PEI-Coated Fe3O4 Nanoparticles Enable Efficient Delivery of Therapeutic siRNA Targeting REST into Glioblastoma Cells
Abstract
1. Introduction
2. Results
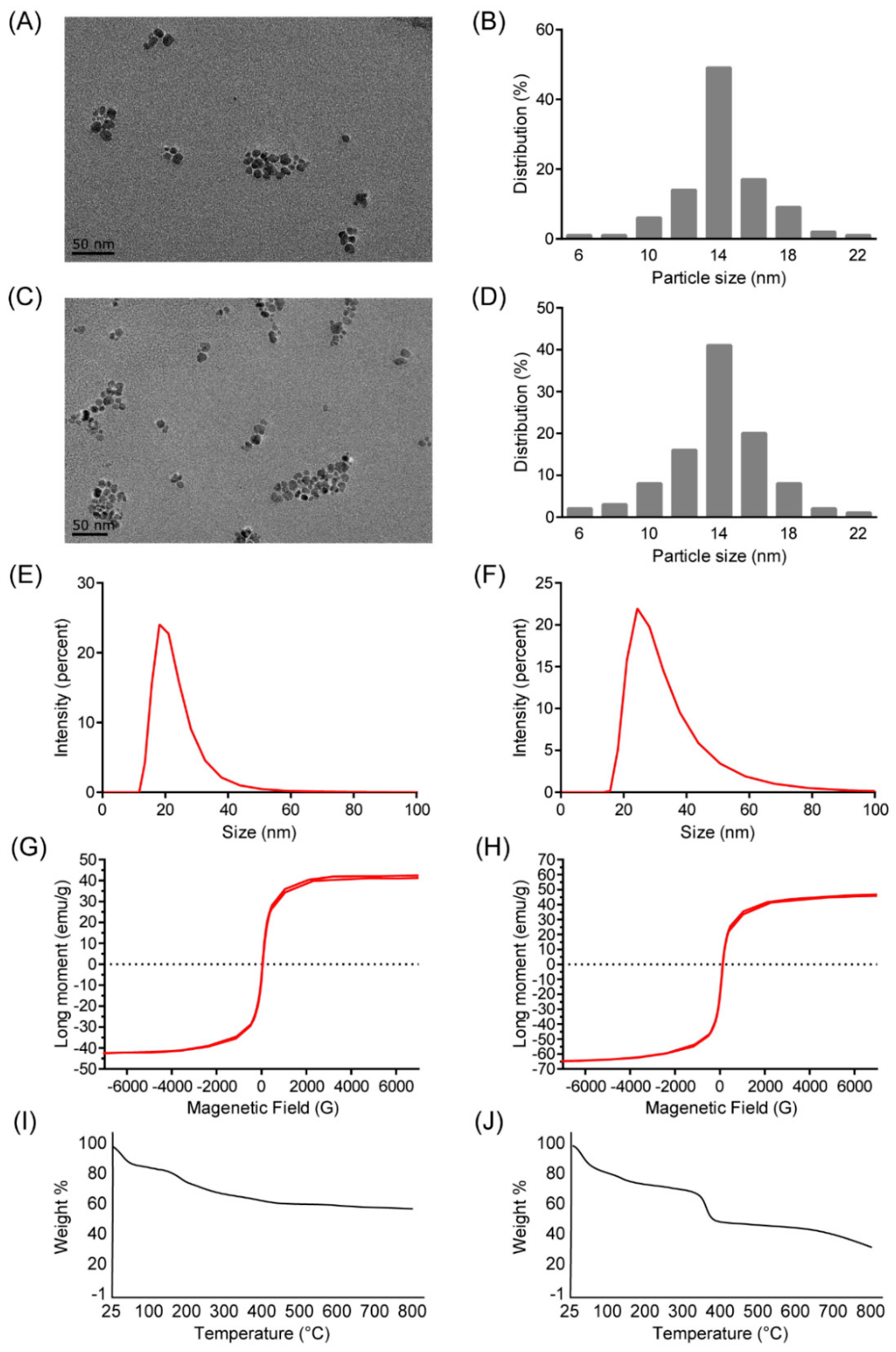
2.1. Characterization of Polyethyleneimine (PEI)-Coated Fe3O4 Nanoparticles (NPs)
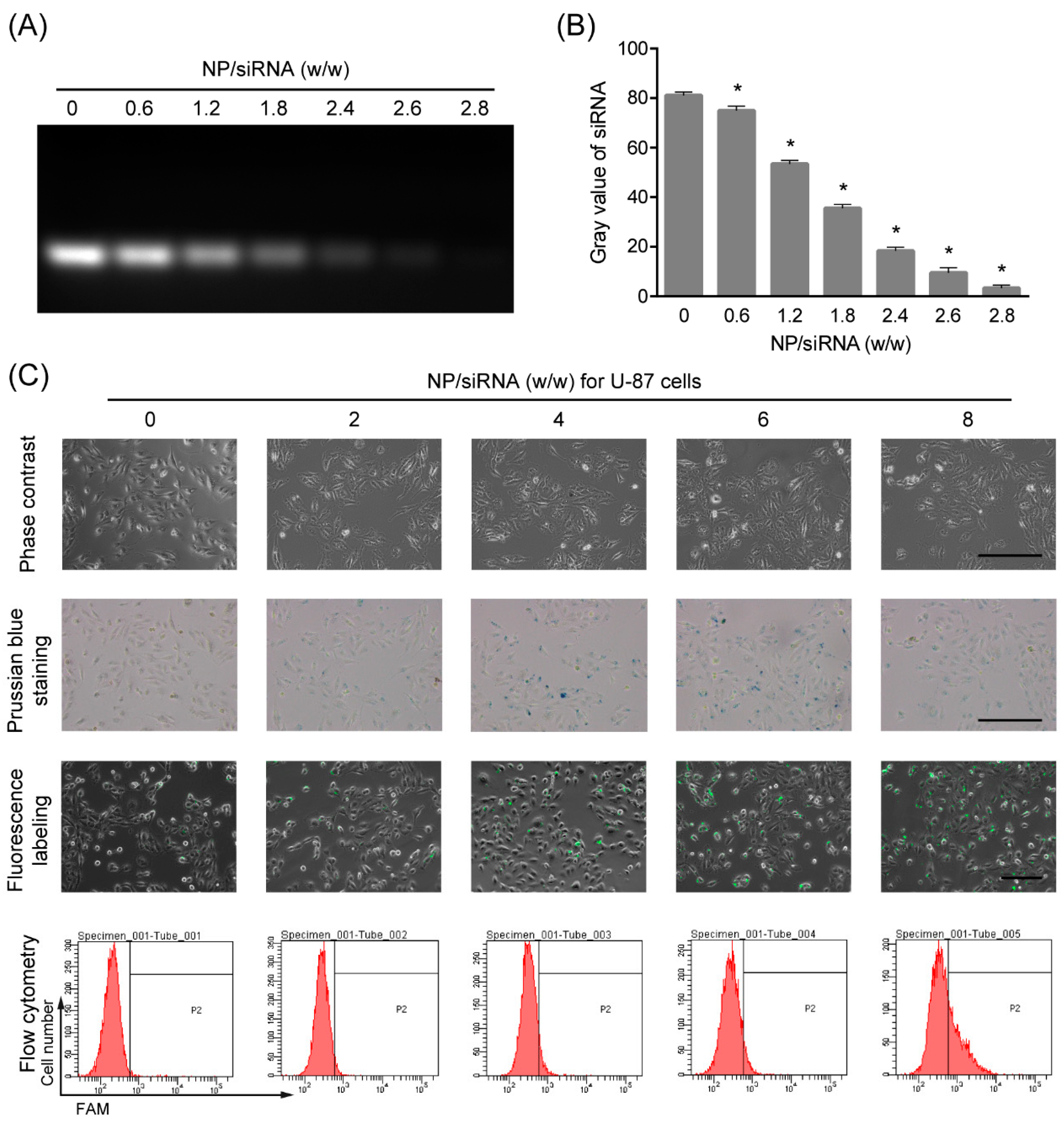
2.2. Interaction between the NP and siRNA
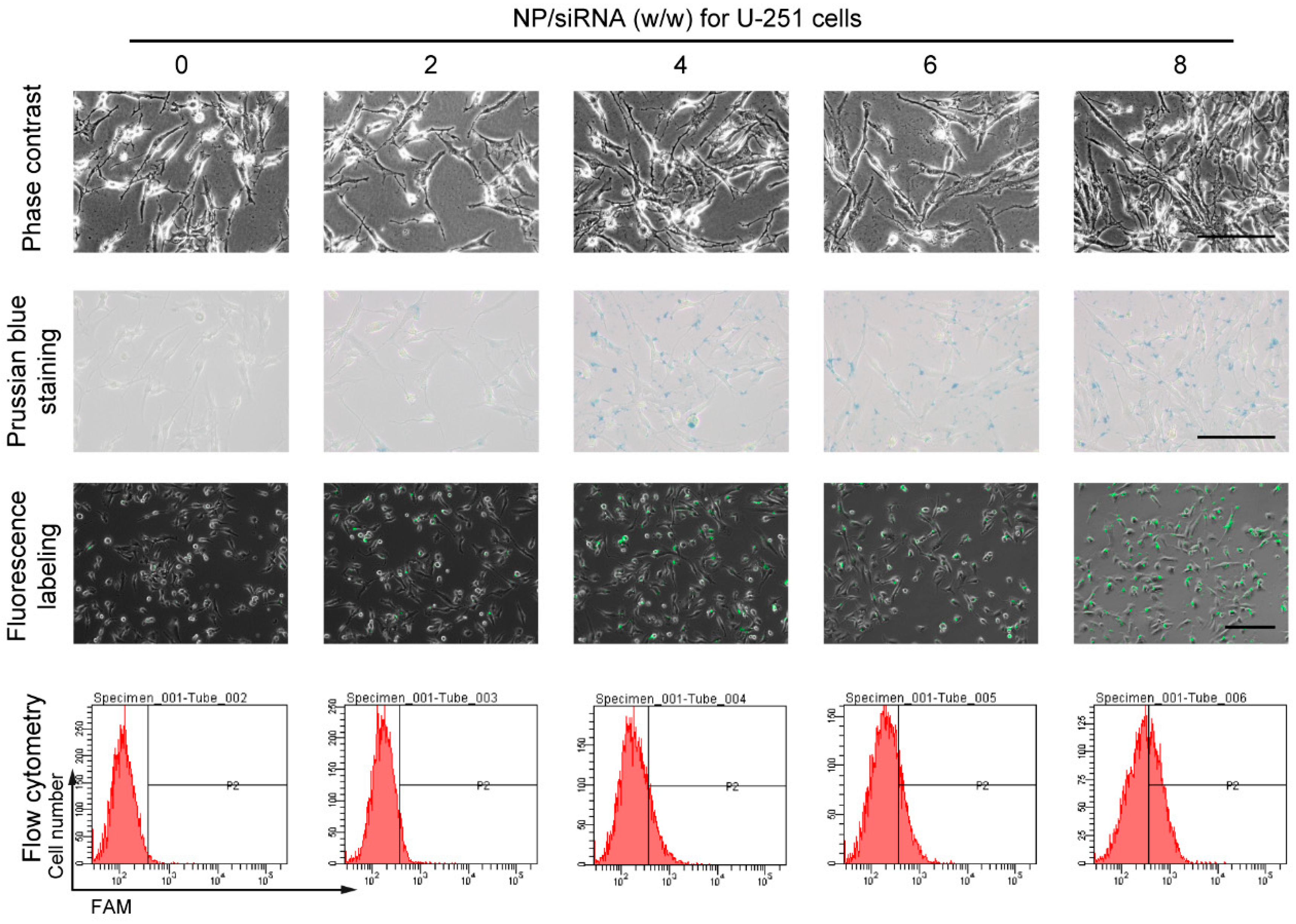
2.3. Cellular Uptake of the NP/siRNA Complexes
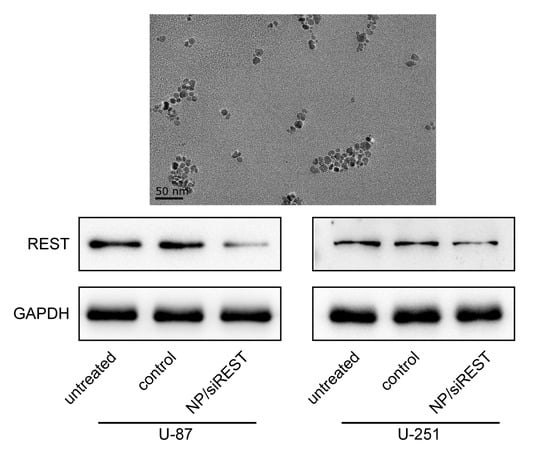
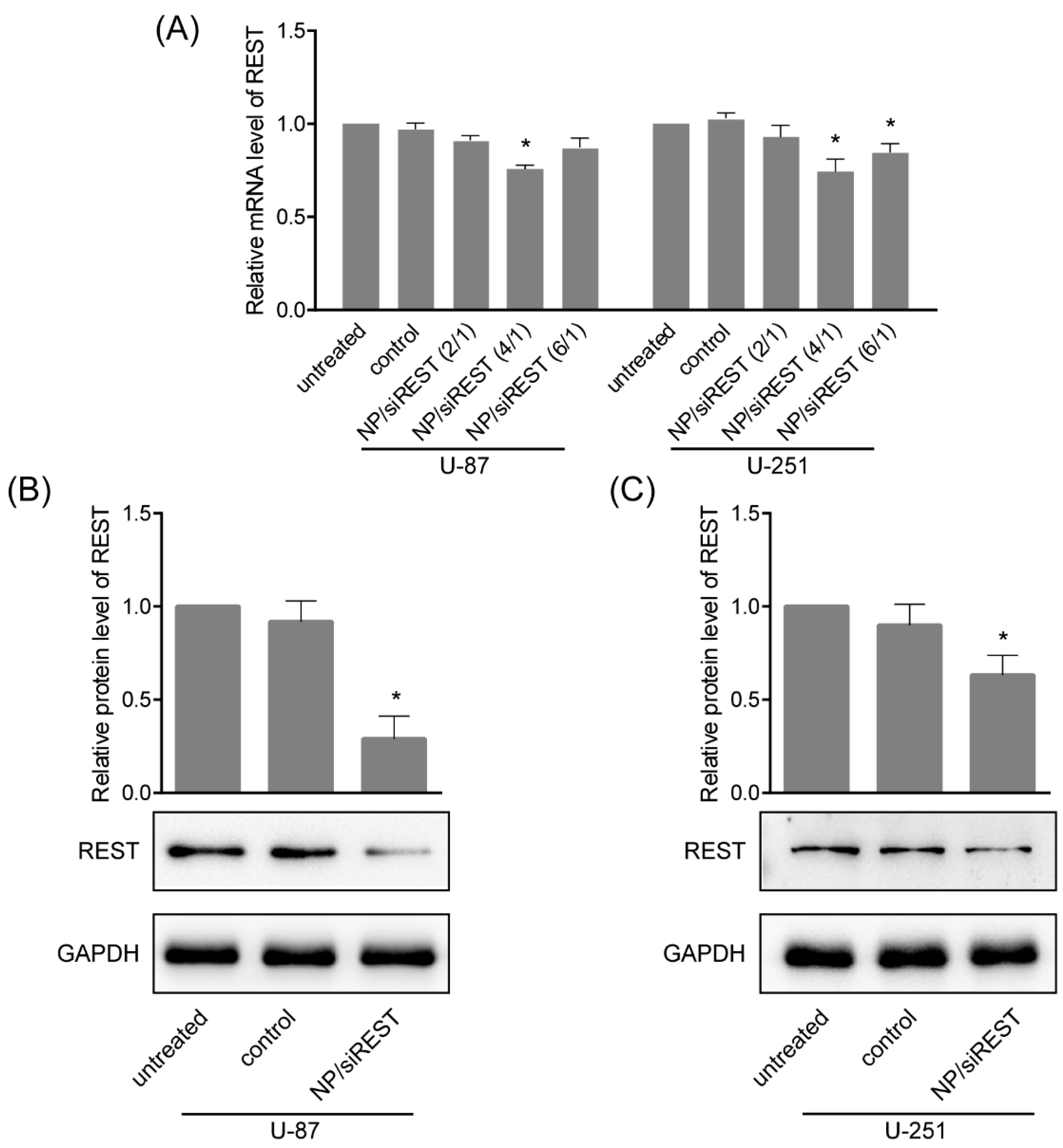
2.4. REST (Repressor Element 1-Silencing Transcription Factor) Silencing Mediated by NP/siRNA Complexes
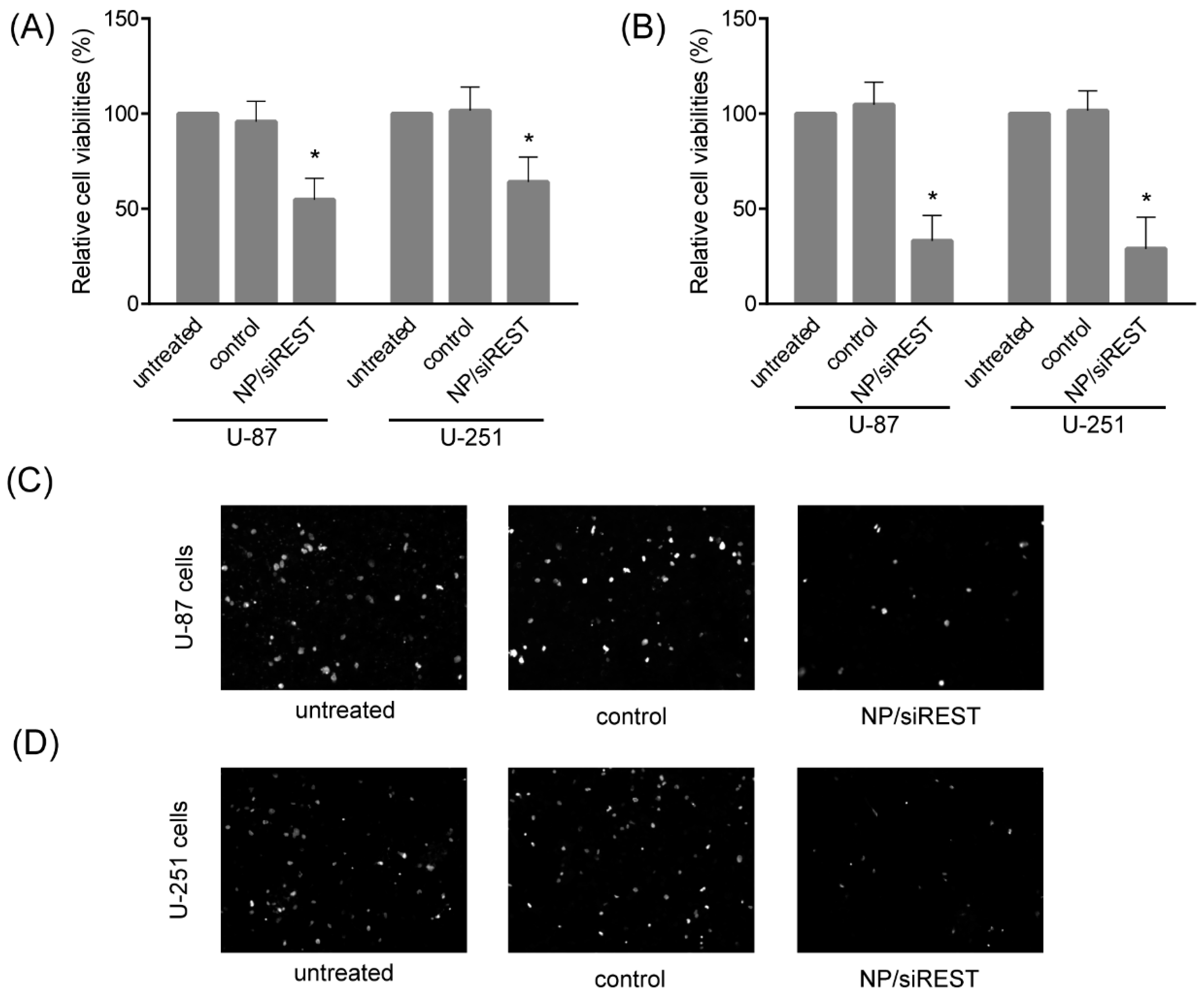
2.5. Anti-Tumor Activity of NP/siRNA Complexes
3. Discussion
4. Materials and Methods
4.1. Synthesis of PEI-Coated Fe3O4 NPs
4.2. NPs Characterization
4.3. Gel Retardation Assay
4.4. Cell Culture
4.5. Transfection of siRNA
4.6. Prussian Blue Staining
4.7. RNA Extraction and Realtime Polymerase Chain Reaction (PCR) Assay
4.8. Protein Extraction and Western Blotting
4.9. Cell Viability Assay
4.10. Transwell Assay
