Graphene Quantum Dots and Enzyme-Coupled Biosensor for Highly Sensitive Determination of Hydrogen Peroxide and Glucose
Abstract
1. Introduction
2. Results and Discussion
2.1. GQDs Characterization
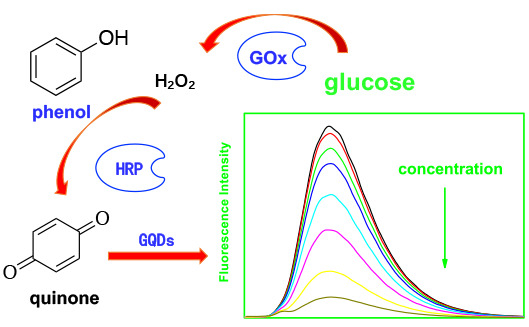
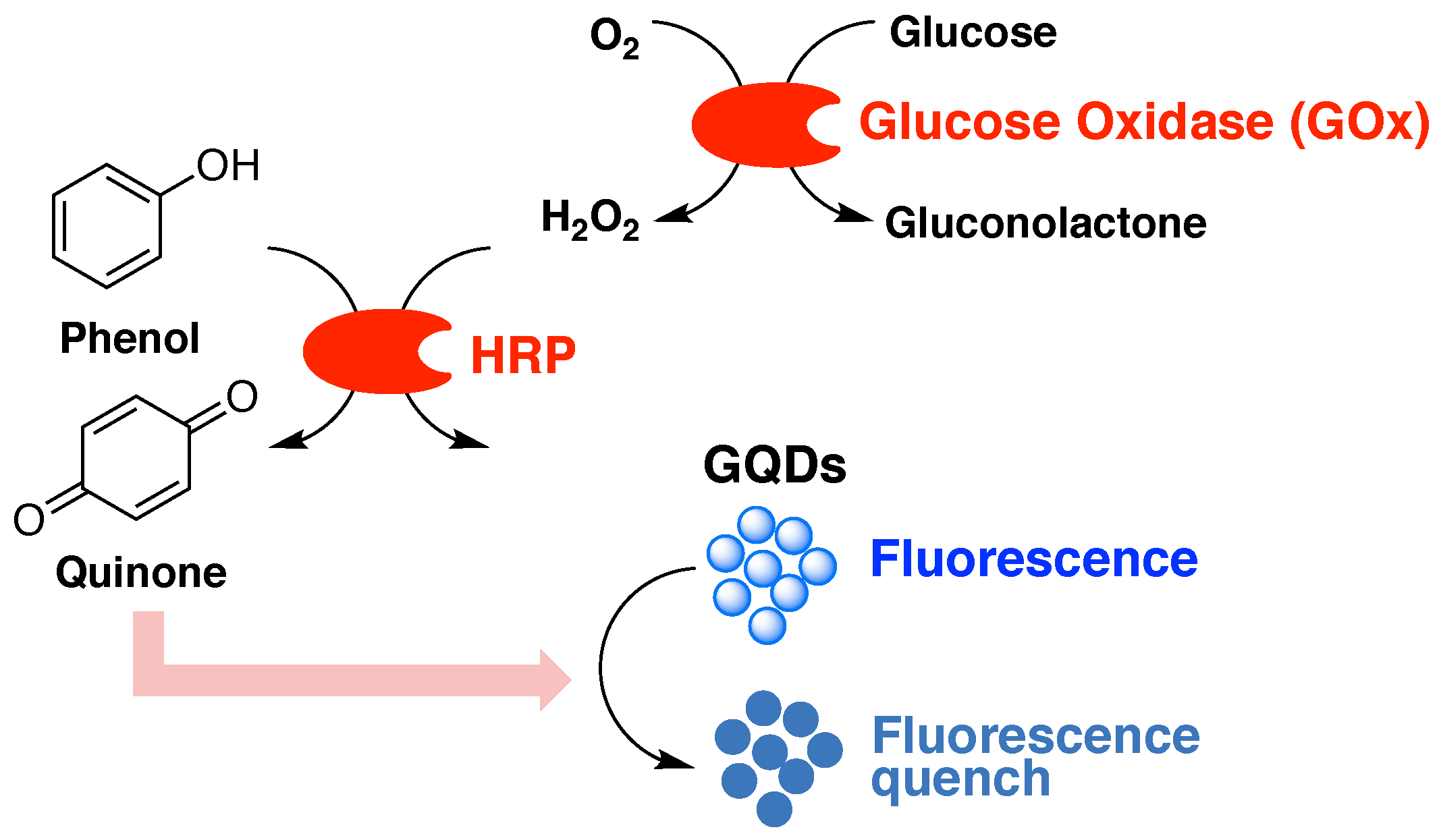
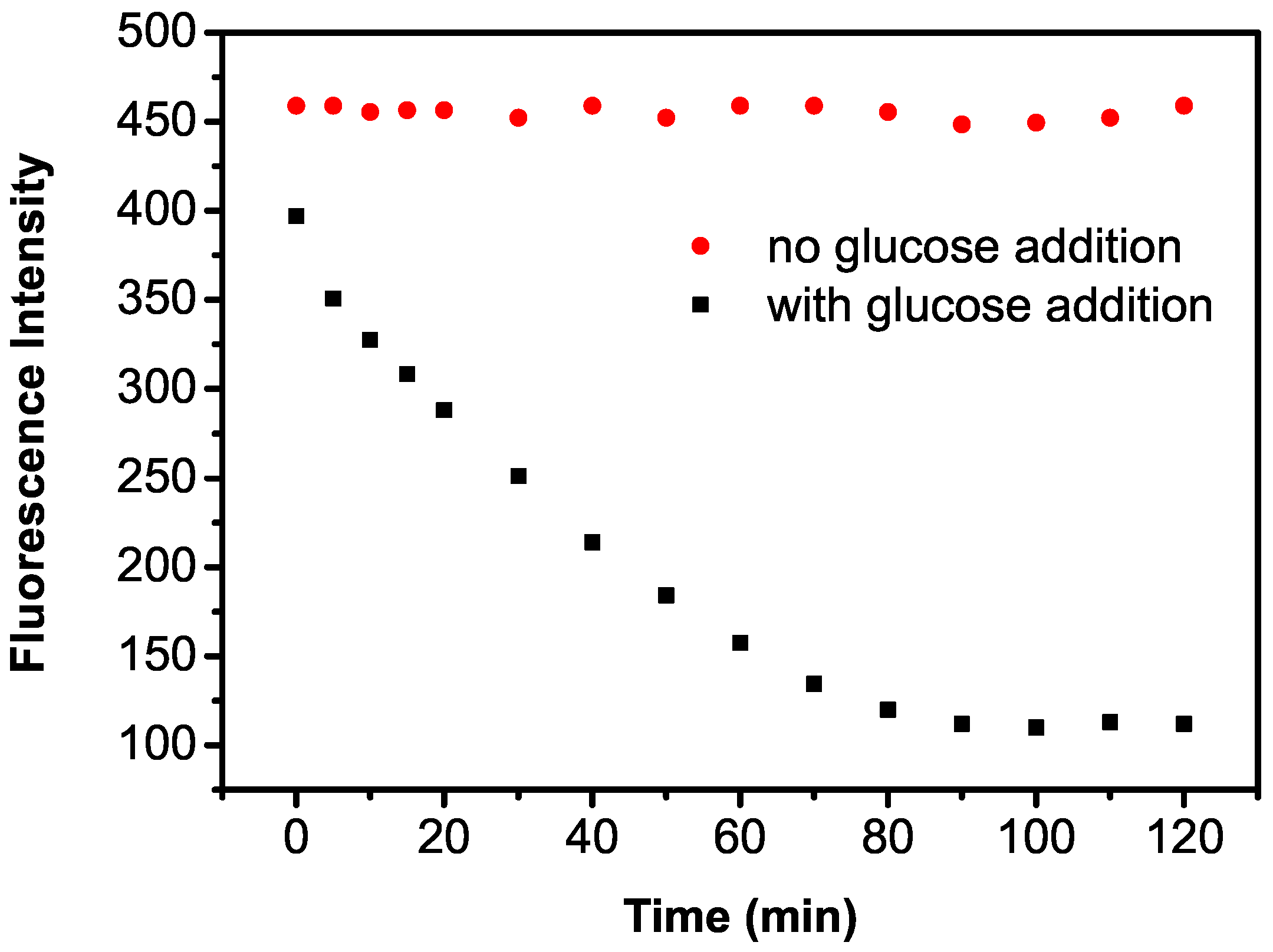
2.2. Detection Principle of the Enzyme-Coupled Biosensor
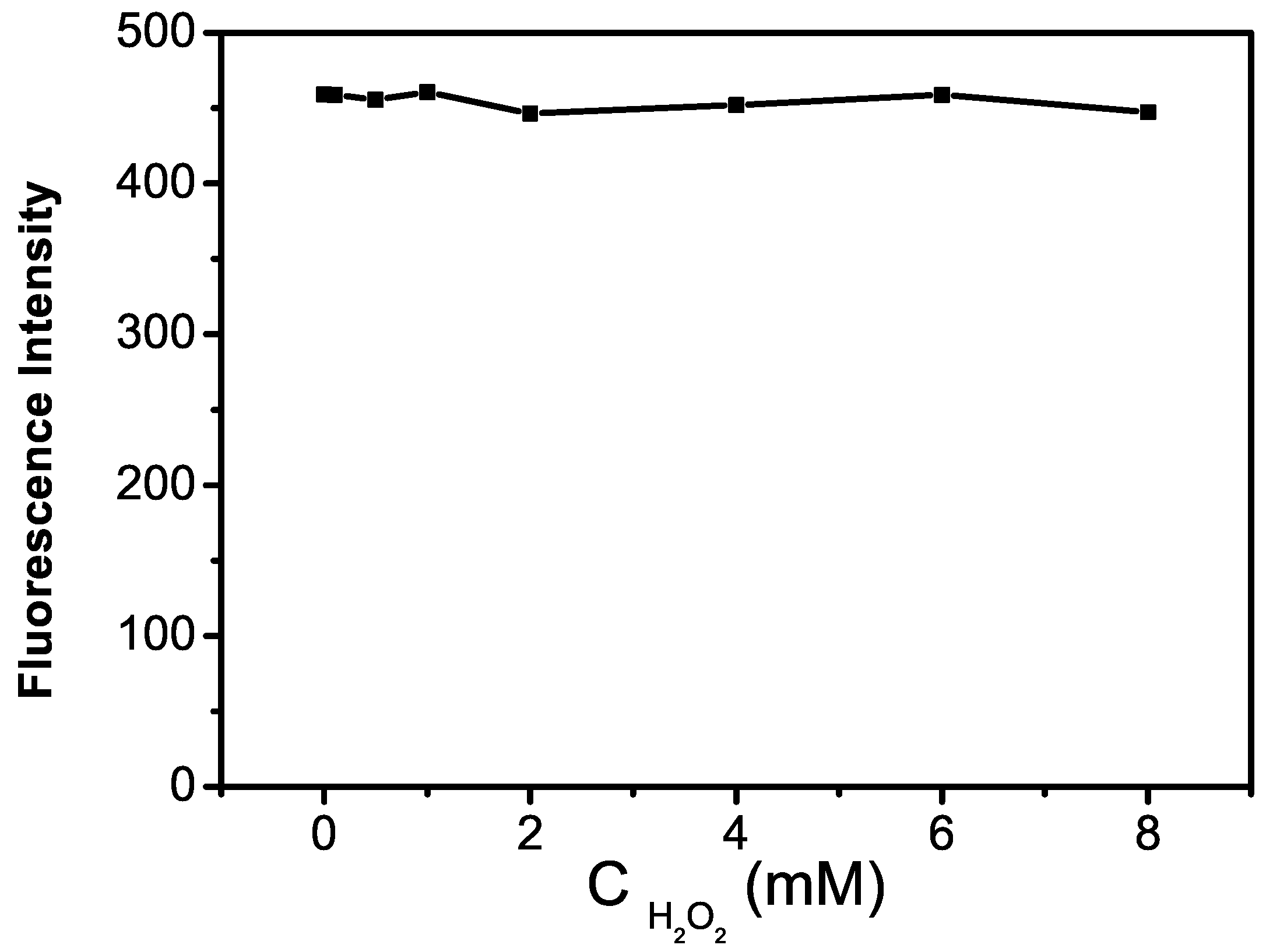
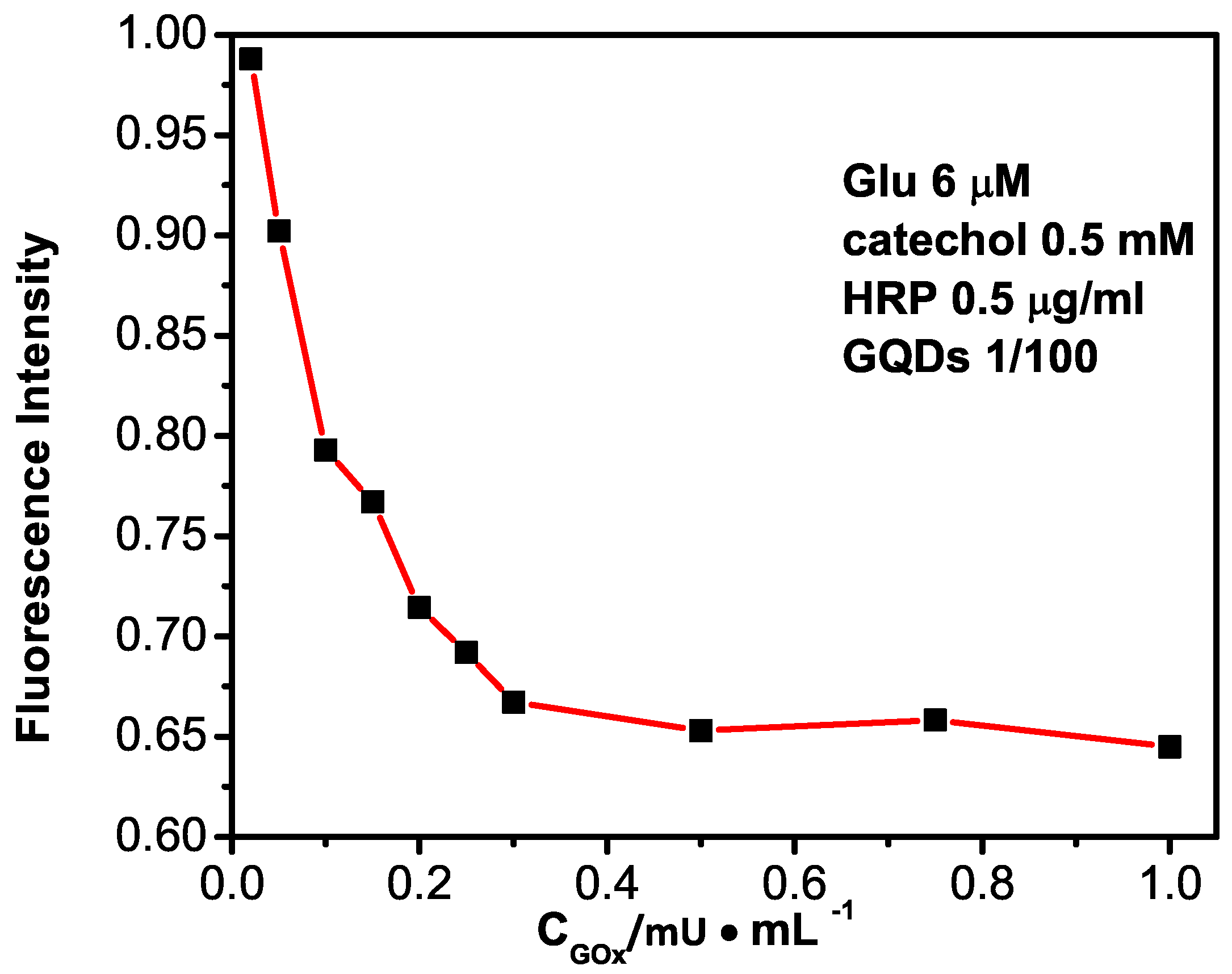
2.3. Optimization of the GQDs–GOx–HRP–Phenol Sensing System
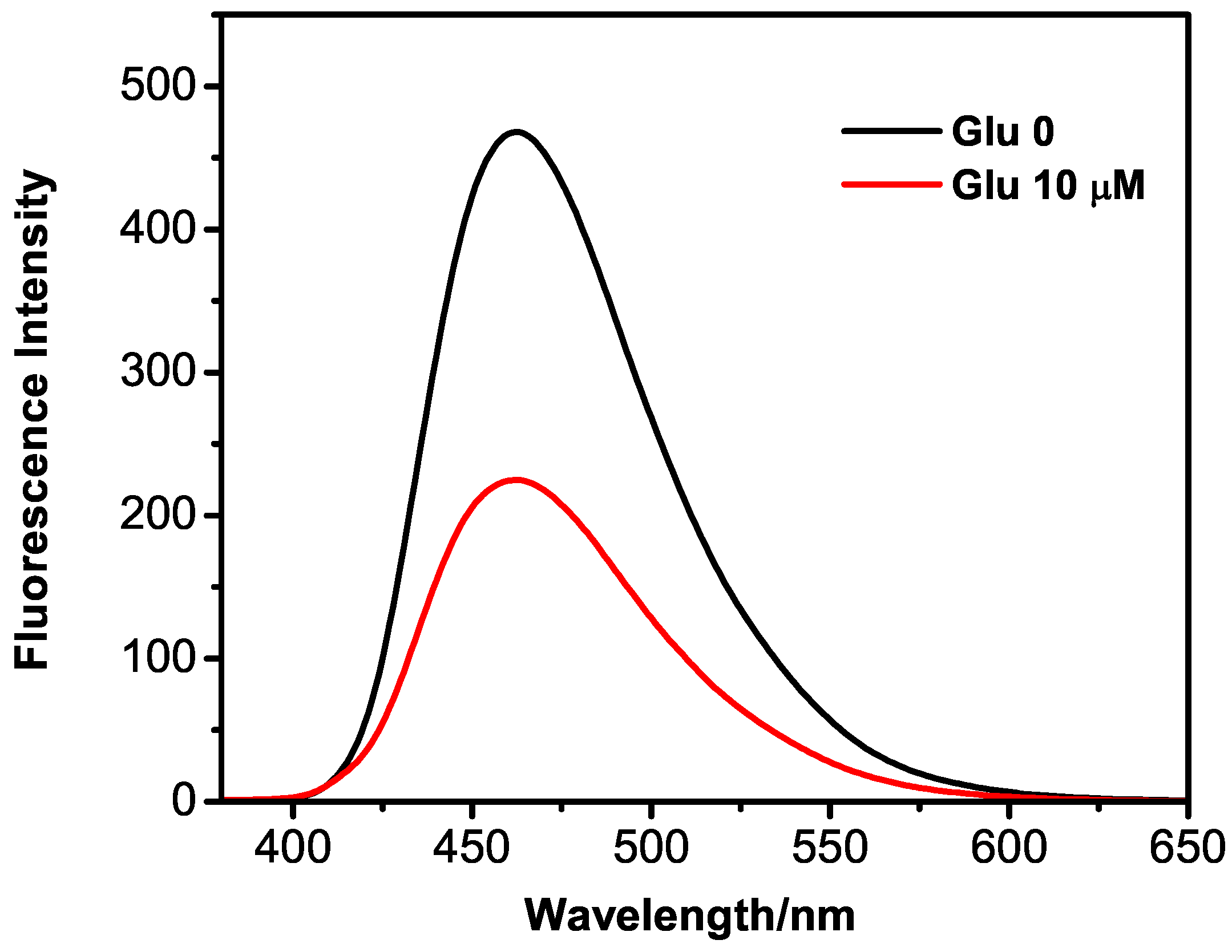
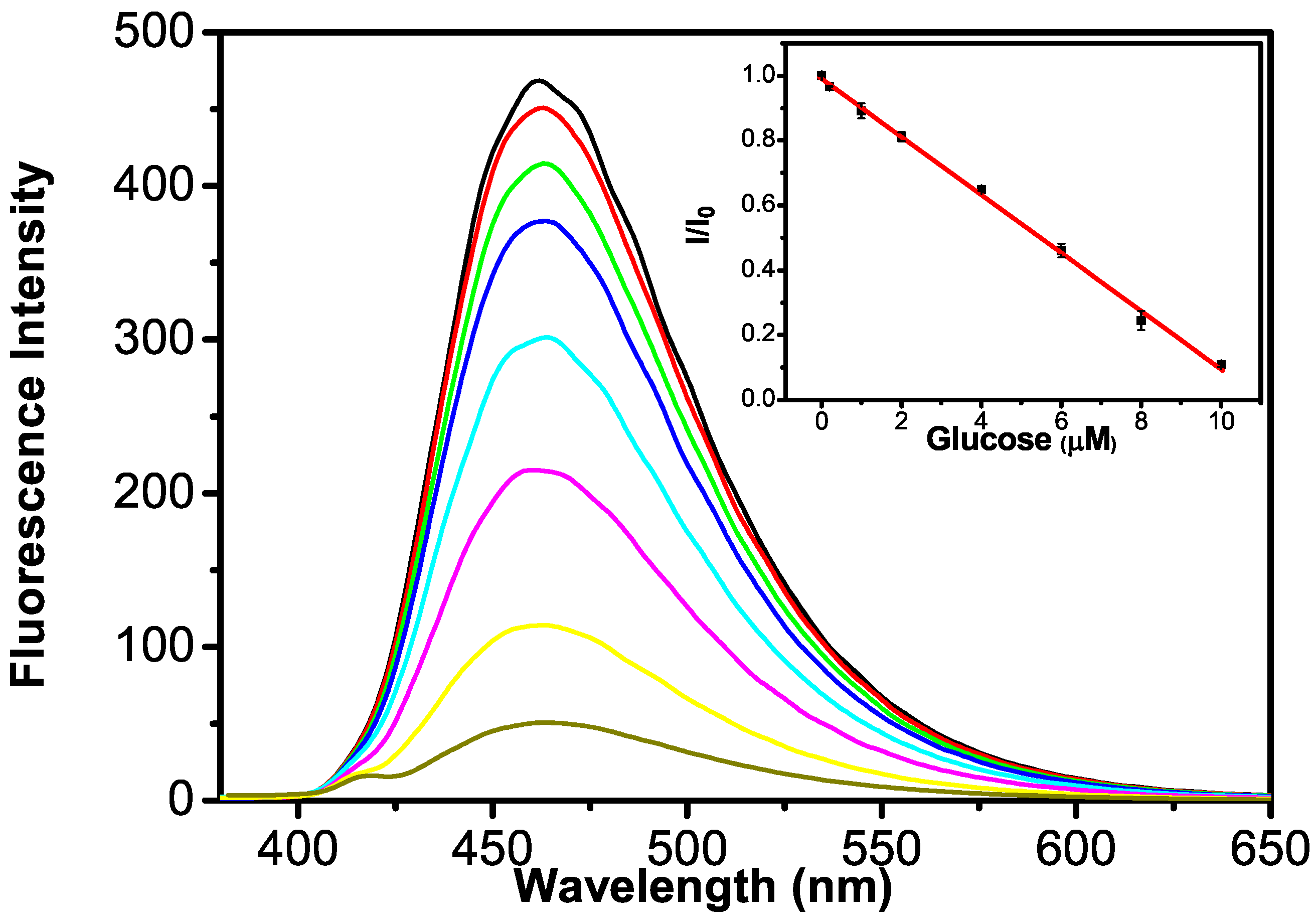
2.4. Detection of Glucose via the GQDs–GOx–HRP–Phenol Sensing System
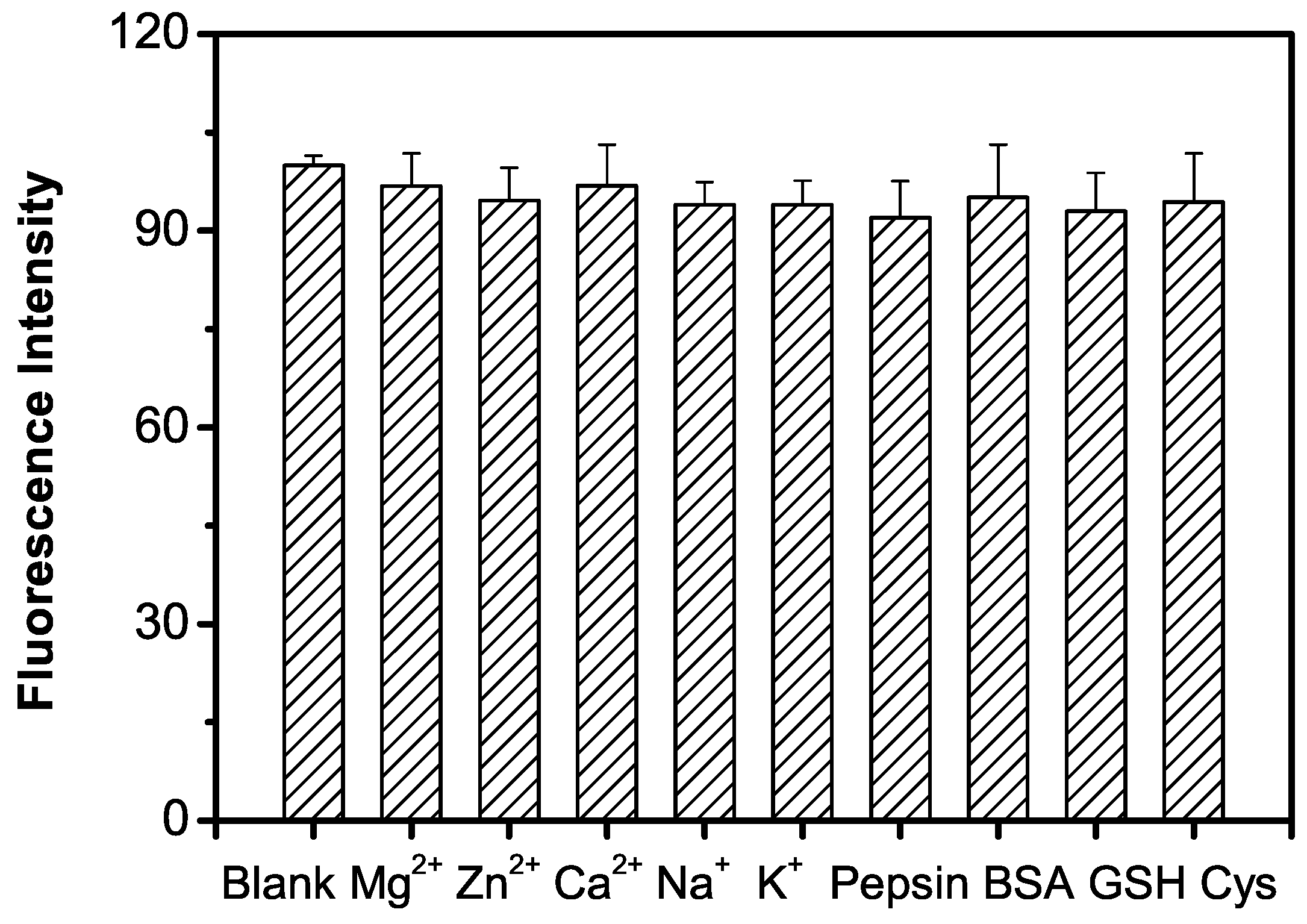
2.5. Selectivity of the GQDs–GOx–HRP–Phenol Sensing System for Detecting Glucose
2.6. Application of Assaying Glucose Concentration in Human Serum Samples
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Instrumentation
3.3. Synthesis of GQDs
3.4. Optimization of the Sensing System
3.5. Assay of Glucose
3.6. Interference Study
3.7. Fluorescence Detection of Glucose in Human Blood Serum
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| GQDs | Graphene quantum dots |
| HRP | horseradish peroxidase |
| GOx | glucose oxidase |
| HO | hydrogen peroxide |
References
- Matz, K.; Keresztes, K.; Tatschl, C.; Nowotny, M.; Dachenhausen, A.; Brainin, M.; Tuomilehto, J. Disorders of glucose metabolism in acute stroke patients: An underrecognized problem. Diabetes Care 2006, 29, 792–797. [Google Scholar] [CrossRef] [PubMed]
- Barzilay, J.I.; Spiekerman, C.F.; Kuller, L.H.; Burke, G.L.; Bittner, V.; Gottdiener, J.S.; Brancati, F.L.; Orchard, T.J.; O’Leary, D.H.; Savage, P.J. Prevalence of clinical and isolated subclinical cardiovascular disease in older adults with glucose disorders-The Cardiovascular Health Study. Diabetes Care 2001, 24, 1233–1239. [Google Scholar] [CrossRef] [PubMed]
