Association between Obesity and Circulating Brain-Derived Neurotrophic Factor (BDNF) Levels: Systematic Review of Literature and Meta-Analysis
Abstract
1. Introduction
2. Results
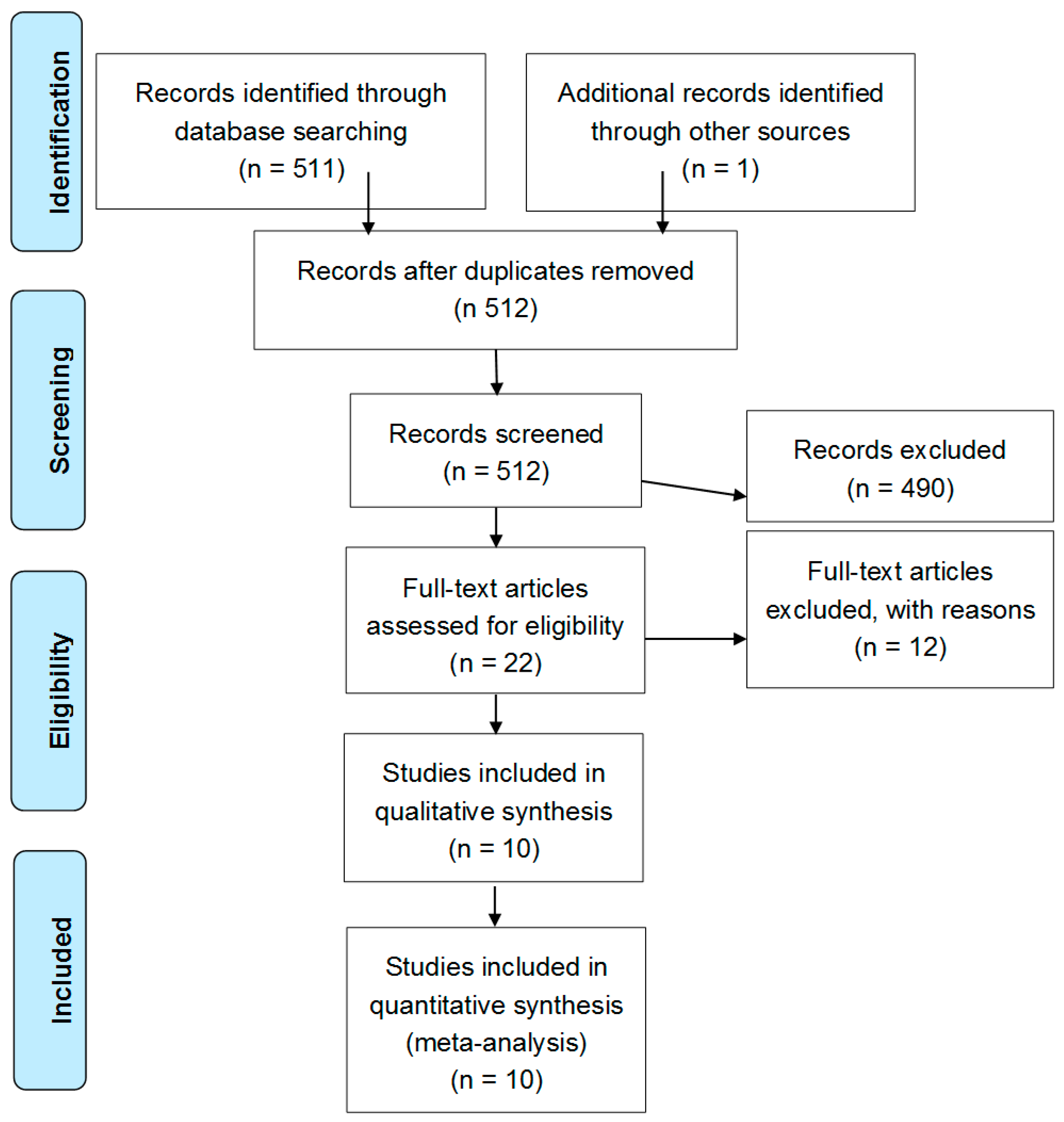
2.1. Literature Search Process
2.2. Study Characteristics
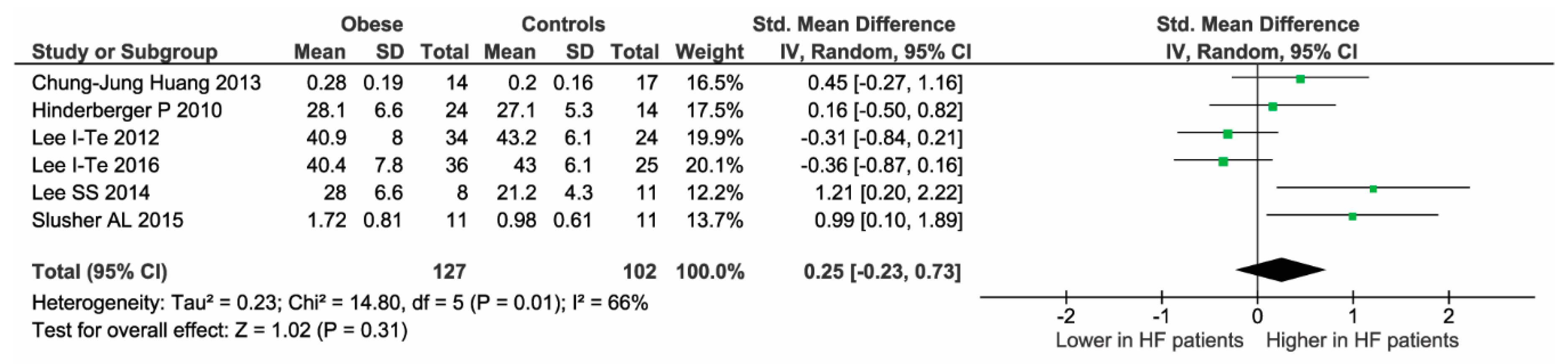
2.3. Meta-Regression Analyses
3. Discussion
4. Materials and Methods
4.1. Search Strategy
4.2. Data Extraction and Quality Assessment
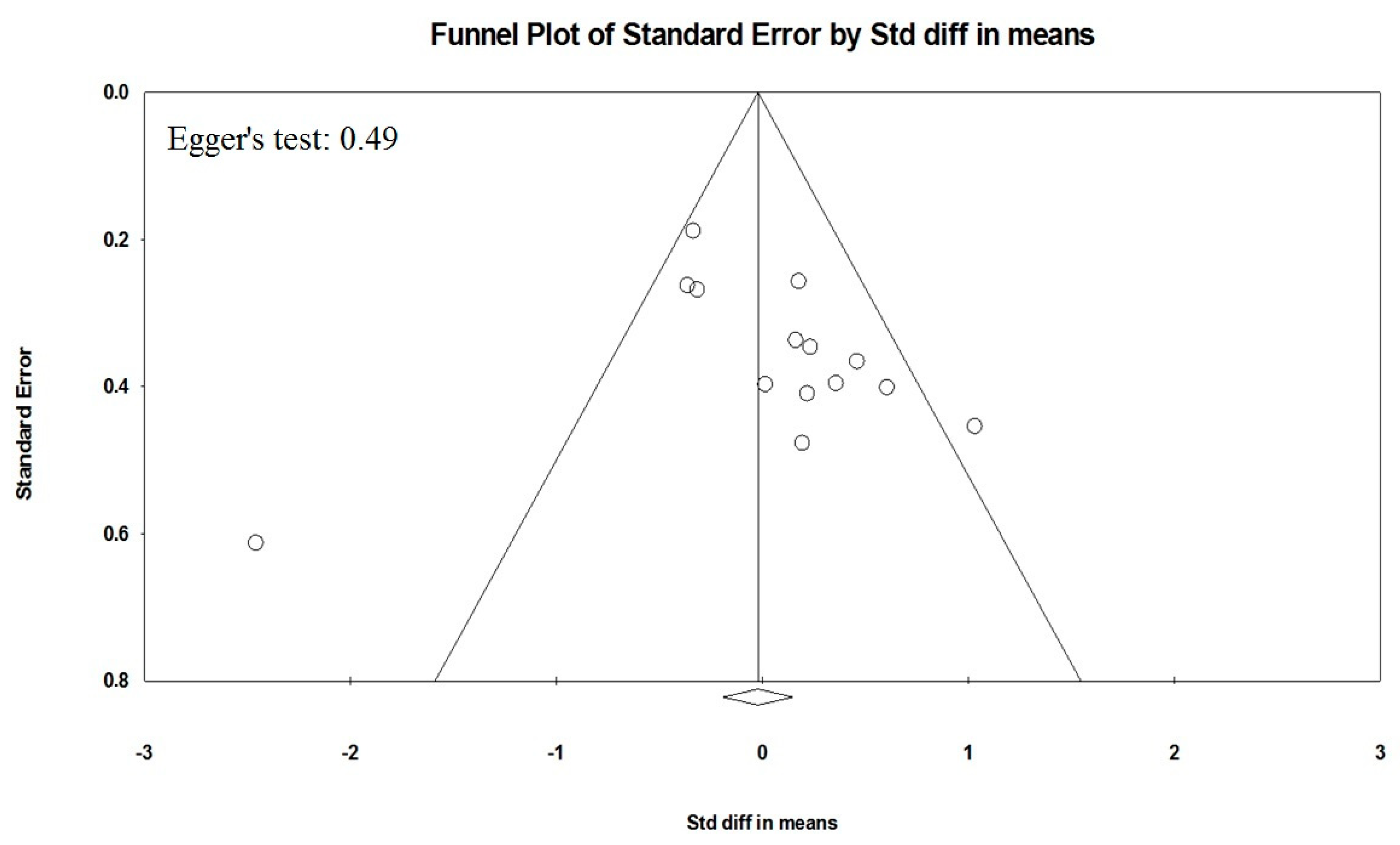
4.3. Statistical Analysis and Risk of Bias Assessment
4.4. Meta Regression Analyses
4.5. Sensitivity Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BDNF | brain-derived neurotrophic factor |
| BMI | body mass index |
| CI | confidence interval |
| CNS CV | central nervous system cardiovascular |
| CVD | cardiovascular disease |
| ELISA | enzyme-linked immunosorbent assay |
| MD | mean difference |
| NOS | Newcastle–Ottawa scale |
| SMD | standard mean difference |
References
- Payne, R.A. Cardiovascular risk. Br. J. Clin. Pharmacol. 2012, 74, 396–410. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, R.; Azevedo, I. Chronic inflammation in obesity and the metabolic syndrome. Mediat. Inflamm. 2010, 2010, 289645. [Google Scholar] [CrossRef] [PubMed]
- Donath, M.Y.; Shoelson, S.E. Type 2 diabetes as an inflammatory disease. Nat. Rev. Immunol. 2011, 11, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Fiore, M.; Chaldakov, G.N.; Rancic, G.; Bełtowski, J.; Tunçel, N.; Aloe, L. An integrated view: Neuroadipocrinology of diabesity. Serbian J. Exp. Clin. Res. 2014, 15, 61–69. [Google Scholar]
- Chaldakov, G. The metabotrophic ngf and BDNF: An emerging concept. Arch. Italiennes Biol. 2011, 149, 257–263. [Google Scholar]
- Kerschensteiner, M.; Gallmeier, E.; Behrens, L.; Leal, V.V.; Misgeld, T.; Klinkert, W.E.; Kolbeck, R.; Hoppe, E.; Oropeza-Wekerle, R.L.; Bartke, I.; et al. Activated human T cells, B cells, and monocytes produce brain-derived neurotrophic factor in vitro and in inflammatory brain lesions: A neuroprotective role of inflammation? J. Exp. Med. 1999, 189, 865–870. [Google Scholar] [CrossRef] [PubMed]
- Sornelli, F.; Fiore, M.; Chaldakov, G.N.; Aloe, L. Adipose tissue-derived nerve growth factor and brain-derived neurotrophic factor: Results from experimental stress and diabetes. Gen. Physiol. Biophys. 2009, 28, 179–183. [Google Scholar] [PubMed]
- Nakahashi, T.; Fujimura, H.; Altar, C.A.; Li, J.; Kambayashi, J.; Tandon, N.N.; Sun, B. Vascular endothelial cells synthesize and secrete brain-derived neurotrophic factor. FEBS Lett. 2000, 470, 113–117. [Google Scholar] [CrossRef]
- Golden, E.; Emiliano, A.; Maudsley, S.; Windham, B.G.; Carlson, O.D.; Egan, J.M.; Driscoll, I.; Ferrucci, L.; Martin, B.; Mattson, M.P. Circulating brain-derived neurotrophic factor and indices of metabolic and cardiovascular health: Data from the baltimore longitudinal study of aging. PLoS ONE 2010, 5, e10099. [Google Scholar] [CrossRef] [PubMed]
