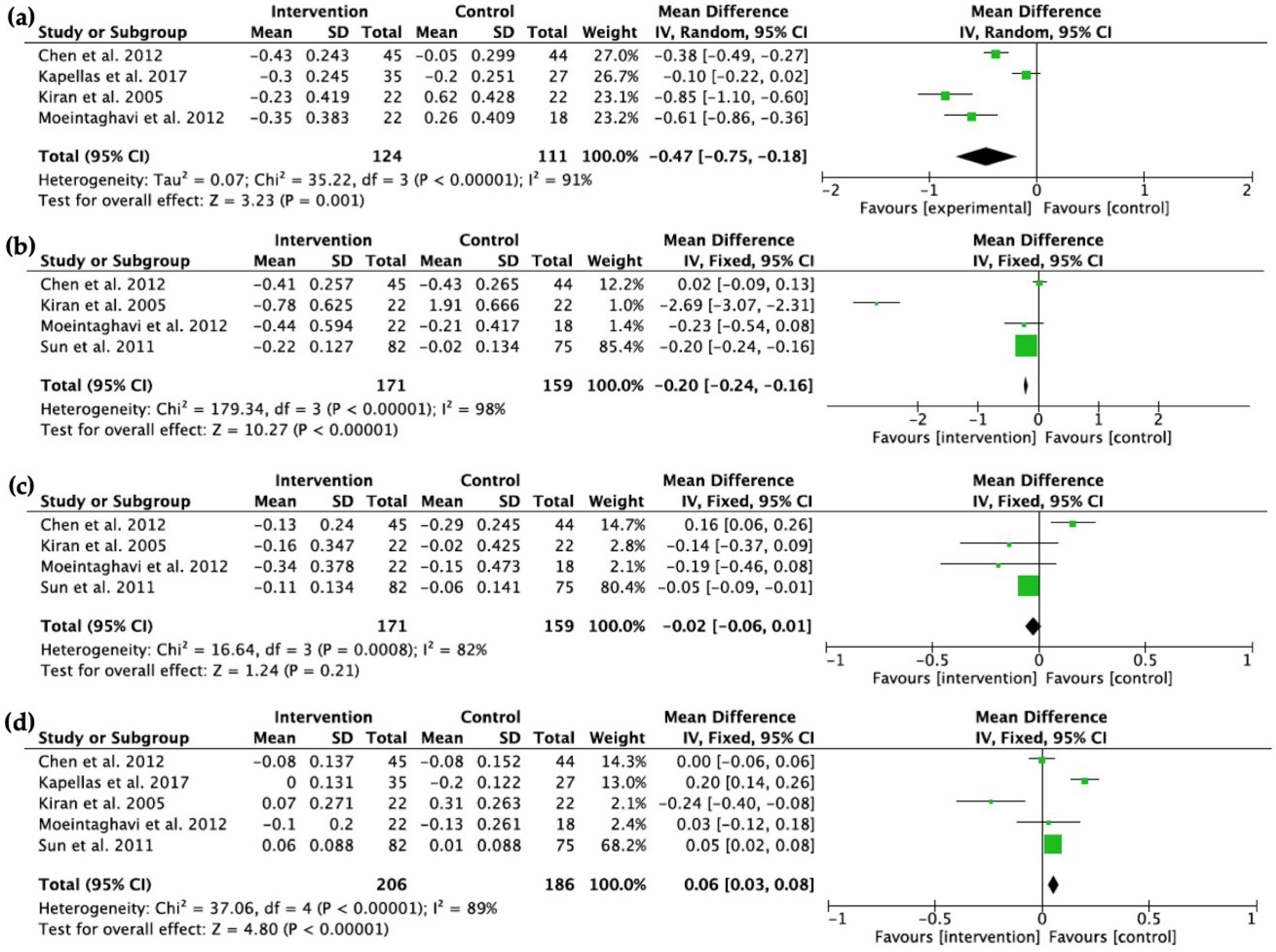
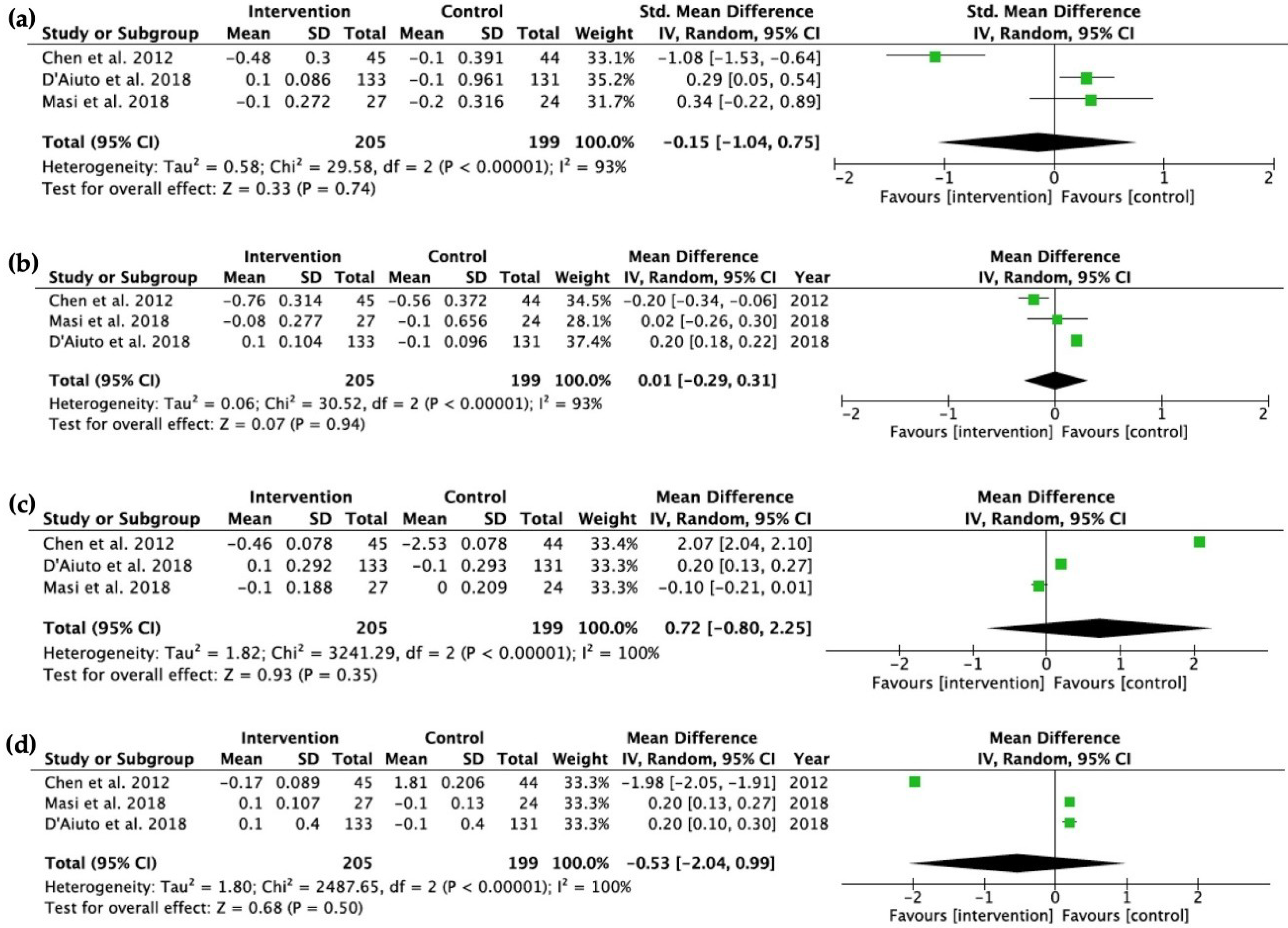
2.4.1. Baseline vs. 3-Month Follow-Up
2.4.2. Baseline vs. 6-Month Follow-Up
2.5. Periodontal Outcomes
3. Discussion
4. Materials and Methods
4.1. Types of Studies
4.2. Types of Participants
4.3. Types of Intervention
4.4. Types of Outcome Measures
4.5. Search Methods
- -
- Patients = individuals with type 2 DM
- -
- Intervention = anti-inflammatory surgical or non-surgical periodontal treatment
- -
- Comparison = no periodontal treatment or only supragingival scaling and polishing
- -
- Outcome = lipid profiles
4.6. Selection of Studies
4.7. Data Extraction
- -
- General study characteristics: authors, year of study, country of origin, intervention/control, number of participants at baseline, follow-up period, diabetes and periodontal inclusion criteria
- -
- Primary outcomes: lipid profiles (total cholesterol, triglycerides, LDL, HDL)
- -
- Secondary outcomes: probing depth and bleeding on probing at baseline and 3- or 6-month follow-ups.
