Drought Stress Tolerance in Wheat and Barley: Advances in Physiology, Breeding and Genetics Research
Abstract
1. Introduction
2. Physiological and Biochemical Responses
2.1. Photosynthesis and Gaseous Exchange
2.2. Water Relations
2.3. Nutrient Relations
2.4. Oxidative Status
2.4.1. Reactive Oxygen Species (ROS)
2.4.2. Antioxidant System
2.5. Osmotic Balance
2.6. Hormonal Effect
3. Advances in Breeding for Drought Tolerance
3.1. Genetic Variation of Drought Tolerance at Different Growth Stages
3.1.1. Drought Tolerance at the Germination Stage
3.1.2. Drought Tolerance at the Seedling Stage
3.1.3. Drought Tolerance at Flowering and Grain Filling Stages
3.2. High-Throughput Phenotyping for Improving Drought Tolerance in Wheat
3.3. The Use of Nanotechnology in Improving and Breeding Drought Tolerance
4. Genetic Landscape of Drought Tolerance in Wheat and Barley
4.1. The Genetic Basis of Drought Tolerance
4.1.1. Quantitative Trait Locus (QTL) of Drought Tolerance
4.1.2. Genomics Analyses of Drought Tolerance
4.1.3. Functional Validation of Drought-Tolerance QTLs and Candidate Genes
4.1.4. Genetic Engineering of Drought-Tolerance Genes in Wheat and Barley
5. The Path Forward: Identifying the Most Drought-Tolerant Genotypes for Further Improvement of Drought Tolerance
Author Contributions
Funding
Conflicts of Interest
References
- FAO World Food and Agriculture. Statistical Yearbook. Available online: http://www.fao.org/3/i3107e/i3107e.pdf (accessed on 20 May 2019).
- Vinocur, B.; Altman, A. Recent advances in engineering plant tolerance to abiotic stress: Achievements and limitations. Curr. Opin. Biotechnol. 2005, 16, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Abid, M.; Tian, Z.; Ata-Ul-Karim, S.T.; Cui, Y.; Liu, Y.; Zahoor, R.; Jiang, D.; Dai, T. Nitrogen Nutrition Improves the Potential of Wheat (Triticum aestivum L.) to Alleviate the Effects of Drought Stress during Vegetative Growth Periods. Front. Plant Sci. 2016, 7, 981. [Google Scholar] [CrossRef] [PubMed]
- Bowne, J.B.; Erwin, T.A.; Juttner, J.; Schnurbusch, T.; Langridge, P.; Bacic, A.; Roessner, U.; Alia; Saradhi, P.; Mohanty, P.; et al. Drought responses of leaf tissues from wheat cultivars of differing drought tolerance at the metabolite level. Mol. Plant 2012, 5, 418–429. [Google Scholar] [CrossRef] [PubMed]
- Baenziger, P.S. Wheat Breeding and Genetics. Ref. Modul. Food Sci. 2016. [Google Scholar]
- Reynolds, P.; Ortiz-Monasterio, J.; McNab, A.; Reynolds, E.M.; Reynolds, M. Application of Physiology in Wheat Breeding. Available online: https://books.google.com.hk/books?hl=zh-TW&lr=&id=PJ1a3yfTgg4C&oi=fnd&pg=PR4&dq=Application+of+Physiology+in+Wheat+Breeding&ots=ldntzpoFEE&sig=AZx6LazRQzYV72WUR-zI8KVY8Bo&redir_esc=y&hl=zh-CN&sourceid=cndr#v=onepage&q=Application%20of%20Physiology%20in%20Wheat%20Breeding&f=false (accessed on 20 May 2019).
- Hussain, W.; Stephen Baenziger, P.; Belamkar, V.; Guttieri, M.J.M.J.; Venegas, J.P.J.P.; Easterly, A.; Sallam, A.; Poland, J. Genotyping-by-Sequencing Derived High-Density Linkage Map and its Application to QTL Mapping of Flag Leaf Traits in Bread Wheat. Sci. Rep. 2017, 7, 16394. [Google Scholar] [CrossRef] [PubMed]
- Thabet, S.G.; Moursi, Y.S.; Karam, M.A.; Graner, A.; Alqudah, A.M. Genetic basis of drought tolerance during seed germination in barley. PLoS ONE 2018, 13, e0206682. [Google Scholar] [CrossRef] [PubMed]
- Ovenden, B.; Milgate, A.; Wade, L.J.; Rebetzke, G.J.; Holland, J.B. Genome-Wide Associations for Water-Soluble Carbohydrate Concentration and Relative Maturity in Wheat Using SNP and DArT Marker Arrays. G3 2017, 7, 2821–2830. [Google Scholar] [CrossRef][Green Version]
- Qaseem, M.F.; Qureshi, R.; Muqaddasi, Q.H.; Shaheen, H.; Kousar, R.; Röder, M.S. Genome-wide association mapping in bread wheat subjected to independent and combined high temperature and drought stress. PLoS One 2018, 13, e0199121. [Google Scholar] [CrossRef]
- Rasheed, A.; Wen, W.; Gao, F.; Zhai, S.; Jin, H.; Liu, J.; Guo, Q.; Zhang, Y.; Dreisigacker, S.; Xia, X.; et al. Development and validation of KASP assays for genes underpinning key economic traits in bread wheat. Theor. Appl. Genet. 2016, 129, 1843–1860. [Google Scholar] [CrossRef]
- Zeng, Z.B.; Teulat, B.; Merah, O.; Sirault, X.; Borries, C.; Waugh, R.; This, D.; Abebe, T.; Guenzi, A.C.; Martin, B.; et al. Detection and validation of novel QTL for shoot and root traits in barley (Hordeum vulgare L.). J. Exp. Bot. 2014, 9, 171–180. [Google Scholar]
- Sukumaran, S.; Reynolds, M.P.; Sansaloni, C. Genome-Wide Association Analyses Identify QTL Hotspots for Yield and Component Traits in Durum Wheat Grown under Yield Potential, Drought, and Heat Stress Environments. Front. Plant Sci 2018, 9, 81. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.S.B.D. Marker-Assisted Plant Breeding: Principles and Practices, 1st ed.; Springer India: Delhi, India, 2015; ISBN 978-81-322-2316-0. [Google Scholar]
- Dawood, M.F.A.; Abeed, A.H.A.; Aldaby, E.E.S. Titanium dioxide nanoparticles model growth kinetic traits of some wheat cultivars under different water regimes. Indian J. Plant Physiol. 2019, 24, 129–140. [Google Scholar] [CrossRef]
- Pandey, V.; Shukla, A. Acclimation and Tolerance Strategies of Rice under Drought Stress. Rice Sci. 2015, 22, 147–161. [Google Scholar] [CrossRef]
- Foyer, C.H. Bundle sheath proteins are more sensitive to oxidative damage than those of the mesophyll in maize leaves exposed to paraquat or low temperatures. J. Exp. Bot. 2000, 51, 123–130. [Google Scholar]
- Amirjani, M.R.; Mahdiyeh, M. Antioxidative and biochemical responses of wheat. J. Agric. Biol. Sci 2013, 8, 291–301. [Google Scholar]
- Kabiri, R.; Nasibi, F.; Farahbakhsh, H. Effect of Exogenous Salicylic Acid on Some Physiological Parameters and Alleviation of Drought Stress in Nigella sativa Plant under Hydroponic Culture. Plant Prot. 2014, 50, 43–51. [Google Scholar] [CrossRef]
- Rama, R.; Nagaraja, R.; Ragimasalawada, M.; Sabbavarapu, M.M.; Nadoor, S.; Patil, J.V. Detection and validation of stay-green QTL in post-rainy sorghum involving widely adapted cultivar, M35-1 and a popular stay-green genotype B35. BMC Genomics 2014, 15, 909. [Google Scholar]
- Centritto, M.; Lauteri, M.; Monteverdi, M.C.; Serraj, R. Leaf gas exchange, carbon isotope discrimination, and grain yield in contrasting rice genotypes subjected to water deficits during the reproductive stage. J. Exp. Bot. 2009, 60, 2325–2339. [Google Scholar] [CrossRef] [PubMed]
- A, M.M.C.; B, J.P.M.; A, J.S.P. Understanding plant responses to drought — from genes to the whole plant. Funct. plant biol. 2003, 239–264. [Google Scholar]
- Alaei, Y. The Effect of Amino Acids on Leaf Chlorophyll Content in Bread Wheat Genotypes under Drought Stress Conditions. Middle-East J. Sci. Res. 2011, 10, 99–101. [Google Scholar]
- Kiliç, H.; Yağbasanlar, T. The Effect of Drought Stress on Grain Yield, Yield Components and some Quality Traits of Durum Wheat (Triticum turgidum ssp. durum). Cultivars 2010, 38, 164–170. [Google Scholar]
- Samarah, N.H.; Alqudah, A.M.; Amayreh, J.A.; McAndrews, G.M. The Effect of Late-terminal Drought Stress on Yield Components of Four Barley Cultivars. J. Agron. Crop Sci. 2009, 195, 427–441. [Google Scholar] [CrossRef]
- Allahverdiyev, T.I. Effect of drought stress on some physiological parameters, yield, yield components of durum (Triticum durum desf.) and bread (Triticum aestivum L.) wheat genotypes. Ekin J. Crop Breed. Genet. 2015, 1, 50–62. [Google Scholar]
- Clarke, J.M.; McCaig, T.N. Evaluation of Techniques for Screening for Drought Resistance in Wheat1. Crop Sci. 1982, 22, 503. [Google Scholar] [CrossRef]
- Izanloo, A.; Condon, A.G.; Langridge, P.; Tester, M.; Schnurbusch, T. Different mechanisms of adaptation to cyclic water stress in two South Australian bread wheat cultivars. J. Exp. Bot. 2008, 59, 3327–3346. [Google Scholar] [CrossRef]
- Lonbani, M.; Arzani, A. Morpho-physiological traits associated with terminal drought- stress tolerance in triticale and wheat. Agronomy Research 2011, 9, 315–329. [Google Scholar]
- Tahara, M.; Carver, B.F.; Johnson, R.C.; Smith, E.L. Relationship between relative water content during reproductive development and winter wheat grain yield. Euphytica 1990, 49, 255–262. [Google Scholar]
- Teulat, B.; Monneveux, P.; Wery, J.; Borries, C.; Souyris, I.; Charrier, A.; This, D. Relationships between relative water content and growth parameters under water stress in barley: a QTL study. New Phytol. 1997, 137, 99–107. [Google Scholar] [CrossRef]