- Amadio, P.; Colombo, G.I.; Tarantino, E.; Gianellini, S.; Ieraci, A.; Brioschi, M.; Banfi, C.; Werba, J.P.; Parolari, A.; Lee, F.S.; et al. Bdnfval66met polymorphism: A potential bridge between depression and thrombosis. Eur. Heart J. 2017, 38, 1426–1435. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.B.; Takahashi, T.; Tokunaga, M.; Wilczynska, A.; Kim, C.J.; Meester, F.D.; Handjieva-Darlenska, T.; Cheema, S.K.; Wilson, D.W.; Milovanovic, B.; et al. Effect of brain derived neurotrophic factor, in relation to diet and lifestyle factors, for prevention of neuropsychiatric and vascular diseases and diabetes. Open Nutr. J. 2014, 7, 5–14. [Google Scholar] [CrossRef]
- Lebrun, B.; Bariohay, B.; Moyse, E.; Jean, A. Brain-derived neurotrophic factor (BDNF) and food intake regulation: A minireview. Auton Neurosci. 2006, 126–127, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Kernie, S.G.; Liebl, D.J.; Parada, L.F. Bdnf regulates eating behavior and locomotor activity in mice. EMBO J. 2000, 19, 1290–1300. [Google Scholar] [CrossRef] [PubMed]
- Lyons, W.E.; Mamounas, L.A.; Ricaurte, G.A.; Coppola, V.; Reid, S.W.; Bora, S.H.; Wihler, C.; Koliatsos, V.E.; Tessarollo, L. Brain-derived neurotrophic factor-deficient mice develop aggressiveness and hyperphagia in conjunction with brain serotonergic abnormalities. Proc. Natl. Acad. Sci. USA 1999, 96, 15239–15244. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Goulding, E.H.; Zang, K.; Cepoi, D.; Cone, R.D.; Jones, K.R.; Tecott, L.H.; Reichardt, L.F. Brain-derived neurotrophic factor regulates energy balance downstream of melanocortin-4 receptor. Nat. Neurosci. 2003, 6, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Yeo, G.S.; Connie Hung, C.C.; Rochford, J.; Keogh, J.; Gray, J.; Sivaramakrishnan, S.; O’Rahilly, S.; Farooqi, I.S. A de novo mutation affecting human trkb associated with severe obesity and developmental delay. Nat. Neurosci. 2004, 7, 1187–1189. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Lin, E.J.; Cahill, M.C.; Wang, C.; Liu, X.; During, M.J. Molecular therapy of obesity and diabetes by a physiological autoregulatory approach. Nat. Med. 2009, 15, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Bariohay, B.; Lebrun, B.; Moyse, E.; Jean, A. Brain-derived neurotrophic factor plays a role as an anorexigenic factor in the dorsal vagal complex. Endocrinology 2005, 146, 5612–5620. [Google Scholar] [CrossRef] [PubMed]
- Motamedi, S.; Karimi, I.; Jafari, F. The interrelationship of metabolic syndrome and neurodegenerative diseases with focus on brain-derived neurotrophic factor (BDNF): Kill two birds with one stone. Metab. Brain Dis. 2017, 32, 651–665. [Google Scholar] [CrossRef] [PubMed]
- Das, U.N. Obesity: Genes, brain, gut and environment. Nutrition 2010, 26, 459–473. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.B.; Williamson, R.; Santini, M.A.; Clemmensen, C.; Ettrup, A.; Rios, M.; Knudsen, G.M.; Aznar, S. Blood BDNF concentrations reflect brain-tissue BDNF levels across species. Int. J. Neuropsychopharmacol. 2011, 14, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Polyakova, M.; Stuke, K.; Schuemberg, K.; Mueller, K.; Schoenknecht, P.; Schroeter, M.L. Bdnf as a biomarker for successful treatment of mood disorders: A systematic & quantitative meta-analysis. J. Affect. Disord. 2015, 174, 432–440. [Google Scholar] [PubMed]
