Molecular Targets of Genistein and Its Related Flavonoids to Exert Anticancer Effects
Abstract
1. Introduction
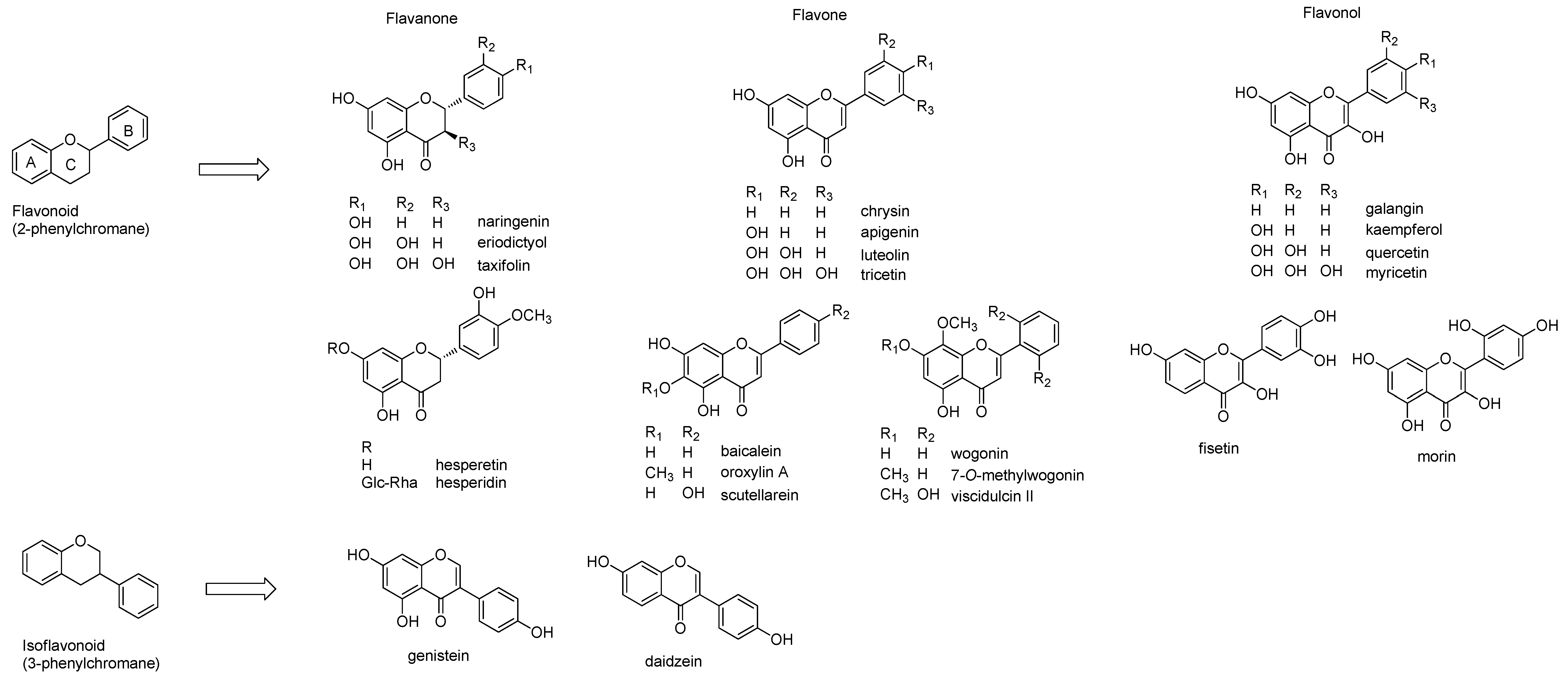
2. Flavonoids
2.1. Structures of Flavonoids
2.2. Anticancer Effects of Flavonoids
2.2.1. Flavones with Anti-Cancer Effects
2.2.1.1. Apigenin
2.2.1.2. Luteolin
2.2.1.3. Oroxylin A
2.2.1.4. Wogonin
2.2.2. Flavonols with Anti-Cancer Effects
2.2.2.1. Kaempferol
2.2.2.2. Quercetin
2.2.2.3. Myricetin
2.2.3. Flavanones with Anti-Cancer Effects
2.2.3.1. Hesperidin
2.2.3.2. Naringin
3. Flavonoids with PLK1 Inhibitory Effects
3.1. 7-O-Methylwogonin
3.2. Baicalein
3.3. Dihydrobaicalein
3.4. Viscidulin II
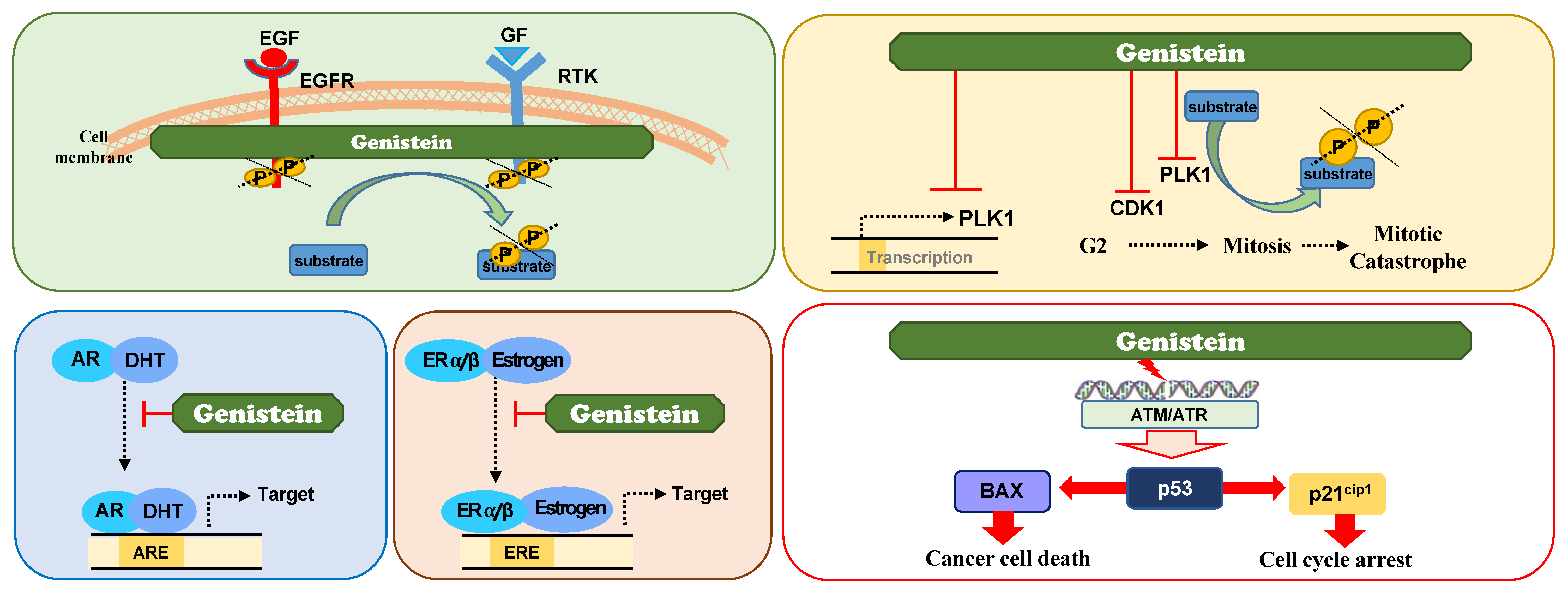
3.5. Genistein
3.5.1. Genistein as A Receptor Tyrosine Kinase (RTK) Inhibitor
3.5.2. Genistein as A Direct PLK1 Kinase Inhibitor
3.5.3. Genistein as A Suppressor of PLK1 Expression
3.5.4. Genistein as A Modulator of Hormone Receptor
3.5.5. Genistein in Clinical Trials for Chemoprevention and Cancer Treatment
3.5.6. New Contrivance for Overcoming the Hurdles of Genistein
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Abotaleb, M.; Samuel, S.M.; Varghese, E.; Varghese, S.; Kubatka, P.; Liskova, A.; Busselberg, D. Flavonoids in Cancer and Apoptosis. Cancers 2018, 11, 28. [Google Scholar] [CrossRef]
- Douglas, C.C.; Johnson, S.A.; Arjmandi, B.H. Soy and its isoflavones: The truth behind the science in breast cancer. Anticancer Agents Med. Chem. 2013, 13, 1178–1187. [Google Scholar] [CrossRef]
- Nwodo, J.N.; Ibezim, A.; Simoben, C.V.; Ntie-Kang, F. Exploring Cancer Therapeutics with Natural Products from African Medicinal Plants, Part II: Alkaloids, Terpenoids and Flavonoids. Anticancer Agents Med. Chem. 2016, 16, 108–127. [Google Scholar] [CrossRef] [PubMed]
- Raffa, D.; Maggio, B.; Raimondi, M.V.; Plescia, F.; Daidone, G. Recent discoveries of anticancer flavonoids. Eur. J. Med. Chem. 2017, 142, 213–228. [Google Scholar] [CrossRef]
- George, V.C.; Dellaire, G.; Rupasinghe, H.P.V. Plant flavonoids in cancer chemoprevention: Role in genome stability. J. Nutr. Biochem. 2017, 45, 1–14. [Google Scholar] [CrossRef]
- Rathee, P.; Chaudhary, H.; Rathee, S.; Rathee, D.; Kumar, V.; Kohli, K. Mechanism of action of flavonoids as anti-inflammatory agents: A review. Inflamm. Allergy Drug Targets 2009, 8, 229–235. [Google Scholar] [CrossRef]
- Babu, P.V.; Liu, D.; Gilbert, E.R. Recent advances in understanding the anti-diabetic actions of dietary flavonoids. J. Nutr. Biochem. 2013, 24, 1777–1789. [Google Scholar] [CrossRef]
- Kawser Hossain, M.; Abdal Dayem, A.; Han, J.; Yin, Y.; Kim, K.; Kumar Saha, S.; Yang, G.M.; Choi, H.Y.; Cho, S.G. Molecular Mechanisms of the Anti-Obesity and Anti-Diabetic Properties of Flavonoids. Int. J. Mol. Sci. 2016, 17, 569. [Google Scholar] [CrossRef]
- Parhiz, H.; Roohbakhsh, A.; Soltani, F.; Rezaee, R.; Iranshahi, M. Antioxidant and anti-inflammatory properties of the citrus flavonoids hesperidin and hesperetin: An updated review of their molecular mechanisms and experimental models. Phytother Res. 2015, 29, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Faggio, C.; Sureda, A.; Morabito, S.; Sanches-Silva, A.; Mocan, A.; Nabavi, S.F.; Nabavi, S.M. Flavonoids and platelet aggregation: A brief review. Eur. J. Pharmacol. 2017, 807, 91–101. [Google Scholar] [CrossRef]
- Spagnuolo, C.; Moccia, S.; Russo, G.L. Anti-inflammatory effects of flavonoids in neurodegenerative disorders. Eur. J. Med. Chem. 2018, 153, 105–115. [Google Scholar] [CrossRef]
- Liu, X.; Erikson, R.L. Polo-like kinase (Plk)1 depletion induces apoptosis in cancer cells. Proc. Natl. Acad. Sci. USA 2003, 100, 5789–5794. [Google Scholar] [CrossRef]
- Liu, X.; Lei, M.; Erikson, R.L. Normal cells, but not cancer cells, survive severe Plk1 depletion. Mol. Cell Biol. 2006, 26, 2093–2108. [Google Scholar] [CrossRef] [PubMed]
- Yim, H.; Erikson, R.L. Polo-like kinase 1 depletion induces DNA damage in early S prior to caspase activation. Mol. Cell Biol. 2009, 29, 2609–2621. [Google Scholar] [CrossRef]
- Yim, H. Current clinical trials with polo-like kinase 1 inhibitors in solid tumors. Anticancer Drugs 2013, 24, 999–1006. [Google Scholar] [CrossRef]
- Yim, H.; Erikson, R.L. Plk1-targeted therapies in TP53- or RAS-mutated cancer. Mutat. Res. Rev. Mutat. Res. 2014, 761, 31–39. [Google Scholar] [CrossRef]
- Schmucker, S.; Sumara, I. Molecular dynamics of PLK1 during mitosis. Mol. Cell Oncol. 2014, 1, e954507. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.W.; Messersmith, W.A.; Dy, G.K.; Weekes, C.D.; Whitworth, A.; Ren, C.; Maniar, M.; Wilhelm, F.; Eckhardt, S.G.; Adjei, A.A.; et al. Phase I study of Rigosertib, an inhibitor of the phosphatidylinositol 3-kinase and Polo-like kinase 1 pathways, combined with gemcitabine in patients with solid tumors and pancreatic cancer. Clin. Cancer Res. 2012, 18, 2048–2055. [Google Scholar] [CrossRef]
- Gutteridge, R.E.; Ndiaye, M.A.; Liu, X.; Ahmad, N. Plk1 Inhibitors in Cancer Therapy: From Laboratory to Clinics. Mol. Cancer Ther. 2016, 15, 1427–1435. [Google Scholar] [CrossRef]
