The Formation and Therapeutic Update of Tumor-Associated Macrophages in Cervical Cancer
Abstract
1. Introduction
2. The Origins and Categories of Macrophages
2.1. Tissue-Resident Macrophages
2.2. Infiltrating Macrophages
3. The Molecules Involved in Forming TAMs
3.1. Tumor-Derived Molecules
3.1.1. Molecules Promoting the Differentiation of Monocytes to TAMs
3.1.2. Molecules Promoting the Activation of TAMs
3.1.3. The role of Oncoproteins on TAMs Formation in CC
3.2. T Cell-Derived Molecules
3.3. Seminal Plasma-Derived Molecules
3.4. Anaerobic Microenvironment
4. Biomarkers of TAMs
5. The Switch from M2 to M1-like Macrophages
6. Development of Therapy by Targeting TAMs
6.1. Macrophages and Its Prognostic Value in CC
6.2. Therapy Targeting the Differentiation of TAMs
6.3. Therapy Targeting the Improvement of TAMs Anti-Tumor Activity
7. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Naga, C.P.; Gurram, L.; Chopra, S.; Mahantshetty, U. The management of locally advanced cervical cancer. Curr. Opin. Oncol. 2018, 30, 323–329. [Google Scholar]
- Bosch, F.X.; Lorincz, A.; Munoz, N.; Meijer, C.J.; Shah, K.V. The causal relation between human papillomavirus and cervical cancer. J. Clin. Pathol. 2002, 55, 244–265. [Google Scholar] [CrossRef] [PubMed]
- Ruttkay-Nedecky, B.; Jimenez Jimenez, A.M.; Nejdl, L.; Chudobova, D.; Gumulec, J.; Masarik, M.; Adam, V.; Kizek, R. Relevance of infection with human papillomavirus: The role of the p53 tumor suppressor protein and E6/E7 zinc finger proteins (Review). Int. J. Oncol. 2013, 43, 1754–1762. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Cao, X. Characteristics and significance of the pre-metastatic niche. Cancer Cell 2016, 30, 668–681. [Google Scholar] [CrossRef] [PubMed]
- Schwertfeger, K.L.; Cowman, M.K.; Telmer, P.G.; Turley, E.A.; McCarthy, J.B. Hyaluronan, inflammation, and breast cancer progression. Front. Immunol. 2015, 6, 236. [Google Scholar] [CrossRef] [PubMed]
- Gunther, J. [Experiences with the macrophage electrophoresis mobility test (MEM test) in the diagnosis of malignant gynecological diseases]. Zent. Bakteriol. Orig. B 1975, 91, 529–539. [Google Scholar]
- Cho, J.W.; Kim, W.W.; Lee, Y.M.; Jeon, M.J.; Kim, W.G.; Song, D.E.; Park, Y.; Chung, K.W.; Hong, S.J.; Sung, T.Y. Impact of tumor-associated macrophages and BRAF(V600E) mutation on clinical outcomes in patients with various thyroid cancers. Head Neck 2019, 41, 686–691. [Google Scholar] [CrossRef]
- Hoeffel, G.; Chen, J.; Lavin, Y.; Low, D.; Almeida, F.F.; See, P.; Beaudin, A.E.; Lum, J.; Low, I.; Forsberg, E.C.; et al. C-Myb(+) erythro-myeloid progenitor-derived fetal monocytes give rise to adult tissue-resident macrophages. Immunity 2015, 42, 665–678. [Google Scholar] [CrossRef] [PubMed]
- Gibbings, S.L.; Goyal, R.; Desch, A.N.; Leach, S.M.; Prabagar, M.; Atif, S.M.; Bratton, D.L.; Janssen, W.; Jakubzick, C.V. Transcriptome analysis highlights the conserved difference between embryonic and postnatal-derived alveolar macrophages. Blood 2015, 126, 1357–1366. [Google Scholar] [CrossRef] [PubMed]
- Kratofil, R.M.; Kubes, P.; Deniset, J.F. Monocyte conversion during inflammation and injury. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Chanmee, T.; Ontong, P.; Konno, K.; Itano, N. Tumor-associated macrophages as major players in the tumor microenvironment. Cancers 2014, 6, 1670–1690. [Google Scholar] [CrossRef] [PubMed]
- Gautier, E.L.; The Immunological Genome Consortium; Shay, T.; Miller, J.; Greter, M.; Jakubzick, C.; Ivanov, S.; Helft, J.; Chow, A.; Elpek, K.G.; et al. Gene-expression profiles and transcriptional regulatory pathways that underlie the identity and diversity of mouse tissue macrophages. Nat. Immunol. 2012, 13, 1118–1128. [Google Scholar] [CrossRef] [PubMed]
- Rantakari, P.; Jäppinen, N.; Lokka, E.; Mokkala, E.; Gerke, H.; Peuhu, E.; Ivaska, J.; Elima, K.; Auvinen, K.; Salmi, M. Fetal liver endothelium regulates the seeding of tissue-resident macrophages. Nature 2016, 538, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Misharin, A.V.; Morales-Nebreda, L.; Reyfman, P.A.; Cuda, C.M.; Walter, J.M.; McQuattie-Pimentel, A.C.; Chen, C.-I.; Anekalla, K.R.; Joshi, N.; Williams, K.J.; et al. Monocyte-derived alveolar macrophages drive lung fibrosis and persist in the lung over the life span. J. Exp. Med. 2017, 214, 2387–2404. [Google Scholar] [CrossRef] [PubMed]
