Regulation of Serum Sphingolipids in Andean Children Born and Living at High Altitude (3775 m)
Abstract
1. Introduction
2. Results
2.1. Biochemical Parameters Assessment
2.2. Anthropometrics and Biochemical Differences
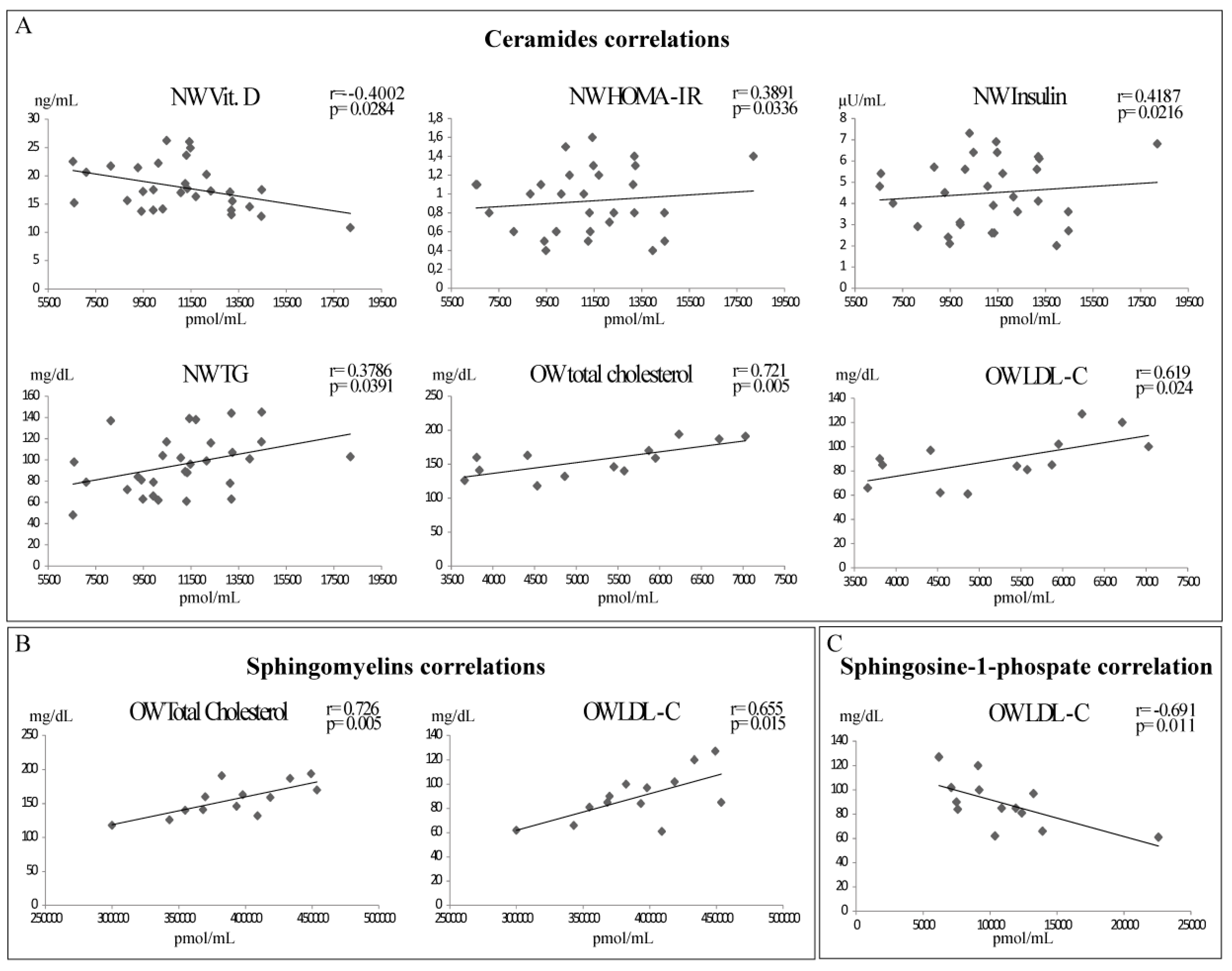
2.3. Sphingolipids and Biochemical Parameters Correlation
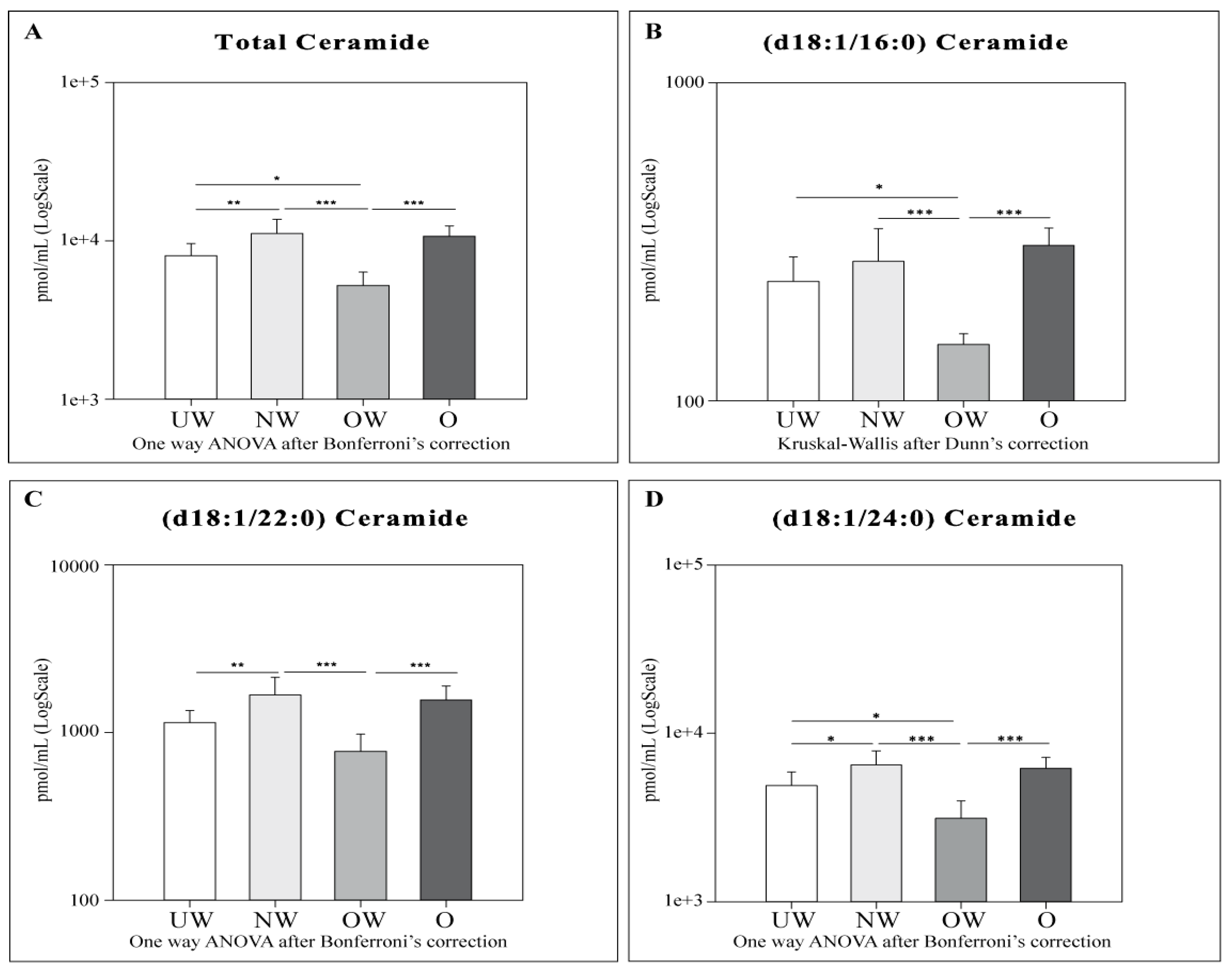
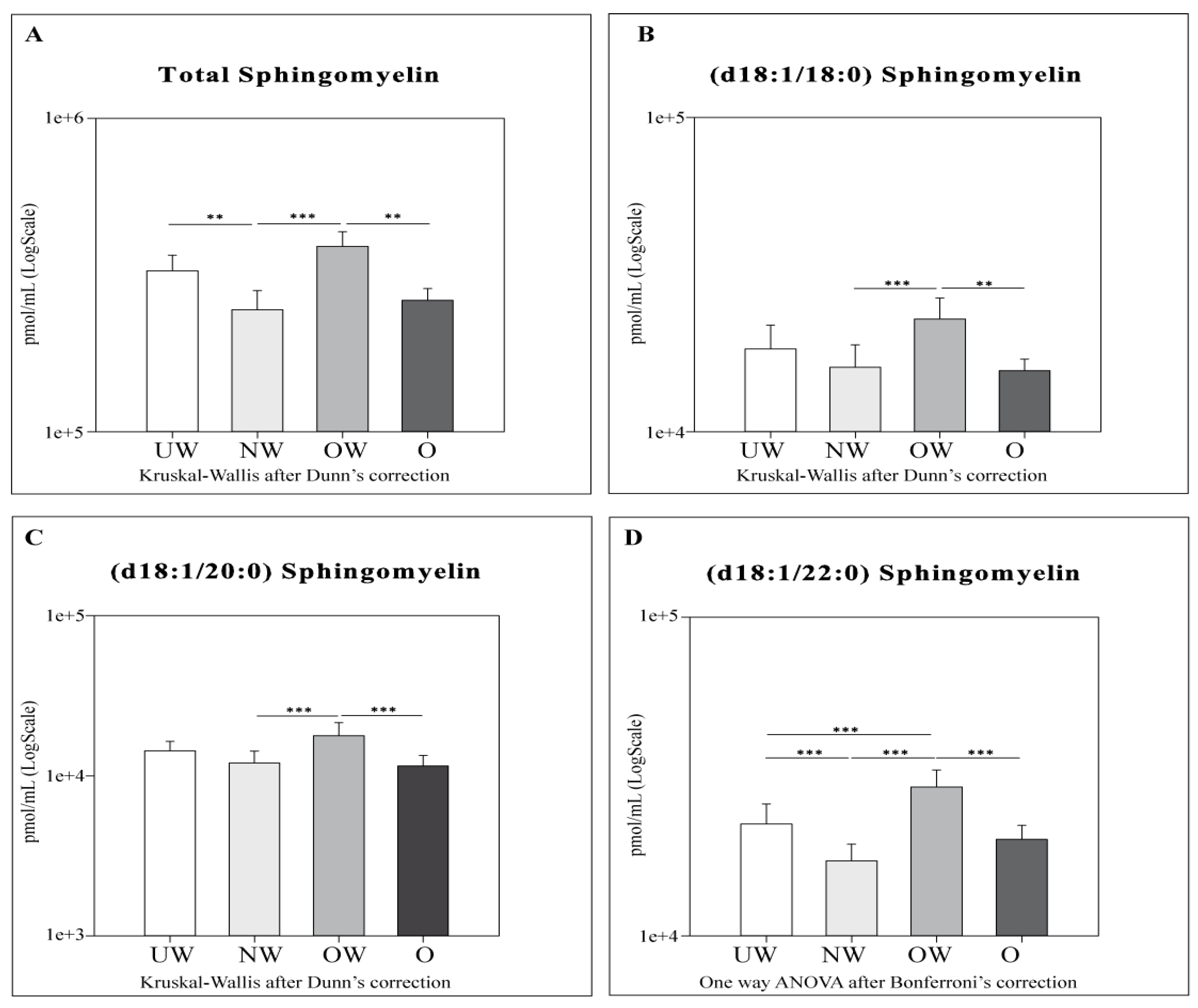
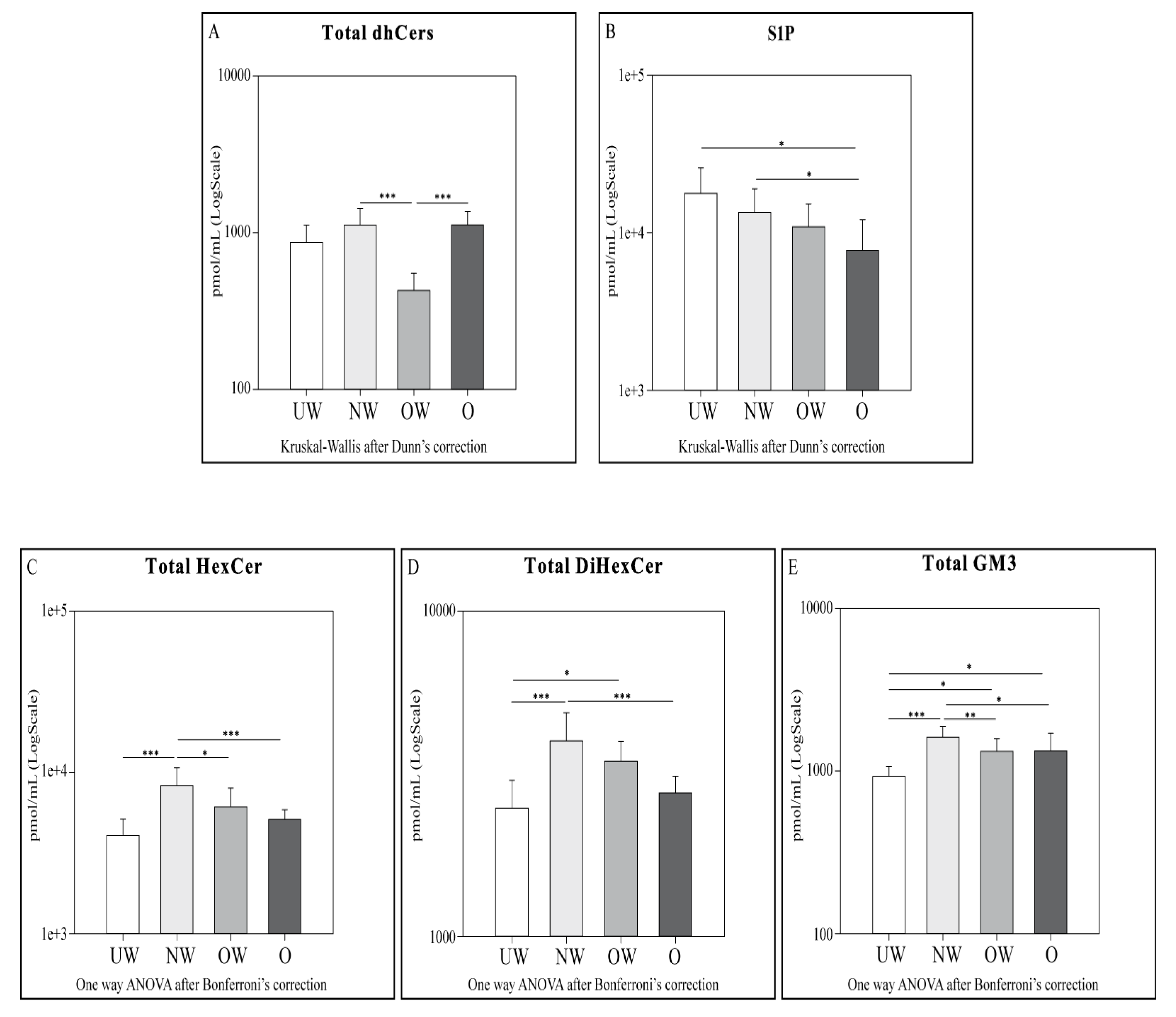
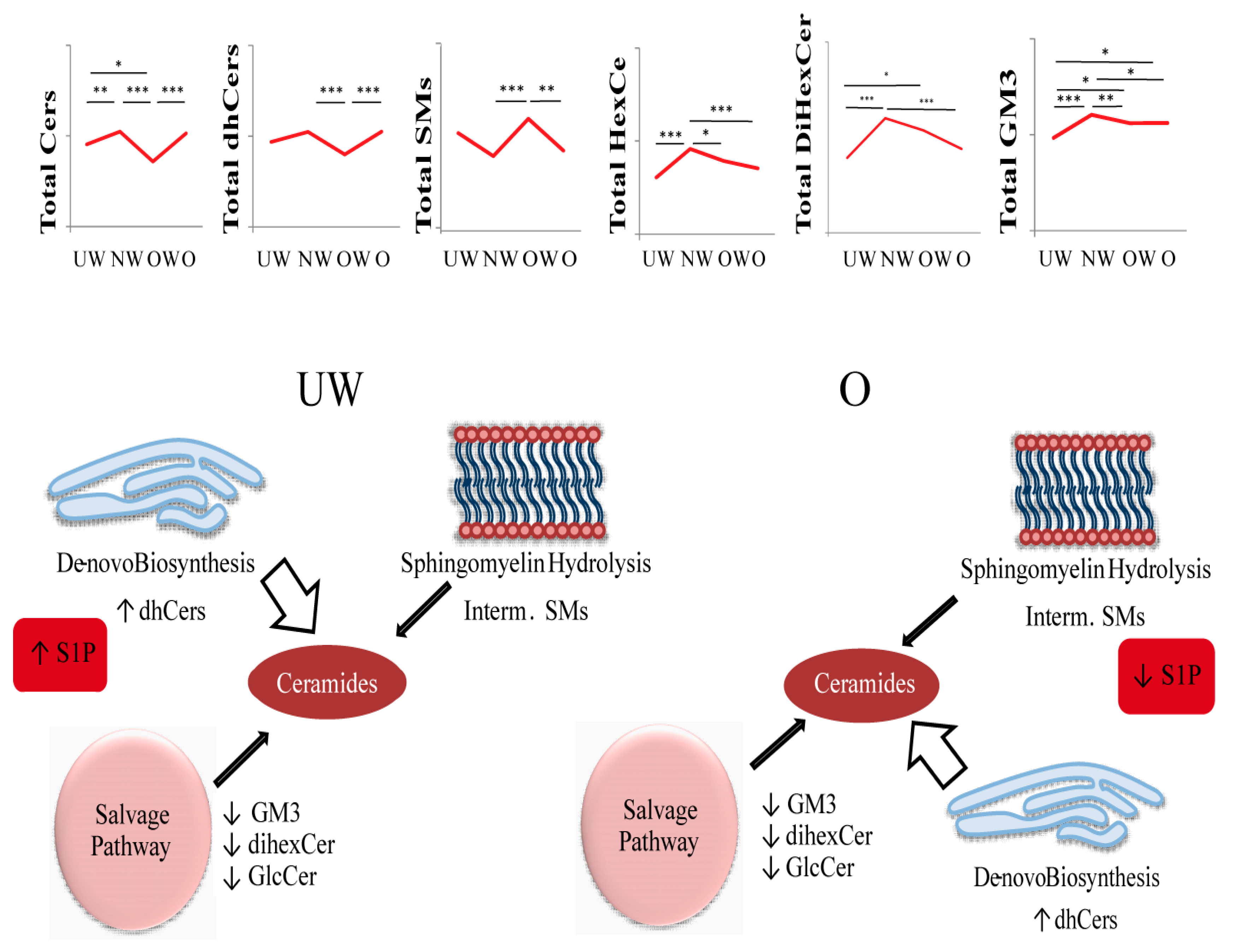
2.4. Sphingolipid Results by LC-MS
To Summarize:
- Cers: UW subjects were found to have lower levels of total Cers, cer (d18:1/22:0), and cer (d18:1/24:0) compared to NW. OW subjects showed lower levels of total Cers and all acyl Cers chains compared to NW and O. UW and OW subjects are characterized by lower levels for total Cers, Cer (d18:1/16:0), and Cer (d18:1/24:0).
- dhCers: Comparing UW and NW groups, lower levels of total SMs and SM (d18:1/22:0) were observed in NW. OW subjects showed lower levels of total dhCers when compared to NW and O groups.
- SMs: OW subjects showed higher levels of total SMs and all acyl SMs chains compared to their NW and O counterparts. OW subjects were also characterized by higher levels of SM (d18:1/22:0) compared to UW subjects only.
- HexCer, diHexCer, and GM3: NW subjects showed higher levels of HexCer, dihexCer, and GM3 compared to UW and O. UW subjects had lower levels of total dihexCer compared to OW subjects and lower levels of total GM3 compared both to OW and O groups. When comparing NW vs. OW, total HexCer and total GM3 were higher in NW subjects.
- S1P: a decrement in S1P levels was observed in O subjects compared to both UW and NW.
3. Discussion
4. Materials and Methods
4.1. Participants and Sample Collection
4.2. Reagents and Chemicals
4.3. Lipid Extraction
4.4. UPLC-MS for Sphingolipids
4.5. MRM Analysis for S1P
