Intracellular Ca2+ Increases and Connexin 43 Hemichannel Opening Are Necessary but Not Sufficient for Thy-1-Induced Astrocyte Migration
Abstract
1. Introduction
2. Results
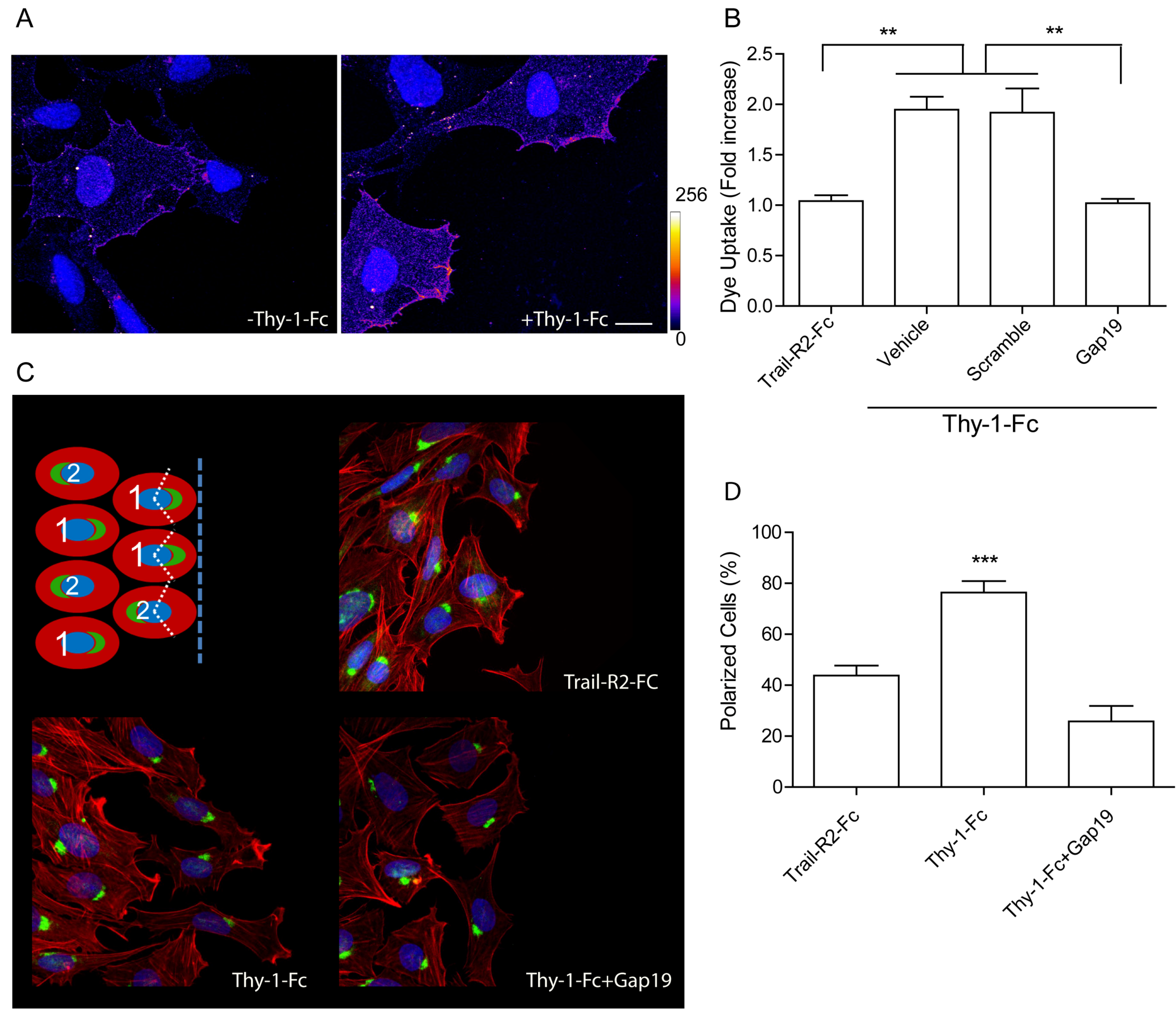
2.1. Thy-1 Enhances Cell Surface Cx43 Levels, Opens Hemichannels, and Induces Cell Polarization
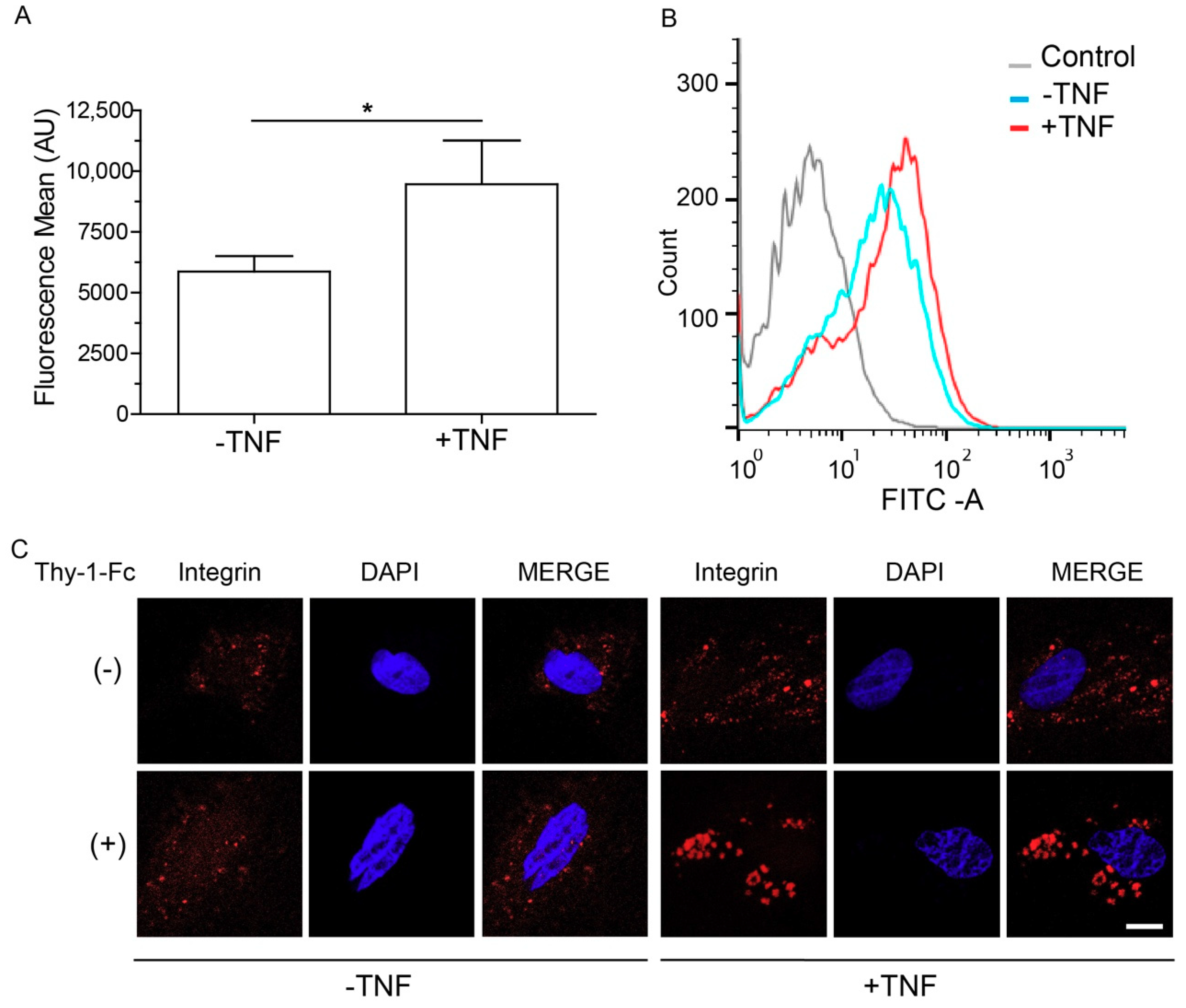
2.2. Levels of αvβ3 Integrin Increase at the Cell Membrane and Form β3 Integrin Receptor Microclusters
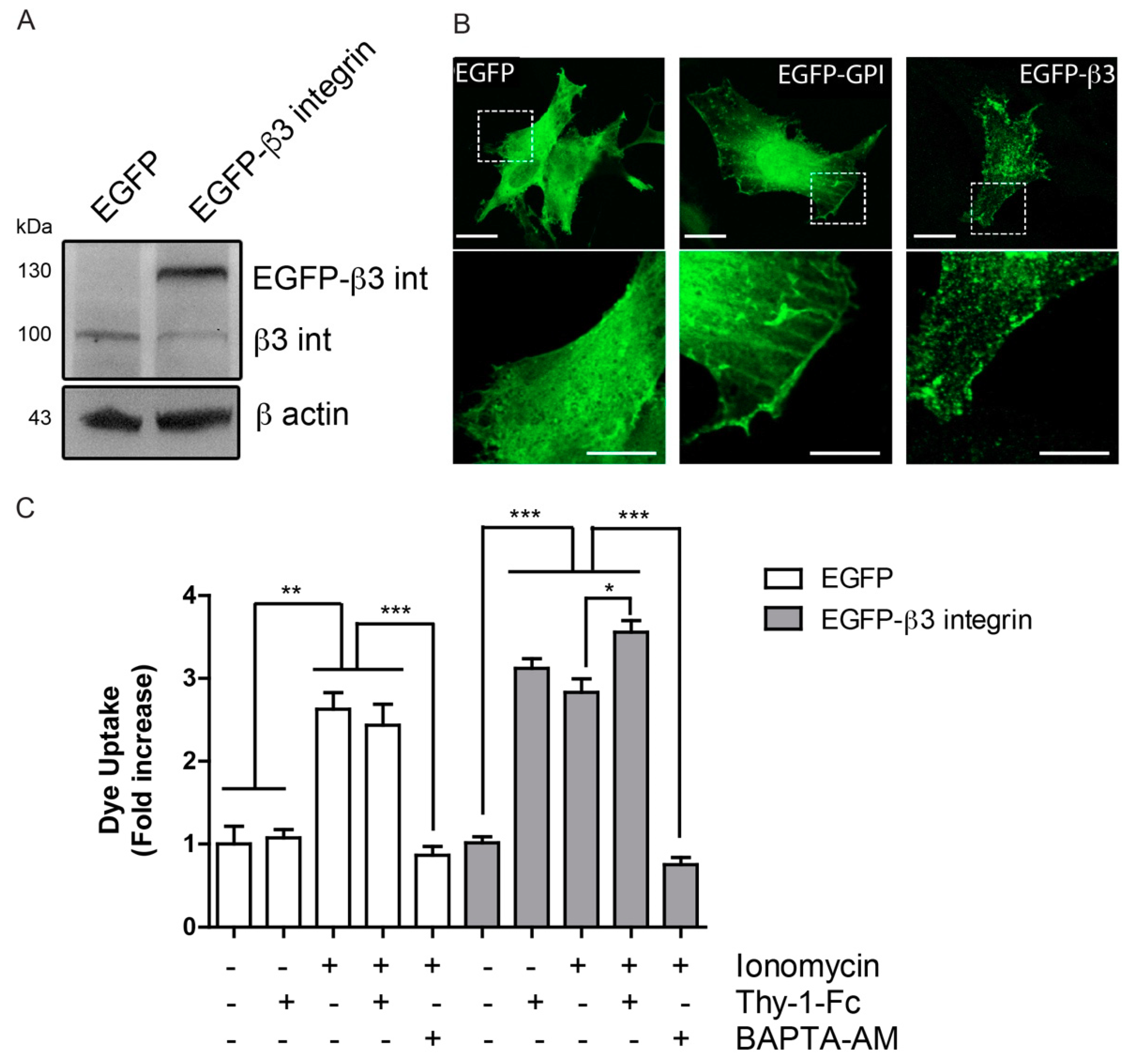
2.3. β3 Integrin Overexpression Leads to Formation of Integrin Microclusters at the Cell Surface
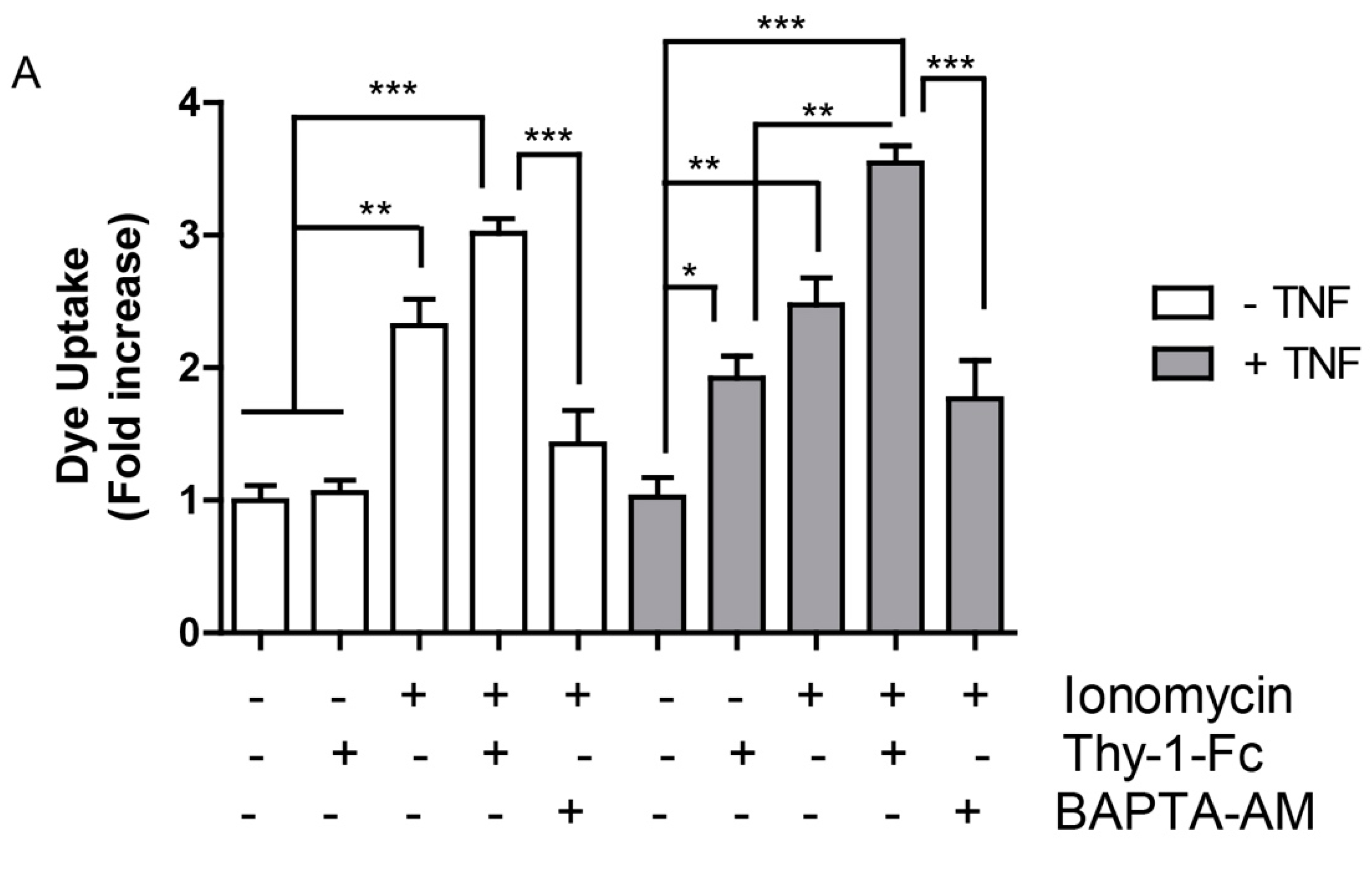
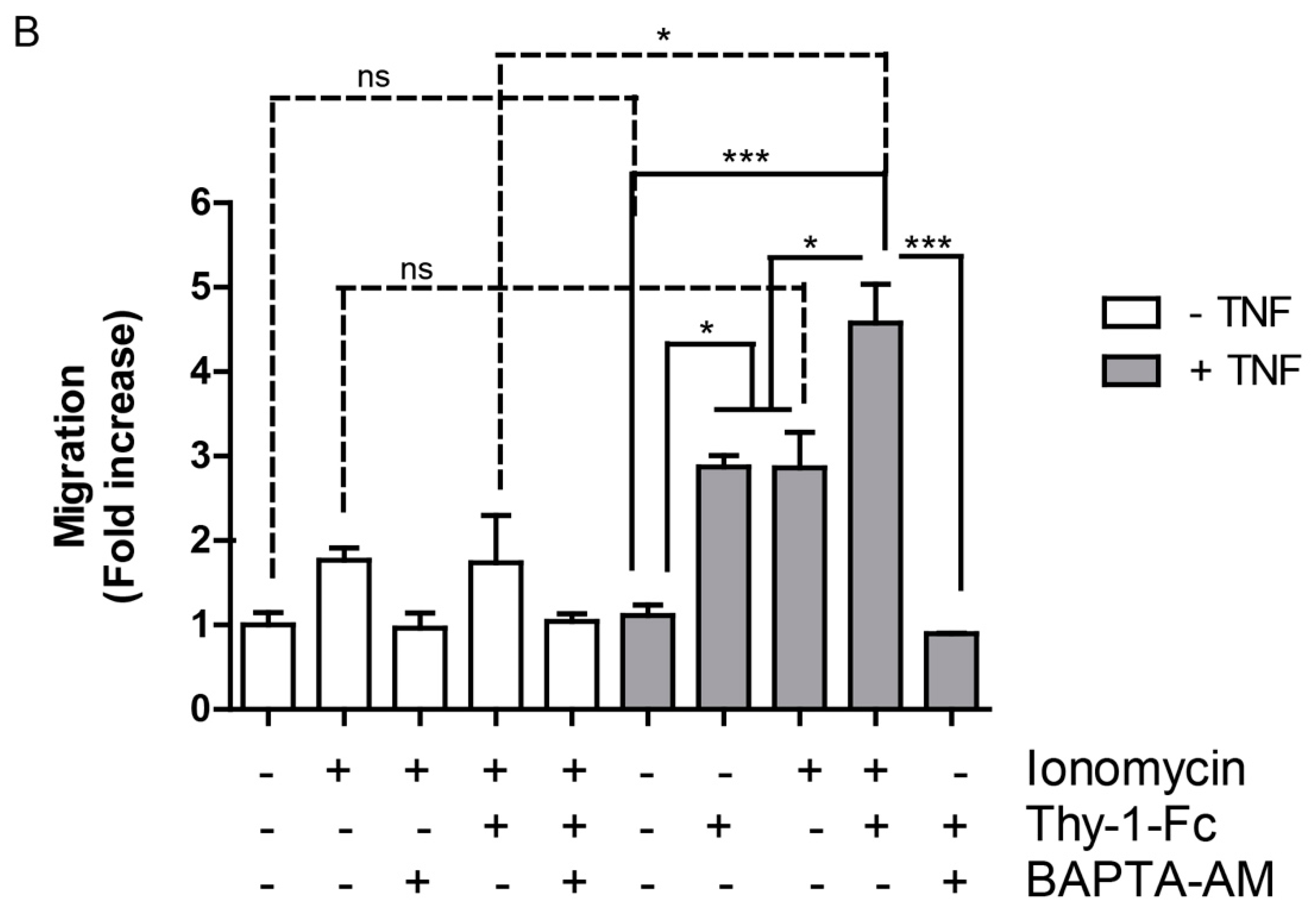
2.4. Hemichannel Opening and [Ca2+]i Increases Are Necessary, but Not Sufficient to Promote Cell Migration
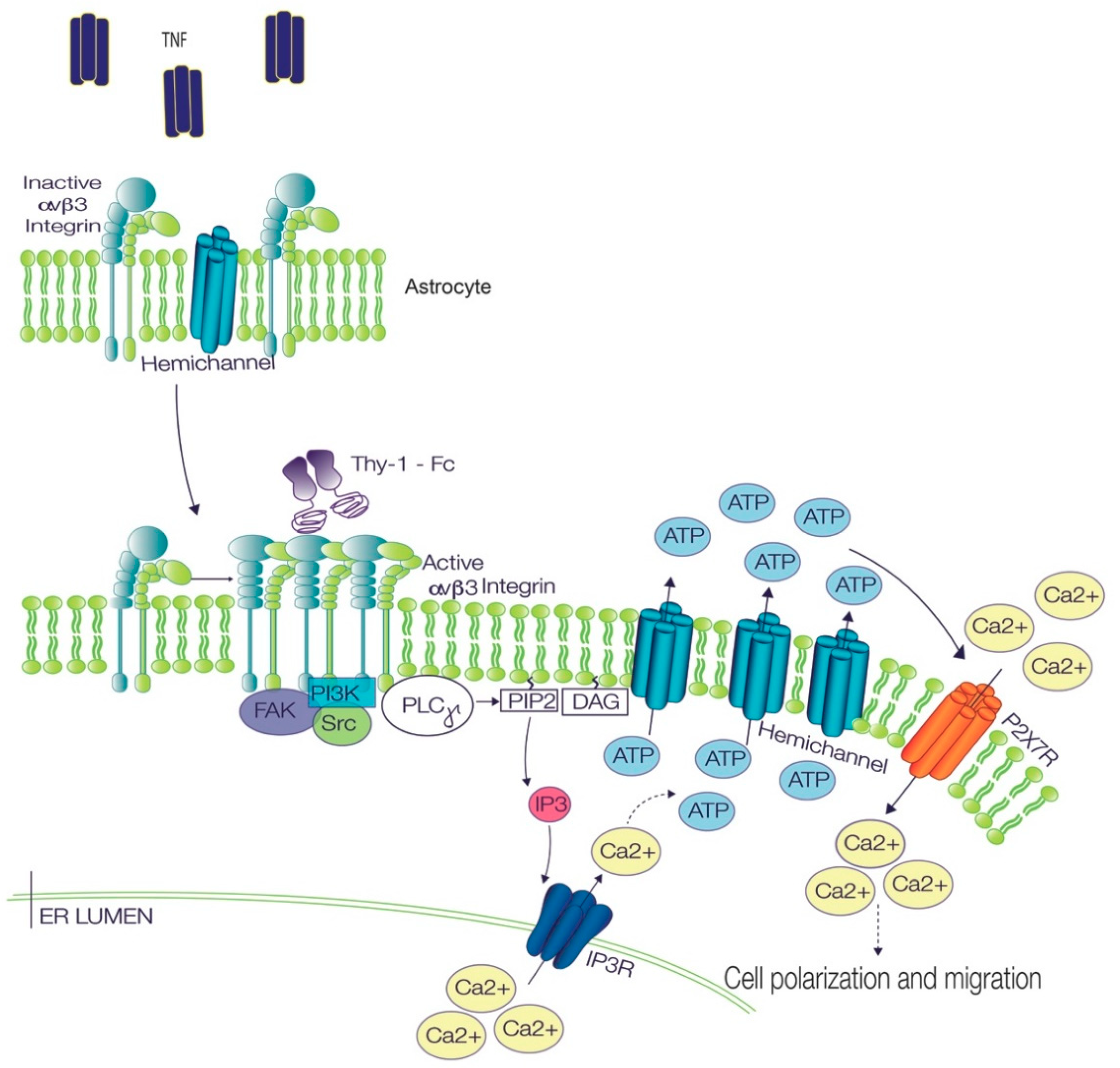
3. Discussion
4. Materials and Methods
4.1. Cell Line and Primary Cultures
4.2. Plasmids and Transfections
4.3. Fusion Proteins
4.4. Western Blot
4.5. Indirect Immunofluorescence
4.6. Dye Uptake
4.7. Cell Polarity Assay
4.8. Transwell Migration Assay
4.9. Cell Cytometry
4.10. Cluster Formation and Confocal Microscopy
4.11. Statistical Analysis
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| TNF | Tumor necrosis factor |
| Cx43 | Connexin 43 |
| IP3R | Inositol 1,4,5-trisphosphate receptor |
| LY | Lucifer yellow |
| Ca2+ | Calcium |
| [Ca2+]i | Intracellular Ca2+ concentration |
| CNS | Central nervous system |
| Px | Pannexin |
References
- Allen, N.J.; Eroglu, C. Cell Biology of Astrocyte-Synapse Interactions. Neuron 2017, 96, 697–708. [Google Scholar] [CrossRef] [PubMed]
