Chemokine CCL25 Induces Migration and Extracellular Matrix Production of Anulus Fibrosus-Derived Cells
Abstract
1. Introduction
2. Results
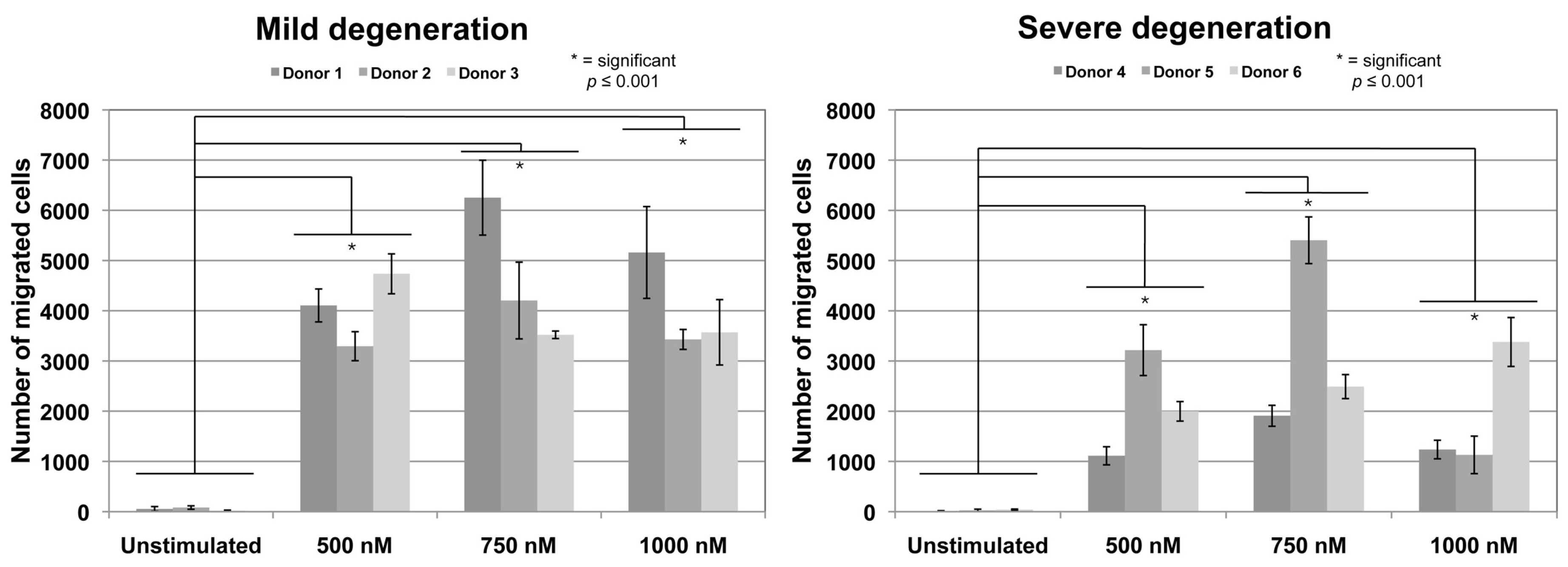
2.1. Chemotaxis Assay
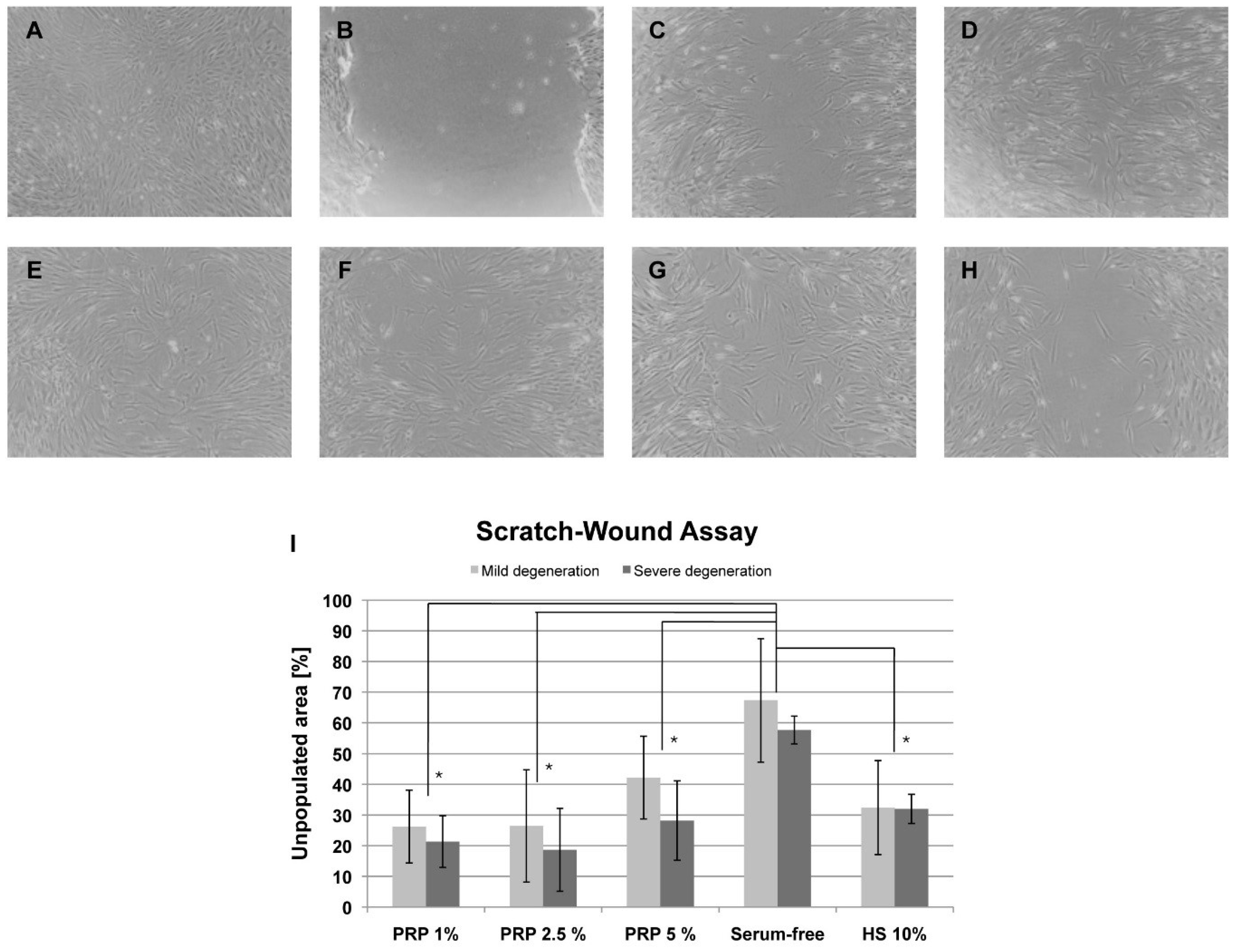
2.2. Scratch-Wound Assay
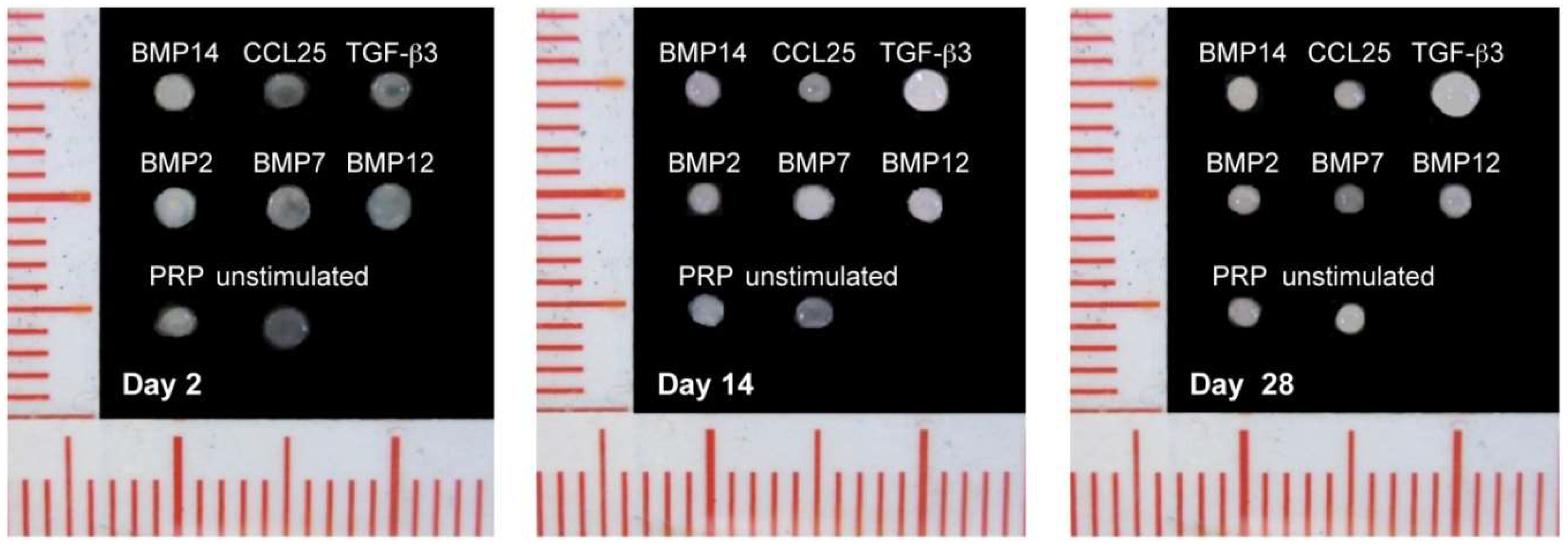
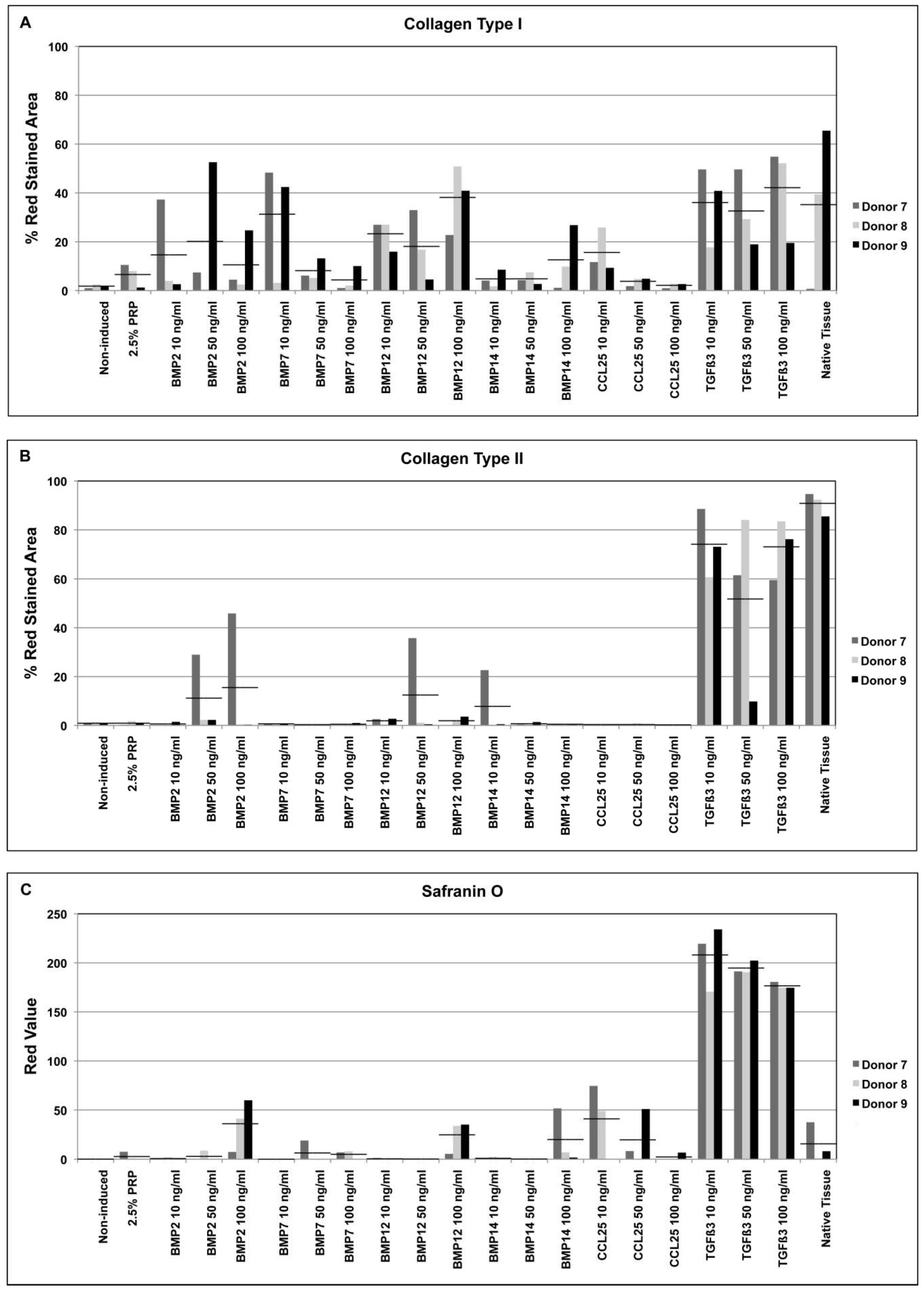
2.3. Factor Screening Assay
3. Discussion
4. Materials and Methods
4.1. Preparation of Human Serum and PRP
4.2. Isolation and Cultivation of Anulus Fibrosus Cells
4.3. Chemotaxis Assay
4.4. Scratch-Wound Assay
4.5. Factor Screening Assay
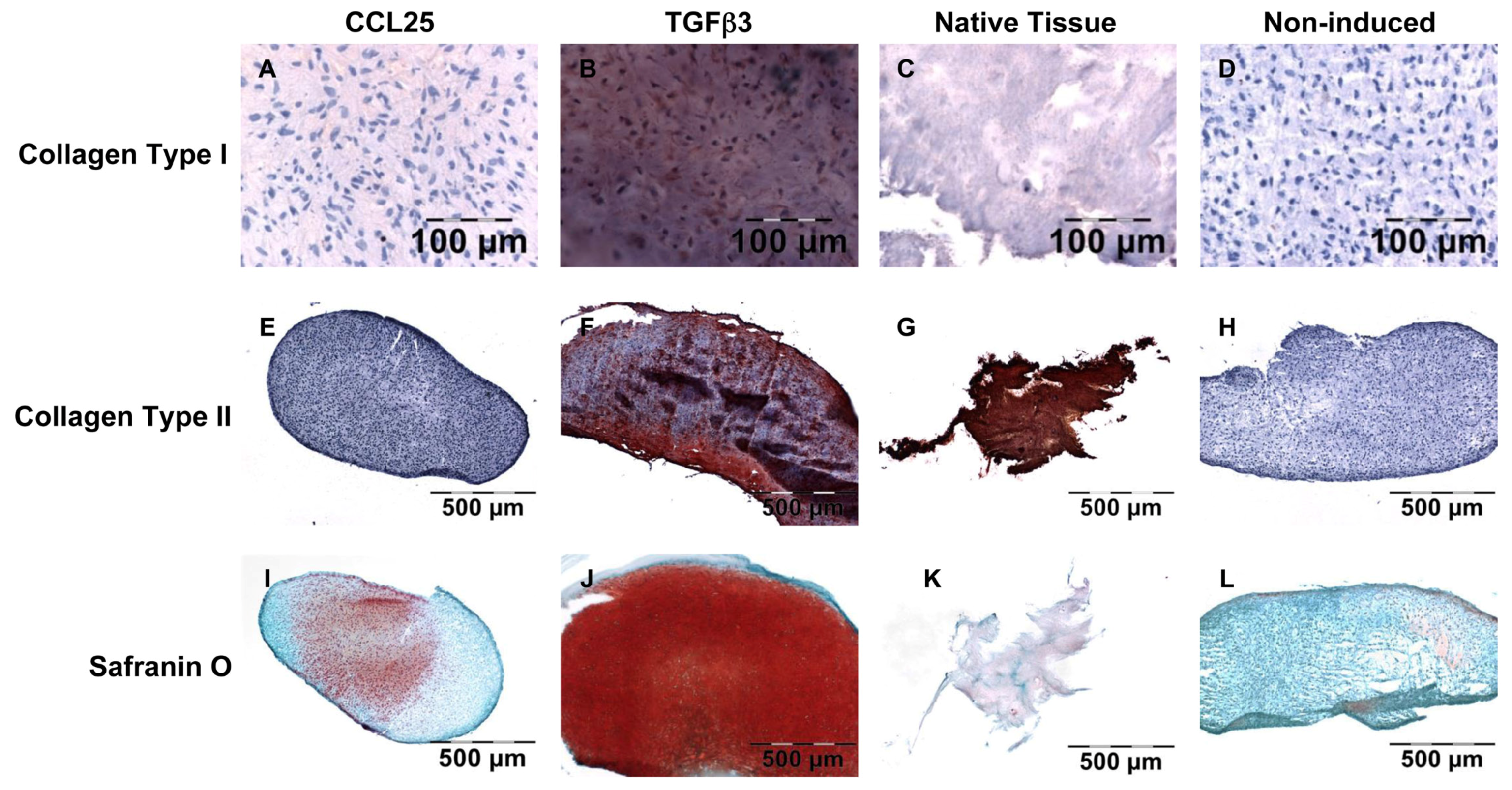
4.6. Histological and Immunohistochemical Analysis of Anulus Fibrosus Matrix Production
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ACD | Acid citrate dextrose |
| AF | Anulus fibrosus |
| BMP | Bone morphogenetic protein |
| CCL | C-C motif chemokine ligand |
| ECM | Extracellular matrix |
| GDF | Growth differentiation factor |
| IVD | Intervertebral disc |
| PG | Proteoglycan |
| PRP | Platelet-rich plasma |
| TECK | Thymus—expressed chemokine |
| TGF | Transforming growth factor |
References
- Schmidt, C.O.; Raspe, H.; Pfingsten, M.; Hasenbring, M.; Basler, H.D.; Eich, W.; Kohlmann, T. Back Pain in the German Adult Population: Prevalence, Severity, and Sociodemographic Correlates in a Multiregional Survey. Spine 2007, 32, 2005–2011. [Google Scholar] [CrossRef] [PubMed]