- Pickup, J.C.; Zhi, Z.L.; Khan, F.; Saxl, T.; Birch, D.J.S. Nanomedicine and its potential in diabetes research and practice. Diabetes Metab. Res. Rev. 2008, 24, 604–610. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, H.H.; Kreft, M.; Jensen, J.; Zorec, R. Insulin Induces an Increase in Cytosolic Glucose Levels in 3T3-L1 Cells with Inhibited Glycogen Synthase Activation. Int. J. Mol. Sci. 2014, 15, 17827–17837. [Google Scholar] [CrossRef] [PubMed]
- Arvand, M.; Hemmati, S. Magnetic nanoparticles embedded with graphene quantum dots and multiwalled carbon nanotubes as a sensing platform for electrochemical detection of progesterone. Int. J. Mol. Sci. 2017, 238, 346–356. [Google Scholar] [CrossRef]
- Shiddiky, M.J.A.; Rauf, S.; Kithva, P.H.; Trau, M. Graphene/quantum dot bionanoconjugates as signal amplifiers in stripping voltammetric detection of EpCAM biomarkers. Biosens. Bioelectron. 2012, 35, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Feng, J.; Zhou, X.; Ren, C.; Li, H.; Chen, X. Colorimetric Detection of Urine Glucose Based ZnFe2O4 Magnetic Nanoparticles. Anal. Chem. 2012, 84, 5753–5758. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.D.; Long, Y.F.; Wang, J.X. Glucose oxidase-functionalized fluorescent gold nanoclusters as probes for glucose. Anal. Chim. Acta 2013, 772, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Shang, L.; Guo, S.; Fang, Y.; Wen, D.; Wang, L.; Yin, J.; Dong, S. Biomolecule-stabilized Au nanoclusters as a fluorescence probe for sensitive detection of glucose. Biosens. Bioelectron. 2011, 26, 1965–1969. [Google Scholar] [CrossRef] [PubMed]
- Sricharoen, P.; Limchoowong, N.; Areerob, Y.; Nuengmatcha, P.; Techawongstien, S.; Chanthai, S. Fe3O4/hydroxyapatite/graphene quantum dots as a novel nano-sorbent for preconcentration of copper residue in Thai food ingredients: Optimization of ultrasound-assisted magnetic solid phase extraction. Ultrason. Sonochem. 2017, 37, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.L.H.; Zhang, L.L.; Gao, H.L.; Yang, W.Y.; Wang, S.; Xing, L.L.; Xue, X.Y. Self-Powered Implantable Skin-Like Glucometer for Real-Time Detection of Blood Glucose Level In Vivo. Nano-Micro Lett. 2018, 10, 32. [Google Scholar] [CrossRef]