- Bardehle, S.; Kruger, M.; Buggenthin, F.; Schwausch, J.; Ninkovic, J.; Clevers, H.; Snippert, H.J.; Theis, F.J.; Meyer-Luehmann, M.; Bechmann, I.; et al. Live imaging of astrocyte responses to acute injury reveals selective juxtavascular proliferation. Nat. Neurosci. 2013, 16, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Bardehle, S.; Krüger, M.; Buggenthin, F.; Schwausch, J.; Ninkovic, J.; Clevers, H.; Snippert, H.J.; Theis, F.J.; Meyer-Luehmann, M.; Bechmann, I.; et al. Elusive roles for reactive astrocytes in neurodegenerative diseases. Front. Cell. Neurosci. 2015, 9, 278. [Google Scholar]
- Lagos-Cabre, R.; Alvarez, A.; Kong, M.; Burgos-Bravo, F.; Cardenas, A.; Rojas-Mancilla, E.; Perez-Nunez, R.; Herrera-Molina, R.; Rojas, F.; Schneider, P.; et al. αvβ3 Integrin regulates astrocyte reactivity. J. Neuroinflamm. 2017, 14, 194. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, A.; Lagos-Cabré, R.; Kong, M.; Cárdenas, A.; Burgos-Bravo, F.; Schneider, P.; Quest, A.F.; Leyton, L. Integrin-mediated transactivation of P2X7R via hemichannel-dependent ATP release stimulates astrocyte migration. Biochim. Biophys. Acta 2016, 1863, 2175–2188. [Google Scholar] [CrossRef] [PubMed]
- Abudara, V.; Roux, L.; Dallérac, G.; Matias, I.; Dulong, J.; Mothet, J.P.; Rouach, N.; Giaume, C. Activated microglia impairs neuroglial interaction by opening Cx43 hemichannels in hippocampal astrocytes. Glia 2015, 63, 795–811. [Google Scholar] [CrossRef] [PubMed]
