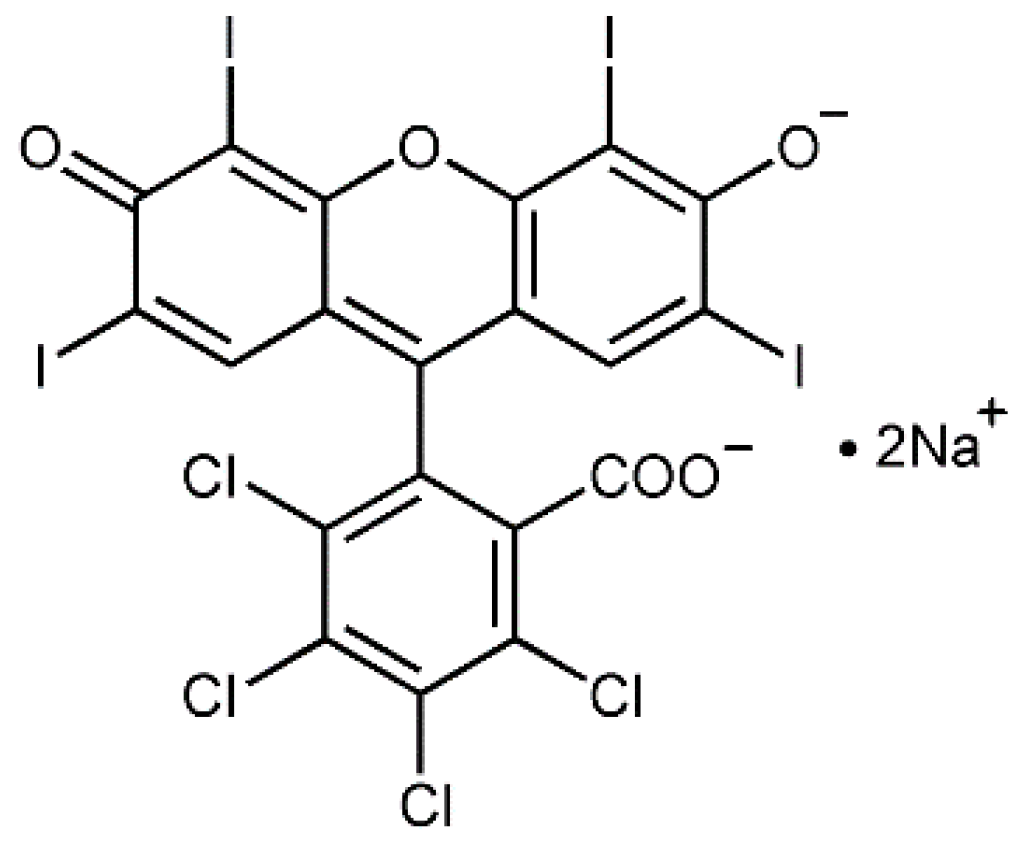
Dark Antibacterial Activity of Rose Bengal
Abstract
1. Introduction
2. Results and Discussion
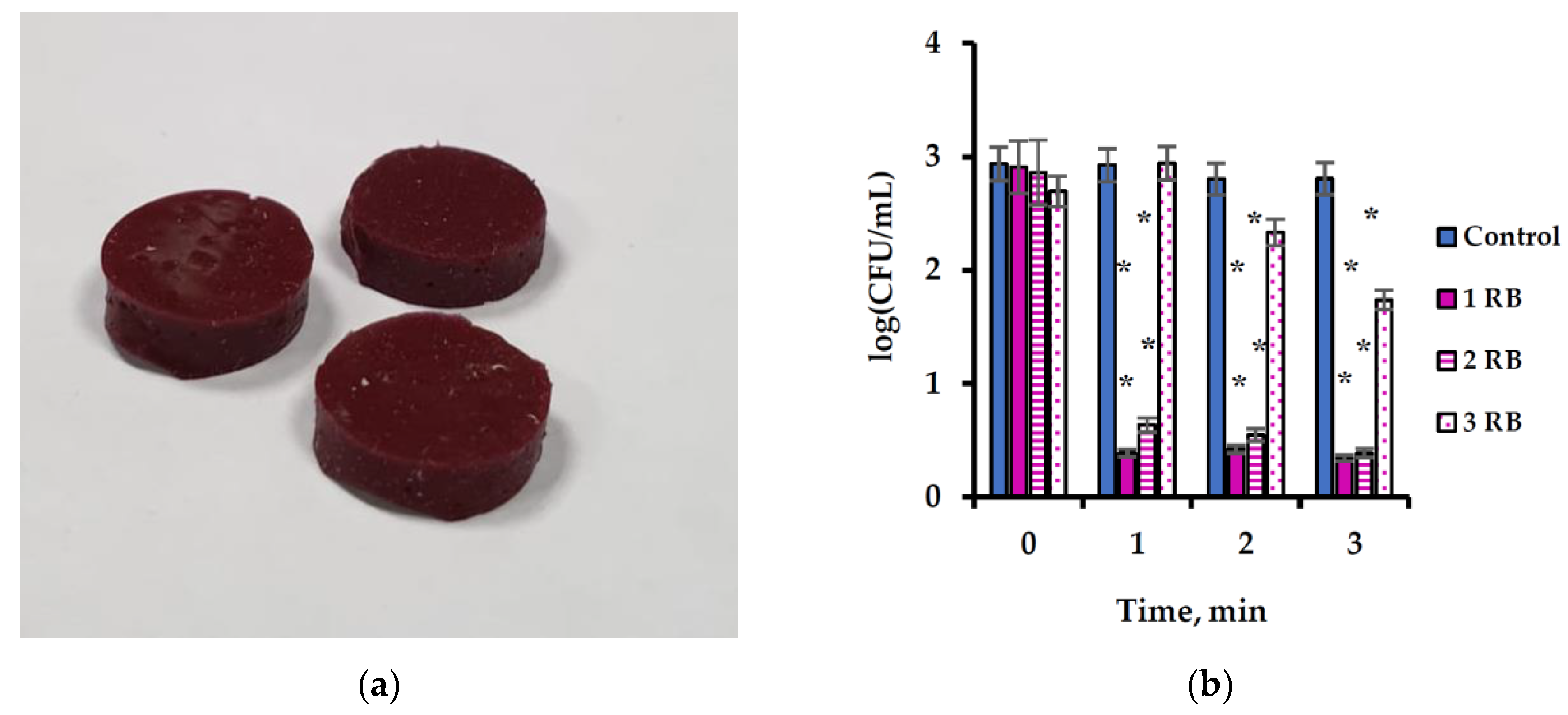
2.1. Dark Activity of Rose Bengal
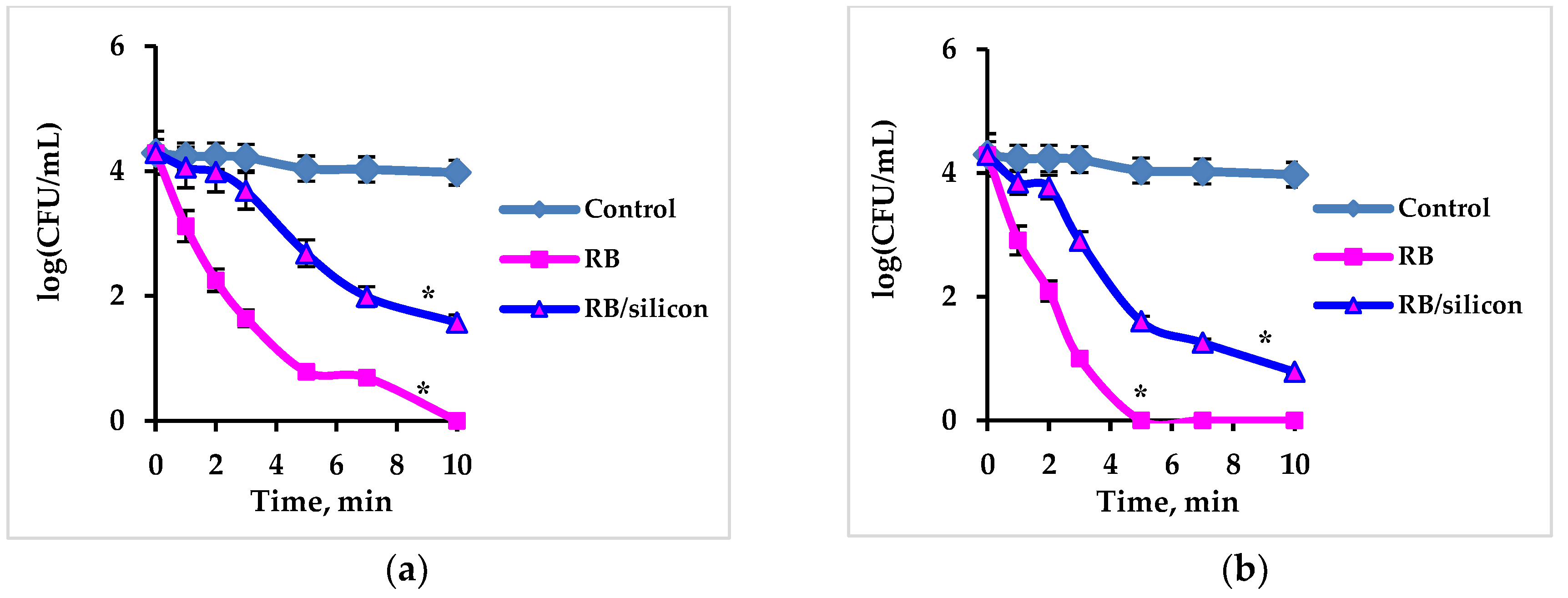
2.2. Ultrasonic Activation of Free and Immobilized Rose Bengal
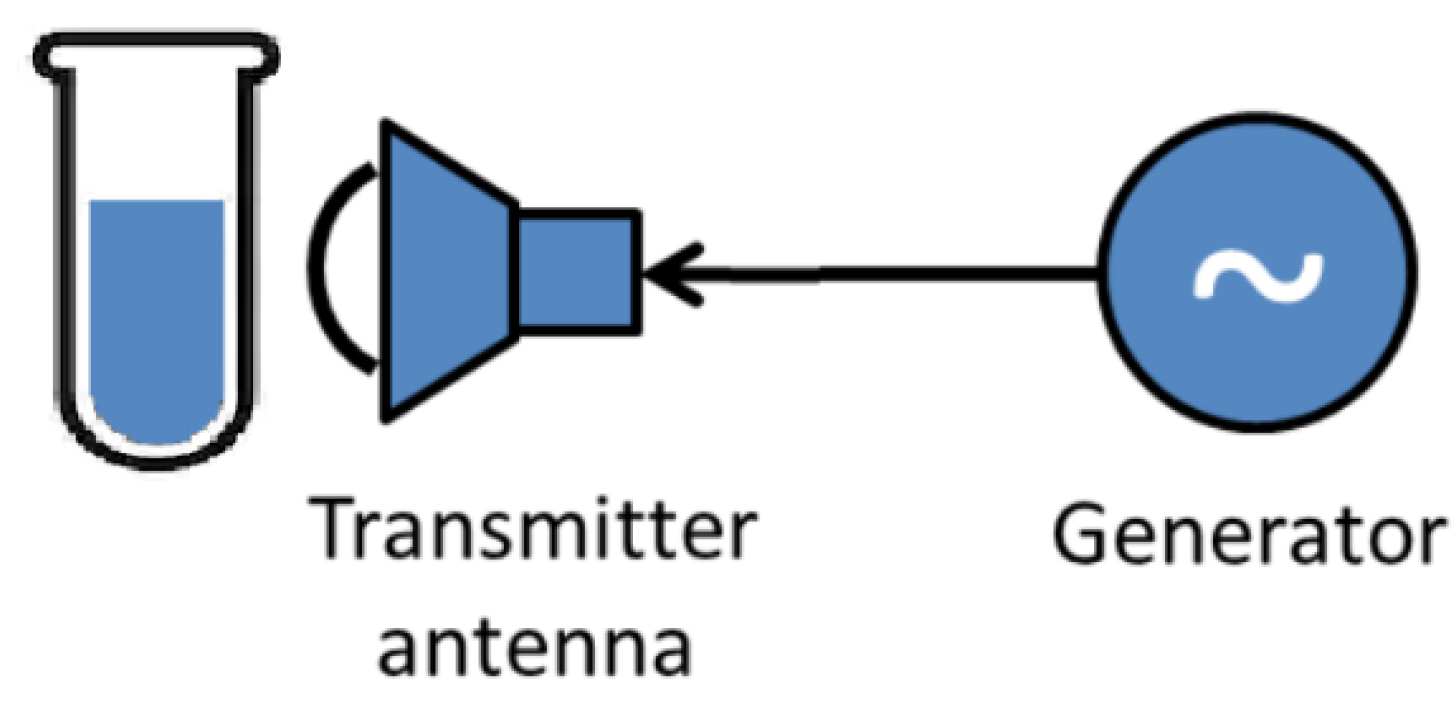
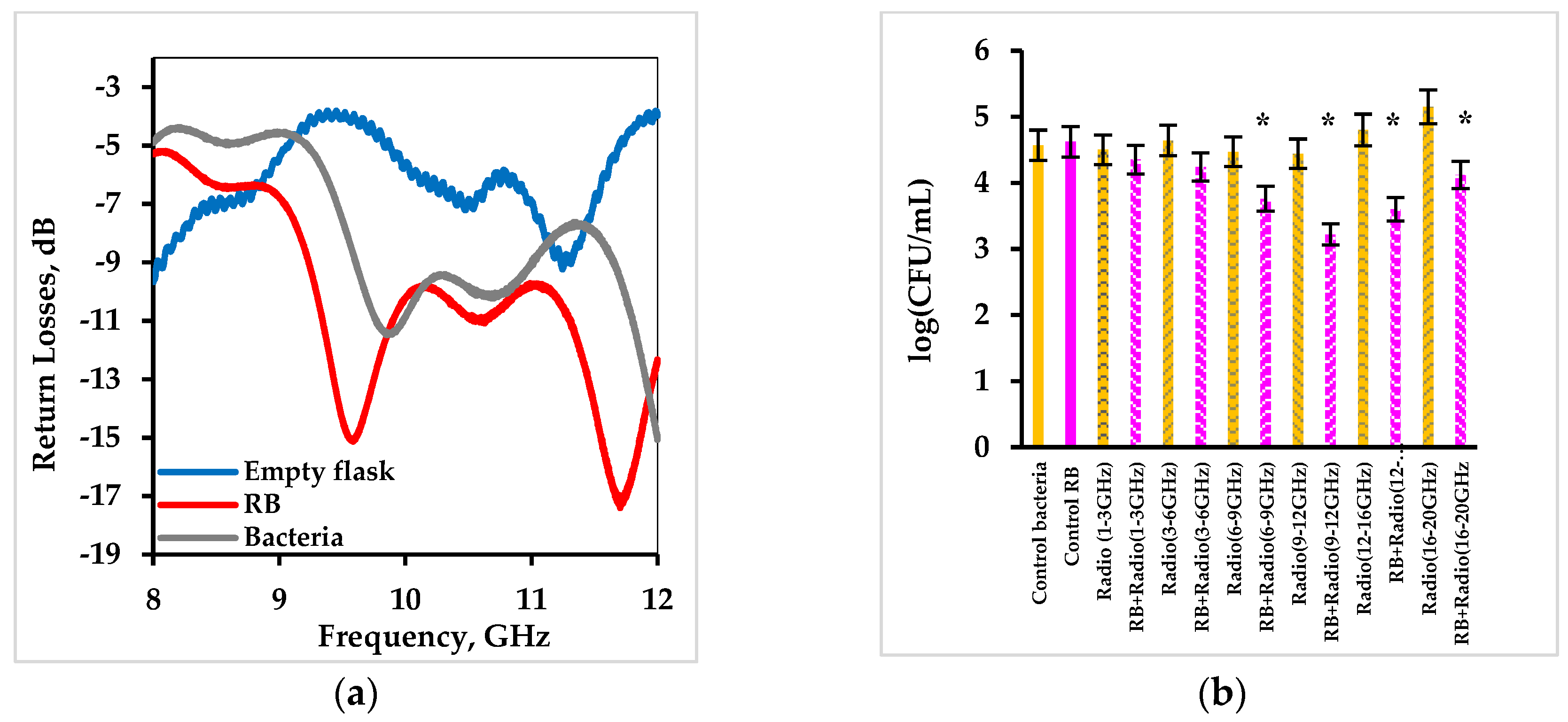
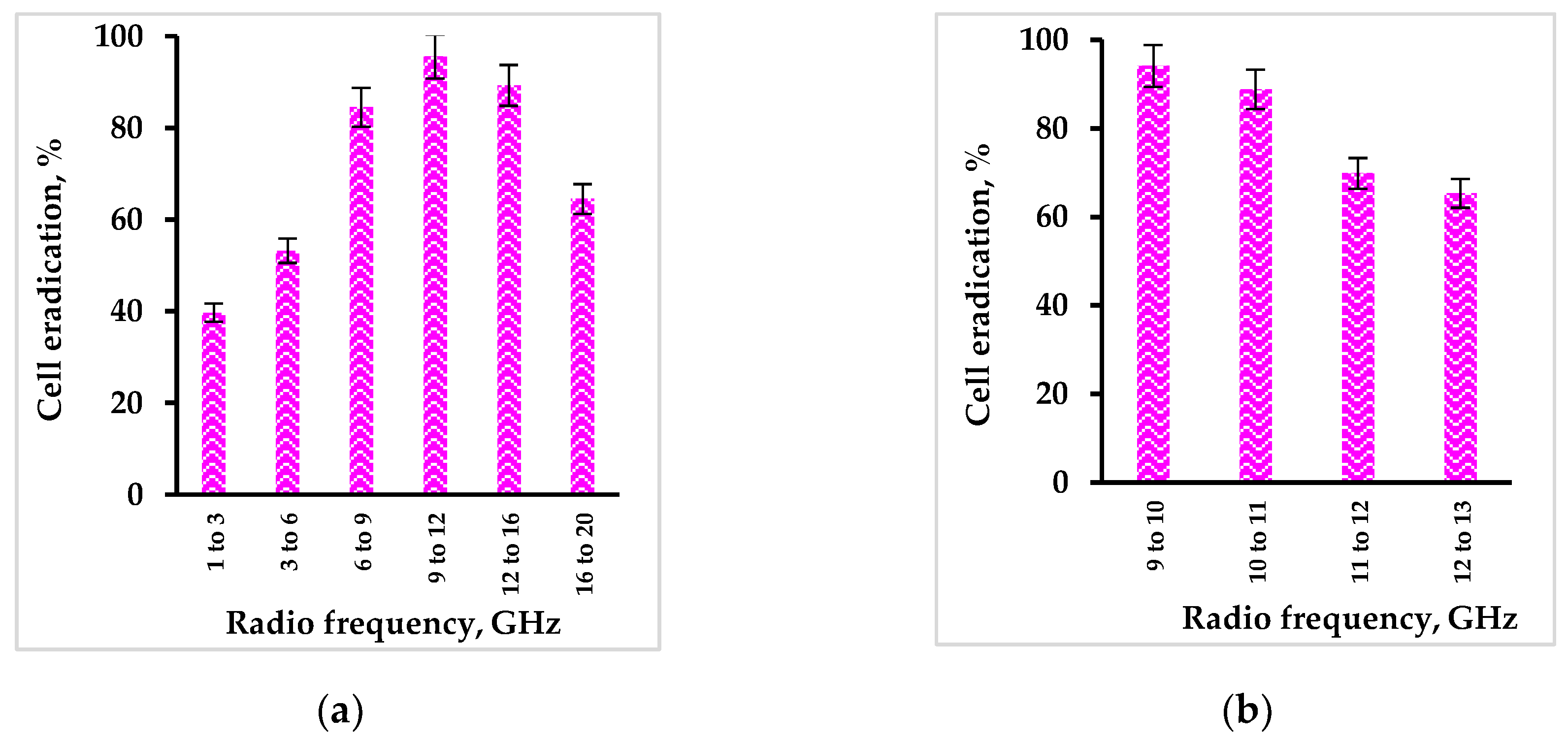
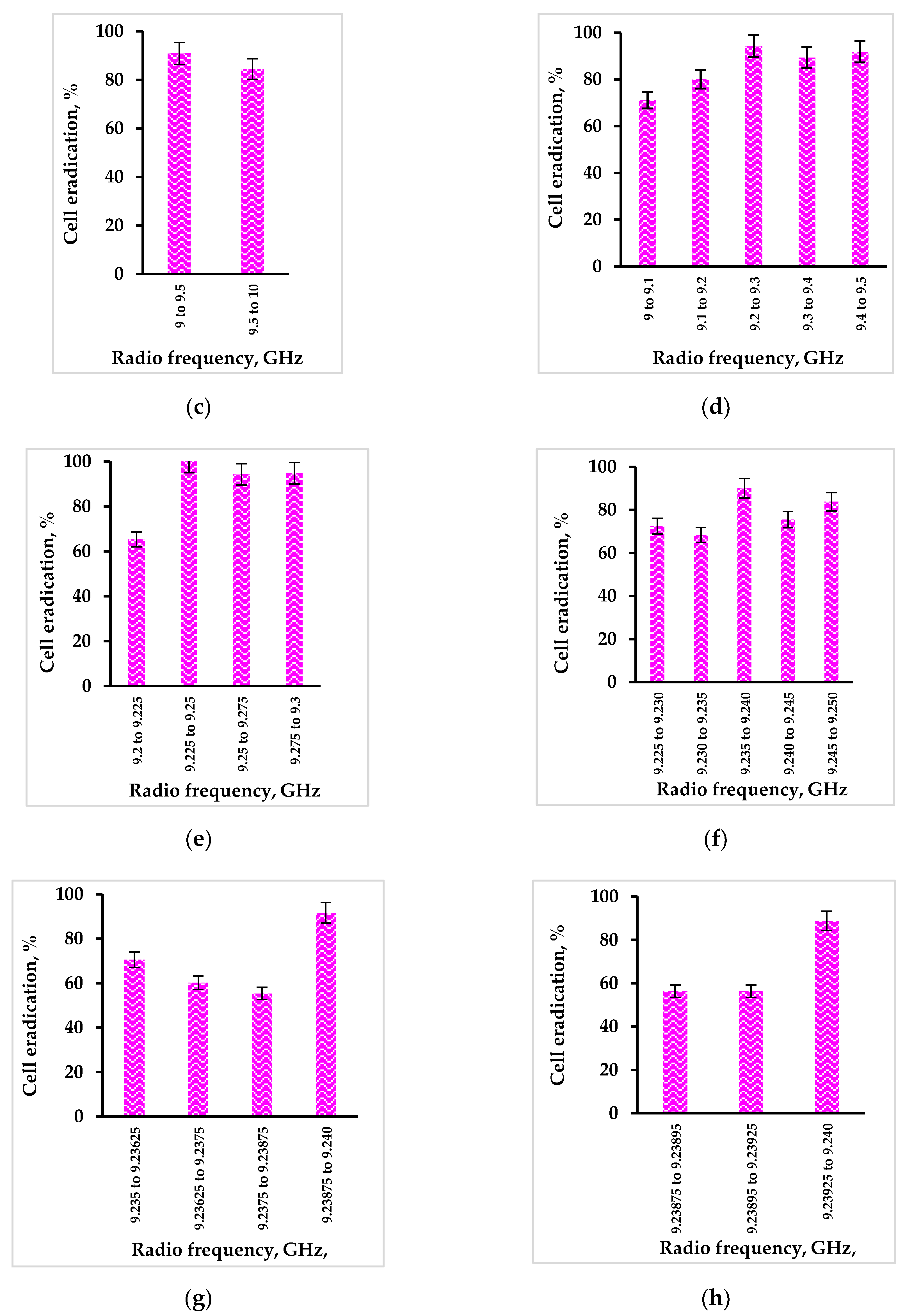
2.3. Activation of Rose Bengal by Radio Waves
3. Materials and Methods
3.1. Materials and Bacterial Strains
3.2. Immobilization of Rose Bengal onto Silicon
3.3. Bacterial Growth
3.4. Examination of Dark Rose Bengal Activity in the Presence of Antibiotics
3.4.1. MIC Determination of Antibiotics
3.4.2. MIC Determination of Rose Bengal in the Presence of Sub-MIC Antibiotic Concentrations
3.5. Ultrasonic Activation of Free and Silicon-Immobilized Rose Bengal
3.6. Activation of Free Rose Bengal by Radio Waves
3.7. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PACT | Photodynamic antimicrobial chemotherapy |
| PS | photosensitizer |
| MIC | minimum inhibitory concentration |
| SE | Standard error |
| CFU | Colony forming units |
| BH | Brain Heart broth |
| BHA | Brain Heart agar |
| OD660 | Optical density at 660 nm |
References
- Maisch, T. A new strategy to destroy antimicrobial resistant microorganisms: Antimicrobial photodynamic treatment. Mini Rev. Med. Chem. 2009, 9, 974–983. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, I.J.; Dougherty, T.J. Basic principles of photodynamic therapy. J. Porphyr. Phthalocyanines 2001, 5, 105–129. [Google Scholar] [CrossRef]
- Yano, S.; Hirohara, S.; Obata, M.; Hagiya, Y.; Ogura, S.; Ikeda, A.; Kataoka, H.; Tanaka, M.; Joh, T. Current states and future views in photodynamic therapy. J. Photochem. Photobiol. C Photochem. Rev. 2011, 12, 46–67. [Google Scholar] [CrossRef]
