Transplantation of Adipose Stromal Cell Sheet Producing Hepatocyte Growth Factor Induces Pleiotropic Effect in Ischemic Skeletal Muscle
Abstract
1. Introduction
2. Results
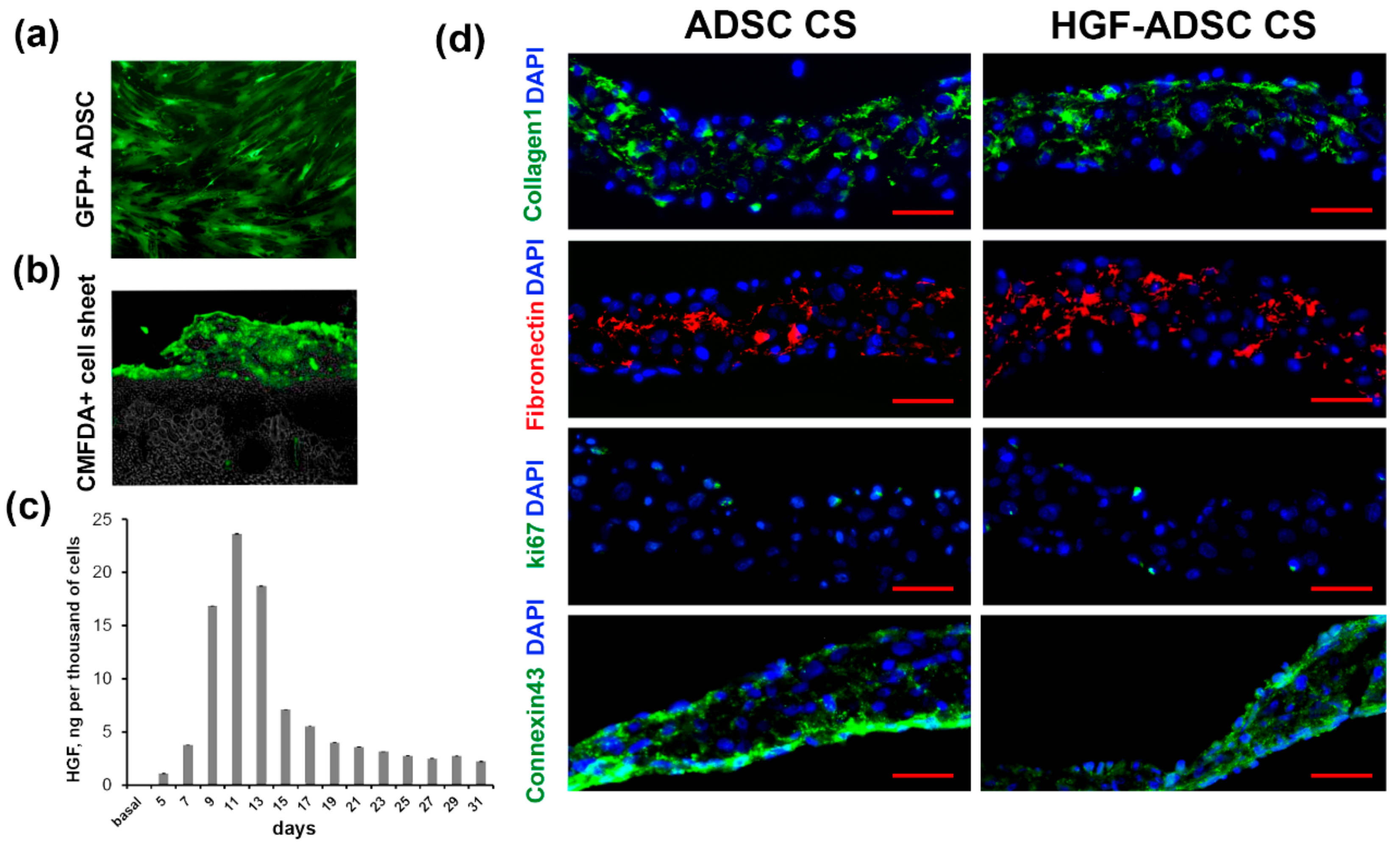
2.1. Characteristics of Cell Sheets Comprised of HGF-Overexpressing ADSC
2.2. Stimulating Effect of Conditioned Medium Collected from HGF-Producing ADSC on Neurite Outgrowth and Glial Cells Migration In Vitro
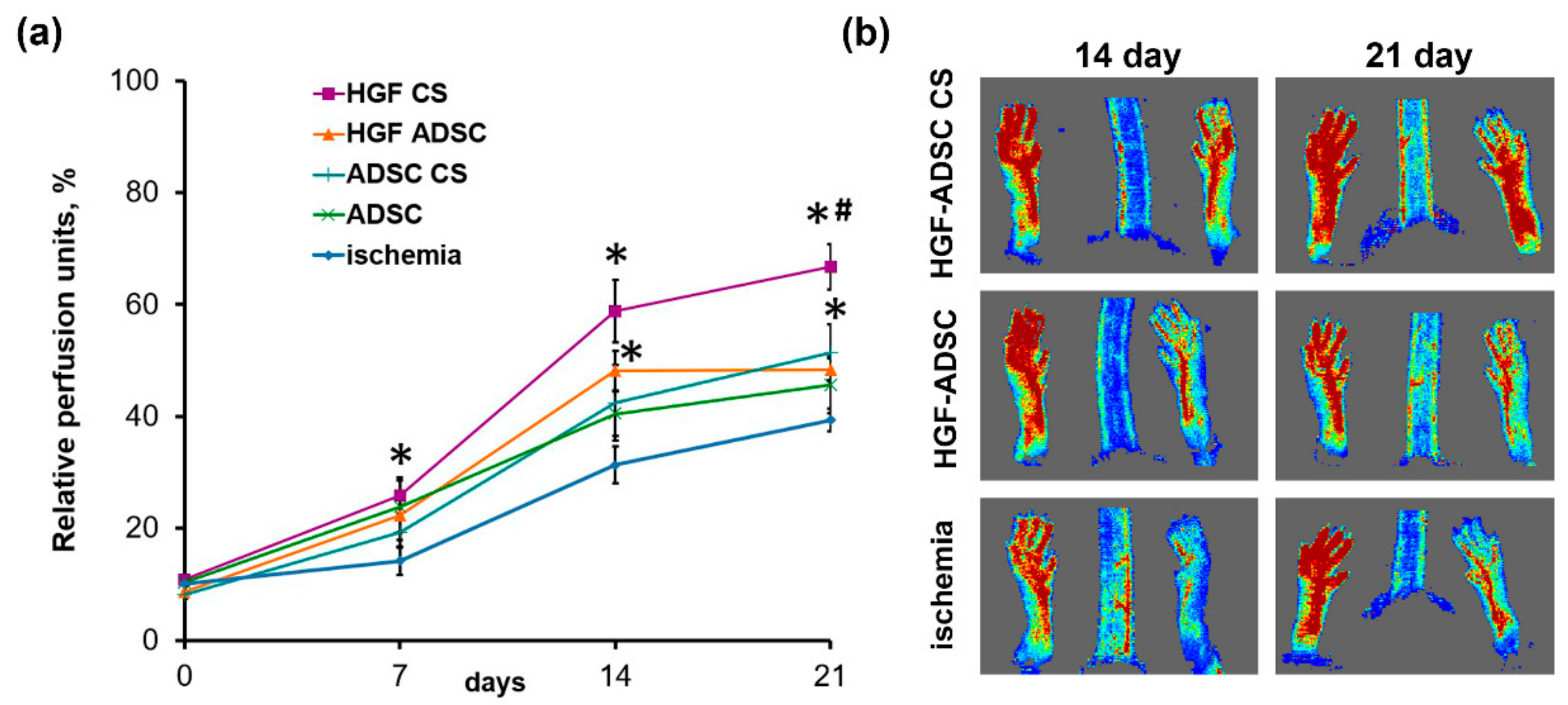
2.3. Blood Flow Recovery Following HGF-Producing ADSC CS Transplantation into Ischemic Limb
2.4. Increased Vascularization of Ischemic Skeletal Muscle after HGF-ADSC CS Transplantation
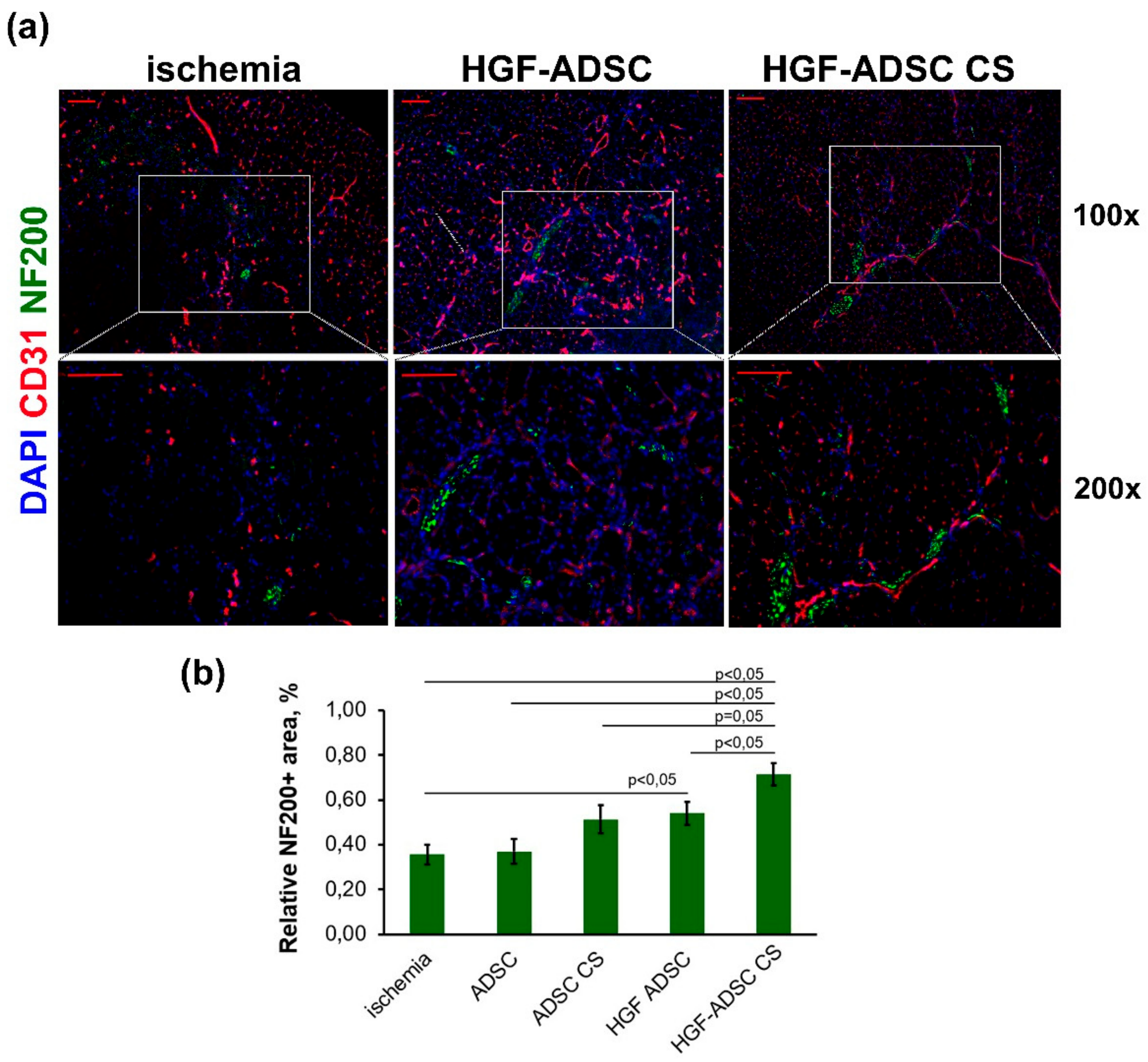
2.5. Neuroprotection in Ischemic Skeletal Muscle Following HGF-Producing CS Transplantation Overexpression
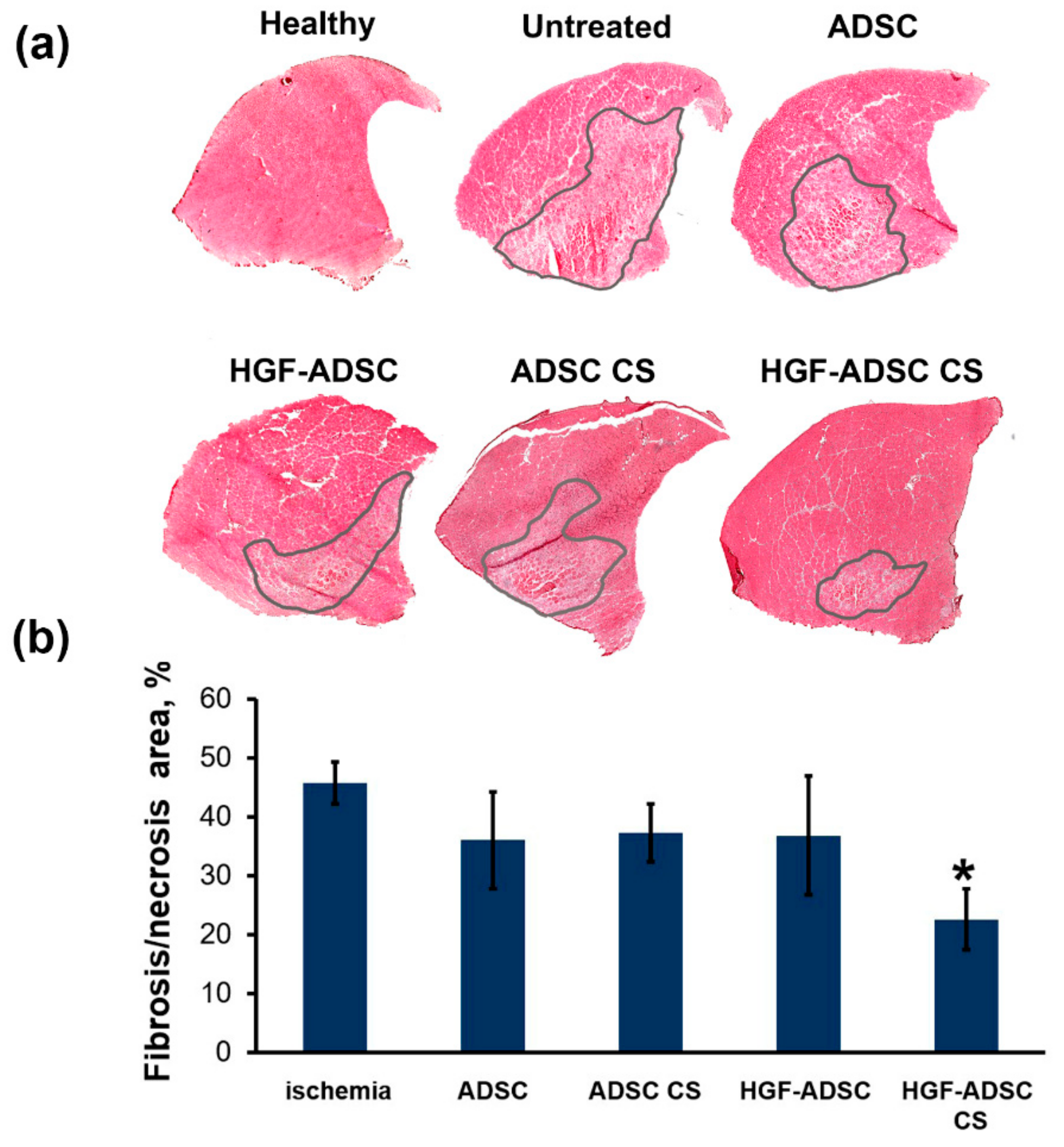
2.6. Reduction of Necrosis and Anti-Fibrotic Effect after ADSC Transplantation to Mice with Limb Ischemia
3. Discussion
- (1)
- Angiogenesis and vascularization of tissue
- (2)
- Neural terminals and innervation
- (3)
- Immune response and control of excessive inflammation
- (4)
- Reduction of fibrosis and necrosis span
4. Materials and Methods
4.1. Cell Cultures
4.2. DNA Constructs, Viral Vectors and Cell Transduction
4.3. Cell Sheets
4.4. Animals
4.5. Dorsal Root Ganglion Explants
4.6. Mouse Hind Limb Ischemia Model and Cell Transplantation
4.7. Laser Doppler Perfusion Measurement
4.8. Histological Analysis
4.9. Statistical Analysis
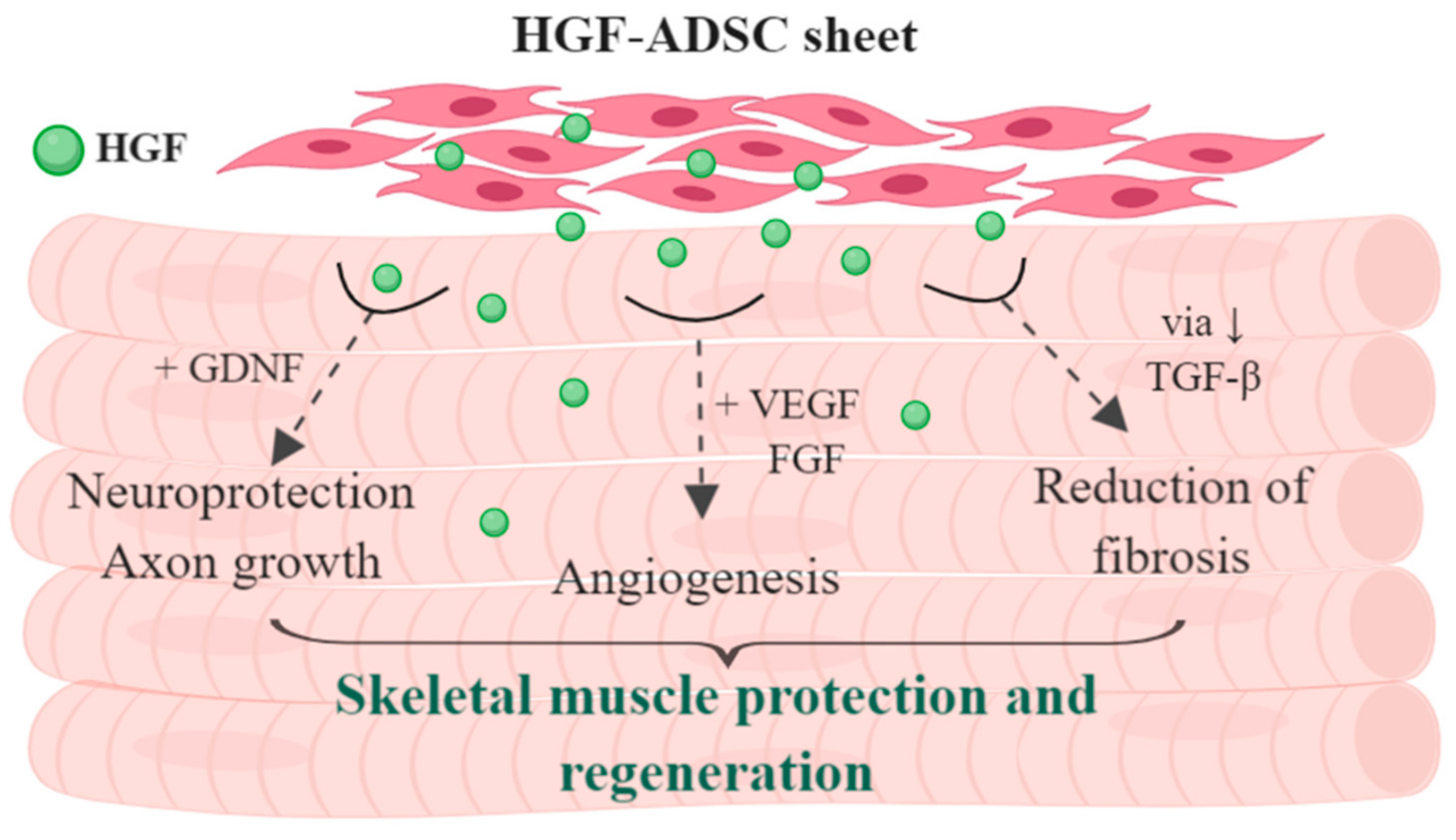
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Makarevich, P.I.; Parfyonova, Y.V. Therapeutic Angiogenesis: Foundations and Practical Application. In Physiologic and Pathologic Angiogenesis—Signaling Mechanisms and Targeted Therapy; Simionescu, D., Simionescu, A., Eds.; IntechOpean: London, UK, 2017; pp. 343–364. [Google Scholar]
- Parikh, P.P.; Liu, Z.-J.; Velazquez, O.C. A Molecular and Clinical Review of Stem Cell Therapy in Critical Limb Ischemia. Stem Cells Int. 2017, 2017, 3750829. [Google Scholar] [CrossRef] [PubMed]
- Ylitalo, K.R.; Sowers, M.; Heeringa, S. Peripheral vascular disease and peripheral neuropathy in individuals with cardiometabolic clustering and obesity: National Health and Nutrition Examination Survey 2001-2004. Diabetes Care 2011, 34, 1642–1647. [Google Scholar] [CrossRef] [PubMed]
