Exosomal miRNA Signatures for Late-Onset Acute Graft-Versus-Host Disease in Allogenic Hematopoietic Stem Cell Transplantation
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
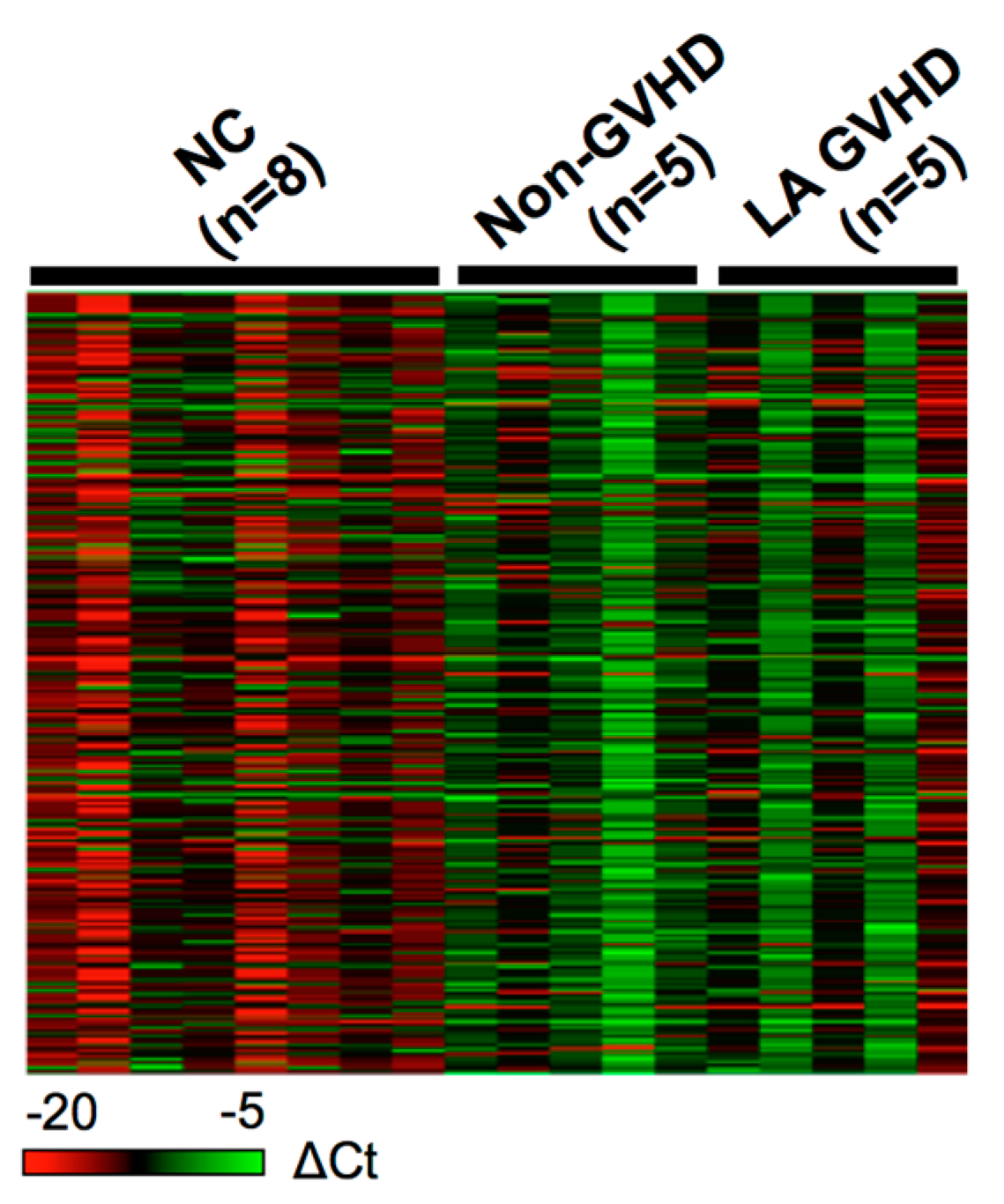
2.2. miRNA Expression Profiling at LA GVHD Diagnosis
2.3. Comparison of Exosomal miRNAs Differentially Expressed among LA GVHD, Non-GVHD, and Healthy Volunteers
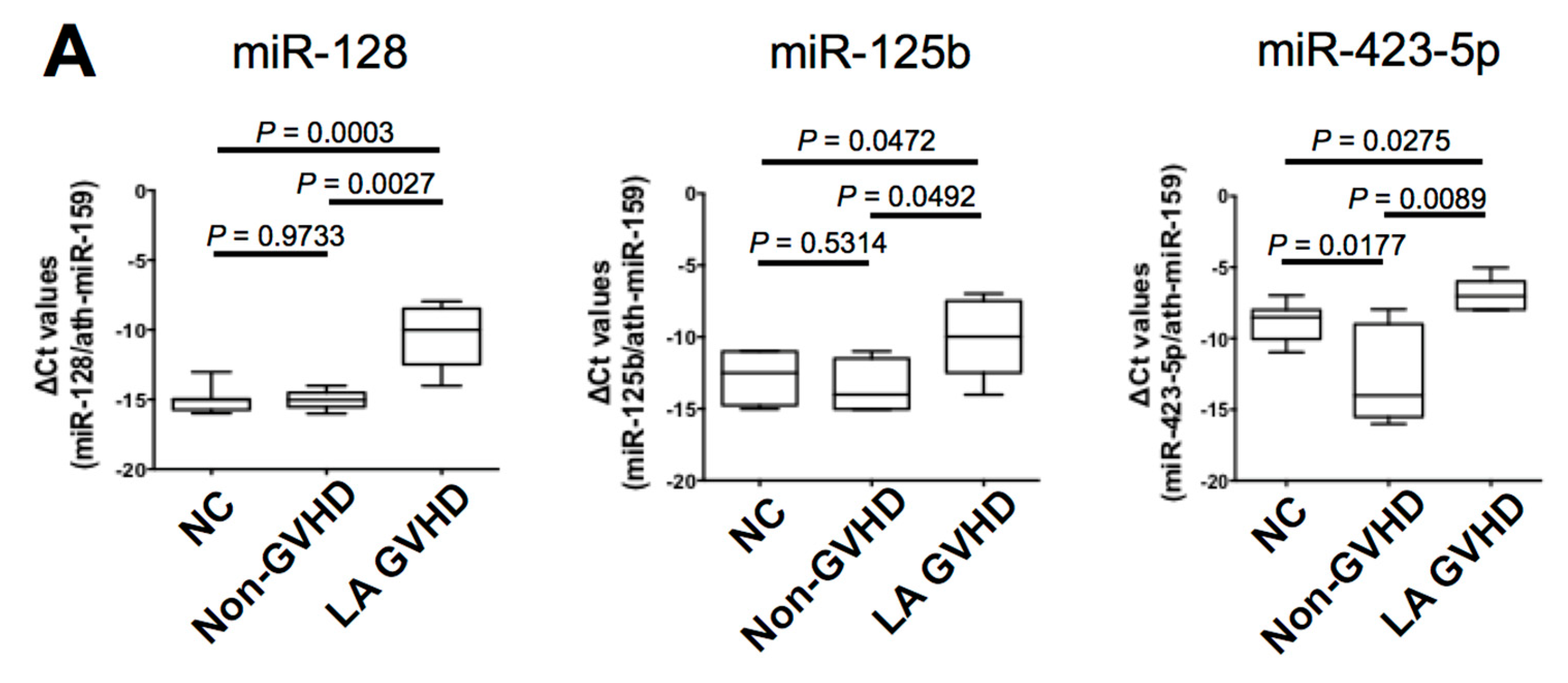
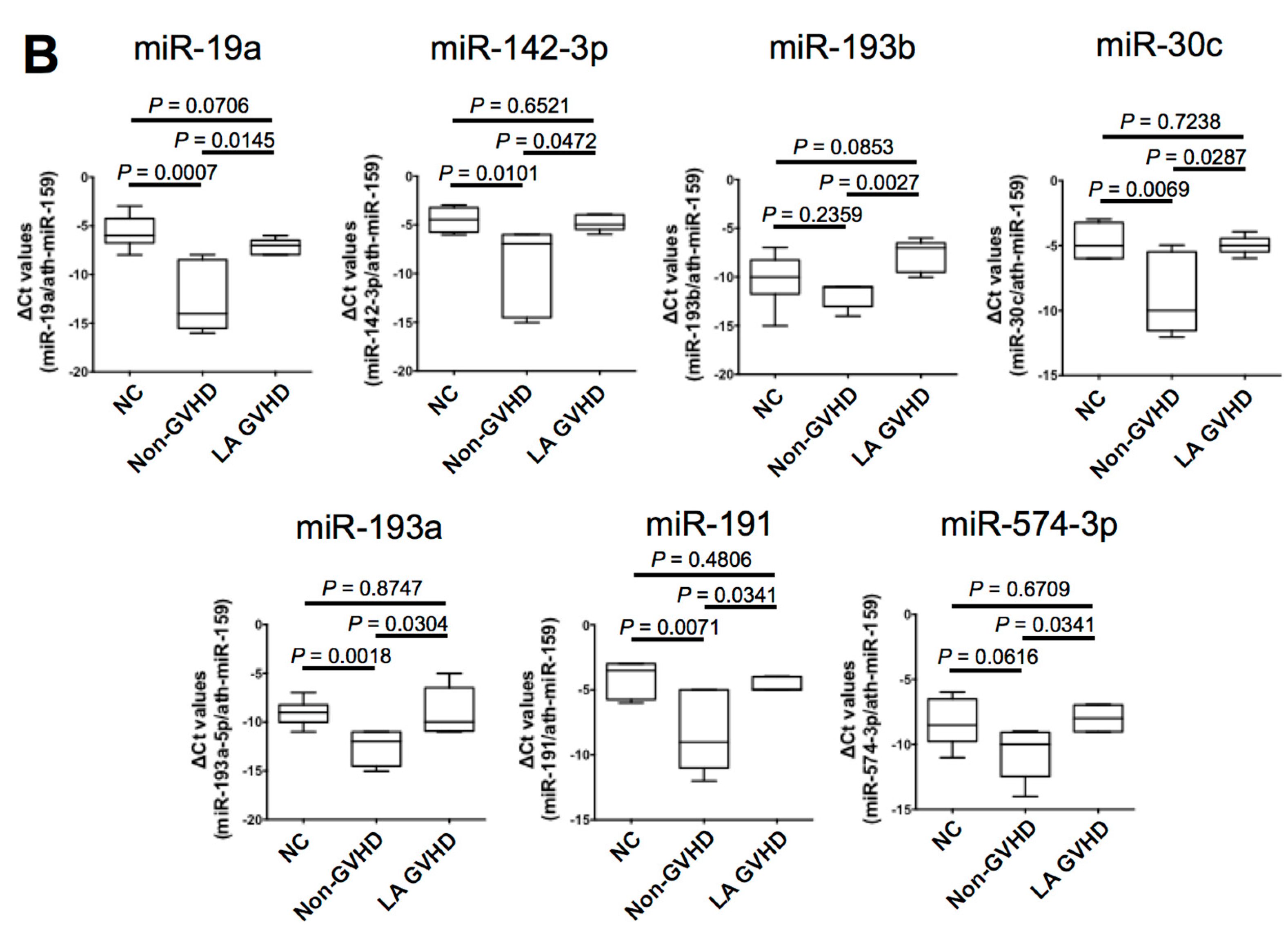
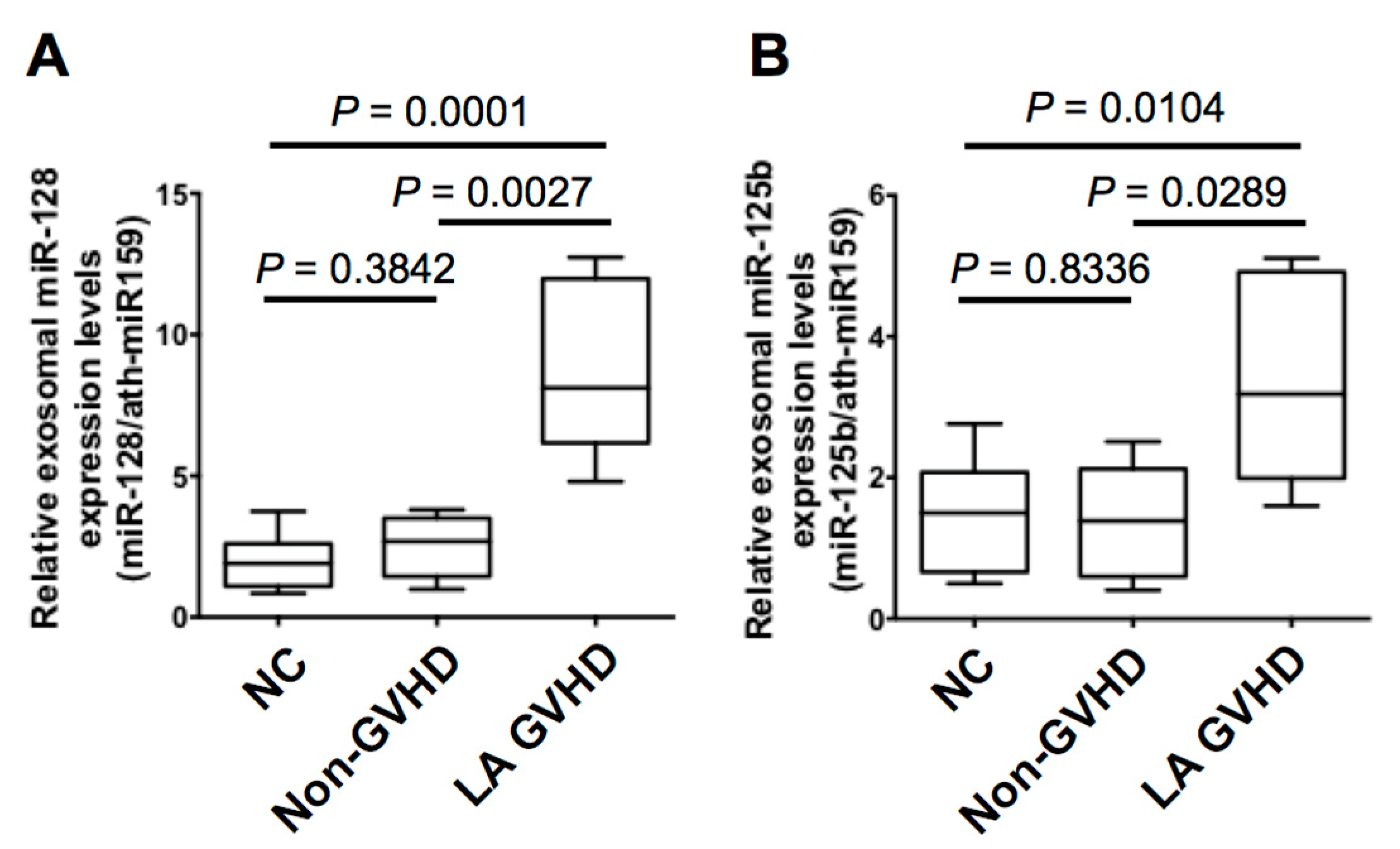
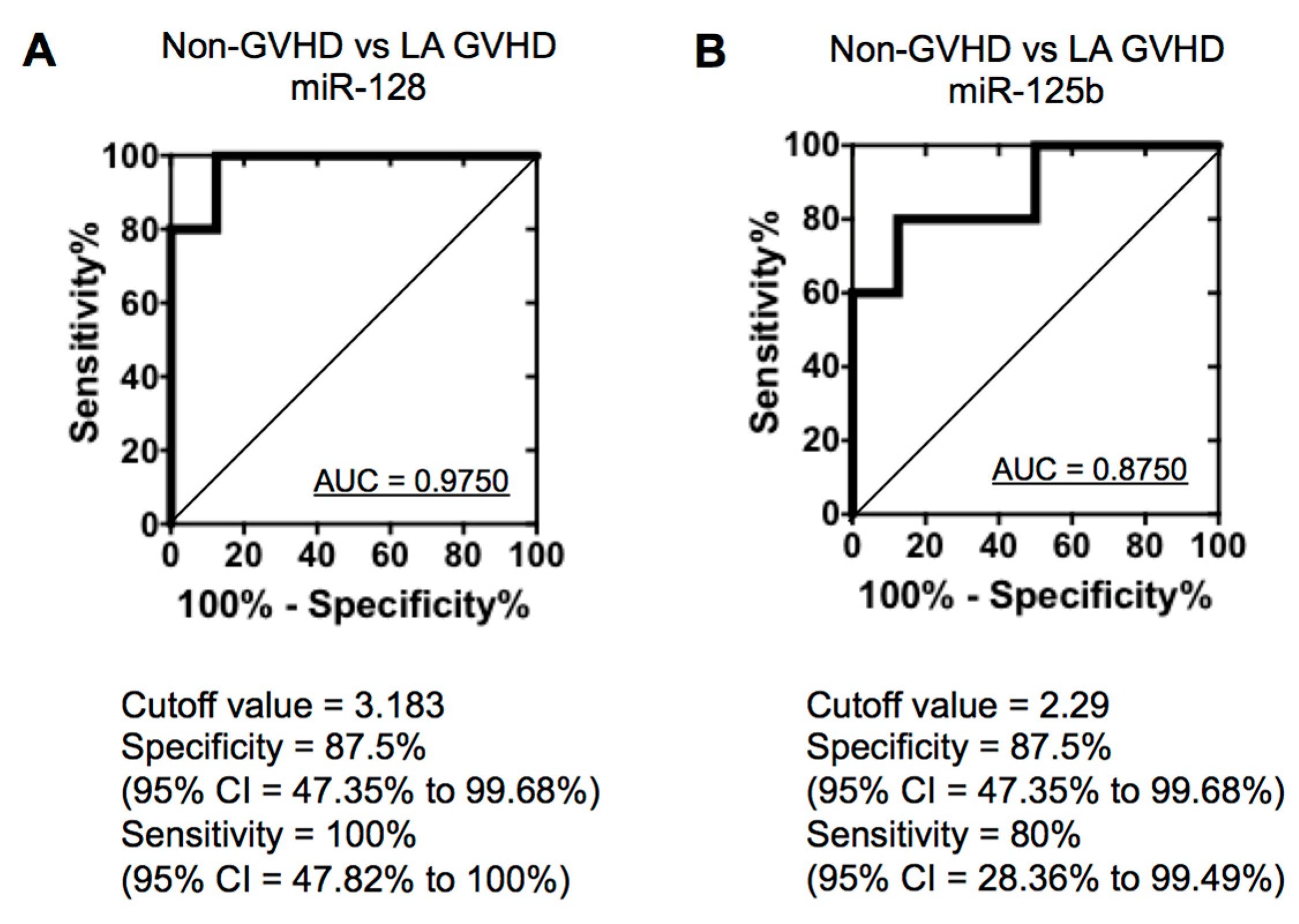
2.4. Quantification of Individual miRNA by Real-Time Quantitive RT-PCR
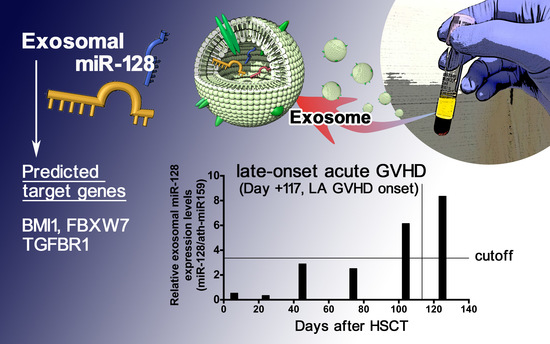
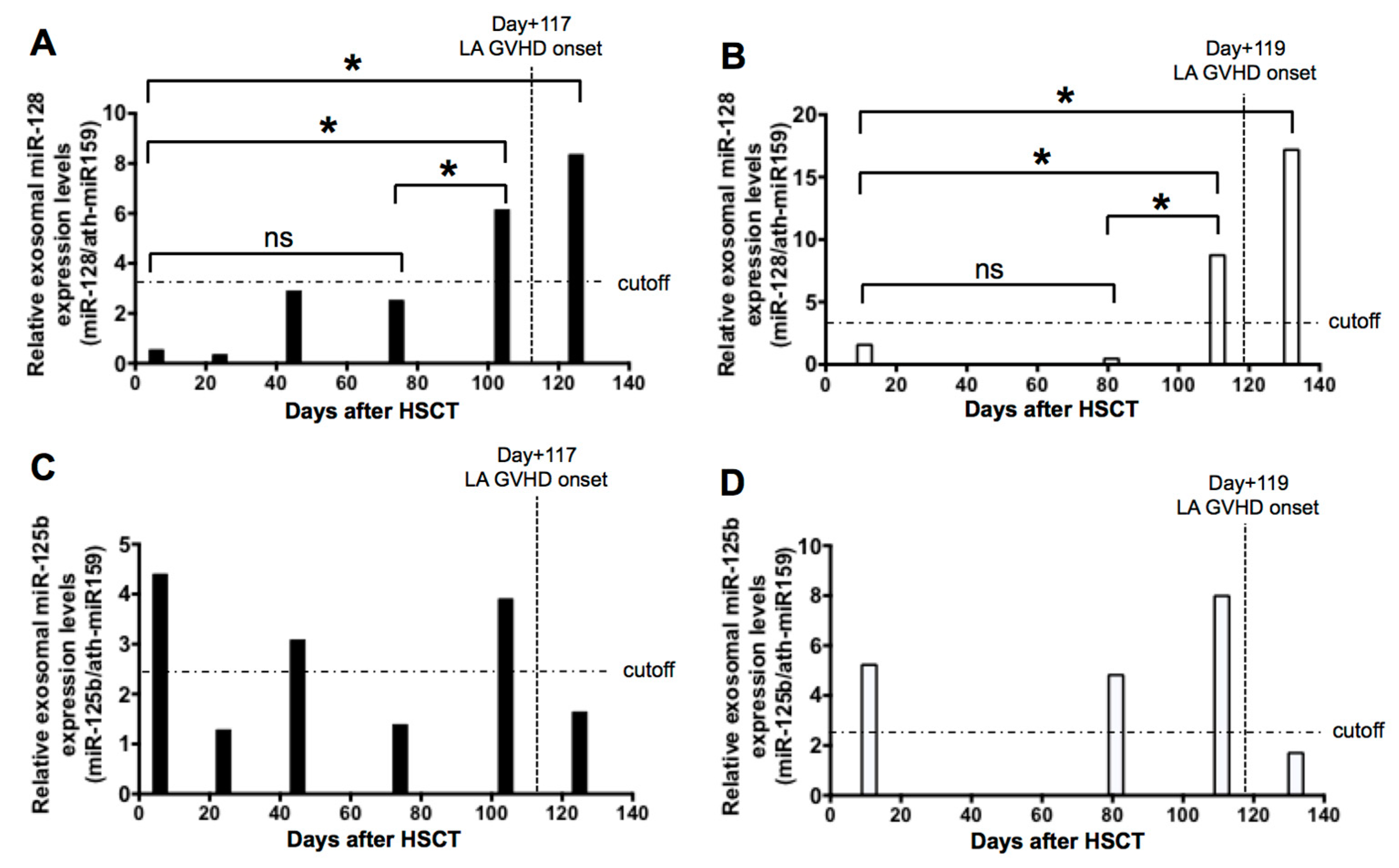
2.5. Sequential Analysis of Exosomal miRNAs Post-Allogenic HSCT
2.6. Prediction of the Target Genes of Candidate miRNAs
3. Discussion
4. Materials and Methods
4.1. Ethics
4.2. Diagnosis of Late-Onset Acute GVHD
4.3. Isolation of Exosome Fractions from Plasma Sample of Patients
4.4. Exosomal miRNA Profiling
4.5. TaqMan Loq-Density Array Screening
4.6. Real-Time Quantitative RT-PCR for Candidate miRNAs
4.7. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Filipovich, A.; Weisdorf, D.; Pavletic, S.; Socie, G.; Wingard, J.R.; Lee, S.J.; Martin, P.; Chien, J.; Przepiorka, D.; Couriel, D.; et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol. Blood Marrow Transplant. 2005, 11, 945–956. [Google Scholar] [CrossRef] [PubMed]
- Omer, A.K.; Weisdorf, D.J.; Lazaryan, A.; Shanley, R.; Blazar, B.R.; MacMillan, M.L.; Brunstein, C.; Bejanyan, N.; Arora, M. Late Acute Graft-versus-Host Disease after Allogeneic Hematopoietic Stem Cell Transplantation. Biol. Blood Marrow Transplant. 2016, 22, 879–883. [Google Scholar] [CrossRef] [PubMed]
