Gene Expression Indicates Altered Immune Modulation and Signaling Pathway Activation in Ovarian Cancer Patients Resistant to Topotecan
Abstract
1. Introduction
2. Results
2.1. Transcriptomic Database for Biomarker Selection
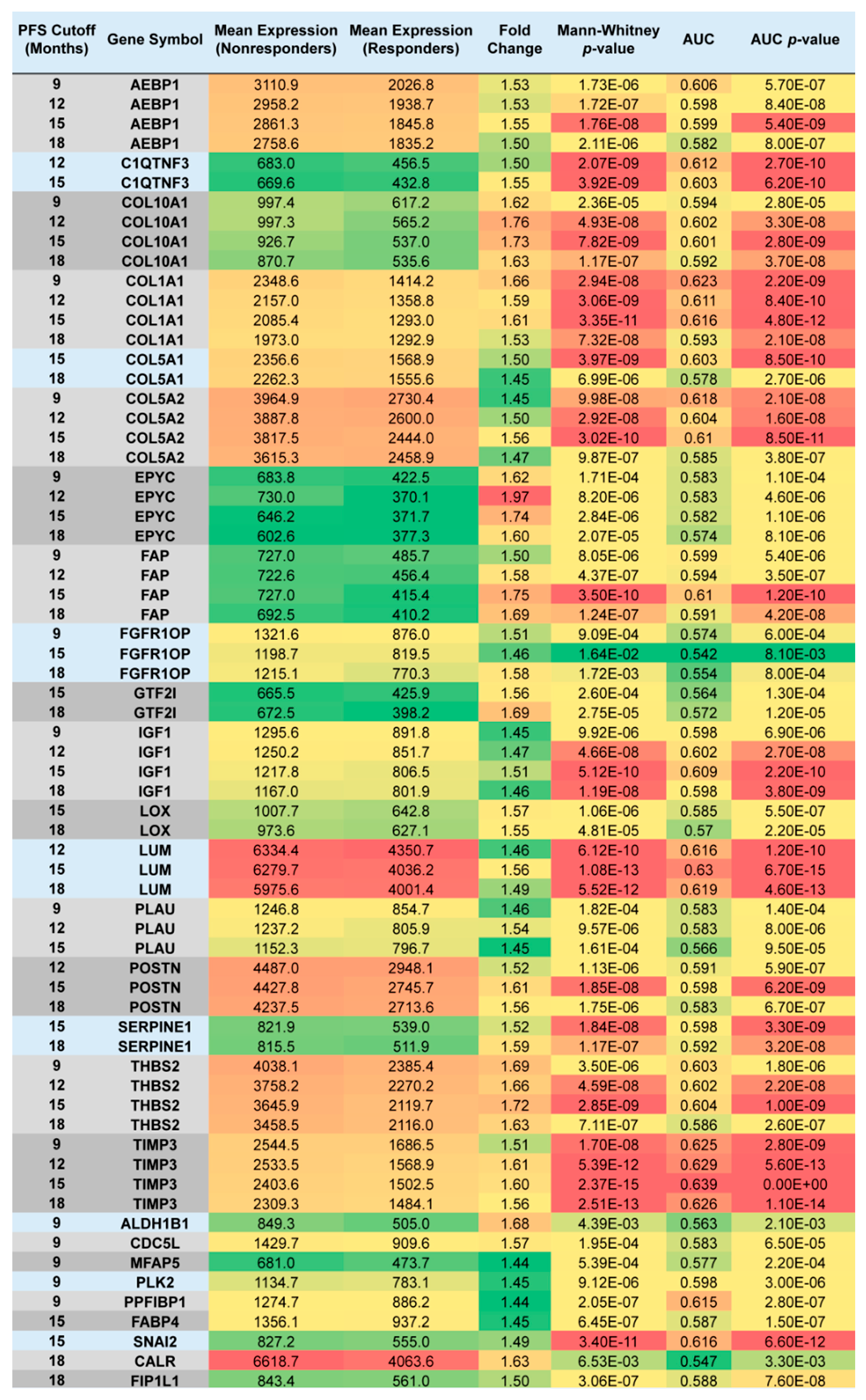
2.2. Upregulated Genes among Nonresponders Treated with Chemotherapy Excluding Topotecan
2.3. Topotecan-Treated EOC Population
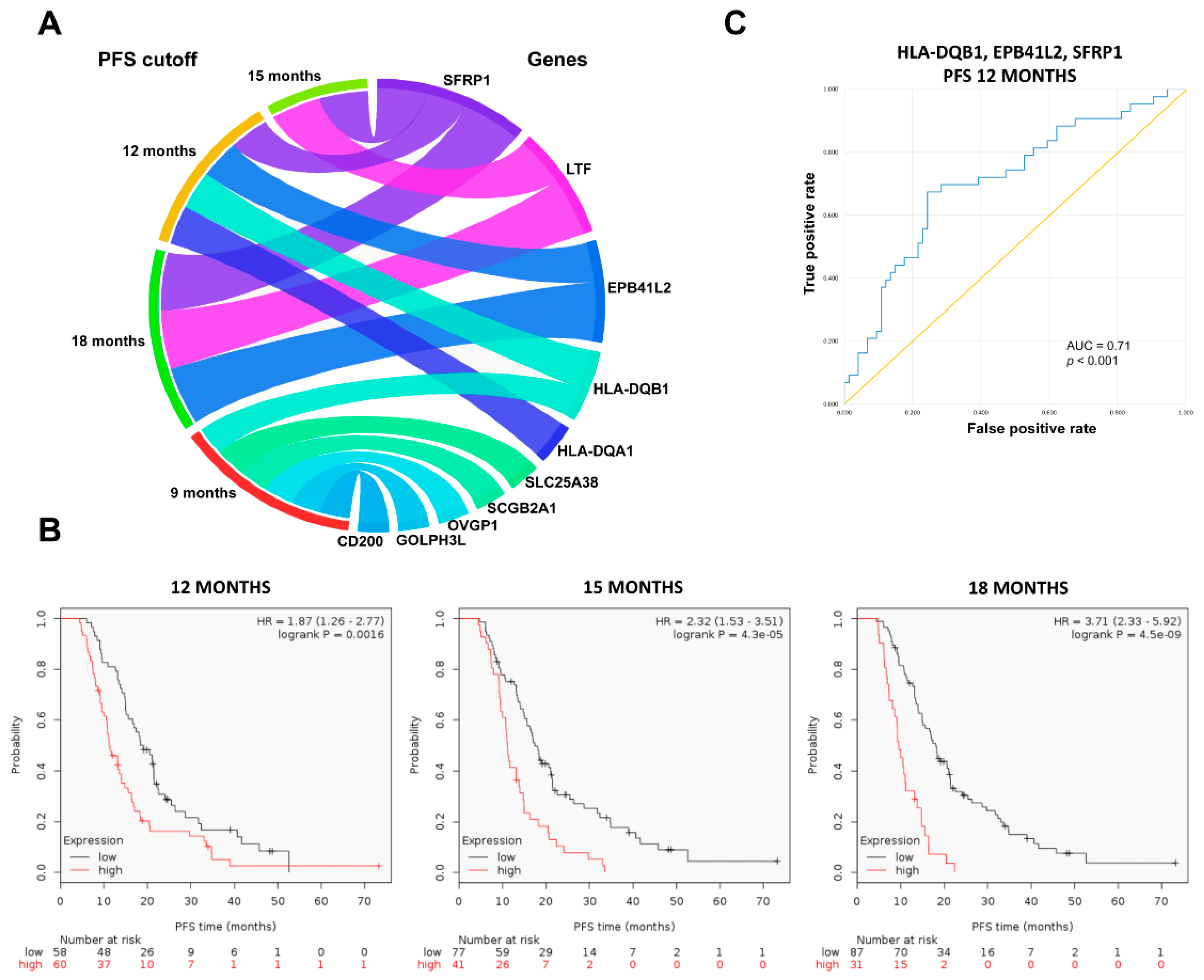
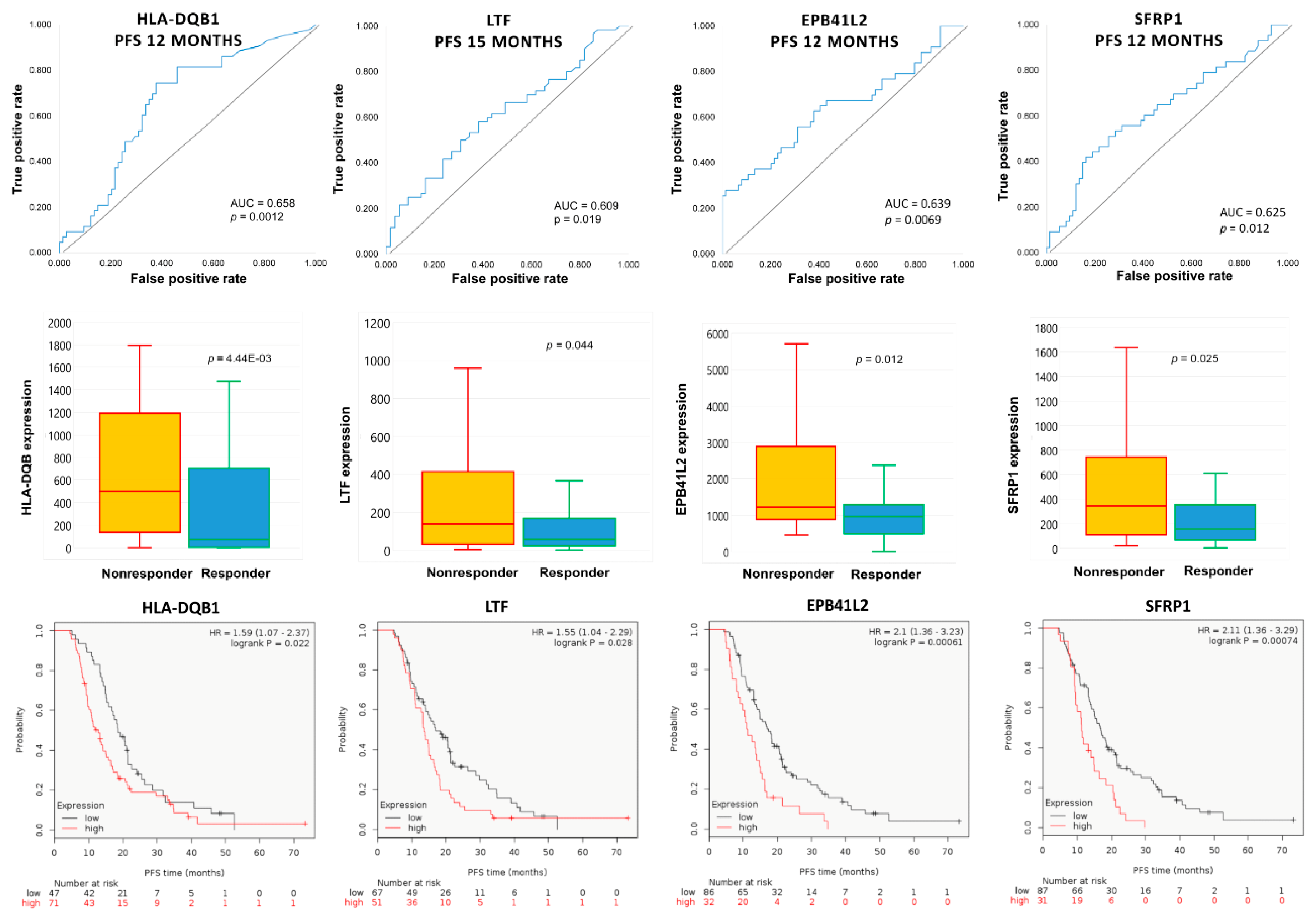
2.4. Upregulated Genes in Topotecan-Treated EOC Population
3. Discussion
4. Methods
4.1. Database Setup
4.2. Clinical Data
4.3. Gene Selecting Algorithm
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Torre, L.A.; Trabert, B.; DeSantis, C.E.; Miller, K.D.; Samimi, G.; Runowicz, C.D.; Gaudet, M.M.; Jemal, A.; Siegel, R.L. Ovarian cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 284–296. [Google Scholar] [CrossRef] [PubMed]
- Menyhart, O.; Fekete, J.T.; Gyorffy, B. Demographic shift disproportionately increases cancer burden in an aging nation: Current and expected incidence and mortality in hungary up to 2030. Clin. Epidemiol. 2018, 10, 1093–1108. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2017. CA Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Barnholtz-Sloan, J.S.; Schwartz, A.G.; Qureshi, F.; Jacques, S.; Malone, J.; Munkarah, A.R. Ovarian cancer: Changes in patterns at diagnosis and relative survival over the last three decades. Am. J. Obstet. Gynecol. 2003, 189, 1120–1127. [Google Scholar] [CrossRef]
- Friedlander, M.L.; Stockler, M.R.; Butow, P.; King, M.T.; McAlpine, J.; Tinker, A.; Ledermann, J.A. Clinical trials of palliative chemotherapy in platinum-resistant or -refractory ovarian cancer: Time to think differently? J. Clin. Oncol. 2013, 31, 2362. [Google Scholar] [CrossRef] [PubMed]
- Wright, A.A.; Bohlke, K.; Armstrong, D.K.; Bookman, M.A.; Cliby, W.A.; Coleman, R.L.; Dizon, D.S.; Kash, J.J.; Meyer, L.A.; Moore, K.N.; et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: Society of gynecologic oncology and american society of clinical oncology clinical practice guideline. Gynecol. Oncol. 2016, 143, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Webb, P.M.; Jordan, S.J. Epidemiology of epithelial ovarian cancer. Best Pract. Res. Clin. Obstet. Gynaecol. 2017, 41, 3–14. [Google Scholar] [CrossRef]
- Davis, A.; Tinker, A.V.; Friedlander, M. “Platinum resistant” ovarian cancer: What is it, who to treat and how to measure benefit? Gynecol. Oncol. 2014, 133, 624–631. [Google Scholar] [CrossRef]
- Sehouli, J.; Stengel, D.; Oskay-Oezcelik, G.; Zeimet, A.G.; Sommer, H.; Klare, P.; Stauch, M.; Paulenz, A.; Camara, O.; Keil, E.; et al. Nonplatinum topotecan combinations versus topotecan alone for recurrent ovarian cancer: Results of a phase III study of the north-eastern german society of gynecological oncology ovarian cancer study group. J. Clin. Oncol. 2008, 26, 3176–3182. [Google Scholar] [CrossRef]