- Galipeau, J.; Sensebe, L. Mesenchymal Stromal Cells: Clinical Challenges and Therapeutic Opportunities. Cell Stem Cell 2018, 22, 824–833. [Google Scholar] [CrossRef] [PubMed]
- Rehman, J.; Traktuev, D.; Li, J.; Merfeld-Clauss, S.; Temm-Grove, C.J.; Bovenkerk, J.E.; Pell, C.L.; Johnstone, B.H.; Considine, R.V.; March, K.L. Secretion of Angiogenic and Antiapoptotic Factors by Human Adipose Stromal Cells. Circulation 2004, 109, 1292–1298. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Du, Z.; Zhao, L.; Feng, D.; Wei, G.; He, Y.; Tan, J.; Lee, W.-H.; Hampel, H.; Dodel, R.; et al. IFATS collection: The conditioned media of adipose stromal cells protect against hypoxia-ischemia-induced brain damage in neonatal rats. Stem Cells 2009, 27, 478–488. [Google Scholar] [CrossRef]
- Rubina, K.; Kalinina, N.; Efimenko, A.; Lopatina, T.; Melikhova, V.; Tsokolaeva, Z.; Sysoeva, V.; Tkachuk, V.; Parfyonova, Y. Adipose stromal cells stimulate angiogenesis via promoting progenitor cell differentiation, secretion of angiogenic factors, and enhancing vessel maturation. Tissue Eng. Part A 2009, 15, 2039–2050. [Google Scholar] [CrossRef] [PubMed]
- Merfeld-Clauss, S.; Gollahalli, N.; March, K.L.; Traktuev, D.O. Adipose tissue progenitor cells directly interact with endothelial cells to induce vascular network formation. Tissue Eng. Part A 2010, 16, 2953–2966. [Google Scholar] [CrossRef] [PubMed]
- Hutton, D.L.; Logsdon, E.A.; Moore, E.M.; Mac Gabhann, F.; Gimble, J.M.; Grayson, W.L. Vascular morphogenesis of adipose-derived stem cells is mediated by heterotypic cell-cell interactions. Tissue Eng. Part A 2012, 18, 1729–1740. [Google Scholar] [CrossRef] [PubMed]
- Traktuev, D.O.; March, K.L.; Tkachuk, V.A.; Parfenova, E.V. Adipose tissue stromal cells—Multipotent cells with therapeutic potential for stimulation of angiogenesis in tissue ischemia. Kardiologiia 2006, 46, 53–63. [Google Scholar]
- Makarevich, P.I.; Boldyreva, M.A.; Gluhanyuk, E.V.; Efimenko, A.Y.; Dergilev, K.V.; Shevchenko, E.K.; Sharonov, G.V.; Gallinger, J.O.; Rodina, P.A.; Sarkisyan, S.S.; et al. Enhanced angiogenesis in ischemic skeletal muscle after transplantation of cell sheets from baculovirus-transduced adipose-derived stromal cells expressing VEGF165. Stem Cell Res. Ther. 2015, 6, 204. [Google Scholar] [CrossRef]