- Cho, B.S.; Lee, S.E.; Song, H.H.; Lee, J.H.; Yahng, S.A.; Eom, K.S.; Kim, Y.-J.; Kim, H.-J.; Lee, S.; Min, C.-K.; et al. Graft-versus-tumor according to type of graft-versus-host disease defined by National Institute of Health consensus criteria and associated outcome. Biol. Blood Marrow Transplant. 2012, 18, 1136–1143. [Google Scholar] [CrossRef] [PubMed]
- Paczesny, S.; Krijanovski, O.I.; Braun, T.M.; Choi, S.W.; Clouthier, S.G.; Kuick, R.; Misek, D.E.; Cooke, K.R.; Kicko, C.L.; Weyand, A.; et al. A biomarker panel for acute graft-versus-host disease. Blood 2009, 113, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Nelson, P.R., Jr.; Khawaja, M.R.; Perkins, S.M.; Mumaw, C.L.; Orschell, C.; Paczesny, S. Prognostic biomarkers for acute graft-versus-host disease risk after cyclophosphamide-fludarabine nonmyeloablative allotransplantation. Biol Blood Marrow Transplant 2014, 20, 1861–1864. [Google Scholar] [CrossRef] [PubMed]
- Vader Lugt, M.T.; Braun, T.M.; Hanash, S.; Ritz, J.; Ho, V.T.; Antin, J.H.; Zhang, Q.; Wong, C.-H.; Wang, H.; Chin, A.; et al. ST2 as a marker for risk of therapy-resistant graft-versus-host disease and death. N. Engl. J. Med. 2013, 369, 529–539. [Google Scholar] [CrossRef] [PubMed]
- McDonald, G.B.; Tabellini, L.; Storer, B.E.; Lawler, R.L.; Martin, P.J.; Hansen, J.A. Plasma biomarkers of acute GVHD and nonrelapse mortality: Predictive value of measurements before GVHD onset and treatment. Blood 2015, 126, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Mathivanan, S.; Ji, H.; Simpson, R.J. Exosome: Extracellular organelles important in intracellular communication. J. Proteom. 2010, 73, 1907–1920. [Google Scholar] [CrossRef] [PubMed]
- Yuan, A.; Farber, E.L.; Rapoport, A.L.; Tejada, D.; Deniskin, R.; Akhmedov, N.B.; Farber, D.B. Transfer of microRNAs by embryonic stem cell microvesicles. PLoS ONE 2009, 4, e4722. [Google Scholar] [CrossRef] [PubMed]