4.8. Quality Assessment
4.9. Data Synthesis
Author Contributions
Funding
Conflicts of Interest
References
- Ericsson, J.S.; Abrahamsson, K.H.; Ostberg, A.L.; Hellström, M.K.; Jönsson, K.; Wennström, J.L. Periodontal health status in Swedish adolescents: An epidemiological, cross-sectional study. Swed. Dent. J. 2009, 33, 131–139. [Google Scholar] [PubMed]
- Li, Y.; Lee, S.; Hujoel, P.; Su, M.; Zhang, W.; Kim, J.; Zhang, Y.P.; De Vizio, W. Prevalence and severity of gingivitis in American adults. Am. J. Dent. 2010, 23, 9. [Google Scholar] [PubMed]
- Negrato, C.A.; Tarzia, O.; Jovanovič, L.; Chinellato, L.E.M. Periodontal disease and diabetes mellitus. J. Appl. Oral Sci. 2013, 21, 1. [Google Scholar] [CrossRef] [PubMed]
- Preshaw, P.; Alba, A.; Herrera, D.; Jepsen, S.; Konstantinidis, A.; Makrilakis, K.; Taylor, R. Periodontitis and diabetes: A two-way relationship. Diabetologia 2012, 55, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Capewell, S.; Buchan, I. Why have sustained increases in obesity and type 2 diabetes not offset declines in cardiovascular mortality over recent decades in Western countries? Nutr. Metab. Cardiovasc. Dis. NMCD 2012, 22, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Fagot-campagna, A.; Rolka, D.B.; Beckles, G.L.; Gregg, E.W.; Narayan, K. Prevalence of lipid abnormalities, awareness, and treatment in US adults with diabetes. Diabetes 2000, 49, A78. [Google Scholar]
- Shen, G.X. Lipid disorders in diabetes mellitus and current management. Curr. Pharm. Anal. 2007, 3, 17–24. [Google Scholar] [CrossRef]
- Nourooz-Zadeh, J.; Rahimi, A.; Tajaddini-Sarmadi, J.; Tritschler, H.; Rosen, P.; Halliwell, B.; Betteridge, D. Relationships between plasma measures of oxidative stress and metabolic control in NIDDM. Diabetologia 1997, 40, 647–653. [Google Scholar] [CrossRef]
- Pickup, J.; Mattock, M.; Chusney, G.; Burt, D. NIDDM as a disease of the innate immune system: association of acute-phase reactants and interleukin-6 with metabolic syndrome X. Diabetologia 1997, 40, 1286. [Google Scholar] [CrossRef]
- Chen, L.; Luo, G.; Xuan, D.; Wei, B.; Liu, F.; Li, J.; Zhang, J. Effects of non-surgical periodontal treatment on clinical response, serum inflammatory parameters, and metabolic control in patients with type 2 diabetes: A randomized study. J. Periodontol. 2012, 83, 435–443. [Google Scholar] [CrossRef]