- Ferrandina, G.; Ludovisi, M.; Lorusso, D.; Pignata, S.; Breda, E.; Savarese, A.; Del Medico, P.; Scaltriti, L.; Katsaros, D.; Priolo, D.; et al. Phase iii trial of gemcitabine compared with pegylated liposomal doxorubicin in progressive or recurrent ovarian cancer. J. Clin. Oncol. 2008, 26, 890–896. [Google Scholar] [CrossRef]
- Peng, L.H.; Chen, X.Y.; Wu, T.X. Topotecan for ovarian cancer. Cochrane Database Syst. Rev. 2008, Cd005589. [Google Scholar]
- Mutch, D.G.; Orlando, M.; Goss, T.; Teneriello, M.G.; Gordon, A.N.; McMeekin, S.D.; Wang, Y.; Scribner, D.R., Jr.; Marciniack, M.; Naumann, R.W.; et al. Randomized phase iii trial of gemcitabine compared with pegylated liposomal doxorubicin in patients with platinum-resistant ovarian cancer. J. Clin. Oncol. 2007, 25, 2811–2818. [Google Scholar] [CrossRef] [PubMed]
- Staker, B.L.; Hjerrild, K.; Feese, M.D.; Behnke, C.A.; Burgin, A.B., Jr.; Stewart, L. The mechanism of topoisomerase i poisoning by a camptothecin analog. Proc. Natl. Acad. Sci. USA 2002, 99, 15387–15392. [Google Scholar] [CrossRef] [PubMed]
- Herzog, T.J. Update on the role of topotecan in the treatment of recurrent ovarian cancer. Oncologist 2002, 7, S3–S10. [Google Scholar] [CrossRef]
- Rubin, E.H.; Li, T.K.; Duann, P.; Liu, L.F. Cellular resistance to topoisomerase poisons. Cancer Treat. Res. 1996, 87, 243–260. [Google Scholar] [PubMed]
- Januchowski, R.; Swierczewska, M.; Sterzynska, K.; Wojtowicz, K.; Nowicki, M.; Zabel, M. Increased expression of several collagen genes is associated with drug resistance in ovarian cancer cell lines. J. Cancer 2016, 7, 1295–1310. [Google Scholar] [CrossRef]
- Januchowski, R.; Sterzyńska, K.; Zaorska, K.; Sosińska, P.; Klejewski, A.; Brązert, M.; Nowicki, M.; Zabel, M. Analysis of mdr genes expression and cross-resistance in eight drug resistant ovarian cancer cell lines. J. Ovarian Res. 2016, 9, 65. [Google Scholar] [CrossRef]
- Denkert, C.; Budczies, J.; Darb-Esfahani, S.; Gyorffy, B.; Sehouli, J.; Konsgen, D.; Zeillinger, R.; Weichert, W.; Noske, A.; Buckendahl, A.C.; et al. A prognostic gene expression index in ovarian cancer—Validation across different independent data sets. J. Pathol. 2009, 218, 273–280. [Google Scholar] [CrossRef]
- Ahmed, A.A.; Mills, A.D.; Ibrahim, A.E.K.; Temple, J.; Blenkiron, C.; Vias, M.; Massie, C.E.; Iyer, N.G.; McGeoch, A.; Crawford, R.; et al. The extracellular matrix protein tgfbi induces microtubule stabilization and sensitizes ovarian cancers to paclitaxel. Cancer Cell 2007, 12, 514–527. [Google Scholar] [CrossRef]