- Umezu, T.; Tadokoro, H.; Azuma, K.; Yoshizawa, S.; Ohyashiki, K.; Ohyashiki, J.H. Exosomal miR-135b shed from hypoxic multiple myeloma cells enhances angiogenesis by targeting factor-inhibiting HIF-1. Blood 2014, 124, 3748–3757. [Google Scholar] [CrossRef] [PubMed]
- Valadi, H.; Ekstrom, K.; Bossios, A.; Sjostrand, M.; Lee, J.J.; Lotvall, J.O. Exosome-mediated transfer of mRNAa and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 2007, 9, 654–659. [Google Scholar] [CrossRef] [PubMed]
- Qu, Z.; Wu, J.; Wu, J.; Ji, A.; Qiang, G.; Jiang, Y.; Jiang, C.; Ding, Y. Exosomal miR-665 as a novel minimally invasive biomarker for hepatocellular carcinoma diagnosis and prognosis. Oncotarget 2017, 8, 80666–80678. [Google Scholar] [CrossRef] [PubMed]
- Sueta, A.; Yamamoto, Y.; Tomiguchi, M.; Takeshita, T.; Yamamoto-Ibusuki, M.; Iwase, H. Differential expression of exosomal miRNAs between breast cancer patients with and without recurrence. Oncotarget 2017, 8, 69934–69944. [Google Scholar] [CrossRef] [PubMed]
- Ranganathan, P.; Heaphy, C.E.; Costinean, S.; Stauffer, N.; Na, C.; Hamadani, M.; Santhanam, R.; Mao, C.; Taylor, P.A.; Sandhu, S.; et al. Regulation of acute graft-versus-host disease by microRNA-155. Blood 2012, 119, 4786–4797. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Robinson, S.N.; Setoyama, T.; Tung, S.S.; D’Abundo, L.; Shah, M.Y.; Yang, H.; Yvon, E.; Shah, N.; Yang, H.; et al. FOXP3 is a direct target of miR15a/16 in umbilical cord blood regulatory T cells. Bone Marrow Transplant 2014, 49, 793–799. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Heinrichs, J.; Bastian, D.; Fu, J.; Nguyen, H.; Schutt, S.; Liu, Y.; Jin, J.; Liu, C.; Li, Q.-J.; et al. MicroRNA-17-92 controls T-cell responses in graft-versus-host disease and leukemia relapse in mice. Blood 2015, 126, 1314–1323. [Google Scholar] [CrossRef] [PubMed]