- D’Aiuto, F.; Gkranias, N.; Bhowruth, D.; Khan, T.; Orlandi, M.; Suvan, J.; Masi, S.; Tsakos, G.; Hurel, S.; Hingorani, A.D.; et al. Systemic effects of periodontitis treatment in patients with type 2 diabetes: A 12 month, single-centre, investigator-masked, randomised trial. Lancet Diabetes Endocrinol. 2018, 6, 954–965. [Google Scholar] [CrossRef]
- Kapellas, K.; Mejia, G.; Bartold, P.M.; Skilton, M.R.; Maple-Brown, L.J.; Slade, G.D.; O’Dea, K.; Brown, A.; Celermajer, D.S.; Jamieson, L.M. Periodontal therapy and glycaemic control among individuals with type 2 diabetes: Reflections from the PerioCardio study. Int. J. Dent. Hyg. 2017, 15, e42–e51. [Google Scholar] [CrossRef] [PubMed]
- Kiran, M.; Arpak, N.; Unsal, E.; Erdogan, M.F. The effect of improved periodontal health on metabolic control in type 2 diabetes mellitus. J. Clin. Periodontol. 2005, 32, 266–272. [Google Scholar] [CrossRef] [PubMed]
- Masi, S.; Orlandi, M.; Parkar, M.; Bhowruth, D.; Kingston, I.; O’Rourke, C.; Virdis, A.; Hingorani, A.; Hurel, S.J.; Donos, N.; et al. Mitochondrial oxidative stress, endothelial function and metabolic control in patients with type II diabetes and periodontitis: A randomised controlled clinical trial. Int. J. Cardiol. 2018, 271, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Moeintaghavi, A.; Arab, H.R.; Bozorgnia, Y.; Kianoush, K.; Alizadeh, M. Non-surgical periodontal therapy affects metabolic control in diabetics: A randomized controlled clinical trial. Aust. Dent. J. 2012, 57, 31–37. [Google Scholar] [CrossRef]
- Sun, W.L.; Chen, L.L.; Zhang, S.Z.; Wu, Y.M.; Ren, Y.Z.; Qin, G.M. Inflammatory cytokines, adiponectin, insulin resistance and metabolic control after periodontal intervention in patients with type 2 diabetes and chronic periodontitis. Intern. Med. (Tokyo, Japan) 2011, 50, 1569–1574. [Google Scholar] [CrossRef]
- Higgins, J.P.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- Sbordone, L.; Ramaglia, L.; Gulletta, E.; Iacono, V. Recolonization of the subgingival microflora after scaling and root planing in human periodontitis. J. Periodontol. 1990, 61, 579–584. [Google Scholar] [CrossRef]