- Ahmed, M.; Asif, M.; Goyal, A. Silicon the Non-Essential Beneficial Plant Nutrient to Enhanced Drought Tolerance in Wheat; Goyal, A., Ed.; IntechOpen: London, UK, 2012. [Google Scholar]
- Balota, M. Excised-leaf water status in Romanian and foreign winter wheat cultivars. I. The physiological and environmental effects on excised-leaf water loss. Rom. Agric. Res 2008, 3, 69–76. [Google Scholar]
- Akrami, M.; Yousefi, Z. Biological Control of Fusarium wilt of Tomato (Solanum lycopersicum) by Trichoderma spp. as Antagonist Fungi. Biol. Forum – An Int. J. 2015, 7, 887–892. [Google Scholar]
- Noman, A.; Ali, Q.; Naseem, J.; Javed, M.T.; Kanwal, H.; Islam, W.; Aqeel, M.; Khalid, N.; Zafar, S.; Tayyeb, M.; et al. Sugar beet extract acts as a natural bio-stimulant for physio-biochemical attributes in water stressed wheat (Triticum aestivum L.). Acta Physiol. Plant. 2018, 40, 110. [Google Scholar] [CrossRef]
- Mobasser, H.R.; Mohammadi, G.N.; Abad, H.H.S.; Rigi, K. Effect of application elements, water stress and variety on nutrients of grain wheat in Zahak region, Iran. JBES 2014, 5, 105–110. [Google Scholar]
- Faye, I.; Diouf, O.; Guisse, A.; Sene, M.; Diallo, N. Characterizing root responses to low phosphorus in pearl millet [Pennisetum glaucum (L.) R. Br.]. Agron. J. 2006, 98, 1187–1194. [Google Scholar] [CrossRef]
- Sardans, J.; Peñuelas, J. Increasing drought decreases phosphorus availability in an evergreen Mediterranean forest. Plant Soil 2004, 267, 367–377. [Google Scholar] [CrossRef]
- Farooq, M.; Hussain, M.; Wahid, A.; Siddique, K.H.M. Plant Responses to Drought Stress; Springer: Berlin/Heidelberg, Germany, 2012; pp. 1–6. [Google Scholar]
- Sardans, J.; Peñuelas, J.; Ogaya, R. Drought’s impact on Ca, Fe, Mg, Mo and S concentration and accumulation patterns in the plants and soil of a Mediterranean evergreen Quercus ilex forest. Biogeochemistry 2008, 87, 49–69. [Google Scholar] [CrossRef]
- Hu, Y.; Schmidhalter, U. Drought and salinity: A comparison of their effects on mineral nutrition of plants. J. Plant Nutr. Soil Sci. 2005, 168, 541–549. [Google Scholar] [CrossRef]
- Havlin, J. Soil Fertility and Fertilizers: An Introduction to Nutrient Management, 6th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 1999; ISBN 0136268064. [Google Scholar]
- Hasanuzzaman, M.; Nahar, K.; Anee, T.I.; Khan, M.I.R.; Fujita, M. Silicon-mediated regulation of antioxidant defense and glyoxalase systems confers drought stress tolerance in Brassica napus L. South Afr. J. Bot. 2018, 115, 50–57. [Google Scholar] [CrossRef]
- Outoukarte, I.; El Keroumi, A.; Dihazi, A.; Naamani, K. Use of morpho-physiological parameters and biochemical markers to select drought tolerant genotypes of durum wheat. J. Plant Stress Phys. 2019, 1–7. [Google Scholar]
- Sharma, P.; Sareen, S.; Saini, M. Shefali Assessing genetic variation for heat stress tolerance in Indian bread wheat genotypes using morpho-physiological traits and molecular markers. Plant Genet. Resour. 2017, 15, 539–547. [Google Scholar] [CrossRef]
- Zhang, Y.-J.; Yang, J.-S.; Guo, S.-J.; Meng, J.-J.; Zhang, Y.-L.; Wan, S.-B.; He, Q.-W.; Li, X.-G. Over-expression of the Arabidopsis CBF1 gene improves resistance of tomato leaves to low temperature under low irradiance. Plant Biol. 2011, 13, 362–367. [Google Scholar] [CrossRef]
- Sánchez-Rodríguez, E.; Rubio-Wilhelmi, M.; Cervilla, L.M.; Blasco, B.; Rios, J.J.; Rosales, M.A.; Ruiz, J.M. Genotypic differences in some physiological parameters symptomatic for oxidative stress under moderate drought in tomato plants. Plant Sci. 2010, 178, 30–40. [Google Scholar] [CrossRef]
- Alam, M.; Hasanuzzaman, M.; Nahar, K.; Fujita, M. Exogenous salicylic acid ameliorates short-term drought stress in mustard (Brassica juncea L.) seedlings by up-regulating the antioxidant defense and glyoxalase system. Aust. J. Crop Sci. 2013, 7, 1053–1063. [Google Scholar]
- Corpas, F.J.; del Río, L.A.; Barroso, J.B. Need of biomarkers of nitrosative stress in plants. Trends Plant Sci. 2007, 12, 436–438. [Google Scholar] [CrossRef] [PubMed]
- Ojeda, A.G.; Escobosa, A.R.C.; Wrobel, K.; Barrientos, E.Y.; Wrobel, K. Effect of Cd (II) and Se (IV) exposure on cellular distribution of both elements and concentration levels of glyoxal and methylglyoxal in Lepidium sativum. Metallomics 2013, 5, 1254–1261. [Google Scholar] [CrossRef] [PubMed]
- Hoque, M.A.; Uraji, M.; Torii, A.; Banu, M.N.A.; Mori, I.C.; Nakamura, Y.; Murata, Y. Methylglyoxal inhibition of cytosolic ascorbate peroxidase from Nicotiana tabacum. J. Biochem. Mol. Toxicol. 2012, 26, 315–321. [Google Scholar] [CrossRef]
- Hossain, M.A.; Piyatida, P.; da Silva, J.A.T.; Fujita, M. Molecular mechanism of heavy metal toxicity and tolerance in plants: central role of glutathione in detoxification of reactive oxygen species and methylglyoxal and in heavy metal chelation. J. Bot. 2012, 2012. [Google Scholar] [CrossRef]
- Alam, M.; Nahar, K.; Hasanuzzaman, M.; Fujita, M. Trehalose-induced drought stress tolerance: A comparative study among different Brassica species. Plant Omics 2014, 7, 271–283. [Google Scholar]
- Nahar, K.; Hasanuzzaman, M.; Alam, M.M.; Rahman, A.; Mahmud, J.A.; Suzuki, T.; Fujita, M. Insights into spermine-induced combined high temperature and drought tolerance in mung bean: osmoregulation and roles of antioxidant and glyoxalase system. Protoplasma 2017, 254, 445–460. [Google Scholar] [CrossRef]
- Guan, L.M.; Scandalios, J.G. Hydrogen peroxide-mediated catalase gene expression in response to wounding. Free Radic. Biol. Med. 2000, 28, 1182–1190. [Google Scholar] [CrossRef]
- Dudziak, K.; Zapalska, M.; Börner, A.; Szczerba, H.; Kowalczyk, K.; Nowak, M. Analysis of wheat gene expression related to the oxidative stress response and signal transduction under short-term osmotic stress. Sci. Rep. 2019, 9, 2743. [Google Scholar] [CrossRef]
- Hasheminasab, H.; Assad, M.T.; Ali, A.; Sahhafi, S.R. Influence of Drought Stress on Oxidative Damage and Antioxidant Defense Systems in Tolerant and Susceptible Wheat Genotypes. J. Agric. Sci. 2012, 4, 625–637. [Google Scholar] [CrossRef]
- Nikolaeva, M.K.; Maevskaya, S.N.; Shugaev, A.G.; Bukhov, N.G. Effect of drought on chlorophyll content and antioxidant enzyme activities in leaves of three wheat cultivars varying in productivity. Russ. J. Plant Physiol. 2010, 57, 87–95. [Google Scholar] [CrossRef]
- Ahmed, M.; Asif, M. Amelioration of Drought in Sorghum (Sorghum bicolor L.) by Silicon. Commun. Soil Sci. Plant Anal. 2014, 45, 470–486. [Google Scholar] [CrossRef]
- Wang, Y.; Frei, M. Agriculture, Ecosystems and Environment Stressed food – The impact of abiotic environmental stresses on crop quality. "Agriculture, Ecosyst. Environ. 2011, 141, 271–286. [Google Scholar] [CrossRef]
- Hossain, M.A.; Fujita, M. Purification of glyoxalase I from onion bulbs and molecular cloning of its cDNA. Biosci. Biotechnol. Biochem. 2009, 73, 2007–2013. [Google Scholar] [CrossRef]
- Cheng, L.; Yang, A.; Jiang, C.; Ren, M.; Zhang, Y.; Feng, Q.; Wang, S.; Guan, Y.; Luo, C. Quantitative trait loci mapping for plant height in tobacco using linkage and association mapping methods. Crop Sci. 2015, 55, 641. [Google Scholar] [CrossRef]
- Harb, A. Identification of Candidate Genes for Drought Stress Tolerance. In Drought Stress Tolerance in Plants, Vol 2: Molecular and Genetic Perspectives; Hossain, A.M., Wani, H.S., Bhattacharjee, S., Burritt, J.D., Tran, P.L.-S., Eds.; Springer: Cham, Switzerland, 2016; pp. 385–414. [Google Scholar]
- Loggini, B.; Scartazza, A. Antioxidative defense system, pigment composition, and photosynthetic efficiency in two wheat cultivars subjected to drought. Plant Physiol. 1999, 119, 1091–1099. [Google Scholar] [CrossRef]
- Pazirandeh, M.S.; Hasanloo, T.; Niknam, V.; Shahbazi, M.; Mabood, H.E.; Ghaffari, A. Effects of drought and methyl jasmonate on antioxidant activities of selected barley genotypes. J. Agrobiol. 2013, 30, 71–82. [Google Scholar]
- Kovács-Bogdán, E.; Nyitrai, P.; Keresztes, Á. How does a little stress stimulate a plant? Plant Signal. Behav. 2014, 5, 354–358. [Google Scholar] [CrossRef]
- Chalker-Scott, L. Environmental Significance of Anthocyanins in Plant Stress Responses. Photochem. Photobiol. 1999, 70, 1–9. [Google Scholar] [CrossRef]
- Ahmed, M.; Asif, M. Silicon the non-essential beneficial plant nutrient to enhanced drought tolerance in wheat. Crop Plant 2012. Available online: https://www.researchgate.net/profile/Aakash_Goyal/publication/224830516_Silicon_the_Non-Essential_Beneficial_Plant_Nutrient_to_Enhanced_Drought_Tolerance_in_Wheat/links/09e4150a6b2078aab9000000.pdf. (accessed on 20 May 2019).