- Newman, J.D.; Turner, A.P.F. Home blood glucose biosensors: A commercial perspective. Biosens. Bioelectron. 2005, 20, 2435–2453. [Google Scholar] [CrossRef] [PubMed]
- Steiner, M.S.; Duerkop, A.; Wolfbeis, O.S. Optical methods for sensing glucose. Chem. Soc. Rev. 2011, 40, 4805–4839. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.Y.; Lan, T.; Zhang, B.C.; Ren, J.C. Gold nanoparticle-enzyme conjugates based FRET for highly sensitive determination of hydrogen peroxide, glucose and uric acid using tyramide reaction. Analyst 2012, 137, 3659–3666. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, C.B.; Peairs, M.J.; Venton, B.J. Review: Carbon nanotube based electrochemical sensors for biomolecules. Anal. Chim. Acta 2010, 662, 105–127. [Google Scholar] [CrossRef] [PubMed]
- Besteman, K.; Lee, J.O.; Wiertz, F.G.M.; Heering, H.A.; Dekker, C. Enzyme-coated carbon nanotubes as single-molecule biosensors. Nano Lett. 2003, 3, 727–730. [Google Scholar] [CrossRef]
- Wang, Y.J.; Xin, B.J.; Duan, X.R.; Xing, G.W.; Wang, S. Assembly of Anionic Conjugated Polymer with 6-O-Modified PNP-beta-Galactoside for Fluorescence Logic-signal-based Multiplex Detections of Enzymes. Macromol. Rapid. Commun. 2010, 31, 1473–1478. [Google Scholar] [CrossRef] [PubMed]
- Azmi, N.E.; Ramli, N.I.; Abdullah, J.; Hamid, M.A.A.; Sidek, H.; Abd Rahman, S.; Ariffin, N.; Yusof, N.A. A simple and sensitive fluorescence based biosensor for the determination of uric acid using H2O2-sensitive quantum dots/dual enzymes. Biosens. Bioelectron. 2015, 67, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Uusitalo, L.M.; Hempel, N. Recent Advances in Intracellular and In Vivo ROS Sensing: Focus on Nanoparticle and Nanotube Applications. Int. J. Mol. Sci. 2012, 13, 10660–10679. [Google Scholar] [CrossRef] [PubMed]