- Bonome, T.; Levine, D.A.; Shih, J.; Randonovich, M.; Pise-Masison, C.A.; Bogomolniy, F.; Ozbun, L.; Brady, J.; Barrett, J.C.; Boyd, J.; et al. A gene signature predicting for survival in suboptimally debulked patients with ovarian cancer. Cancer Res. 2008, 68, 5478–5486. [Google Scholar] [CrossRef]
- Ferriss, J.S.; Kim, Y.; Duska, L.; Birrer, M.; Levine, D.A.; Moskaluk, C.; Theodorescu, D.; Lee, J.K. Multi-gene expression predictors of single drug responses to adjuvant chemotherapy in ovarian carcinoma: Predicting platinum resistance. PLoS ONE 2012, 7, e30550. [Google Scholar] [CrossRef]
- Yoshihara, K.; Tsunoda, T.; Shigemizu, D.; Fujiwara, H.; Hatae, M.; Fujiwara, H.; Masuzaki, H.; Katabuchi, H.; Kawakami, Y.; Okamoto, A.; et al. High-risk ovarian cancer based on 126-gene expression signature is uniquely characterized by downregulation of antigen presentation pathway. Clin. Cancer Res. 2012, 18, 1374–1385. [Google Scholar] [CrossRef]
- Koti, M.; Gooding, R.J.; Nuin, P.; Haslehurst, A.; Crane, C.; Weberpals, J.; Childs, T.; Bryson, P.; Dharsee, M.; Evans, K.; et al. Identification of the IGF1/PI3K/NF κB/ERK gene signalling networks associated with chemotherapy resistance and treatment response in high-grade serous epithelial ovarian cancer. BMC Cancer 2013, 13, 549. [Google Scholar] [CrossRef] [PubMed]
- Lisowska, K.M.; Olbryt, M.; Dudaladava, V.; Pamuła-Piłat, J.; Kujawa, K.; Grzybowska, E.; Jarząb, M.; Student, S.; Rzepecka, I.K.; Jarząb, B.; et al. Gene expression analysis in ovarian cancer—Faults and hints from DNA microarray study. Front. Oncol. 2014, 4, 6. [Google Scholar] [CrossRef] [PubMed]
- Uehara, Y.; Oda, K.; Ikeda, Y.; Koso, T.; Tsuji, S.; Yamamoto, S.; Asada, K.; Sone, K.; Kurikawa, R.; Makii, C.; et al. Correction: Integrated copy number and expression analysis identifies profiles of whole-arm chromosomal alterations and subgroups with favorable outcome in ovarian clear cell carcinomas. PLoS ONE 2015, 10, e0132751. [Google Scholar] [CrossRef] [PubMed]
- Tothill, R.W.; Tinker, A.V.; George, J.; Brown, R.; Fox, S.B.; Lade, S.; Johnson, D.S.; Trivett, M.K.; Etemadmoghadam, D.; Locandro, B.; et al. Novel molecular subtypes of serous and endometrioid ovarian cancer linked to clinical outcome. Clin. Cancer Res. 2008, 14, 5198–5208. [Google Scholar] [CrossRef]
- Integrated genomic analyses of ovarian carcinoma. Nature 2011, 474, 609–615. [CrossRef]
- Bussard, K.M.; Mutkus, L.; Stumpf, K.; Gomez-Manzano, C.; Marini, F.C. Tumor-associated stromal cells as key contributors to the tumor microenvironment. Breast Cancer Res. 2016, 18, 84. [Google Scholar] [CrossRef]
- Hanahan, D.; Coussens, L.M. Accessories to the crime: Functions of cells recruited to the tumor microenvironment. Cancer Cell 2012, 21, 309–322. [Google Scholar] [CrossRef] [PubMed]