- Calzadilla, P.I.; Gazquez, A.; Maiale, S.J.; Ruiz, O.A.; Bernardina, M.A. Polyamines as indicators and modulators of the abiotic stress in plants. In Plant adaptation to environmental change: Significance of amino acids and their derivatives; CABI: Wallingford, UK, 2014. [Google Scholar]
- Basu, S.; Roychoudhury, A.; Saha, P.P.; Sengupta, D.N. Comparative analysis of some biochemical responses of three indica rice varieties during polyethylene glycol-mediated water stress exhibits distinct varietal differences. Acta Physiol. Plant. 2010, 32, 551–563. [Google Scholar] [CrossRef]
- Isaksson, C.; Andersson, S. Oxidative stress does not influence carotenoid mobilization and plumage pigmentation. Proc. R. Soc. B. Biol. Sci. 2008, 275, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.D.; Dhuria, N.; Rakhra, G.; Mamik, S. Accumulation of water stress-responsive class-III type of boiling stable peroxidases (BsPOD) in different cultivars of wheat (Triticum aestivum). Acta Biol. Szeged. 2014, 58, 115–122. [Google Scholar]
- Loutfy, N.; El-Tayeb, M.A.; Hassanen, A.M.; Moustafa, M.F.M.; Sakuma, Y.; Inouhe, M. Changes in the water status and osmotic solute contents in response to drought and salicylic acid treatments in four different cultivars of wheat (Triticum aestivum). J. Plant Res. 2012, 125, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Hussain, H.A.; Hussain, S.; Khaliq, A.; Ashraf, U.; Anjum, S.A.; Men, S.; Wang, L. Chilling and Drought Stresses in Crop Plants: Implications, Cross Talk, and Potential Management Opportunities. Front. Plant Sci. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Farshadfar, E.; Ghasempour, H.; Vaezi, H. Molecular aspects of drought tolerance in bread wheat (T. aestivum). PJBS 2008, 11, 118–122. [Google Scholar] [CrossRef]
- Hammad, S.A.; Ali, O.A. Physiological and biochemical studies on drought tolerance of wheat plants by application of amino acids and yeast extract. Ann. Agric. Sci 2014, 59, 133–145. [Google Scholar] [CrossRef]
- Sara, K.; Abbaspour, H.; Sinaki, J.M.; Makarian, H. Effects of Water Deficit and Chitosan Spraying on Osmotic Adjustment and Soluble Protein of Cultivars Castor Bean (Ricinus communis L.). J. Stress Physiol. Biochem. 2012, 8, 160–169. [Google Scholar]
- Al-jebory, E.I. Effect of water stress on carbohydrate metabolism during Pisum sativum seedlings growth. Euphrates J. Agric. Sci. 2012, 4, 1–12. [Google Scholar]
- Gurumurthy, S.; Sarkar, B.; Vanaja, M.; Lakshmi, J.; Yadav, S.K.; Maheswari, M. Morpho-physiological and biochemical changes in black gram (Vigna mungo L. Hepper) genotypes under drought stress at flowering stage. Acta Physiol. Plant. 2019, 41, 42. [Google Scholar] [CrossRef]
- Nazarli, H.; Faraji, F. Response of proline, soluble sugars and antioxidant enzymes in wheat (Triticum aestivum L.) to different irrigation regimes in greenhouse condition. Cercet. agronom. Moldova 2011, 44, 27–33. [Google Scholar] [CrossRef]
- Marcińska, I.; Czyczyło-Mysza, I.; Skrzypek, E.; Filek, M.; Grzesiak, S.; Grzesiak, M.T.; Janowiak, F.; Hura, T.; Dziurka Michałand Dziurka, K.; Nowakowska, A.; et al. Impact of osmotic stress on physiological and biochemical characteristics in drought-susceptible and drought-resistant wheat genotypes. Acta Physiol. Plant. 2013, 35, 451–461. [Google Scholar] [CrossRef]
- Zali, A.G.; Ehsanzadeh, P. Exogenous proline improves osmoregulation, physiological functions, essential oil, and seed yield of fennel. Ind. Crop. prod. 2018, 111, 133–140. [Google Scholar] [CrossRef]
- Gou, W.; Tian, L.; Ruan, Z.; Zheng, P.; Chen, F.; Zhang, L.; Cui, Z.; Zheng, P.; Li, Z.; Gao, M.; et al. Accumulation of choline and glycinebetaine and drought stress tolerance induced in maize (Zea mays) by three plant growth promoting rhizobacteria (PGPR). Pak. J. Bot. 2015, 47, 581–586. [Google Scholar]
- Huseynova, I.M.; Rustamova, S.M.; Suleymanov, S.Y.; Aliyeva, D.R.; Mammadov, A.C.; Aliyev, J.A. Drought-induced changes in photosynthetic apparatus and antioxidant components of wheat (Triticum durum Desf.) varieties. Photosynth. Res. 2016, 130, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Abdul, M.; Faisal, R.; Hui, L.; Yang, L.J.; Xian, Z.H. Assessment of Drought Tolerance of Some Triticum L. Species through Physiological Indices. Czech J. Genet. Plant Breed. 2012, 2012, 178–184. [Google Scholar]
- Thompson, A.J.; Andrews, J.; Mulholland, B.J.; McKee, J.M.T.; Hilton, H.W.; Black, C.R.; Taylor, I.B. Overproduction of abscisic acid in tomato increases transpiration efficiency and root hydraulic conductivity and influences leaf expansion. Plant Physiol. 2007, 143, 1905–1917. [Google Scholar] [CrossRef] [PubMed]
- Lata, C.; Prasad, M. Role of DREBs in regulation of abiotic stress responses in plants. J. Exp. Bot. 2011, 62, 4731–4748. [Google Scholar] [CrossRef]
- Schachtman, D.P.; Goodger, J.Q. Chemical root to shoot signaling under drought. Trends Plant sci. 2 0018, 13, 281–287. [Google Scholar] [CrossRef]
- Suzuki, N.; Miller, G.; Salazar, C.; Mondal, H.A.; Shulaev, E.; Cortes, D.F.; .Shuman, J.L.; Luo, X.; Shah, J.; Schlauch, K.; et al. Temporal-spatial interaction between reactive oxygen species and abscisic acid regulates rapid systemic acclimation in plants. Plant Cell 2013, 25, 3553–3569. [Google Scholar] [CrossRef]
- Xu, W.; Jia, L.; Shi, W.; Liang, J.; Zhou, F.; Li, Q.; Zhang, J. Abscisic acid accumulation modulates auxin transport in the root tip to enhance proton secretion for maintaining root growth under moderate water stress. New Phytol. 2013, 197, 139–150. [Google Scholar] [CrossRef]
- Lamaoui, M.; Jemo, M.; Datla, R.; Bekkaoui, F. Heat and Drought Stresses in Crops and Approaches for Their Mitigation. Front. Chem. 2018, 6, 26. [Google Scholar] [CrossRef] [PubMed]
- Reddy, A.R.; Chaitanya, K.V.; Vivekanandan, M. Drought-induced responses of photosynthesis and antioxidant metabolism in higher plants. Plant Physiol. 2004, 161, 1189–1202. [Google Scholar] [CrossRef]
- Zhu, J.K. Salt and drought stress signal transduction in plants. Annu. Rev. Plant Biol. 2002, 53, 247–273. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhang, J.; Wang, Z.; Xu, G.; Zhu, Q. Activities of key enzymes in sucrose-to-starch conversion in wheat grains subjected to water deficit during grain filling. Plant Physiol. 2004, 135, 1621–1629. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.L.; Jing, R.L.; Chang, X.P.; Li, W. Identification of quantitative trait loci and environmental interactions for accumulation and remobilization of water-soluble carbohydrates in wheat (Triticum aestivum L.) stems. Genetics 2007, 176, 571–584. [Google Scholar] [CrossRef] [PubMed]
- De Campos, M.K.F.; de Carvalho, K.; de Souza, F.S.; Marur, C.J.; Pereira, L.F.P.; Filho, J.C.B.; Vieira, L.G.E. Drought tolerance and antioxidant enzymatic activity in transgenic ‘Swingle citrumelo plants over-accumulating proline’. Environ. Exp. Bot. 2011, 72, 242–250. [Google Scholar] [CrossRef]
- Valluru, R.; Davies, W.J.; Reynolds, M.P.; Dodd, I.C. Foliar Abscisic Acid-To-Ethylene Accumulation and Response Regulate Shoot Growth Sensitivity to Mild Drought in Wheat. Front. Plant Sci. 2016, 7, 461. [Google Scholar] [CrossRef]
- Bennani, S.; Nsarellah, N.; Birouk, A.; Ouabbou, H.; Tadesse, W. Effective Selection Criteria for Screening Drought Tolerant and High Yielding Bread Wheat Genotypes. Univers. J. Agric. Res. 2016, 4, 134–142. [Google Scholar] [CrossRef]
- Bernardo, R. Molecular Markers and Selection for Complex Traits in Plants: Learning from the Last 20 Years. Crop Sci. 2008, 48, 1649. [Google Scholar] [CrossRef]
- Eltaher, S.; Sallam, A.; Belamkar, V.; Emara, H.A.; Nower, A.A.; Salem, K.F.M.; Poland, J.; Baenziger, P.S. Genetic Diversity and Population Structure of F3:6 Nebraska Winter Wheat Genotypes Using Genotyping-By-Sequencing. Front. Genet. 2018, 9, 76. [Google Scholar] [CrossRef]
- Reinert, S.; Kortz, A.; Léon, J.; Naz, A.A. Genome-Wide Association Mapping in the Global Diversity Set Reveals New QTL Controlling Root System and Related Shoot Variation in Barley. Front. Plant Sci. 2016, 7, 1061. [Google Scholar] [CrossRef] [PubMed]
- Mwadzingeni, L.; Shimelis, H.; Rees, D.J.G.; Tsilo, T.J. Genome-wide association analysis of agronomic traits in wheat under drought-stressed and non-stressed conditions. PLoS ONE 2017, 12, e0171692. [Google Scholar] [CrossRef] [PubMed]
- Sallam, A.; Mourad, A.M.I.; Hussain, W.; Stephen Baenziger, P. Genetic variation in drought tolerance at seedling stage and grain yield in low rainfall environments in wheat (Triticum aestivum L.). Euphytica 2018, 214, 169. [Google Scholar] [CrossRef]
- Sallam, A.; Dhanapal, A.P.; Liu, S. Association mapping of winter hardiness and yield traits in faba bean (Vicia faba L.). Crop Pasture Sci. 2016, 67, 55. [Google Scholar] [CrossRef]
- Srivastava, L.M. Plant growth and development: hormones and environment. Ann Bot. 2003, 92, 846. [Google Scholar]
- Kerr, J.M. Sustainable Development of Rainfed Agriculture in India. Available online: https://ageconsearch.umn.edu/record/16104/ (accessed on 20 May 2019).