4.11. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Stupp, R.; Mason, W.P.; van Den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Dwivedi, T. A simplified overview of world health organization classification update of central nervous system tumors 2016. J. Neurosci. Rural Pract. 2017, 8, 629–641. [Google Scholar] [CrossRef] [PubMed]
- Omuro, A.; DeAngelis, L.M. Glioblastoma and other malignant gliomas: A clinical review. JAMA 2013, 310, 1842–1850. [Google Scholar] [CrossRef] [PubMed]
- Grossman, S.A.; Ye, X.; Piantadosi, S.; Desideri, S.; Nabors, L.B.; Rosenfeld, M.; Fisher, J.; Consortium, N.C. Survival of patients with newly diagnosed glioblastoma treated with radiation and temozolomide in research studies in the united states. Clin. Cancer Res. 2010, 16, 2443–2449. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Vader, P.; Schiffelers, R.M. Extracellular vesicles for nucleic acid delivery: Progress and prospects for safe RNA-based gene therapy. Gene Ther. 2017, 24, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Lam, J.K.; Chow, M.Y.; Zhang, Y.; Leung, S.W. Sirna versus mirna as therapeutics for gene silencing. Mol. Ther. Nucleic Acids 2015, 4, e252. [Google Scholar] [CrossRef] [PubMed]