- Makarevich, P.; Boldyreva, M.; Dergilev, K.V.; Gluhanyuk, E.; Gallinger, J.O.; Efimenko, A.; Tkachuk, V.A.; Parfyonova, Y.V. Transplantation of cell sheets from adipose-derived mesenchymal stromal cells effectively induces angiogenesis in ischemic skeletal muscle. Genes Cells 2015, 10, 68–77. [Google Scholar]
- Shevchenko, E.K.; Makarevich, P.I.; Tsokolaeva, Z.I.; Boldyreva, M.A.; Sysoeva, V.Y.; Tkachuk, V.A.; Parfyonova, Y.V. Transplantation of modified human adipose derived stromal cells expressing VEGF165 results in more efficient angiogenic response in ischemic skeletal muscle. J. Transl. Med. 2013, 11, 138. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Mizuno, S. The discovery of Hepatocyte Growth Factor (HGF) and its significance for cell biology, life sciences and clinical medicine. Proc. Jpn Acad. Ser. B Phys. Bol. Sci. 2010, 86, 588–610. [Google Scholar] [CrossRef]
- Mungunsukh, O.; McCart, E.A.; Day, R.M. Hepatocyte Growth Factor Isoforms in Tissue Repair, Cancer, and Fibrotic Remodeling. Biomedicines 2014, 2, 301–326. [Google Scholar] [CrossRef] [PubMed]
- Imamura, R.; Matsumoto, K. Hepatocyte growth factor in physiology and infectious diseases. Cytokine 2017, 98, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Makarevich, P.I.; Dergilev, K.V.; Tsokolaeva, Z.I.; Boldyreva, M.A.; Shevchenko, E.K.; Gluhanyuk, E.V.; Gallinger, J.O.; Menshikov, M.Y.; Parfyonova, Y.V. Angiogenic and pleiotropic effects of VEGF165 and HGF combined gene therapy in a rat model of myocardial infarction. PLoS ONE 2018, 13, 1–25. [Google Scholar] [CrossRef]
- Boldyreva, M.A.; Bondar, I.V.; Stafeev, I.S.; Makarevich, P.I.; Beloglazova, I.B.; Zubkova, E.S.; Shevchenko, E.K.; Molokotina, Y.D.; Karagyaur, M.N.; Ratner, E.I.; et al. Plasmid-based gene therapy with hepatocyte growth factor stimulates peripheral nerve regeneration after traumatic injury. Biomed. Pharmacother. 2018, 101, 682–690. [Google Scholar] [CrossRef]
- Baldari, S.; Di Rocco, G.; Piccoli, M.; Pozzobon, M.; Muraca, M.; Toietta, G. Challenges and Strategies for Improving the Regenerative Effects of Mesenchymal Stromal Cell-Based Therapies. Int. J. Mol. Sci. 2017, 18, 2087. [Google Scholar] [CrossRef]