- Kido, É.A.; Ferreira-Neto, J.R.C.; Pandolfi, V.; de Melo Souza, A.C.; Benko-Iseppon, A.M. Drought Stress Tolerance in Plants: Insights from Transcriptomic Studies. In Drought Stress Tolerance in Plants, Vol 2; Springer: Cham, Switzerland, 2016; pp. 153–185. [Google Scholar]
- Khazayi, H.; Kafi, M.; Masumi, A. Physiological effects of stress induced by polyethylene glycol on germination of chickpea genotypes. J. Agron. Res. Iran 2008, 2, 453. [Google Scholar]
- Gallagher, J.N.; Biscoe, P.V.; Hunter, B. Effects of drought on grain growth. Nature 1976, 264, 541–542. [Google Scholar] [CrossRef]
- Hameed, A.; Goher, M.; Iqbal, N. Evaluation of Seedling Survivability and Growth Response as Selection Criteria for Breeding Drought Tolerance in Wheat. Cereal Res. Commun. 2010, 38, 193–202. [Google Scholar] [CrossRef]
- Basu, S.; Ramegowda, V.; Kumar, A.; Pereira, A. Plant adaptation to drought stress. F1000Research 2016, 5. [Google Scholar] [CrossRef]
- Muir, C.D.; Thomas-Huebner, M. Constraint around Quarter-Power Allometric Scaling in Wild Tomatoes (Solanum sect. Lycopersicon; Solanaceae). Am. Nat. 2015, 186, 421–433. [Google Scholar] [CrossRef]
- Drira, M.; Hanin, M.; Masmoudi, K.; Brini, F.; Amara, I.; Odena, A.; Eliandre-Oliveira, E.; Moreno, A.; Masmoudi, K.; Pagès, M.; et al. Comparison of full-length and conserved segments of wheat dehydrin DHN-5 overexpressed in Arabidopsis thaliana showed different responses to abiotic and biotic stress. Funct. Plant Biol. 2016, 43, 1048. [Google Scholar] [CrossRef]
- Rebetzke, G.J.; Jimenez-Berni, J.A.; Bovill, W.D.; Deery, D.M.; James, R.A. High-throughput phenotyping technologies allow accurate selection of stay-green. J. Exp. Bot. 2016, 67, 4919–4924. [Google Scholar] [CrossRef] [PubMed]
- Harb, A.; Krishnan, A.; Ambavaram, M.M.R.; Pereira, A. Molecular and physiological analysis of drought stress in Arabidopsis reveals early responses leading to acclimation in plant growth. Plant Physiol. 2010, 154, 1254–1271. [Google Scholar] [CrossRef] [PubMed]
- Falconer, D.; Mackay, T. Introduction to Quantitative Genetics, 4th ed.Prentice Hall: Harlow, UK, 1996; Available online: https://www.cabdirect.org/cabdirect/abstract/19601603365 (accessed on 20 May 2019).
- Sallam, A.; Martsch, R.; Moursi, Y.S. Genetic variation in morpho-physiological traits associated with frost tolerance in faba bean (Vicia faba L.). Euphytica 2015, 205, 395–408. [Google Scholar] [CrossRef]
- Sallam, A.; Hussain, W.; Belamkar, V.; Baenziger, S.P. Molecular genetic dissection to improve drought tolerance in winter wheat using QTL mapping. In Proceedings of the Plant Science Retreat, University of Nebraska-Lincoln, Nebraska City, NE, USA, 14–15 October 2016. [Google Scholar]
- Sallam, A.; Hamed, E.-S.; Hashad, M.; Omara, M. Inheritance of stem diameter and its relationship to heat and drought tolerance in wheat (Triticum aestivum L.). J. Plant Breed. Crop Sci. 2014, 6, 11–23. [Google Scholar]
- Fernandez, G.C.J. Effective Selection Criteria for Assessing Stress Tolerance. In Proceedings of the International Symposium on Adaptation of Vegetables and Other Food Crops in Temperature and Water Stress, Taibei, Taiwan, 13–16 August 1992. [Google Scholar]
- Lazar, M.D.; Salisbury, C.D.; Worrall, W.D. Variation in drought susceptibility among closely related wheat lines. F. Crop. Res. 1995, 41, 147–153. [Google Scholar] [CrossRef]
- Kumar Joshi, R.; Nayak, S. Gene pyramiding-A broad spectrum technique for developing durable stress resistance in crops. Biotechnol. Mol. Biol. Rev. 2010, 5, 51–60. [Google Scholar]
- Sallam, A.; Hashad, M.; Hamed, E.-S.; Omara, M. Genetic variation of stem characters in wheat and their relation to kernel weight under drought and heat stresses. J. Crop Sci Biotechnol. 2015, 18. [Google Scholar]
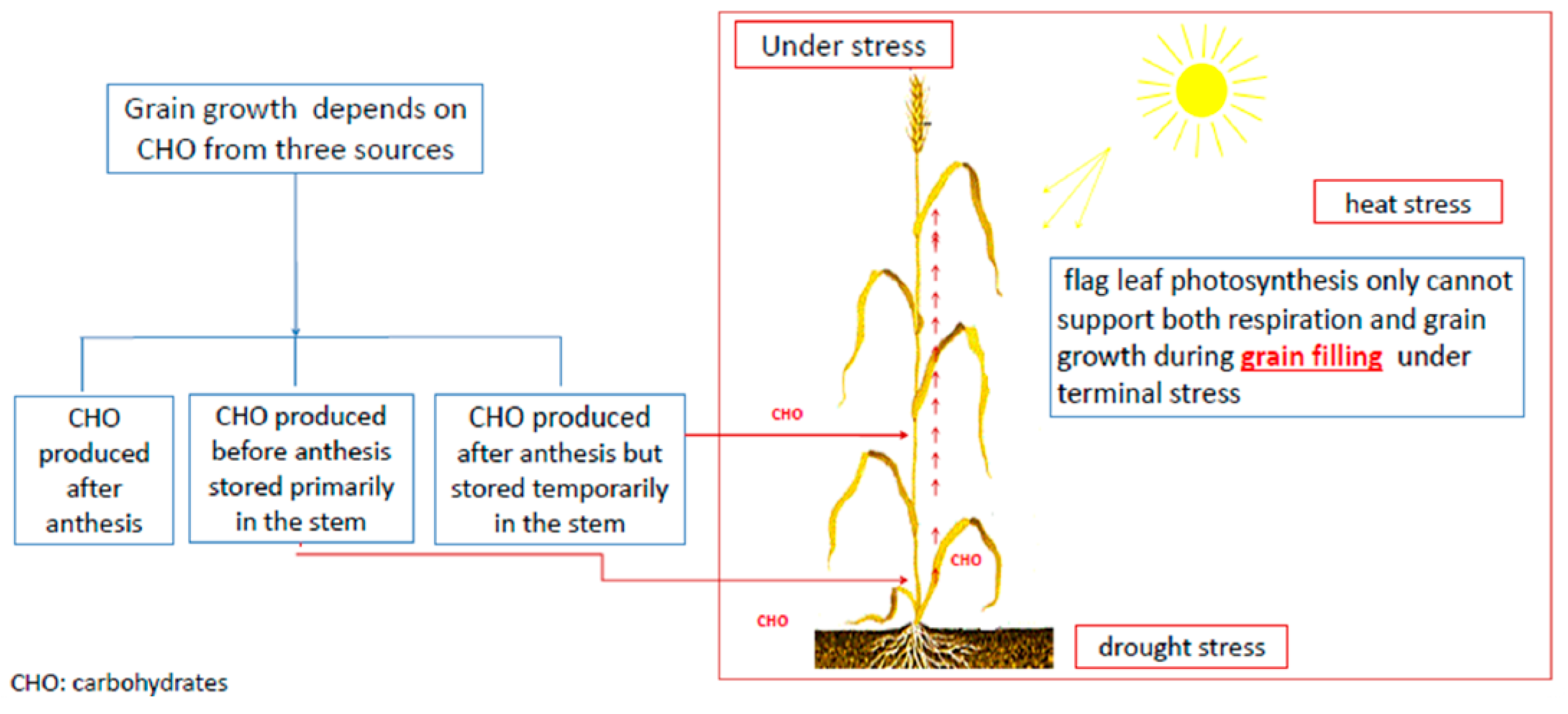
- Kobata, T.; Palta, J.A.; Turner, N.C. Rate of Development of Postanthesis Water Deficits and Grain Filling of Spring Wheat. Crop Sci 1992, 32, 1238. [Google Scholar] [CrossRef]
- WARDLAW, I.F.; WILLENBRINK, J. Mobilization of fructan reserves and changes in enzyme activities in wheat stems correlate with water stress during kernel filling. New Phytol. 2000, 148, 413–422. [Google Scholar] [CrossRef]
- Gent, M.P.N.; Kiyomoto, R.K. Comparison of Canopy and Flag Leaf Net Carbon Dioxide Exchange of 1920 and 1977 New York Winter Wheats1. Crop Sci. 1985, 25, 81. [Google Scholar] [CrossRef]
- Gent, M.P.N. Photosynthate Reserves during Grain Filling in Winter Wheat. Agron. J. 1994, 86, 159. [Google Scholar] [CrossRef]
- Ehdaie, B.; Alloush, G.A.; Madore, M.A.; Waines, J.G. Genotypic Variation for Stem Reserves and Mobilization in Wheat. Crop Sci 2006, 46, 2093. [Google Scholar] [CrossRef]
- Xue, G.-P.; McIntyre, C.L.; Jenkins, C.L.D.; Glassop, D.; van Herwaarden, A.F.; Shorter, R. Molecular dissection of variation in carbohydrate metabolism related to water-soluble carbohydrate accumulation in stems of wheat. Plant Physiol. 2008, 146, 441–454. [Google Scholar] [CrossRef] [PubMed]
- Araus, J.L.; Cairns, J.E. Field high-throughput phenotyping: the new crop breeding frontier. Trends Plant Sci 2014, 19, 52–61. [Google Scholar] [CrossRef]
- Winterhalter, L.; Mistele, B.; Schmidhalter, U. Evaluation of active and passive sensor systems in the field to phenotype maize hybrids with high-throughput. F. Crop. Res. 2013, 154, 236–245. [Google Scholar] [CrossRef]
- Erdle, K.; Mistele, B.; Schmidhalter, U. Comparison of active and passive spectral sensors in discriminating biomass parameters and nitrogen status in wheat cultivars. F. Crop. Res. 2011, 124, 74–84. [Google Scholar] [CrossRef]
- Hackl, H.; Baresel, J.P.; Mistele, B.; Hu, Y.; Schmidhalter, U. A Comparison of Plant Temperatures as Measured by Thermal Imaging and Infrared Thermometry. J. Agron. Crop Sci 2012, 198, 415–429. [Google Scholar] [CrossRef]
- Cairns, J.E.; Sonder, K.; Zaidi, P.H.; Verhulst, N.; Mahuku, G.; Babu, R.; Nair, S.K.; Das, B.; Govaerts, B.; Vinayan, M.T.; et al. Maize Production in a Changing Climate: Impacts, Adaptation, and Mitigation Strategies. Adv. Agron. 2012, 114, 1–58. [Google Scholar]
- Kipp, S.; Mistele, B.; Baresel, P.; Schmidhalter, U. High-throughput phenotyping early plant vigour of winter wheat. Eur. J. Agron. 2014, 52, 271–278. [Google Scholar] [CrossRef]
- Richard, C.; Hickey, L.; Fletcher, S.; Chenu, K.; Borrell, A.; Christopher, J. High-throughput Phenotyping of Wheat Seminal Root Traits in a Breeding Context. Procedia Environ. Sci 2015, 29, 102–103. [Google Scholar] [CrossRef]
- Ghanem, M.E.; Lè Ne Marrou, H.; Sinclair, T.R. Physiological phenotyping of plants for crop improvement. Trends Plant Sci 2015, 20, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Jsarotia, P.; Kashyap, P.L.; Bhardwaj, A.K.; Kumar, S.; Singh, G.P. Nanotechnology Scope and Applications for Wheat Production and Quality Enhancement:A Review of Recent Advances. Wheat Barley Res. 2018, 10, 137–150. [Google Scholar] [CrossRef]