- Zuckerman, J.E.; Davis, M.E. Clinical experiences with systemically administered siRNA-based therapeutics in cancer. Nat. Rev. Drug Discov. 2015, 14, 843–856. [Google Scholar] [CrossRef] [PubMed]
- Aigner, A.; Kogel, D. Nanoparticle/sirna-based therapy strategies in glioma: Which nanoparticles, which sirnas? Nanomedicine 2018, 13, 89–103. [Google Scholar] [CrossRef] [PubMed]
- Lukawska, A.; Jagoo, Z.; Kozlowski, G.; Turgut, Z.; Kosai, H.; Sheets, A.; Bixel, T.; Wheatley, A.; Abdulkin, P.; Knappett, B.; et al. Ac magnetic heating of superparamagnetic Fe and Co nanoparticles. Defect Diffus. Forum 2013, 336, 159–167. [Google Scholar] [CrossRef]
- Yoo, B.; Ifediba, M.A.; Ghosh, S.; Medarova, Z.; Moore, A. Combination treatment with theranostic nanoparticles for glioblastoma sensitization to tmz. Mol. Imaging Biol. 2014, 16, 680–689. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.; Qiu, J.; Sun, W.; Yang, J.; Shen, M.; Wang, L.; Shi, X. Multifunctional pei-entrapped gold nanoparticles enable efficient delivery of therapeutic siRNA into glioblastoma cells. Biomater. Sci. 2017, 5, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.B.; Wang, J.; Wang, Z.; Wang, R.; Song, L.; Zhang, T.; Lin, X.W.; Shi, P.; Xin, H.C.; Pang, X.N. Polyethyleneimine-coated Fe3O4 nanoparticles for efficient sirna delivery to human mesenchymal stem cells derived from different tissues. Sci. Adv. Mater. 2015, 7, 1058–1064. [Google Scholar] [CrossRef]
- Rockowitz, S.; Lien, W.H.; Pedrosa, E.; Wei, G.; Lin, M.; Zhao, K.; Lachman, H.M.; Fuchs, E.; Zheng, D. Comparison of rest cistromes across human cell types reveals common and context-specific functions. PLoS Comput. Biol. 2014, 10, e1003671. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Li, Y.; Wang, R.; Li, Y.; Shi, P.; Kan, Z.; Pang, X. Inhibition of rest suppresses proliferation and migration in glioblastoma cells. Int. J. Mol. Sci. 2016, 17, 664. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Kim, A.; Miyata, K.; Kataoka, K. Recent progress in development of siRNA delivery vehicles for cancer therapy. Adv. Drug Deliv. Rev. 2016, 104, 61–77. [Google Scholar] [CrossRef] [PubMed]
- Hendruschk, S.; Wiedemuth, R.; Aigner, A.; Topfer, K.; Cartellieri, M.; Martin, D.; Kirsch, M.; Ikonomidou, C.; Schackert, G.; Temme, A. RNA interference targeting survivin exerts antitumoral effects in vitro and in established glioma xenografts in vivo. Neuro Oncol. 2011, 13, 1074–1089. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Zhang, W.; Shen, Y.; Huang, Q.; Zhou, D.; Guo, S. Efficient RNA delivery by integrin-targeted glutathione responsive polyethyleneimine capped gold nanorods. Acta Biomater. 2015, 23, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Khan, O.F.; Suva, M.L.; Dong, B.; Panek, W.K.; Xiao, T.; Wu, M.; Han, Y.; Ahmed, A.U.; Balyasnikova, I.V.; et al. Multiplexed rnai therapy against brain tumor-initiating cells via lipopolymeric nanoparticle infusion delays glioblastoma progression. Proc. Natl. Acad. Sci. USA 2017, 114, E6147–E6156. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.; Wu, Y.; Alves, C.S.; Shi, X. Efficient delivery of therapeutic siRNA into glioblastoma cells using multifunctional dendrimer-entrapped gold nanoparticles. Nanomedicine 2016, 11, 3103–3115. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zhu, M.; Yu, Y.; Qiu, L.; Zhang, Y.; He, L.; Zhang, J. Brain rest/nrsf is not only a silent repressor but also an active protector. Mol. Neurobiol. 