- McQuade, D.T.; Pullen, A.E.; Swager, T.M. Conjugated polymer-based chemical sensors. Chem. Rev. 2000, 100, 2537–2574. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, J.G.; Jaffe, J.S.; Schulman, E.S.; Raible, D.G. A highly sensitive fluorescent micro-assay of H2O2 release from activated human leukocytes using a dihydroxyphenoxazine derivative. J. Immunol. Methods 1997, 202, 133–141. [Google Scholar] [CrossRef]
- Geim, A.K. Graphene: Status and Prospects. Science 2009, 324, 1530–1534. [Google Scholar] [CrossRef] [PubMed]
- Forsyth, R.; Devadoss, A.; Guy, O.J. Graphene Field Effect Transistors for Biomedical Applications: Current Status and Future Prospects. Diagnostics 2017, 7, 45. [Google Scholar] [CrossRef] [PubMed]
- Najafabadi, A.T. Emerging applications of graphene and its derivatives in carbon capture and conversion: Current status and future prospects. Renew. Sustain. Energy Rev. 2015, 41, 1515–1545. [Google Scholar] [CrossRef]
- Bacon, M.; Bradley, S.J.; Nann, T. Graphene Quantum Dots. Part. Part. Syst. Charact. 2014, 31, 415–428. [Google Scholar] [CrossRef]
- Dai, H.J. Carbon nanotubes: Opportunities and challenges. Surf. Sci. 2017, 500, 218–241. [Google Scholar] [CrossRef]
- Ponomarenko, L.A.; Schedin, F.; Katsnelson, M.I.; Yang, R.; Hill, E.; Novoselov, K.S.; Geim, A.K. Chaotic dirac billiard in graphene quantum dots. Science 2008, 320, 356–358. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, Y.L.; Xu, T.; Liao, H.B.; Yao, C.J.; Liu, Y.; Li, Z.; Chen, Z.W.; Pan, D.Y.; Sun, L.T.; et al. Gram-scale synthesis of single-crystalline graphene quantum dots with superior optical properties. Nat. Commun. 2014, 5, 5357. [Google Scholar] [CrossRef] [PubMed]
- Samareh, J.A.; Siochi, E.J. Systems analysis of carbon nanotubes: Opportunities and challenges for space applications. Nanotechnology 2017, 28, 372001. [Google Scholar] [CrossRef] [PubMed]
- Chong, Y.; Ma, Y.F.; Shen, H.; Tu, X.L.; Zhou, X.; Xu, J.Y.; Dai, J.W.; Fan, S.J.; Zhang, Z.J. The in vitro and in vivo toxicity of graphene quantum dots. Biomaterials 2014, 35, 5041–5048. [Google Scholar] [CrossRef] [PubMed]