- James, K.; Harvey, J.; Bradbury, A.W.; Hargreave, T.B.; Cullen, R.T. The effect of seminal plasma on macrophage function—A possible contributory factor in sexually transmitted disease. AIDS Res. 1983, 1, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.T.; Teshima, H.; Shimizu, Y.; Hasumi, K.; Masubuchi, K.; Suzuki, M. [Sizofiran and recombinant interferon γ stimulate peritoneal macrophages obtained from patients with gynecologic malignancies—Increased secretion of tumor necrosis factor, IL-1 and interferon-γ]. Gan Kagaku Ryoho 1990, 17, 1365–1369. [Google Scholar]
- Rojas, J.; Salazar, J.; Martinez, M.S.; Palmar, J.; Bautista, J.; Chavez-Castillo, M.; Gomez, A.; Bermúdez, V. Macrophage heterogeneity and plasticity: Impact of macrophage biomarkers on atherosclerosis. Scientifica (Cairo) 2015, 2015, 851252. [Google Scholar] [CrossRef]
- Gordon, S. Alternative activation of macrophages. Nat. Rev. Immunol. 2003, 3, 23–35. [Google Scholar] [CrossRef]
- Hao, N.-B.; Lü, M.-H.; Fan, Y.-H.; Cao, Y.-L.; Zhang, Z.-R.; Yang, S.-M. Macrophages in tumor microenvironments and the progression of tumors. Clin. Dev. Immunol. 2012, 2012, 948098. [Google Scholar] [CrossRef]
- Ma, X.; Wu, D.; Zhou, S.; Wan, F.; Liu, H.; Xu, X.; Xu, X.; Zhao, Y.; Tang, M. The pancreatic cancer secreted REG4 promotes macrophage polarization to M2 through EGFR/AKT/CREB pathway. Oncol. Rep. 2016, 35, 189–196. [Google Scholar] [CrossRef]
- Chen, P.-C.; Cheng, H.-C.; Wang, J.; Wang, S.-W.; Tai, H.-C.; Lin, C.-W.; Tang, C.-H. Prostate cancer-derived CCN3 induces M2 macrophage infiltration and contributes to angiogenesis in prostate cancer microenvironment. Oncotarget 2014, 5, 1595–1608. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Xu, Y.; Zhong, J.; Wang, H.; Weng, M.; Cheng, Q.; Wu, Q.; Sun, Z.; Jiang, H.; Zhu, M.; et al. MFHAS1 promotes colorectal cancer progress by regulating polarization of tumor-associated macrophages via STAT6 signaling pathway. Oncotarget 2016, 7, 78726–78735. [Google Scholar] [CrossRef] [PubMed]
- Mazzoni, M.; Mauro, G.; Erreni, M.; Romeo, P.; Minna, E.; Vizioli, M.G.; Belgiovine, C.; Rizzetti, M.G.; Pagliardini, S.; Avigni, R.; et al. Senescent thyrocytes and thyroid tumor cells induce M2-like macrophage polarization of human monocytes via a PGE2-dependent mechanism. J. Exp. Clin. Cancer Res. 2019, 38, 208. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K. [Effect of gynecologic cancer sera on functions of monocyte from healthy volunteers]. Nihon Sanka Fujinka Gakkai Zasshi 1982, 34, 1528–1534. [Google Scholar] [PubMed]
- De Vos van Steenwijk, P.J.; Ramwadhdoebe, T.H.; Goedemans, R.; Doorduijn, E.M.; van Ham, J.J.; Gorter, A.; van Hall, T.; Kuijjer, M.L.; van Poelgeest, M.I.; van der Burg, S.H.; et al. Tumor-infiltrating CD14-positive myeloid cells and CD8-positive T-cells prolong survival in patients with cervical carcinoma. Int. J. Cancer 2013, 133, 2884–2894. [Google Scholar] [CrossRef]
- Pedraza-Brindis, E.J.; Sánchez-Reyes, K.; Hernández-Flores, G.; Bravo-Cuellar, A.; Jave-Suárez, L.F.; Aguilar-Lemarroy, A.; Gómez-Lomelí, P.; López-López, B.A.; Ortiz-Lazareno, P.C. Culture supernatants of cervical cancer cells induce an M2 phenotypic profile in THP-1 macrophages. Cell Immunol. 2016, 310, 42–52. [Google Scholar] [CrossRef]
- Li, L.; Yu, S.; Zang, C. Low necroptosis process predicts poor treatment outcome of human papillomavirus positive cervical cancers by decreasing tumor-associated macrophages M1 polarization. Gynecol. Obstet. Investig. 2018, 83, 259–267. [Google Scholar] [CrossRef]
- Heusinkveld, M.; van Steenwijk, P.J.; Goedemans, R.; Ramwadhdoebe, T.H.; Gorter, A.; Welters, M.J.; van Hall, T.; van der Burg, S.H. M2 macrophages induced by prostaglandin E2 and IL-6 from cervical carcinoma are switched to activated M1 macrophages by CD4+ Th1 cells. J. Immunol. 2011, 187, 1157–1165. [Google Scholar] [CrossRef]
- Stelma, T.; Leaner, V.D. KPNB1-mediated nuclear import is required for motility and inflammatory transcription factor activity in cervical cancer cells. Oncotarget 2017, 8, 32833–32847. [Google Scholar] [CrossRef]
- Sierra-Filardi, E.; Nieto, C.; Domínguez-Soto, Á.; Barroso, R.; Sánchez-Mateos, P.; Puig-Kroger, A.; López-Bravo, M.; Joven, J.; Ardavín, C.; Rodríguez-Fernández, J.L.; et al. CCL2 shapes macrophage polarization by GM-CSF and M-CSF: Identification of CCL2/CCR2-dependent gene expression profile. J. Immunol. 2014, 192, 3858–3867. [Google Scholar] [CrossRef]
- Riley, J.K.; Takeda, K.; Akira, S.; Schreiber, R.D. Interleukin-10 receptor signaling through the JAK-STAT pathway. Requirement for two distinct receptor-derived signals for anti-inflammatory action. J. Biol. Chem. 1999, 274, 16513–16521. [Google Scholar] [CrossRef] [PubMed]