2017, 54, 541–550. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.P.; Zhang, Y.; Zhang, S.; Xia, J.G.; Liu, J.W.; Xu, K.; Gu, N. Preparation and characterization of water-soluble monodisperse magnetic iron oxide nanoparticles via surface double-exchange with dmsa. Colloids Surf. A 2008, 316, 210–216. [Google Scholar] [CrossRef]
- Wang, C.Y.; Hong, J.M.; Chen, G.; Zhang, Y.; Gu, N. Facile method to synthesize oleic acid-capped magnetite nanoparticles. Chin. Chem. Lett. 2010, 21, 179–182. [Google Scholar] [CrossRef]
- Zhang, D.; Wang, R.; Wang, Z.; Dong, J.; Shi, P.; Zhang, T.; Lin, X.; Pang, X. In vitro toxicity of pei-coated Fe3o4 nanoparticles in hacat cells. Mater. Focus 2014, 3, 145–148. [Google Scholar] [CrossRef]
| Gene | Primer Sequence | Product Lengths (bp) |
|---|---|---|
| REST | Forward: 5′-CGCCCATATAAATGTGAACTTTGTC-3′ Reverse: 5′-GGCGGGTTACTTCATGTTGATTAG-3′ | 145 |
| GAPDH | Forward: 5′-GCACCGTCAAGGCTGAGAAC-3′ Reverse: 5′-TGGTGAAGACGCCAGTGGA-3′ | 138 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, R.; Degirmenci, V.; Xin, H.; Li, Y.; Wang, L.; Chen, J.; Hu, X.; Zhang, D. PEI-Coated