- Gumireddy, K.; Reddy, M.V.; Cosenza, S.C.; Boominathan, R.; Baker, S.J.; Papathi, N.; Jiang, J.; Holland, J.; Reddy, E.P. ON01910, a non-ATP-competitive small molecule inhibitor of Plk1, is a potent anticancer agent. Cancer Cell 2005, 7, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Woo, S.U.; Jang, H.R.; Chin, Y.W.; Yim, H. 7-O-Methylwogonin from Scutellaria baicalensis Disturbs Mitotic Progression by Inhibiting Plk1 Activity in Hep3B Cells. Planta Med. 2019, 85, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.B.; Woo, S.U.; Chin, Y.W.; Jang, Y.J.; Yim, H. Sensitivity of TP53-Mutated Cancer Cells to the Phytoestrogen Genistein Is Associated with Direct Inhibition of Plk1 Activity. J. Cell Physiol. 2017, 232, 2818–2828. [Google Scholar] [CrossRef] [PubMed]
- Harborne, J.B. The Flavonoids: Advances in Research Since 1986, 1st ed.; Chapman & Hall: London, UK; New York, NY, USA, 1994; p. xii. 676 p. [Google Scholar]
- Li, Y.; Fang, H.; Xu, W. Recent advance in the research of flavonoids as anticancer agents. Mini Rev. Med. Chem. 2007, 7, 663–678. [Google Scholar] [PubMed]
- Ren, W.; Qiao, Z.; Wang, H.; Zhu, L.; Zhang, L. Flavonoids: Promising anticancer agents. Med. Res. Rev. 2003, 23, 519–534. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Yuan, X.; Wang, J.; Feng, Y.; Ji, F.; Li, Z.; Bian, J. A review on flavones targeting serine/threonine protein kinases for potential anticancer drugs. Bioorg. Med. Chem. 2019, 27, 677–685. [Google Scholar] [CrossRef]
- Le Marchand, L. Cancer preventive effects of flavonoids—A review. Biomed. Pharmacother. 2002, 56, 296–301. [Google Scholar] [CrossRef]
- Nijveldt, R.J.; van Nood, E.; van Hoorn, D.E.; Boelens, P.G.; van Norren, K.; van Leeuwen, P.A. Flavonoids: A review of probable mechanisms of action and potential applications. Am. J. Clin. Nutr. 2001, 74, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.P.; Agarwal, R. Natural flavonoids targeting deregulated cell cycle progression in cancer cells. Curr. Drug Targets 2006, 7, 345–354. [Google Scholar] [CrossRef]
- Sun, Q.; Lu, N.N.; Feng, L. Apigetrin inhibits gastric cancer progression through inducing apoptosis and regulating ROS-modulated STAT3/JAK2 pathway. Biochem. Biophys. Res. Commun. 2018, 498, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Krisanapun, C.; Lee, S.H.; Nualsanit, T.; Sams, C.; Peungvicha, P.; Baek, S.J. Molecular targets of apigenin in colorectal cancer cells: Involvement of p21, NAG-1 and p53. Eur. J. Cancer 2010, 46, 3365–3374. [Google Scholar] [CrossRef] [PubMed]
- Gao, A.M.; Zhang, X.Y.; Ke, Z.P. Apigenin sensitizes BEL-7402/ADM cells to doxorubicin through inhibiting miR-101/Nrf2 pathway. Oncotarget 2017, 8, 82085–82091. [Google Scholar] [CrossRef]
- Chen, C.H.; Huang, T.S.; Wong, C.H.; Hong, C.L.; Tsai, Y.H.; Liang, C.C.; Lu, F.J.; Chang, W.H. Synergistic anti-cancer effect of baicalein and silymarin on human hepatoma HepG2 Cells. Food Chem. Toxicol. 2009, 47, 638–644. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.T.; Li, J.; Haung, H.H.; Liu, H.; Han, M.; Ramachandran, S.; Li, C.Q.; Sharp, W.W.; Hamann, K.J.; Yuan, C.S.; et al. Baicalein protects against doxorubicin-induced cardiotoxicity by attenuation of mitochondrial oxidant injury and JNK activation. J. Cell Biochem. 2011, 112, 2873–2881. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Ling, Y.; Chen, Y.; Li, C.L.; Feng, F.; You, Q.D.; Lu, N.; Guo, Q.L. Flavonoid baicalein suppresses adhesion, migration and invasion of MDA-MB-231 human breast cancer cells. Cancer Lett. 2010, 297, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Dou, J.; Wang, Z.; Ma, L.; Peng, B.; Mao, K.; Li, C.; Su, M.; Zhou, C.; Peng, G. Baicalein and baicalin inhibit colon cancer using two distinct fashions of apoptosis and senescence. Oncotarget 2018, 9, 20089–20102. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; He, S.; Sun, X. HSP70 desensitizes osteosarcoma cells to baicalein and protects cells from undergoing apoptosis. Apoptosis 2014, 19, 1269–1280. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Yang, Y.; Li, Y.; Cao, Y.; Tang, L.; Chen, F.; Xia, J. Baicalein inhibits cervical cancer progression via downregulating long noncoding RNA BDLNR and its downstream PI3K/Akt pathway. Int. J. Biochem. Cell Biol. 2018, 94, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.C.; Chen, C.Y.; Lin, C.R.; Yang, T.Y.; Chen, T.H.; Wu, L.C.; Wu, C.C. Luteolin attenuates TGF-beta1-induced epithelial-mesenchymal transition of lung cancer cells by interfering in the PI3K/Akt-NF-kappaB-Snail pathway. Life Sci. 2013, 93, 924–933. [Google Scholar] [CrossRef] [PubMed]
- Im, E.; Yeo, C.; Lee, E.O. Luteolin induces caspase-dependent apoptosis via inhibiting the AKT/osteopontin pathway in human hepatocellular carcinoma SK-Hep-1 cells. Life Sci. 2018, 209, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Jiang, W.; Yu, D.; Yan, Z. Luteolin inhibits proliferation and induces apoptosis of human melanoma cells in vivo and in vitro by suppressing MMP-2 and MMP-9 through the PI3K/AKT pathway. Food Funct. 2019, 10, 703–712. [Google Scholar] [CrossRef] [PubMed]
- Chian, S.; Thapa, R.; Chi, Z.; Wang, X.J.; Tang, X. Luteolin inhibits the Nrf2 signaling pathway and tumor growth in vivo. Biochem. Biophys. Res. Commun. 2014, 447, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Wang, H.; Jia, Y.; Pan, H.; Ding, H. Luteolin induces apoptosis by ROS/ER stress and mitochondrial dysfunction in gliomablastoma. Cancer Chemother. Pharmacol. 2017, 79, 1031–1041. [Google Scholar] [CrossRef] [PubMed]