- Ramírez de Arellano, A.; Riera Leal, A.; Lopez-Pulido, E.I.; González-Lucano, L.R.; Macías Barragan, J.; del Toro Arreola, S.; García-Chagollan, M.; Palafox-Sánchez, C.A.; Muñoz-Valle, J.F.; Pereira-Suárez, A.L. A 60 kDa prolactin variant secreted by cervical cancer cells modulates apoptosis and cytokine production. Oncol. Rep. 2018, 39, 1253–1260. [Google Scholar] [CrossRef] [PubMed]
- Sodhi, A.; Tripathi, A. Prolactin and growth hormone induce differential cytokine and chemokine profile in murine peritoneal macrophages in vitro: Involvement of p-38 MAP kinase, STAT3 and NF-kappaB. Cytokine 2008, 41, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Cameron, D.J. Specificity of macrophage-mediated cytotoxicity: Role of target and effector cell fucose. Immunol. Lett. 1985, 11, 39–44. [Google Scholar] [CrossRef]
- Rigo, A.; Gottardi, M.; Zamò, A.; Mauri, P.; Bonifacio, M.; Krampera, M.; Damiani, E.; Pizzolo, G.; Vinante, F. Macrophages may promote cancer growth via a GM-CSF/HB-EGF paracrine loop that is enhanced by CXCL12. Mol. Cancer 2010, 9, 273. [Google Scholar] [CrossRef] [PubMed]
- Yaddanapudi, K.; Putty, K.; Rendon, B.E.; Lamont, G.J.; Faughn, J.D.; Satoskar, A.; Lasnik, A.; Eaton, J.W.; Mitchell, R.A. Control of tumor-associated macrophage alternative activation by macrophage migration inhibitory factor. J. Immunol. 2013, 190, 2984–2993. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Bae, J.S. Tumor-associated macrophages and neutrophils in tumor microenvironment. Mediat. Inflamm. 2016, 2016, 6058147. [Google Scholar] [CrossRef]
- Nees, M.; Geoghegan, J.M.; Hyman, T.; Frank, S.; Miller, L.; Woodworth, C.D. Papillomavirus type 16 oncogenes downregulate expression of interferon-responsive genes and upregulate proliferation-associated and NF-kappaB-responsive genes in cervical keratinocytes. J. Virol. 2001, 75, 4283–4296. [Google Scholar] [CrossRef]
- Divya, C.S.; Pillai, M.R. Antitumor action of curcumin in human papillomavirus associated cells involves downregulation of viral oncogenes, prevention of NFkB and AP-1 translocation, and modulation of apoptosis. Mol. Carcinog. 2006, 45, 320–332. [Google Scholar] [CrossRef]
- Zijlmans, H.J.; Fleuren, G.J.; Baelde, H.J.; Eilers, P.H.; Kenter, G.G.; Gorter, A. The absence of CCL2 expression in cervical carcinoma is associated with increased survival and loss of heterozygosity at 17q11.2. J. Pathol. 2006, 208, 507–517. [Google Scholar] [CrossRef]
- Alcocer-González, J.M.; Berumen, J.; Tamez-Guerra, R.; Bermúdez-Morales, V.; Peralta-Zaragoza, O.; Hernández-Pando, R.; Moreno, J.; Gariglio, P.; Madrid-Marina, V. In vivo expression of immunosuppressive cytokines in human papillomavirus-transformed cervical cancer cells. Viral. Immunol. 2006, 19, 481–491. [Google Scholar] [CrossRef] [PubMed]
- Farzaneh, F.; Roberts, S.; Mandal, D.; Ollier, B.; Winters, U.; Kitchener, H.; Brabin, L. The IL-10-1082G polymorphism is associated with clearance of HPV infection. BJOG 2006, 113, 961–964. [Google Scholar] [CrossRef] [PubMed]
- Brower, V. Researchers attempting to define role of cytokines in cancer risk. J. Natl. Cancer Inst. 2005, 97, 1175–1177. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mantovani, A.; Sica, A.; Sozzani, S.; Allavena, P.; Vecchi, A.; Locati, M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. 2004, 25, 677–686. [Google Scholar] [CrossRef]
- Peghini, B.C.; Abdalla, D.R.; Barcelos, A.C.M.; Teodoro, L.D.G.V.L.; Murta, E.F.C.; Michelin, M.A. Local cytokine profiles of patients with cervical intraepithelial and invasive neoplasia. Hum. Immunol. 2012, 73, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Riteau, B.; Rouas-Freiss, N.; Menier, C.; Paul, P.; Dausset, J.; Carosella, E.D. HLA-G2, -G3, and -G4 isoforms expressed as nonmature cell surface glycoproteins inhibit NK and antigen-specific CTL cytolysis. J. Immunol. 2001, 166, 5018–5026. [Google Scholar] [CrossRef]
- Rodríguez, J.A.; Galeano, L.; Palacios, D.M.; Gómez, C.; Serrano, M.L.; Bravo, M.M.; Combita, A.L. Altered HLA class I and HLA-G expression is associated with IL-10 expression in patients with cervical cancer. Pathobiology 2012, 79, 72–83. [Google Scholar] [CrossRef] [PubMed]
- Vesce, F.; Biondi, C. α-l-Fucosidase activity in endometrial, cervical and ovarian cancer. Eur. J. Gynaecol. Oncol. 1983, 4, 135–138. [Google Scholar] [PubMed]
- Chopra, V.; Dinh, T.V.; Hannigan, E.V. Circulating serum levels of cytokines and angiogenic factors in patients with cervical cancer. Cancer Investig. 1998, 16, 152–159. [Google Scholar] [CrossRef]
- Zijlmans, H.J.; Fleuren, G.J.; Baelde, H.J.; Eilers, P.H.; Kenter, G.G.; Gorter, A. Role of tumor-derived proinflammatory cytokines GM-CSF, TNF-alpha, and IL-12 in the migration and differentiation of antigen-presenting cells in cervical carcinoma. Cancer 2007, 109, 556–565. [Google Scholar] [CrossRef]
