Formation of γH2AX and pATM Foci in Human Mesenchymal Stem Cells Exposed to Low Dose-Rate Gamma-Radiation
Abstract
1. Introduction
2. Results
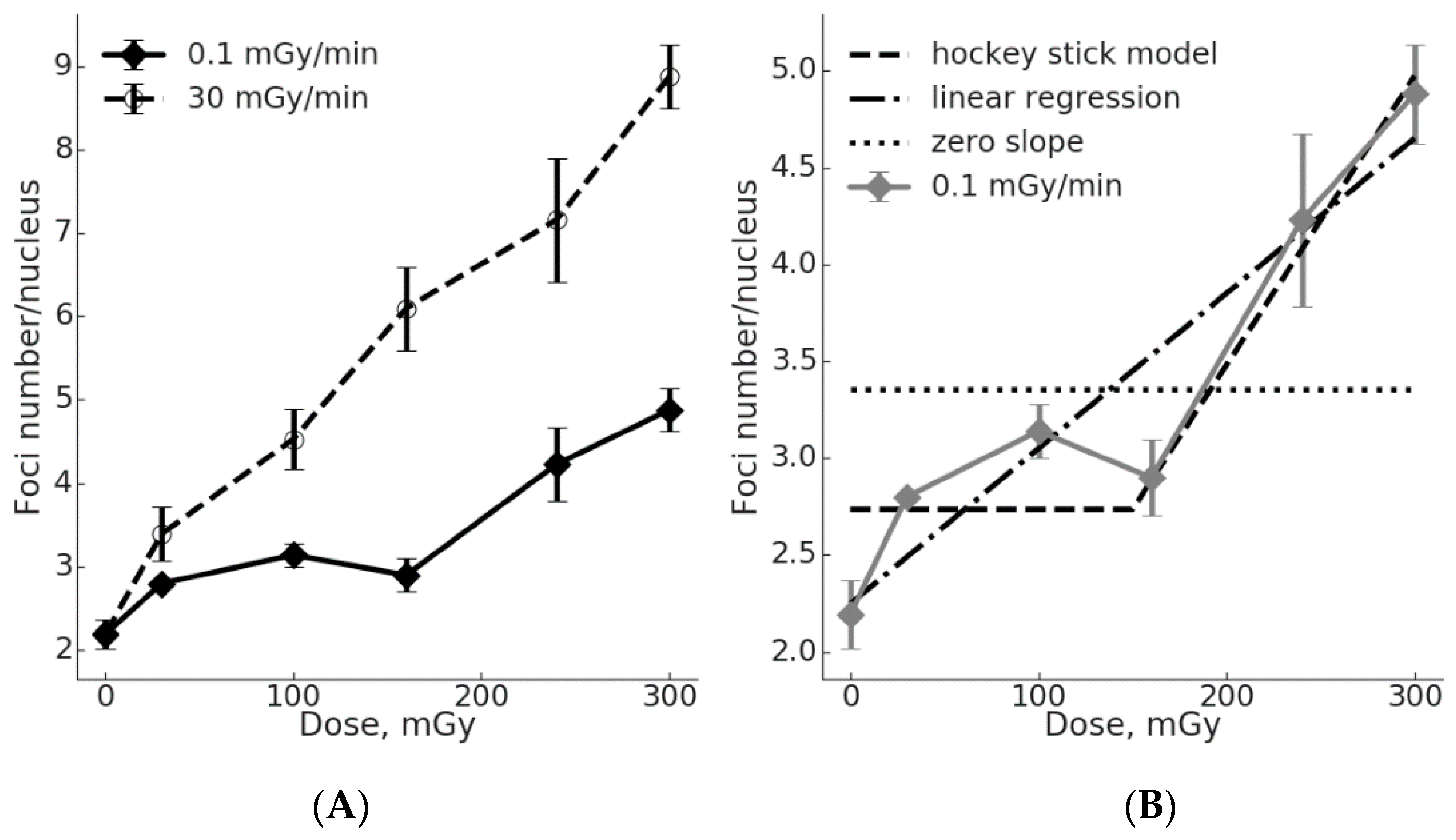
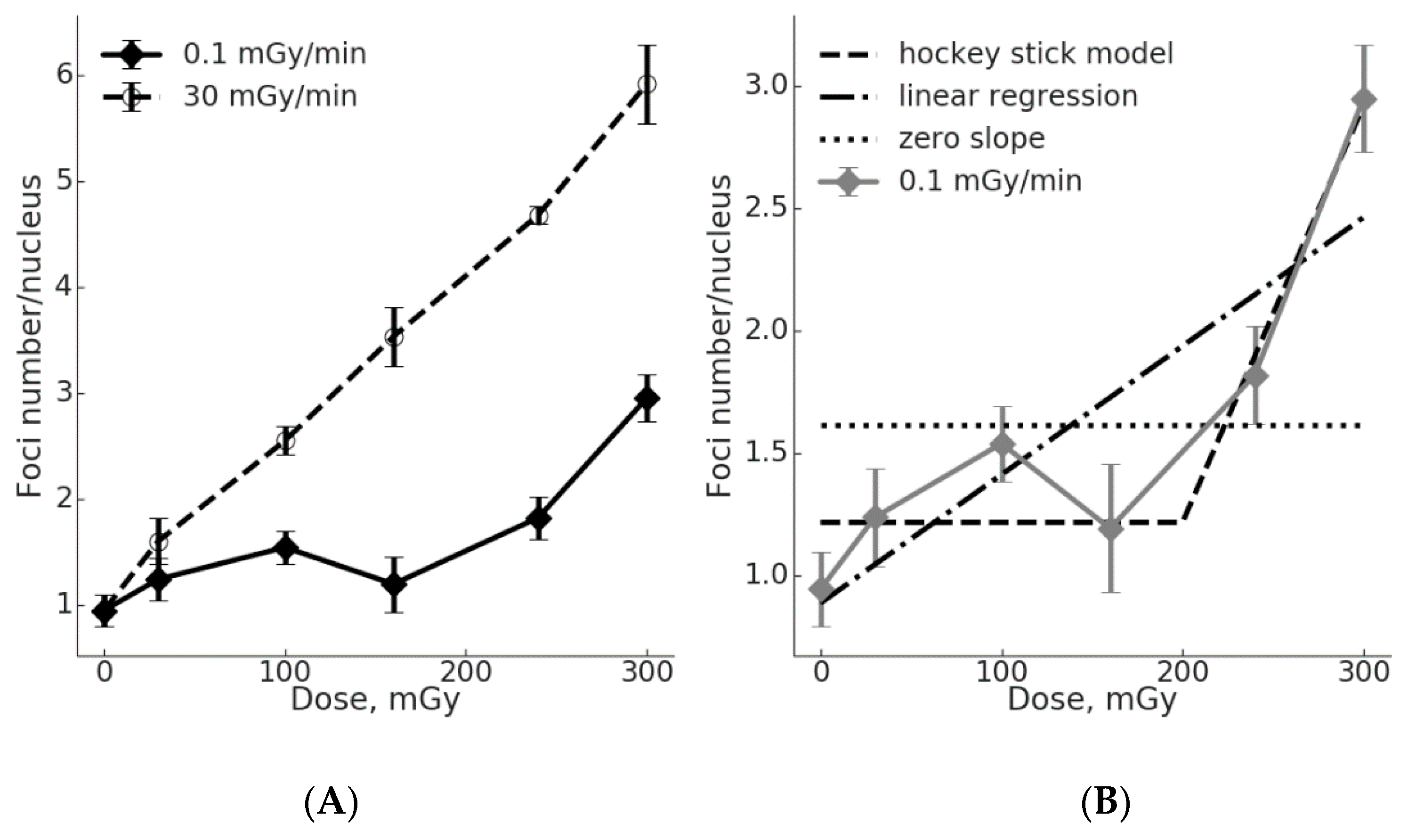
2.1. Low γH2AX Foci Yield in MSCs During Chronic Irradiation
2.2. Comparison of the γH2AX Foci Yields per Radiation Dose Unit for Acute vs. Chronic Exposure
2.3. The Threshold for ATM Activation upon Chronic Irradiation
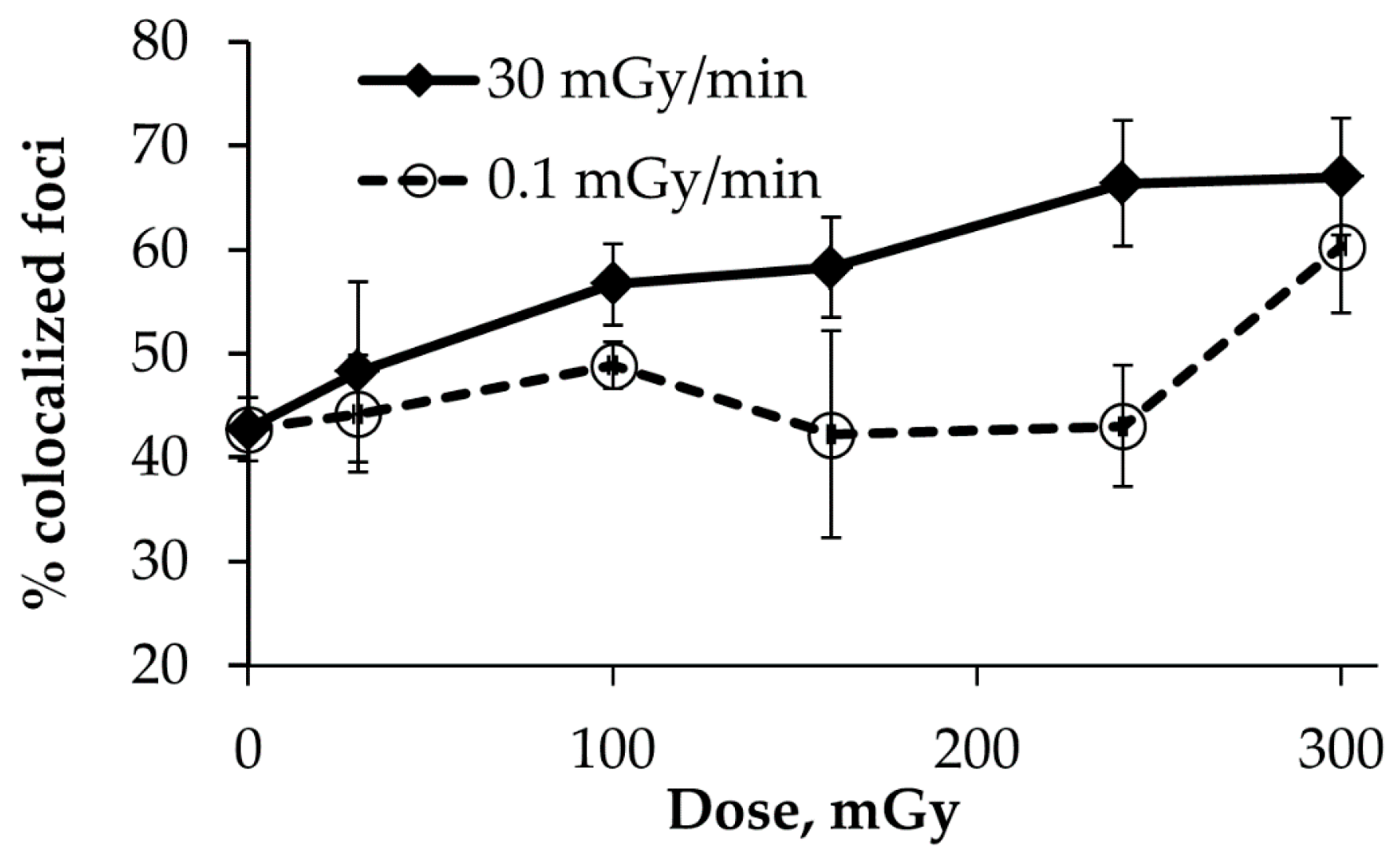
2.4. Co-localization of γH2AX and pATM Foci
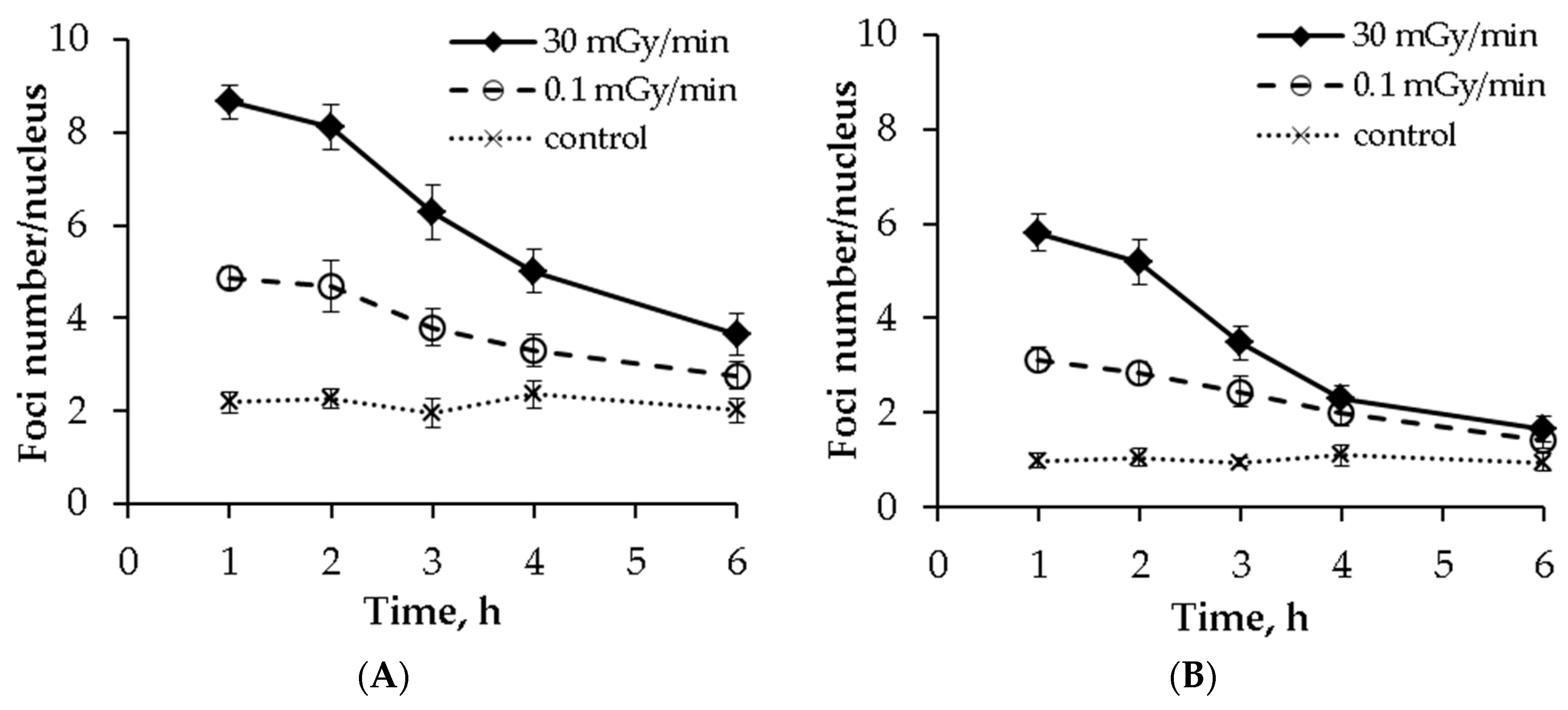
2.5. Post-irradiation Kinetics of γH2AX and pATM Foci Numbers
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. The Irradiation Sources and Dosimetry
4.3. Cell Irradiation
4.4. Foci Detection and Analysis
4.5. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Ruhm, W.; Eidemuller, M.; Kaiser, J.C. Biologically-based mechanistic models of radiation-related carcinogenesis applied to epidemiological data. Int. J. Radiat. Biol. 2017, 93, 1093–1117. [Google Scholar] [CrossRef] [PubMed]