- Pejcic, A.; Kesic, L.; Brkic, Z.; Pesic, Z.; Mirkovic, D. Effect of periodontal treatment on lipoproteins levels in plasma in patients with periodontitis. South. Med. J. 2011, 104, 547–552. [Google Scholar] [CrossRef]
- Oz, S.G.; Fentoglu, O.; Kilicarslan, A.; Guven, G.S.; Tanriover, M.D.; Aykac, Y.; Sozen, T. Beneficial effects of periodontal treatment on metabolic control of hypercholesterolemia. South. Med. J. 2007, 100, 686–692. [Google Scholar] [CrossRef]
- Feingold, K.R.; Staprans, I.; Memon, R.; Moser, A.; Shigenaga, J.; Doerrler, W.; Dinarello, C.; Grunfeld, C. Endotoxin rapidly induces changes in lipid metabolism that produce hypertriglyceridemia: low doses stimulate hepatic triglyceride production while high doses inhibit clearance. J. Lipid Res. 1992, 33, 1765–1776. [Google Scholar]
- Fentoglu, O.; Bozkurt, F.Y. The bi-directional relationship between periodontal disease and hyperlipidemia. Eur. J. Dent. 2008, 2, 142. [Google Scholar]
- Cutler, C.W.; Eke, P.; Arnold, R.R.; Van Dyke, T.E. Defective neutrophil function in an insulin-dependent diabetes mellitus patient. A case report. J. Periodontol. 1991, 62, 394–401. [Google Scholar] [CrossRef]
- Stratton, I.M.; Adler, A.I.; Neil, H.A.W.; Matthews, D.R.; Manley, S.E.; Cull, C.A.; Hadden, D.; Turner, R.C.; Holman, R.R. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): Prospective observational study. BMJ 2000, 321, 405–412. [Google Scholar] [CrossRef]
- Borgnakke, W.S. Does treatment of periodontal disease influence systemic disease? Dent. Clin. 2015, 59, 885–917. [Google Scholar] [CrossRef]
- Teeuw, W.J.; Gerdes, V.E.; Loos, B.G. Effect of periodontal treatment on glycemic control of diabetic patients: A systematic review and meta-analysis. Diabetes Care 2010, 33, 421–427. [Google Scholar] [CrossRef]
- Iwamoto, Y.; Nishimura, F.; Nakagawa, M.; Sugimoto, H.; Shikata, K.; Makino, H.; Fukuda, T.; Tsuji, T.; Iwamoto, M.; Murayama, Y. The effect of antimicrobial periodontal treatment on circulating tumor necrosis factor-alpha and glycated hemoglobin level in patients with type 2 diabetes. J. Periodontol. 2001, 72, 774–778. [Google Scholar] [CrossRef]