- Fernandes, B.S.; Steiner, J.; Berk, M.; Molendijk, M.L.; Gonzalez-Pinto, A.; Turck, C.W.; Nardin, P.; Gonçalves, C.A. Peripheral brain-derived neurotrophic factor in schizophrenia and the role of antipsychotics: Meta-analysis and implications. Mol. Psychiatry 2015, 20, 1108–1119. [Google Scholar] [CrossRef] [PubMed]
- Sanada, K.; Zorrilla, I.; Iwata, Y.; Bermúdez-Ampudia, C.; Graff-Guerrero, A.; Martínez-Cengotitabengoa, M.; González-Pinto, A. The efficacy of non-pharmacological interventions on brain-derived neurotrophic factor in schizophrenia: A systematic review and meta-analysis. Int. J. Mol. Sci. 2016, 17, E1766. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Jin, Y.; Wang, J.; Weng, X.; Li, C. Serum brain-derived neurotrophic factor (BDNF) levels in schizophrenia: A systematic review. Shanghai Arch. Psychiatry 2012, 24, 250–261. [Google Scholar] [PubMed]
- Green, M.J.; Matheson, S.L.; Shepherd, A.; Weickert, C.S.; Carr, V.J. Brain-derived neurotrophic factor levels in schizophrenia: A systematic review with meta-analysis. Mol. Psychiatry 2011, 16, 960–972. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, B.S.; Molendijk, M.L.; Köhler, C.A.; Soares, J.C.; Leite, C.M.; Machado-Vieira, R.; Ribeiro, T.L.; Silva, J.C.; Sales, P.M.; Quevedo, J.; et al. Peripheral brain-derived neurotrophic factor (BDNF) as a biomarker in bipolar disorder: A meta-analysis of 52 studies. BMC Med. 2015, 13, 289. [Google Scholar] [CrossRef] [PubMed]
- Roth, C.L.; Elfers, C.; Gebhardt, U.; Müller, H.L.; Reinehr, T. Brain-derived neurotrophic factor and its relation to leptin in obese children before and after weight loss. Metabolism 2013, 62, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Celik Guzel, E.; Bakkal, E.; Guzel, S.; Eroglu, H.E.; Acar, A.; Kuçukyalcin, V.; Topcu, B. Can low brain-derived neurotrophic factor levels be a marker of the presence of depression in obese women? Neuropsychiatr. Dis. Treat. 2014, 10, 2079–2086. [Google Scholar] [CrossRef] [PubMed]
- Slusher, A.L.; Whitehurst, M.; Zoeller, R.F.; Mock, J.T.; Maharaj, A.; Huang, C.J. Brain-derived neurotrophic factor and substrate utilization following acute aerobic exercise in obese individuals. J. Neuroendocrinol. 2015, 27, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Hinderberger, P.; Rullmann, M.; Drabe, M.; Luthardt, J.; Becker, G.A.; Blüher, M.; Regenthal, R.; Sabri, O.; Hesse, S. The effect of serum BDNF levels on central serotonin transporter availability in obese versus non-obese adults: A [(11)c]dasb positron emission tomography study. Neuropharmacology 2016, 110, 530–536. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.T.; Wang, J.S.; Fu, C.P.; Lin, S.Y.; Sheu, W.H. Relationship between body weight and the increment in serum brain-derived neurotrophic factor after oral glucose challenge in men with obesity and metabolic syndrome: A prospective study. Medicine 2016, 95, e5260. [Google Scholar] [CrossRef] [PubMed]
- Gajewska, E.; Sobieska, M.; Łojko, D.; Wieczorowska-Tobis, K.; Suwalska, A. Obesity itself does not influence BDNF serum levels in adults. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 3246–3250. [Google Scholar] [PubMed]