- Zhang, J.; Jiang, C.; Figueiró Longo, J.P.; Azevedo, R.B.; Zhang, H.; Muehlmann, L.A. An updated overview on the development of new photosensitizers for anticancer photodynamic therapy. Acta Pharm. Sin. B 2018, 8, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Konaté, K.; Mavoungou, J.F.; Lepengué, A.N.; Aworet-Samseny, R.R.R.; Hilou, A.; Souza, A.; Dicko, M.H.; Batchi, B.M. Antibacterial activity against β-lactamase producing methicillin and ampicillin-resistants Staphylococcus aureus: Fractional inhibitory concentration index (FICI) determination. Ann. Clin. Microbiol. Antimicrob. 2012, 11, 18. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, C.M.; Donnelly, R.F.; Elborn, J.S.; Magee, N.D.; Tunney, M.M. Photodynamic antimicrobial chemotherapy (PACT) in combination with antibiotics for treatment of Burkholderia cepacia complex infection. J. Photochem. Photobiol. B 2012, 106, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Di Poto, A.; Sbarra, M.S.; Provenza, G.; Visai, L.; Speziale, P. The effect of photodynamic treatment combined with antibiotic action or host defence mechanisms on Staphylococcus aureus biofilms. Biomaterials 2009, 30, 3158–3166. [Google Scholar] [CrossRef]
- Almeida, J.; Tomé, J.P.; Neves, M.G.; Tomé, A.C.; Cavaleiro, J.A.; Cunha, Â.; Costa, L.; Faustino, M.A.; Almeida, A. Photodynamic inactivation of multidrug-resistant bacteria in hospital wastewaters: Influence of residual antibiotics. Photochem. Photobiol. Sci. 2014, 13, 626–633. [Google Scholar] [CrossRef]
- Nisnevitch, M.; Valkov, A.; Nakonechny, F.; Gutterman, M.; Nitzan, Y. Antibiotics combined with photosensitizers-a novel approach to antibacterial treatment. In Antibiotic Therapy: New Developments; Turner, A., Hall, J., Eds.; Nova Science Inc.: New York, NY, USA, 2013; pp. 63–88. ISBN 978-1-62808-171-8. [Google Scholar]