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| UW | Underweight |
| NW | Normal weight |
| OW | Overweight |
| O | Obese |
| LC-MS/MS | Liquid Chromatography Mass Spectrometry/Mass spectrometry |
| Cers | Ceramides |
| S1P | Sphingosine 1 phosphate |
| SMs | Sphingomyelins |
| HDL-C | High density lipoprotein-cholesterol |
| LDL-C | Low density lipoprotein-cholesterol |
| A.s.l | Above sea level |
| HIF | Hypoxia inducible factor |
| EPAS1 | Endothelial PAS Domain Protein 1 |
| EGLN1 | Egl-9 Family Hypoxia Inducible Factor 1 |
| NOS2A | Nitric oxide synthase 2 |
| CH | Chicoana |
| SAC | San Antonio de los Cobres |
| BA | Buenos Aires |
| CerS | Ceramide Synthases |
| SphK | Sphingosine Kinase |
| ER | Endoplasmic reticulum |
| HOMA-IR | Homeostatic model assessment of insulin resistance |
| TC | Total Cholesterol |
| TG | Triglycerides |
| Vit. D | Vitamin D |
| dhCers | Dihydroceramides |
| HexCer | Hexosylceramides |
| DiHexCer | DiHexosylceramides |
| GM3 | Monosialodihexosylganglioside |
| BMI | Body Mass Index |
| apoM | Apolipoprotein M |
| eNOS | Endothelial nitric oxide synthase |
| NO | Nitric oxide |
| UPR | Unfolded protein response |
| 2,3-BPG | 2,3-diphosphoglycerate |
| NEU3 | Neuraminidase 3 |
| PCO2 | Partial pressure of carbon dioxide |
| PO2 | Partial pressure of oxygen |
| SO2 | Haemoglobin-O2 saturation |
| n-NOS | Nitric oxide synthase |
| PBMCs | Peripheral blood mononuclear cells |
| SRM | Selected Reaction Monitoring |
| CDC | Centers for Disease Control and Prevention |
| BHT | 3,5-Di-tert-4-butylhydroxytoluene |
References
- Ge, R.-L.; Simonson, T.S.; Gordeuk, V.; Prchal, J.T.; McClain, D.A. Metabolic aspects of high-altitude adaptation in Tibetans. Exp. Physiol. 2015, 100, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.G. Measuring high-altitude adaptation. J. Appl. Physiol. 2017, 123, 1371–1385. [Google Scholar] [CrossRef] [PubMed]