- Garré, J.M.; Yang, G.; Bukauskas, F.F.; Bennett, M.V. FGF-1 Triggers Pannexin-1 Hemichannel Opening in Spinal Astrocytes of Rodents and Promotes Inflammatory Responses in Acute Spinal Cord Slices. J. Neurosci. 2016, 36, 4785–4801. [Google Scholar] [CrossRef] [PubMed]
- Henriquez, M.; Herrera-Molina, R.; Valdivia, A.; Alvarez, A.; Kong, M.; Munoz, N.; Eisner, V.; Jaimovich, E.; Schneider, P.; Quest, A.F.; et al. ATP release due to Thy-1-integrin binding induces P2X7-mediated calcium entry required for focal adhesion formation. J. Cell Sci. 2011, 124, 1581–1588. [Google Scholar] [CrossRef] [PubMed]
- De Vuyst, E.; Wang, N.; Decrock, E.; De Bock, M.; Vinken, M.; Van Moorhem, M.; Lai, C.; Culot, M.; Rogiers, V.; Cecchelli, R.; et al. Ca(2+) regulation of connexin 43 hemichannels in C6 glioma and glial cells. Cell Calcium 2009, 46, 176–187. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.M.; Gilula, N.B. The gap junction communication channel. Cell 1996, 84, 381–388. [Google Scholar] [CrossRef]
- Homkajorn, B.; Sims, N.R.; Muyderman, H. Connexin 43 regulates astrocytic migration and proliferation in response to injury. Neurosci. Lett. 2010, 486, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Clasadonte, J.; Scemes, E.; Wang, Z.; Boison, D.; Haydon, P.G. Connexin 43-Mediated Astroglial Metabolic Networks Contribute to the Regulation of the Sleep-Wake Cycle. Neuron 2017, 95, 1365–1380. [Google Scholar] [CrossRef] [PubMed]
- Retamal, M.A.; Froger, N.; Palacios-Prado, N.; Ezan, P.; Sáez, P.J.; Sáez, J.C.; Giaume, C. Cx43 hemichannels and gap junction channels in astrocytes are regulated oppositely by proinflammatory cytokines released from activated microglia. J. Neurosci. 2007, 27, 13781–13792. [Google Scholar] [CrossRef] [PubMed]
