The Cancer Prevention, Anti-Inflammatory and Anti-Oxidation of Bioactive Phytochemicals Targeting the TLR4 Signaling Pathway
Abstract
1. Introduction
2. Inflammation and Cancer Progression
3. Toll-Like Receptor
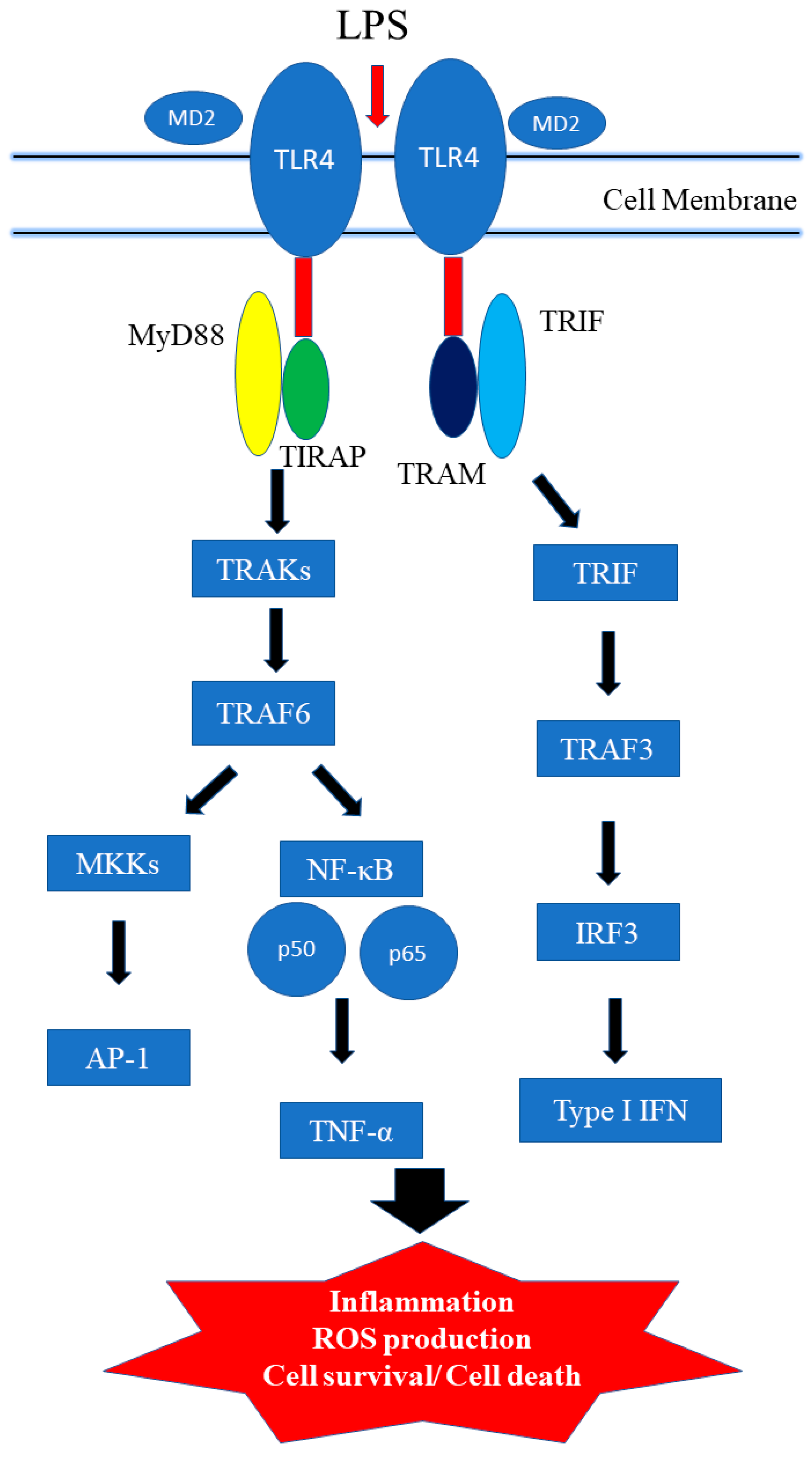
3.1. TLR4 Signaling Pathways
3.2. TLR4, NF-κB in Inflammatory Process and Tumorigenesis
4. Bioactive Phytochemicals Targeting TLR4 and Oxidative Stress
4.1. Zingiberaceae Family
4.1.1. Curcumin
4.1.2. Ginger
4.2. EGCG
4.3. Luteolin
4.4. Quercetin
4.5. Resveratrol
4.6. Caffeic acid Phenethyl Ester
4.7. Xanthohumol
4.8. Genistein
4.9. Berberine
4.10. Sulforaphane
5. Prospective and Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kimbrell, D.A.; Beutler, B. The evolution and genetics of innate immunity. Nat. Rev. Genet. 2001, 2, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V. Innate lymphoid cells: New paradigm in immunology of inflammation. Immunol. Lett. 2014, 157, 23–37. [Google Scholar] [CrossRef] [PubMed]
- Kolaczkowska, E.; Kubes, P. Neutrophil recruitment and function in health and inflammation. Nat. Rev. Immunol. 2013, 13, 159–175. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, S.; Rosowski, E.E.; Huttenlocher, A. Neutrophil migration in infection and wound repair: Going forward in reverse. Nat. Rev. Immunol. 2016, 16, 378–391. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Sharma, A. Innate immunity in sepsis pathogenesis and its modulation: New immunomodulatory targets revealed. J. Chemother. 2008, 20, 672–683. [Google Scholar] [CrossRef] [PubMed]
- Murillo, L.S.; Morre, S.A.; Pena, A.S. Toll-like receptors and NOD/CARD proteins: Pattern recognition receptors are key elements in the regulation of immune response. Drugs Today 2003, 39, 415–438. [Google Scholar] [CrossRef] [PubMed]
- Areschoug, T.; Gordon, S. Pattern recognition receptors and their role in innate immunity: Focus on microbial protein ligands. Contrib. Microbiol. 2008, 15, 45–60. [Google Scholar] [PubMed]
- Kumagai, Y.; Akira, S. Identification and functions of pattern-recognition receptors. J. Allergy Clin. Immunol. 2010, 125, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.; Zhang, H.; Fu, Y.; Zhu, Y.; Kong, L.; Chen, L.; Zhao, F.; Yu, L.; Chen, X. TLR4/MyD88 signaling determines the metastatic potential of breast cancer cells. Mol. Med. Rep. 2018, 18, 3411–3420. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yu, K.; Zhang, X.; Yu, S. Dual functional roles of the MyD88 signaling in colorectal cancer development. Biomed. Pharmacother. 2018, 107, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.H.; Kho, D.T.; O’Carroll, S.J.; Angel, C.E.; Graham, E.S. The functional and inflammatory response of brain endothelial cells to toll-like receptor agonists. Sci. Rep. 2018, 8, 10102. [Google Scholar] [CrossRef] [PubMed]
- Dajon, M.; Iribarren, K.; Cremer, I. Toll-like receptor stimulation in cancer: A pro- and anti-tumor double-edged sword. Immunobiology 2017, 222, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, D.S.; Selvam, A.P.; Rajeswari, V.D. Effective anti-cancer property of Pouteria sapota leaf on breast cancer cell lines. Biochem. Biophys. Rep. 2018, 15, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Koh, Y.C.; Yang, G.; Lai, C.S.; Weerawatanakorn, M.; Pan, M.H. Chemopreventive effects of phytochemicals and medicines on M1/M2 polarized macrophage role in inflammation-related diseases. Int. J. Mol. Sci. 2018, 19, 2208. [Google Scholar] [CrossRef] [PubMed]
- Koo, J.E.; Park, Z.Y.; Kim, N.D.; Lee, J.Y. Sulforaphane inhibits the engagement of LPS with TLR4/MD2 complex by preferential binding to Cys133 in MD2. Biochem. Biophys. Res. Commun. 2013, 434, 600–605. [Google Scholar] [CrossRef] [PubMed]
- Bjorkbacka, H.; Fitzgerald, K.A.; Huet, F.; Li, X.; Gregory, J.A.; Lee, M.A.; Ordija, C.M.; Dowley, N.E.; Golenbock, D.T.; Freeman, M.W. The induction of macrophage gene expression by LPS predominantly utilizes Myd88-independent signaling cascades. Physiol. Genom. 2004, 19, 319–330. [Google Scholar] [CrossRef] [PubMed]
- Minihane, A.M.; Vinoy, S.; Russell, W.R.; Baka, A.; Roche, H.M.; Tuohy, K.M.; Teeling, J.L.; Blaak, E.E.; Fenech, M.; Vauzour, D.; et al. Low-grade inflammation, diet composition and health: Current research evidence and its translation. Br. J. Nutr. 2015, 114, 999–1012. [Google Scholar] [CrossRef] [PubMed]