- Gutterman, M.; Valkov, A.; Nisnevitch, M. Sensitization of bacteria to antibiotics by combined antibiotic-photodynamic treatment. FEBS J. 2014, 281, 166. [Google Scholar]
- Ilizirov, Y.; Formanovsky, A.; Mikhura, I.; Paitan, Y.; Nakonechny, F.; Nisnevitch, M. Effect of photodynamic antibacterial chemotherapy combined with antibiotics on Gram-positive and Gram-negative bacteria. Molecules 2018, 23, 3152. [Google Scholar] [CrossRef]
- O’Riordan, K.; Akilov, O.E.; Hasan, T. The potential for photodynamic therapy in the treatment of localized infections. Photodiagn. Photodyn. Ther. 2005, 2, 247–262. [Google Scholar] [CrossRef]
- Nakonechny, F.; Firer, M.A.; Nitzan, Y.; Nisnevitch, M. Intracellular antimicrobial photodynamic therapy: A novel technique for efficient eradication of pathogenic bacteria. Photochem. Photobiol. 2010, 86, 1350–1355. [Google Scholar] [CrossRef] [PubMed]
- Nakonechny, F.; Nisnevitch, M.; Nitzan, Y.; Firer, M.A. New techniques in antimicrobial photodynamic therapy: Scope of application and overcoming drug resistance in nosocomial infections. In Science Against Microbial Pathogens: Communicating Current Research and Technological Advances; Microbiology book series; Mendez-Vilas, A., Ed.; Formatex: Badajoz, Spain, 2011; Volume 1, pp. 684–691. ISBN 978-84-939843-1-1. [Google Scholar]
- Nisnevitch, M.; Nakonechny, F.; Firer, M.; Nitzan, Y. Photodynamic antimicrobial chemotherapy with photosensitizers in liposomes under external and chemoluminescent excitation. FEBS J. 2009, 276, 332. [Google Scholar]