- Deying, W.; Feng, G.; Shumei, L.; Hui, Z.; Ming, L.; Hongqing, W. CAF-derived HGF promotes cell proliferation and drug resistance by up-regulating the c-Met/PI3K/Akt and GRP78 signalling in ovarian cancer cells. Biosci. Rep. 2017, 37, BSR20160470. [Google Scholar] [CrossRef]
- Li, X.Y.; Hu, S.Q.; Xiao, L. The cancer-associated fibroblasts and drug resistance. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 2112–2119. [Google Scholar]
- Kraman, M.; Bambrough, P.J.; Arnold, J.N.; Roberts, E.W.; Magiera, L.; Jones, J.O.; Gopinathan, A.; Tuveson, D.A.; Fearon, D.T. Suppression of antitumor immunity by stromal cells expressing fibroblast activation protein-α. Science 2010, 330, 827–830. [Google Scholar] [CrossRef]
- Lloyd, K.L.; Cree, I.A.; Savage, R.S. Prediction of resistance to chemotherapy in ovarian cancer: A systematic review. BMC Cancer 2015, 15, 117. [Google Scholar] [CrossRef] [PubMed]
- Matondo, A.; Jo, Y.H.; Shahid, M.; Choi, T.G.; Nguyen, M.N.; Nguyen, N.N.Y.; Akter, S.; Kang, I.; Ha, J.; Maeng, C.H.; et al. The prognostic 97 chemoresponse gene signature in ovarian cancer. Sci. Rep. 2017, 7, 9689. [Google Scholar] [CrossRef]
- Cheon, D.J.; Tong, Y.; Sim, M.S.; Dering, J.; Berel, D.; Cui, X.; Lester, J.; Beach, J.A.; Tighiouart, M.; Walts, A.E.; et al. A collagen-remodeling gene signature regulated by TGF-β signaling is associated with metastasis and poor survival in serous ovarian cancer. Clin. Cancer Res. 2014, 20, 711–723. [Google Scholar] [CrossRef]
- Anderson, M.S.; Miller, J. Invariant chain can function as a chaperone protein for class II major histocompatibility complex molecules. Proc. Natl. Acad. Sci. USA 1992, 89, 2282–2286. [Google Scholar] [CrossRef]
- van den Elsen, P.J.; Holling, T.M.; Kuipers, H.F.; van der Stoep, N. Transcriptional regulation of antigen presentation. Curr. Opin. Immunol. 2004, 16, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Van den Hoorn, T.; Paul, P.; Jongsma, M.L.; Neefjes, J. Routes to manipulate MHC class II antigen presentation. Curr. Opin. Immunol. 2011, 23, 88–95. [Google Scholar] [CrossRef]
- Esteban, F.; Ruiz-Cabello, F.; Concha, A.; Perez-Ayala, M.; Sanchez-Rozas, J.A.; Garrido, F. HLA-DR expression is associated with excellent prognosis in squamous cell carcinoma of the larynx. Clin. Exp. Metastasis 1990, 8, 319–328. [Google Scholar] [CrossRef]
- Kunihiro, M.; Tanaka, S.; Haruma, K.; Yoshihara, M.; Sumii, K.; Kajiyama, G.; Shimamoto, F. Combined expression of HLA-DR antigen and proliferating cell nuclear antigen correlate with colorectal cancer prognosis. Oncology 1998, 55, 326–333. [Google Scholar] [CrossRef]
