Novel Bioactive and Antibacterial Acrylic Bone Cement Nanocomposites Modified with Graphene Oxide and Chitosan
Abstract
1. Introduction
2. Results and Discussion
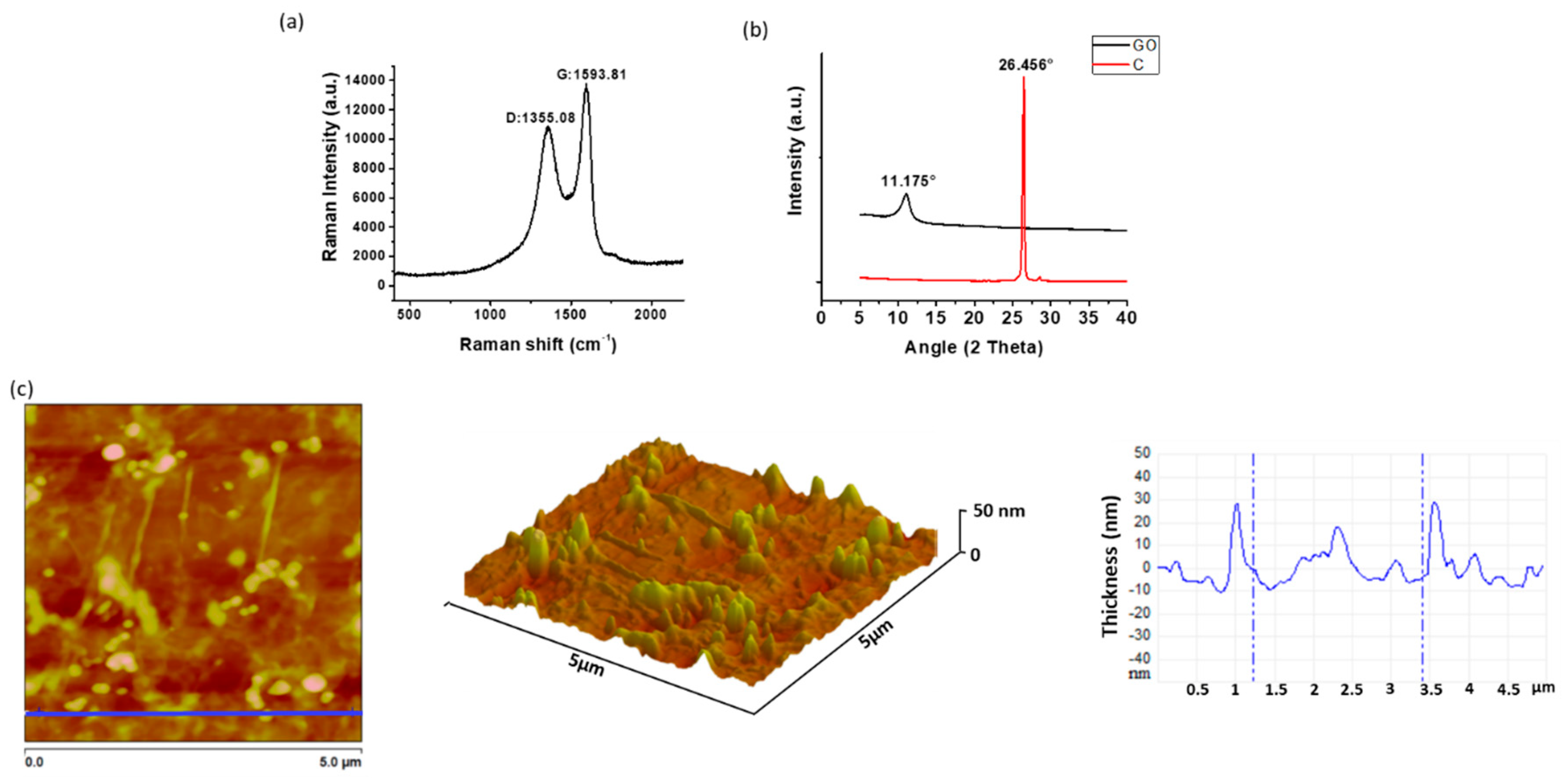
2.1. Characterization of Graphene Oxide
2.2. Characterization of ABC
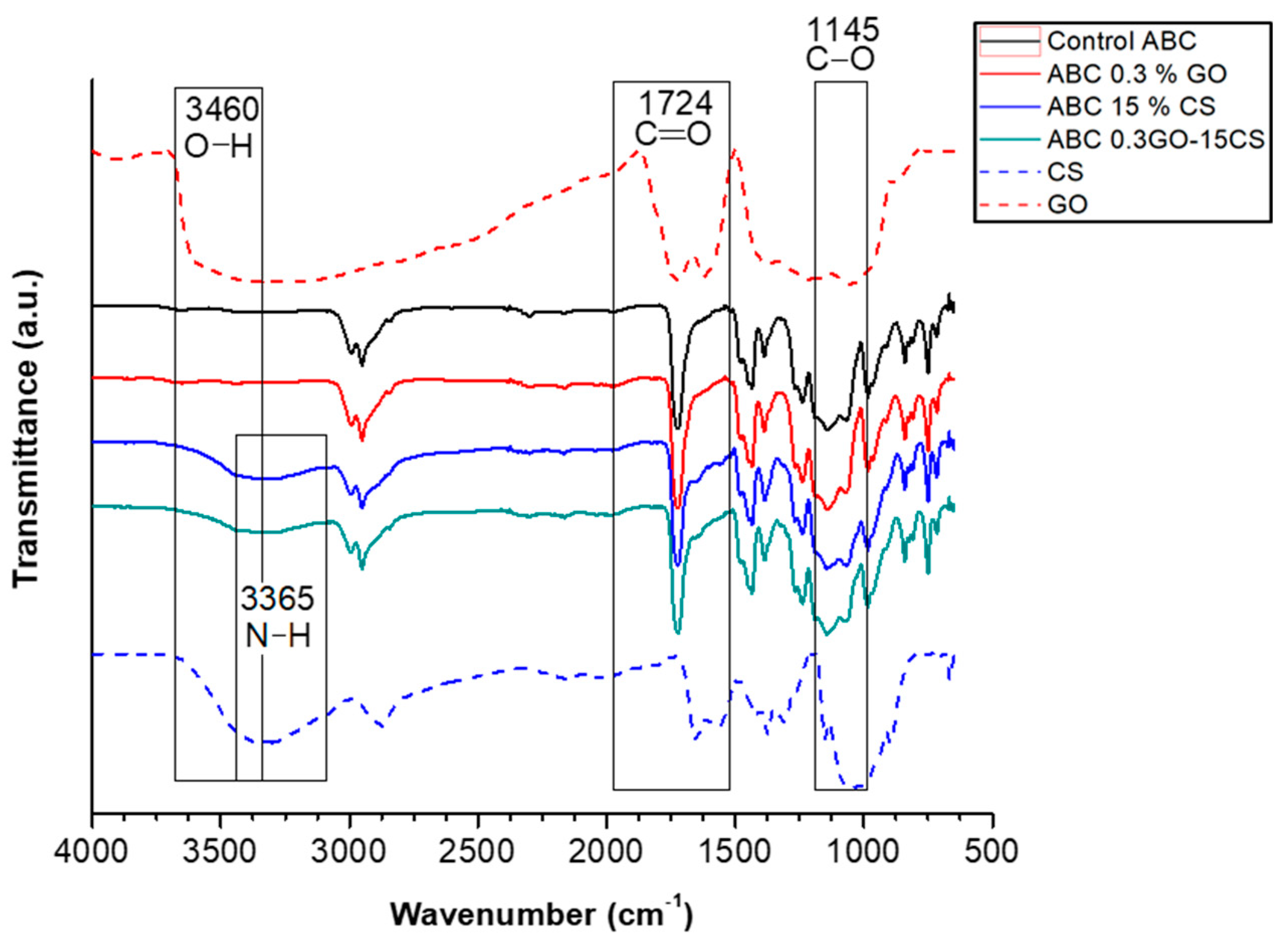
2.2.1. Chemical Characterization
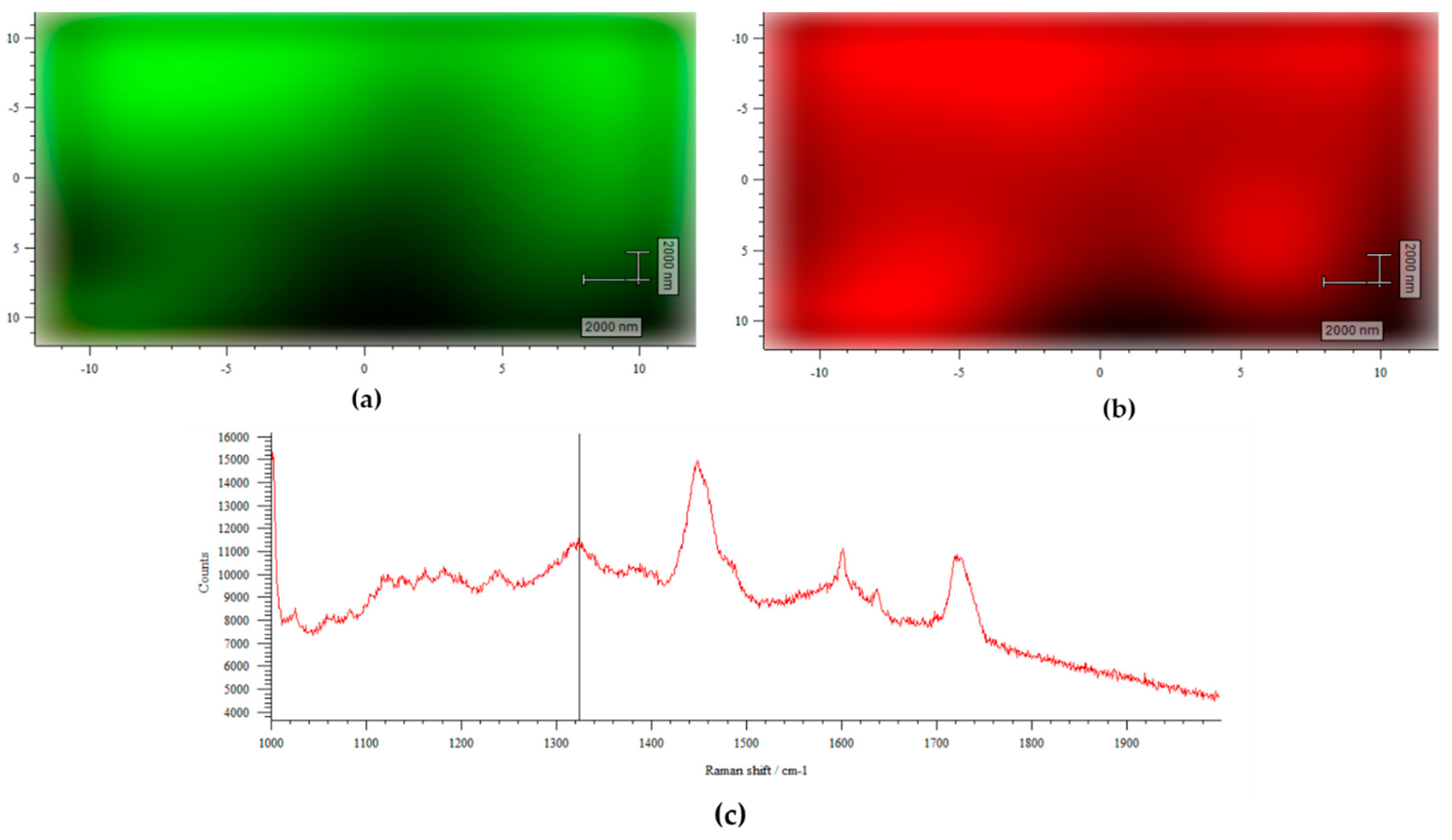
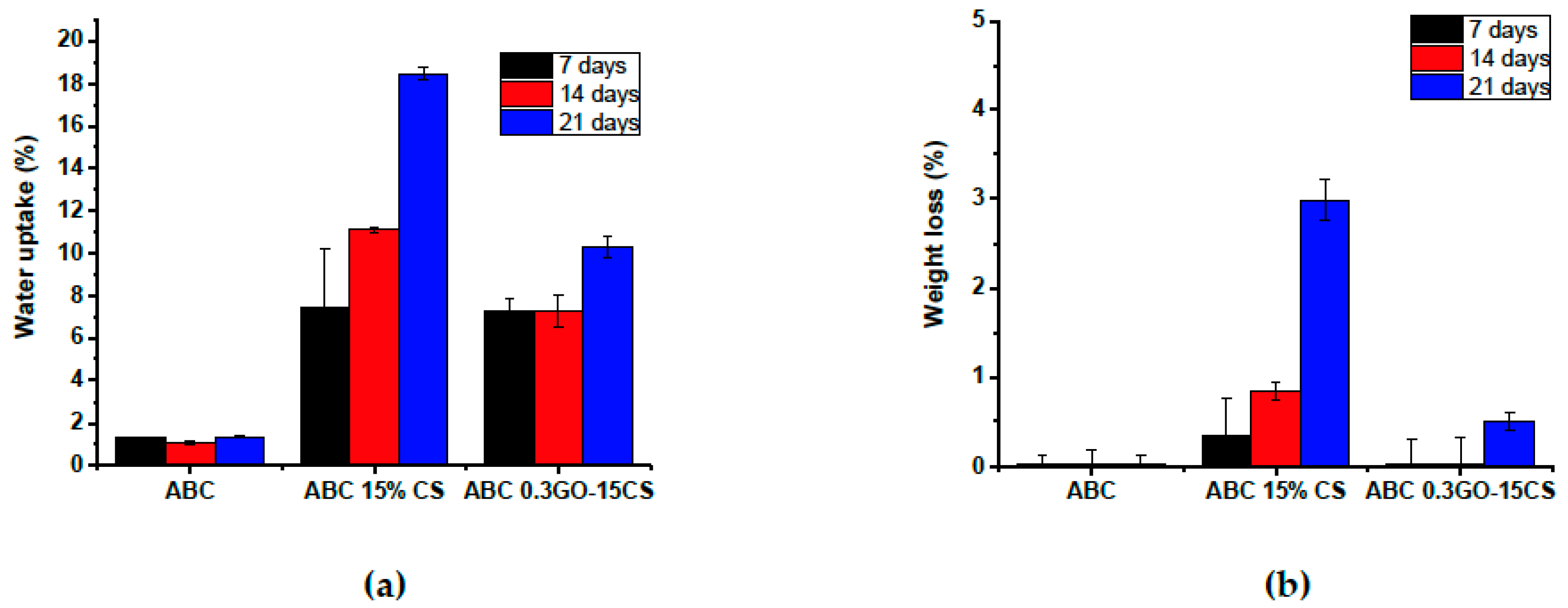
2.2.2. Physical Characterization
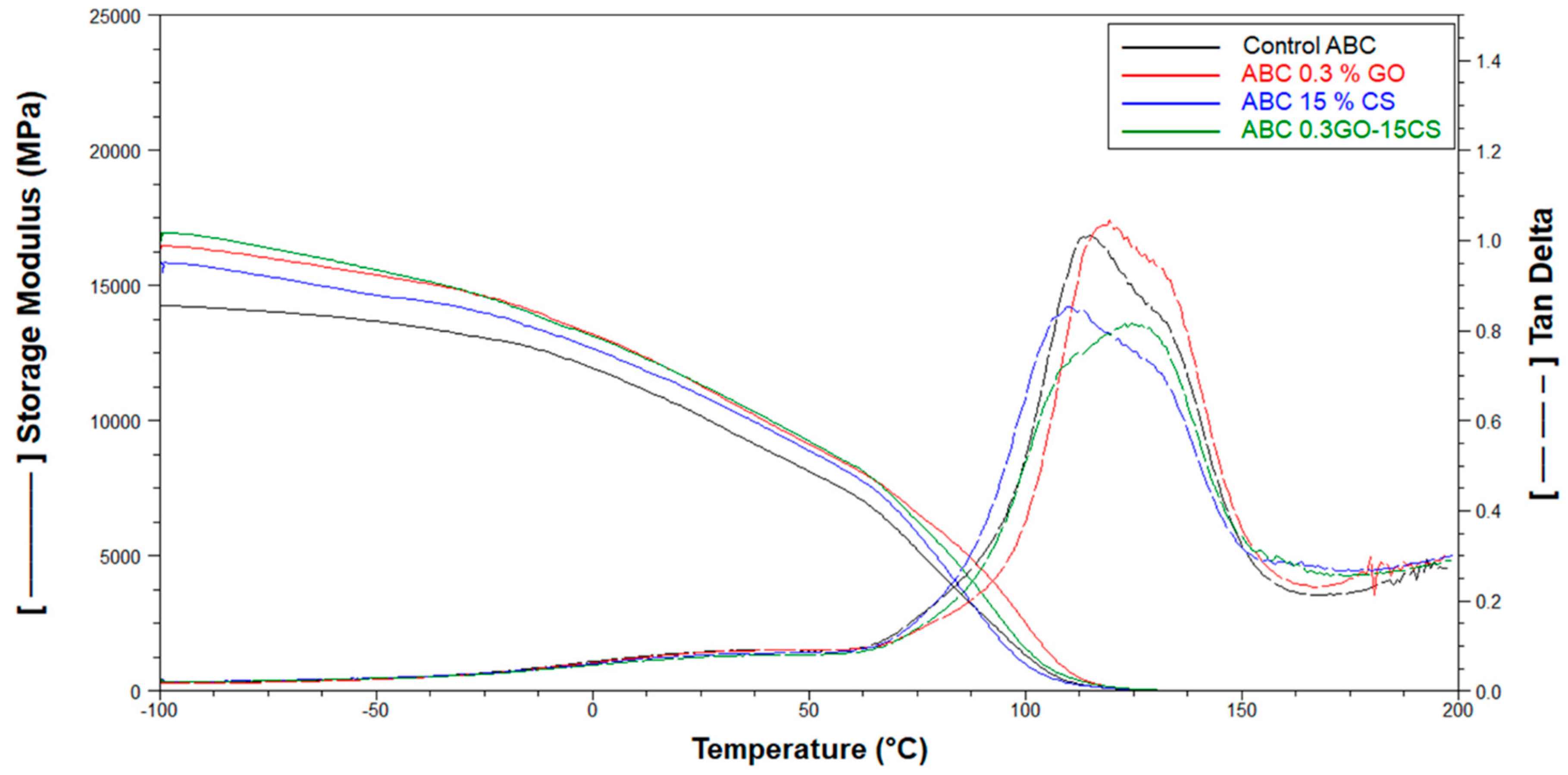
2.2.3. Thermal Characterization
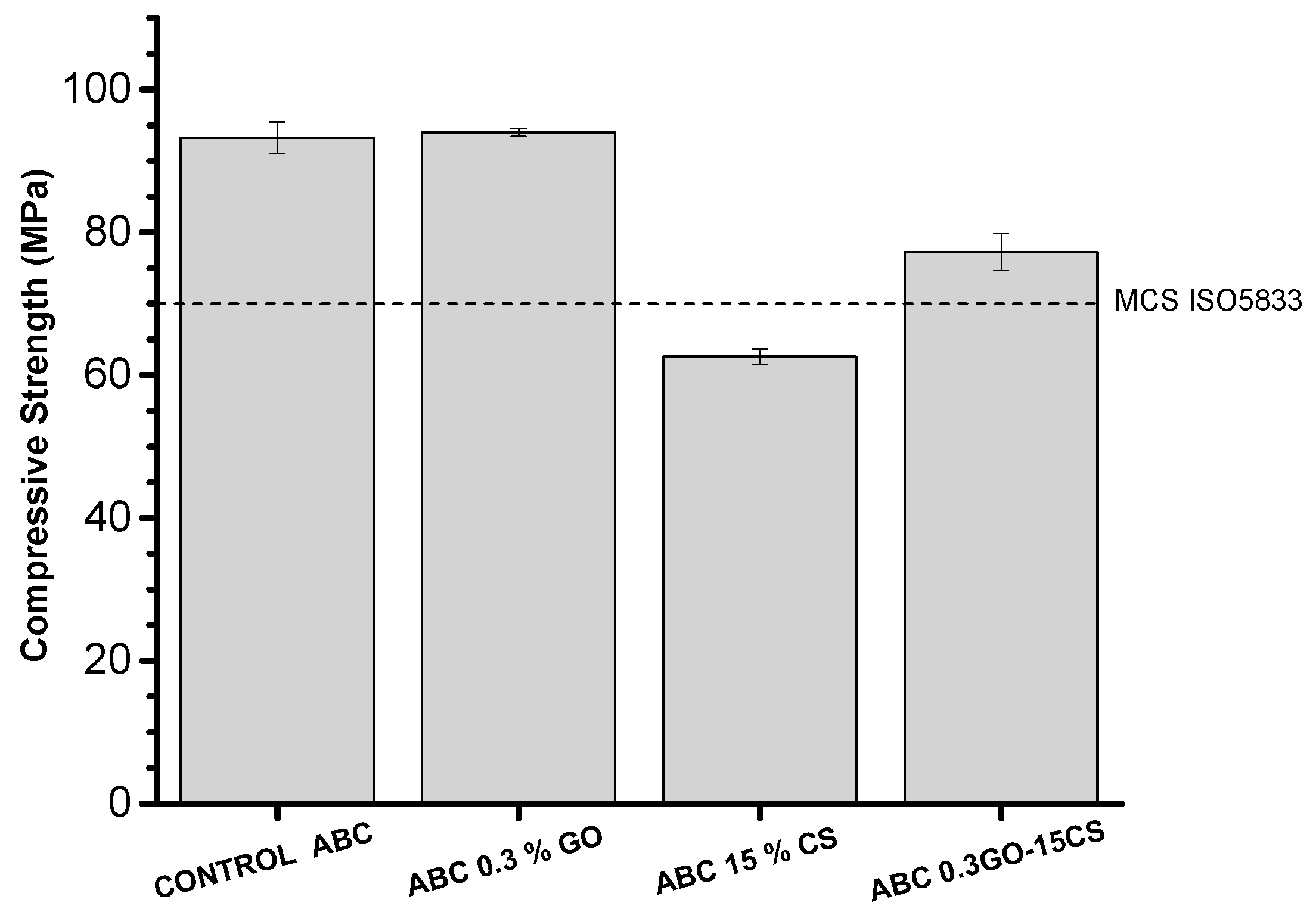
2.2.4. Mechanical Characterization
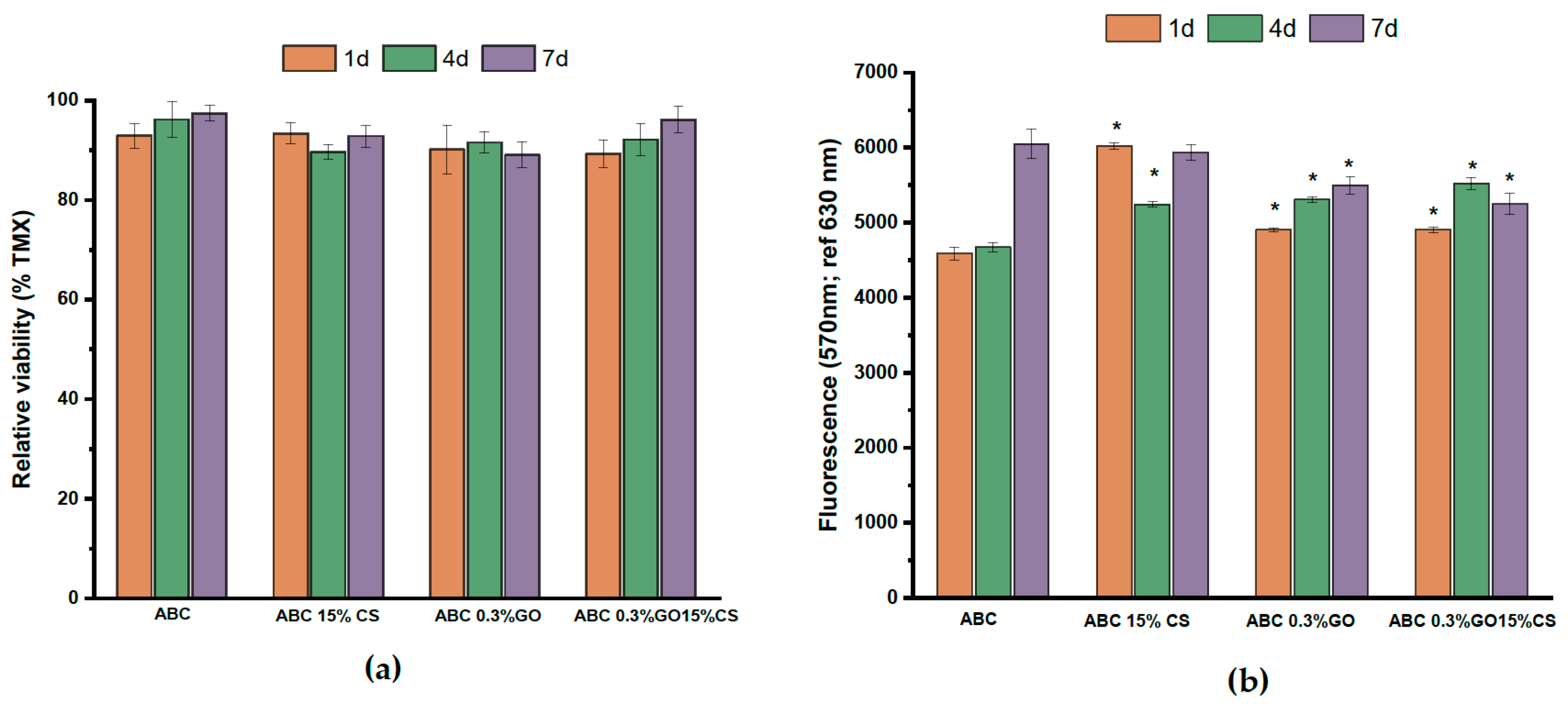
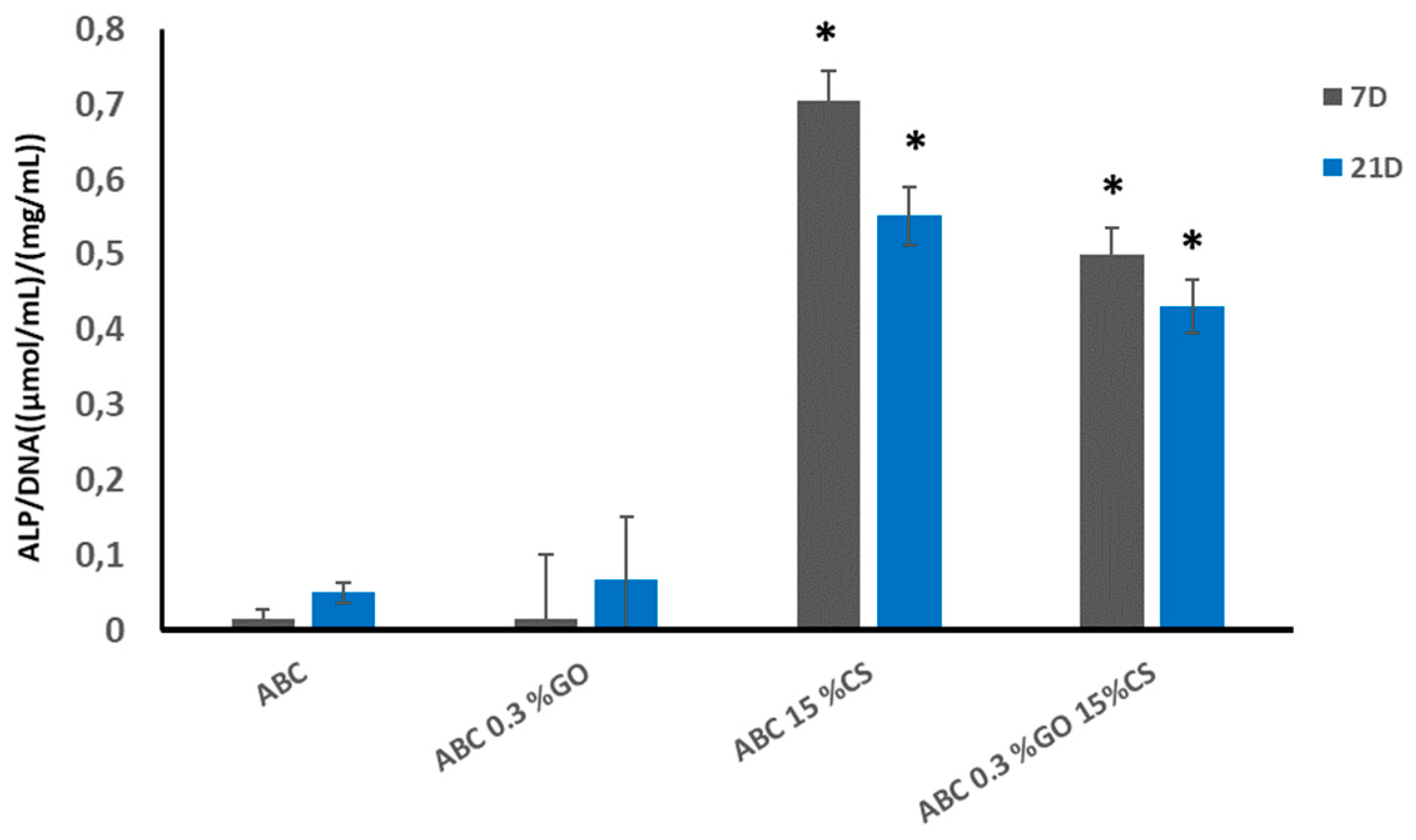
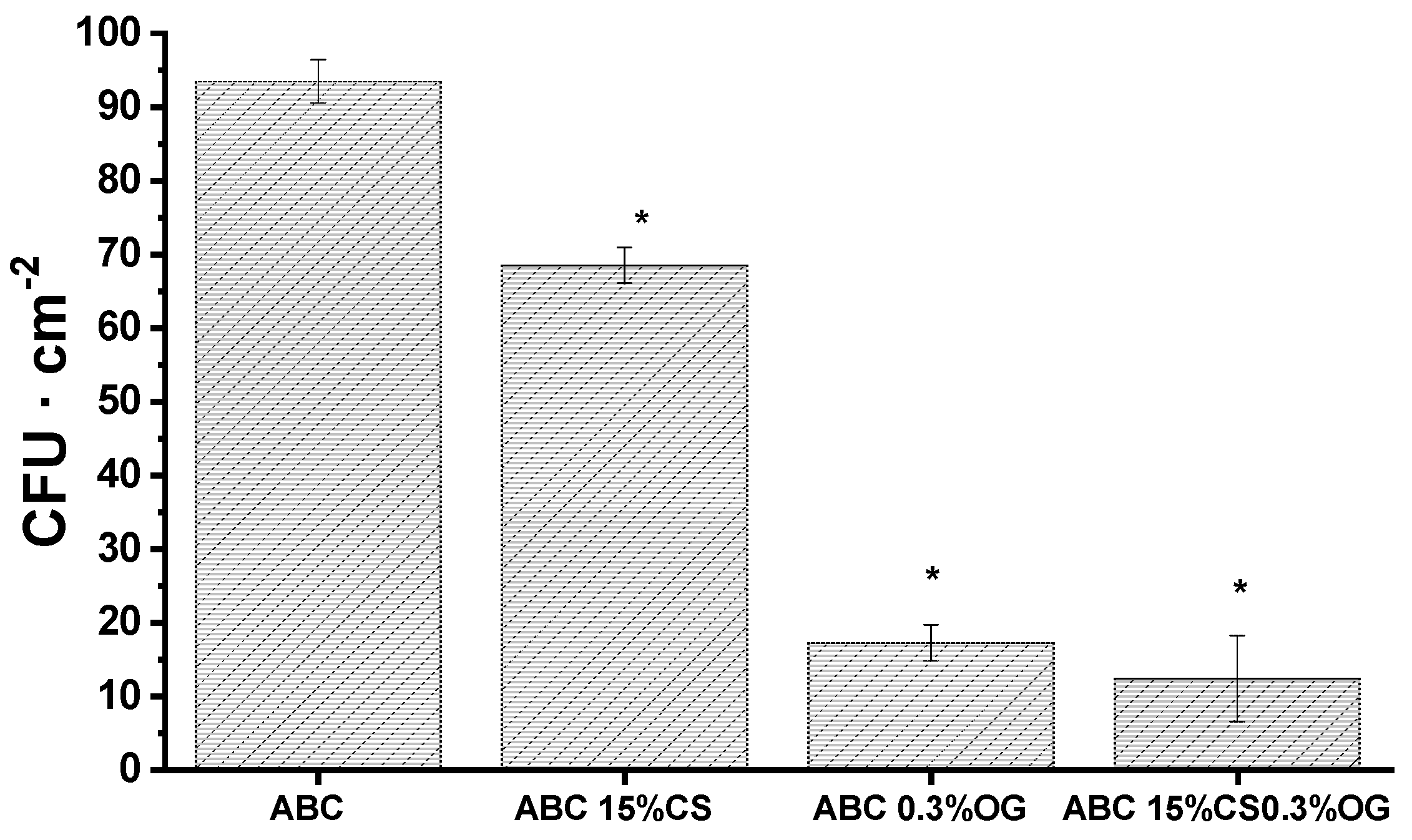
2.2.5. Biological Characterization
3. Materials and Methods
3.1. Materials
3.2. Synthesis of GO
3.3. Characterization of GO
3.4. Preparation of ABC
3.5. Characterization of ABC
3.5.1. Chemical Characterization
3.5.2. Physical Characterization
3.5.3. Thermal Characterization
3.5.4. Mechanical Characterization
3.5.5. Biological Characterization
MTT Assay
Alamar Blue Assay
ALP Assay
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Deb, S.; Koller, G. Chapter 8. Acrylic bone cement: Genesis and evolution. In Orthopaedic Bone Cement; Deb, S., Ed.; Woodhead Publishing Limited: Cambridge, UK, 2008; pp. 167–182. [Google Scholar]
- González, C.M.; Obando, C.M.; Jaramillo, C.A.; de Jesús Peña, A.; Paz, A.; Acosta, J.L. Resultados clínicos de pacientes intervenidos con reemplazos articulares de cadera y rodilla en la Clínica Soma, año 2010. Rev. Colomb. Ortop. Traumatol. 2014, 28, 4–8. [Google Scholar] [CrossRef]
- Ghavimi, S.A.A.; Lungren, E.S.; Faulkner, T.J.; Josselet, M.A.; Wu, Y.; Sun, Y.; Ulery, B.D. Inductive co-crosslinking of cellulose nanocrystal/chitosan hydrogels for the treatment of vertebral compression fractures. Int. J. Biol. Macromol. 2019, 130, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Hendriks, J.G.E.; van Horn, J.R.; van der Mei, H.C.; Busscher, H.J. Backgrounds of antibiotic-loaded bone cement and prosthesis-related infection. Biomaterials 2004, 25, 545–556. [Google Scholar] [CrossRef]