- Murray, A.J.; Montgomery, H.E.; Feelisch, M.; Grocott, M.P.W.; Martin, D.S. Metabolic adjustment to high-altitude hypoxia: From genetic signals to physiological implications. Biochem. Soc. Trans. 2018, 46, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Beall, C.M. Andean, Tibetan, and Ethiopian patterns of adaptation to high-altitude hypoxia. Integr. Comp. Biol. 2006, 46, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Simonson, T.S.; Huff, C.D.; Witherspoon, D.J.; Prchal, J.T.; Jorde, L.B. Adaptive genetic changes related to haemoglobin concentration in native high-altitude Tibetans. Exp. Physiol. 2015, 100, 1263–1268. [Google Scholar] [CrossRef] [PubMed]
- Bigham, A.W.; Mao, X.; Mei, R.; Brutsaert, T.; Wilson, M.J.; Julian, C.G.; Parra, E.J.; Akey, J.M.; Moore, L.G.; Shriver, M.D. Identifying positive selection candidate loci for high-altitude adaptation in Andean populations. Hum. Genom. 2009, 4, 79–90. [Google Scholar] [PubMed]
- Julian, C.G.; Moore, L.G. Human Genetic Adaptation to High Altitude: Evidence from the Andes. Genes 2019, 10, 150. [Google Scholar] [CrossRef]
- Hirschler, V.; Maccallini, G.; Aranda, C.; Molinari, C. San Antonio de los Cobres Study Group Dyslipidemia without Obesity in Indigenous Argentinean Children Living at High Altitude. J. Pediatr. 2012, 161, 646.e1–651.e1. [Google Scholar] [CrossRef]
- Beall, C.M.; Brittenham, G.M.; Macuaga, F.; Barragan, M. Variation in hemoglobin concentration among samples of high-altitude natives in the Andes and the Himalayas. Am. J. Hum. Biol. 1990, 2, 639–651. [Google Scholar] [CrossRef]
- Beall, C.M.; Brittenham, G.M.; Strohl, K.P.; Blangero, J.; Williams-Blangero, S.; Goldstein, M.C.; Decker, M.J.; Vargas, E.; Villena, M.; Soria, R.; et al. Hemoglobin concentration of high-altitude Tibetans and Bolivian Aymara. Am. J. Phys. Anthropol. 1998, 106, 385–400. [Google Scholar] [CrossRef]