- Mu, R.; Qi, Q.; Gu, H.; Wang, J.; Yang, Y.; Rong, J.; Liu, W.; Lu, N.; You, Q.; Guo, Q. Involvement of p53 in oroxylin A-induced apoptosis in cancer cells. Mol. Carcinog. 2009, 48, 1159–1169. [Google Scholar] [CrossRef] [PubMed]
- Li, H.N.; Nie, F.F.; Liu, W.; Dai, Q.S.; Lu, N.; Qi, Q.; Li, Z.Y.; You, Q.D.; Guo, Q.L. Apoptosis induction of oroxylin A in human cervical cancer HeLa cell line in vitro and in vivo. Toxicology 2009, 257, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Qiao, C.; Wei, L.; Dai, Q.; Zhou, Y.; Yin, Q.; Li, Z.; Xiao, Y.; Guo, Q.; Lu, N. UCP2-related mitochondrial pathway participates in oroxylin A-induced apoptosis in human colon cancer cells. J. Cell Physiol. 2015, 230, 1054–1063. [Google Scholar] [CrossRef]
- Lu, Z.; Lu, N.; Li, C.; Li, F.; Zhao, K.; Lin, B.; Guo, Q. Oroxylin A inhibits matrix metalloproteinase-2/9 expression and activation by up-regulating tissue inhibitor of metalloproteinase-2 and suppressing the ERK1/2 signaling pathway. Toxicol Lett. 2012, 209, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Yao, Y.; Zhao, K.; Huang, Y.; Zhou, Y.; Zhao, L.; Guo, Q.; Lu, N. Oroxylin A inhibits invasion and migration through suppressing ERK/GSK-3β signaling in snail-expressing non-small-cell lung cancer cells. Mol. Carcinog. 2016, 55, 2121–2134. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Zhao, K.; Li, G.; Yao, J.; Dai, Q.; Hui, H.; Li, Z.; Guo, Q.; Lu, N. Oroxylin A inhibits hypoxia-induced invasion and migration of MCF-7 cells by suppressing the Notch pathway. Anticancer Drugs 2014, 25, 778–789. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Ding, Q.; Ding, Y.; Lu, L.; Wang, X.; Zhang, Y.; Zhang, X.; Guo, Q.; Zhao, L. Oroxylin A reverses the drug resistance of chronic myelogenous leukemia cells to imatinib through CXCL12/CXCR7 axis in bone marrow microenvironment. Mol. Carcinog. 2017, 56, 863–876. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Miao, H.; Li, W.; Yao, J.; Sun, Y.; Li, Z.; Zhao, L.; Guo, Q. CXCL12/CXCR4 axis confers adriamycin resistance to human chronic myelogenous leukemia and oroxylin A improves the sensitivity of K562/ADM cells. Biochem. Pharmacol. 2014, 90, 212–225. [Google Scholar] [CrossRef]
- Li, X.; Miao, H.; Zhang, Y.; Li, W.; Li, Z.; Zhou, Y.; Zhao, L.; Guo, Q. Bone marrow microenvironment confers imatinib resistance to chronic myelogenous leukemia and oroxylin A reverses the resistance by suppressing Stat3 pathway. Arch. Toxicol. 2015, 89, 121–136. [Google Scholar] [CrossRef] [PubMed]
- Feng, Q.; Wang, H.; Pang, J.; Ji, L.; Han, J.; Wang, Y.; Qi, X.; Liu, Z.; Lu, L. Prevention of Wogonin on Colorectal Cancer Tumorigenesis by Regulating p53 Nuclear Translocation. Front. Pharmacol. 2018, 9, 1356. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zhang, L.; Wu, Y.; Dai, Q.; Zhou, Y.; Li, Z.; Yang, L.; Guo, Q.; Lu, N. Selective anti-tumor activity of wogonin targeting the Warburg effect through stablizing p53. Pharmacol. Res. 2018, 135, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Fu, P.; Du, F.; Liu, Y.; Hong, Y.; Yao, M.; Zheng, S. Wogonin increases doxorubicin sensitivity by down-regulation of IGF-1R/AKT signaling pathway in human breast cancer. Cell Mol. Biol. 2015, 61, 123–127. [Google Scholar] [PubMed]
- Hu, C.; Xu, M.; Qin, R.; Chen, W.; Xu, X. Wogonin induces apoptosis and endoplasmic reticulum stress in HL-60 leukemia cells through inhibition of the PI3K-AKT signaling pathway. Oncol. Rep. 2015, 33, 3146–3154. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chen, P.; Lu, N.; Ling, Y.; Chen, Y.; Hui, H.; Lu, Z.; Song, X.; Li, Z.; You, Q.; Guo, Q. Inhibitory effects of wogonin on the invasion of human breast carcinoma cells by downregulating the expression and activity of matrix metalloproteinase-9. Toxicology 2011, 282, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Hong, M.; Cheng, H.; Song, L.; Wang, W.; Wang, Q.; Xu, D.; Xing, W. Wogonin Suppresses the Activity of Matrix Metalloproteinase-9 and Inhibits Migration and Invasion in Human Hepatocellular Carcinoma. Molecules 2018, 23, 384. [Google Scholar] [CrossRef] [PubMed]
- Huynh, D.L.; Kwon, T.; Zhang, J.J.; Sharma, N.; Gera, M.; Ghosh, M.; Kim, N.; Kim Cho, S.; Lee, D.S.; Park, Y.H.; Jeong, D.K. Wogonin suppresses stem cell-like traits of CD133 positive osteosarcoma cell via inhibiting matrix metallopeptidase-9 expression. BMC Complement. Altern. Med. 2017, 17, 304. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Yao, J.; Wang, F.; Zhou, M.; Zhou, Y.; Wang, H.; Wei, L.; Zhao, L.; Li, Z.; Lu, N.; et al. Wogonin inhibits tumor angiogenesis via degradation of HIF-1α protein. Toxicol. Appl. Pharmacol. 2013, 271, 144–155. [Google Scholar] [CrossRef]
- Yang, L.; Wang, Q.; Li, D.; Zhou, Y.; Zheng, X.; Sun, H.; Yan, J.; Zhang, L.; Lin, Y.; Wang, X. Wogonin enhances antitumor activity of tumor necrosis factor-related apoptosis-inducing ligand in vivo through ROS-mediated downregulation of cFLIPL and IAP proteins. Apoptosis 2013, 18, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Zhang, Y.; Li, W.; Miao, H.; Zhang, H.; Zhou, Y.; Li, Z.; You, Q.; Zhao, L.; Guo, Q. Wogonin reverses multi-drug resistance of human myelogenous leukemia K562/A02 cells via downregulation of MRP1 expression by inhibiting Nrf2/ARE signaling pathway. Biochem. Pharmacol. 2014, 92, 220–234. [Google Scholar] [CrossRef] [PubMed]