- Almad, A.A.; Doreswamy, A.; Gross, S.K.; Richard, J.P.; Huo, Y.; Haughey, N.; Maragakis, N.J. Connexin 43 in astrocytes contributes to motor neuron toxicity in amyotrophic lateral sclerosis. Glia 2016, 64, 1154–1169. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Wu, Y.; Xie, F.; Zhong, Y.; Wang, Y.; Xu, M.; Feng, J.; Charish, J.; Monnier, P.P.; Qin, X. RGMa mediates reactive astrogliosis and glial scar formation through TGFβ1/Smad2/3 signaling after stroke. Cell Death Differ. 2018, 1. [Google Scholar] [CrossRef] [PubMed]
- Yi, C.; Koulakoff, A.; Giaume, C. Astroglial connexins as a therapeutic target for Alzheimer’s disease. Curr. Pharm. Des. 2017, 23, 4958–4968. [Google Scholar] [CrossRef] [PubMed]
- Hamadi, A.; Giannone, G.; Takeda, K.; Rondé, P. Glutamate involvement in calcium-dependent migration of astrocytoma cells. Cancer Cell Int. 2014, 14, 42. [Google Scholar] [CrossRef] [PubMed]
- Cebecauer, M.; Spitaler, M.; Sergé, A.; Magee, A.I. Signalling complexes and clusters: Functional advantages and methodological hurdles. J. Cell Sci. 2010, 123, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Kong, M.; Muñoz, N.; Valdivia, A.; Alvarez, A.; Herrera-Molina, R.; Cárdenas, A.; Schneider, P.; Burridge, K.; Quest, A.F.; Leyton, L. Thy-1-mediated cell-cell contact induces astrocyte migration through the engagement of αvβ3 integrin and syndecan-4. Biochim. Biophys. Acta 2013, 1833, 1409–1420. [Google Scholar] [CrossRef] [PubMed]