- Krockenberger, M.; Engel, J.B.; Kolb, J.; Dombrowsky, Y.; Häusler, S.F.; Kohrenhagen, N.; Dietl, J.; Wischhusen, J.; Honig, A. Macrophage migration inhibitory factor expression in cervical cancer. J. Cancer Res. Clin. Oncol. 2010, 136, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Beswick, E.J.; Reyes, V.E. CD74 in antigen presentation, inflammation, and cancers of the gastrointestinal tract. World J. Gastroenterol. 2009, 15, 2855–2861. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Lian, J.; Tao, H.; Shang, H.; Zhang, L. Correlation of macrophage migration inhibitory factor gene polymorphism with the risk of early-stage cervical cancer and lymphatic metastasis. Oncol. Lett. 2011, 2, 1261–1267. [Google Scholar] [CrossRef] [PubMed]
- Cheng, R.J.; Deng, W.G.; Niu, C.B.; Li, Y.Y.; Fu, Y. Expression of macrophage migration inhibitory factor and CD74 in cervical squamous cell carcinoma. Int. J. Gynecol. Cancer 2011, 21, 1004–1012. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.Z.; Dai, B.; Chen, J.; Luo, Q.; Liu, X.Y.; Lin, Q.X.; Li, X.H.; Huang, W.; Yu, X.Y. Loss of macrophage migration inhibitory factor impairs the growth properties of human HeLa cervical cancer cells. Cell Prolif. 2011, 44, 582–590. [Google Scholar] [CrossRef] [PubMed]
- Corraliza, I.M.; Soler, G.; Eichmann, K.; Modolell, M. Arginase induction by suppressors of nitric oxide synthesis (IL-4, IL-10 and PGE2) in murine bone-marrow-derived macrophages. Biochem. Biophys. Res. Commun. 1995, 206, 667–673. [Google Scholar] [CrossRef]
- Taylor, P.; Martinez-Pomares, L.; Stacey, M.; Lin, H.-H.; Brown, G.; Gordon, S.; Taylor, P. Macrophage receptors and immune recognition. Annu. Rev. Immunol. 2005, 23, 901–944. [Google Scholar] [CrossRef]
- Chen, Z.; Terai, M.; Fu, L.; Herrero, R.; DeSalle, R.; Burk, R.D. Diversifying selection in human papillomavirus type 16 lineages based on complete genome analyses. J. Virol. 2005, 79, 7014–7023. [Google Scholar] [CrossRef]
- Bravo, I.G.; Felez-Sanchez, M. Papillomaviruses: Viral evolution, cancer and evolutionary medicine. Evol. Med. Public Health 2015, 2015, 32–51. [Google Scholar] [CrossRef]
- Doorbar, J. The E4 protein; structure, function and patterns of expression. Virology 2013, 445, 80–98. [Google Scholar] [CrossRef]
- Galloway, D.A.; Laimins, L.A. Human papillomaviruses: Shared and distinct pathways for pathogenesis. Curr. Opin. Virol. 2015, 14, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Doorbar, J.; Egawa, N.; Griffin, H.; Kranjec, C.; Murakami, I. Human papillomavirus molecular biology and disease association. Rev. Med. Virol. 2015, 25 (Suppl. 1), 2–23. [Google Scholar] [CrossRef] [PubMed]
- Wallace, N.A.; Galloway, D.A. Novel functions of the human papillomavirus E6 oncoproteins. Annu. Rev. Virol. 2015, 2, 403–423. [Google Scholar] [CrossRef] [PubMed]
- Riethdorf, S.; Riethdorf, L.; Richter, N.; Loning, T. Expression of the MCP-1 gene and the HPV 16 E6/E7 oncogenes in squamous cell carcinomas of the cervix uteri and metastases. Pathobiology 1998, 66, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Berti, F.C.B.; Pereira, A.P.L.; Cebinelli, G.C.M.; Trugilo, K.P.; de Oliveira, K.B. The role of interleukin 10 in human papilloma virus infection and progression to cervical carcinoma. Cytokine Growth Factor Rev. 2017, 34, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Pahler, J.C.; Tazzyman, S.; Erez, N.; Chen, Y.-Y.; Murdoch, C.; Nozawa, H.; Lewis, C.E.; Hanahan, D. Plasticity in tumor-promoting inflammation: Impairment of macrophage recruitment evokes a compensatory neutrophil response. Neoplasia 2008, 10, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.M.; Kim, S.H.; Lee, Y.I.; Seo, M.; Kim, S.Y.; Song, Y.S.; Kim, W.H.; Juhnn, Y.S. Human papillomavirus E5 protein induces expression of the EP4 subtype of prostaglandin E2 receptor in cyclic AMP response element-dependent pathways in cervical cancer cells. Carcinogenesis 2009, 30, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Subbaramaiah, K.; Dannenberg, A.J. Cyclooxygenase-2 transcription is regulated by human papillomavirus 16 E6 and E7 oncoproteins: Evidence of a corepressor/coactivator exchange. Cancer Res. 2007, 67, 3976–3985. [Google Scholar] [CrossRef] [PubMed]
- Bolpetti, A.; Silva, J.S.; Villa, L.L.; Lepique, A.P. Interleukin-10 production by tumor infiltrating macrophages plays a role in Human Papillomavirus 16 tumor growth. BMC Immunol. 2010, 11, 27. [Google Scholar] [CrossRef]