- Das, U.N. Arachidonic acid in health and disease with focus on hypertension and diabetes mellitus: A review. J. Adv. Res. 2018, 11, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Allavena, P.; Sica, A.; Solinas, G.; Porta, C.; Mantovani, A. The inflammatory micro-environment in tumor progression: The role of tumor-associated macrophages. Crit. Rev. Oncol. Hematol. 2008, 66, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Balkwill, F. TNF-α in promotion and progression of cancer. Cancer Metastasis Rev. 2006, 25, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Echizen, K.; Oshima, H.; Nakayama, M.; Oshima, M. The inflammatory microenvironment that promotes gastrointestinal cancer development and invasion. Adv. Biol. Regul. 2018, 68, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Bravo, D.; Hoare, A.; Soto, C.; Valenzuela, M.A.; Quest, A.F. Helicobacter pylori in human health and disease: Mechanisms for local gastric and systemic effects. World J. Gastroenterol. 2018, 24, 3071–3089. [Google Scholar] [CrossRef] [PubMed]
- Leung, W.K.; Wong, I.O.L.; Cheung, K.S.; Yeung, K.F.; Chan, E.W.; Wong, A.Y.S.; Chen, L.; Wong, I.C.K.; Graham, D.Y. Effects of helicobacter pylori treatment on incidence of gastric cancer in older individuals. Gastroenterology 2018, 155, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Doorakkers, E.; Lagergren, J.; Engstrand, L.; Brusselaers, N. Helicobacter pylori eradication treatment and the risk of gastric adenocarcinoma in a Western population. Gut 2018. [Google Scholar] [CrossRef] [PubMed]
- Madden, C.R.; Finegold, M.J.; Slagle, B.L. Hepatitis B virus X protein acts as a tumor promoter in development of diethylnitrosamine-induced preneoplastic lesions. J. Virol. 2001, 75, 3851–3858. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Luo, D.Z.; Liu, L.; Feng, Z.B.; Guo, F.; Li, P. Hepatic local micro-environmental immune status in hepatocellular carcinoma and cirrhotic tissues. West Indian Med. J. 2006, 55, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Karin, M. Nuclear factor-κB in cancer development and progression. Nature 2006, 441, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, L.; Vanden Berghe, W.; Haegeman, G. Regulation of NF-κB transcriptional activity. Cancer Treat. Res. 2006, 130, 89–102. [Google Scholar] [PubMed]
- De Freitas, R.P.; Greatti, V.R.; Alcalde, M.P.; Cavenago, B.C.; Vivan, R.R.; Duarte, M.A.; Weckwerth, A.C.; Weckwerth, P.H. Effect of the association of nonsteroidal anti-inflammatory and antibiotic drugs on antibiofilm activity and pH of calcium hydroxide pastes. J. Endod. 2017, 43, 131–134. [Google Scholar] [CrossRef] [PubMed]
- Meintjes, G.; Scriven, J.; Marais, S. Management of the immune reconstitution inflammatory syndrome. Curr. HIV/AIDS Rep. 2012, 9, 238–250. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R.; Janeway, C., Jr. The Toll receptor family and microbial recognition. Trends Microbiol. 2000, 8, 452–456. [Google Scholar] [CrossRef]
- Medzhitov, R.; Janeway, C., Jr. Innate immune recognition: Mechanisms and pathways. Immunol. Rev. 2000, 173, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R.; Janeway, C.A., Jr. Innate immune recognition and control of adaptive immune responses. Semin. Immunol. 1998, 10, 351–353. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Akira, S. Role of toll-like receptor in innate immunity. Tanpakushitsu Kakusan Koso 2002, 47 (Suppl. 16), 2097–2102. [Google Scholar] [PubMed]
- Takeda, K.; Akira, S. Toll-like receptors. Curr. Protoc. Immunol. 2015, 109, 1–10. [Google Scholar]
- Takeda, K.; Akira, S. Toll-like receptors in innate immunity. Int. Immunol. 2005, 17, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lavelle, E.C.; Murphy, C.; O’Neill, L.A.; Creagh, E.M. The role of TLRs, NLRs, and RLRs in mucosal innate immunity and homeostasis. Mucosal Immunol. 2010, 3, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Akira, S. The roles of TLRs, RLRs and NLRs in pathogen recognition. Int. Immunol. 2009, 21, 317–337. [Google Scholar] [CrossRef] [PubMed]
- McClure, R.; Massari, P. TLR-dependent human mucosal epithelial cell responses to microbial pathogens. Front. Immunol. 2014, 5, 386–399. [Google Scholar] [CrossRef] [PubMed]
