Dietary Supplementation with Oleum Cinnamomi Improves Intestinal Functions in Piglets
Abstract
:1. Introduction
2. Results
2.1. Growth Performance and Diarrhoea Incidence
2.2. Concentrations of Hormones, IGF-1 and PGE2 and in Plasma
2.3. Intestinal Histology
2.4. DNA Levels, RNA/DNA, and Protein/DNA in the Intestinal Mucosa
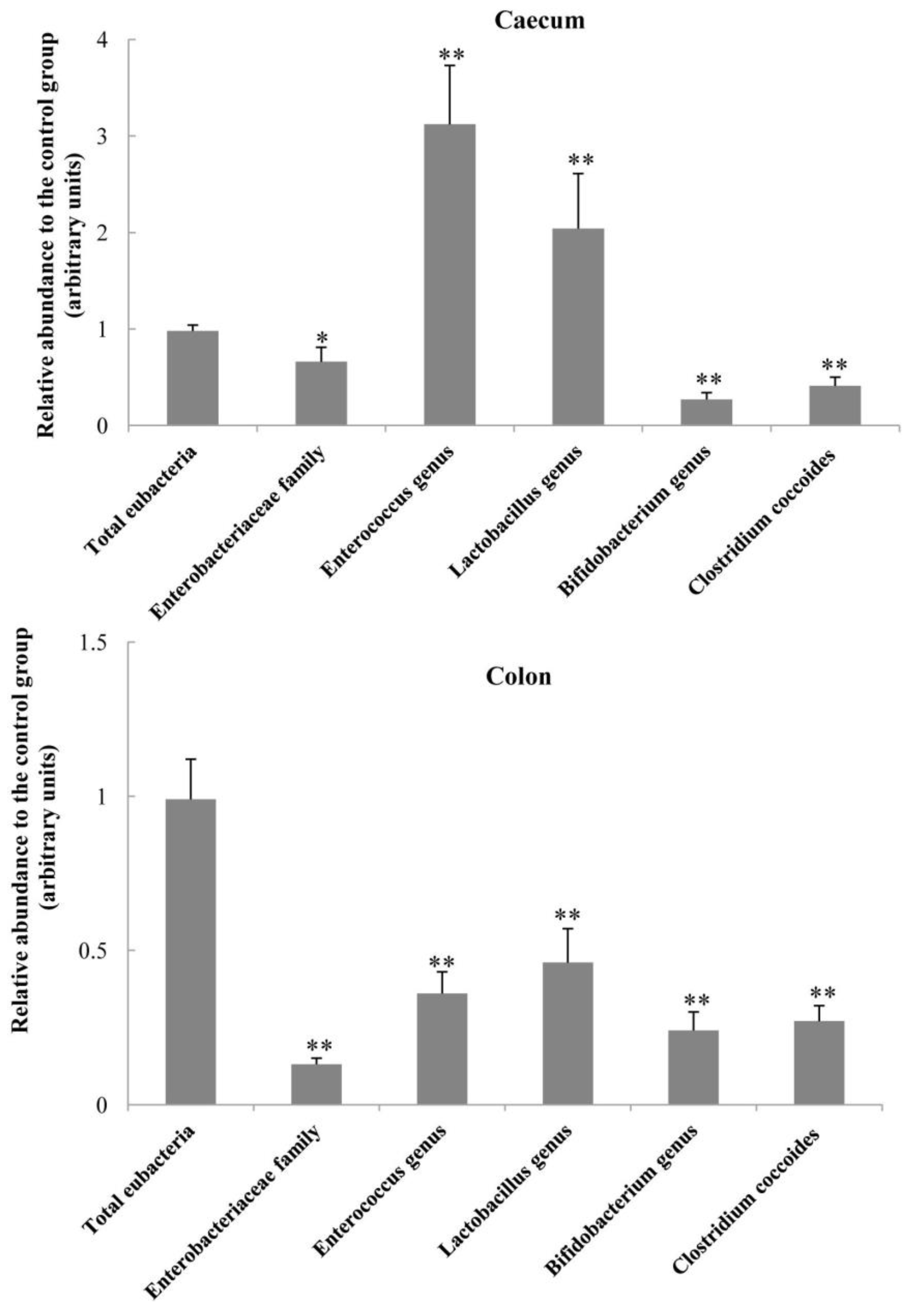
2.5. Bacterial Populations in the Caecum and Colon Digesta
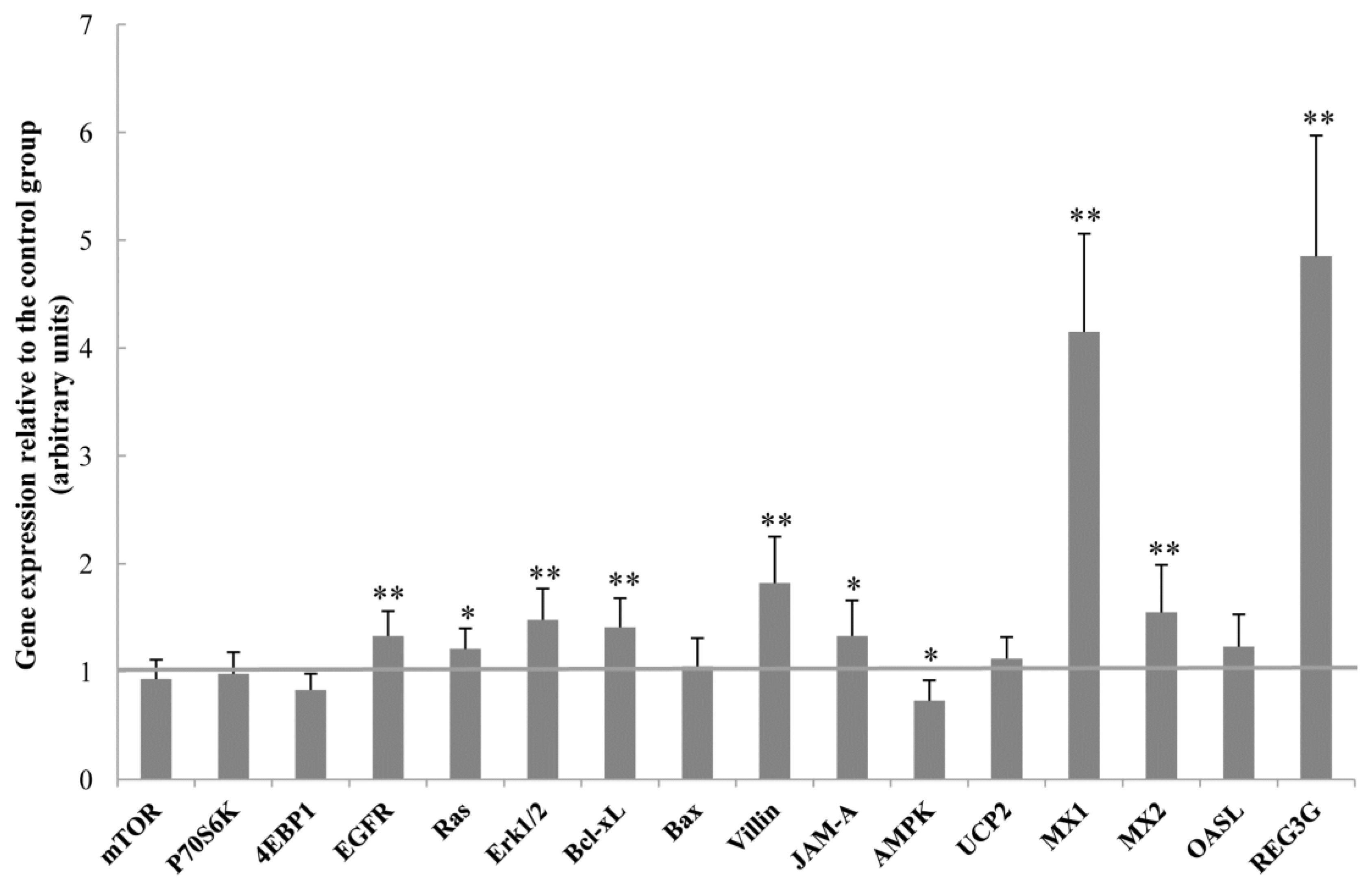
2.6. Gene Expression in the Jejunal Mucosa
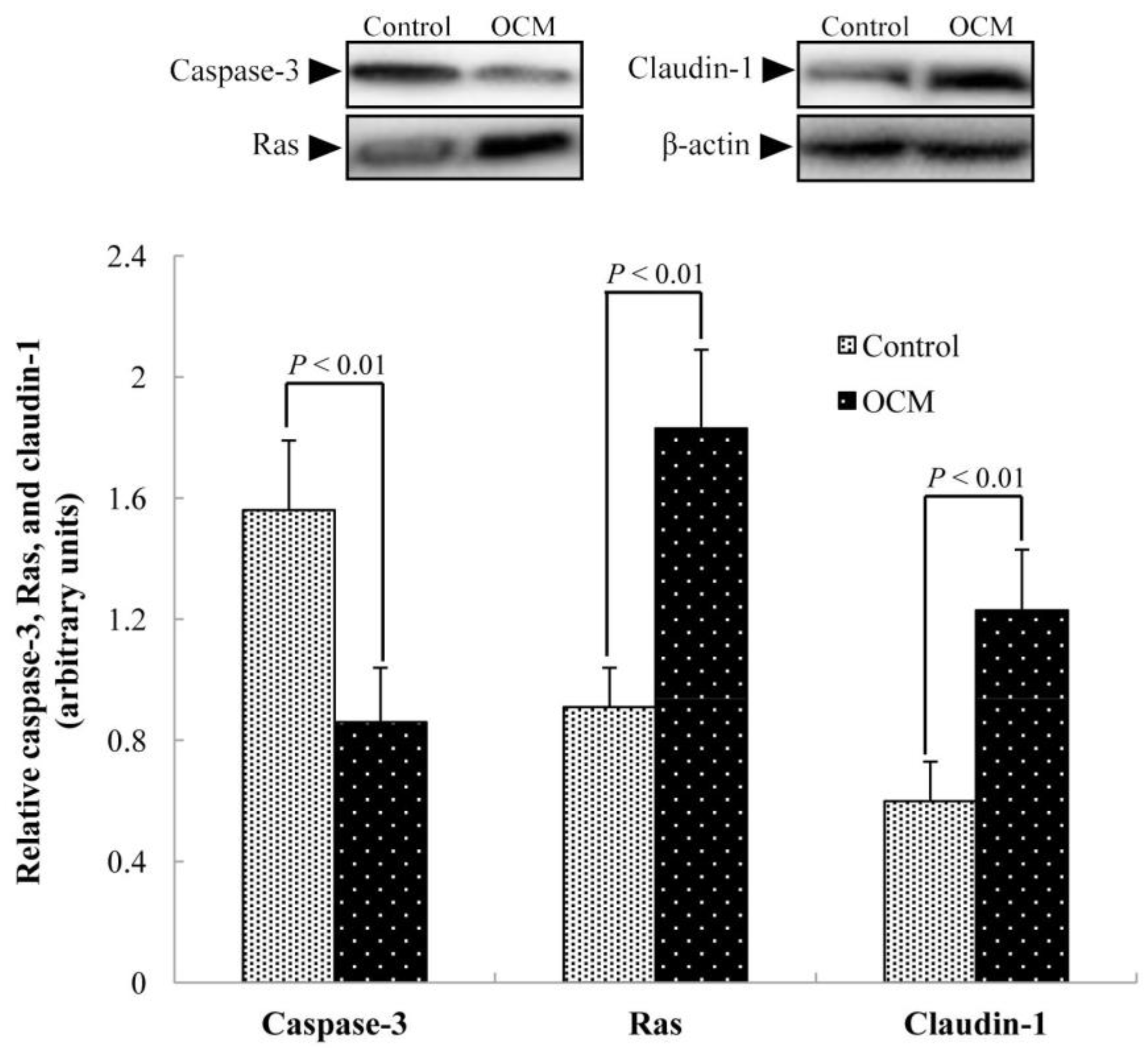
2.7. Abundance of Caspase-3, Ras, and Claudin-1 Proteins in the Jejunal Mucosa