- Wenig, C.M.; Schmidt, C.O.; Kohlmann, T.; Schweikert, B. Costs of Back Pain in Germany. Eur. J. Pain 2009, 13, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Parker, S.L.; Mendenhall, S.K.; Godil, S.S.; Sivasubramanian, P.; Cahill, K.; Ziewacz, J.; McGirt, M.J. Incidence of Low Back Pain After Lumbar Discectomy for Herniated Disc and Its Effect on Patient-Reported Outcomes. Clin. Orthop. Relat. Res. 2015, 473, 1988–1999. [Google Scholar] [CrossRef] [PubMed]
- Parker, S.L.; Grahovac, G.; Vukas, D.; Vilendecic, M.; Ledic, D.; McGirt, M.J.; Carragee, E.J. Effect of an Annular Closure Device (Barricaid) on Same-Level Recurrent Disk Herniation and Disk Height Loss after Primary Lumbar Discectomy: Two-Year Results of a Multicenter Prospective Cohort Study. Clin. Spine Surg. 2016, 29, 454–460. [Google Scholar] [CrossRef] [PubMed]
- Lange, N.; Meyer, B.; Shiban, E. Symptomatic Annulus-Repair-Device Loosening due to a Low-Grade Infection. Acta Neurochir. (Wien) 2018, 160, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Ledet, E.H.; Jeshuran, W.; Glennon, J.C.; Shaffrey, C.; de Deyne, P.; Belden, C.; Kallakury, B.; Carl, A.L. Small Intestinal Submucosa for Anular Defect Closure: Long-Term Response in an in vivo Sheep Model. Spine 2009, 34, 1457–1463. [Google Scholar] [CrossRef] [PubMed]
- Pirvu, T.; Blanquer, S.B.; Benneker, L.M.; Grijpma, D.W.; Richards, R.G.; Alini, M.; Eglin, D.; Grad, S.; Li, Z. A Combined Biomaterial and Cellular Approach for Annulus Fibrosus Rupture Repair. Biomaterials 2015, 42, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.P.; Oegema, T.R., Jr.; Bradford, D.S. Stimulation of Mature Canine Intervertebral Disc by Growth Factors. Spine 1991, 16, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Tim Yoon, S.; Su Kim, K.; Li, J.; Soo Park, J.; Akamaru, T.; Elmer, W.A.; Hutton, W.C. The Effect of Bone Morphogenetic Protein-2 on Rat Intervertebral Disc Cells in vitro. Spine 2003, 28, 1773–1780. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; An, H.S.; Song, S.; Toofanfard, M.; Masuda, K.; Andersson, G.B.; Thonar, E.J. Growth Factor Osteogenic Protein-1: Differing Effects on Cells from Three Distinct Zones in the Bovine Intervertebral Disc. Am. J. Phys. Med. Rehabilit. 2004, 83, 515–521. [Google Scholar] [CrossRef]
- Chujo, T.; An, H.S.; Akeda, K.; Miyamoto, K.; Muehleman, C.; Attawia, M.; Andersson, G.; Masuda, K. Effects of Growth Differentiation Factor-5 on the Intervertebral Disc—In Vitro Bovine Study and in vivo Rabbit Disc Degeneration Model Study. Spine 2006, 31, 2909–2917. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Anderson, D.G.; Phillips, F.M.; Thonar, E.J.; He, T.C.; Pietryla, D.; An, H.S. Comparative Effects of Bone Morphogenetic Proteins and Sox9 Overexpression on Matrix Accumulation by Bovine Anulus Fibrosus Cells: Implications for Anular Repair. Spine 2007, 32, 2515–2520. [Google Scholar] [CrossRef] [PubMed]
- Imai, Y.; Miyamoto, K.; An, H.S.; Thonar, E.J.; Andersson, G.B.; Masuda, K. Recombinant Human Osteogenic Protein-1 Upregulates Proteoglycan Metabolism of Human Anulus Fibrosus and Nucleus Pulposus Cells. Spine 2007, 32, 1303–1309. [Google Scholar] [CrossRef] [PubMed]