- Kumar, H.; Kawai, T.; Akira, S. Toll-like receptors and innate immunity. Biochem. Biophys. Res. Commun. 2009, 388, 621–625. [Google Scholar] [CrossRef] [PubMed]
- Coorens, M.; Schneider, V.A.F.; de Groot, A.M.; van Dijk, A.; Meijerink, M.; Wells, J.M.; Scheenstra, M.R.; Veldhuizen, E.J.A.; Haagsman, H.P. Cathelicidins inhibit Escherichia coli-induced TLR2 and TLR4 activation in a viability-dependent manner. J. Immunol. 2017, 199, 1418–1428. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Sakabe, J.; Kamiya, K.; Funakoshi, A.; Tokura, Y. The Vitamin D3 analogue calcipotriol suppresses CpG-activated TLR9-MyD88 signalling in murine plasmacytoid dendritic cells. Clin. Exp. Dermatol. 2018, 43, 445–448. [Google Scholar] [CrossRef] [PubMed]
- Rahimifard, M.; Maqbool, F.; Moeini-Nodeh, S.; Niaz, K.; Abdollahi, M.; Braidy, N.; Nabavi, S.M.; Nabavi, S.F. Targeting the TLR4 signaling pathway by polyphenols: A novel therapeutic strategy for neuroinflammation. Ageing Res. Rev. 2017, 36, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Balistreri, C.R.; Ruvolo, G.; Lio, D.; Madonna, R. Toll-like receptor-4 signaling pathway in aorta aging and diseases: “Its double nature”. J. Mol. Cell. Cardiol. 2017, 110, 38–53. [Google Scholar] [CrossRef] [PubMed]
- Cen, X.; Liu, S.; Cheng, K. The Role of Toll-Like Receptor in Inflammation and Tumor Immunity. Front. Pharmacol. 2018, 9, 878. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, K.A.; Palsson-McDermott, E.M.; Bowie, A.G.; Jefferies, C.A.; Mansell, A.S.; Brady, G.; Brint, E.; Dunne, A.; Gray, P.; Harte, M.T.; et al. Mal (MyD88-adapter-like) is required for toll-like receptor-4 signal transduction. Nature 2001, 413, 78–83. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, L.A. Toll-like receptor signal transduction and the tailoring of innate immunity: A role for Mal? Trends Immunol. 2002, 23, 296–300. [Google Scholar] [CrossRef]
- Schilling, D.; Thomas, K.; Nixdorff, K.; Vogel, S.N.; Fenton, M.J. Toll-like receptor 4 and Toll-IL-1 receptor domain-containing adapter protein (TIRAP)/myeloid differentiation protein 88 adapter-like (Mal) contribute to maximal IL-6 expression in macrophages. J. Immunol. 2002, 169, 5874–5880. [Google Scholar] [CrossRef] [PubMed]
- Vogel, S.N.; Fenton, M. Toll-like receptor 4 signalling: New perspectives on a complex signal-transduction problem. Biochem. Soc. Trans. 2003, 31 Pt 3, 664–668. [Google Scholar] [CrossRef]
- Nagai, Y.; Akashi, S.; Nagafuku, M.; Ogata, M.; Iwakura, Y.; Akira, S.; Kitamura, T.; Kosugi, A.; Kimoto, M.; Miyake, K. Essential role of MD-2 in LPS responsiveness and TLR4 distribution. Nat. Immunol. 2002, 3, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Keating, S.E.; Maloney, G.M.; Moran, E.M.; Bowie, A.G. IRAK-2 participates in multiple toll-like receptor signaling pathways to NFκB via activation of TRAF6 ubiquitination. J. Biol. Chem. 2007, 282, 33435–33443. [Google Scholar] [CrossRef] [PubMed]
- Doyle, S.L.; O’Neill, L.A. Toll-like receptors: From the discovery of NFκB to new insights into transcriptional regulations in innate immunity. Biochem. Pharmacol. 2006, 72, 1102–1113. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Akira, S. Microbial recognition by Toll-like receptors. J. Dermatol. Sci. 2004, 34, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Zhou, H.; Feng, P.; Zhou, X.; Wen, H.; Xie, X.; Shen, H.; Zhu, X. Reduced expression of toll-like receptor 4 inhibits human breast cancer cells proliferation and inflammatory cytokines secretion. J. Exp. Clin. Cancer Res. 2010, 29, 92. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Zhang, Y.; Zhang, Q.; Wang, F.; Zhang, D. Toll-like receptors and prostate cancer. Front. Immunol. 2014, 5, 352. [Google Scholar] [CrossRef] [PubMed]
- Moradi-Marjaneh, R.; Hassanian, S.M.; Fiuji, H.; Soleimanpour, S.; Ferns, G.A.; Avan, A.; Khazaei, M. Toll like receptor signaling pathway as a potential therapeutic target in colorectal cancer. J. Cell. Physiol. 2018, 233, 5613–5622. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, N.; Nasti, T.H.; Long, J.A.; Naseemuddin, M.; Lucas, A.P.; Xu, H.; Elmets, C.A. Protective role of Toll-like receptor 4 during the initiation stage of cutaneous chemical carcinogenesis. Cancer Res. 2008, 68, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Kuzmich, N.N.; Sivak, K.V.; Chubarev, V.N.; Porozov, Y.B.; Savateeva-Lyubimova, T.N.; Peri, F. TLR4 signaling pathway modulators as potential therapeutics in inflammation and sepsis. Vaccines 2017, 5, 34. [Google Scholar] [CrossRef] [PubMed]