- Xiao, B.; Wang, Y.; Li, W.; Baker, M.; Guo, J.; Corbet, K.; Tsalik, E.L.; Li, Q.-J.; Palmer, S.M.; Wood, C.W.; et al. Plasma microRNA signature as a noninvasive biomarker for acute graft-versus-host disease. Blood 2013, 122, 3365–3375. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhao, X.; Ye, X.; Luo, H.; Zhao, T.; Diao, Y.; Zhang, H.; Lv, M.; Zhang, W.; Huang, X.; et al. Plasma microRNA-586 is a new biomarker for acute graft-versus-host disease. Ann. Hematol. 2015, 94, 1505–1514. [Google Scholar] [CrossRef] [PubMed]
- Crossland, R.E.; Norden, J.; Juric, M.K.; Green, K.; Pearce, K.F.; Lendrem, C.; Greinix, H.T.; Dickinson, A.M. Expression of Serum microRNAs is Altered During Acute Graft-versus-Host Disease. Front. Immunol. 2017, 8, 308. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Oravecz-Wilson, K.; Mathewson, N.; Wang, Y.; McEachin, R.; Liu, C.; Toubai, T.; Wu, J.; Rossi, C.; Braun, T.; et al. Mature T cell responses are controlled by microRNA-142. J. Clin. Investig. 2015, 125, 2825–2840. [Google Scholar] [CrossRef] [PubMed]
- Godlewski, J.; Nowicki, M.O.; Bronisz, A.; Williams, S.; Otsuki, A.; Nuovo, G.; Raychaudhury, A.; Newton, H.B.; Chiocca, E.A.; Lawler, S. Targeting of the Bmi-1 oncogene/stem cell renewal factor by microRNA-128 inhibits glioma proliferation and self-renewal. Cancer Res. 2008, 68, 9125–9130. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.G.; Zhao, Y.; Sethi, P.; Li, Y.Y.; Mahta, A.; Culicchia, F.; Lukiw, W.J. Micro-RNA-128 (miRNA-128) down-regulation in glioblastoma targets ARP5 (ANGPTL6), Bmi-1 and E2F-3a, key regulators of brain cell proliferation. J. Neurooncol. 2010, 98, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.; Zhang, T.; Liu, C.; Badeaux, M.A.; Liu, B.; Liu, R.F.; Jeter, C.; Chen, X.; Vlassov, A.V.; Tang, D.G. miRNA-128 suppresses prostate cancer by inhibiting BMI-1 to inhibit tumor-initiating cells. Cancer Res. 2014, 74, 4183–4195. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; He, X.; Zou, J.; Guo, P.; Jiang, S.; Lv, N.; Alexseyev, Y.; Luo, L.; Luo, Z. Negative regulation of Bmi-1 by AMPK and implication in cancer progression. Oncotarget 2016, 7, 6188–6200. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.X.; Wilfred, B.R.; Hu, Y.; Stromberg, A.J.; Nelson, P.T. Anti-Argonaute RIP-Chip shows that miRNA transfections alter global patterns of mRNA recruitment to microribonucleoprotein complexes. RNA 2010, 16, 394–404. [Google Scholar] [CrossRef] [PubMed]
- Hafner, M.; Landthaler, M.; Burger, L.; Khorshid, M.; Hausser, J.; Berninger, P.; Rothballer, A.; Ascano, M., Jr.; Jungkamp, A.-C.; Munschauer, M.; et al. Transcriptome-wide identification of RNA-binding protein and microRNA target sites by PAR-CLIP. Cell 2010, 141, 129–141. [Google Scholar] [CrossRef] [PubMed]
- Lia, G.; Brunello, L.; Bruno, S.; Carpanetto, A.; Omede, P.; Festuccia, M.; Tosti, L.; Maffini, E.; Giaccone, L.; Arpinati, M.; et al. Extracellular vesicles as potentiall biomarkers of acute graft-vs-host disease. Leukemia 2018, 32, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Ohyashiki, K.; Umezu, T.; Katagiri, S.; Kobayashi, C.; Azuma, K.; Tauchi, T.; Okabe, S.; Fukuoka, Y.; Oyashika, J.H. Down-regulation of plasma miR-125 in chronic myeloid leukemia patients with successful discontinuation of imatinib. Int. J. Mol. Sci. 2016, 17, 570. [Google Scholar] [CrossRef] [PubMed]
- Kordelas, L.; Rebmann, V.; Ludwig, A.K.; Radtke, S.; Ruesing, J.; Doeppner, T.R.; Epple, M.; Horn, P.A.; Beelen, D.W.; Giebel, B. MSC-derived exosomes: a novel tool to treat therapy-refractory graft-versus-host disease. Leukemia 2014, 28, 970–973. [Google Scholar] [CrossRef] [PubMed]