- Avalos, A.M.; Valdivia, A.D.; Muñoz, N.; Herrera-Molina, R.; Tapia, J.C.; Lavandero, S.; Chiong, M.; Burridge, K.; Schneider, P.; Quest, A.F.; et al. Neuronal Thy-1 induces astrocyte adhesion by engaging syndecan-4 in a cooperative interaction with αvβ3 integrin that activates PKCα and RhoA. J. Cell Sci. 2009, 122, 3462–3471. [Google Scholar] [CrossRef] [PubMed]
- Derangeon, M.; Bourmeyster, N.; Plaisance, I.; Pinet-Charvet, C.; Chen, Q.; Duthe, F.; Popoff, M.R.; Sarrouilhe, D.; Hervé, J.C. RhoA GTPase and F-actin dynamically regulate the permeability of Cx43-made channels in rat cardiac myocytes. J. Biol. Chem. 2008, 283, 30754–30765. [Google Scholar] [CrossRef] [PubMed]
- Lauf, U.; Giepmans, B.N.; Lopez, P.; Braconnot, S.; Chen, S.C.; Falk, M.M. Dynamic trafficking and delivery of connexons to the plasma membrane and accretion to gap junctions in living cells. Proc. Natl. Acad. Sci. USA 2002, 99, 10446–10451. [Google Scholar] [CrossRef] [PubMed]
- Ghézali, G.; Calvo, C.F.; Pillet, L.E.; Llense, F.; Ezan, P.; Pannasch, U.; Bemelmans, A.P.; Manneville, S.E.; Rouach, N. Connexin 30 controls astroglial polarization during postnatal brain development. Development 2018, 145, dev155275. [Google Scholar] [CrossRef] [PubMed]
- Dhein, S.; Schreiber, A.; Steinbach, S.; Apel, D.; Salameh, A.; Schlegel, F.; Kostelka, M.; Dohmen, P.M.; Mohr, F.W. Mechanical control of cell biology. Effects of cyclic mechanical stretch on cardiomyocyte cellular organization. Prog. Biophys. Mol. Biol. 2014, 115, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, A.M.; Fernandez, S.; Carrero, P.; Garcia-Garcia, M.; Torres-Aleman, I. Calcineurin in reactive astrocytes plays a key role in the interplay between proinflammatory and anti-inflammatory signals. J. Neurosci. 