- Dei Cas, M.; Ghidoni, R. Cancer prevention and therapy with polyphenols: Sphingolipid-mediated mechanisms. Nutrients 2018, 10, 940. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.; Chinkwo, K.A.; Santhakumar, A.B.; Blanchard, C.L. Inhibitory effects of pulse bioactive compounds on cancer development pathways. Diseases 2018, 6, 72. [Google Scholar] [CrossRef] [PubMed]
- Hasani-Ranjbar, S.; Jouyandeh, Z.; Abdollahi, M. A systematic review of anti-obesity medicinal plants—An update. J. Diabetes Metab. Disord. 2013, 12, 28. [Google Scholar] [CrossRef] [PubMed]
- Hasani-Ranjbar, S.; Larijani, B.; Abdollahi, M. A systematic review of the potential herbal sources of future drugs effective in oxidant-related diseases. Inflamm. Allergy Drug Targets 2009, 8, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, M.; Varoni, E.M.; Salehi, B.; Sharifi-Rad, J.; Matthews, K.R.; Ayatollahi, S.A.; Kobarfard, F.; Ibrahim, S.A.; Mnayer, D.; Zakaria, Z.A.; et al. Plants of the genus zingiber as a source of bioactive phytochemicals: From tradition to pharmacy. Molecules 2017, 22, 2145. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, T. Curcumin as a functional food-derived factor: Degradation products, metabolites, bioactivity, and future perspectives. Food Funct. 2018, 9, 705–714. [Google Scholar] [CrossRef] [PubMed]
- Kocaadam, B.; Sanlier, N. Curcumin, an active component of turmeric (Curcuma longa), and its effects on health. Crit. Rev. Food Sci. Nutr. 2017, 57, 2889–2895. [Google Scholar] [CrossRef] [PubMed]
- Lestari, M.L.; Indrayanto, G. Curcumin. Profiles Drug Subst. Excip. Relat. Methodol. 2014, 39, 113–204. [Google Scholar] [PubMed]
- Jamwal, R. Bioavailable curcumin formulations: A review of pharmacokinetic studies in healthy volunteers. J. Integr. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Gao, R.; Cao, Y.; Guo, M.; Wei, Z.; Zhou, E.; Li, Y.; Yao, M.; Yang, Z.; Zhang, N. Curcumin attenuates inflammatory responses by suppressing TLR4-mediated NF-κB signaling pathway in lipopolysaccharide-induced mastitis in mice. Int. Immunopharmacol. 2014, 20, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Meng, Z.; Yan, C.; Deng, Q.; Gao, D.F.; Niu, X.L. Curcumin inhibits LPS-induced inflammation in rat vascular smooth muscle cells in vitro via ROS-relative TLR4-MAPK/NF-κB pathways. Acta Pharmacol. Sin. 2013, 34, 901–911. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.T.; Bian, C.; Yuan, J.C.; Chu, W.H.; Xiang, X.; Chen, F.; Wang, C.S.; Feng, H.; Lin, J.K. Curcumin attenuates acute inflammatory injury by inhibiting the TLR4/MyD88/NF-κB signaling pathway in experimental traumatic brain injury. J. Neuroinflamm. 2014, 11, 59. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Zhan, L.; Liao, H.; Chen, L.; Lv, X. Curcumin improves TNBS-induced colitis in rats by inhibiting IL-27 expression via the TLR4/NF-κB signaling pathway. Plant. Med. 2013, 79, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Ren, B.; Luo, S.; Tian, X.; Jiang, Z.; Zou, G.; Xu, F.; Yin, T.; Huang, Y.; Liu, J. Curcumin inhibits liver cancer by inhibiting DAMP molecule HSP70 and TLR4 signaling. Oncol. Rep. 2018, 40, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Oboh, G.; Akinyemi, A.J.; Ademiluyi, A.O. Antioxidant and inhibitory effect of red ginger (Zingiber officinale var. Rubra) and white ginger (Zingiber officinale Roscoe) on Fe2+ induced lipid peroxidation in rat brain in vitro. Exp. Toxicol. Pathol. 2012, 64, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.M.; Kao, C.L.; Tseng, Y.T.; Lo, Y.C.; Chen, C.Y. Ginger Phytochemicals Inhibit Cell Growth and Modulate Drug Resistance Factors in Docetaxel Resistant Prostate Cancer Cell. Molecules 2017, 22, 1477. [Google Scholar] [CrossRef] [PubMed]
- Kubra, I.R.; Rao, L.J. An impression on current developments in the technology, chemistry, and biological activities of ginger (Zingiber officinale Roscoe). Crit. Rev. Food Sci. Nutr. 2012, 52, 651–688. [Google Scholar] [CrossRef] [PubMed]