- Moretti, S.; Pinzi, C.; Berti, E.; Spallanzani, A.; Chiarugi, A.; Boddi, V.; Reali, U.M.; Giannotti, B. In situ expression of transforming growth factor beta is associated with melanoma progression and correlates with ki67, HLA-DR and β3 integrin expression. Melanoma Res. 1997, 7, 313–321. [Google Scholar] [CrossRef]
- Pollack, M.S.; Livingston, P.O. Hla and dr antigen frequencies in melanoma patients: Possible relation to disease prognosis. Tissue Antigens 1985, 26, 262–265. [Google Scholar] [CrossRef]
- Zhou, Q.; Xu, J.; Zhao, J.; Zhang, S.; Pan, W. Downregulation of CD99 and upregulation of human leukocyte antigen class II promote tumor aggravation and poor survival in patients with osteosarcomas. Oncotargets Ther. 2014, 7, 477–484. [Google Scholar]
- Sato, E.; Olson, S.H.; Ahn, J.; Bundy, B.; Nishikawa, H.; Qian, F.; Jungbluth, A.A.; Frosina, D.; Gnjatic, S.; Ambrosone, C.; et al. Intraepithelial CD8+ tumor-infiltrating lymphocytes and a high CD8+/regulatory T cell ratio are associated with favorable prognosis in ovarian cancer. Proc. Natl. Acad. Sci. USA 2005, 102, 18538–18543. [Google Scholar] [CrossRef]
- Koskinen, W.J.; Partanen, J.; Vaheri, A.; Aaltonen, L.M. HLA-DRB1, -DQB1 alleles in head and neck carcinoma patients. Tissue Antigens 2006, 67, 237–240. [Google Scholar] [CrossRef]
- Kubler, K.; Arndt, P.F.; Wardelmann, E.; Krebs, D.; Kuhn, W.; van der Ven, K. HLA-class II haplotype associations with ovarian cancer. Int. J. Cancer 2006, 119, 2980–2985. [Google Scholar] [CrossRef]
- Kubler, K.; Arndt, P.F.; Wardelmann, E.; Landwehr, C.; Krebs, D.; Kuhn, W.; van der Ven, K. Genetic alterations of HLA-class II in ovarian cancer. Int. J. Cancer 2008, 123, 1350–1356. [Google Scholar] [CrossRef]
- Ward, P.P.; Uribe-Luna, S.; Conneely, O.M. Lactoferrin and host defense. Biochem. Cell Biol. 2002, 80, 95–102. [Google Scholar] [CrossRef]
- Hu, L.; Hu, X.; Long, K.; Gao, C.; Dong, H.L.; Zhong, Q.; Gao, X.M.; Gong, F.Y. Extraordinarily potent proinflammatory properties of lactoferrin-containing immunocomplexes against human monocytes and macrophages. Sci. Rep. 2017, 7, 4230. [Google Scholar] [CrossRef]
- Gao, C.H.; Dong, H.L.; Tai, L.; Gao, X.M. Lactoferrin-containing immunocomplexes drive the conversion of human macrophages from M2- into M1-like phenotype. Front. Immunol. 2018, 9, 37. [Google Scholar] [CrossRef]
- Farnaud, S.; Evans, R.W. Lactoferrin—A multifunctional protein with antimicrobial properties. Mol. Immunol. 2003, 40, 395–405. [Google Scholar] [CrossRef]
- Miller, L.D.; Coffman, L.G.; Chou, J.W.; Black, M.A.; Bergh, J.; D’Agostino, R., Jr.; Torti, S.V.; Torti, F.M. An iron regulatory gene signature predicts outcome in breast cancer. Cancer Res. 2011, 71, 6728–6737. [Google Scholar] [CrossRef]
- Gibbons, J.A.; Kanwar, R.K.; Kanwar, J.R. Lactoferrin and cancer in different cancer models. Front. Biosci. 2011, 3, 1080–1088. [Google Scholar] [CrossRef]