- Frese, L.; Dijkman, P.E.; Hoerstrup, S.P. Adipose Tissue-Derived Stem Cells in Regenerative Medicine. Transfus. Med. Hemother. 2016, 43, 268–274. [Google Scholar] [CrossRef]
- Qomi, R.T.; Sheykhhasan, M. Adipose-derived stromal cell in regenerative medicine: A review. World J. Stem Cells 2017, 9, 107. [Google Scholar] [CrossRef]
- Inampudi, C.; Akintoye, E.; Ando, T.; Briasoulis, A. Angiogenesis in peripheral arterial disease. Curr. Opin. Pharmacol. 2018, 39, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Abdul Wahid, S.F.; Ismail, N.A.; Wan Jamaludin, W.F.; Muhamad, N.A.; Abdul Hamid, M.K.A.; Harunarashid, H.; Lai, N.M. Autologous cells derived from different sources and administered using different regimens for “no-option” critical lower limb ischaemia patients. Cochrane Database Syst. Rev. 2018, 8, CD010747. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, Y.; Miyagawa, S.; Toda, K.; Saito, A.; Sakata, Y.; Sawa, Y. Myocardial regenerative therapy using a scaffold-free skeletal-muscle-derived cell sheet in patients with dilated cardiomyopathy even under a left ventricular assist device: A safety and feasibility study. Surg. Today 2018, 48, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kim, Y.; Park, J.; Hwang, N.S.; Lee, Y.K.; Hwang, Y. Recent Advances in Engineered Stem Cell-Derived Cell Sheets for Tissue Regeneration. Polymers 2019, 11, E209. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Yamato, M.; Mitani, G.; Takagaki, T.; Hamahashi, K.; Nakamura, Y.; Ishihara, M.; Matoba, R.; Kobayashi, H.; Okano, T.; et al. Combined surgery and chondrocyte cell-sheet transplantation improves clinical and structural outcomes in knee osteoarthritis. NPJ Regen. Med. 2019, 4, 4. [Google Scholar] [CrossRef] [PubMed]
- Hsu, M.-N.; Liao, H.-T.; Li, K.-C.; Chen, H.-H.; Yen, T.-C.; Makarevich, P.; Parfyonova, Y.; Hu, Y.-C. Adipose-derived stem cell sheets functionalized by hybrid baculovirus for prolonged GDNF expression and improved nerve regeneration. Biomaterials 2017, 140, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Yeh, T.-S.; Fang, Y.-H.D.; Lu, C.-H.; Chiu, S.-C.; Yeh, C.-L.; Yen, T.-C.; Parfyonova, Y.; Hu, Y.-C. Baculovirus-transduced, VEGF-expressing adipose-derived stem cell sheet for the treatment of myocardium infarction. Biomaterials 2014, 35, 174–184. [Google Scholar] [CrossRef] [PubMed]