- Li, X.H.; McGrath, K.C.; Tran, V.H.; Li, Y.M.; Duke, C.C.; Roufogalis, B.D.; Heather, A.K. Attenuation of Proinflammatory Responses by S-[6]-Gingerol via Inhibition of ROS/NF-κB/COX2 Activation in HuH7 Cells. Evid. Based Complement. Alternat. Med. 2013, 2013, 146142. [Google Scholar] [CrossRef] [PubMed]
- Dongare, S.; Gupta, S.K.; Mathur, R.; Saxena, R.; Mathur, S.; Agarwal, R.; Nag, T.C.; Srivastava, S.; Kumar, P. Zingiber officinale attenuates retinal microvascular changes in diabetic rats via anti-inflammatory and antiangiogenic mechanisms. Mol. Vis. 2016, 22, 599–609. [Google Scholar] [PubMed]
- Tuorkey, M.J. Cancer therapy with phytochemicals: Present and future perspectives. Biomed. Environ. Sci. 2015, 28, 808–819. [Google Scholar] [CrossRef]
- Han, Q.; Yuan, Q.; Meng, X.; Huo, J.; Bao, Y.; Xie, G. 6-Shogaol attenuates LPS-induced inflammation in BV2 microglia cells by activating PPAR-γ. Oncotarget 2017, 8, 42001–42006. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.H.; Hsieh, M.C.; Hsu, P.C.; Ho, S.Y.; Lai, C.S.; Wu, H.; Sang, S.; Ho, C.T. 6-Shogaol suppressed lipopolysaccharide-induced up-expression of iNOS and COX-2 in murine macrophages. Mol. Nutr. Food Res. 2008, 52, 1467–1477. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.I.; Lee, J.K.; Youn, H.S. Inhibition of homodimerization of toll-like receptor 4 by 6-shogaol. Mol. Cells 2009, 27, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Park, S.J.; Lee, M.Y.; Son, B.S.; Youn, H.S. TBK1-targeted suppression of TRIF-dependent signaling pathway of Toll-like receptors by 6-shogaol, an active component of ginger. Biosci. Biotechnol. Biochem. 2009, 73, 1474–1478. [Google Scholar] [CrossRef] [PubMed]
- Koh, E.M.; Kim, H.J.; Kim, S.; Choi, W.H.; Choi, Y.H.; Ryu, S.Y.; Kim, Y.S.; Koh, W.S.; Park, S.Y. Modulation of macrophage functions by compounds isolated from Zingiber officinale. Plant. Med. 2009, 75, 148–151. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.Y.; Park, S.H.; Lee, M.; Kim, H.J.; Ryu, S.Y.; Kim, N.D.; Hwang, B.Y.; Hong, J.T.; Han, S.B.; Kim, Y. 1-Dehydro-[10]-gingerdione from ginger inhibits IKKβ activity for NF-κB activation and suppresses NF-κB-regulated expression of inflammatory genes. Br. J. Pharmacol. 2012, 167, 128–140. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Kyeong, M.S.; Hwang, Y.; Ryu, S.Y.; Han, S.B.; Kim, Y. Inhibition of LPS binding to MD-2 co-receptor for suppressing TLR4-mediated expression of inflammatory cytokine by 1-dehydro-10-gingerdione from dietary ginger. Biochem. Biophys. Res. Commun. 2012, 419, 735–740. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, C.; Artacho, R.; Gimenez, R. Beneficial effects of green tea—A review. J. Am. Coll. Nutr. 2006, 25, 79–99. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Mukhtar, H. Tea and health: Studies in humans. Curr. Pharm. Des. 2013, 19, 6141–6147. [Google Scholar] [CrossRef] [PubMed]
- Khoi, P.N.; Park, J.S.; Kim, J.H.; Xia, Y.; Kim, N.H.; Kim, K.K.; Jung, Y.D. (−)-Epigallocatechin-3-gallate blocks nicotine-induced matrix metalloproteinase-9 expression and invasiveness via suppression of NF-κB and AP-1 in endothelial cells. Int. J. Oncol. 2013, 43, 868–876. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Chiou, Y.S.; Pan, M.H.; Shahidi, F. Anti-inflammatory activity of lipophilic epigallocatechin gallate (EGCG) derivatives in LPS-stimulated murine macrophages. Food Chem. 2012, 134, 742–748. [Google Scholar] [CrossRef] [PubMed]
- Youn, H.S.; Lee, J.Y.; Saitoh, S.I.; Miyake, K.; Kang, K.W.; Choi, Y.J.; Hwang, D.H. Suppression of MyD88- and TRIF-dependent signaling pathways of Toll-like receptor by (−)-epigallocatechin-3-gallate, a polyphenol component of green tea. Biochem. Pharmacol. 2006, 72, 850–859. [Google Scholar] [CrossRef] [PubMed]
- Byun, E.B.; Mi, S.; Kim, J.H.; Song, D.S.; Lee, B.S.; Park, J.N.; Park, S.H.; Park, C.; Jung, P.M.; Sung, N.Y.; et al. Epigallocatechin-3-gallate-mediated Tollip induction through the 67-kDa laminin receptor negatively regulating TLR4 signaling in endothelial cells. Immunobiology 2014, 219, 866–872. [Google Scholar] [CrossRef] [PubMed]