- Stich, S.; Loch, A.; Leinhase, I.; Neumann, K.; Kaps, C.; Sittinger, M.; Ringe, J. Human Periosteum-Derived Progenitor Cells Express Distinct Chemokine Receptors and Migrate Upon Stimulation with CCL2, CCL25, CXCL8, CXCL12, and CXCL13. Eur. J. Cell Biol. 2008, 87, 365–376. [Google Scholar] [CrossRef] [PubMed]
- Binger, T.; Stich, S.; Andreas, K.; Kaps, C.; Sezer, O.; Notter, M.; Sittinger, M.; Ringe, J. Migration Potential and Gene Expression Profile of Human Mesenchymal Stem Cells Induced by CCL25. Exp. Cell Res. 2009, 315, 1468–1479. [Google Scholar] [CrossRef] [PubMed]
- Freeman, B.J.; Kuliwaba, J.S.; Jones, C.F.; Shu, C.C.; Colloca, C.J.; Zarrinkalam, M.R.; Mulaibrahimovic, A.; Gronthos, S.; Zannettino, A.C.; Howell, S. Allogeneic Mesenchymal Precursor Cells Promote Healing in Postero-Lateral Annular Lesions and Improve Indices of Lumbar Intervertebral Disc Degeneration in an Ovine Model. Spine 2016, 41, 1331–1339. [Google Scholar] [CrossRef] [PubMed]
- Kruger, J.P.; Freymannx, U.; Vetterlein, S.; Neumann, K.; Endres, M.; Kaps, C. Bioactive Factors in Platelet-Rich Plasma Obtained by Apheresis. Transfus. Med. Hemother. 2013, 40, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Pirvu, T.N.; Schroeder, J.E.; Peroglio, M.; Verrier, S.; Kaplan, L.; Richards, R.G.; Alini, M.; Grad, S. Platelet-Rich Plasma Induces Annulus Fibrosus Cell Proliferation and Matrix Production. Eur. Spine J. 2014, 23, 745–753. [Google Scholar] [CrossRef] [PubMed]