- Han, J.C.; Muehlbauer, M.J.; Cui, H.N.; Newgard, C.B.; Haqq, A.M. Lower brain-derived neurotrophic factor in patients with prader-willi syndrome compared to obese and lean control subjects. J. Clin. Endocrinol. Metab. 2010, 95, 3532–3536. [Google Scholar] [CrossRef] [PubMed]
- Corripio, R.; Gónzalez-Clemente, J.M.; Jacobo, P.S.; Silvia, N.; Lluis, G.; Joan, V.; Assumpta, C. Plasma brain-derived neurotrophic factor in prepubertal obese children: Results from a 2-year lifestyle intervention programme. Clin. Endocrinol. 2012, 77, 715–720. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.T.; Lee, W.J.; Tsai, I.C.; Liang, K.W.; Lin, S.Y.; Wan, C.J.; Fu, C.P.; Sheu, W.H. Brain-derived neurotrophic factor not associated with metabolic syndrome but inversely correlated with vascular cell adhesion molecule-1 in men without diabetes. Clin. Chim. Acta Int. J. Clin. Chem. 2012, 413, 944–948. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.S.; Yoo, J.H.; Kang, S.; Woo, J.H.; Shin, K.O.; Kim, K.B.; Cho, S.Y.; Roh, H.T.; Kim, Y.I. The effects of 12 weeks regular aerobic exercise on brain-derived neurotrophic factor and inflammatory factors in juvenile obesity and type 2 diabetes mellitus. J. Phys. Ther. Sci. 2014, 26, 1199–1204. [Google Scholar] [CrossRef] [PubMed]
- Levinger, I.; Goodman, C.; Matthews, V.; Hare, D.L.; Jerums, G.; Garnham, A.; Selig, S. Bdnf, metabolic risk factors, and resistance training in middle-aged individuals. Med. Sci. Sports Exerc. 2008, 40, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.J.; Mari, D.C.; Whitehurst, M.; Slusher, A.; Wilson, A.; Shibata, Y. Brain-derived neurotrophic factor expression ex vivo in obesity. Physiol. Behav. 2014, 123, 76–79. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.J.; Cao, P.J.; Bian, W.H.; Li, M.E.; Zhou, R.; Zhang, L.Y.; Yang, M.Z. Bdnf levels in adipose tissue and hypothalamus were reduced in mice with msg-induced obesity. Nutr. Neurosci. 2015, 18, 376–382. [Google Scholar] [CrossRef] [PubMed]
- Fox, E.A.; Biddinger, J.E.; Jones, K.R.; McAdams, J.; Worman, A. Mechanism of hyperphagia contributing to obesity in brain-derived neurotrophic factor knockout mice. Neuroscience 2013, 229, 176–199. [Google Scholar] [CrossRef] [PubMed]
- Han, J.C.; Liu, Q.R.; Jones, M.; Levinn, R.L.; Menzie, C.M.; Jefferson-George, K.S.; Adler-Wailes, D.C.; Sanford, E.L.; Lacbawan, F.L.; Uhl, G.R.; et al. Brain-derived neurotrophic factor and obesity in the wagr syndrome. N. Engl. J. Med. 2008, 359, 918–927. [Google Scholar] [CrossRef] [PubMed]
- Gray, J.; Yeo, G.S.; Cox, J.J.; Morton, J.; Adlam, A.L.; Keogh, J.M.; Yanovski, J.A.; El Gharbawy, A.; Han, J.C.; Tung, Y.C.; et al. Hyperphagia, severe obesity, impaired cognitive function, and hyperactivity associated with functional loss of one copy of the brain-derived neurotrophic factor (BDNF) gene. Diabetes 2006, 55, 3366–3371. [Google Scholar] [CrossRef] [PubMed]
- Lommatzsch, M.; Zingler, D.; Schuhbaeck, K.; Schloetcke, K.; Zingler, C.; Schuff-Werner, P.; Virchow, J.C. The impact of age, weight and gender on BDNF levels in human platelets and plasma. Neurobiol. Aging 2005, 26, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Radka, S.F.; Holst, P.A.; Fritsche, M.; Altar, C.A. Presence of brain-derived neurotrophic factor in brain and human and rat but not mouse serum detected by a sensitive and specific immunoassay. Brain Res. 1996, 709, 122–301. [Google Scholar] [CrossRef]