- Hobo, K.; Shimizu, T.; Sekine, H.; Shin’oka, T.; Okano, T.; Kurosawa, H. Therapeutic angiogenesis using tissue engineered human smooth muscle cell sheets. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 637–643. [Google Scholar] [CrossRef]
- Kaga, T.; Kawano, H.; Sakaguchi, M.; Nakazawa, T.; Taniyama, Y.; Morishita, R. Hepatocyte growth factor stimulated angiogenesis without inflammation: Differential actions between hepatocyte growth factor, vascular endothelial growth factor and basic fibroblast growth factor. Vascul. Pharmacol. 2012, 57, 3–9. [Google Scholar] [CrossRef]
- Miller, K.J.; Thaloor, D.; Matteson, S.; Pavlath, G.K. Hepatocyte growth factor affects satellite cell activation and differentiation in regenerating skeletal muscle. Am. J. Physiol. Cell Physiol. 2000, 278, C174–181. [Google Scholar] [CrossRef]
- Fu, X.; Wang, H.; Hu, P. Stem cell activation in skeletal muscle regeneration. Cell. Mol. Life Sci. 2015, 72, 1663–1677. [Google Scholar] [CrossRef] [PubMed]
- Duan, H.-F.; Wu, C.-T.; Wu, D.-L.; Lu, Y.; Liu, H.-J.; Ha, X.-Q.; Zhang, Q.-W.; Wang, H.; Jia, X.-X.; Wang, L.-S. Treatment of myocardial ischemia with bone marrow-derived mesenchymal stem cells overexpressing hepatocyte growth factor. Mol. Ther. 2003, 8, 467–474. [Google Scholar] [CrossRef]
- Yang, Z.-J.; Ma, D.-C.; Wang, W.; Xu, S.-L.; Zhang, Y.-Q.; Chen, B.; Zhou, F.; Zhu, T.-B.; Wang, L.-S.; Xu, Z.-Q.; et al. Experimental study of bone marrow-derived mesenchymal stem cells combined with hepatocyte growth factor transplantation via noninfarct-relative artery in acute myocardial infarction. Gene Ther. 2006, 13, 1564–1568. [Google Scholar] [CrossRef][Green Version]
- Su, G.-H.; Sun, Y.-F.; Lu, Y.-X.; Shuai, X.-X.; Liao, Y.-H.; Liu, Q.-Y.; Han, J.; Luo, P. Hepatocyte growth factor gene-modified bone marrow-derived mesenchymal stem cells transplantation promotes angiogenesis in a rat model of hindlimb ischemia. J. Huazhong Univ. Sci. Technol. Med. Sci. 2013, 33, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.-Z.; Nonoguchi, N.; Ikeda, N.; Watanabe, T.; Furutama, D.; Miyazawa, D.; Funakoshi, H.; Kajimoto, Y.; Nakamura, T.; Dezawa, M.; et al. Novel therapeutic strategy for stroke in rats by bone marrow stromal cells and ex vivo HGF gene transfer with HSV-1 vector. J. Cereb. Blood Flow Metab. 2006, 26, 1176–1188. [Google Scholar] [CrossRef] [PubMed]