- Taran, N.; Storozhenko, V.; Svietlova, N.; Batsmanova, L.; Shvartau, V.; Kovalenko, M. Effect of Zinc and Copper Nanoparticles on Drought Resistance of Wheat Seedlings. Nanoscale Res. Lett. 2017, 12, 60. [Google Scholar] [CrossRef] [PubMed]
- Olkhovych, O.; Volkogon, M.; Taran, N.; Batsmanova, L.; Kravchenko, I. The Effect of Copper And Zinc Nanoparticles on the Growth Parameters, Contents of Ascorbic Acid, and Qualitative Composition of Amino Acids and Acylcarnitines in Pistia stratiotes L. (Araceae). Nanoscale Res. Lett. 2016, 11, 218. [Google Scholar] [CrossRef] [PubMed]
- Konotop, I.O.; Kovalenko, M.S.; Ulynets, V.Z.; Meleshko, A.O.; Batsmanova, L.M.; Taran, N.I. Phytotoxicity of colloidal solutions of metal-containing nanoparticles. Tsitol. Genet. 2014, 48, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Ghorbanian, H.; Janmohammadi, M.; Ebadi-Segherloo, A.; Sabaghnia, N. Genotypic response of barley to exogenous application of nanoparticles under water stress condition. Ann. Univ. Mariae Curie-Sklodowska, Sect. C – Biol. 2019, 72, 15–27. [Google Scholar] [CrossRef]
- Shafea, A.; Dawood, M.; Zidan, M. Wheat seedlings traits as affected by soaking at titanium dioxide nanoparticles. Environ. Earth Ecol. 2017, 1, 102–111. [Google Scholar] [CrossRef]
- Abdelsalam, N.R.; Abdel-Megeed, A.; Ali, H.M.; Salem, M.Z.M.; Al-Hayali, M.F.A.; Elshikh, M.S. Genotoxicity effects of silver nanoparticles on wheat (Triticum aestivum L.) root tip cells. Ecotoxicol. Environ. Saf. 2018, 155, 76–85. [Google Scholar] [CrossRef]
- Fleury, D.; Jefferies, S.; Kuchel, H.; Langridge, P. Genetic and genomic tools to improve drought tolerance in wheat. J. Exp. Bot. 2010, 61, 3211–3222. [Google Scholar] [CrossRef]
- Hu, H.; Xiong, L. Genetic engineering and breeding of drought-resistant crops. Annu. Rev. Plant Biol. 2014, 65, 715–741. [Google Scholar] [CrossRef] [PubMed]
- Tricker, P.J.; ElHabti, A.; Schmidt, J.; Fleury, D. The physiological and genetic basis of combined drought and heat tolerance in wheat. J. Exp. Bot. 2018, 69, 3195–3210. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.K.; Balyan, H.S.; Gahlaut, V. QTL Analysis for Drought Tolerance in Wheat: Present Status and Future Possibilities. Agronomy-Basel 2017, 7, 5. [Google Scholar] [CrossRef]
- Acuna-Galindo, M.A.; Mason, R.E.; Subramanian, N.K.; Hays, D.B. Meta-Analysis of Wheat QTL Regions Associated with Adaptation to Drought and Heat Stress. Crop Sci 2015, 55, 477–492. [Google Scholar] [CrossRef]
- Mathews, K.L.; Malosetti, M.; Chapman, S.; McIntyre, L.; Reynolds, M.; Shorter, R.; van Eeuwijk, F. Multi-environment QTL mixed models for drought stress adaptation in wheat. Theor. Appl. Genet. 2008, 117, 1077–1091. [Google Scholar] [CrossRef] [PubMed]
- Von Korff, M.; Grando, S.; Del Greco, A.; This, D.; Baum, M.; Ceccarelli, S. Quantitative trait loci associated with adaptation to Mediterranean dryland conditions in barley. Theor. Appl. Genet. 2008, 117, 653–669. [Google Scholar] [CrossRef]
- Maccaferri, M.; Sanguineti, M.C.; Corneti, S.; Ortega, J.L.A.; Salem, M.B.; Bort, J.; DeAmbrogio, E.; del Moral, L.F.G.; Demontis, A.; El-Ahmed, A.; et al. Quantitative Trait Loci for Grain Yield and Adaptation of Durum Wheat (Triticum durum Desf.) Across a Wide Range of Water Availability. Genetics 2008, 178, 489–511. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, C.L.; Mathews, K.L.; Rattey, A.; Chapman, S.C.; Drenth, J.; Ghaderi, M.; Reynolds, M.; Shorter, R. Molecular detection of genomic regions associated with grain yield and yield-related components in an elite bread wheat cross evaluated under irrigated and rainfed conditions. Theor. Appl. Genet. 2010, 120, 527–541. [Google Scholar] [CrossRef]
- Quarrie, S.; Gulli, M.; Calestani, C.; Steed, A.; Marmiroli, N. Location of a gene regulating drought-induced abscisic acid production on the long arm of chromosome 5A of wheat. Theor. Appl. Genet. 1994, 89, 794–800. [Google Scholar] [CrossRef]
- Obsa, B.T.; Eglinton, J.; Coventry, S.; March, T.; Langridge, P.; Fleury, D. Genetic analysis of developmental and adaptive traits in three doubled haploid populations of barley (Hordeum vulgare L.). Theor. Appl. Genet. 2016, 129, 1139–1151. [Google Scholar] [CrossRef]
- Kalladan, R.; Worch, S.; Rolletschek, H.; Harshavardhan, V.T.; Kuntze, L.; Seiler, C.; Sreenivasulu, N.; Roder, M.S. Identification of quantitative trait loci contributing to yield and seed quality parameters under terminal drought in barley advanced backcross lines. Mol. Breed. 2013, 32, 71–90. [Google Scholar] [CrossRef]
- Malosetti, M.; Voltas, J.; Romagosa, I.; Ullrich, S.E.; van Eeuwijk, F.A. Mixed models including environmental covariables for studying QTL by environment interaction. Euphytica 2004, 137, 139–145. [Google Scholar] [CrossRef]
- Samarah, N.; Alqudah, A. Effects of late-terminal drought stress on seed germination and vigor of barley (Hordeum vulgare L.). Arch. Agron. Soil Sci 2011, 57, 27–32. [Google Scholar] [CrossRef]
- Czyczyło-Mysza, I.; Marcińska, I.; Skrzypek, E.; Cyganek, K.; Juzoń, K.; Karbarz, M. QTL mapping for germination of seeds obtained from previous wheat generation under drought. Open Life Sci. 2014, 9, 374. [Google Scholar] [CrossRef]
- Nakamura, S.; Pourkheirandish, M.; Morishige, H.; Sameri, M.; Sato, K.; Komatsuda, T. Quantitative Trait Loci and Maternal Effects Affecting the Strong Grain Dormancy of Wild Barley (Hordeum vulgare ssp. spontaneum). Front Plant Sci 2017, 8, 1840. [Google Scholar] [CrossRef]
- Campoli, C.; von Korff, M. Genetic Control of Reproductive Development in Temperate Cereals. In The Molecular Genetics of Floral Transition and Flower Development; Fabio, F., Ed.; Elsevier: Amsterdam, The Netherlands, 2014; Volume 72, pp. 131–158. [Google Scholar]
- Tahmasebi, S.; Heidari, B.; Pakniyat, H.; McIntyre, C.L. Mapping QTLs associated with agronomic and physiological traits under terminal drought and heat stress conditions in wheat (Triticum aestivum L.). Genome 2017, 60, 26–45. [Google Scholar] [CrossRef] [PubMed]
- Mora, F.; Quitral, Y.A.; Matus, I.; Russell, J.; Waugh, R.; Del Pozo, A. SNP-Based QTL Mapping of 15 Complex Traits in Barley under Rain-Fed and Well-Watered Conditions by a Mixed Modeling Approach. Front. Plant Sci 2016, 7, 909. [Google Scholar] [CrossRef]
- Arifuzzaman, M.; Sayed, M.A.; Muzammil, S.; Pillen, K.; Schumann, H.; Naz, A.A.; Leon, J. Detection and validation of novel QTL for shoot and root traits in barley (Hordeum vulgare L.). Mol. Breed. 2014, 34, 1373–1387. [Google Scholar] [CrossRef]
- Ocaña-Moral, S.; Gutiérrez, N.; Torres, A.M.; Madrid, E. Saturation mapping of regions determining resistance to Ascochyta blight and broomrape in faba bean using transcriptome-based SNP genotyping. Theor. Appl. Genet. 2017, 130, 2271–2282. [Google Scholar] [CrossRef]