- Amadio, P.; Sandrini, L.; Ieraci, A.; Tremoli, E.; Barbieri, S.S. Effect of clotting duration and temperature on BDNF measurement in human serum. Int. J. Mol. Sci. 2017, 18, E1987. [Google Scholar] [CrossRef] [PubMed]
- Fujimura, H.; Altar, C.A.; Chen, R.; Nakamura, T.; Nakahashi, T.; Kambayashi, J.; Sun, B.; Tandon, N.N. Brain-derived neurotrophic factor is stored in human platelets and released by agonist stimulation. Thromb. Haemost. 2002, 87, 728–734. [Google Scholar] [CrossRef] [PubMed]
- Amadio, P.; Baldassarre, D.; Sandrini, L.; Weksler, B.B.; Tremoli, E.; Barbieri, S.S. Effect of cigarette smoke on monocyte procoagulant activity: Focus on platelet-derived brain-derived neurotrophic factor (BDNF). Platelets 2017, 28, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Serra-Millàs, M. Are the changes in the peripheral brain-derived neurotrophic factor levels due to platelet activation? World J. Psychiatry 2016, 6, 84–101. [Google Scholar] [CrossRef] [PubMed]
- Kishino, A.; Katayama, N.; Ishige, Y.; Yamamoto, Y.; Ogo, H.; Tatsuno, T.; Mine, T.; Noguchi, H.; Nakayama, C. Analysis of effects and pharmacokinetics of subcutaneously administered BDNF. Neuroreport 2001, 12, 1067–1072. [Google Scholar] [CrossRef] [PubMed]
- Polyakova, M.; Schlögl, H.; Sacher, J.; Schmidt-Kassow, M.; Kaiser, J.; Stumvoll, M.; Kratzsch, J.; Schroeter, M.L. Stability of BDNF in human samples stored up to 6 months and correlations of serum and edta-plasma concentrations. Int. J. Mol. Sci. 2017, 18, E1189. [Google Scholar] [CrossRef] [PubMed]
- Trajkovska, V.; Marcussen, A.B.; Vinberg, M.; Hartvig, P.; Aznar, S.; Knudsen, G.M. Measurements of brain-derived neurotrophic factor: Methodological aspects and demographical data. Brain Res. Bull. 2007, 73, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Tsuchimine, S.; Sugawara, N.; Ishioka, M.; Yasui-Furukori, N. Preanalysis storage conditions influence the measurement of brain-derived neurotrophic factor levels in peripheral blood. Neuropsychobiology 2014, 69, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Eyileten, C.; Kaplon-Cieslicka, A.; Mirowska-Guzel, D.; Malek, L.; Postula, M. Antidiabetic effect of brain-derived neurotrophic factor and its association with inflammation in type 2 diabetes mellitus. J. Diabetes Res. 2017, 2017, 2823671. [Google Scholar] [CrossRef] [PubMed]
- Krabbe, K.S.; Nielsen, A.R.; Krogh-Madsen, R.; Plomgaard, P.; Rasmussen, P.; Erikstrup, C.; Fischer, C.P.; Lindegaard, B.; Petersen, A.M.; Taudorf, S.; et al. Brain-derived neurotrophic factor (BDNF) and type 2 diabetes. Diabetologia 2007, 50, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Noble, E.E.; Billington, C.J.; Kotz, C.M.; Wang, C. The lighter side of BDNF. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 300, R1053–R1069. [Google Scholar] [CrossRef] [PubMed]