- Tu, L.Y.; Bai, H.H.; Cai, J.Y.; Deng, S.P. The mechanism of kaempferol induced apoptosis and inhibited proliferation in human cervical cancer SiHa cell: From macro to nano. Scanning 2016, 38, 644–653. [Google Scholar] [CrossRef]
- Qin, Y.; Cui, W.; Yang, X.; Tong, B. Kaempferol inhibits the growth and metastasis of cholangiocarcinoma in vitro and in vivo. Acta Biochim. Biophys. Sin. 2016, 48, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kim, J.H. Kaempferol Inhibits Pancreatic Cancer Cell Growth and Migration through the Blockade of EGFR-Related Pathway In Vitro. PLoS ONE 2016, 11, e0155264. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Gao, M.; Wang, J. Kaempferol inhibits cancer cell growth by antagonizing estrogen-related receptor alpha and γ activities. Cell Biol. Int. 2013, 37, 1190–1196. [Google Scholar] [PubMed]
- Kim, T.W.; Lee, S.Y.; Kim, M.; Cheon, C.; Ko, S.G. Kaempferol induces autophagic cell death via IRE1-JNK-CHOP pathway and inhibition of G9a in gastric cancer cells. Cell Death Dis. 2018, 9, 875. [Google Scholar] [CrossRef] [PubMed]
- Klimaszewska-Wisniewska, A.; Halas-Wisniewska, M.; Izdebska, M.; Gagat, M.; Grzanka, A.; Grzanka, D. Antiproliferative and antimetastatic action of quercetin on A549 non-small cell lung cancer cells through its effect on the cytoskeleton. Acta Histochem. 2017, 119, 99–112. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.K.; Park, S.Y.; Kim, Y.M.; Lee, W.S.; Park, O.J. AMP kinase/cyclooxygenase-2 pathway regulates proliferation and apoptosis of cancer cells treated with quercetin. Exp. Mol. Med. 2009, 41, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; Lee, Y.H.; Sharma, A.R.; Park, J.B.; Jagga, S.; Sharma, G.; Lee, S.S.; Nam, J.S. Quercetin induces apoptosis and cell cycle arrest in triple-negative breast cancer cells through modulation of Foxo3a activity. Korean J. Physiol. Pharmacol. 2017, 21, 205–213. [Google Scholar] [CrossRef]
- Balakrishnan, S.; Bhat, F.A.; Raja Singh, P.; Mukherjee, S.; Elumalai, P.; Das, S.; Patra, C.R.; Arunakaran, J. Gold nanoparticle-conjugated quercetin inhibits epithelial-mesenchymal transition, angiogenesis and invasiveness via EGFR/VEGFR-2-mediated pathway in breast cancer. Cell Prolif. 2016, 49, 678–697. [Google Scholar] [CrossRef] [PubMed]
- Cao, C.; Sun, L.; Mo, W.; Sun, L.; Luo, J.; Yang, Z.; Ran, Y. Quercetin Mediates β-Catenin in Pancreatic Cancer Stem-Like Cells. Pancreas 2015, 44, 1334–1339. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Choi, Y.J.; Park, S.H.; Nam, M.J. Potential role of nucleoside diphosphate kinase in myricetin-induced selective apoptosis in colon cancer HCT-15cells. Food Chem. Toxicol. 2018, 116, 315–322. [Google Scholar] [CrossRef]
- Ma, L.; Cao, X.; Wang, H.; Lu, K.; Wang, Y.; Tu, C.; Dai, Y.; Meng, Y.; Li, Y.; Yu, P.; et al. Discovery of Myricetin as a Potent Inhibitor of Human Flap Endonuclease 1, Which Potentially Can Be Used as Sensitizing Agent against HT-29 Human Colon Cancer Cells. J. Agric. Food Chem. 2019, 67, 1656–1665. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y.; Park, S.Y.; Choung, S.Y. Enhancing effects of myricetin on the osteogenic differentiation of human periodontal ligament stem cells via BMP-2/Smad and ERK/JNK/p38 mitogen-activated protein kinase signaling pathway. Eur. J. Pharmacol. 2018, 834, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Ye, C.; Zhang, C.; Huang, H.; Yang, B.; Xiao, G.; Kong, D.; Tian, Q.; Song, Q.; Song, Y.; Tan, H.; et al. The Natural Compound Myricetin Effectively Represses the Malignant Progression of Prostate Cancer by Inhibiting PIM1 and Disrupting the PIM1/CXCR4 Interaction. Cell Physiol. Biochem. 2018, 48, 1230–1244. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Cui, S.X.; Sun, S.Y.; Shi, W.N.; Song, Z.Y.; Wang, S.Q.; Yu, X.F.; Gao, Z.H.; Qu, X.J. Chemoprevention of intestinal tumorigenesis by the natural dietary flavonoid myricetin in APCMin/+ mice. Oncotarget 2016, 7, 60446–60460. [Google Scholar]
- Pandey, P.; Sayyed, U.; Tiwari, R.K.; Siddiqui, M.H.; Pathak, N.; Bajpai, P. Hesperidin Induces ROS-Mediated Apoptosis along with Cell Cycle Arrest at G2/M Phase in Human Gall Bladder Carcinoma. Nutr. Cancer 2019, 71, 676–687. [Google Scholar] [CrossRef] [PubMed]
- Elango, R.; Athinarayanan, J.; Subbarayan, V.P.; Lei, D.K.Y.; Alshatwi, A.A. Hesperetin induces an apoptosis-triggered extrinsic pathway and a p53- independent pathway in human lung cancer H522 cells. J. Asian Nat. Prod. Res. 2018, 20, 559–569. [Google Scholar] [CrossRef]
- Xia, R.; Xu, G.; Huang, Y.; Sheng, X.; Xu, X.; Lu, H. Hesperidin suppresses the migration and invasion of non-small cell lung cancer cells by inhibiting the SDF-1/CXCR-4 pathway. Life Sci. 2018, 201, 111–120. [Google Scholar] [CrossRef]
- Mo’men, Y.S.; Hussein, R.M.; Kandeil, M.A. Involvement of PI3K/Akt pathway in the protective effect of hesperidin against a chemically induced liver cancer in rats. J. Biochem. Mol. Toxicol. 2019, e22305. [Google Scholar] [CrossRef]