- Nakonechny, F.; Nisnevitch, M.M.; Nitzan, Y.; Nisnevitch, M. Sonodynamic excitation of Rose Bengal for eradication of Gram-positive and Gram-negative bacteria. Biomed. Res. Int. 2013, 2013, 684930. [Google Scholar] [CrossRef] [PubMed]
- Alves, F.; Pavarina, A.C.; Mima, E.G.O.; McHale, A.P.; Callan, J.F. Antimicrobial sonodynamic and photodynamic therapies against Candida albicans. Biofouling 2018, 34, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Harris, F.; Dennison, S.R.; Phoenix, D.A. Using sound for microbial eradication—Light at the end of the tunnel? FEMS Microbiol. Lett. 2014, 356, 20–22. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Wang, D.J.; Liu, B.M.; Wang, X.; He, L.-L.; Wang, J.; Xu, S.-K. The influence of ultrasound on the fluoroquinolones antibacterial activity. Ultrason. Sonochem. 2011, 18, 1052–1056. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Pan, H.; Wu, G.; Yang, Z.; Yi, J. Ultrasound may be exploited for the treatment of microbial diseases. Med. Hypotheses 2009, 73, 18–19. [Google Scholar] [CrossRef] [PubMed]
- Valkov, A.; Nakonechny, F.; Nisnevitch, M. Polymer-immobilized photosensitizers for continuous eradication of bacteria. Int. J. Mol. Sci. 2014, 15, 14984–14996. [Google Scholar] [CrossRef]
- Valkov, A.; Nakonechny, F.; Nisnevitch, M. Antibacterial properties of Rose Bengal immobilized in polymer supports. Appl. Mech. Mater. 2015, 719, 21–24. [Google Scholar] [CrossRef]
- Valkov, A.; Raik, K.A.; Mualem-Sinai, Y.; Nakonechny, F.; Nisnevitch, M. Water disinfection by immobilized photosensitizers. Water 2019, 11, 26. [Google Scholar] [CrossRef]
- Tamarov, K.P.; Osminkina, L.A.; Zinovyev, S.V.; Maximova, K.A.; Kargina, J.V.; Gongalsky, M.B.; Ryabchikov, Y.; Al-Kattan, A.; Sviridov, A.P.; Sentis, M.; et al. Radio frequency radiation-induced hyperthermia using Si nanoparticle-based sensitizers for mild cancer therapy. Sci. Rep. 2014, 4, 7034. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.J.; Lee, H.K.; Kwon, K.B.; Kim, H.J.; Hong, S.T. Transferrin as a thermosensitizer in radiofrequency hyperthermia for cancer treatment. Sci. Rep. 2018, 8, 13505. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Wang, L.; Gao, J.; Liu, Y.; Zhang, J.; Ma, R.; Liu, R.; Zhang, Z. A fullerene-based multi-functional nanoplatform for cancer theranostic applications. Biomaterials 2014, 35, 5771–5784. [Google Scholar] [CrossRef] [PubMed]
- Rauf, M.A.; Graham, J.P.; Bukallah, S.B.; Al-Saedi, M.A. Solvatochromic behavior on the absorption and fluorescence spectra of Rose Bengal dye in various solvents. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2009, 72, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Schäfer, M.; Schmitz, C.; Facius, R.; Horneck, G.; Milow, B.; Funken, K.H.; Ortner, J. Systematic study of parameters influencing the action of Rose Bengal with visible light on bacterial cells: Comparison between the biological effect and singlet-oxygen production. Photochem. Photobiol. 2000, 71, 514–523. [Google Scholar] [CrossRef]
- Nisnevitch, M.; Nakonechny, F.; Nitzan, Y. Photodynamic antimicrobial chemotherapy by liposome-encapsulated water-soluble photosensitizers. Russ. J. Biorgan. Chem. 2010, 36, 363–369. [Google Scholar] [CrossRef]
- Nitzan, Y.; Nisnevitch, M. Special features of Gram-positive bacterial eradication by photosensitizers. Recent Pat. Anti-Infect. Drug Discov. 2013, 8, 88–99. [Google Scholar] [CrossRef]
- Bond, J.S.; Francis, S.H.; Park, J.H. An essential histidine in the catalytic activities of 3-phosphoglyceraldehyde dehydrogenase. J. Biol. Chem. 1970, 245, 1041–1053. [Google Scholar]