2007, 27, 8745–8756. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Ortiz, J.M.; Serrano-Pérez, M.C.; Pastor, M.D.; Martín, E.D.; Calvo, S.; Rincón, M.; Tranque, P. Mechanical lesion activates newly identified NFATc1 in primary astrocytes: Implication of ATP and purinergic receptors. Eur. J. Neurosci. 2008, 27, 2453–2465. [Google Scholar] [CrossRef] [PubMed]
- Kar, P.; Samanta, K.; Kramer, H.; Morris, O.; Bakowski, D.; Parekh, A.B. Dynamic assembly of a membrane signaling complex enables selective activation of NFAT by Orai1. Curr. Biol. 2014, 24, 1361–1368. [Google Scholar] [CrossRef] [PubMed]
- Burgos-Bravo, F.; Figueroa, N.L.; Casanova-Morales, N.; Quest, A.F.G.; Wilson, C.A.M.; Leyton, L. Single-molecule measurements of the effect of force on Thy-1/αvβ3 integrin interaction using non-purified proteins. Mol. Biol. Cell 2017. [Google Scholar] [CrossRef]
- Taubenberger, A.; Cisneros, D.A.; Friedrichs, J.; Puech, P.H.; Muller, D.J.; Franz, C.M. Revealing early steps of α2β1 integrin-mediated adhesion to collagen type I by using single-cell force spectroscopy. Mol. Biol. Cell 2007, 18, 1634–1644. [Google Scholar] [CrossRef] [PubMed]
- Cluzel, C.; Saltel, F.; Lussi, J.; Paulhe, F.; Imhof, B.A.; Wehrle-Haller, B. The mechanisms and dynamics of αvβ3 integrin clustering in living cells. J. Cell Biol. 2005, 171, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Foletti, A.; Alghisi, G.C.; Ponsonnet, L.; Rüegg, C. Isolated integrin β3 subunit cytoplasmic domains require membrane anchorage and the NPXY motif to recruit to adhesion complexes but do not discriminate between β1- and β3-positive complexes. Thromb. Haemost. 