- Ohyashiki, J.H.; Ohtsuki, K.; Mizoguchi, I.; Yoshimoto, T.; Katagiri, S.; Umezu, T.; Ohyashiki, K. Downregulated microRNA-148b in circulating PBMCs in chronic myeloid leukemia patients with undetectable minimal residual disease: A possible biomarker to discontinue imatinib safely. Drug Des. Dev. Ther. 2014, 8, 1151–1159. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa, S.; Ohyashiki, J.H.; Ohyashiki, M.; Umezu, T.; Suzuki, K.; Inagaki, A.; Lida, S.; Ohyashiki, K. Downregulated plasma miR-92a levels have clinical impact on multiple myeloma and related disorders. Blood Cancer J. 2012, 2, e53. [Google Scholar] [CrossRef] [PubMed]
- Ohyashiki, K.; Umezu, T.; Yoshizawa, S.; Ito, Y.; Ohyashiki, M.; Kawashima, H.; Tanaka, M.; Kuroda, M.; Ohyashiki, J.H. Clinical impact of down-regulated plasma miR-92a levels in non-Hodgkin’s lymphoma. PLoS ONE 2011, 6, e16408. [Google Scholar] [CrossRef] [PubMed]
- Smirnova, L.; Gräfe, A.; Seiler, A.; Schumacher, S.; Nitsch, R.; Wulczyn, F.G. Regulation of miRNA expression during neural cell specification. Eur. J. Neurosci. 2005, 21, 1469–1477. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.P.; Poisson, L.M.; Bhat, V.B.; Fermin, D.; Zhao, R.; Kalyana-Sundaram, S.; Michailidis, G.; Nesvizhskii, A.I.; Omenn, G.S.; Chinnaiyan, A.M.; et al. Quantitative proteomic profiling of prostate cancer reveals a role for miR-128 in prostate cancer. Mol. Cell Proteom. 2010, 9, 298–312. [Google Scholar] [CrossRef] [PubMed]
- Woo, H.H.; László, C.F.; Greco, S.; Chambers, S.K. Regulation of colony stimulating factor-1 expression and ovarian cancer cell behavior in vitro by miR-128 and miR-152. Mol. Cancer 2012, 11, 58. [Google Scholar] [CrossRef] [PubMed]