- Ogrodowicz, P.; Adamski, T.; Mikolajczak, K.; Kuczynska, A.; Surma, M.; Krajewski, P.; Sawikowska, A.; Gorny, A.G.; Gudys, K.; Szarejko, I.; et al. QTLs for earliness and yield-forming traits in the Lubuski x CamB barley RIL population under various water regimes. J. Appl. Genet. 2017, 58, 49–65. [Google Scholar] [CrossRef]
- Xu, Y.F.; Li, S.S.; Li, L.H.; Ma, F.F.; Fu, X.Y.; Shi, Z.L.; Xu, H.X.; Ma, P.T.; An, D.G. QTL mapping for yield and photosynthetic related traits under different water regimes in wheat. Mol. Breed. 2017, 37, 34. [Google Scholar] [CrossRef]
- Alqudah, A.M.; Samarah, N.H.; Mullen, R.E. Drought Stress Effect on Crop Pollination, Seed Set, Yield and Quality. In Alternative Farming Systems, Biotechnology, Drought Stress and Ecological Fertilisation; Lichtfouse, E., Ed.; Springer: Dordrecht, The Netherlands, 2011; Volume 6, pp. 193–213. [Google Scholar]
- Parent, B.; Shahinnia, F.; Maphosa, L.; Berger, B.; Rabie, H.; Chalmers, K.; Kovalchuk, A.; Langridge, P.; Fleury, D. Combining field performance with controlled environment plant imaging to identify the genetic control of growth and transpiration underlying yield response to water-deficit stress in wheat. J. Exp. Bot. 2015, 66, 5481–5492. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Chang, X.; Jing, R. Genetic analysis of carbon isotope discrimination and its relation to yield in a wheat doubled haploid population. J. Integr. Plant Biol. 2011, 53, 719–730. [Google Scholar] [CrossRef] [PubMed]
- Honsdorf, N.; March, T.J.; Pillen, K. QTL controlling grain filling under terminal drought stress in a set of wild barley introgression lines. PLoS ONE 2017, 12, e0185983. [Google Scholar] [CrossRef] [PubMed]
- Peleg, Z.V.I.; Fahima, T.; Krugman, T.; Abbo, S.; Yakir, D.A.N.; Korol, A.B.; Saranga, Y. Environment Genomic dissection of drought resistance in durum wheat× wild emmer wheat recombinant inbreed line population. Plant Cell Environ. 2009, 32, 758–779. [Google Scholar] [CrossRef]
- Kadam, S.; Singh, K.; Shukla, S.; Goel, S.; Vikram, P.; Pawar, V.; Gaikwad, K.; Khanna-Chopra, R.; Singh, N. Genomic associations for drought tolerance on the short arm of wheat chromosome 4B. Funct Integr Genomics 2012, 12, 447–464. [Google Scholar] [CrossRef] [PubMed]
- Peleg, Z.; Blumwald, E. Hormone balance and abiotic stress tolerance in crop plants. Curr. Opin. Plant Biol. 2011, 14, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Guo, P.; Baum, M.; Grando, S.; Ceccarelli, S.; Bai, G.; Li, R.; von Korff, M.; Varshney, R.K.; Graner, A.; Valkoun, J. Differentially expressed genes between drought-tolerant and drought-sensitive barley genotypes in response to drought stress during the reproductive stage. J. Exp. Bot. 2009, 60, 3531–3544. [Google Scholar] [CrossRef]
- Teulat, B.; Merah, O.; Sirault, X.; Borries, C.; Waugh, R.; This, D. QTLs for grain carbon isotope discrimination in field-grown barley. Theor. Appl. Genet. 2002, 106, 118–126. [Google Scholar] [CrossRef]
- Diab, A.A.; Teulat-Merah, B.; This, D.; Ozturk, N.Z.; Benscher, D.; Sorrells, M.E. Identification of drought-inducible genes and differentially expressed sequence tags in barley. Theor. Appl. Genet. 2004, 109, 1417–1425. [Google Scholar] [CrossRef]
- Shahinnia, F.; Le Roy, J.; Laborde, B.; Sznajder, B.; Kalambettu, P.; Mahjourimajd, S.; Tilbrook, J.; Fleury, D. Genetic association of stomatal traits and yield in wheat grown in low rainfall environments. BMC Plant Biol. 2016, 16, 150. [Google Scholar] [CrossRef] [PubMed]
- Sayed, M.A.; Schumann, H.; Pillen, K.; Naz, A.A.; Leon, J. AB-QTL analysis reveals new alleles associated to proline accumulation and leaf wilting under drought stress conditions in barley (Hordeum vulgare L.). BMC Genet. 2012, 13, 61. [Google Scholar] [CrossRef] [PubMed]
- Naz, A.A.; Arifuzzaman, M.; Muzammil, S.; Pillen, K.; Leon, J. Wild barley introgression lines revealed novel QTL alleles for root and related shoot traits in the cultivated barley (Hordeum vulgare L.). BMC Genet. 2014, 15, 107. [Google Scholar] [CrossRef] [PubMed]
- Iehisa, J.C.; Matsuura, T.; Mori, I.C.; Takumi, S. Identification of quantitative trait locus for abscisic acid responsiveness on chromosome 5A and association with dehydration tolerance in common wheat seedlings. J. Plant Physiol. 2014, 171, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Castro, A.M.; Tacaliti, M.S.; Gimenez, D.; Tocho, E.; Dobrovolskaya, O.; Vasicek, A.; Collado, M.; Snape, J.W.; Borner, A. Mapping quantitative trait loci for growth responses to exogenously applied stress induced hormones in wheat. Euphytica 2008, 164, 719–727. [Google Scholar] [CrossRef]
- Li, W.-T.; Liu, C.; Liu, Y.-X.; Pu, Z.-E.; Dai, S.-F.; Wang, J.-R.; Lan, X.-J.; Zheng, Y.-L.; Wei, Y.-M. Meta-analysis of QTL associated with tolerance to abiotic stresses in barley. Euphytica 2012, 189, 31–49. [Google Scholar] [CrossRef]
- Jain, N.; Singh, G.P.; Singh, P.K.; Ramya, P.; Krishna, H.; Ramya, K.T.; Todkar, L.; Amasiddha, B.; Kumar, K.C.P.; Vijay, P. Molecular approaches for wheat improvement under drought and heat stress. Indian J. Genet. Plant Breed. 2014. [Google Scholar] [CrossRef]
- Ain, Q.U.; Rasheed, A.; Anwar, A.; Mahmood, T.; Imtiaz, M.; Mahmood, T.; Xia, X.; He, Z.; Quraishi, U.M. Genome-wide association for grain yield under rainfed conditions in historical wheat cultivars from Pakistan. Front. Plant Sci 2015, 6, 743. [Google Scholar] [CrossRef]
- Beyer, S.; Daba, S.; Tyagi, P.; Bockelman, H.; Brown-Guedira, G.; Mohammadi, M. Loci and candidate genes controlling root traits in wheat seedlings-a wheat root GWAS. Funct. Integr. Genomics 2019, 19, 91–107. [Google Scholar] [CrossRef]
- Lozada, D.N.; Mason, R.E.; Babar, M.A.; Carver, B.F.; Guedira, G.B.; Merrill, K.; Arguello, M.N.; Acuna, A.; Vieira, L.; Holder, A.; et al. Association mapping reveals loci associated with multiple traits that affect grain yield and adaptation in soft winter wheat. Euphytica 2017, 213, 222. [Google Scholar] [CrossRef]
- Petrarulo, M.; Marone, D.; Ferragonio, P.; Cattivelli, L.; Rubiales, D.; De Vita, P.; Mastrangelo, A.M. Genetic analysis of root morphological traits in wheat. Mol. Genet. Genomics 2015, 290, 785–806. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Verma, B.; Kumar, N.; Chahota, R.K.; Rathour, R.; Sharma, S.K.; Bhatia, S.; Sharma, T.R. Construction of intersubspecific molecular genetic map of lentil based on ISSR, RAPD and SSR markers. J. Genet. 2012, 91, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Liu, J.; Zhang, Y.; Geng, H.; Rasheed, A.; Xiao, Y.; Cao, S.; Fu, L.; Yan, J.; Wen, W.; et al. Genome-Wide Association of Stem Water Soluble Carbohydrates in Bread Wheat. PLoS ONE 2016, 11, e0164293. [Google Scholar] [CrossRef] [PubMed]
- Bhatta, M.; Morgounov, A.; Belamkar, V.; Baenziger, P.S. Genome-Wide Association Study Reveals Novel Genomic Regions for Grain Yield and Yield-Related Traits in Drought-Stressed Synthetic Hexaploid Wheat. Int. J. Mol. Sci 2018, 19, 3011. [Google Scholar] [CrossRef] [PubMed]