- International Commission on Radiological Protection; Valentin, J. The 2007 Recommendations of the International Commission on Radiological Protection; Icrp publication 103; Elsevier: Oxford, UK, 2007; Volume 37, pp. 1–332. [Google Scholar]
- Mladenov, E.; Magin, S.; Soni, A.; Iliakis, G. DNA double-strand-break repair in higher eukaryotes and its role in genomic instability and cancer: Cell cycle and proliferation-dependent regulation. Semin. Cancer Biol. 2016, 37–38, 51–64. [Google Scholar] [CrossRef] [PubMed]
- Hoeijmakers, J.H. Genome maintenance mechanisms for preventing cancer. Nature 2001, 411, 366–374. [Google Scholar] [CrossRef]
- van Gent, D.C.; Hoeijmakers, J.H.; Kanaar, R. Chromosomal stability and the DNA double-stranded break connection. Nat. Rev. Genet. 2001, 2, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Kotenko, K.V.; Bushmanov, A.Y.; Ozerov, I.V.; Guryev, D.V.; Anchishkina, N.A.; Smetanina, N.M.; Arkhangelskaya, E.Y.; Vorobyeva, N.Y.; Osipov, A.N. Changes in the number of double-strand DNA breaks in chinese hamster v79 cells exposed to gamma-radiation with different dose rates. Int. J. Mol. Sci. 2013, 14, 13719–13726. [Google Scholar] [CrossRef]