- Mahmoud, A.M.; Mohammed, H.M.; Khadrawy, S.M.; Galaly, S.R. Hesperidin protects against chemically induced hepatocarcinogenesis via modulation of Nrf2/ARE/HO-1, PPARγ and TGF-β1/Smad3 signaling, and amelioration of oxidative stress and inflammation. Chem. Biol. Interact. 2017, 277, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Peng, W.; Hu, S.; Deng, J. miR-126/VCAM-1 regulation by naringin suppresses cell growth of human non-small cell lung cancer. Oncol. Lett. 2018, 16, 4754–4760. [Google Scholar] [CrossRef]
- Yoshinaga, A.; Kajiya, N.; Oishi, K.; Kamada, Y.; Ikeda, A.; Chigwechokha, P.K.; Kibe, T.; Kishida, M.; Kishida, S.; Komatsu, M.; et al. NEU3 inhibitory effect of naringin suppresses cancer cell growth by attenuation of EGFR signaling through GM3 ganglioside accumulation. Eur. J. Pharmacol. 2016, 782, 21–29. [Google Scholar] [CrossRef]
- Chandrika, B.B.; Steephan, M.; Kumar, T.R.S.; Sabu, A.; Haridas, M. Hesperetin and Naringenin sensitize HER2 positive cancer cells to death by serving as HER2 Tyrosine Kinase inhibitors. Life Sci. 2016, 160, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Lian, G.Y.; Wang, Q.M.; Tang, P.M.; Zhou, S.; Huang, X.R.; Lan, H.Y. Combination of Asiatic Acid and Naringenin Modulates NK Cell Anti-cancer Immunity by Rebalancing Smad3/Smad7 Signaling. Mol. Ther. 2018, 26, 2255–2266. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Lim, W.; Bazer, F.W.; Song, G. Naringenin suppresses growth of human placental choriocarcinoma via reactive oxygen species-mediated P38 and JNK MAPK pathways. Phytomedicine 2018, 50, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Aroui, S.; Aouey, B.; Chtourou, Y.; Meunier, A.C.; Fetoui, H.; Kenani, A. Naringin suppresses cell metastasis and the expression of matrix metalloproteinases (MMP-2 and MMP-9) via the inhibition of ERK-P38-JNK signaling pathway in human glioblastoma. Chem. Biol. Interact. 2016, 244, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Aroui, S.; Najlaoui, F.; Chtourou, Y.; Meunier, A.C.; Laajimi, A.; Kenani, A.; Fetoui, H. Naringin inhibits the invasion and migration of human glioblastoma cell via downregulation of MMP-2 and MMP-9 expression and inactivation of p38 signaling pathway. Tumour. Biol. 2016, 37, 3831–3839. [Google Scholar] [CrossRef]
- Ming, H.; Chuang, Q.; Jiashi, W.; Bin, L.; Guangbin, W.; Xianglu, J. Naringin targets Zeb1 to suppress osteosarcoma cell proliferation and metastasis. Aging 2018, 10, 4141–4151. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wang, Y.; Zhang, L.; Chen, L.; Du, Y.; Ye, T.; Shi, X. Naringenin exerts anti-angiogenic effects in human endothelial cells: Involvement of ERRα/VEGF/KDR signaling pathway. Fitoterapia 2016, 111, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Song, H.M.; Park, G.H.; Eo, H.J.; Jeong, J.B. Naringenin-Mediated ATF3 Expression Contributes to Apoptosis in Human Colon Cancer. Biomol. Ther. 2016, 24, 140–146. [Google Scholar] [CrossRef]
- Akiyama, T.; Ishida, J.; Nakagawa, S.; Ogawara, H.; Watanabe, S.; Itoh, N.; Shibuya, M.; Fukami, Y. Genistein, a specific inhibitor of tyrosine-specific protein kinases. J. Biol. Chem. 1987, 262, 5592–5595. [Google Scholar]
- Peterson, G.; Barnes, S. Genistein inhibits both estrogen and growth factor-stimulated proliferation of human breast cancer cells. Cell Growth Differ. 1996, 7, 1345–1351. [Google Scholar]
- Spinozzi, F.; Pagliacci, M.C.; Migliorati, G.; Moraca, R.; Grignani, F.; Riccardi, C.; Nicoletti, I. The natural tyrosine kinase inhibitor genistein produces cell cycle arrest and apoptosis in Jurkat T-leukemia cells. Leuk. Res. 1994, 18, 431–439. [Google Scholar] [CrossRef]
- Choi, Y.H.; Zhang, L.; Lee, W.H.; Park, K.Y. Genistein-induced G2/M arrest is associated with the inhibition of cyclin B1 and the induction of p21 in human breast carcinoma cells. Int. J. Oncol. 1998, 13, 391–396. [Google Scholar] [CrossRef]
- Yan, G.R.; Zou, F.Y.; Dang, B.L.; Zhang, Y.; Yu, G.; Liu, X.; He, Q.Y. Genistein-induced mitotic arrest of gastric cancer cells by downregulating KIF20A, a proteomics study. Proteomics 2012, 12, 2391–2399. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.L.; Kung, M.L.; Chow, N.H.; Su, S.J. Genistein arrests hepatoma cells at G2/M phase: Involvement of ATM activation and upregulation of p21waf1/cip1 and Wee1. Biochem. Pharmacol. 2004, 67, 717–726. [Google Scholar] [CrossRef]
- Seo, Y.J.; Kim, B.S.; Chun, S.Y.; Park, Y.K.; Kang, K.S.; Kwon, T.G. Apoptotic effects of genistein, biochanin-A and apigenin on LNCaP and PC-3 cells by p21 through transcriptional inhibition of polo-like kinase-1. J. Korean Med. Sci. 2011, 26, 1489–1494. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Gong, P.; Madak-Erdogan, Z.; Martin, T.; Jeyakumar, M.; Carlson, K.; Khan, I.; Smillie, T.J.; Chittiboyina, A.G.; Rotte, S.C.; et al. Mechanisms enforcing the estrogen receptor β selectivity of botanical estrogens. FASEB J. 2013, 27, 4406–4418. [Google Scholar] [CrossRef] [PubMed]