- Beall, C.M.; Reichsman, A.B. Hemoglobin levels in a Himalayan high altitude population. Am. J. Phys. Anthropol. 1984, 63, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Beall, C.M.; Decker, M.J.; Brittenham, G.M.; Kushner, I.; Gebremedhin, A.; Strohl, K.P. An Ethiopian pattern of human adaptation to high-altitude hypoxia. Proc. Natl. Acad. Sci. USA 2002, 99, 17215–17218. [Google Scholar] [CrossRef] [PubMed]
- Tashi, T.; Scott Reading, N.; Wuren, T.; Zhang, X.; Moore, L.G.; Hu, H.; Tang, F.; Shestakova, A.; Lorenzo, F.; Burjanivova, T.; et al. Gain-of-function EGLN1 prolyl hydroxylase (PHD2 D4E:C127S) in combination with EPAS1 (HIF-2α) polymorphism lowers hemoglobin concentration in Tibetan highlanders. J. Mol. Med. 2017, 95, 665–670. [Google Scholar] [CrossRef] [PubMed]
- Bigham, A.W.; Lee, F.S. Human high-altitude adaptation: Forward genetics meets the HIF pathway. Genes Dev. 2014, 28, 2189–2204. [Google Scholar] [CrossRef] [PubMed]
- Hirschler, V.; Gonzalez, C.; Maccallini, G.; Hidalgo, M.; Molinari, C. Comparison Between HDL-C Levels in Argentine Indigenous Children Living at High Altitudes and U.S. Children. Diabetes Technol. Ther. 2016, 18, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Hirschler, V.; Maccallini, G.; Molinari, C.; Hidalgo, M.; Intersimone, P.; Gonzalez, C. Type 2 diabetes markers in indigenous Argentinean children living at different altitudes. AIMS Public Health 2018, 5, 440–453. [Google Scholar] [CrossRef]
- Hirschler, V.; Molinari, C.; Maccallini, G.; Intersimone, P.; Gonzalez, C.D. Blood Pressure Levels Among Indigenous Children Living at Different Altitudes. Appl. Physiol. Nutr. Metab. 2018, 44, 659–664. [Google Scholar] [CrossRef]
- Millán, J.; Pintó, X.; Muñoz, A.; Zúñiga, M.; Rubiés-Prat, J.; Pallardo, L.F.; Masana, L.; Mangas, A.; Hernández-Mijares, A.; González-Santos, P.; et al. Lipoprotein ratios: Physiological significance and clinical usefulness in cardiovascular prevention. Vasc. Health Risk Manag. 2009, 5, 757–765. [Google Scholar]