- Leyns, L.; Bouwmeester, T.; Kim, S.H.; Piccolo, S.; de Robertis, E.M. Frzb-1 is a secreted antagonist of wnt signaling expressed in the spemann organizer. Cell 1997, 88, 747–756. [Google Scholar] [CrossRef]
- Surana, R.; Sikka, S.; Cai, W.; Shin, E.M.; Warrier, S.R.; Tan, H.J.; Arfuso, F.; Fox, S.A.; Dharmarajan, A.M.; Kumar, A.P. Secreted frizzled related proteins: Implications in cancers. Biochim. Et Biophys. Acta 2014, 1845, 53–65. [Google Scholar] [CrossRef]
- Vincent, K.M.; Postovit, L.-M. A pan-cancer analysis of secreted frizzled-related proteins: Re-examining their proposed tumour suppressive function. Sci. Rep. 2017, 7, 42719. [Google Scholar] [CrossRef]
- Kardum, V.; Karin, V.; Glibo, M.; Skrtic, A.; Martic, T.N.; Ibisevic, N.; Skenderi, F.; Vranic, S.; Serman, L. Methylation-associated silencing of SFRP1 gene in high-grade serous ovarian carcinomas. Ann. Diagn. Pathol. 2017, 31, 45–49. [Google Scholar] [CrossRef]
- Davis, M.; Li, J.; Knight, E.; Eldridge, S.R.; Daniels, K.K.; Bushel, P.R. Toxicogenomics profiling of bone marrow from rats treated with topotecan in combination with oxaliplatin: A mechanistic strategy to inform combination toxicity. Front. Genet. 2015, 6, 14. [Google Scholar] [CrossRef]
- Lehmann, B.D.; Bauer, J.A.; Chen, X.; Sanders, M.E.; Chakravarthy, A.B.; Shyr, Y.; Pietenpol, J.A. Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J. Clin. Investig. 2011, 121, 2750–2767. [Google Scholar] [CrossRef]
- Qu, Y.; Ray, P.S.; Li, J.; Cai, Q.; Bagaria, S.P.; Moran, C.; Sim, M.S.; Zhang, J.; Turner, R.R.; Zhu, Z.; et al. High levels of secreted frizzled-related protein 1 correlate with poor prognosis and promote tumourigenesis in gastric cancer. Eur. J. Cancer 2013, 49, 3718–3728. [Google Scholar] [CrossRef]
- Uhlen, M.; Zhang, C.; Lee, S.; Sjostedt, E.; Fagerberg, L.; Bidkhori, G.; Benfeitas, R.; Arif, M.; Liu, Z.; Edfors, F.; et al. A pathology atlas of the human cancer transcriptome. Science 2017, 357. [Google Scholar] [CrossRef]
- Gautier, L.; Møller, M.; Friis-Hansen, L.; Knudsen, S. Alternative mapping of probes to genes for affymetrix chips. BMC Bioinform. 2004, 5, 111. [Google Scholar] [CrossRef]
- Sims, A.H.; Smethurst, G.J.; Hey, Y.; Okoniewski, M.J.; Pepper, S.D.; Howell, A.; Miller, C.J.; Clarke, R.B. The removal of multiplicative, systematic bias allows integration of breast cancer gene expression datasets —improving meta-analysis and prediction of prognosis. BMC Med. Genom. 2008, 1, 42. [Google Scholar] [CrossRef]
- Carey, V. ROC: Utilities for ROC, with uarray focus. R package version 1.58.0. enhancements HRfCl. 2018. Available online: http://bioconductor.org/packages/ROC/ (accessed on 5 January 2019).