- Ginebra, M.-P.; Gil, F.-J.; Planell, J.-A. Acrylic bone cement: Influence of time and environment on physical properties. In Integrated Biomaterials Science; Barbucci, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2002; pp. 569–588. [Google Scholar]
- Parra-Ruíz, F.J.; González-Gómez, A.; Fernández-Gutiérrez, M.; Parra, J.; García-García, J.; Azuara, G.; Molina-Crisol, M. Development of advanced antibiotic-loaded bone cement spacers for arthroplasty associated infections. Int. J. Pharm. 2017, 522, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Román, J.S.; Vázquez-Lasa, B.; Aguilar, M.R. Self-curing systems for regenerative medicine. In Polymers in Regenerative Medicine. Biomedical Applications from Nano-to Macro-Structures; Pradas, M.M., Vicent, M.J., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015; pp. 207–233. [Google Scholar]
- Rojo, L.; Deb, S. Polymer Therapeutics in Relation to Dentistry. Front. Oral Biol. 2015, 17, 13–21. [Google Scholar] [PubMed]
- Rojo, L.; Vazquez, B.; Deb, S.; Roman, J.S. Eugenol derivatives immobilized in auto-polymerizing formulations as an approach to avoid inhibition interferences and improve biofunctionality in dental and orthopedic cements. Acta Biomater. 2009, 5, 1616–1625. [Google Scholar] [CrossRef] [PubMed]
- Palla-Rubio, B.; Araújo-Gomes, N.; Fernández-Gutiérrez, M.; Rojo, L.; Suay, J.; Gurruchaga, M.; Goñi, I. Synthesis and characterization of silica-chitosan hybrid materials as antibacterial coatings for titanium implants. Carbohydr. Polym. 2019, 203, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, S.; Tamayo, J.A.; Ospina, J.D.; Navia Porras, D.P.; Valencia Zapata, M.E.; Hernandez, J.H.M.; Grande Tovar, C.D. Antimicrobial Films Based on Nanocomposites of Chitosan/Poly (vinyl alcohol)/Graphene Oxide for Biomedical Applications. Biomolecules 2019, 9. [Google Scholar] [CrossRef]
- Khandaker, M.; Vaughan, M.B.; Morris, T.L.; White, J.J.; Meng, Z. Effect of additive particles on mechanical, thermal, and cell functioning properties of poly (methyl methacrylate ) cement. Int. J. Nanomed. 2014, 9, 2699. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Niu, L.; Li, J.; Du, J.; Wei, Y.; Hu, Y.; Wang, K. Reinforced chitosan membranes by microspheres for guided bone regeneration. J. Mech. Behav. Biomed. Mater. 2018, 81, 195–201. [Google Scholar] [CrossRef]