- Varshney, R.K.; Paulo, M.J.; Grando, S.; van Eeuwijk, F.A.; Keizer, L.C.P.; Guo, P.; Ceccarelli, S.; Kilian, A.; Baum, M.; Graner, A. Genome wide association analyses for drought tolerance related traits in barley (Hordeum vulgare L.). F. Crop. Res. 2012, 126, 171–180. [Google Scholar] [CrossRef]
- Jabbari, M.; Fakheri, B.A.; Aghnoum, R.; Mahdi Nezhad, N.; Ataei, R. GWAS analysis in spring barley (Hordeum vulgare L.) for morphological traits exposed to drought. PLoS ONE 2018, 13, e0204952. [Google Scholar] [CrossRef]
- Al-Abdallat, A.M.; Karadsheh, A.; Hadadd, N.I.; Akash, M.W.; Ceccarelli, S.; Baum, M.; Hasan, M.; Jighly, A.; Elenein, J.M.A.; Abu Elenein, J.M. Assessment of genetic diversity and yield performance in Jordanian barley (Hordeum vulgare L.) landraces grown under Rainfed conditions. BMC Plant Biol. 2017, 17, 191. [Google Scholar] [CrossRef]
- Song, S.-Y.; Chen, Y.; Chen, J.; Dai, X.-Y.; Zhang, W.-H. Physiological mechanisms underlying OsNAC5-dependent tolerance of rice plants to abiotic stress. Planta 2011, 234, 331–345. [Google Scholar] [CrossRef]
- Wehner, G.; Balko, C.; Humbeck, K.; Zyprian, E.; Ordon, F. Expression profiling of genes involved in drought stress and leaf senescence in juvenile barley. BMC Plant Biol. 2016, 16, 3. [Google Scholar] [CrossRef]
- Wojcik-Jagla, M.; Fiust, A.; Koscielniak, J.; Rapacz, M. Association mapping of drought tolerance-related traits in barley to complement a traditional biparental QTL mapping study. Theor. Appl. Genet. 2018, 131, 167–181. [Google Scholar] [CrossRef]
- Abou-Elwafa, S.F. Association mapping for drought tolerance in barley at the reproductive stage. C. R. Biol. 2016, 339, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Wehner, G.G.; Balko, C.C.; Enders, M.M.; Humbeck, K.K.; Ordon, F.F. Identification of genomic regions involved in tolerance to drought stress and drought stress induced leaf senescence in juvenile barley. BMC Plant Biol 2015, 15, 125. [Google Scholar] [CrossRef] [PubMed]
- Jafarzadeh, J.; Bonnett, D.; Jannink, J.L.; Akdemir, D.; Dreisigacker, S.; Sorrells, M.E. Breeding Value of Primary Synthetic Wheat Genotypes for Grain Yield. PLoS ONE 2016, 11, e0162860. [Google Scholar] [CrossRef] [PubMed]
- Samarah, N.H. Understanding How Plants Respond to Drought Stress at the Molecular and Whole Plant Levels. In Drought Stress Tolerance in Plants, Vol 2: Molecular and Genetic Perspectives; Hossain, A.M., Wani, H.S., Bhattacharjee, S., Burritt, J.D., Tran, P.L.-S., Eds.; Springer: Cham, Switzerland, 2016; pp. 1–37. [Google Scholar]
- Bi, H.; Luang, S.; Li, Y.; Bazanova, N.; Morran, S.; Song, Z.; Perera, M.A.; Hrmova, M.; Borisjuk, N.; Lopato, S. Identification and characterization of wheat drought-responsive MYB transcription factors involved in the regulation of cuticle biosynthesis. J. Exp. Bot. 2016, 67, 5363–5380. [Google Scholar] [CrossRef] [PubMed]
- Saeid, M.A.R.; Tarek, G.A.; Leena, A.I.; Naser, M.S.; Maen, K.H.; Hazem, S.H. Cloning and expression patterns of the HvP5CS gene from barley (Hordeum vulgare). J. Food Agric. Environ. 2011, 9, 279–284. [Google Scholar]
- Bahieldin, A.; Mahfouz, H.T.; Eissa, H.F.; Saleh, O.M.; Ramadan, A.M.; Ahmed, I.A.; Dyer, W.E.; El-Itriby, H.A.; Madkour, M.A. Field evaluation of transgenic wheat plants stably expressing the HVA1 gene for drought tolerance. Physiol. Plant. 2005, 123, 421–427. [Google Scholar] [CrossRef]
- Abebe, T.; Guenzi, A.C.; Martin, B.; Cushman, J.C. Tolerance of mannitol-accumulating transgenic wheat to water stress and salinity. Plant Physiol. 2003, 131, 1748–1755. [Google Scholar] [CrossRef]
- Shavrukov, Y.; Baho, M.; Lopato, S.; Langridge, P. The Ta DREB 3 transgene transferred by conventional crossings to different genetic backgrounds of bread wheat improves drought tolerance. Plant Biotechnol. J. 2016, 14, 313–322. [Google Scholar] [CrossRef]
- Zhou, R.; Hyldgaard, B.; Yu, X.; Rosenqvist, E.; Ugarte, R.M.; Yu, S.; Wu, Z.; Ottosen, C.-O.; Zhao, T. Phenotyping of faba beans (Vicia faba L.) under cold and heat stresses using chlorophyll fluorescence. Euphytica 2018, 214, 68. [Google Scholar] [CrossRef]
- Morran, S.; Eini, O.; Pyvovarenko, T.; Parent, B.; Singh, R.; Ismagul, A.; Eliby, S.; Shirley, N.; Langridge, P.; Lopato, S. Improvement of stress tolerance of wheat and barley by modulation of expression of DREB/CBF factors. Plant Biotechnol. J. 2011, 9, 230–249. [Google Scholar] [CrossRef]
- Al Abdallat, A.M.; Ayad, J.Y.; Abu Elenein, J.M.; Al Ajlouni, Z.; Harwood, W.A. Overexpression of the transcription factor HvSNAC1 improves drought tolerance in barley (Hordeum vulgare L.). Mol. Breed. 2014, 33, 401–414. [Google Scholar] [CrossRef]
- Hussain, W.; Guttieri, M.J.; Belamkar, V.; Poland, J.; Sallam, A.; Baenziger, P.S. Registration of a Bread Wheat Recombinant Inbred Line Mapping Population Derived from a Cross Between ‘Harry’ and ‘Wesley’. J. Plant Regist. 2018, 12, 411–414. [Google Scholar] [CrossRef]
- Sallam, A.; Arbaoui, M.; El-Esawi, M.; Abshire, N.; Martsch, R. Identification and Verification of QTL Associated with Frost Tolerance Using Linkage Mapping and GWAS in Winter Faba Bean. Front. Plant Sci. 2016, 7, 1098. [Google Scholar] [CrossRef]
- Sallam, A.; Amro, A.; EL-Akhdar, A.; Dawood, M.F.A.; Kumamaru, T.; Stephen Baenziger, P. Genetic diversity and genetic variation in morpho-physiological traits to improve heat tolerance in Spring barley. Mol. Biol. Rep. 2018, 45, 2441–2453. [Google Scholar] [CrossRef] [PubMed]

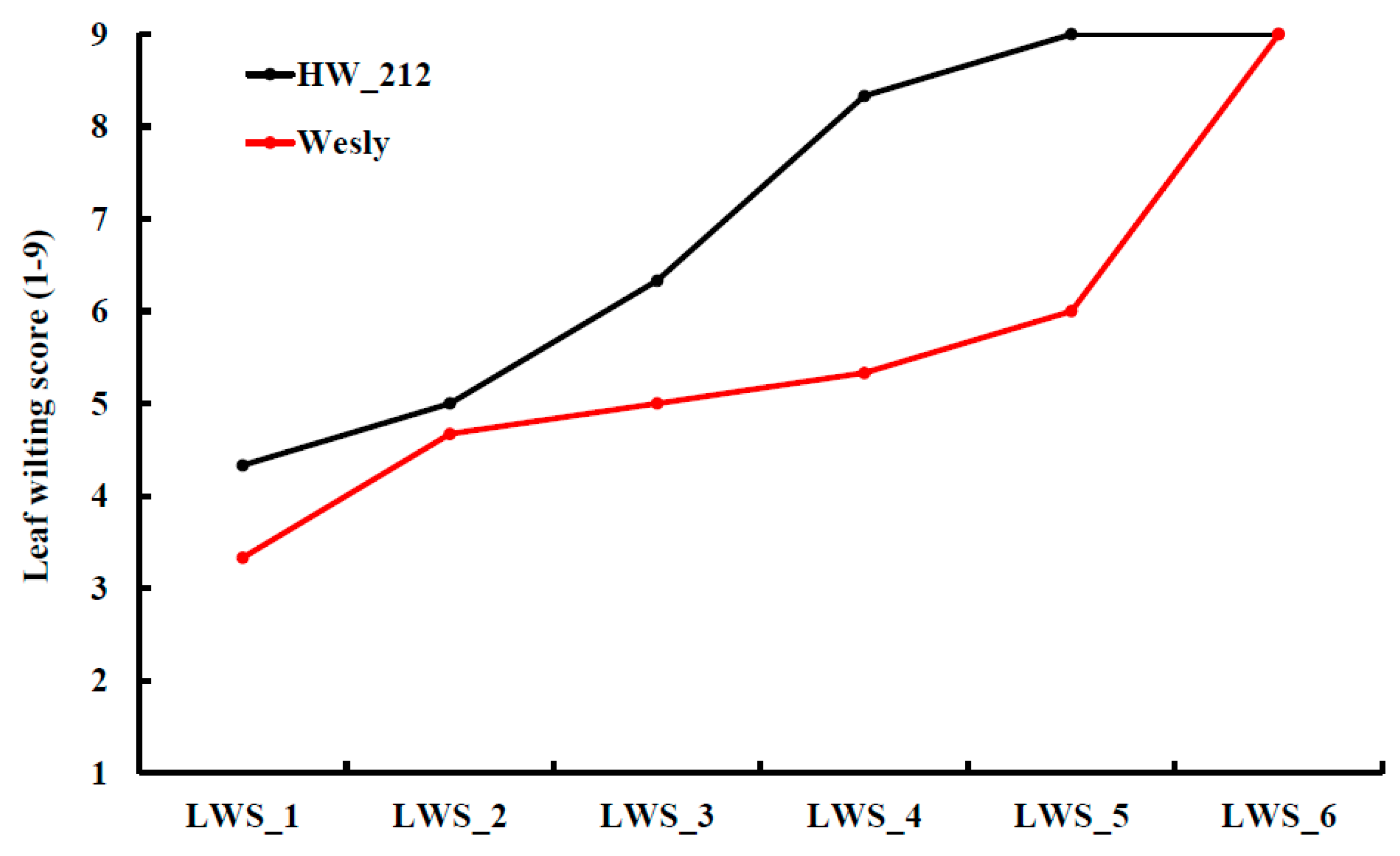


| Traits | Crop | Reference |
|---|---|---|
| Superoxide dismutase | Wheat | [62] |
| Barley | [63] | |
| Glutathione reductase | Wheat | [64] Shan et al. 2018 |
| Glutathione peroxidase | Wheat | [64,65] |
| Ascorbate peroxidase | Wheat | Shan et al. 2018 |
| Barley | [63] | |
| Monodehydroascorbate reductase (MDHAR) | Wheat | Shan et al. 2018 |
| Dehydroascorbate reductase (DHAR) | Wheat | Shan et al. 2018 |
| Catalase | Wheat | [62,66] |
| Barley | [65] | |
| Guaiacol peroxidase | Wheat | [66] |
| Barley | [65] | |
| PAL | Wheat | [66] |
| GST | Wheat | [66] |
| Traits | Crop | Chr. | Reference |
|---|---|---|---|
| Agronomic traits | |||
| Grain yield | Wheat | 1B, 1D, 3B, 4A, 6D, 7D | [162] |
| Barley | 1H, 2H, 3H, 6H | [163,164] | |
| Grain weight spike−1 | Wheat | 1B, 1D | [167] |
| Barley | 2H, 4H, 5H, 6H | [166,171] | |
| Thousand grain weight | Wheat | 1B, 1D, 2A, 2B, 3A, 3B, 4A, 4D, 6A, 6D, 7B, 7D | [167,172] |
| Barley | 2H, 5H, 7H | [169] | |
| Grain number m−2 | Wheat | 1B, 5A, 5B, 7D | [162] |
| Grain number spike−1 | Wheat | 1A, 2A, 2B, 3A, 6B | [167,172] |
| Barley | 2H, 3H, 4H, 5H, 6H | [163,164,166] | |
| Harvest index | Wheat | 1B, 2D, 4BS, 5A | [167] |
| Barley | 1H | [169] | |
| Spike number plant−1 | Wheat | 1A, 2A, 2B, 2D, 4B, 5A, 7B | [167] |
| Barley | 2H, 5H, 6H | [167] | |
| Spikelet compactness | Wheat | 1A, 1B, 2B, 5A, 5B, 6A, 6B, 7A | [167,172] |
| Spikelet number spike−1 | Wheat | 1B, 1D, 2B, 3B, 4B, 5A, 6B, 7D | [167,172] |
| Barley | 2H, 5H, 6H | [151] | |