- Huang da, W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using david bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef]
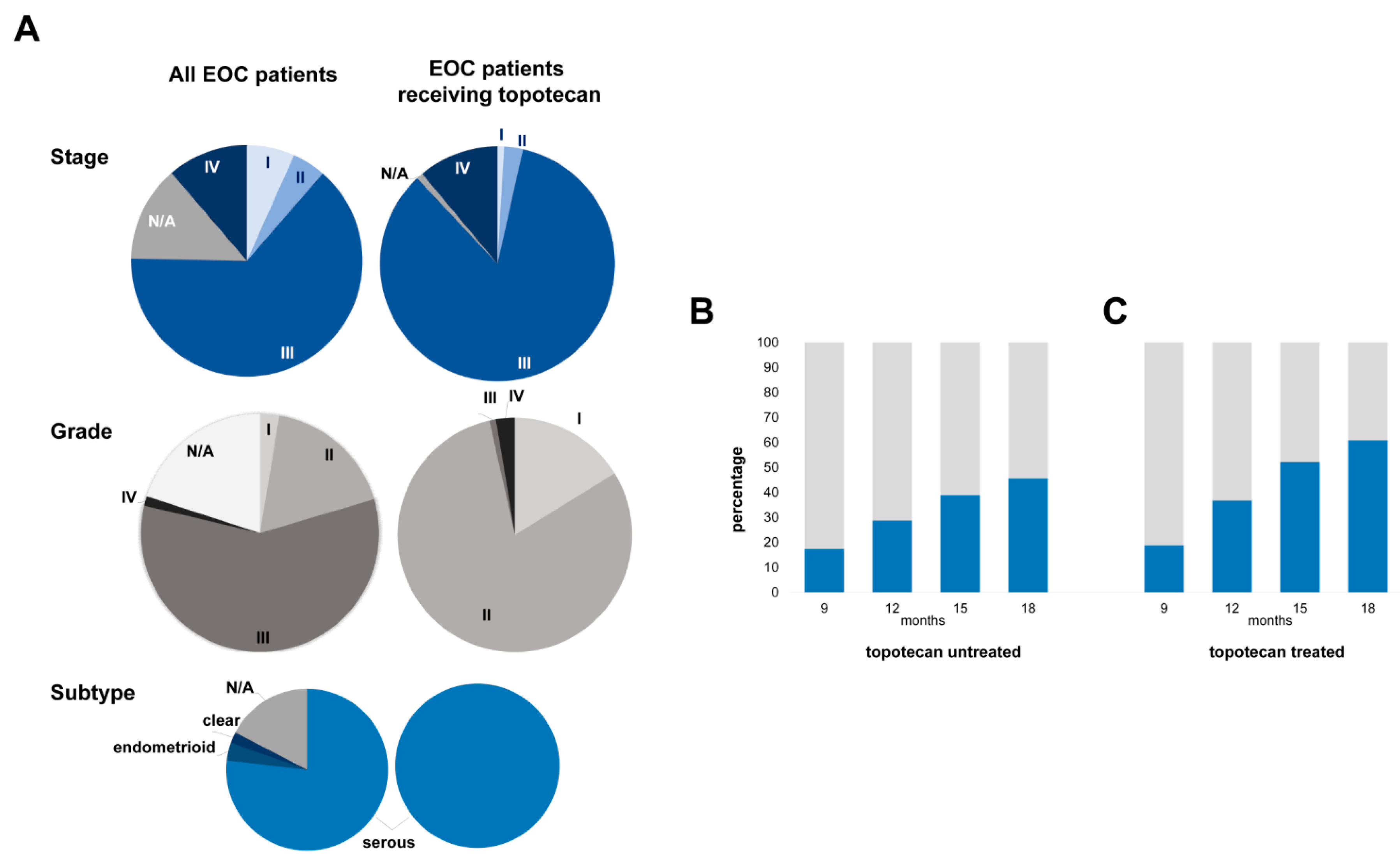

| Characteristics | All EOC Patients (%) | Topotecan-Treated EOC Patients (%) | ||
|---|---|---|---|---|
| Stage | ||||
| I | 6.7 | 0.9 | ||
| II | 4.7 | 2.6 | ||
| III | 64.0 | 84.6 | ||
| IV | 11.3 | 11.1 | ||
| N/A | 13.4 | 0.9 | ||
| Grade | ||||
| I | 2.6 | 16.2 | ||
| II | 17.8 | 80.3 | ||
| III | 58.3 | 0.9 | ||
| IV | 1.3 | 2.6 | ||
| N/A | 20.0 | |||
| Subtype | ||||
| Serous | 76.9 | 100 | ||
| Endometrioid | 3.6 | |||
| Clear | 2.2 | |||
| N/A | 17.4 | |||
| Debulking | ||||
| Optimal | 48.5 | 74.4 | ||
| Suboptimal | 32.0 | 22.2 | ||
| N/A | 19.5 | 3.4 | ||
| Progression | (median follow up 16.3 months) | 68.1 | (median follow up 14.8 months) | 88.0 |
| Treatment | ||||
| Platinum | 87.7 | 100 | ||
| Taxol | 49.8 | |||
| Combined platinum and taxol | 48.6 | 75.2 | ||
| Bevacizumab | 3.5 | 12.8 | ||
| Docetaxel | 7.4 | 30.8 | ||
| Gemcitabine | 9.1 | 62.4 | ||
| Paclitaxel | 15.9 | 25.6 | ||
| Topotecan | 8.2 | 100 |
| Term | Count | p-Value | Genes | Bonferroni |
|---|---|---|---|---|
| Extracellular matrix organization | 9 | 3.90 × 10−10 | COL5A1, COL1A1, LOX, POSTN, COL5A2, COL10A1, LUM, SERPINE1, MFAP5 | 1.31 × 10−7 |
| Collagen fibril organization | 5 | 3.58 × 10−7 | COL5A1, COL1A1, LOX, COL5A2, LUM | 1.20 × 10−4 |
| Skeletal system development | 6 | 1.93 × 10−6 | IGF1, COL1A1, POSTN, COL5A2, COL10A1, AEBP1 | 6.45 × 10−4 |
| Cellular response to fibroblast growth factor stimulus | 4 | 1.30 × 10−5 | COL1A1, POSTN, SNAI2, CDC5L | 4.35 × 10−3 |
| Collagen catabolic process | 4 | 1.29 × 10−4 | COL5A1, COL1A1, COL5A2, COL10A1 | 4.23 × 10−2 |
| PFS Cutoff (Months) | Gene Symbol | Mean Expression (Nonresponder) | Mean Expression (Responder) | Fold Change | Mann–Whitney p-Value | AUC | AUC p-Value |
|---|---|---|---|---|---|---|---|
| 9 | CD200 | 1718.6 | 1165.7 | 1.47 | 2.13 × 10−2 | 0.658 | 8.20 × 10−3 |
| 12 | EPB41L2 | 1918.2 | 1160.9 | 1.65 | 1.24 × 10−2 | 0.639 | 6.90 × 10−3 |
| 18 | EPB41L2 | 1703.6 | 1036.1 | 1.64 | 7.70 × 10−3 | 0.648 | 1.80 × 10−3 |
| 9 | GOLPH3L | 2289.1 | 1583.0 | 1.45 | 2.34 × 10−2 | 0.656 | 6.50 × 10−3 |