- Eyre, D.R.; Muir, H. Quantitative Analysis of Types I and II Collagens in Human Intervertebral Discs at Various Ages. Biochim. Biophys. Acta 1977, 492, 29–42. [Google Scholar] [CrossRef]
- Urban, J.P.; Roberts, S. Degeneration of the Intervertebral Disc. Arthritis Res. Ther. 2003, 5, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Hondke, S.; Cabraja, M.; Kruger, J.P.; Stich, S.; Hartwig, T.; Sittinger, M.; Endres, M. Proliferation, Migration, and ECM Formation Potential of Human Annulus Fibrosus Cells Is Independent of Degeneration Status. Cartilage 2018. [Google Scholar] [CrossRef] [PubMed]
- Mwale, F.; Roughley, P.; Antoniou, J. Distinction between the Extracellular Matrix of the Nucleus Pulposus and Hyaline Cartilage: A Requisite for Tissue Engineering of Intervertebral Disc. Eur. Cell Mater. 2004, 8, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Akeda, K.; An, H.S.; Pichika, R.; Attawia, M.; Thonar, E.J.; Lenz, M.E.; Uchida, A.; Masuda, K. Platelet-Rich Plasma (PRP) Stimulates the Extracellular Matrix Metabolism of Porcine Nucleus Pulposus and Anulus Fibrosus Cells Cultured in Alginate Beads. Spine 2006, 31, 959–966. [Google Scholar] [CrossRef] [PubMed]