- Qian, Z.S.; Shan, X.Y.; Chai, L.J.; Chen, J.R.; Peng, H. A fluorescent nanosensor based on graphene quantum dots-aptamer probe and graphene oxide platform for detection of lead (II) ion. Biosens. Bioelectron. 2015, 68, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Vasilescu, I.; Eremia, S.A.V.; Kusko, M.; Radoi, A.; Vasile, E.; Radu, G.L. Molybdenum disulphide and graphene quantum dots as electrode modifiers for laccase biosensor. Biosens. Bioelectron. 2016, 75, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Shang, W.H.; Zhang, X.Y.; Zhang, M.; Fan, Z.T.; Sun, Y.T.; Han, M.; Fan, L.Z. The uptake mechanism and biocompatibility of graphene quantum dots with human neural stem cells. Nanoscale 2014, 6, 5799–5806. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.T.; Zhou, S.X.; Garcia, C.; Fan, L.Z.; Zhou, J.B. pH-Responsive fluorescent graphene quantum dots for fluorescence-guided cancer surgery and diagnosis. Nanoscale 2017, 9, 4928–4933. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y.X.; Zhang, M.; Fu, Q.Q.; Liu, R.; Shi, G.Y. Highly sensitive and selective fluorescent detection of cerebral lead(II) based on graphene quantum dot conjugates. Chem. Commun. 2013, 49, 10599–10601. [Google Scholar] [CrossRef] [PubMed]
- Na, W.D.; Qu, Z.Y.; Chen, X.Q.; Su, X.G. A turn-on fluorescent probe for sensitive detection of sulfide anions and ascorbic acid by using sulfanilic acid and glutathione functionalized graphene quantum dots. Sens. Actuators B 2018, 256, 48–54. [Google Scholar] [CrossRef]
- Wang, B.J.; Zhuo, S.J.; Chen, L.Y.; Zhang, Y.J. Fluorescent graphene quantum dot nanoprobes for the sensitive and selective detection of mercury ions. Spectrochim. Acta Part A 2014, 131, 384–387. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Qin, A.M.; Chen, S.P.; Liao, L.; Qin, J.K.; Zhang, K.Y. Manganese(II) enhanced fluorescent nitrogen-doped graphene quantum dots: A facile and efficient synthesis and their applications for bioimaging and detection of Hg2+ ions. RSC Adv. 2018, 8, 5902–5911. [Google Scholar] [CrossRef]