- Ahsan, S.M.; Thomas, M.; Reddy, K.K.; Gopal, S.; Asthana, A.; Bhatnagar, I. Chitosan as biomaterial in drug delivery and tissue engineering. Int. J. Biol. Macromol. 2018, 110, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, A.; Sittinger, M.; Risbud, M.V. Chitosan: A versatile biopolymer for orthopaedic tissue-engineering. Biomaterials 2005, 26, 5983–5990. [Google Scholar] [CrossRef] [PubMed]
- Unal, S.; Arslan, S.; Gokce, T.; Melek, B.; Karademir, B. Design and characterization of polycaprolactone-gelatin-graphene oxide scaffolds for drug in fl uence on glioblastoma cells. Eur. Polym. J. 2019, 115, 157–165. [Google Scholar] [CrossRef]
- Gong, M.; Zhao, Q.; Dai, L.; Li, Y.; Jiang, T. Fabrication of polylactic acid/hydroxyapatite/graphene oxide composite and their thermal stability, hydrophobic and mechanical properties. J. Asian Ceram. Soc. J. 2017, 5, 160–168. [Google Scholar] [CrossRef]
- Palmieri, V.; Papi, M.; Conti, C.; Ciasca, G.; Maulucci, G.; De Spirito, M. The future development of bacteria fighting medical devices: The role of graphene oxide. Expert Rev. Med. Devices 2016, 13, 1013–1019. [Google Scholar] [CrossRef]
- Dutt, S.; Bhaskar, R.; Singh, H.; Yadav, I. Development of a nanocomposite scaffold of gelatin—alginate—graphene oxide for bone tissue engineering. Int. J. Biol. Macromol. 2019, 133, 592–602. [Google Scholar]
- Paz, E.; Forriol, F.; del Real, J.C.; Dunne, N. Graphene oxide versus graphene for optimization of PMMA bone cement for orthopedic applications. Mater. Sci. Eng. C 2017, 77, 1003–1011. [Google Scholar] [CrossRef]
- Amin, M.; Mirzadeh, H.; Mahdavi, H.; Rabiee, A.; Mohebbi-kalhori, D.; Baghaban, M. Graphene oxide containing chitosan scaffolds for cartilage tissue engineering. Int. J. Biol. Macromol. 2019, 127, 396–405. [Google Scholar]
- Pei, S.; Wei, Q.; Huang, K.; Cheng, H.M.; Ren, W. Green synthesis of graphene oxide by seconds timescale water electrolytic oxidation. Nat. Commun. 2018, 9, 145. [Google Scholar] [CrossRef]