- Thompson, P.D.; Panza, G.; Zaleski, A.; Taylor, B. Statin-associated side effects. J. Am. Coll. Cardiol. 2016, 67, 2395–2410. [Google Scholar] [CrossRef]
- Feingold, K.R.; Grunfeld, C. Cholesterol Lowering Drugs. MDText 2000. [Google Scholar]
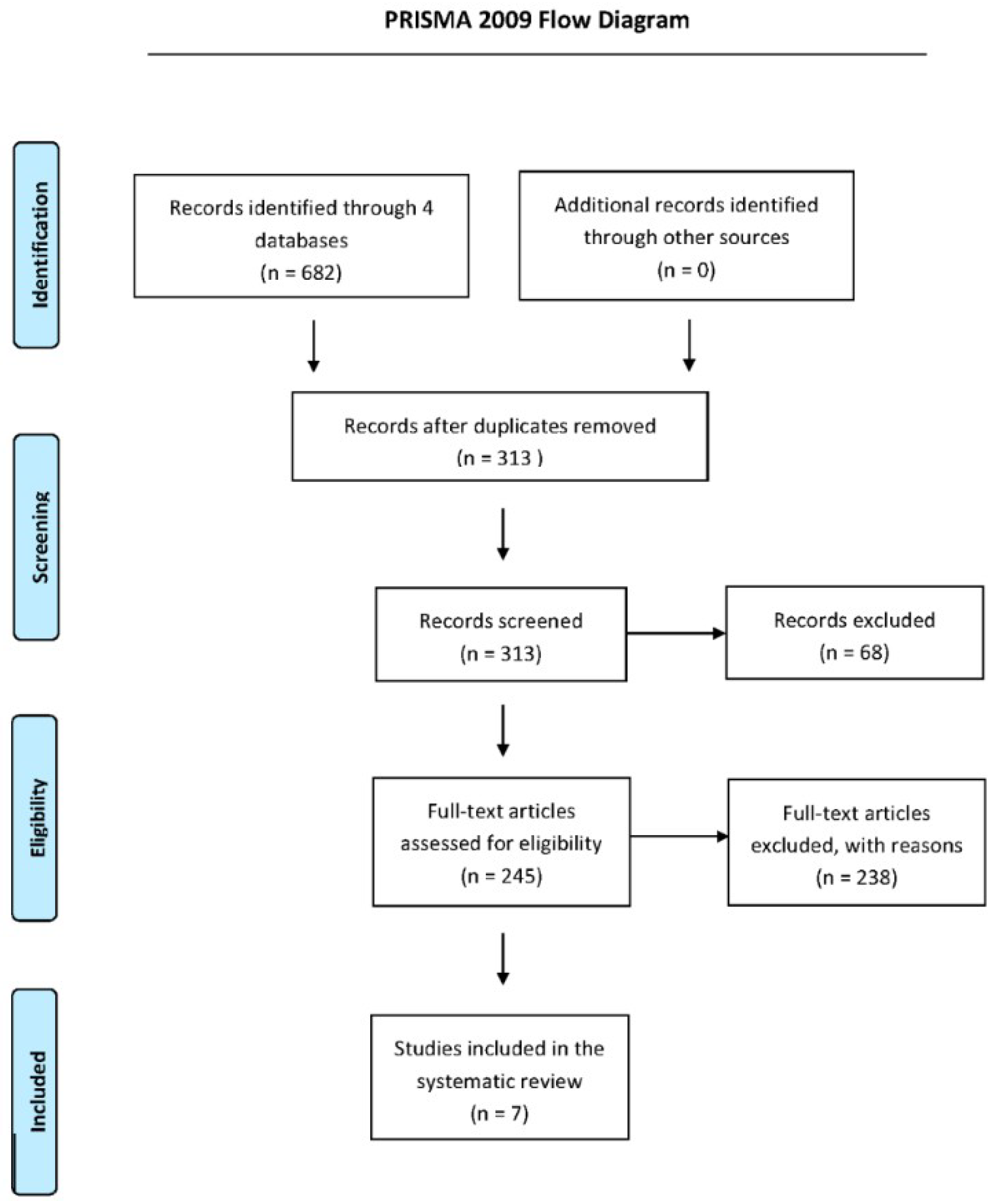
| Author | Country | Intervention/Control | Participants at Baseline (n) | Follow Up TIME (months) | Diabetes Inclusion Criteria | Periodontal Inclusion Criteria |
|---|---|---|---|---|---|---|
| D’Aiuto et al. 2018 [11] | United Kingdom | SPT + NSPT | 133 | 12 | Type 2 DM for >6 months (WHO diagnostic criteria) | >20 periodontal pockets with PD > 4mm and alveolar bone loss > 30% |
| Supragingival SRP | 131 | |||||
| Masi et al. 2018 [14] | United Kingdom | NSPT | 27 | 6 | Diagnosed type 2 DM (WHO criteria) | >15 remaining teeth and >20 sites with PD >5mm |
| Supragingival SRP | 24 | |||||
| Kapellas et al. 2017 [12] | Australia | NSPT | 35 | 3 | HbA1c > 6.5% or >47.5 mmol/mol | Joint Centers for Disease Control and Prevention and American Academy of Periodontology case definition |
| No treatment | 27 | |||||
| Chen et al. 2012 [10] | China | NSPT | 45 | 6 | Type 2 DM for >12 months | American Academy of Periodontology criteria |
| No treatment | 44 | |||||
| Moeintaghavi et al. 2012 [15] | Iran | NSPT | 22 | 3 | HbA1c > 7% | Mild-moderate periodontitis- American Academy of Periodontology criteria |
| No treatment | 18 | |||||
| Sun et al. 2011 [16] | China | NSPT | 82 | 3 | Diagnosed type 2 DM for >12 months and HbA1c 7.5–9.5% | >20 remaining teeth, PD > 5 mm, more than 30% teeth CAL over 4 mm, or over 60% teeth with PD > 4 mm and CAL > 3 mm |
| No treatment | 75 | |||||
| Kiran et al. 2005 [13] | Turkey | NSPT | 22 | 3 | HbA1c 6–8% | Not specified |
| No treatment | 22 |
| Author | Groups | Baseline | 3 Month Follow Up | 6 Month Follow Up | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TC | TG | LDL | HDL | TC | TG | LDL | HDL | TC | TG | LDL | HDL | |||||||||||||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | |||
| D’Aiuto et al. 2018 [11] | Intervention | SPT+NSPT | 4.2 | 1 * | 1.6 | 1.2 * | 2.2 | 0.9 * | 1.2 | 0.4 * | 4.3 | 0.1 * | 1.7 | 0.1 * | 2.3 | 0.1 * | 1.3 | 0.0 * | ||||||||
| Control | SG SRP | 4.3 | 1.1 * | 1.6 | 1.1 * | 2.4 | 0.9 * | 1.3 | 0.4 * | 4.2 | 0.1 * | 1.5 | 0.1 * | 2.3 | 0.1 * | 1.2 | 0.0 * | |||||||||