- Zhao, X.E.; Lei, C.H.; Gao, Y.; Gao, H.; Zhu, S.Y.; Yang, X.; You, J.M.; Wang, H. A ratiometric fluorescent nanosensor for the detection of silver ions using graphene quantum dots. Sens. Actuators B 2017, 253, 239–246. [Google Scholar] [CrossRef]
- Liu, R.; Yang, R.; Qu, C.J.; Mao, H.C.; Hu, Y.; Li, J.J.; Qu, L.B. Synthesis of glycine-functionalized graphene quantum dots as highly sensitive and selective fluorescent sensor of ascorbic acid in human serum. Sens. Actuators B 2017, 241, 644–651. [Google Scholar] [CrossRef]
- Li, Y.X.; Huang, H.; Ma, Y.H.; Tong, J. Highly sensitive fluorescent detection of dihydroxybenzene based on graphene quantum dots. Sens. Actuators B 2014, 205, 227–233. [Google Scholar] [CrossRef]
- He, Y.; Wang, X.; Sun, J.; Jiao, S.; Chen, H.; Gao, F.; Wang, L. Fluorescent blood glucose monitor by hemin-functionalized graphene quantum dots based sensing system. Anal. Chim. Acta 2014, 810, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Wang, L.M.; Huang, C.S.; Su, W.; Xiao, Q. Label-free and ratiometric fluorescent nanosensor based on amino-functionalized graphene quantum dots coupling catalytic G-quadruplex/hemin DNAzyme for ultrasensitive recognition of human telomere DNA. Sens. Actuators B 2017, 245, 648–655. [Google Scholar] [CrossRef]
- Minisci, F.; Citterio, A.; Vismara, E.; Fontana, F.; Debernardinis, S.; Correale, M. Facile and Convenient Syntheses of Quinones from Phenols. J. Org. Chem. 1989, 54, 728–731. [Google Scholar] [CrossRef]
- Huggett, A.S.G.; Nixoh, D.A. Use of glucose oxidase, peroxidase, and o-dianisidine in determination of blood and urinary glucose. Lancet 1957, 273, 368. [Google Scholar] [CrossRef]
- Yuan, J.; Guo, W.; Wang, E. Utilizing a CdTe quantum dots-enzyme hybrid system for the determination of both phenolic compounds and hydrogen peroxide. Anal. Chem. 2008, 80, 1141–1145. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Sun, R.L.; Ni, Y.N.; Kokot, S. A novel fluorescent probe involving a graphene quantum dot-enzyme hybrid system for the analysis of hydroquinone in the presence of toxic resorcinol and catechol. Anal. Methods 2014, 6, 7420–7425. [Google Scholar] [CrossRef]