- Liang, F.; Kina, S.; Takemoto, H.; Matayoshi, A.; Phonaphonh, T.; Sunagawa, N.; Arakaki, K.; Arasaki, A.; Kuang, H.; Sunakawa, H. HPV16E6-dependent c-fos expression contributes to AP-1 complex formation in SiHa cells. Mediat. Inflamm. 2011, 2011, 263216. [Google Scholar] [CrossRef]
- Balkwill, F.; Mantovani, A.; Balkwill, F. Inflammation and cancer: Back to Virchow? Lancet 2001, 357, 539–545. [Google Scholar] [CrossRef]
- Tartour, E.; Fossiez, F.; Joyeux, I.; Galinha, A.; Gey, A.; Claret, E.; Sastre-Garau, X.; Couturier, J.; Mosseri, V.; Vives, V.; et al. Interleukin 17, a T-cell-derived cytokine, promotes tumorigenicity of human cervical tumors in nude mice. Cancer Res. 1999, 59, 3698–3704. [Google Scholar] [PubMed]
- Yang, W.; Lu, Y.-P.; Yang, Y.-Z.; Kang, J.-R.; Jin, Y.-D.; Wang, H.-W.; Lu, Y.; Kang, J.; Jin, Y.; Wang, H. Expressions of programmed death (PD)-1 and PD-1 ligand (PD-L1) in cervical intraepithelial neoplasia and cervical squamous cell carcinomas are of prognostic value and associated with human papillomavirus status. J. Obstet. Gynaecol. Res. 2017, 43, 1602–1612. [Google Scholar] [CrossRef] [PubMed]
- Makita, N.; Hizukuri, Y.; Yamashiro, K.; Murakawa, M.; Hayashi, Y. IL-10 enhances the phenotype of M2 macrophages induced by IL-4 and confers the ability to increase eosinophil migration. Int. Immunol. 2015, 27, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Sales, K.J.; Katz, A.A.; Millar, R.P.; Jabbour, H.N. Seminal plasma activates cyclooxygenase-2 and prostaglandin E2 receptor expression and signalling in cervical adenocarcinoma cells. Mol. Hum. Reprod. 2002, 8, 1065–1070. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Muller, M.; Sales, K.J.; Katz, A.A.; Jabbour, H.N. Seminal plasma promotes the expression of tumorigenic and angiogenic genes in cervical adenocarcinoma cells via the E-series prostanoid 4 receptor. Endocrinology 2006, 147, 3356–3365. [Google Scholar] [CrossRef]
- Rametse, C.L.; Adefuye, A.O.; Olivier, A.J.; Curry, L.; Gamieldien, H.; Burgers, W.A.; Lewis, D.A.; Williamson, A.-L.; Katz, A.A.; Passmore, J.-A.S. Inflammatory cytokine profiles of semen influence cytokine responses of cervicovaginal epithelial cells. Front. Immunol. 2018, 9, 2721. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.J.; Wu, S.; Yan, R.M.; Fan, L.S.; Yu, L.; Zhang, Y.M.; Wei, W.F.; Zhou, C.F.; Wu, X.G.; Zhong, M.; et al. The role of the hypoxia-Nrp-1 axis in the activation of M2-like tumor-associated macrophages in the tumor microenvironment of cervical cancer. Mol. Carcinog. 2019, 58, 388–397. [Google Scholar] [CrossRef]
- Adurthi, S.; Mukherjee, G.; Krishnamurthy, H.; Sudhir, K.; Bafna, U.D.; Umadevi, K.; Jayshree, R.S. Functional tumor infiltrating TH1 and TH2 effectors in large early-stage cervical cancer are suppressed by regulatory T cells. Int. J. Gynecol. Cancer 2012, 22, 1130–1137. [Google Scholar] [CrossRef]
- Akiyama, S.K.; Yamada, S.S.; Yamada, K.M. Analysis of the role of glycosylation of the human fibronectin receptor. J. Biol. Chem. 1989, 264, 18011–18018. [Google Scholar]
- Jiang, S.; Yang, Y.; Fang, M.; Li, X.; Yuan, X.; Yuan, J. Co-evolution of tumor-associated macrophages and tumor neo-vessels during cervical cancer invasion. Oncol. Lett. 2016, 12, 2625–2631. [Google Scholar] [CrossRef] [PubMed]
- Schoppmann, S.F.; Birner, P.; Stöckl, J.; Kalt, R.; Ullrich, R.; Caucig, C.; Kriehuber, E.; Nagy, K.; Alitalo, K.; Kerjaschki, D. Tumor-associated macrophages express lymphatic endothelial growth factors and are related to peritumoral lymphangiogenesis. Am. J. Pathol. 2002, 161, 947–956. [Google Scholar] [CrossRef]
- Prata, T.T.M.; Bonin, C.M.; Ferreira, A.M.T.; Padovani, C.T.J.; Fernandes, C.E.D.S.; Machado, A.P.; Tozetti, I.A. Local immunosuppression induced by high viral load of human papillomavirus: Characterization of cellular phenotypes producing interleukin-10 in cervical neoplastic lesions. Immunology 2015, 146, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.; Cai, J.; Mao, M.; Fang, Y.; Huang, Z.; Jia, J.; Li, T.; Xu, L.; Wang, J.; Zhou, J.; et al. Tumor-associated macrophages induce lymphangiogenesis in cervical cancer via interaction with tumor cells. APMIS 2014, 122, 1059–1069. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Kang, S.; Chen, X.; Kim, E.J.; Kim, J.; Kim, N.; Kim, J.; Jin, M.M. Tumor suppression via paclitaxel-loaded drug carriers that target inflammation marker upregulated in tumor vasculature and macrophages. Biomaterials 2013, 34, 598–605. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, R.; Ozhegov, E.; Berg, Y.W.V.D.; Aronow, B.J.; Franco, R.S.; Palascak, M.B.; Fallon, J.T.; Ruf, W.; Versteeg, H.; Bogdanov, V.; et al. Splice variants of tissue factor promote monocyte-endothelial interactions by triggering the expression of cell adhesion molecules via integrin-mediated signaling. J. Thromb. Haemost. 2011, 9, 2087–2096. [Google Scholar] [CrossRef] [PubMed]