- Hegewald, A.A.; Neumann, K.; Kalwitz, G.; Freymann, U.; Endres, M.; Schmieder, K.; Kaps, C.; Thome, C. The Chemokines CXCL10 and XCL1 Recruit Human Annulus Fibrosus Cells. Spine 2012, 37, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Endres, M.; Andreas, K.; Kalwitz, G.; Freymann, U.; Neumann, K.; Ringe, J.; Sittinger, M.; Kaps, C. Chemokine Profile of Synovial Fluid from Normal, Osteoarthritis and Rheumatoid Arthritis Patients: CCL25, CXCL10 and XCL1 Recruit Human Subchondral Mesenchymal Progenitor Cells. Osteoarthr. Cartil. 2010, 18, 1458–1466. [Google Scholar] [CrossRef] [PubMed]
- Mishima, Y.; Lotz, M. Chemotaxis of Human Articular Chondrocytes and Mesenchymal Stem Cells. J. Orthop. Res. 2008, 26, 1407–1412. [Google Scholar] [CrossRef] [PubMed]
- Haschtmann, D.; Ferguson, S.J.; Stoyanov, J.V. BMP-2 and TGF-β3 do not Prevent Spontaneous Degeneration in Rabbit Disc Explants but Induce Ossification of the Annulus Fibrosus. Eur. Spine J. 2012, 21, 1724–1733. [Google Scholar] [CrossRef] [PubMed]
- Burkus, J.K.; Gornet, M.F.; Dickman, C.A.; Zdeblick, T.A. Anterior Lumbar Interbody Fusion Using rhBMP-2 with Tapered Interbody Cages. J. Spinal Disord. Tech. 2002, 15, 337–349. [Google Scholar] [CrossRef] [PubMed]
- Hustedt, J.W.; Blizzard, D.J. The Controversy Surrounding Bone Morphogenetic Proteins in the Spine: A Review of Current Research. Yale J. Biol. Med. 2014, 87, 549–561. [Google Scholar] [PubMed]
- Vaccaro, A.R.; Patel, T.; Fischgrund, J.; Anderson, D.G.; Truumees, E.; Herkowitz, H.; Phillips, F.; Hilibrand, A.; Albert, T.J. A 2-Year Follow-Up Pilot Study Evaluating the Safety and efficacy of op-1 putty (rhbmp-7) as an Adjunct to Iliac Crest Autograft in Posterolateral Lumbar Fusions. Eur. Spine J. 2005, 14, 623–629. [Google Scholar] [CrossRef] [PubMed]