- Cai, N.; Tan, L.; Li, Y.; Xia, T.; Hu, T.; Su, X. Biosensing platform for the detection of uric acid based on graphene quantum dots and Gquadruplex/hemin DNAzyme. Anal. Chim. Acta 2017, 965, 96. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.J.; Qu, K.G.; Zhao, C.; Ren, J.S.; Qu, X.G. Graphene Oxide: Intrinsic Peroxidase Catalytic Activity and Its Application to Glucose Detection. Adv. Mater. 2010, 22, 2206–2210. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.Y.; Huang, P.C.; Wu, F.Y. Gold-platinum bimetallic nanoclusters with enhanced peroxidase-like activity and their integrated agarose hydrogel-based sensing platform for the colorimetric analysis of glucose levels in serum. Analys 2017, 142, 4106–4115. [Google Scholar] [CrossRef] [PubMed]
- Razmi, H.; Mohammad-Rezaei, R. Graphene quantum dots as a new substrate for immobilization and direct electrochemistry of glucose oxidase: Application to sensitive glucose determination. Biosens. Bioelectron. 2013, 41, 498–504. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.H.; Deng, J.H.; Zhang, Y.Y.; Li, H.T.; Yao, S.Z. Label-free Si quantum dots as photoluminescence probes for glucose detection. Chem. Commun. 2013, 49, 612–614. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.L.; Yin, B.C.; Wu, X.; Ye, B.C. Simple and Cost-Effective Glucose Detection Based on Carbon Nanodots Supported on Silver Nanoparticles. Anal. Chem. 2017, 89, 1323–1328. [Google Scholar] [CrossRef] [PubMed]
- Molaabasi, F.; Hosseinkhani, S.; Moosavi-Movahedi, A.A.; Shamsipur, M. Hydrogen peroxide sensitive hemoglobin-capped gold nanoclusters as a fluorescence enhancing sensor for the label-free detection of glucose. RSC Adv. 2015, 5, 33123–33135. [Google Scholar] [CrossRef]
- Zhang, R.Z.; He, S.J.; Zhang, C.M.; Chen, W. Three-dimensional Fe- and N-incorporated carbon structures as peroxidase mimics for fluorescence detection of hydrogen peroxide and glucose. J. Phys. Chem. B 2015, 3, 4146–4154. [Google Scholar] [CrossRef]