- Silveira, C.R.; Cipelli, M.; Manzine, C.; Rabelo-Santos, S.H.; Zeferino, L.C.; Rodriguez, G.R.; de Assis, J.B.; Hebster, S.; Bernadinelli, I.; Laginha, F.; et al. Swainsonine, an alpha-mannosidase inhibitor, may worsen cervical cancer progression through the increase in myeloid derived suppressor cells population. PLoS ONE 2019, 14, e0213184. [Google Scholar] [CrossRef]
- Lepique, A.P.; Daghastanli, K.R.; Cuccovia, I.M.; Villa, L.L. HPV16 tumor-associated macrophages suppress antitumor T cell responses. Clin. Cancer Res. 2009, 15, 4391–4400. [Google Scholar] [CrossRef]
- Sánchez-Reyes, K.; Bravo-Cuellar, A.; Hernández-Flores, G.; Lerma-Díaz, J.M.; Jave-Suarez, L.F.; Gómez-Lomeli, P.; de Celis, R.; Aguilar-Lemarroy, A.; Domínguez-Rodríguez, J.R.; Ortiz-Lazareno, P.C. Cervical cancer cell supernatants induce a phenotypic switch from U937-derived macrophage-activated M1 state into M2-like suppressor phenotype with change in Toll-like receptor profile. Biomed. Res. Int. 2014, 2014, 683068. [Google Scholar] [CrossRef]
- Petrillo, M.; Zannoni, G.F.; Martinelli, E.; Anchora, L.P.; Ferrandina, G.; Tropeano, G.; Fagotti, A.; Scambia, G. Polarisation of tumor-associated macrophages toward M2 phenotype correlates with poor response to chemoradiation and reduced survival in patients with locally advanced cervical cancer. PLoS ONE 2015, 10, e0136654. [Google Scholar] [CrossRef]
- Li, Y.; Huang, G.; Zhang, S. Associations between intratumoral and peritumoral M2 macrophage counts and cervical squamous cell carcinoma invasion patterns. Int. J. Gynaecol. Obstet. 2017, 139, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.J.; Han, L.F.; Wu, X.G.; Wei, W.F.; Wu, L.F.; Yi, H.Y.; Yan, R.M.; Bai, X.Y.; Zhong, M.; Yu, Y.H.; et al. Clinical significance of CD163+ and CD68+ tumor-associated macrophages in high-risk HPV-related cervical cancer. J. Cancer 2017, 8, 3868–3875. [Google Scholar] [CrossRef] [PubMed]
- Rotman, J.; Mom, C.H.; Jordanova, E.S.; de Gruijl, T.D.; Kenter, G.G. ‘DURVIT’: A phase-I trial of single low-dose durvalumab (Medi4736) IntraTumourally injected in cervical cancer: Safety, toxicity and effect on the primary tumour- and lymph node microenvironment. BMC Cancer 2018, 18, 888. [Google Scholar] [CrossRef]
- Swangphon, P.; Pientong, C.; Sunthamala, N.; Bumrungthai, S.; Azuma, M.; Kleebkaow, P.; Tangsiriwatthana, T.; Sangkomkamhang, U.; Kongyingyoes, B.; Ekalaksananan, T. Correlation of circulating CD64(+)/CD163(+) monocyte ratio and stroma/peri-tumoral CD163(+) monocyte density with human papillomavirus infected cervical lesion severity. Cancer Microenviron. 2017, 10, 77–85. [Google Scholar] [CrossRef]
- Li, L.; Ma, Y.; Liu, S.; Zhang, J.; Xu, X.Y. Interleukin 10 promotes immune response by increasing the survival of activated CD8(+) T cells in human papillomavirus 16-infected cervical cancer. Tumour Biol. 2016, 37, 16093–16101. [Google Scholar] [CrossRef] [PubMed]
- Heeren, A.M.; Punt, S.; Bleeker, M.C.; Gaarenstroom, K.N.; Van Der Velden, J.; Kenter, G.G.; De Gruijl, T.D.; Jordanova, E.S. Prognostic effect of different PD-L1 expression patterns in squamous cell carcinoma and adenocarcinoma of the cervix. Mod. Pathol. 2016, 29, 753–763. [Google Scholar] [CrossRef]
- Punt, S.; Thijssen, V.L.; Vrolijk, J.; De Kroon, C.D.; Gorter, A.; Jordanova, E.S. Galectin-1, -3 and -9 expression and clinical significance in squamous cervical cancer. PLoS ONE 2015, 10, e0129119. [Google Scholar] [CrossRef]
- Stone, S.C.; Rossetti, R.A.M.; Alvarez, K.L.F.; Carvalho, J.P.; Margarido, P.F.R.; Baracat, E.C.; Tacla, M.; Boccardo, E.; Yokochi, K.; Lorenzi, N.P.; et al. Lactate secreted by cervical cancer cells modulates macrophage phenotype. J. Leukoc. Biol. 2019, 105, 1041–1054. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, K.L.; Beldi, M.; Sarmanho, F.; Rossetti, R.A.; Silveira, C.R.; Mota, G.R.; Andreoli, M.A.; de Carvalho Caruso, E.D.; Kamillos, M.F.; Souza, A.M.; et al. Local and systemic immunomodulatory mechanisms triggered by Human Papillomavirus transformed cells: A potential role for G-CSF and neutrophils. Sci. Rep. 2017, 7, 9002. [Google Scholar] [CrossRef]
- Strachan, D.C.; Ruffell, B.; Oei, Y.; Bissell, M.J.; Coussens, L.M.; Pryer, N.; Daniel, D. CSF1R inhibition delays cervical and mammary tumor growth in murine models by attenuating the turnover of tumor-associated macrophages and enhancing infiltration by CD8(+) T cells. Oncoimmunology 2013, 2, e26968. [Google Scholar] [CrossRef]