- Martínez, V.G.; Lopez, R.M.; Velasco, C.B. Estudio de la estabilidad del óxido de grafeno con el tiempo. Final master thesis degree. University of Oviedo. Oviedo, Spain. Master’s Degree, University of Oviedo, Oviedo, Spain, 2013. [Google Scholar]
- Goumri, M.; Poilâne, C.; Ruterana, P.; Doudou, B.B.; Wéry, J.; Bakour, A.; Baitoul, M. Synthesis and characterization of nanocomposites films with graphene oxide and reduced graphene oxide nanosheets. Chin. J. Phys. 2017, 55, 412–422. [Google Scholar] [CrossRef]
- Sánchez-Valdes, S.; Zapata-Domínguez, A.G.; Martinez-Colunga, J.G.; Mendez-Nonell, J.; Ramos de Valle, L.F.; Espinoza-Martinez, A.B.; Ramirez-Vargas, E. Influence of functionalized polypropylene on polypropylene/graphene oxide nanocomposite properties. Polym. Compos. 2016, 39, 1361–1369. [Google Scholar] [CrossRef]
- Valencia, C.; Valencia, C.; Zuluaga, F.; Valencia, M.; Mina, J.; Grande-Tovar, C. Synthesis and Application of Scaffolds of Chitosan-Graphene Oxide by the Freeze-Drying Method for Tissue Regeneration. Molecules 2018, 23, 2651. [Google Scholar] [CrossRef] [PubMed]
- Quan, C.; Tang, Y.; Liu, Z.; Rao, M.; Zhang, W.; Liang, P.; Jiang, Q. Effect of modification degree of nanohydroxyapatite on biocompatibility and mechanical property of injectable poly(methyl methacrylate)-based bone cement. J. Biomed. Mater. Res. Part. B Appl. Biomater. 2016, 104, 576–584. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.; Su, Q.; Liu, Z.; Liang, P.; Wu, N.; Quan, C.; Jiang, Q. Preparation and characterization of a poly(methyl methacrylate) based composite bone cement containing poly (acrylate-co-silane) modified hydroxyapatite nanoparticles. J. Appl. Polym. Sci. 2014, 131. [Google Scholar] [CrossRef]
- Ma, X.; Qiao, C.; Zhang, J.; Xu, J. Effect of sorbitol content on microstructure and thermal properties of chitosan films. Int. J. Biol. Macromol. 2018, 119, 1294–1297. [Google Scholar] [CrossRef] [PubMed]
- Riyadh, S.; Khalil, K.; Aljuhani, A. Chitosan-MgO Nanocomposite: One Pot Preparation and Its Utility as an Eco-friendly Biocatalyst in the Synthesis of Thiazoles and [1,3,4] thiadiazoles. Nanomaterials 2018, 8, 928. [Google Scholar] [CrossRef]