2005, 94, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Hermosilla, T.; Munoz, D.; Herrera-Molina, R.; Valdivia, A.; Munoz, N.; Nham, S.U.; Schneider, P.; Burridge, K.; Quest, A.F.; Leyton, L. Direct Thy-1/αvβ3 integrin interaction mediates neuron to astrocyte communication. Biochim. Biophys. Acta 2008, 1783, 1111–1120. [Google Scholar] [CrossRef] [PubMed]
- Leyton, L.; Schneider, P.; Labra, C.V.; Rüegg, C.; Hetz, C.A.; Quest, A.F.; Bron, C. Thy-1 binds to integrin β3 on astrocytes and triggers formation of focal contact sites. Curr. Biol. 2001, 11, 1028–1038. [Google Scholar] [CrossRef]
- Schneider, P. Production of recombinant TRAIL and TRAIL receptor: Fc chimeric proteins. Methods Enzymol. 2000, 322, 325–345. [Google Scholar] [PubMed]
- Mendoza, P.; Ortiz, R.; Díaz, J.; Quest, A.F.; Leyton, L.; Stupack, D.; Torres, V.A. Rab5 activation promotes focal adhesion disassembly, migration and invasiveness in tumor cells. J. Cell Sci. 2013, 126, 3835–3847. [Google Scholar] [CrossRef] [PubMed]
- Urra, H.; Torres, V.A.; Ortiz, R.J.; Lobos, L.; Díaz, M.I.; Díaz, N.; Härtel, S.; Leyton, L.; Quest, A.F. Caveolin-1-enhanced motility and focal adhesion turnover require tyrosine-14 but not accumulation to the rear in metastatic cancer cells. PLoS ONE 2012, 7, e33085. [Google Scholar] [CrossRef] [PubMed]