- Makarevich, P.; Tsokolaeva, Z.; Shevelev, A.; Rybalkin, I.; Shevchenko, E.; Beloglazova, I.; Vlasik, T.; Tkachuk, V.; Parfyonova, Y. Combined transfer of human VEGF165 and HGF genes renders potent angiogenic effect in ischemic skeletal muscle. PLoS ONE 2012, 7, e38776. [Google Scholar] [CrossRef] [PubMed]
- Dai, C.; Liu, Y. Hepatocyte growth factor antagonizes the profibrotic action of TGF-beta1 in mesangial cells by stabilizing Smad transcriptional corepressor TGIF. J. Am. Soc. Nephrol. 2004, 15, 1402–1412. [Google Scholar] [CrossRef]
- Yang, J.; Liu, Y. Blockage of tubular epithelial to myofibroblast transition by hepatocyte growth factor prevents renal interstitial fibrosis. J. Am. Soc. Nephrol. 2002, 13, 96–107. [Google Scholar]
- Yu, Y.; Lu, L.; Qian, X.; Chen, N.; Yao, A.; Pu, L.; Zhang, F.; Li, X.; Kong, L.; Sun, B.; et al. Antifibrotic effect of hepatocyte growth factor-expressing mesenchymal stem cells in small-for-size liver transplant rats. Stem Cells Dev. 2010, 19, 903–914. [Google Scholar] [CrossRef]
- Nakamura, T.; Mizuno, S.; Matsumoto, K.; Sawa, Y.; Matsuda, H.; Nakamura, T. Myocardial protection from ischemia/reperfusion injury by endogenous and exogenous HGF. J. Clin. Invest. 2000, 106, 1511–1519. [Google Scholar] [CrossRef]
- Morishita, R.; Nakamura, S.; Hayashi, S.; Taniyama, Y.; Moriguchi, A.; Nagano, T.; Taiji, M.; Noguchi, H.; Takeshita, S.; Matsumoto, K.; et al. Therapeutic angiogenesis induced by human recombinant hepatocyte growth factor in rabbit hind limb ischemia model as cytokine supplement therapy. Hypertens 1999, 33, 1379–1384. [Google Scholar] [CrossRef]
- Eichmann, A.; Brunet, I. Arterial innervation in development and disease. Sci. Transl. Med. 2014, 6, 252ps9. [Google Scholar] [CrossRef] [PubMed]
- Chalothorn, D.; Zhang, H.; Clayton, J.A.; Thomas, S.A.; Faber, J.E. Catecholamines augment collateral vessel growth and angiogenesis in hindlimb ischemia. Am. J. Physiol. Heart Circ. Physiol. 2005, 289, H947–959. [Google Scholar] [CrossRef] [PubMed]