- Iughetti, L.; Casarosa, E.; Predieri, B.; Patianna, V.; Luisi, S. Plasma brain-derived neurotrophic factor concentrations in children and adolescents. Neuropeptides 2011, 45, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, K. Ethnic differences in the serum levels of probdnf, a precursor of brain-derived neurotrophic factor (BDNF), in mood disorders. Eur. Arch. Psychiatry Clin. Neurosci. 2016, 266, 285–287. [Google Scholar] [CrossRef] [PubMed]
- Akbarian, S.A.; Salehi-Abargouei, A.; Pourmasoumi, M.; Kelishadi, R.; Nikpour, P.; Heidari-Beni, M. Association of brain-derived neurotrophic factor gene polymorphisms with body mass index: A systematic review and meta-analysis. Adv. Med. Sci. 2017, 63, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Yeebo, M.F. Ethnic differences in BDNF val66met polymorphism. Br. J. Psychiatry 2015, 207, 363. [Google Scholar] [CrossRef] [PubMed]
- Zuccato, C.; Marullo, M.; Vitali, B.; Tarditi, A.; Mariotti, C.; Valenza, M.; Lahiri, N.; Wild, E.J.; Sassone, J.; Ciammola, A.; et al. Brain-derived neurotrophic factor in patients with huntington’s disease. PLoS ONE 2011, 6, e22966. [Google Scholar] [CrossRef] [PubMed]
- Polacchini, A.; Metelli, G.; Francavilla, R.; Baj, G.; Florean, M.; Mascaretti, L.G.; Tongiorgi, E. A method for reproducible measurements of serum BDNF: Comparison of the performance of six commercial assays. Sci. Rep. 2015, 5, 17989. [Google Scholar] [CrossRef] [PubMed]
- Marosi, K.; Mattson, M.P. BDNF mediates adaptive brain and body responses to energetic challenges. Trends Endocrinol. Metab. 2014, 25, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Di Minno, A.; Turnu, L.; Porro, B.; Squellerio, I.; Cavalca, V.; Tremoli, E.; Di Minno, M.N. 8-hydroxy-2-deoxyguanosine levels and cardiovascular disease: A systematic review and meta-analysis of the literature. Antioxid. Redox Signal. 2016, 24, 548–555. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The prisma statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Faraone, S.V. Interpreting estimates of treatment effects: Implications for managed care. P T 2008, 33, 700–711. [Google Scholar] [PubMed]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.; Egger, M.; Smith, G.D. Systematic reviews in health care: Investigating and dealing with publication and other biases in meta-analysis. BMJ 2001, 323, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Duval, S.; Tweedie, R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 2000, 56, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle–Ottawa Scale (Nos) for Assessing the Quality of Non-Randomized Studies in Meta-Analysis; Ottawa Hospital Research Institute: Ottawa, ON, Canada, 2000. [Google Scholar]
| Author, Country | Study Design | Type of Pathology | BDNF Measurement | Cases (n) | Controls (n) | BDNF (Ng/ML) Cases Mean | BDNF (ng/mL) Controls Mean | BDNF (ng/mL) Weighted Average | Age (Years) Cases | Age (Years) Controls | Age (Years) Mean | Males (%) | BMI (kg/m2) Cases | BMI (kg/m2) Controls | BMI (kg/m2) Mean | WC (cm) Cases | WC (cm) Controls | WC (cm) Mean |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lee I-Te 2016, Taiwan [32] | Prospective | Obesity and MetS | Serum (ELISA) | 36 | 25 | 40.40 | 43.00 | 41.47 | 44 | 39 | 41.95 | 100.0 | 33.5 | 22.5 | 29.0 | 109.1 | 81.9 | 98 |