- Osipov, A.N.; Grekhova, A.; Pustovalova, M.; Ozerov, I.V.; Eremin, P.; Vorobyeva, N.; Lazareva, N.; Pulin, A.; Zhavoronkov, A.; Roumiantsev, S.; et al. Activation of homologous recombination DNA repair in human skin fibroblasts continuously exposed to x-ray radiation. Oncotarget 2015, 6, 26876–26885. [Google Scholar] [CrossRef][Green Version]
- Shibata, A. Regulation of repair pathway choice at two-ended DNA double-strand breaks. Mutat. Res. 2017, 803–805, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Brooks, A.L.; Hoel, D.G.; Preston, R.J. The role of dose rate in radiation cancer risk: Evaluating the effect of dose rate at the molecular, cellular and tissue levels using key events in critical pathways following exposure to low let radiation. Int. J. Radiat. Biol. 2016, 92, 405–426. [Google Scholar] [CrossRef]
- Tsvetkova, A.; Ozerov, I.V.; Pustovalova, M.; Grekhova, A.; Eremin, P.; Vorobyeva, N.; Eremin, I.; Pulin, A.; Zorin, V.; Kopnin, P.; et al. Gammah2ax, 53bp1 and rad51 protein foci changes in mesenchymal stem cells during prolonged x-ray irradiation. Oncotarget 2017, 8, 64317–64329. [Google Scholar] [CrossRef]