| Sterile spikelet number spike−1 | Wheat | 7A | [167] |
| Fertile spikelet spike−1 | Wheat | 2A | [167] |
| Spike length | Wheat | 2B, 7A, 7B | [167] |
| Biomass | Wheat | 1B | [167] |
| Barley | 2H | [167] | |
| Shoot biomass | Wheat | 4B | [173] |
| Plant height | Wheat | 1B, 4B, 7D | [167,172] |
| Spike length | Wheat | 2B, 7A, 7B | [167] |
| Lateral spikelet traits | Barley | 1H, 2H, 3H, 5H, 6H | [166] |
| Physiological traits | |||
| leaf area, growth rate, transpiration efficiency, water-use efficiency | Wheat | 2A, 2D, 3A, 4B, 6A, | [169] |
| Early vigor, leaf rolling, leaf waxiness, leaf chlorophyll content | Barley | 1H, 2H, 3H, 4H, 5H | [155] |
| Carbon isotope ratio, osmotic potential, chlorophyll content, flag leaf rolling index | wheat | 2B, 4A, 5A, 7B | [174] |
| Chlorophyll and chlorophyll fluorescence parameters | Barley | 2H, 4H, 6H, 7H | [175] |
| Grain carbon isotope discrimination | Barley | 2H, 3H, 6H, 7H | [176] |
| Relative water content | Barley | 6HL | [176] |
| Water-soluble carbohydrate | Barley | 4H | [177] |
| Water-soluble carbohydrate | Wheat | 1A, 1D, 2D, 4A, 6B, 7B, 7D | [95] |
| Stomatal density, index, aperture area, length; Guard cell area and length | wheat | 2B, 4AS, 5AS, 7AL, 7BL; 1BL, 4BS, 5BS, 7AS | [178] |
| Stomatal conductance, Net photosynthetic rate | wheat | 5A, 6B | [167] |
| Leaf wilting | Barley | 1H, 2H, 3H, 4H | [179] |
| Root length | Wheat | 2D, 4B, 5D, 6B | [173] |
| Barley | 2H, 3H,5H | [180] | |
| Root biomass | Wheat | 2D, 4BS | [173] |
| Barley | 1H, 2H, 3H, 4H, 5H, 7H | [180] | |
| Metabolite traits | |||
| Proline content | Barley | 3H, 4H, 5H, 6H | [179] |
| Abscisic acid (ABA) | Wheat | 1B, 2A, 3A, 4D, 5A, 6D, 7B | [181] |
| Jasmonic acid (JA), salicylic acid (SA), ethylene | Wheat | 6A | [182] |
| Traits | Crop | Chr. (pos. (cM or bp*)) | Reference |
|---|---|---|---|
| Agronomic traits | |||
| Grain yield | Wheat | 1A(140), 1B(99), 2B(18), 3B(133), 6A(54), 7B(39–40) | [13] |
| Barley | 1H(133–134), 3H(153–155) | [192] | |
| Grain weight | Wheat | 1A(298646355), 1A(522189599), 2A(758448348),2B(47837996), 2D(617414673), 3A(610441472), 4A(7441672), 4A(73454791), 5A(423673926), 6A(615815033), 7A(30902570), 7A(691163940), 7A(14787746)* | [191] |
| Thousand-grain weight | Wheat | 2A(66–70), 3A(69–74) | [13] |
| Barley | 2H(45–46), 6H(134) | [192] | |
| Grain number spike−1 | Wheat | 2D(128), 4A(132) | [102] |
| Barley | 3H(126–127), 5H(130–131), 6H(44–45) | [193] | |
| Harvest index | Wheat | 3B(194–195), 6B(83) | [185] |
| Barley | 2H(106–107), | [194] | |
| Spikelet number spike−1 | Wheat | 1B(239), 2B(107), 2D(128), 4B(1), 5B(1), 6B(1) | [102] |
| Barley | 7H(106–107) | [194] | |
| Biomass | Wheat | 1A(85–86), 4B(101), 4D(30), 6B(90) | [185] |
| Barley | 1H(87–92), 5H(46–47) | [195] | |
| Plant height | Wheat | 1A(116–117), 1B(51), 2A(45), 2B(79, 107), 2D(128), 3A(9), 4B(31–32), 5B (65), 6A(12), 7A(88), 7B(59) | [107,108] |
| Barley | 5H(86–87) | [193] | |
| Spike length | Wheat | 1B(184), 2B(107–108), 2D(128), 3A(1), 4B(1), 5B(117), 6A(1), 6B(1), 7A(1), 7D(197~206) | [10,97] |
| Barley | 1H(64–65), 2H(3–4, 14–15), 6H(95–96) | [193] | |
| Physiological traits | |||
| Flag leaf area | Wheat | 1A(516732460–575597761), 1B(58989138), 1D(278097355), 2A(29874199), 2A(764065400), 2D(35564010), 4D(54054104), 5A(587423540), 6B(120860110–120860130), 6B(643131336–674558588), 7D(10009696), 7D(558932149), 7D(638535043–638535045)* | [191] |
| Flag leaf length | Wheat | 1B(62791605–667135914), 1D(382219667), 2A(29874199), 2B(140752747), 2D(642055122–71578532), 4A(612662321), 6D(1771825), 6D(463762312), 7B(520419132–68562846)* | [191] |
| Barley | 2H(117–122), 3H(125–126), 4H(68–69), 6H(95–96) | [193] | |
| Flag leaf width | Wheat | 1A(516732460), 1B(453278609–554003233), 1D(16816400), 2B(16009609), 2B(48030550), 2D(32992152), 4B(534722043), 6B(119525401), 6B(220551194), 6B(26200560–320552308), 6B(677338037–73535204), 6D(16376439)* | [191] |
| Barley | 4H(125), 5H(12) | [193] | |
| Branched root length | Wheat | 1A(474451217), 2B(165520954), 6B(292760947, 353776019, 42406493) * | [186] |
| Root diameter | Wheat | 5A(561134164), 5B(699669413– 700035453)* | [186] |
| Root dry matter | Wheat | 1A(508184675), 5B(712600907)* | [186] |
| Root length | Wheat at flowering | 2D(620326979), 3B(757480752), 5B(669373985–669374027), 6A(169248262–169248303), 6D(241296319), 6D(431108774–445773103),7A(94404310)* | [191] |
| Barley at seedling | 1H(46–48), 2H(12–13, 114) | [8] | |
| Seedling shoot length | Barley | 1H(46–48), 2H(12–13, 114) | [8] |
| Seminal axis length | Wheat | 5B(658559755– 711277563) | [186] |
| Stem water soluble carbohydrates | Wheat | 1A(54–58), 1B(159–160), 2B(69–72), 3A(26), 3B(81–83), 3D(130), 4B(62–63) | [190] |
| Water-soluble carbohydrate accumulation | Wheat | 1A)68–69), 1B(11–12), 1D(83–86), 2D (40–41), 4A(62–63) | [9] |
| Germination and seed viability | Barley | 1H(46–48) | [8] |
| Leaf senescenc | Barley | 1H(188–119), 2H(131–132), 3H(142–143), 6H(64–65), 7H(40–41, 81–82) | [196] |
| Water use efficiency, Water content and Relative water content | Barley | 2H(118–119), 3H(24–25), 4H(49–55), 5H(48–49, 147–148) | [197] |
| Net photosynthesis rate, intercellular CO2 concentration, stomatal conductivity | Barley | 3H(51–52), 4H(43–49, 51–52) | [197] |
| Leaf wilting | 3H(49–50), 4H(72–73), 5H(53–54), 6H(75–76), 7H(93–94, 125–126) | [198] | |
| Relative water content | 2H(51–52, 137–138), 7H(88–89, 125–126, 147–148) | [198] | |
| Metabolite traits | |||
| The total content of soluble sugars | Barley | 1H(95) | [199] |
| Osmolality | Barley | 1H(116), 2H(51.8), 3H(2.4), 4H(52.3), 5H(46.5), 6H(10.3), 7H(106.5) | [195] |
| Proline accumulation | Barley | 1H(49–50), 2H(137–138), 3H(1–2, 144–145), 7H(147–154) | [198] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sallam, A.; Alqudah, A.M.; Dawood, M.F.A.; Baenziger, P.S.; Börner, A. Drought Stress Tolerance in Wheat and Barley: Advances in Physiology, Breeding and Genetics Research. Int. J. Mol. Sci. 2019, 20, 3137. https://doi.org/10.3390/ijms20133137
Sallam A, Alqudah AM, Dawood MFA, Baenziger PS, Börner A. Drought Stress Tolerance in Wheat and Barley: Advances in Physiology, Breeding and Genetics Research. International Journal of Molecular Sciences. 2019; 20(13):3137. https://doi.org/10.3390/ijms20133137
Chicago/Turabian StyleSallam, Ahmed, Ahmad M. Alqudah, Mona F. A. Dawood, P. Stephen Baenziger, and Andreas Börner. 2019. "Drought Stress Tolerance in Wheat and Barley: Advances in Physiology, Breeding and Genetics Research" International Journal of Molecular Sciences 20, no. 13: 3137. https://doi.org/10.3390/ijms20133137
APA StyleSallam, A., Alqudah, A. M., Dawood, M. F. A., Baenziger, P. S., & Börner, A. (2019). Drought Stress Tolerance in Wheat and Barley: Advances in Physiology, Breeding and Genetics Research. International Journal of Molecular Sciences, 20(13), 3137. https://doi.org/10.3390/ijms20133137