- Paz, E.; Abenojar, J.; Ballesteros, Y.; Forriol, F.; Dunne, N.; del Real, J.C. Mechanical and thermal behavior of an acrylic bone cement modified with a triblock copolymer. J. Mater. Sci. Mater. Med. 2016, 27. [Google Scholar] [CrossRef]
- Islas-Blancas, M.E.; Cervantes, J.M.; Vargas-Coronado, R.; Cauich-Rodriguez, J.V.; Vera-Graziano, R.; Martinez-Richa, A. Characterization of bone cement prepared with functionalized methacrylates and hydroxyapatite. J. Biomater. Sci. Polym. Ed. 2001, 12, 893–910. [Google Scholar] [CrossRef]
- Kuehn, K.-D.; Ege, W.; Gopp, U. Acrylic bone cement: Composition and properties. Orthop. Clin. N. Am. 2005, 36, 17–28. [Google Scholar] [CrossRef]
- Gonalves, G.; Cruz, S.M.A.; Ramalho, A.; Grácio, J.; Marques, P.A.A.P. Graphene oxide versus functionalized carbon nanotubes as a reinforcing agent in a PMMA/HA bone cement. Nanoscale 2012, 4, 2937–2945. [Google Scholar] [CrossRef]
- Khandaker, M.; Riahinezhad, S.; Jamadagni, H.G.; Morris, T.L.; Coles, A.V.; Vaughan, M.B. Use of Polycaprolactone Electrospun Nanofibers as A Coating for Poly (methyl methacrylate) Bone Cement. Nanomaterials 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Rojo, L.; Gharibi, B.; McLister, R.; Meenan, B.J.; Deb, S. Self-assembled monolayers of alendronate on Ti6Al4V alloy surfaces enhance osteogenesis in mesenchymal stem cells. Sci. Rep. 2016, 6, 30548. [Google Scholar] [CrossRef] [PubMed]
- Croll, H.; Soroush, A.; Pillsbury, M.E.; Castrillón, S.R.-V. Graphene oxide surface modification of polyamide reverse osmosis membranes for improved N-nitrosodimethylamine (NDMA) removal. Sep. Purif. Technol. 2019, 210, 973–980. [Google Scholar] [CrossRef]
- Pawar, P.B.; Shukla, S.; Saxena, S. Graphene oxide—Polyvinyl alcohol nanocomposite based electrode material for supercapacitors. J. Power Sources 2016, 321, 102–105. [Google Scholar] [CrossRef]
- Severin, I.; Boiko, V.; Moiseyenko, V.; Olenchuk, M.; Goushcha, A.; Barillé, R.; Dovbeshko, G. Optical properties of graphene oxide coupled with 3D opal based photonic crystal. Opt. Mater. 2018, 86, 326–330. [Google Scholar] [CrossRef]