- Roth, P.; Wischhsen, J.; Happold, C.; Chandran, P.A.; Hofer, S.; Eisele, G.; Weller, M.; Keller, A. A specific miRNA signature in the peripheral blood of glioblastoma patients. J. Neurochem. 2011, 118, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Razo, A.; Dontje, B.; Vellenga, E.; de Haan, G.; Schuringa, J.J. Long-term maintenance of human hematopoietic stem/progenitor cells by expression of BMI1. Blood 2008, 111, 2621–2630. [Google Scholar] [CrossRef] [PubMed]

| Diagnosis | Age (y) | Gender | Donor | HLA Sero Compatibility | Stem Cell Source | Conditioning | GVHD Prophylaxis | Days of LA GVHD Onset | Days of Sampling | Target Organ | Grade | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Late-Onset Acute GVHD Group | ||||||||||||
| PN 1 | AML | 33 | M | Matched unrelated | 6/6 | BM | Myeloablative | CsA + st-MTX | +238 | +248 | Liver | IV |
| PN 2 | NHL | 38 | M | Matched related | 6/6 | PBSC | Reduced intensity | CsA + st-MTX | +180 | +221 | Liver | IV |
| PN 3 | AML | 57 | M | Mismatched | 5/6 | BM | Myeloablative | Tac + st-MTX | +117 | +136 | Gut | III |
| PN 4 | ALL | 56 | F | Mismatched | 4/6 | Umbilical cord blood | Myeloablative | Tac | +119 | +132 | Skin + gut | IV |
| PN 5 | ALL | 60 | F | Mismatched | 4/6 | Umbilical cord blood | Myeloablative | Tac | +165 | +174 | gut | II |
| Non-GVHD Group | ||||||||||||
| PN 6 | ALL | 62 | M | Matched unrelated | 6/6 | BM | Reduced intensity | CsA + st-MTX | +42 | |||
| PN 7 | ALL | 26 | F | Matched unrelated | 6/6 | BM | Myeloablative | CsA + st-MTX | +41 | |||
| PN 8 | AML | 60 | M | Mismatched | 4/6 | Umbilical cord blood | Myeloablative | Tac | +49 | |||
| PN 9 | AML | 62 | M | Mismatched | 5/6 | PBSC | Reduced intensity | Tac + st-MTX | +41 | |||
| PN 10 | AML | 70 | F | Mismatched | 4/6 | Umbilical cord blood | Reduced intensity | Tac | +45 | |||
| Exosomal miRNAs | Fold Change | p Value |
|---|---|---|
| hsa-miR-423-5p | 49.07 | 0.000198 |
| hsa-miR-19a | 37.33 | 0.000013 |
| hsa-miR-142-3p | 28.35 | 0.000198 |