- Li, Q.; Liu, L.; Zhang, Q.; Liu, S.; Ge, D.; You, Z. Interleukin-17 indirectly promotes M2 macrophage differentiation through stimulation of COX-2/PGE2 pathway in the cancer cells. Cancer Res. Treat. 2014, 46, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Sica, A.; Bronte, V. Altered macrophage differentiation and immune dysfunction in tumor development. J. Clin. Investig. 2007, 117, 1155–1166. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.V.; Ajay, A.K.; Mohammad, N.; Malvi, P.; Chaube, B.; Meena, A.S.; Bhat, M.K. Proteasomal inhibition sensitizes cervical cancer cells to mitomycin C-induced bystander effect: The role of tumor microenvironment. Cell Death Dis. 2015, 6, e1934. [Google Scholar] [CrossRef] [PubMed]
- Osawa, T.; Muramatsu, M.; Wang, F.; Tsuchida, R.; Kodama, T.; Minami, T.; Shibuya, M. Increased expression of histone demethylase JHDM1D under nutrient starvation suppresses tumor growth via down-regulating angiogenesis. Proc. Natl. Acad. Sci. USA 2011, 108, 20725–20729. [Google Scholar] [CrossRef] [PubMed]
- Heeren, A.M.; Kenter, G.G.; Jordanova, E.S.; de Gruijl, T.D. CD14(+) macrophage-like cells as the linchpin of cervical cancer perpetrated immune suppression and early metastatic spread: A new therapeutic lead? Oncoimmunology 2015, 4, e1009296. [Google Scholar] [CrossRef] [PubMed]
- Kawachi, A.; Yoshida, H.; Kitano, S.; Ino, Y.; Kato, T.; Hiraoka, N. Tumor-associated CD204(+) M2 macrophages are unfavorable prognostic indicators in uterine cervical adenocarcinoma. Cancer Sci. 2018, 109, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Berens, M.E.; Welander, C.E. Direct and indirect effects of human recombinant γ-interferon on tumor cells in a clonogenic assay. Cancer Res. 1986, 46, 1142–1147. [Google Scholar] [PubMed]
- Ohshika, Y.; Umesaki, N.; Sugawa, T. Immunomodulating capacity of the monocyte-macrophage system in patients with uterine cervical cancer. Nihon Sanka Fujinka Gakkai Zasshi 1988, 40, 601–608. [Google Scholar] [PubMed]
- Dijkgraaf, E.M.; Heusinkveld, M.; Tummers, B.; Vogelpoel, L.T.C.; Goedemans, R.; Jha, V.; Nortier, J.W.R.; Welters, M.J.P.; Kroep, J.R.; Van Der Burg, S.H. Chemotherapy alters monocyte differentiation to favor generation of cancer-supporting M2 macrophages in the tumor microenvironment. Cancer Res. 2013, 73, 2480–2492. [Google Scholar] [CrossRef] [PubMed]
- Van der Sluis, T.C.; Sluijter, M.; van Duikeren, S.; West, B.L.; Melief, C.J.; Arens, R.; van der Burg, S.H.; van Hall, T. Therapeutic peptide vaccine-induced CD8 T cells strongly modulate intratumoral macrophages required for tumor regression. Cancer Immunol. Res. 2015, 3, 1042–1051. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, K.; Nishimura, R.; Kinugasa, M.; Okamura, M.; Kimura, A.; Ohtsu, F.; Takeuchi, K.; Mizuhira, V. [Electron microscopic and immunological studies concerning the effect on the antitumor activity of sizofiran (SPG) combined with radiotherapy for cervical cancer]. Nihon Gan Chiryo Gakkai Shi 1990, 25, 2549–2561. [Google Scholar] [PubMed]
- Wetzel, K.; Menten, P.; Van Damme, J.; Giese, N.; Vecchi, A.; Sozzani, S.; Cornelis, J.J.; Rommelaere, J.; Dinsart, C. Transduction of human MCP-3 by a parvoviral vector induces leukocyte infiltration and reduces growth of human cervical carcinoma cell xenografts. J. Gene Med. 2001, 3, 326–337. [Google Scholar] [CrossRef] [PubMed]
- Nagy, T.; Glavinas, H.; Szincsák, N.; Hunyadi, J.; Jánossy, T.; Duda, E.; Vizler, C.; Juhász, I. Tumor cells expressing membrane-bound tumor necrosis factor activate macrophages and have a compromised growth in immunosuppressed and immunodeficient mice. Cancer Lett. 2003, 196, 49–56. [Google Scholar] [CrossRef]
- Rosales, C.; Graham, V.V.; Rosas, G.A.; Merchant, H.; Rosales, R. A recombinant vaccinia virus containing the papilloma E2 protein promotes tumor regression by stimulating macrophage antibody-dependent cytotoxicity. Cancer Immunol. Immunother. 2000, 49, 347–360. [Google Scholar] [CrossRef] [PubMed]
- Lou, P.-J.; Cheng, W.-F.; Chung, Y.-C.; Cheng, C.-Y.; Chiu, L.-H.; Young, T.-H. PMMA particle-mediated DNA vaccine for cervical cancer. J. Biomed. Mater. Res. A 2009, 88, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Jantová, S.; Paulovičová, E.; Paulovičová, L.; Janošková, M.; Pánik, M.; Milata, V. Immunobiological efficacy and immunotoxicity of novel synthetically prepared fluoroquinolone ethyl 6-fluoro-8-nitro-4-oxo-1,4-dihydroquinoline-3-carboxylate. Immunobiology 2018, 223, 81–93. [Google Scholar] [CrossRef] [PubMed]