- Recalde, A.; Richart, A.; Guerin, C.; Cochain, C.; Zouggari, Y.; Yin, K.H.W.; Vilar, J.; Drouet, I.; Levy, B.; Varoquaux, O.; et al. Sympathetic nervous system regulates bone marrow-derived cell egress through endothelial nitric oxide synthase activation: Role in postischemic tissue remodeling. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 643–653. [Google Scholar] [CrossRef] [PubMed]
- Katayama, Y.; Battista, M.; Kao, W.-M.; Hidalgo, A.; Peired, A.J.; Thomas, S.A.; Frenette, P.S. Signals from the sympathetic nervous system regulate hematopoietic stem cell egress from bone marrow. Cell 2006, 124, 407–421. [Google Scholar] [CrossRef] [PubMed]
- Henderson, C.E.; Yamamoto, Y.; Livet, J.; Arce, V.; Garces, A.; DeLapeyriere, O. Role of neurotrophic factors in motoneuron development. J. Physiol. Paris 1998, 92, 279–281. [Google Scholar] [CrossRef]
- Iida, H.; Schmeichel, A.M.; Wang, Y.; Schmelzer, J.D.; Low, P.A. Schwann cell is a target in ischemia-reperfusion injury to peripheral nerve. Muscle Nerve 2004, 30, 761–766. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.; Ju, R.; Mao, M. Therapeutic potential of hepatocyte growth factor against cerebral ischemia (Review). Exp. Ther. Med. 2015, 9, 283–288. [Google Scholar] [CrossRef]
- Carmeliet, P.; Tessier-Lavigne, M. Common mechanisms of nerve and blood vessel wiring. Nature 2005, 436, 193–200. [Google Scholar] [CrossRef]
- Molokotina, Y.D.; Boldyreva, M.A.; Stafeev, I.S.; Semina, E.V.; Shevchenko, E.K.; Zubkova, E.S.; Beloglazova, I.B.; Parfyonova, Y.V. The combined action of GDNF and HGF upregulates axonal growth via increase in Erk1/2 phosphorylation. Bull. Exp. Biol. Med. 2019, (in press).
- Hou, S.; Tian, W.; Xu, Q.; Cui, F.; Zhang, J.; Lu, Q.; Zhao, C. The enhancement of cell adherence and inducement of neurite outgrowth of dorsal root ganglia co-cultured with hyaluronic acid hydrogels modified with Nogo-66 receptor antagonist in vitro. Neuroscience 2006, 137, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Wang, Y.-N.; Cui, X.-L.; Fang, S.-Y.; Ge, J.-Y.; Sun, Y.; Liu, Z.-H. The role and mechanism of action of activin A in neurite outgrowth of chicken embryonic dorsal root ganglia. J. Cell Sci. 2012, 125, 1500–1507. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Li, Z. The effects of target skeletal muscle cells on dorsal root ganglion neuronal outgrowth and migration in vitro. PLoS ONE 2013, 8, e52849. [Google Scholar] [CrossRef] [PubMed]