- Hsieh, Y.H.; Huang, Y.J.; Jin, J.S.; Yu, L.; Yang, H.; Jiang, C.; Wang, B.; Tai, P.C. Mechanisms of Rose Bengal inhibition on SecA ATPase and ion channel activities. Biochem. Biophys. Res. Commun. 2014, 454, 308–312. [Google Scholar] [CrossRef][Green Version]
- Shrestha, A.; Hamblin, M.R.; Kishen, A. Photoactivated rose bengal functionalized chitosan nanoparticles produce antibacterial/biofilm activity and stabilize dentin-collagen. Nanomedicine 2014, 10, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Coulson, A.F.; Yonetani, T. Interaction of rose bengal with apo-hemoproteins. An essential histidine residue in cytochrome c peroxidase. Eur. J. Biochem. 1972, 26, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Nakonieczna, J.; Wolnikowska, K.; Ogonowska, P.; Neubauer, D.; Bernat, A.; Kamysz, W. Rose bengal-mediated photoinactivation of multidrug resistant Pseudomonas aeruginosa is enhanced in the presence of antimicrobial peptides. Front. Microbiol. 2018, 9, 1949. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho Goulart, R.; Bolean, M.; Bolean, M.; de Paiva Paulino, T.; Thedei, G., Jr.; Souza, S.L.; Tedesco, A.C.; Ciancaglini, P. Original Articles Photodynamic Therapy in Planktonic and Biofilm Cultures of Aggregatibacter actinomycetemcomitans. Photomed. Laser Surg. 2010, 28, S53–S60. [Google Scholar] [CrossRef] [PubMed]
- Sabbahi, S.; Ben Ayed, L.; Jemli, M. Staphylococcus aureus photodynamic inactivation mechanisms by rose bengal: Use of antioxidants and spectroscopic study. Appl. Water Sci. 2018, 8, 56. [Google Scholar] [CrossRef]
- Kotra, L.P.; Haddad, J.; Mobashery, S. Aminoglycosides: Perspectives on mechanisms of action and resistance and strategies to counter resistance. Antimicrob. Agents Chemother. 2000, 44, 3249–3256. [Google Scholar] [CrossRef] [PubMed]
- Krause, K.M.; Serio, A.W.; Kane, T.R.; Connolly, L.E. Aminoglycosides: An overview. Cold Spring Harb. Perspect. Med. 2016, 6, a027029. [Google Scholar] [CrossRef]