- Pustovalova, M.; Astrelina capital Te, C.; Grekhova, A.; Vorobyeva, N.; Tsvetkova, A.; Blokhina, T.; Nikitina, V.; Suchkova, Y.; Usupzhanova, D.; Brunchukov, V.; et al. Residual gammah2ax foci induced by low dose x-ray radiation in bone marrow mesenchymal stem cells do not cause accelerated senescence in the progeny of irradiated cells. Aging 2017, 9, 2397–2410. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Li, P.; Gong, Z.; Shultz, L.D.; Ren, G. Mesenchymal stem cells: From regeneration to cancer. Pharmacol. Ther. 2019. [Google Scholar] [CrossRef]
- Papaccio, F.; Paino, F.; Regad, T.; Papaccio, G.; Desiderio, V.; Tirino, V. Concise review: Cancer cells, cancer stem cells, and mesenchymal stem cells: Influence in cancer development. Stem Cells Transl. Med. 2017, 6, 2115–2125. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Wang, G.; Chen, R.; Hua, Y.; Cai, Z. Mesenchymal stem cells in the osteosarcoma microenvironment: Their biological properties, influence on tumor growth, and therapeutic implications. Stem Cell Res. Ther. 2018, 9, 22. [Google Scholar] [CrossRef]
- La Noce, M.; Paino, F.; Mele, L.; Papaccio, G.; Regad, T.; Lombardi, A.; Papaccio, F.; Desiderio, V.; Tirino, V. Hdac2 depletion promotes osteosarcoma’s stemness both in vitro and in vivo: A study on a putative new target for cscs directed therapy. J. Exp. Clin. Cancer Res. CR 2018, 37, 296. [Google Scholar] [CrossRef]
- Yao, D.; Dai, C.; Peng, S. Mechanism of the mesenchymal-epithelial transition and its relationship with metastatic tumor formation. Mol. Cancer Res. MCR 2011, 9, 1608–1620. [Google Scholar] [CrossRef]
- Xiang, C.; Chen, J.; Fu, P. Hgf/met signaling in cancer invasion: The impact on cytoskeleton remodeling. Cancers 2017, 9, 44. [Google Scholar] [CrossRef]
- Della Corte, C.M.; Fasano, M.; Papaccio, F.; Ciardiello, F.; Morgillo, F. Role of hgf-met signaling in primary and acquired resistance to targeted therapies in cancer. Biomedicines 2014, 2, 345–358. [Google Scholar] [CrossRef] [PubMed]
- Andrzejewska, A.; Lukomska, B.; Janowski, M. Mesenchymal Stem Cells: From Roots to Boost. Stem Cells. in press.