- Vaccaro, A.R.; Whang, P.G.; Patel, T.; Phillips, F.M.; Anderson, D.G.; Albert, T.J.; Hilibrand, A.S.; Brower, R.S.; Kurd, M.F.; Appannagari, A.; et al. The Safety and Efficacy of OP-1 (rhBMP-7) as a Replacement for Iliac Crest Autograft for Posterolateral Lumbar Arthrodesis: Minimum 4-Year Follow-Up of a Pilot Study. Spine J. 2008, 8, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Magit, D.P.; Maak, T.; Trioano, N.; Raphael, B.; Hamouria, Q.; Polzhofer, G.; Drespe, I.; Albert, T.J.; Grauer, J.N. Healos/Recombinant Human Growth and Differentiation Factor-5 Induces Posterolateral Lumbar Fusion in a New Zealand White Rabbit Model. Spine 2006, 31, 2180–2188. [Google Scholar] [CrossRef] [PubMed]
- Kennon, J.C.; Awad, M.E.; Chutkan, N.; DeVine, J.; Fulzele, S. Current Insights on Use of Growth Factors as Therapy for Intervertebral Disc Degeneration. Biomol. Concepts 2018, 9, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Pfirrmann, C.W.; Metzdorf, A.; Zanetti, M.; Hodler, J.; Boos, N. Magnetic Resonance Classification of Lumbar Intervertebral Disc Degeneration. Spine 2001, 26, 1873–1878. [Google Scholar] [PubMed]
- Geback, T.; Schulz, M.M.; Koumoutsakos, P.; Detmar, M. TScratch: A Novel and Simple Software Tool for Automated Analysis of Monolayer Wound Healing Assays. BioTechniques 2009, 46, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Schlichting, N.; Dehne, T.; Mans, K.; Endres, M.; Stuhlmuller, B.; Sittinger, M.; Kaps, C.; Ringe, J. Suitability of Porcine Chondrocyte Micromass Culture to Model Osteoarthritis in Vitro. Mol. Pharm. 