- Davidson, B.; Goldberg, I.; Kopolovic, J. Inflammatory response in cervical intraepithelial neoplasia and squamous cell carcinoma of the uterine cervix. Pathol. Res. Pract. 1997, 193, 491–495. [Google Scholar] [CrossRef]
- Al-Saleh, W.; Delvenne, P.; Arrese, J.E.; Boniver, J.; Nikkels, A.F. Inverse modulation of intraepithelial Langerhans’ cells and stromal macrophage/dendrocyte populations in human papillomavirus-associated squamous intraepithelial lesions of the cervix. Virchows Arch. 1995, 427, 41–48. [Google Scholar] [CrossRef]
- Heller, D.S.; Hameed, M.; Cracchiolo, B.; Wiederkehr, M.; Scott, D.; Skurnick, J.; Ammar, N.; Lambert, W.C. Presence and quantification of macrophages in squamous cell carcinoma of the cervix. Int. J. Gynecol. Cancer 2003, 13, 67–70. [Google Scholar] [CrossRef]
- Davidson, B.; Goldberg, I.; Gotlieb, W.H.; Lerner-Geva, L.; Ben-Baruch, G.; Agulansky, L.; Novikov, I.; Kopolovic, J. Macrophage infiltration and angiogenesis in cervical squamous cell carcinoma—Clinicopathologic correlation. Acta Obstet. Gynecol. Scand. 1999, 78, 240–244. [Google Scholar] [CrossRef]
- Van Damme, J.; Proost, P.; Lenaerts, J.P.; Opdenakker, G. Structural and functional identification of two human, tumor-derived monocyte chemotactic proteins (MCP-2 and MCP-3) belonging to the chemokine family. J. Exp. Med. 1992, 176, 59–65. [Google Scholar] [CrossRef] [PubMed]
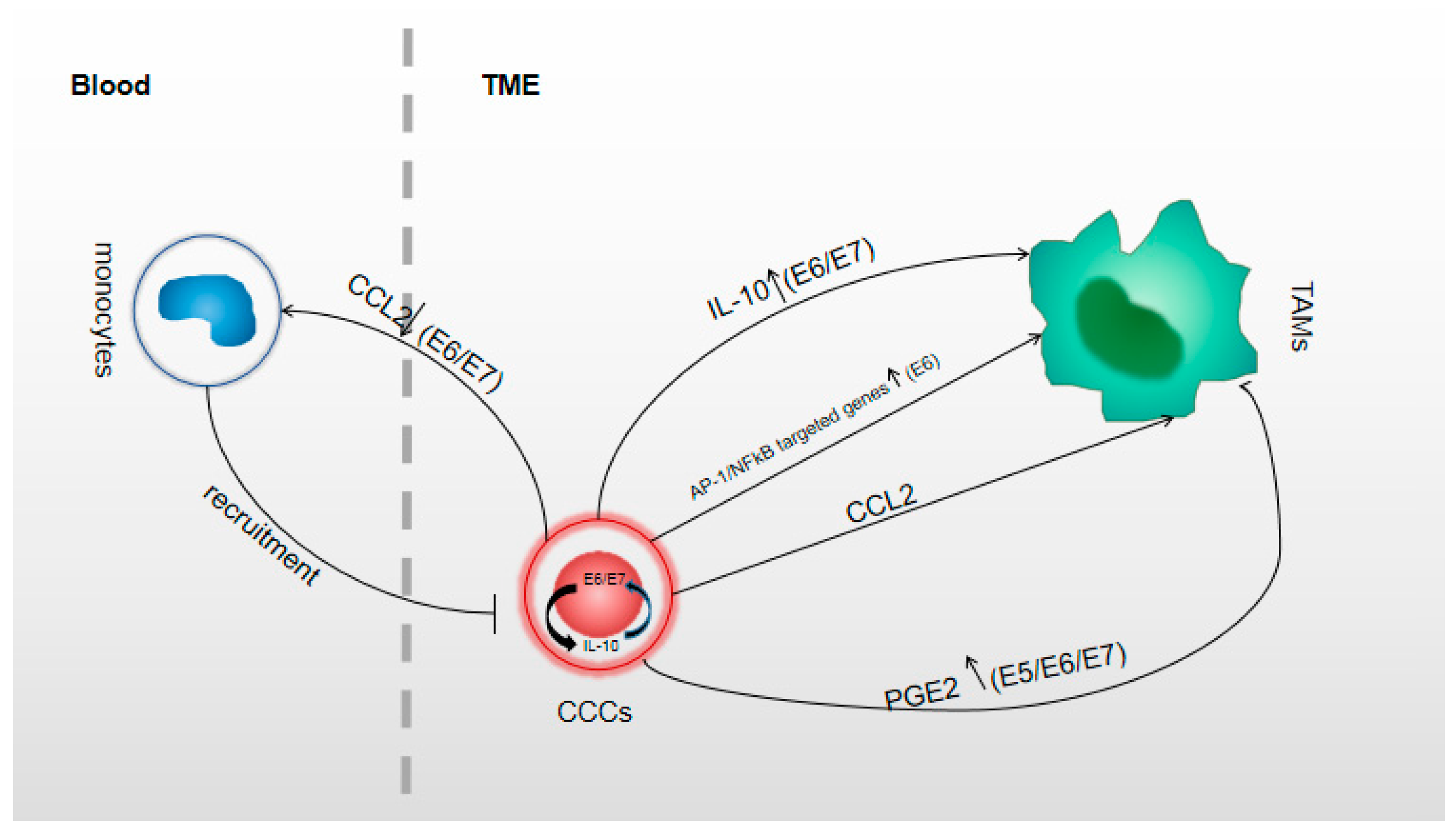
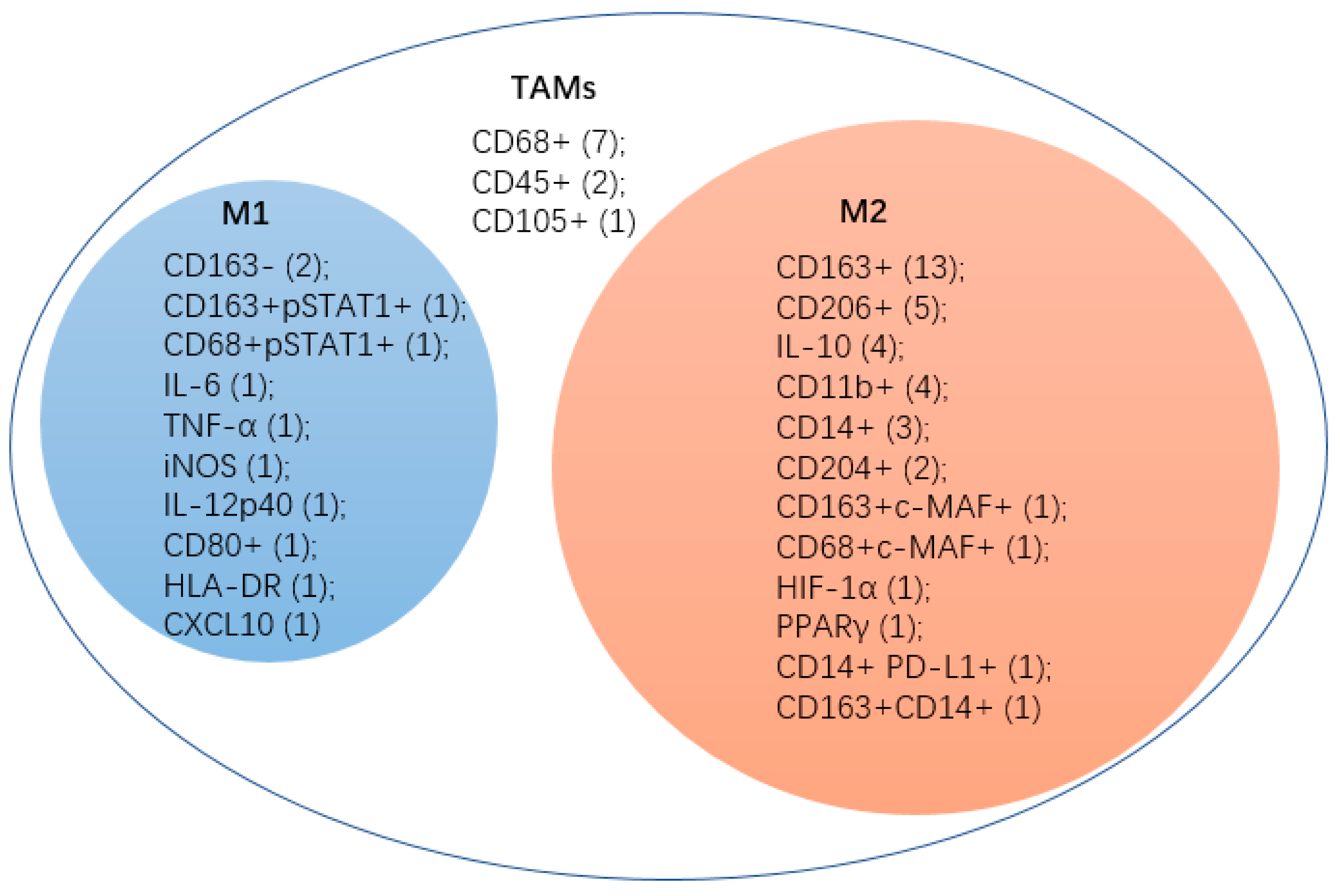
| Categories | Characteristics |
|---|---|
| Tissue-resident macrophages | |
| Infiltrating macrophages |
|
| Categories | Name of Molecules | Potential Mechanism |
|---|---|---|
| Molecules promoting the differentiation of monocytes to TAMs | PGE2 and IL-6 [28] | |
| Karyopherin β1 | Regulating the expression of transcriptional factors NFκB and AP-1 [29] | |
| CCL2 /MCP-1 | ||
| IL-10 | Inhibit the classic activation of macrophages through the JAK1/Tyk2/STAT3 pathway [31] | |
| PRL | ||
| Molecules promoting the activation of TAMs | FUCA-1 | |
| GM-CSF | Promoting TAMs to release pro-tumor factors [35] | |
| MIF [36] | ||
| IL-10 |
| Source | Name of Molecules | Potential Mechanism |
|---|---|---|
| T cell | IL-17 | |
| IL-4 | Promoting M2a subtype polarization by inducing a transcriptional factor KLF4 [73] | |
| IL-10 | Promoting M2c subtype polarization [44] | |
| IL-4 and IL-10 | Promoting M2a subtype polarization [74] | |
| Seminal plasma | Increasing the expression of COX2, PGE2, IL-6 in CC cells [75,76,77] | |
| Anaerobic microenvironment | Promoting M2 polarization by overexpressing Nrp-1 in CC cells [78] |
| Component | Potential Mechanism | |
|---|---|---|
| Targeting the differentiation of TAMs | CD4+ Th1 cells | Promoting TAMs to secret costimulatory molecules and the expression of CCR7 [28] |
| rIFN-γ | Promoting TAMs to produce a diffusible tumoricidal substance [107] |
| Molecules | Potential Mechanism | |
|---|---|---|
| Targeting the differentiation of TAMs | Ind. | Inhibiting the expression of PG [108] |
| tocilizumab | Blocking IL-6R [109] | |
| rIFN-γ | Simulating the role of IFN-γ to induce M2 to M1-like macrophages [16] | |
| SLP | Inducing T cells to inflow [110] | |
| Improvement in anti-tumor activity of TAMs | SPG | Inducing the cytotoxic activity of macrophage [111] |
| MCP-3 | Activating the phagocytic ability of macrophages [112] | |
| Pre-TNF | Increasing phagocytosis [113] | |
| bovine papilloma virus antibody | Inducing the cytotoxic activity of macrophage [114] | |
| PMMA | Stimulating TAMs to produce TNF-α [115] | |
| 6FN | Inducing TAMs to release anti-tumor cytokines [116] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Q.; Steger, A.; Mahner, S.; Jeschke, U.; Heidegger, H. The Formation and Therapeutic Update of Tumor-Associated Macrophages in Cervical Cancer. Int. J. Mol. Sci. 2019, 20, 3310. https://doi.org/10.3390/ijms20133310
Wang Q, Steger A, Mahner S, Jeschke U, Heidegger H. The Formation and Therapeutic Update of Tumor-Associated Macrophages in Cervical Cancer. International Journal of Molecular Sciences. 2019; 20(13):3310. https://doi.org/10.3390/ijms20133310
Chicago/Turabian StyleWang, Qun, Alexander Steger, Sven Mahner, Udo Jeschke, and Helene Heidegger. 2019. "The Formation and Therapeutic Update of Tumor-Associated Macrophages in Cervical Cancer" International Journal of Molecular Sciences 20, no. 13: 3310. https://doi.org/10.3390/ijms20133310
APA StyleWang, Q., Steger, A., Mahner, S., Jeschke, U., & Heidegger, H. (2019). The Formation and Therapeutic Update of Tumor-Associated Macrophages in Cervical Cancer. International Journal of Molecular Sciences, 20(13), 3310. https://doi.org/10.3390/ijms20133310