- Gomes, J.P.A.; Assoni, A.F.; Pelatti, M.; Coatti, G.; Okamoto, O.K.; Zatz, M. Deepening a simple question: Can mscs be used to treat cancer? Anticancer Res. 2017, 37, 4747–4758. [Google Scholar] [PubMed]
- Li, J.H.; Fan, W.S.; Wang, M.M.; Wang, Y.H.; Ren, Z.G. Effects of mesenchymal stem cells on solid tumor metastasis in experimental cancer models: A systematic review and meta-analysis. J. Transl. Med. 2018, 16, 113. [Google Scholar] [CrossRef] [PubMed]
- Manda, K.; Kavanagh, J.N.; Buttler, D.; Prise, K.M.; Hildebrandt, G. Low dose effects of ionizing radiation on normal tissue stem cells. Mutat. Res. Rev. Mutat. Res. 2014, 761, 6–14. [Google Scholar] [CrossRef]
- Rogakou, E.P.; Boon, C.; Redon, C.; Bonner, W.M. Megabase chromatin domains involved in DNA double-strand breaks in vivo. J. Cell Biol. 1999, 146, 905–916. [Google Scholar] [CrossRef] [PubMed]
- Stiff, T.; O’Driscoll, M.; Rief, N.; Iwabuchi, K.; Lobrich, M.; Jeggo, P.A. Atm and DNA-pk function redundantly to phosphorylate h2ax after exposure to ionizing radiation. Cancer Res. 2004, 64, 2390–2396. [Google Scholar] [CrossRef]
- Burma, S.; Chen, B.P.; Murphy, M.; Kurimasa, A.; Chen, D.J. Atm phosphorylates histone h2ax in response to DNA double-strand breaks. J. Biol. Chem. 2001, 276, 42462–42467. [Google Scholar] [CrossRef]
- Bakkenist, C.J.; Kastan, M.B. DNA damage activates atm through intermolecular autophosphorylation and dimer dissociation. Nature 2003, 421, 499–506. [Google Scholar] [CrossRef]
- Flegal, M.; Blimkie, M.S.; Wyatt, H.; Bugden, M.; Surette, J.; Klokov, D. Measuring DNA damage and repair in mouse splenocytes after chronic in vivo exposure to very low doses of beta- and gamma-radiation. J. Vis. Exp. 2015, 101, e52912. [Google Scholar] [CrossRef] [PubMed]
- Blimkie, M.S.J.; Fung, L.C.W.; Petoukhov, E.S.; Girard, C.; Klokov, D. Repair of DNA double-strand breaks is not modulated by low-dose gamma radiation in c57bl/6j mice. Radiat. Res. 2014, 181, 548–559. [Google Scholar] [CrossRef]
- Osipov, A.N.; Pustovalova, M.; Grekhova, A.; Eremin, P.; Vorobyova, N.; Pulin, A.; Zhavoronkov, A.; Roumiantsev, S.; Klokov, D.Y.; Eremin, I. Low doses of x-rays induce prolonged and atm-independent persistence of gammah2ax foci in human gingival mesenchymal stem cells. Oncotarget 2015, 6, 27275–27287. [Google Scholar] [CrossRef][Green Version]
- Calabrese, E.J. The threshold vs lnt showdown: Dose rate findings exposed flaws in the lnt model part 1. The russell-muller debate. Environ. Res. 2017, 154, 435–451. [Google Scholar] [CrossRef]
- Ruhm, W.; Woloschak, G.E.; Shore, R.E.; Azizova, T.V.; Grosche, B.; Niwa, O.; Akiba, S.; Ono, T.; Suzuki, K.; Iwasaki, T.; et al. Dose and dose-rate effects of ionizing radiation: A discussion in the light of radiological protection. Radiat. Environ. Biophys. 2015, 54, 379–401. [Google Scholar] [CrossRef]
- World Health Organization. Global report on fukushima nuclear accident details health risks. Relev. Epidemiol. Hebd. 2013, 88, 115–116. [Google Scholar]
- Aurengo, A.; Averbeck, D.; Bonnin, A.; LeGuen, B.; Masse, R.; Monier, R.; Tubiana, M.; Valleron, A.J.; de Vathaire, F. Dose-Effect Relationships and Estimation of the Carcinogenic Effects of Low Doses of Ionizing Radiation; Académie des Sciences–Académie Nationale de Médecine: Paris, France, 2005. [Google Scholar]
- Siegel, J.A.; Sacks, B.; Pennington, C.W.; Welsh, J.S. Dose optimization to minimize radiation risk for children undergoing ct and nuclear medicine imaging is misguided and detrimental. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2017, 58, 865–868. [Google Scholar] [CrossRef] [PubMed]