- Díaz, J.; Mendoza, P.; Ortiz, R.; Díaz, N.; Leyton, L.; Stupack, D.; Quest, A.F.; Torres, V.A. Rab5 is required in metastatic cancer cells for Caveolin-1-enhanced Rac1 activation, migration and invasion. J. Cell Sci. 2014, 127, 2401–2406. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lagos-Cabré, R.; Brenet, M.; Díaz, J.; Pérez, R.D.; Pérez, L.A.; Herrera-Molina, R.; Quest, A.F.G.; Leyton, L. Intracellular Ca2+ Increases and Connexin 43 Hemichannel Opening Are Necessary but Not Sufficient for Thy-1-Induced Astrocyte Migration. Int. J. Mol. Sci. 2018, 19, 2179. https://doi.org/10.3390/ijms19082179
Lagos-Cabré R, Brenet M, Díaz J, Pérez RD, Pérez LA, Herrera-Molina R, Quest AFG, Leyton L. Intracellular Ca2+ Increases and Connexin 43 Hemichannel Opening Are Necessary but Not Sufficient for Thy-1-Induced Astrocyte Migration. International Journal of Molecular Sciences. 2018; 19(8):2179. https://doi.org/10.3390/ijms19082179
Chicago/Turabian StyleLagos-Cabré, Raúl, Marianne Brenet, Jorge Díaz, Ramón D. Pérez, Leonardo A. Pérez, Rodrigo Herrera-Molina, Andrew F. G. Quest, and Lisette Leyton. 2018. "Intracellular Ca2+ Increases and Connexin 43 Hemichannel Opening Are Necessary but Not Sufficient for Thy-1-Induced Astrocyte Migration" International Journal of Molecular Sciences 19, no. 8: 2179. https://doi.org/10.3390/ijms19082179
APA StyleLagos-Cabré, R., Brenet, M., Díaz, J., Pérez, R. D., Pérez, L. A., Herrera-Molina, R., Quest, A. F. G., & Leyton, L. (2018). Intracellular Ca2+ Increases and Connexin 43 Hemichannel Opening Are Necessary but Not Sufficient for Thy-1-Induced Astrocyte Migration. International Journal of Molecular Sciences, 19(8), 2179. https://doi.org/10.3390/ijms19082179