| Masi et al. 2018 [14] | Intervention | NSPT | 4.3 | 1.1 | 1.48 | 1.13 | 2.3 | 0.9 | 1.3 | 0.4 | 4.2 | 0.9 | 1.4 | 0.9 | 2.2 | 0.7 | 1.4 | 0.4 | ||||||||
| Control | SG SRP | 4.3 | 1 | 2.3 | 2.6 | 2 | 0.9 | 1.3 | 0.4 | 4.1 | 1.1 | 2.2 | 1.9 | 2 | 0.8 | 1.2 | 0.5 | |||||||||
| Kapellas et al. 2017 [12] | Intervention | NSPT | 4.8 | 1.1 | 1 | 0.3 | 4.5 | 1 | 1 | 0.3 | ||||||||||||||||
| Control | No tx | 4.6 | 0.8 | 1.1 | 0.2 | 4.4 | 0.9 | 0.9 | 0.2 | |||||||||||||||||
| Chen et al. 2012 [10] | Intervention | NSPT | 2.63 | 1.32 | 6.02 | 1.57 | 3.5 | 1.3 | 1.31 | 0.46 | 2.2 | 1.33 | 5.61 | 1.41 | 3.37 | 1.3 | 1.23 | 0.39 | 2.15 | 1.93 | 5.26 | 1.41 | 3.04 | 1.23 | 1.14 | 0.39 |
| Control | No tx | 2.35 | 1.78 | 6.37 | 1.87 | 3.79 | 1.48 | 1.44 | 0.53 | 2.3 | 2.16 | 5.94 | 1.22 | 3.5 | 1.17 | 1.36 | 0.49 | 2.25 | 1.98 | 5.81 | 1.61 | 1.26 | 0.5 | 3.25 | 1.27 | |
| Moeintaghavi et al. 2012 [15] | Intervention | NSPT | 10.66 | 1.5 | 7.66 | 4.54 | 6.6 | 1.38 | 2.55 | 0.51 | 10.31 | 1.72 | 7.22 | 3.22 | 6.26 | 1.76 | 2.45 | 0.37 | ||||||||
| Control | No tx | 10.69 | 1.5 | 8.4 | 1.51 | 6.51 | 1.9 | 2.57 | 0.69 | 10.95 | 1.51 | 8.19 | 1.62 | 6.36 | 2.12 | 2.44 | 0.54 | |||||||||
| Sun et al. 2011 [16] | Intervention | NSPT | 2.07 | 0.69 | 3.32 | 0.71 | 1.17 | 0.29 | 1.85 | 0.64 | 3.21 | 0.76 | 1.23 | 0.33 | ||||||||||||
| Control | No tx | 2.1 | 0.68 | 3.37 | 0.74 | 1.15 | 0.28 | 2.08 | 0.66 | 3.31 | 0.75 | 1.16 | 0.3 | |||||||||||||
| Kiran et al. 2005 [13] | Intervention | NSPT | 10.4 | 2.13 | 7.6 | 5.5 | 6.31 | 1.35 | 2.87 | 0.77 | 10.17 | 1.73 | 6.82 | 3.1 | 6.15 | 1.73 | 2.94 | 0.85 | ||||||||
| Control | No tx | 9.95 | 1.95 | 7.26 | 3.8 | 5.96 | 1.81 | 2.54 | 0.73 | 10.57 | 2.07 | 9.17 | 5.95 | 5.94 | 2.16 | 2.85 | 0.79 | |||||||||
| Author | Groups | Baseline | 3-month Follow Up | 6-month Follow Up | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PD | BOP | PD | BOP | PD | BOP | |||||||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | |||
| D’Aiuto et al. 2018 [11] | Intervention | SPT + NSPT | 3.9 | 0.1 * | 65 | 2.0 * | 2.9 | 0.1 * | 33.0 | 2.0 * | ||||
| Control | SG SRP | 3.9 | 0.1 * | 63 | 2.0 * | 3.7 | 0.1 * | 57.0 | 2.0 * | |||||