2014, 11, 2092–2105. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stich, S.; Möller, A.; Cabraja, M.; Krüger, J.P.; Hondke, S.; Endres, M.; Ringe, J.; Sittinger, M. Chemokine CCL25 Induces Migration and Extracellular Matrix Production of Anulus Fibrosus-Derived Cells. Int. J. Mol. Sci. 2018, 19, 2207. https://doi.org/10.3390/ijms19082207
Stich S, Möller A, Cabraja M, Krüger JP, Hondke S, Endres M, Ringe J, Sittinger M. Chemokine CCL25 Induces Migration and Extracellular Matrix Production of Anulus Fibrosus-Derived Cells. International Journal of Molecular Sciences. 2018; 19(8):2207. https://doi.org/10.3390/ijms19082207
Chicago/Turabian StyleStich, Stefan, Anke Möller, Mario Cabraja, Jan Philipp Krüger, Sylvia Hondke, Michaela Endres, Jochen Ringe, and Michael Sittinger. 2018. "Chemokine CCL25 Induces Migration and Extracellular Matrix Production of Anulus Fibrosus-Derived Cells" International Journal of Molecular Sciences 19, no. 8: 2207. https://doi.org/10.3390/ijms19082207
APA StyleStich, S., Möller, A., Cabraja, M., Krüger, J. P., Hondke, S., Endres, M., Ringe, J., & Sittinger, M. (2018). Chemokine CCL25 Induces Migration and Extracellular Matrix Production of Anulus Fibrosus-Derived Cells. International Journal of Molecular Sciences, 19(8), 2207. https://doi.org/10.3390/ijms19082207