- Kato, H.; Komagoe, K.; Nakanishi, Y.; Inoue, T.; Katsu, T. Xanthene dyes induce membrane permeabilization of bacteria and erythrocytes by photoinactivation. Photochem. Photobiol. 2012, 88, 423–431. [Google Scholar] [CrossRef]
- Bryan, L.E.; Kwan, S. Roles of ribosomal binding, membrane potential, and electron transport in bacterial uptake of streptomycin and gentamicin. Antimicrob. Agents Chemother. 1983, 23, 835–845. [Google Scholar] [CrossRef]
- Wang, M.; Tang, T. Surface treatment strategies to combat implant-related infection from the beginning. J. Orthop. Transl. 2018, 17, 42–54. [Google Scholar] [CrossRef]
- Schierholz, J.M.; Beuth, J. Implant infections: A haven for opportunistic bacteria. J. Hosp. Infect. 2001, 49, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Hatanaka, S.; Mitome, H.; Yasui, K.; Hayashi, S. Single bubble sonochemiluminescence in aqueous luminol solutions. J. Am. Chem. Soc. 2002, 124, 10250–10251. [Google Scholar] [CrossRef] [PubMed]
- Mišík, V.; Riesz, P. Free radical intermediates in sonodynamic therapy. Ann. N. Y. Acad. Sci. 2000, 899, 335–348. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, W.; Honda, H.; Feril, L.B., Jr.; Kudo, N.; Kondo, T. Comparison between sonodynamic effect and photodynamic effect with photo sensitizers on free radical formation and cell killing. Ultrason. Sonochem. 2006, 13, 535–542. [Google Scholar] [CrossRef] [PubMed]
- McCaughan, B.; Rouanet, C.; Fowley, C.; Nomikou, N.; McHale, A.P.; McCarron, P.A.; Callan, J.F. Enhanced ROS production and cell death through combined photo- and sono-activation of conventional photosensitising drugs. Bioorg. Med. Chem. Lett. 2011, 21, 5750–5752. [Google Scholar] [CrossRef] [PubMed]