- Pihlajamaa, P.; Zhang, F.P.; Saarinen, L.; Mikkonen, L.; Hautaniemi, S.; Janne, O.A. The phytoestrogen genistein is a tissue-specific androgen receptor modulator. Endocrinology 2011, 152, 4395–4405. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.T.; Wang, C.Y.; Yang, R.C.; Chu, C.J.; Wu, H.T.; Pang, J.H. Wogonin, an active compound in Scutellaria baicalensis, induces apoptosis and reduces telomerase activity in the HL-60 leukemia cells. Phytomedicine 2010, 17, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Benerjee, S.; Li, Y.; Wang, Z.; Sarkar, F.H. Multi-targeted therapy of cancer by genistein. Cancer Lett. 2008, 269, 226–242. [Google Scholar] [CrossRef]
- Plochmann, K.; Korte, G.; Koutsilieri, E.; Richling, E.; Riederer, P.; Rethwilm, A.; Schreier, P.; Scheller, C. Structure-activity relationships of flavonoid-induced cytotoxicity on human leukemia cells. Arch Biochem. Biophys. 2007, 460, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Yang, L.J.; Wang, P.; He, Y.J.; Huang, J.M.; Liu, H.W.; Shen, X.F.; Wang, F. Dietary apigenin potentiates the inhibitory effect of interferon-alpha on cancer cell viability through inhibition of 26S proteasome-mediated interferon receptor degradation. Food Nutr. Res. 2016, 60, 31288. [Google Scholar] [CrossRef] [PubMed]
- Imran, M.; Rauf, A.; Abu-Izneid, T.; Nadeem, M.; Shariati, M.A.; Khan, I.A.; Imran, A.; Orhan, I.E.; Rizwan, M.; Atif, M.; et al. Luteolin, a flavonoid, as an anticancer agent: A review. Biomed. Pharmacother. 2019, 112, 108612. [Google Scholar] [CrossRef]
- Maruthanila, V.L.; Elancheran, R.; Roy, N.K.; Bhattacharya, A.; Kunnumakkara, A.B.; Kabilan, S.; Kotoky, J. In silico Molecular Modelling of Selected Natural Ligands and their Binding Features with Estrogen Receptor Alpha. Curr. Comput. Aided. Drug Des. 2019, 15, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Guo, Q.; Zhao, L. Overview of Oroxylin A: A Promising Flavonoid Compound. Phytother. Res. 2016, 30, 1765–1774. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, A.; Ravanan, P. Kaempferol mitigates Endoplasmic Reticulum Stress Induced Cell Death by targeting caspase 3/7. Sci. Rep. 2018, 8, 2189. [Google Scholar] [CrossRef]
- Pang, X.; Fu, W.; Wang, J.; Kang, D.; Xu, L.; Zhao, Y.; Liu, A.L.; Du, G.H. Identification of Estrogen Receptor alpha Antagonists from Natural Products via In Vitro and In Silico Approaches. Oxid. Med. Cell Longev. 2018, 2018, 6040149. [Google Scholar] [CrossRef]
- Rauf, A.; Imran, M.; Khan, I.A.; Ur-Rehman, M.; Gilani, S.A.; Mehmood, Z.; Mubarak, M.S. Anticancer potential of quercetin: A comprehensive review. Phytother Res. 2018, 32, 2109–2130. [Google Scholar] [CrossRef] [PubMed]
- Devi, K.P.; Rajavel, T.; Habtemariam, S.; Nabavi, S.F.; Nabavi, S.M. Molecular mechanisms underlying anticancer effects of myricetin. Life Sci. 2015, 142, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Chen, H.; Lu, W.; Wu, Y.; Wu, X.; Xia, D.; Zhu, J. Myricetin Induces Protective Autophagy by Inhibiting the Phosphorylation of mTOR in HepG2 Cells. Anat. Rec. 2018, 301, 786–795. [Google Scholar] [CrossRef] [PubMed]
- AlAjmi, M.F.; Rehman, M.T.; Hussain, A.; Rather, G.M. Pharmacoinformatics approach for the identification of Polo-like kinase-1 inhibitors from natural sources as anti-cancer agents. Int. J. Biol. Macromol. 2018, 116, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Ameer, B.; Weintraub, R.A.; Johnson, J.V.; Yost, R.A.; Rouseff, R.L. Flavanone absorption after naringin, hesperidin, and citrus administration. Clin. Pharmacol. Ther. 1996, 60, 34–40. [Google Scholar] [CrossRef]
- Bharti, S.; Rani, N.; Krishnamurthy, B.; Arya, D.S. Preclinical evidence for the pharmacological actions of naringin: A review. Planta Med. 2014, 80, 437–451. [Google Scholar] [CrossRef]
- Ye, F.; Jiang, S.; Volshonok, H.; Wu, J.; Zhang, D.Y. Molecular mechanism of anti-prostate cancer activity of Scutellaria baicalensis extract. Nutr. Cancer 2007, 57, 100–110. [Google Scholar] [CrossRef]
- Ye, F.; Wang, H.; Jiang, S.; Wu, J.; Shao, J.; Cheng, X.; Tu, Y.; Zhang, D.Y. Quality evaluation of commercial extracts of Scutellaria baicalensis. Nutr. Cancer 2004, 49, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Nan, J.X.; Park, E.J.; Kim, Y.C.; Ko, G.; Sohn, D.H. Scutellaria baicalensis inhibits liver fibrosis induced by bile duct ligation or carbon tetrachloride in rats. J. Pharm. Pharmacol. 2002, 54, 555–563. [Google Scholar] [CrossRef]
- Sonoda, M.; Nishiyama, T.; Matsukawa, Y.; Moriyasu, M. Cytotoxic activities of flavonoids from two Scutellaria plants in Chinese medicine. J. Ethnopharmacol. 2004, 91, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Dong, Y.; Gao, Y.; Du, Z.; Wang, Y.; Cheng, P.; Chen, A.; Huang, H. The Fascinating Effects of Baicalein on Cancer: A Review. Int. J. Mol. Sci. 2016, 17, 1681. [Google Scholar] [CrossRef]
- Pidgeon, G.P.; Kandouz, M.; Meram, A.; Honn, K.V. Mechanisms controlling cell cycle arrest and induction of apoptosis after 12-lipoxygenase inhibition in prostate cancer cells. Cancer Res. 2002, 62, 2721–2727. [Google Scholar] [PubMed]
- Ling, Y.; Chen, Y.; Chen, P.; Hui, H.; Song, X.; Lu, Z.; Li, C.; Lu, N.; Guo, Q. Baicalein potently suppresses angiogenesis induced by vascular endothelial growth factor through the p53/Rb signaling pathway leading to G1/S cell cycle arrest. Exp. Biol. Med. 2011, 236, 851–858. [Google Scholar] [CrossRef] [PubMed]
- Chao, J.I.; Su, W.C.; Liu, H.F. Baicalein induces cancer cell death and proliferation retardation by the inhibition of CDC2 kinase and survivin associated with opposite role of p38 mitogen-activated protein kinase and AKT. Mol. Cancer Ther. 2007, 6, 3039–3048. [Google Scholar] [CrossRef]