| Slusher AL 2015, United States [30] | Case-control/Prospective | Obesity | Plasma (ELISA) | 11 | 11 | 1.72 | 0.98 | 1.35 | 22.91 | 23.27 | 23.09 | 40.9 | 35.72 | 21.89 | 28.8 | 99.32 | 71.36 | 85.3 |
| Hinderberger P 2016, Germany [31] | Case-control | Obesity | Serum (ELISA) | 24 | 14 | 28.10 | 27.10 | 27.73 | 36.9 | 36.1 | 36.61 | 26.3 | 40.7 | 22.5 | 34.0 | ND | ND | ND |
| Gajewska E 2014, Poland [33] | Case-control | Obesity | Serum (ELISA) | 15 | 11 | 23.89 | 23.83 | 23.86 | ≤65 | ≤65 | ≤65 | 0 | >40 | ND | ND | ND | ND | ND |
| 39 | 25 | 24.11 | 23.22 | 23.76 | >66 | >66 | >66 | 0 | >40 | ND | ND | ND | ND | ND | ||||
| 12 | 7 | 23.72 | 22.95 | 23.44 | ≤65 | ≤65 | ≤65 | 100 | >40 | ND | ND | ND | ND | ND | ||||
| 14 | 21 | 24.74 | 23.94 | 23.99 | >66 | >66 | >66 | 100 | >40 | ND | ND | ND | ND | ND | ||||
| Han JC 2010, United States [34] | Cross-sectional | Obesity | Serum (ELISA) | 13 | 13 | 17.60 | 14.20 | 15.90 | 12.3 | 12.4 | 12.35 | 92 | 33.1 | 17.2 | 25.2 | ND | ND | ND |
| Corripio R 2012, Spain [35] | Prospective | Obesity | Plasma (ELISA) | 73 | 47 | 57.70 | 78.50 | 65.85 | 8.03 | 7.74 | 7.92 | 55.8 | 26.5 | 16.2 | 22.5 | 81.3 | 57.5 | 72.0 |
| Lee I-Te 2012, Taiwan [36] | Case-control | Obesity and MetS | Serum (ELISA) | 34 | 24 | 40.90 | 43.20 | 41.85 | 41 | 39 | 40.17 | 100 | 33.9 | 22.5 | 29.2 | 109.3 | 82.2 | 98.1 |
| Lee SS 2014, Korea [37] | Prospective | Obesity | Serum (ELISA) | 8 | 11 | 8.00 | 21.20 | 15.64 | 16.3 | 16.4 | 16.36 | 65 | 27.47 | 22.35 | 24.5 | ND | ND | ND |
| Levinger I 2007, Australia [38] | Prospective | Obesity and MetS | Plasma (ELISA) | 14 | 10 | 1.03 | 0.70 | 0.89 | 51.9 | 48.9 | 50.65 | 54 | 30.3 | 23.8 | 27.6 | 99.4 | 81 | 91.7 |
| Chun-Jung Huang 2014, United States [39] | Case-control | Obesity | Plasma (ELISA) | 14 | 17 | 0.28 | 0.20 | 0.24 | 22.64 | 22.94 | 22.80 | 35 | 38.18 | 21.21 | 28.9 | 111.46 | 68.74 | 88.0 |
| Number of Studies | Number of Patients | Effect Size | Test for Subgroup Differences | |
|---|---|---|---|---|
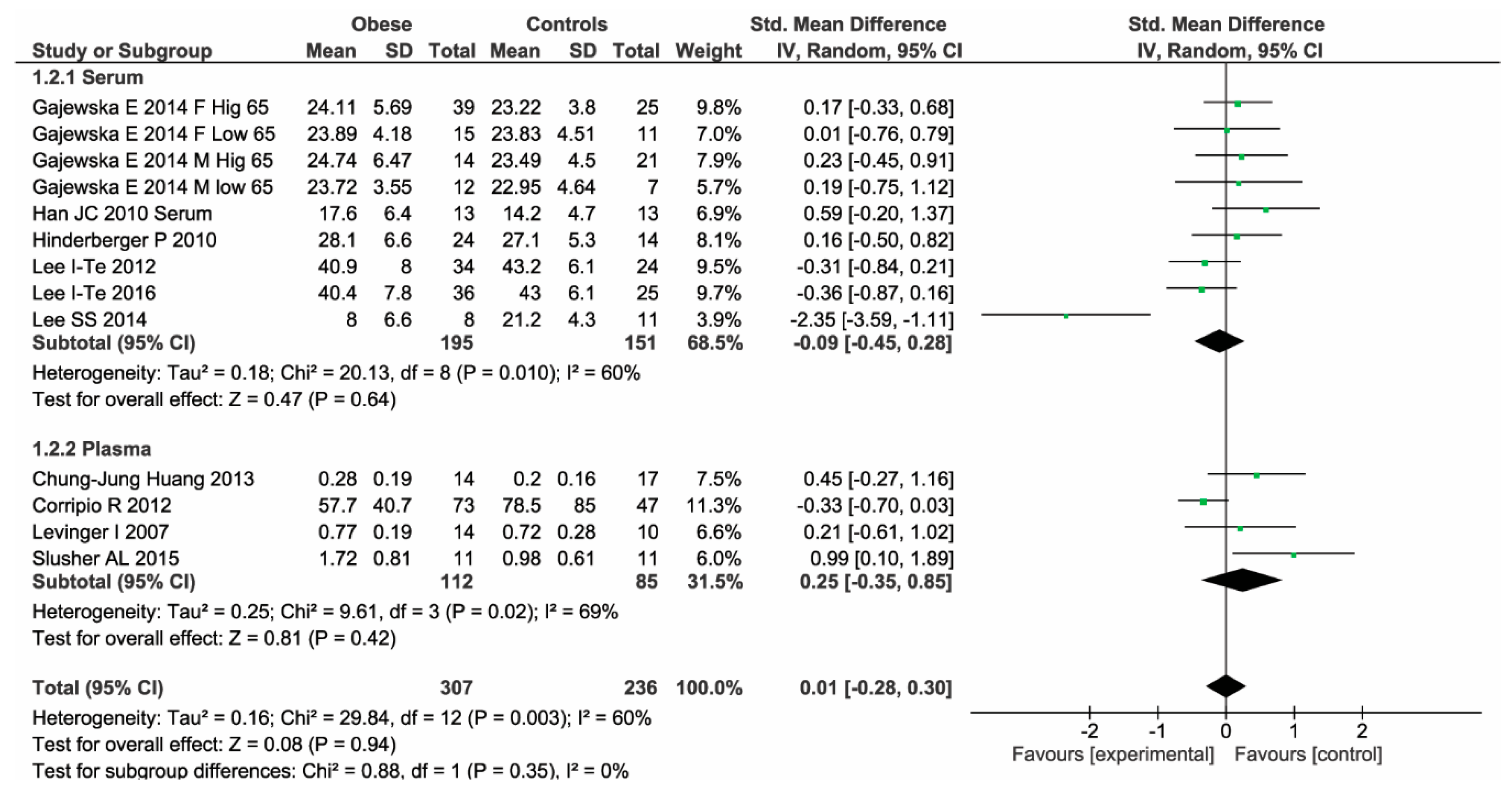
| Serum | 6 | 195 cases 151 controls | MD: −0.93; 95% CI: −3.34, 1.48, p = 0.45, I2: 74%, p = 0.0001 | Chi2: 0.88, p = 0.35 |
| Plasma | 4 | 112 cases 85 controls | MD: 0.15; 95% CI: −0.09, 0.39, p = 0.23, I2: 58%, p = 0.07 |
| Definition of Cases | Representativeness of Cases | Selection of Controls | Definition of Controls | Comparability of Cases and Controls | Same Method of Ascertainment | Quality | |
|---|---|---|---|---|---|---|---|
| Lee I-Te, 2016 [31] |  |  | - |  |  |  | 5 |
| Slusher AL, 2015 [29] |  | - | - | - |  |  | 3 |
| Hinderberger P, 2016 [32] |  | - | - |  |  |  | 4 |
| Gajewska E, 2014 [33] | - | - | - | - |  | - | 1 |
| Han JC, 2010 [34] | - | - | - | - |  | - | 1 |
| Corripio R, 2012 [35] |  | - | - | - |  | - | 2 |
| Lee I-TE, 2012 [36] |  |  | - |  |  | - | 4 |
| Lee SS, 2014 [37] |  | - | - |  |  | - | 3 |
| Levinger I, 2007 [38] | - | - | - | - | - |  | 1 |
| Chun-Jung Huang 2013 [39] |  |  | - |  | - | - | 3 |
| Author | Sample | Information Reported about Blood Collection | Patients Condition | Information Reported about Procedure to Obtain Plasma/Serum | Conservation | Assay Kit |