| Method | System | Linear Range (M) | LOD (M) | Reference |
|---|---|---|---|---|
| Colorimetry | Graphene oxide/GOx/TMB | 1–20 | 1 | [49] |
| Colorimetry | Au–PtNCs/GOx/TMB | 5–55 | 2.4 | [50] |
| Electrochemistry | Graphene quantum dots/GOx | 5–1270 | 1.73 | [51] |
| Fluorometry | Hemin–functionalized GQDs/GOx | 9–300 | 0.1 | [42] |
| Fluorometry | Si quantum dots/GOx | 5–650 | 0.68 | [52] |
| Fluorometry | C–dots/AgNPs/GOx | 2–100 | 1.39 | [53] |
| Fluorometry | Hb–AuNCs/GOx | 0.5–1000 | 1.65 | [54] |
| Fluorometry | TA/Fe–Phen–CFs | 0.5–200 | 0.19 | [55] |
| Fluorometry | Graphene quantum dots/GOx/HRP/phenol | 0.2–10 | 0.08 | This paper |
| Samples | Added Glucose (M) | Measured ( M) | Recovery (%) | RSD (%) |
|---|---|---|---|---|
| Serum 1 | 2 | 1.97 | 98.5 | 3.94 |
| Serum 2 | 5 | 5.11 | 102.0 | 2.44 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, B.; Shen, J.; Huang, Y.; Liu, Z.; Zhuang, H. Graphene Quantum Dots and Enzyme-Coupled Biosensor for Highly Sensitive Determination of Hydrogen Peroxide and Glucose. Int. J. Mol. Sci. 2018, 19, 1696. https://doi.org/10.3390/ijms19061696
Wang B, Shen J, Huang Y, Liu Z, Zhuang H. Graphene Quantum Dots and Enzyme-Coupled Biosensor for Highly Sensitive Determination of Hydrogen Peroxide and Glucose. International Journal of Molecular Sciences. 2018; 19(6):1696. https://doi.org/10.3390/ijms19061696
Chicago/Turabian StyleWang, Bingdi, Jing Shen, Yanjun Huang, Zhenning Liu, and Hong Zhuang. 2018. "Graphene Quantum Dots and Enzyme-Coupled Biosensor for Highly Sensitive Determination of Hydrogen Peroxide and Glucose" International Journal of Molecular Sciences 19, no. 6: 1696. https://doi.org/10.3390/ijms19061696
APA StyleWang, B., Shen, J., Huang, Y., Liu, Z., & Zhuang, H. (2018). Graphene Quantum Dots and Enzyme-Coupled Biosensor for Highly Sensitive Determination of Hydrogen Peroxide and Glucose. International Journal of Molecular Sciences, 19(6), 1696. https://doi.org/10.3390/ijms19061696