- Zaczek, A.; Brandt, B.; Bielawski, K.P. The diverse signaling network of EGFR, HER2, HER3 and HER4 tyrosine kinase receptors and the consequences for therapeutic approaches. Histol. Histopathol. 2005, 20, 1005–1015. [Google Scholar]
- Capdevila, J.; Elez, E.; Macarulla, T.; Ramos, F.J.; Ruiz-Echarri, M.; Tabernero, J. Anti-epidermal growth factor receptor monoclonal antibodies in cancer treatment. Cancer Treat. Rev. 2009, 35, 354–363. [Google Scholar] [CrossRef]
- Gao, J.; Li, H.R.; Jin, C.; Jiang, J.H.; Ding, J.Y. Strategies to overcome acquired resistance to EGFR TKI in the treatment of non-small cell lung cancer. Clin. Transl. Oncol. 2019, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Barnes, S.; Peterson, T.G. Biochemical targets of the isoflavone genistein in tumor cell lines. Proc. Soc. Exp. Biol. Med. 1995, 208, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Matsukawa, Y.; Marui, N.; Sakai, T.; Satomi, Y.; Yoshida, M.; Matsumoto, K.; Nishino, H.; Aoike, A. Genistein arrests cell cycle progression at G2-M. Cancer Res. 1993, 53, 1328–1331. [Google Scholar] [PubMed]
- Ahmed, A.A.; Goldsmith, J.; Fokt, I.; Le, X.F.; Krzysko, K.A.; Lesyng, B.; Bast, R.C., Jr.; Priebe, W. A genistein derivative, ITB-301, induces microtubule depolymerization and mitotic arrest in multidrug-resistant ovarian cancer. Cancer Chemother. Pharmacol. 2011, 68, 1033–1044. [Google Scholar] [CrossRef] [PubMed]
- Cappelletti, V.; Fioravanti, L.; Miodini, P.; di Fronzo, G. Genistein blocks breast cancer cells in the G(2)M phase of the cell cycle. J. Cell Biochem. 2000, 79, 594–600. [Google Scholar] [CrossRef]
- Nakayama, Y.; Saito, Y.; Soeda, S.; Iwamoto, E.; Ogawa, S.; Yamagishi, N.; Kuga, T.; Yamaguchi, N. Genistein induces cytokinesis failure through RhoA delocalization and anaphase chromosome bridging. J. Cell Biochem. 2014, 115, 763–771. [Google Scholar] [CrossRef]
- Ismail, I.A.; Kang, K.S.; Lee, H.A.; Kim, J.W.; Sohn, Y.K. Genistein-induced neuronal apoptosis and G2/M cell cycle arrest is associated with MDC1 up-regulation and PLK1 down-regulation. Eur. J. Pharmacol. 2007, 575, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Acharya, B.R.; Bhattacharyya, B.; Chakrabarti, G. Genistein arrests cell cycle progression of A549 cells at the G2/M phase and depolymerizes interphase microtubules through binding to a unique site of tubulin. Biochemistry 2010, 49, 1702–1712. [Google Scholar] [CrossRef]
- Brzezinski, A.; Debi, A. Phytoestrogens: The “natural” selective estrogen receptor modulators? Eur. J. Obstet. Gynecol. Reprod. Biol. 1999, 85, 47–51. [Google Scholar] [CrossRef]
- Kuiper, G.G.; Lemmen, J.G.; Carlsson, B.; Corton, J.C.; Safe, S.H.; van der Saag, P.T.; van der Burg, B.; Gustafsson, J.A. Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor β. Endocrinology 1998, 139, 4252–4263. [Google Scholar] [CrossRef]
- Paruthiyil, S.; Parmar, H.; Kerekatte, V.; Cunha, G.R.; Firestone, G.L.; Leitman, D.C. Estrogen receptor β inhibits human breast cancer cell proliferation and tumor formation by causing a G2 cell cycle arrest. Cancer Res. 2004, 64, 423–428. [Google Scholar] [CrossRef]
- Jordan, V.C. The new biology of estrogen-induced apoptosis applied to treat and prevent breast cancer. Endocr. Relat. Cancer 2015, 22, R1–R31. [Google Scholar] [CrossRef]
- Obiorah, I.E.; Fan, P.; Jordan, V.C. Breast cancer cell apoptosis with phytoestrogens is dependent on an estrogen-deprived state. Cancer Prev. Res. 2014, 7, 939–949. [Google Scholar] [CrossRef] [PubMed]
- Shike, M.; Doane, A.S.; Russo, L.; Cabal, R.; Reis-Filho, J.S.; Gerald, W.; Cody, H.; Khanin, R.; Bromberg, J.; Norton, L. The effects of soy supplementation on gene expression in breast cancer: A randomized placebo-controlled study. J. Natl. Cancer Inst. 2014, 106, dju189. [Google Scholar] [CrossRef]
- Montgomery, J.S.; Price, D.K.; Figg, W.D. The androgen receptor gene and its influence on the development and progression of prostate cancer. J. Pathol. 2001, 195, 138–146. [Google Scholar] [CrossRef]
- Davis, J.N.; Muqim, N.; Bhuiyan, M.; Kucuk, O.; Pienta, K.J.; Sarkar, F.H. Inhibition of prostate specific antigen expression by genistein in prostate cancer cells. Int. J. Oncol. 2000, 16, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Basak, S.; Pookot, D.; Noonan, E.J.; Dahiya, R. Genistein down-regulates androgen receptor by modulating HDAC6-Hsp90 chaperone function. Mol. Cancer Ther. 2008, 7, 3195–3202. [Google Scholar] [CrossRef] [PubMed]
- Lazarevic, B.; Boezelijn, G.; Diep, L.M.; Kvernrod, K.; Ogren, O.; Ramberg, H.; Moen, A.; Wessel, N.; Berg, R.E.; Egge-Jacobsen, W.; et al. Efficacy and safety of short-term genistein intervention in patients with localized prostate cancer prior to radical prostatectomy: A randomized, placebo-controlled, double-blind Phase 2 clinical trial. Nutr. Cancer 2011, 63, 889–898. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, N.; Song, Y.H.; De, R. Recent progress on biocompatible nanocarrier-based genistein delivery systems in cancer therapy. J. Drug Target. 2019, 27, 394–407. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Ren, K.; Song, Z.; Li, D.; Quan, M.; Zheng, Y.; Cao, J.; Zeng, W.; Zou, H. 7-Difluoromethoxyl-5′,4′-di-n-octyl genistein inhibits the stem-like characteristics of gastric cancer stem-like cells and reverses the phenotype of epithelial-mesenchymal transition in gastric cancer cells. Onocol. Rep. 2016, 36, 1157–1165. [Google Scholar] [CrossRef] [PubMed]