- Hong Byun, E.; Fujimura, Y.; Yamada, K.; Tachibana, H. TLR4 signaling inhibitory pathway induced by green tea polyphenol epigallocatechin-3-gallate through 67-kDa laminin receptor. J. Immunol. 2010, 185, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Kuo, M.Y.; Liao, M.F.; Chen, F.L.; Li, Y.C.; Yang, M.L.; Lin, R.H.; Kuan, Y.H. Luteolin attenuates the pulmonary inflammatory response involves abilities of antioxidation and inhibition of MAPK and NFκB pathways in mice with endotoxin-induced acute lung injury. Food Chem. Toxicol. 2011, 49, 2660–2666. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.K.; Kim, S.Y.; Kim, Y.S.; Lee, W.H.; Hwang, D.H.; Lee, J.Y. Suppression of the TRIF-dependent signaling pathway of Toll-like receptors by luteolin. Biochem. Pharmacol. 2009, 77, 1391–1400. [Google Scholar] [CrossRef] [PubMed]
- Spagnuolo, C.; Moccia, S.; Russo, G.L. Anti-inflammatory effects of flavonoids in neurodegenerative disorders. Eur. J. Med. Chem. 2018, 153, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, S.; Sudhakaran, P.R.; Helen, A. Quercetin attenuates atherosclerotic inflammation and adhesion molecule expression by modulating TLR-NF-κB signaling pathway. Cell Immunol. 2016, 310, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.S.; Jeong, D.; Yi, Y.S.; Lee, B.H.; Kim, T.W.; Htwe, K.M.; Kim, Y.D.; Yoon, K.D.; Hong, S.; Lee, W.S.; et al. Myrsine seguinii ethanolic extract and its active component quercetin inhibit macrophage activation and peritonitis induced by LPS by targeting to Syk/Src/IRAK-1. J. Ethnopharmacol. 2014, 151, 1165–1174. [Google Scholar] [CrossRef] [PubMed]
- Elshaer, M.; Chen, Y.; Wang, X.J.; Tang, X. Resveratrol: An overview of its anti-cancer mechanisms. Life Sci. 2018, 207, 340–349. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.W.; Bode, A.M.; Dong, Z. Molecular targets of phytochemicals for cancer prevention. Nat. Rev. Cancer 2011, 11, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Cianciulli, A.; Calvello, R.; Cavallo, P.; Dragone, T.; Carofiglio, V.; Panaro, M.A. Modulation of NF-κB activation by resveratrol in LPS treated human intestinal cells results in downregulation of PGE2 production and COX-2 expression. Toxicol. In Vitro 2012, 26, 1122–1128. [Google Scholar] [CrossRef] [PubMed]
- Youn, H.S.; Lee, J.Y.; Fitzgerald, K.A.; Young, H.A.; Akira, S.; Hwang, D.H. Specific inhibition of MyD88-independent signaling pathways of TLR3 and TLR4 by resveratrol: Molecular targets are TBK1 and RIP1 in TRIF complex. J. Immunol. 2005, 175, 3339–3346. [Google Scholar] [CrossRef] [PubMed]
- Sebai, H.; Ristorcelli, E.; Sbarra, V.; Hovsepian, S.; Fayet, G.; Aouani, E.; Lombardo, D. Protective effect of resveratrol against LPS-induced extracellular lipoperoxidation in AR42J cells partly via a Myd88-dependent signaling pathway. Arch. Biochem. Biophys. 2010, 495, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Panaro, M.A.; Carofiglio, V.; Acquafredda, A.; Cavallo, P.; Cianciulli, A. Anti-inflammatory effects of resveratrol occur via inhibition of lipopolysaccharide-induced NF-κB activation in Caco-2 and SW480 human colon cancer cells. Br. J. Nutr. 2012, 108, 1623–1632. [Google Scholar] [CrossRef] [PubMed]
- Jakus, P.B.; Kalman, N.; Antus, C.; Radnai, B.; Tucsek, Z.; Gallyas, F., Jr.; Sumegi, B.; Veres, B. TRAF6 is functional in inhibition of TLR4-mediated NF-κB activation by resveratrol. J. Nutr. Biochem. 2013, 24, 819–823. [Google Scholar] [CrossRef] [PubMed]
- Murtaza, G.; Karim, S.; Akram, M.R.; Khan, S.A.; Azhar, S.; Mumtaz, A.; Bin Asad, M.H. Caffeic acid phenethyl ester and therapeutic potentials. Biomed. Res. Int. 2014, 2014, 145342. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.C.; Lin, Y.L.; Liang, Y.C.; Yang, Y.H.; Lee, J.H.; Yu, H.H.; Wu, W.M.; Chiang, B.L. The effect of caffeic acid phenethyl ester on the functions of human monocyte-derived dendritic cells. BMC Immunol. 2009, 10, 39. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Koo, J.E.; Seo, Y.J.; Tyagi, N.; Jeong, E.; Choi, J.; Lim, K.M.; Park, Z.Y.; Lee, J.Y. Suppression of Toll-like receptor 4 activation by caffeic acid phenethyl ester is mediated by interference of LPS binding to MD2. Br. J. Pharmacol. 2013, 168, 1933–1945. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Wang, Y.; Yin, X.; Liu, X.; Xuan, H. Ethanol extract of propolis and its constituent caffeic acid phenethyl ester inhibit breast cancer cells proliferation in inflammatory microenvironment by inhibiting TLR4 signal pathway and inducing apoptosis and autophagy. BMC Complement Altern. Med. 2017, 17, 471. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Sun, W.; Wu, T.; Lu, R.; Shi, B. Caffeic acid phenethyl ester attenuates lipopolysaccharide-stimulated proinflammatory responses in human gingival fibroblasts via NF-κB and PI3K/Akt signaling pathway. Eur. J. Pharmacol. 2017, 794, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Nozawa, H.; Daikonnya, A.; Kondo, K.; Kitanaka, S. Inhibitors of nitric oxide production from hops (Humulus lupulus L.). Biol. Pharm. Bull. 2003, 26, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.C.; Kim, H.J.; Kim, Y.J.; Lee, K.Y.; Choi, H.J.; Lee, I.S.; Kang, B.Y. Differential anti-inflammatory pathway by xanthohumol in IFN-γ and LPS-activated macrophages. Int. Immunopharmacol. 2008, 8, 567–753. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Xiao, B.; Chen, L.; Bai, B.; Zhang, Y.; Xu, Z.; Fu, L.; Liu, Z.; Li, X.; Zhao, Y.; et al. Discovery of new MD2-targeted anti-inflammatory compounds for the treatment of sepsis and acute lung injury. Eur. J. Med. Chem. 2017, 139, 726–740. [Google Scholar] [CrossRef] [PubMed]
- Jia, Z.; Babu, P.V.; Si, H.; Nallasamy, P.; Zhu, H.; Zhen, W.; Misra, H.P.; Li, Y.; Liu, D. Genistein inhibits TNF-α-induced endothelial inflammation through the protein kinase pathway A and improves vascular inflammation in C57BL/6 mice. Int. J. Cardiol. 2013, 168, 2637–2645. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Liu, K.; Huang, F.; Zhang, D.; Guo, X.; Wang, M.; Liu, B. Positive and negative regulation of insulin action by genistein in the endothelium. J. Nutr. Biochem. 2013, 24, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.W.; Lee, H.H.; Han, M.H.; Kim, G.Y.; Kim, W.J.; Choi, Y.H. Anti-inflammatory effects of genistein via suppression of the toll-like receptor 4-mediated signaling pathway in lipopolysaccharide-stimulated BV2 microglia. Chem. Biol. Interact. 2014, 212, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.; Bilitewski, U. Effect of genistein on the TLR and MAPK transduction cascades in lipopolysaccharide -stimulated macrophages. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 2014, 30, 233–236. [Google Scholar] [PubMed]
- Llaneza, P.; Gonzalez, C.; Fernandez-Inarrea, J.; Alonso, A.; Diaz, F.; Arnott, I.; Ferrer-Barriendos, J. Soy isoflavones, diet and physical exercise modify serum cytokines in healthy obese postmenopausal women. Phytomedicine 2011, 18, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.Y.; Chen, C.S.; Wu, S.N.; Jong, Y.J.; Lo, Y.C. Berberine activates Nrf2 nuclear translocation and protects against oxidative damage via a phosphatidylinositol 3-kinase/Akt-dependent mechanism in NSC34 motor neuron-like cells. Eur. J. Pharm. Sci. 2012, 46, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.Y.; Chen, L.; Yang, L.; Yu, X.; Kou, J.P.; Yu, B.Y. Berberine inhibits LPS-induced TF procoagulant activity and expression through NF-κB/p65, Akt and MAPK pathway in THP-1 cells. Pharmacol. Rep. 2014, 66, 480–484. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liang, Y.; He, C. Anticancer activities and mechanisms of heat-clearing and detoxicating traditional Chinese herbal medicine. Chin. Med. 2017, 12, 20. [Google Scholar] [CrossRef] [PubMed]
- Feng, M.; Zou, Z.; Zhou, X.; Hu, Y.; Ma, H.; Xiao, Y.; Li, X.; Ye, X. Comparative effect of berberine and its derivative 8-cetylberberine on attenuating atherosclerosis in ApoE(−/−) mice. Int. Immunopharmacol. 2017, 43, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.; Chen, X.; Liu, L.; Zhao, Y.; Huang, W.J.; Zhang, Q.; Miao, G.G.; Chen, W.; Xie, H.G.; Cao, C.C. Berberine ameliorates chronic kidney injury caused by atherosclerotic renovascular disease through the suppression of NFκB signaling pathway in rats. PLoS ONE 2013, 8, e59794. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.E.; Ying Chang, M.; Wei, J.Y.; Chen, Y.J.; Maa, M.C.; Leu, T.H. Berberine reduces Toll-like receptor-mediated macrophage migration by suppression of Src enhancement. Eur. J. Pharmacol. 2015, 757, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Li, G.X.; Wang, X.M.; Jiang, T.; Gong, J.F.; Niu, L.Y.; Li, N. Berberine prevents intestinal mucosal barrier damage during early phase of sepsis in rat through the toll-like receptors signaling pathway. Korean J. Physiol. Pharmacol. 