| hsa-miR-128 | 24.79 | 0.000111 |
| hsa-miR-193b | 16.12 | 0.000209 |
| hsa-miR-30c | 14.33 | 0.003157 |
| hsa-miR-193a | 12.56 | 0.000111 |
| hsa-miR-191 | 12.3 | 0.000081 |
| hsa-miR-125b | 10.7 | 0.001064 |
| hsa-miR-574-3p | 6.13 | 0.000198 |
| miRNA | Experimentally Validated Targets by Reporter Assay (miRTarBase) |
|---|---|
| hsa-miR-128 | BMI1, FBXW7, DCX, RELN, WEE1, TGFBR1, NEK2, SREBF1, SREBF2, ABCA1, ABCG1, RXRA |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoshizawa, S.; Umezu, T.; Saitoh, Y.; Gotoh, M.; Akahane, D.; Kobayashi, C.; Ohyashiki, J.H.; Ohyashiki, K. Exosomal miRNA Signatures for Late-Onset Acute Graft-Versus-Host Disease in Allogenic Hematopoietic Stem Cell Transplantation. Int. J. Mol. Sci. 2018, 19, 2493. https://doi.org/10.3390/ijms19092493
Yoshizawa S, Umezu T, Saitoh Y, Gotoh M, Akahane D, Kobayashi C, Ohyashiki JH, Ohyashiki K. Exosomal miRNA Signatures for Late-Onset Acute Graft-Versus-Host Disease in Allogenic Hematopoietic Stem Cell Transplantation. International Journal of Molecular Sciences. 2018; 19(9):2493. https://doi.org/10.3390/ijms19092493
Chicago/Turabian StyleYoshizawa, Seiichiro, Tomohiro Umezu, Yuu Saitoh, Moritaka Gotoh, Daigo Akahane, Chiaki Kobayashi, Junko H. Ohyashiki, and Kazuma Ohyashiki. 2018. "Exosomal miRNA Signatures for Late-Onset Acute Graft-Versus-Host Disease in Allogenic Hematopoietic Stem Cell Transplantation" International Journal of Molecular Sciences 19, no. 9: 2493. https://doi.org/10.3390/ijms19092493
APA StyleYoshizawa, S., Umezu, T., Saitoh, Y., Gotoh, M., Akahane, D., Kobayashi, C., Ohyashiki, J. H., & Ohyashiki, K. (2018). Exosomal miRNA Signatures for Late-Onset Acute Graft-Versus-Host Disease in Allogenic Hematopoietic Stem Cell Transplantation. International Journal of Molecular Sciences, 19(9), 2493. https://doi.org/10.3390/ijms19092493