- McLaughlin, T.; Abbasi, F.; Cheal, K.; Chu, J.; Lamendola, C.; Reaven, G. Use of metabolic markers to identify overweight individuals who are insulin resistant. Ann. Intern. Med. 2003, 139, 802–809. [Google Scholar] [CrossRef]
- Hanak, V.; Munoz, J.; Teague, J.; Stanley, A.; Bittner, V. Accuracy of the triglyceride to high-density lipoprotein cholesterol ratio for prediction of the low-density lipoprotein phenotype B. Am. J. Cardiol. 2004, 94, 219–222. [Google Scholar] [CrossRef]
- Crowder, C.M. Cell biology. Ceramides--friend or foe in hypoxia? Science 2009, 324, 343–344. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rahar, B.; Chawla, S.; Pandey, S.; Bhatt, A.N.; Saxena, S. Sphingosine-1-phosphate pretreatment amends hypoxia-induced metabolic dysfunction and impairment of myogenic potential in differentiating C2C12 myoblasts by stimulating viability, calcium homeostasis and energy generation. J. Physiol. Sci. 2018, 68, 137–151. [Google Scholar] [CrossRef] [PubMed]
- Turpin, S.M.; Nicholls, H.T.; Willmes, D.M.; Mourier, A.; Brodesser, S.; Wunderlich, C.M.; Mauer, J.; Xu, E.; Hammerschmidt, P.; Brönneke, H.S.; et al. Obesity-Induced CerS6-Dependent C16:0 Ceramide Production Promotes Weight Gain and Glucose Intolerance. Cell Metab. 2014, 20, 678–686. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, S.; Milstien, S. Sphingosine-1-phosphate: An enigmatic signalling lipid. Nat. Rev. Mol. Cell Biol. 2003, 4, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Pitson, S.M.; Moretti, P.A.B.; Zebol, J.R.; Lynn, H.E.; Xia, P.; Vadas, M.A.; Wattenberg, B.W. Activation of sphingosine kinase 1 by ERK1/2-mediated phosphorylation. EMBO J. 2003, 22, 5491–5500. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, N.; Okada, T.; Hayashi, S.; Fujita, T.; Jahangeer, S.; Nakamura, S. Sphingosine kinase 2 is a nuclear protein and inhibits DNA synthesis. J. Biol. Chem. 2003, 278, 46832–46839. [Google Scholar] [CrossRef] [PubMed]