- Grudzenski, S.; Raths, A.; Conrad, S.; Rube, C.E.; Lobrich, M. Inducible response required for repair of low-dose radiation damage in human fibroblasts. Proc. Natl. Acad. Sci. USA 2010, 107, 14205–14210. [Google Scholar] [CrossRef]
- Horn, S.; Barnard, S.; Rothkamm, K. Gamma-h2ax-based dose estimation for whole and partial body radiation exposure. PLoS ONE 2011, 6, e25113. [Google Scholar] [CrossRef] [PubMed]
- de Feraudy, S.; Revet, I.; Bezrookove, V.; Feeney, L.; Cleaver, J.E. A minority of foci or pan-nuclear apoptotic staining of gammah2ax in the s phase after uv damage contain DNA double-strand breaks. Proc. Natl. Acad. Sci. USA 2010, 107, 6870–6875. [Google Scholar] [CrossRef]
- Blackford, A.N.; Jackson, S.P. Atm, atr, and DNA-pk: The trinity at the heart of the DNA damage response. Mol. Cell 2017, 66, 801–817. [Google Scholar] [CrossRef]
- Morgan, W.F.; Bair, W.J. Issues in low dose radiation biology: The controversy continues. A perspective. Radiat. Res. 2013, 179, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Collis, S.J.; Schwaninger, J.M.; Ntambi, A.J.; Keller, T.W.; Nelson, W.G.; Dillehay, L.E.; Deweese, T.L. Evasion of early cellular response mechanisms following low level radiation-induced DNA damage. J. Biol. Chem. 2004, 279, 49624–49632. [Google Scholar] [CrossRef]
- Ishizaki, K.; Hayashi, Y.; Nakamura, H.; Yasui, Y.; Komatsu, K.; Tachibana, A. No induction of p53 phosphorylation and few focus formation of phosphorylated h2ax suggest efficient repair of DNA damage during chronic low-dose-rate irradiation in human cells. J. Radiat. Res. 2004, 45, 521–525. [Google Scholar] [CrossRef]
- Osipov, A.N.; Klokov, D.Y.; Elakov, A.L.; Rozanova, O.M.; Zaichkina, S.I.; Aptikaeva, G.F.; Akhmadieva, A. Comparison in vivo study of genotoxic action of high- versus very low dose-rate gamma-irradiation. Nonlinearity Biol. Toxicol. Med. 2004, 2, 223–232. [Google Scholar] [CrossRef]
- Osipov, A.N.; Buleeva, G.; Arkhangelskaya, E.; Klokov, D. In vivo gamma-irradiation low dose threshold for suppression of DNA double strand breaks below the spontaneous level in mouse blood and spleen cells. Mutat. Res. 2013, 756, 141–145. [Google Scholar] [CrossRef]
- Bong, J.J.; Kang, Y.M.; Shin, S.C.; Choi, S.J.; Lee, K.M.; Kim, H.S. Differential expression of thymic DNA repair genes in low-dose-rate irradiated akr/j mice. J. Vet. Sci. 2013, 14, 271–279. [Google Scholar] [CrossRef][Green Version]
- Shin, S.C.; Kang, Y.M.; Kim, H.S. Life span and thymic lymphoma incidence in high- and low-dose-rate irradiated akr/j mice and commonly expressed genes. Radiat. Res. 2010, 174, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Vilenchik, M.M.; Knudson, A.G. Endogenous DNA double-strand breaks: Production, fidelity of repair, and induction of cancer. Proc. Natl. Acad. Sci. USA 2003, 100, 12871–12876. [Google Scholar] [CrossRef] [PubMed]
- Zeman, M.K.; Cimprich, K.A. Causes and consequences of replication stress. Nat. Cell Biol. 2014, 16, 2–9. [Google Scholar] [CrossRef]
- Saleh-Gohari, N.; Bryant, H.E.; Schultz, N.; Parker, K.M.; Cassel, T.N.; Helleday, T. Spontaneous homologous recombination is induced by collapsed replication forks that are caused by endogenous DNA single-strand breaks. Mol. Cell. Biol. 2005, 25, 7158–7169. [Google Scholar] [CrossRef]
- Beels, L.; Bacher, K.; De Wolf, D.; Werbrouck, J.; Thierens, H. Gamma-h2ax foci as a biomarker for patient x-ray exposure in pediatric cardiac catheterization: Are we underestimating radiation risks? Circulation 2009, 120, 1903–1909. [Google Scholar] [CrossRef]