- International Standard. ISO 5833: Implants for Surgery—Acrylic Resin Cement; International Standard: Geneva, Switzerland, 2002; pp. 1–22. [Google Scholar]
- Lin, L.C.; Chang, S.J.; Kuo, S.M.; Chen, S.F.; Kuo, C.H. Evaluation of chitosan/β -tricalcium phosphate microspheres as a constituent to PMMA cement. J. Mater. Sci. Mater. Med. 2005, 16, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Franco-Marquès, E.; Mendez, J.A.; Girones, J.; Pelach, M.A. Thermal and dynamic mechanical characterization of acrylic bone cement modified with biodegradable polymers. J. Appl. Polym. Sci. 2013, 128, 3455–3464. [Google Scholar] [CrossRef]
- Pelto, J.; Verho, T.; Ronkainen, H.; Kaunisto, K.; Metsäjoki, J.; Seitsonen, J.; Karttunen, M. Matrix morphology and the particle dispersion in HDPE nanocomposites with enhanced wear resistance. Polym. Test. 2019, 77, 105897. [Google Scholar] [CrossRef]
- Martino, S.; D’Angelo, F.; Armentano, I.; Kenny, J.M.; Orlacchio, A. Stem cell-biomaterial interactions for regenerative medicine. Biotechnol. Adv. 2012, 30, 338–351. [Google Scholar] [CrossRef]
- Deb, S.; Silvio, L.D.; Vazquez, B.; Roman, J.S. Water absorption characteristics and cytotoxic and biological evaluation of bone cements formulated with a novel activator. J. Biomed. Mater. Res. 1999, 48, 719–725. [Google Scholar] [CrossRef]
- Espigares, I.; Elvira, C.; Mano, J.F.; Vázquez, B.; Román, J.S.; Reis, R.L. New partially degradable and bioactive acrylic bone cements based on starch blends and ceramic fillers. Biomaterials 2002, 23, 1883–1895. [Google Scholar] [CrossRef]
- Naveed, M.; Phil, L.; Sohail, M.; Hasnat, M.; Baig, M.M.F.A.; Ihsan, A.U.; Husain, M.I. Chitosan oligosaccharide (COS): An overview. Int. J. Biol. Macromol. 2019, 129, 827–843. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Unagolla, J.M.; Jayasuriya, A.C. Enhanced cell functions on graphene oxide incorporated 3D printed polycaprolactone scaffolds. Mater. Sci. Eng. C 2019, 102. [Google Scholar] [CrossRef] [PubMed]
- Shang, L.; Qi, Y.; Lu, H.; Pei, H.; Li, Y.; Qu, L.; Zhang, W. Graphene and Graphene Oxide for Tissue Engineering and Regeneration. In Theranostic Bionanomaterials; Cui, W., Zhao, X., Eds.; Elsevier Inc.: Cambridge, UK, 2019; pp. 165–186. [Google Scholar]