- Le Stunff, H.; Giussani, P.; Maceyka, M.; Lépine, S.; Milstien, S.; Spiegel, S. Recycling of sphingosine is regulated by the concerted actions of sphingosine-1-phosphate phosphohydrolase 1 and sphingosine kinase 2. J. Biol. Chem. 2007, 282, 34372–34380. [Google Scholar] [CrossRef] [PubMed]
- Frangioudakis, G.; Garrard, J.; Raddatz, K.; Nadler, J.L.; Mitchell, T.W.; Schmitz-Peiffer, C. Saturated- and n-6 polyunsaturated-fat diets each induce ceramide accumulation in mouse skeletal muscle: Reversal and improvement of glucose tolerance by lipid metabolism inhibitors. Endocrinology 2010, 151, 4187–4196. [Google Scholar] [CrossRef]
- Bikman, B.T.; Summers, S.A. Ceramides as modulators of cellular and whole-body metabolism. J. Clin. Investig. 2011, 121, 4222–4230. [Google Scholar] [CrossRef]
- Boslem, E.; MacIntosh, G.; Preston, A.M.; Bartley, C.; Busch, A.K.; Fuller, M.; Laybutt, D.R.; Meikle, P.J.; Biden, T.J. A lipidomic screen of palmitate-treated MIN6 β-cells links sphingolipid metabolites with endoplasmic reticulum (ER) stress and impaired protein trafficking. Biochem. J. 2011, 435, 267–276. [Google Scholar] [CrossRef]
- Lei, X.; Zhang, S.; Emani, B.; Barbour, S.E.; Ramanadham, S. A link between endoplasmic reticulum stress-induced β-cell apoptosis and the group VIA Ca2+-independent phospholipase A2 (iPLA2β). Diabetes. Obes. Metab. 2010, 12 (Suppl. 2), 93–98. [Google Scholar] [CrossRef] [PubMed]
- Cnop, M.; Foufelle, F.; Velloso, L.A. Endoplasmic reticulum stress, obesity and diabetes. Trends Mol. Med. 2012, 18, 59–68. [Google Scholar] [CrossRef] [PubMed]
- National Cholesterol Education Program (NCEP): Highlights of the report of the Expert Panel on Blood Cholesterol Levels in Children and Adolescents. Pediatrics 1992, 89, 495–501.
- Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents. Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents: Summary report. Pediatrics 2011, 128, S213. [Google Scholar]
- Hirschler, V.; Maccallini, G.; Sanchez, M.S.; Castaño, L.; Molinari, C. Improvement in High-Density Lipoprotein Cholesterol Levels in Argentine Indian School Children after Vitamin D Supplementation. Horm. Res. Paediatr. 2013, 80, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Bischoff-Ferrari, H.A.; Giovannucci, E.; Willett, W.C.; Dietrich, T.; Dawson-Hughes, B. Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. Am. J. Clin. Nutr. 2006, 84, 18–28. [Google Scholar] [CrossRef]
- Hirschler, V.; Molinari, C.; Maccallini, G.; Intersimone, P.; Gonzalez, C.D. Vitamin D Levels and Cardiometabolic Markers in Indigenous Argentinean Children Living at Different Altitudes. Glob. Pediatr. Health 2019, 6. [Google Scholar] [CrossRef]
- Mohanna, S.; Baracco, R.; Seclén, S. Lipid Profile, Waist Circumference, and Body Mass Index in a High Altitude Population. High Alt. Med. Biol. 2006, 7, 245–255. [Google Scholar] [CrossRef]
- Sun, K.; Zhang, Y.; D’alessandro, A.; Nemkov, T.; Song, A.; Wu, H.; Liu, H.; Adebiyi, M.; Huang, A.; Wen, Y.E.; et al. ARTICLE Sphingosine-1-phosphate promotes erythrocyte glycolysis and oxygen release for adaptation to high-altitude hypoxia. Nat. Publ. Gr. 2016, 7, 12086. [Google Scholar]
- Okajima, F. Plasma lipoproteins behave as carriers of extracellular sphingosine 1-phosphate: Is this an atherogenic mediator or an anti-atherogenic mediator? Biochim. Biophys. Acta 2002, 1582, 132–137. [Google Scholar] [CrossRef]
- Christoffersen, C.; Obinata, H.; Kumaraswamy, S.B.; Galvani, S.; Ahnstrom, J.; Sevvana, M.; Egerer-Sieber, C.; Muller, Y.A.; Hla, T.; Nielsen, L.B.; et al. Endothelium-protective sphingosine-1-phosphate provided by HDL-associated apolipoprotein M. Proc. Natl. Acad. Sci. USA 2011, 108, 9613–9618. [Google Scholar] [CrossRef] [PubMed]
- Nofer, J.-R.; van der Giet, M.; Tölle, M.; Wolinska, I.; von Wnuck Lipinski, K.; Baba, H.A.; Tietge, U.J.; Gödecke, A.; Ishii, I.; Kleuser, B.; et al. HDL induces NO-dependent vasorelaxation via the lysophospholipid receptor S1P3. J. Clin. Investig. 2004, 113, 569–581. [Google Scholar] [CrossRef] [PubMed]
- Daiber, A.; Xia, N.; Steven, S.; Oelze, M.; Hanf, A.; Kröller-Schön, S.; Münzel, T.; Li, H.; Daiber, A.; Xia, N.; et al. New Therapeutic Implications of Endothelial Nitric Oxide Synthase (eNOS) Function/Dysfunction in Cardiovascular Disease. Int. J. Mol. Sci. 2019, 20, 187. [Google Scholar] [CrossRef] [PubMed]
- Leone, R.; Giussani, P.; De Palma, S.; Fania, C.; Capitanio, D.; Vasso, M.; Brioschi, L.; Riboni, L.; Viani, P.; Gelfi, C. Proteomic analysis of human glioblastoma cell lines differently resistant to a nitric oxide releasing agent. Mol. Biosyst. 2015, 11, 1612–1621. [Google Scholar] [CrossRef] [PubMed]
- Saponaro, C.; Gaggini, M.; Carli, F.; Gastaldelli, A. The Subtle Balance between Lipolysis and Lipogenesis: A Critical Point in Metabolic Homeostasis. Nutrients 2015, 7, 9453–9474. [Google Scholar] [CrossRef] [PubMed]
- Al-Daghri, N.M.; Torretta, E.; Capitanio, D.; Fania, C.; Guerini, F.R.; Sabico, S.B.; Clerici, M.; Gelfi, C. Intermediate and low abundant protein analysis of vitamin D deficient obese and non-obese subjects by MALDI-profiling. Sci. Rep. 2017, 7, 12633. [Google Scholar] [CrossRef] [PubMed]
- Gelfi, C.; Vasso, M.; Cerretelli, P. Diversity of human skeletal muscle in health and disease: Contribution of proteomics. J. Proteomics 2011, 74, 774–795. [Google Scholar] [CrossRef] [PubMed]
- Yazıcı, D.; Sezer, H. Insulin Resistance, Obesity and Lipotoxicity; Springer: Cham, Switzerland, 2017; pp. 277–304. [Google Scholar]
- Yoshizumi, S.; Suzuki, S.; Hirai, M.; Hinokio, Y.; Yamada, T.; Yamada, T.; Tsunoda, U.; Aburatani, H.; Yamaguchi, K.; Miyagi, T.; et al. Increased hepatic expression of ganglioside-specific sialidase, NEU3, improves insulin sensitivity and glucose tolerance in mice. Metabolism 2007, 56, 420–429. [Google Scholar] [CrossRef]
- Capitanio, D.; Fania, C.; Torretta, E.; Viganò, A.; Moriggi, M.; Bravatà, V.; Caretti, A.; Levett, D.Z.H.; Grocott, M.P.W.; Samaja, M.; et al. TCA cycle rewiring fosters metabolic adaptation to oxygen restriction in skeletal muscle from rodents and humans. Sci. Rep. 2017, 7, 9723. [Google Scholar] [CrossRef]
- Xie, C.; Yagai, T.; Luo, Y.; Liang, X.; Chen, T.; Wang, Q.; Sun, D.; Zhao, J.; Ramakrishnan, S.K.; Sun, L.; et al. Activation of intestinal hypoxia-inducible factor 2α during obesity contributes to hepatic steatosis. Nat. Med. 2017, 23, 1298–1308. [Google Scholar] [CrossRef]
- Raichur, S.; Wang, S.T.; Chan, P.W.; Li, Y.; Ching, J.; Chaurasia, B.; Dogra, S.; Öhman, M.K.; Takeda, K.; Sugii, S.; et al. CerS2 Haploinsufficiency Inhibits β-Oxidation and Confers Susceptibility to Diet-Induced Steatohepatitis and Insulin Resistance. Cell Metab. 2014, 20, 687–695. [Google Scholar] [CrossRef] [PubMed]
- Chaurasia, B.; Kaddai, V.A.; Lancaster, G.I.; Henstridge, D.C.; Sriram, S.; Galam, D.L.A.; Gopalan, V.; Prakash, K.N.B.; Velan, S.S.; Bulchand, S.; et al. Adipocyte Ceramides Regulate Subcutaneous Adipose Browning, Inflammation, and Metabolism. Cell Metab. 2016, 24, 820–834. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Lu, H.; Yang, J.; Xiang, H.; Peng, H. Sphingosine 1-phosphate in metabolic syndrome (Review). Int. J. Mol. Med. 2016, 38, 1030–1038. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, M.; Frej, C.; Holmér, A.; Guo, L.J.; Tran, S.; Dahlbäck, B. High-Density Lipoprotein–Associated Apolipoprotein M Limits Endothelial Inflammation by Delivering Sphingosine-1-Phosphate to the Sphingosine-1-Phosphate Receptor 1. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Obinata, H.; Hla, T. Sphingosine 1-phosphate and inflammation. Int. Immunol. 2019, 31. [Google Scholar] [CrossRef]
- Sattler, K.; Levkau, B. Sphingosine-1-phosphate as a mediator of high-density lipoprotein effects in cardiovascular protection. Cardiovasc. Res. 2008, 82, 201–211. [Google Scholar] [CrossRef]
- Guerrini, G.; Morabito, A.; Samaja, M. Detection of Haemoglobins with Abnormal Oxygen Affinity by Single Blood Gas Analysis and 2,3-Diphosphoglycerate Measurement. Clin. Chem. Lab. Med. 2000, 38, 951–954. [Google Scholar] [CrossRef][Green Version]
- Kuczmarski, R.J.; Ogden, C.L.; Guo, S.S.; Grummer-Strawn, L.M.; Flegal, K.M.; Mei, Z.; Wei, R.; Curtin, L.R.; Roche, A.F.; Johnson, C.L. 2000 CDC Growth Charts for the United States: Methods and development. Vital Health Stat. 11 2002, 1–190. Available online: https://www.cdc.gov/nchs/data/series/sr_11/sr11_246.pdf (accessed on 1 June 2019).