- Boldyreva, M.; Makarevich, P.; Rafieva, L.M.; Beloglazova, I.; Dergilev, K.V.; Kostrov, S.V.; Parfyonova, Y.V. Delivery of nerve growth factor (NGF) gene via recombinant plasmid vector induces angiogenesis in murine ischemic hind limb. Genes Cells 2014, 9, 81–87. [Google Scholar]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boldyreva, M.A.; Shevchenko, E.K.; Molokotina, Y.D.; Makarevich, P.I.; Beloglazova, I.B.; Zubkova, E.S.; Dergilev, K.V.; Tsokolaeva, Z.I.; Penkov, D.; Hsu, M.-N.; et al. Transplantation of Adipose Stromal Cell Sheet Producing Hepatocyte Growth Factor Induces Pleiotropic Effect in Ischemic Skeletal Muscle. Int. J. Mol. Sci. 2019, 20, 3088. https://doi.org/10.3390/ijms20123088
Boldyreva MA, Shevchenko EK, Molokotina YD, Makarevich PI, Beloglazova IB, Zubkova ES, Dergilev KV, Tsokolaeva ZI, Penkov D, Hsu M-N, et al. Transplantation of Adipose Stromal Cell Sheet Producing Hepatocyte Growth Factor Induces Pleiotropic Effect in Ischemic Skeletal Muscle. International Journal of Molecular Sciences. 2019; 20(12):3088. https://doi.org/10.3390/ijms20123088
Chicago/Turabian StyleBoldyreva, Maria A., Evgeny K. Shevchenko, Yuliya D. Molokotina, Pavel I. Makarevich, Irina B. Beloglazova, Ekaterina S. Zubkova, Konstantin V. Dergilev, Zoya I. Tsokolaeva, Dmitry Penkov, Mu-Nung Hsu, and et al. 2019. "Transplantation of Adipose Stromal Cell Sheet Producing Hepatocyte Growth Factor Induces Pleiotropic Effect in Ischemic Skeletal Muscle" International Journal of Molecular Sciences 20, no. 12: 3088. https://doi.org/10.3390/ijms20123088
APA StyleBoldyreva, M. A., Shevchenko, E. K., Molokotina, Y. D., Makarevich, P. I., Beloglazova, I. B., Zubkova, E. S., Dergilev, K. V., Tsokolaeva, Z. I., Penkov, D., Hsu, M.-N., Hu, Y.-C., & Parfyonova, Y. V. (2019). Transplantation of Adipose Stromal Cell Sheet Producing Hepatocyte Growth Factor Induces Pleiotropic Effect in Ischemic Skeletal Muscle. International Journal of Molecular Sciences, 20(12), 3088. https://doi.org/10.3390/ijms20123088