- European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID). Deteremination of minimum inhibitory concentrations (MICs) of antibacterial agents by broth dilution. Clin. Microbiol. Infect. 2003, 9, 1–7. [Google Scholar] [CrossRef]


| Antimicrobial | MIC 1, mg/mL | MIC 1 of Rose Bengal, mg/mL, in the Presence of Sub-MIC of Antibiotics |
|---|---|---|
| Rose Bengal | 0.125 ± 0.001 | NR 2 |
| Ciprofloxacin hydrochloride | 0.025 ± 0.001 | 0.0312 ± 0.0006 |
| Levofloxacin | 0.025 ± 0.001 | 0.0312 ± 0.006 |
| Gentamicin sulfate | 0.005 ± 0.001 | 0.0011 ± 0.0001 |
| Methicillin | 0.0025 ± 0.0008 | 0.0625 ± 0.0013 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakonechny, F.; Barel, M.; David, A.; Koretz, S.; Litvak, B.; Ragozin, E.; Etinger, A.; Livne, O.; Pinhasi, Y.; Gellerman, G.; et al. Dark Antibacterial Activity of Rose Bengal. Int. J. Mol. Sci. 2019, 20, 3196. https://doi.org/10.3390/ijms20133196
Nakonechny F, Barel M, David A, Koretz S, Litvak B, Ragozin E, Etinger A, Livne O, Pinhasi Y, Gellerman G, et al. Dark Antibacterial Activity of Rose Bengal. International Journal of Molecular Sciences. 2019; 20(13):3196. https://doi.org/10.3390/ijms20133196
Chicago/Turabian StyleNakonechny, Faina, Margarita Barel, Arad David, Simor Koretz, Boris Litvak, Elena Ragozin, Ariel Etinger, Oz Livne, Yosef Pinhasi, Gary Gellerman, and et al. 2019. "Dark Antibacterial Activity of Rose Bengal" International Journal of Molecular Sciences 20, no. 13: 3196. https://doi.org/10.3390/ijms20133196
APA StyleNakonechny, F., Barel, M., David, A., Koretz, S., Litvak, B., Ragozin, E., Etinger, A., Livne, O., Pinhasi, Y., Gellerman, G., & Nisnevitch, M. (2019). Dark Antibacterial Activity of Rose Bengal. International Journal of Molecular Sciences, 20(13), 3196. https://doi.org/10.3390/ijms20133196