Fe3O4 Nanoparticles Enable Efficient Delivery of Therapeutic siRNA Targeting REST into Glioblastoma Cells. Int. J. Mol. Sci. 2018, 19, 2230. https://doi.org/10.3390/ijms19082230
Wang R, Degirmenci V, Xin H, Li Y, Wang L, Chen J, Hu X, Zhang D. PEI-Coated Fe3O4 Nanoparticles Enable Efficient Delivery of Therapeutic siRNA Targeting REST into Glioblastoma Cells. International Journal of Molecular Sciences. 2018; 19(8):2230. https://doi.org/10.3390/ijms19082230
Chicago/Turabian StyleWang, Rui, Volkan Degirmenci, Hongchuan Xin, Ying Li, Liping Wang, Jiayu Chen, Xiaoyu Hu, and Dianbao Zhang. 2018. "PEI-Coated Fe3O4 Nanoparticles Enable Efficient Delivery of Therapeutic siRNA Targeting REST into Glioblastoma Cells" International Journal of Molecular Sciences 19, no. 8: 2230. https://doi.org/10.3390/ijms19082230
APA StyleWang, R., Degirmenci, V., Xin, H., Li, Y., Wang, L., Chen, J., Hu, X., & Zhang, D. (2018). PEI-Coated Fe3O4 Nanoparticles Enable Efficient Delivery of Therapeutic siRNA Targeting REST into Glioblastoma Cells. International Journal of Molecular Sciences, 19(8), 2230. https://doi.org/10.3390/ijms19082230