- Magnusson, P.; Larsson, L.; Magnusson, M.; Davie, M.W.; Sharp, C.A. Isoforms of bone alkaline phosphatase: Characterization and origin in human trabecular and cortical bone. J. Bone Miner. Res. 1999, 14, 1926–1933. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, S.; Chawla, A.; Vairamani, M.; Sastry, T.P.; Subramanian, K.S.; Selvamurugan, N. Scaffolds containing chitosan, gelatin and graphene oxide for bone tissue regeneration in vitro and in vivo. Int. J. Biol. Macromol. 2017, 104, 1975–1985. [Google Scholar] [CrossRef] [PubMed]
- López Tenorio, D.; Valencia, C.H.; Valencia, C.; Zuluaga, F.; Valencia, M.E.; Mina, J.H.; Grande Tovar, C.D. Evaluation of the Biocompatibility of CS-Graphene Oxide Compounds In Vivo. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Peng, H.; Wang, X.; Shao, F.; Yuan, Z.; Han, H. Graphene oxide exhibits broad-spectrum antimicrobial activity against bacterial phytopathogens and fungal conidia by intertwining and membrane perturbation. Nanoscale 2014, 6, 1879–1889. [Google Scholar] [CrossRef]
- Ye, S.; Shao, K.; Li, Z.; Guo, N.; Zuo, Y.; Li, Q.; Han, H. Antiviral Activity of Graphene Oxide: How Sharp Edged Structure and Charge Matter. ACS Appl. Mater. Interfaces 2015, 7, 21571–21579. [Google Scholar] [CrossRef] [PubMed]
- Perreault, F.; de Faria, A.F.; Nejati, S.; Elimelech, M. Antimicrobial Properties of Graphene Oxide Nanosheets: Why Size Matters. ACS Nano 2015, 9, 7226–7236. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Grande, C.D.; Rodrigues, D.F. Biodegradation of graphene oxide-polymer nanocomposite films in wastewater. Environ. Sci. Nano 2017, 4, 1808–1816. [Google Scholar] [CrossRef]
- Mangadlao, J.D.; de Leon, A.C.C.; Felipe, M.J.L.; Cao, P.; Advincula, P.A.; Advincula, R.C. Grafted Carbazole-Assisted Electrodeposition of Graphene Oxide. ACS Appl. Mater. Interfaces 2015, 7, 10266–10274. [Google Scholar] [CrossRef] [PubMed]
- American Society of Testing Materials. ASTM F1635-16, Standard Test Method for in Vitro Degradation Testing of Hydrolytically Degradable Polymer Resins and Fabricated Forms for Surgical Implants; American Society of Testing Materials: West Conshohocken, PA, USA, 2016; pp. 1–5. [Google Scholar]