| Flavonoid (Class) | Molecular targets | Model | Ref |
|---|---|---|---|
| Apigenin (Flavones) | JAK2/STAT3 | SGC-7901 (stomach), xenograft mouse model | [31] |
| p53/p21Cip1 | HCT-116 (colon), APCMin/+ mouse model | [32] | |
| Nrf2 | BEL7402/ADM (liver) | [33] | |
| Baicalein (Flavones) | Rb/E2F/cyclin-CDK4/p53 | HepG2 (liver) | [34] |
| MAPKs | HCT116, SW480 (colon), MG-63 (osteosarcoma) MDA-MB-231, MCF-7 (breast), xenograft model | [35,36,37,38] | |
| PI3K/AKT | HeLa, SiHa (cervix), MG-63 (osteosarcoma) | [38,39] | |
| Luteolin (Flavones) | AKT | A375 (skin), A549 (lung), SK-Hep-1 (liver), xenograft mouse model | [40,41,42] |
| Nrf2 | A549 (lung), xenograft mouse model | [43] | |
| ROS/ER stress | U251MG, U87MG (glioma), xenograft mouse model | [44] | |
| Oroxylin A (Flavones) | p53 | HepG2 (liver) | [45] |
| Bcl-2 | HCT-116 (colon), HeLa (cervix), xenograft model | [46,47] | |
| ERK/MAPK | MDA-MB-231 (breast), A549 (lung) | [48,49] | |
| Notch pathway | MCF-7 (breast) | [50] | |
| CXCL12 | K562, KU812, xenograft mouse model | [51,52] | |
| STAT3 | K562, xenograft mouse model | [53] | |
| Wogonin (Flavones) | p53 | HCT-116, xenograft mouse model | [54,55] |
| PI3K-AKT | MCF-7 (breast), HL-60 (leukemia) | [56,57] | |
| MMP-9 | MHCC97L, PLC/PRF/5, CD133+CAL72 | [58,59,60] | |
| HIF-1α | MCF-7, MDA-MB-231 (breast), xenograft model | [61] | |
| cFLIPL and IAP | A549 (lung), xenograft mouse model | [62] | |
| Nrf2/ARE | K562/A02 (leukemia) | [63] | |
| Kaempferol (Flavonols) | PI3K/AKT | HeLa (cervix), HCCC9810, QBC939 (liver), xenograft mouse model | [64,65] |
| EGFR/ERK | Miapaca-2, Panc-1, SNU-213 (pancreas) | [66] | |
| ERRα/γ | HeLa (cervix), HepG2 (liver), A549 (lung) | [67] | |
| IRE1-JNK-CHOP | AGS, SNU-638 (stomach) | [68] | |
| Quercetin (Flavonols) | BCL2/BAX | A549 (lung) | [69] |
| AMPK/COX-2 | MCF-7 (breast), HT-29 (colon) | [70] | |
| p53 | MDA-MB-231 (breast) | [71] | |
| EGFR/VEGFR2 | MCF-7, MDA-MB-231 (breast) | [72] | |
| β-catenin | Pancreatic cancer stem-like cells | [73] | |
| Myricetin (Flavonols) | NDPK | HCT-15 (colon) | [74] |
| mTOR/AKT | HepG2 (liver) | [75] | |
| BMP-2/Smad, MAPKs | Human periodontal ligament stem cells | [76] | |
| PIM1/CXCR4 | PC3, DU145 (prostate), xenograft mouse model | [77] | |
| Wnt/β-catenin | APCMin/+ mouse model | [78] | |
| Hesperidin (Flavanones) | p53, Bax, caspases-3 | H522 (lung), gall bladder carcinoma cell | [79,80] |
| SDF-1/CXCR-4 | A549 (lung) | [81] | |
| PI3K/AKT | Rat (liver) | [82] | |
| Nrf2/ARE/HO-1 | Rat (liver) | [83] | |
| Naringin (Flavanones) | AKT/mTOR | H69AR (lung) | [84] |
| EGFR, NEU3 | A549 (lung) | [85] | |
| HER2 | SK-BR-3, MDA-MB-231 (breast) | [86] | |
| Smad3/Smad7 | Mouse melanoma | [87] | |
| MAPKs | JAR, JEG-3 (placenta) U87, U373, U251 (glioma) | [88,89,90] | |
| Zeb1 | MG63, U2OS (osteosarcoma) | [91] | |
| ERRα/VEGF/KDR | HUVECs (endothelium) | [92] | |
| ATF3 | HCT116, SW480 (colon) | [93] | |
| Genistein (Isoflavonones) | EGFR | A431 (skin), MCF-7, BT20, ZR-75-1 (breast) | [94,95] |
| CDK | Jurkat T (leukemia), MCF-7, MDA-MB-231 (breast) | [96,97] | |
| KIF20A | SGC-79019 (stomach) | [98] | |
| PLK1 | MCF-7, BT20 (breast), H1299 (lung), HeLa (cervix) HepG2 (hepatoma), LNCaP, PC-3 (prostate) | [23,99,100] | |
| ER α/β | MCF-7 (breast) | [101] | |
| AR | Mouse tissues | [102] |
| Signaling Pathway | Flavonoids | Ref |
|---|---|---|
| MAPK pathway | Baicalein Oroxylin A Myricetin Naringin | [36,37,38] [49] [76] [88,89,90] |
| PI3K/AKT pathway | Baicalein Luteolin Wogonin Kaempferol Hesperidin Naringin | [39] [40] [56,57] [64,65] [82] [84] |
| p53 pathway | Apigenin Baicalein Oroxylin A Wogonin Kaempferol Quercetin Hesperidin Genistein | [32] [34] [45] [54,55] [64] [71] [80] [99] |
| Apoptosis | Oroxylin A Wogonin Kaempferol Quercetin Myricetin Hesperidin | [46,47] [103,104] [64,65] [69] [74] [79,80] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chae, H.-S.; Xu, R.; Won, J.-Y.; Chin, Y.-W.; Yim, H. Molecular Targets of Genistein and Its Related Flavonoids to Exert Anticancer Effects. Int. J. Mol. Sci. 2019, 20, 2420. https://doi.org/10.3390/ijms20102420
Chae H-S, Xu R, Won J-Y, Chin Y-W, Yim H. Molecular Targets of Genistein and Its Related Flavonoids to Exert Anticancer Effects. International Journal of Molecular Sciences. 2019; 20(10):2420. https://doi.org/10.3390/ijms20102420
Chicago/Turabian StyleChae, Hee-Sung, Rong Xu, Jae-Yeon Won, Young-Won Chin, and Hyungshin Yim. 2019. "Molecular Targets of Genistein and Its Related Flavonoids to Exert Anticancer Effects" International Journal of Molecular Sciences 20, no. 10: 2420. https://doi.org/10.3390/ijms20102420
APA StyleChae, H.-S., Xu, R., Won, J.-Y., Chin, Y.-W., & Yim, H. (2019). Molecular Targets of Genistein and Its Related Flavonoids to Exert Anticancer Effects. International Journal of Molecular Sciences, 20(10), 2420. https://doi.org/10.3390/ijms20102420