2015, 19, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Youn, H.S.; Kim, Y.S.; Park, Z.Y.; Kim, S.Y.; Choi, N.Y.; Joung, S.M.; Seo, J.A.; Lim, K.M.; Kwak, M.K.; Hwang, D.H.; et al. Sulforaphane suppresses oligomerization of TLR4 in a thiol-dependent manner. J. Immunol. 2010, 184, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Liu, X.; Bao, H.; Zhang, Y.; Wang, X.; Shi, K.; Pang, Q. Effects of sulforaphane on Toll-like receptor 4/myeloid differentiation factor 88 pathway of monocyte-derived macrophages from patients with chronic obstructive pulmonary disease. Zhonghua Jie He He Hu Xi Za Zhi 2014, 37, 250–254. [Google Scholar] [PubMed]
- Kim, S.Y.; Jeong, E.; Joung, S.M.; Lee, J.Y. PI3K/Akt contributes to increased expression of Toll-like receptor 4 in macrophages exposed to hypoxic stress. Biochem. Biophys. Res. Commun. 2012, 419, 466–471. [Google Scholar] [CrossRef] [PubMed]
- Scior, T.; Alexander, C.; Zaehringer, U. Reviewing and identifying amino acids of human, murine, canine and equine TLR4/MD-2 receptor complexes conferring endotoxic innate immunity activation by LPS/lipid A, or antagonistic effects by Eritoran, in contrast to species-dependent modulation by lipid IVa. Comput. Struct. Biotechnol. J. 2013, 5, e201302012. [Google Scholar] [PubMed]
| Effects | Tumor Type |
|---|---|
| Anti-tumor | Skin cancer, mammary cancer, lung cancer |
| Pro-tumor | Breast cancer, lung cancer, head and neck cancer, prostate cancer |
| Compound | Chemical Structure | Molecular Targets |
|---|---|---|
| Curcumin |  | ↓TNF-α, ↓IL-1, ↓IL-6, ↓IL-8, ↓IL-12, ↓MCP-1, ↓IL-1β, ↓TLR4 ↓MAPK ↓NF-κB, ↓HSP70 |
| 6-gingerol | 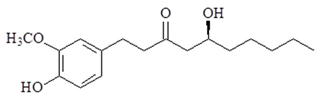 | ↓NF-κB, ↓COX-2, ↓TNF-α, ↓TLR4, ↓ICAM, ↓VCAM, ↓iNOS, ↓VEGF |
| 6-Shogaol |  | ↓TNF-α, ↓IL-1β, ↓IL-6, ↓PGE2, ↓iNOS, ↓COX-2, ↓NF-κB, ↓cyclinD1, ↓survivin, ↓cIAP-1, ↓XIAP, ↓TLR4 |
| 1-Dehydro-10-gingerdione | 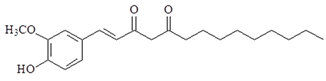 | ↓NO, ↓NF-κB, ↓TNF-α, ↓IL-1β, ↓IRF3, ↓IFN-β, ↓IP-10, ↓MD2 |
| EGCG | 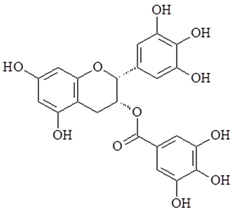 | ↓NF-κB, ↓AP-1, ↓COX-2, ↓NO synthase, ↓TNF-α, ↓TLR4, ↑Tollip |
| Luteolin | 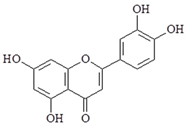 | ↓NF-κB, ↓TNF-α, ↓ICAM-1, ↓TBK1 |
| Quercetin | 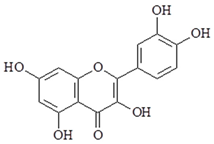 | ↓NF-κB, ↓COX-2, ↓NO, ↓PGE2, ↓iNOS, ↓TNF-α, ↓IL-1β, ↓IL-6, ↓VCAM-1, ↓ICAM-1, ↓MCP-1, ↓TLR2, ↓TLR4, ↓PI3K |
| Resveratrol | 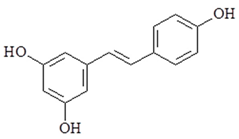 | ↓COX-2, ↓iNOS, ↓NF-κB, ↓TBK1, ↓RIP1, ↓TRAF6, ↓MAPK, ↓AKT, ↓TLR4 |
| Caffeic acid phenethyl ester | 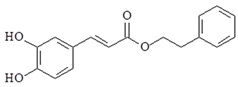 | ↓IL-12, ↓TLR4, ↓MyD88, ↓IRAK4, ↓TRIF, ↓NF-κB, ↓IL-6, ↓IL-8, ↓iNOS, ↓COX-2, ↓PI3K, ↓AKT |
| Xanthohumol | 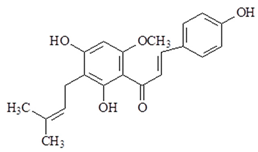 | ↓iNOS, ↓NO, ↓IFN-γ, ↓TLR4, ↓MD-2 |
| Genistein | 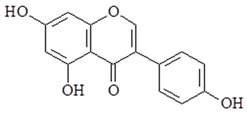 | ↓IL-6, ↓TNF-α, ↓NF-κB, ↓NO, ↓PGE2, ↓TNF-α, ↓TLR4, ↓IFN-β, ↓IL-10, ↓IL-1α, ↓IL-1β, ↓TNF-α, ↓CSF-2, ↓CSF-3, ↓CCL2, ↓CXCL10 |
| Berberine | 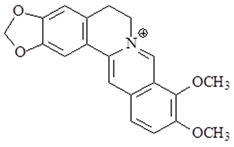 | ↓IL-1β, ↓TNF-α, ↓iNOS, ↓ICAM-1, ↓IL-6, ↓NF-κB, ↓TLR 2, ↓TLR 4, ↓TLR 9 |
| Sulforaphane | 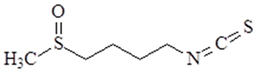 | ↓NF-κB, ↓TNF-α, ↓IL-6, ↓AKT, ↓HIF-1α, ↓TLR4 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, C.-Y.; Kao, C.-L.; Liu, C.-M. The Cancer Prevention, Anti-Inflammatory and Anti-Oxidation of Bioactive Phytochemicals Targeting the TLR4 Signaling Pathway. Int. J. Mol. Sci. 2018, 19, 2729. https://doi.org/10.3390/ijms19092729
Chen C-Y, Kao C-L, Liu C-M. The Cancer Prevention, Anti-Inflammatory and Anti-Oxidation of Bioactive Phytochemicals Targeting the TLR4 Signaling Pathway. International Journal of Molecular Sciences. 2018; 19(9):2729. https://doi.org/10.3390/ijms19092729
Chicago/Turabian StyleChen, Chung-Yi, Chiu-Li Kao, and Chi-Ming Liu. 2018. "The Cancer Prevention, Anti-Inflammatory and Anti-Oxidation of Bioactive Phytochemicals Targeting the TLR4 Signaling Pathway" International Journal of Molecular Sciences 19, no. 9: 2729. https://doi.org/10.3390/ijms19092729
APA StyleChen, C.-Y., Kao, C.-L., & Liu, C.-M. (2018). The Cancer Prevention, Anti-Inflammatory and Anti-Oxidation of Bioactive Phytochemicals Targeting the TLR4 Signaling Pathway. International Journal of Molecular Sciences, 19(9), 2729. https://doi.org/10.3390/ijms19092729