| Formulation | Residual Monomer (%) | Tg by DMA (°C) |
|---|---|---|
| Control ABC | 1.1 | 114.8 |
| ABC 0.3 % GO | 1.3 | 119.6 |
| ABC 15 % CS | 1.6 | 109.5 |
| ABC 0.3GO-15CS | 3.6 | 124.5 |
| Formulation | Solid Phase (% w/w) | Liquid Phase (% w/w) | ||||||
|---|---|---|---|---|---|---|---|---|
| PMMA | BaSO4 | BPO | CS | MMA | DEAEM/ DEAEA | DMPT | GO | |
| Control ABC | 88 | 10 | 2 | 0 | 95.5 | 2 | 2.5 | 0 |
| ABC 0.3 % GO | 88 | 10 | 2 | 0 | 95.2 | 2 | 2.5 | 0.3 |
| ABC 15 % CS | 73 | 10 | 2 | 15 | 95.5 | 2 | 2.5 | 0 |
| ABC 0.3GO-15CS | 73 | 10 | 2 | 15 | 95.2 | 2 | 2.5 | 0.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valencia Zapata, M.E.; Mina Hernandez, J.H.; Grande Tovar, C.D.; Valencia Llano, C.H.; Diaz Escobar, J.A.; Vázquez-Lasa, B.; San Román, J.; Rojo, L. Novel Bioactive and Antibacterial Acrylic Bone Cement Nanocomposites Modified with Graphene Oxide and Chitosan. Int. J. Mol. Sci. 2019, 20, 2938. https://doi.org/10.3390/ijms20122938
Valencia Zapata ME, Mina Hernandez JH, Grande Tovar CD, Valencia Llano CH, Diaz Escobar JA, Vázquez-Lasa B, San Román J, Rojo L. Novel Bioactive and Antibacterial Acrylic Bone Cement Nanocomposites Modified with Graphene Oxide and Chitosan. International Journal of Molecular Sciences. 2019; 20(12):2938. https://doi.org/10.3390/ijms20122938
Chicago/Turabian StyleValencia Zapata, Mayra Eliana, José Herminsul Mina Hernandez, Carlos David Grande Tovar, Carlos Humberto Valencia Llano, José Alfredo Diaz Escobar, Blanca Vázquez-Lasa, Julio San Román, and Luis Rojo. 2019. "Novel Bioactive and Antibacterial Acrylic Bone Cement Nanocomposites Modified with Graphene Oxide and Chitosan" International Journal of Molecular Sciences 20, no. 12: 2938. https://doi.org/10.3390/ijms20122938
APA StyleValencia Zapata, M. E., Mina Hernandez, J. H., Grande Tovar, C. D., Valencia Llano, C. H., Diaz Escobar, J. A., Vázquez-Lasa, B., San Román, J., & Rojo, L. (2019). Novel Bioactive and Antibacterial Acrylic Bone Cement Nanocomposites Modified with Graphene Oxide and Chitosan. International Journal of Molecular Sciences, 20(12), 2938. https://doi.org/10.3390/ijms20122938