- Beels, L.; Werbrouck, J.; Thierens, H. Dose response and repair kinetics of gamma-h2ax foci induced by in vitro irradiation of whole blood and t-lymphocytes with x- and gamma-radiation. Int. J. Radiat. Biol. 2010, 86, 760–768. [Google Scholar] [CrossRef] [PubMed]
- Neumaier, T.; Swenson, J.; Pham, C.; Polyzos, A.; Lo, A.T.; Yang, P.; Dyball, J.; Asaithamby, A.; Chen, D.J.; Bissell, M.J.; et al. Evidence for formation of DNA repair centers and dose-response nonlinearity in human cells. Proc. Natl. Acad. Sci. USA 2012, 109, 443–448. [Google Scholar] [CrossRef]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The international society for cellular therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Lychagin, A.A.; Ulyanenko, S.E.; Koryakin, S.N.; Ulyanenko, L.N.; Chernukha, A.E.; Pugachev, R.M.; Brovin, A.I. Methodological support of radiobiological experiments on the “panorama” facility. Radiobiol. Radioecol. 2018, 58, 214–221. [Google Scholar]
- Lutz, W.K.; Lutz, R.W. Statistical model to estimate a threshold dose and its confidence limits for the analysis of sublinear dose-response relationships, exemplified for mutagenicity data. Mutat. Res. 2009, 678, 118–122. [Google Scholar] [CrossRef] [PubMed]
| Dose Rate, mGy/min | Dose, mGy | ||||
|---|---|---|---|---|---|
| 30 | 100 | 160 | 240 | 300 | |
| 0.1 | 1.28 ± 0.11 | 1.43 ± 0.18 | 1.32 ± 0.20 | 1.93 ± 0.36 | 2.23 ± 0.30 |
| 30.0 | 1.55 ± 0.27 | 2.07 ± 0.33 | 2.78 ± 0.45 | 3.27 ± 0.60 | 4.05 ± 0.50 |
| Dose Rate, mGy/min | Dose, mGy | ||||
|---|---|---|---|---|---|
| 30 | 100 | 160 | 240 | 300 | |
| 0.1 | 2.03 ± 0.74 | 0.95 ± 0.37 | 0.44 ± 0.26 | 0.85 ± 0.30 | 0.90 ± 0.19 |
| 30.0 | 4.01 ± 1.85 | 2.34 ± 0.65 | 2.44 ± 0.54 | 2.07 ± 0.48 | 2.23 ± 0.30 |
| Dose Rate, mGy/min | Dose, mGy | ||||
|---|---|---|---|---|---|
| 30 | 100 | 160 | 240 | 300 | |
| 0.1 | 0.98 ± 1.22 | 0.59 ± 0.34 | 0.16 ± 0.27 | 0.36 ± 0.16 | 0.67 ± 0.16 |
| 30.0 | 2.18 ± 1.35 | 1.60 ± 0.37 | 1.62 ± 0.35 | 1.56 ± 0.18 | 1.66 ± 0.26 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ulyanenko, S.; Pustovalova, M.; Koryakin, S.; Beketov, E.; Lychagin, A.; Ulyanenko, L.; Kaprin, A.; Grekhova, A.; M. Ozerova, A.; V. Ozerov, I.; et al. Formation of γH2AX and pATM Foci in Human Mesenchymal Stem Cells Exposed to Low Dose-Rate Gamma-Radiation. Int. J. Mol. Sci. 2019, 20, 2645. https://doi.org/10.3390/ijms20112645
Ulyanenko S, Pustovalova M, Koryakin S, Beketov E, Lychagin A, Ulyanenko L, Kaprin A, Grekhova A, M. Ozerova A, V. Ozerov I, et al. Formation of γH2AX and pATM Foci in Human Mesenchymal Stem Cells Exposed to Low Dose-Rate Gamma-Radiation. International Journal of Molecular Sciences. 2019; 20(11):2645. https://doi.org/10.3390/ijms20112645
Chicago/Turabian StyleUlyanenko, Stepan, Margarita Pustovalova, Sergey Koryakin, Evgenii Beketov, Anatolii Lychagin, Liliya Ulyanenko, Andrey Kaprin, Anna Grekhova, Alexandra M. Ozerova, Ivan V. Ozerov, and et al. 2019. "Formation of γH2AX and pATM Foci in Human Mesenchymal Stem Cells Exposed to Low Dose-Rate Gamma-Radiation" International Journal of Molecular Sciences 20, no. 11: 2645. https://doi.org/10.3390/ijms20112645
APA StyleUlyanenko, S., Pustovalova, M., Koryakin, S., Beketov, E., Lychagin, A., Ulyanenko, L., Kaprin, A., Grekhova, A., M. Ozerova, A., V. Ozerov, I., Vorobyeva, N., Shegay, P., Ivanov, S., Leonov, S., Klokov, D., & Osipov, A. N. (2019). Formation of γH2AX and pATM Foci in Human Mesenchymal Stem Cells Exposed to Low Dose-Rate Gamma-Radiation. International Journal of Molecular Sciences, 20(11), 2645. https://doi.org/10.3390/ijms20112645