| Masi et al. 2018 [14] | Intervention | NSPT | 3.9 | 0.8 | 70.0 | 20.0 | ||||||||
| Control | SG SRP | 3.6 | 0.7 | 72.0 | 15.0 | |||||||||
| Kapellas et al. 2017 [12] | Intervention | NSPT | ||||||||||||
| Control | No treatment | |||||||||||||
| Chen et al. 2012 [10] | Intervention | NSPT | 2.57 | 0.66 | 32.42 | 16.63 | 2.2 | 0.39 | 12.13 | 8.24 | 2.1 | 0.39 | 12.02 | 8.99 |
| Control | No treatment | 2.47 | 0.57 | 34.01 | 18.91 | 2.38 | 0.47 | 28.53 | 14.42 | 2.42 | 0.5 | 28.37 | 13.5 | |
| Moeintaghavi et al. 2012 [15] | Intervention | NSPT | 2.31 | 0.65 | 2.21 | 0.6 | ||||||||
| Control | No treatment | 2.06 | 0.24 | 2.33 | 0.3 | |||||||||
| Sun et al. 2011 [16] | Intervention | NSPT | 4.53 | 0.83 | 2.97 | 0.78 | ||||||||
| Control | No treatment | 4.49 | 0.85 | 4.28 | 0.81 | |||||||||
| Kiran et al. 2005 [13] | Intervention | NSPT | 2.29 | 0.49 | 54.38 | 18.75 | 1.8 | 0.25 | 23.9 | 12.73 | ||||
| Control | No treatment | 2.24 | 0.7 | 50.48 | 26.1 | 2.26 | 0.63 | 51.91 | 27.38 | |||||
| Total | Intervention | 3.3 | 0.6 | 55.5 | 16.1 | 2.3 | 0.5 | 18.0 | 10.7 | 2.6 | 0.5 | 27.0 | 13.1 | |
| Control | 3.1 | 0.6 | 54.9 | 17.8 | 2.8 | 0.3 | 40.2 | 21.9 | 3.1 | 0.5 | 47.8 | 13.0 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garde, S.; Akhter, R.; Nguyen, M.A.; Chow, C.K.; Eberhard, J. Periodontal Therapy for Improving Lipid Profiles in Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2019, 20, 3826. https://doi.org/10.3390/ijms20153826
Garde S, Akhter R, Nguyen MA, Chow CK, Eberhard J. Periodontal Therapy for Improving Lipid Profiles in Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences. 2019; 20(15):3826. https://doi.org/10.3390/ijms20153826
Chicago/Turabian StyleGarde, Siddharth, Rahena Akhter, Mai Anh Nguyen, Clara K. Chow, and Joerg Eberhard. 2019. "Periodontal Therapy for Improving Lipid Profiles in Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis" International Journal of Molecular Sciences 20, no. 15: 3826. https://doi.org/10.3390/ijms20153826
APA StyleGarde, S., Akhter, R., Nguyen, M. A., Chow, C. K., & Eberhard, J. (2019). Periodontal Therapy for Improving Lipid Profiles in Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences, 20(15), 3826. https://doi.org/10.3390/ijms20153826