- Cingolani, F.; Casasampere, M.; Sanllehí, P.; Casas, J.; Bujons, J.; Fabrias, G. Inhibition of dihydroceramide desaturase activity by the sphingosine kinase inhibitor SKI II. J. Lipid Res. 2014, 55, 1711–1720. [Google Scholar] [CrossRef]
| Anthropometric and Biochemical Parameters | UW | NW | OW | O |
|---|---|---|---|---|
| N. | 7 (43.7 %) | 30 (44.1 %) | 13 (65 %) | 9 (50%) |
| Age | 11 (8.5/11) | 9 (8/11) | 11 (10/11) | 9 (8/9) |
| Weight (Kg) | 25.2 (21.8/27.1) | 27.2 (22.6/32.6) | 45 (37.3/49.5) ***,+++ | 38.6 (37.6/42) **,++ |
| Height (Cm) | 134 (124/138.5) | 129.5 (121.7/140.7) | 145 (134/149) | 130 (124/137) |
| BMI | 14 (13.7/14.3) | 15.765 (14.9/16.9) | 20.9 (18.6/22) ***,++ | 23.9 (22.5/25.6) ***,+++ |
| Gender (M) | 4 (57,1 %) | 15 (50 %) | 7 (53,8 %) | 4 (44,4 %) |
| TC (mg/dL) | 148 (128.5/163) | 170 (166/180.5) | 159 (140/170) | 174 (148/193) |
| HDL-C (mg/dL) | 43 (41/53) | 50.5 (45/59.5) yy | 45 (40/50) | 42 (32/46) |
| TC/HDL-C | 3.1 (2.8/3.3) | 3.35 (2.9/3.7) | 3.7 (3.2/3.9) | 4.1 (3.7/4.8) *,++ |
| LDL-C (mg/dL) | 81 (74/90.5) | 99.5 (94.2/104.7) | 85 (81/100) | 101 (89/104) |
| Vit. D (ng/mL) | 14.2 (14/16.1) | 17.2 (14.6/21.2) | 16.2 (12.7/18.8) | 15.8 (14.3/18.3) |
| Glycaemia (mg/dL) | 88 (82/92) | 83.5 (79.5/87.7) | 85 (81/88) | 84 (81/88) |
| HOMA-IR | 1 (0.9/1.2) | 0.9 (0.6/1.1) | 1.7 (1.1/1.7) + | 1.5 (1.3/2) ++ |
| Insulin (µU/mL) | 5.1 (3.9/5.6) | 4.4 (3/5.6) | 7.7 (5.5/8.7) + | 7.2 (6.4/9.3) ++ |
| TG (mg/dL) | 83 (72.5/95.5) | 97 (78.2/113.7) | 90 (86/122) | 129 (97/143) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barbacini, P.; Casas, J.; Torretta, E.; Capitanio, D.; Maccallini, G.; Hirschler, V.; Gelfi, C. Regulation of Serum Sphingolipids in Andean Children Born and Living at High Altitude (3775 m). Int. J. Mol. Sci. 2019, 20, 2835. https://doi.org/10.3390/ijms20112835
Barbacini P, Casas J, Torretta E, Capitanio D, Maccallini G, Hirschler V, Gelfi C. Regulation of Serum Sphingolipids in Andean Children Born and Living at High Altitude (3775 m). International Journal of Molecular Sciences. 2019; 20(11):2835. https://doi.org/10.3390/ijms20112835
Chicago/Turabian StyleBarbacini, Pietro, Josefina Casas, Enrica Torretta, Daniele Capitanio, Gustavo Maccallini, Valeria Hirschler, and Cecilia Gelfi. 2019. "Regulation of Serum Sphingolipids in Andean Children Born and Living at High Altitude (3775 m)" International Journal of Molecular Sciences 20, no. 11: 2835. https://doi.org/10.3390/ijms20112835
APA StyleBarbacini, P., Casas, J., Torretta, E., Capitanio, D., Maccallini, G., Hirschler, V., & Gelfi, C. (2019). Regulation of Serum Sphingolipids in Andean Children Born and Living at High Altitude (3775 m). International Journal of Molecular Sciences, 20(11), 2835. https://doi.org/10.3390/ijms20112835