| 12 | HLA-DQA1 | 1353.5 | 908.2 | 1.49 | 1.94 × 10−2 | 0.63 | 8.30 × 10−3 |
| 9 | HLA-DQB1 | 1243.8 | 830.4 | 1.50 | 2.42 × 10−2 | 0.655 | 8.20 × 10−3 |
| 12 | HLA-DQB1 | 1202.6 | 737.0 | 1.63 | 4.45 × 10−3 | 0.658 | 1.20 × 10−3 |
| 15 | LTF | 1034.3 | 316.5 | 3.27 | 4.38 × 10−2 | 0.609 | 1.90 × 10−2 |
| 18 | LTF | 908.5 | 352.8 | 2.58 | 4.13 × 10−2 | 0.613 | 1.70 × 10−2 |
| 9 | OVGP1 | 1975.9 | 362.2 | 5.46 | 7.95 × 10−3 | 0.682 | 5.90 × 10−3 |
| 9 | SCGB2A1 | 11433.0 | 7001.9 | 1.63 | 3.31 × 10−2 | 0.646 | 2.00 × 10−2 |
| 12 | SFRP1 | 882.5 | 401.5 | 2.20 | 2.46 × 10−2 | 0.625 | 1.20 × 10−2 |
| 15 | SFRP1 | 727.7 | 393.8 | 1.85 | 1.49 × 10−2 | 0.632 | 5.80 × 10−3 |
| 18 | SFRP1 | 679.6 | 394.3 | 1.72 | 3.23 × 10−2 | 0.619 | 1.40 × 10−2 |
| 9 | SLC25A38 | 1838.8 | 1101.3 | 1.67 | 2.55 × 10−2 | 0.707 | 1.90 × 10−4 |
| Gene Combinations | AUC Values | p-Values |
|---|---|---|
| HLA-DQB1 + EPB41L2 | 0.679 | 0.001 |
| HLA-DQB1 + SFRP1 | 0.696 | <0.001 |
| EPB41L2 + SFRP1 | 0.675 | 0.002 |
| HLA-DQB1 + EPB41L2 + SFRP1 | 0.71 | <0.001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Menyhárt, O.; Fekete, J.T.; Győrffy, B. Gene Expression Indicates Altered Immune Modulation and Signaling Pathway Activation in Ovarian Cancer Patients Resistant to Topotecan. Int. J. Mol. Sci. 2019, 20, 2750. https://doi.org/10.3390/ijms20112750
Menyhárt O, Fekete JT, Győrffy B. Gene Expression Indicates Altered Immune Modulation and Signaling Pathway Activation in Ovarian Cancer Patients Resistant to Topotecan. International Journal of Molecular Sciences. 2019; 20(11):2750. https://doi.org/10.3390/ijms20112750
Chicago/Turabian StyleMenyhárt, Otília, János Tibor Fekete, and Balázs Győrffy. 2019. "Gene Expression Indicates Altered Immune Modulation and Signaling Pathway Activation in Ovarian Cancer Patients Resistant to Topotecan" International Journal of Molecular Sciences 20, no. 11: 2750. https://doi.org/10.3390/ijms20112750
APA StyleMenyhárt, O., Fekete, J. T., & Győrffy, B. (2019). Gene Expression Indicates Altered Immune Modulation and Signaling Pathway Activation in Ovarian Cancer Patients Resistant to Topotecan. International Journal of Molecular Sciences, 20(11), 2750. https://doi.org/10.3390/ijms20112750