|---|---|---|---|---|---|---|
| Lee I-Te 2016 [31] | Serum | ND | ND | ND | ND | ELISA Quantikine; R&D Systems, Inc., Minneapolis, MN |
| Slusher AL 2015 [29] | Plasma | Blood collected in EDTA tubes (BD vacutainer) | ND | Immediately centriguged at 1000× g for 20 min at room temperature | −80 °C until measurements (no reported storage time) | Promega, Madison, WI |
| Hinderberger P 2016 [32] | Serum | Blood collected in two timeframes: 8–10 a.m. and 2–4 p.m. | ND | 2 h of clotting | ND | ELISA Quantikine; R&D Systems, Inc., Minneapolis, MN |
| Gajewska E 2014 [33] | Serum | ND | ND | ND | ND | ELISA Quantikine; R&D Systems, Inc., Minneapolis, MN |
| Han JC 2010 [34] | Serum | Serum obtained using serum separator tubes | ND | 30 min of clotting | −70 °C until measurements (no reported storage time) | ELISA Quantikine; R&D Systems, Inc., Minneapolis, MN |
| Corripio R 2012 [35] | Plasma | ND | Samples obtained after 12 h of fasting | ND | −80 °C until measurements (no reported storage time) | Human BDNF ELISA Kit, RayBiotech, Inc, Norcross, GA |
| Lee I-TE 2012 [36] | Serum | ND | ND | ND | ND | ELISA Quantikine; R&D Systems, Inc., Minneapolis, MN |
| Lee SS 2014 [37] | Serum | ND | Samples obtained after 12 h of fasting | Centrifugation at 3000 rpm for 10 min | −80 °C until measurements (no reported storage time) | ELISA Quantikine; R&D Systems, Inc., Minneapolis, MN |
| Levinger I 2007, Australia [38] | Plasma | ND | Samples obtained after 3 h of fasting | ND | −20 °C until measurements (no reported storage time) | ELISA Quantikine; R&D Systems, Inc., Minneapolis, MN |
| Chun-Jung Huang 2013 [39] | Plasma | Blood collected in EDTA tubes | ND | Centriguged at 2000× g for 15 min at 4 °C | −80 °C until measurements (no reported storage time) | Abcam, Cambridge, MA |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sandrini, L.; Di Minno, A.; Amadio, P.; Ieraci, A.; Tremoli, E.; Barbieri, S.S. Association between Obesity and Circulating Brain-Derived Neurotrophic Factor (BDNF) Levels: Systematic Review of Literature and Meta-Analysis. Int. J. Mol. Sci. 2018, 19, 2281. https://doi.org/10.3390/ijms19082281
Sandrini L, Di Minno A, Amadio P, Ieraci A, Tremoli E, Barbieri SS. Association between Obesity and Circulating Brain-Derived Neurotrophic Factor (BDNF) Levels: Systematic Review of Literature and Meta-Analysis. International Journal of Molecular Sciences. 2018; 19(8):2281. https://doi.org/10.3390/ijms19082281
Chicago/Turabian StyleSandrini, Leonardo, Alessandro Di Minno, Patrizia Amadio, Alessandro Ieraci, Elena Tremoli, and Silvia S. Barbieri. 2018. "Association between Obesity and Circulating Brain-Derived Neurotrophic Factor (BDNF) Levels: Systematic Review of Literature and Meta-Analysis" International Journal of Molecular Sciences 19, no. 8: 2281. https://doi.org/10.3390/ijms19082281
APA StyleSandrini, L., Di Minno, A., Amadio, P., Ieraci, A., Tremoli, E., & Barbieri, S. S. (2018). Association between Obesity and Circulating Brain-Derived Neurotrophic Factor (BDNF) Levels: Systematic Review of Literature and Meta-Analysis. International Journal of Molecular Sciences, 19(8), 2281. https://doi.org/10.3390/ijms19082281