3. Discussion
4. Materials and Methods
4.1. Animals and Treatments
4.2. Intestinal Sample Collection
4.3. Plasma Hormones, IGF-1 and PGE2
4.4. Intestinal Histology
4.5. Intestinal Mucosal DNA, RNA and Protein
4.6. Microbial DNA Extraction
4.7. QPCR Analyses for Mucosal Gene Expression and Intestinal Bacteria
4.8. Protein Immunoblot Analysis
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AMPK | AMP-Activated Protein Kinase |
| ADFI | Average Daily Feed Intake |
| ADG | Average Daily Gain |
| Bax | Bcl-2 Associated X Protein |
| Bcl-xL | B-Cell Lymphoma-Extra Large |
| EGFR | Epithelial Growth Factor Receptor |
| Erk1/2 | Extracellular Signal-Regulated Kinase 1/2 |
| GAPDH | Glyceraldehyde-3-Phosphate Dehydrogenase |
| JAM-A | Junctional Adhesion Molecule A |
| LPS | Lipopolysaccharide |
| mTOR | Mammalian Target of Rapamycin |
| MX1 | Myxovirus Resistance 1 |
| OASL | 2′-5′-Oligoadenylate Synthetase-Like Protein |
| OCM | Oleum Cinnamomi |
| P70S6K | Ribosomal Protein S6 Kinase |
| REG3G | Regenerating Islet-Derived Protein 3 Gamma |
| RPL4 | Ribosomal Protein L4 |
| UCP2 | Uncoupling Protein 2 |
| 4EBP1 | 4E-Binding Protein-1 |
References
- Si, W.; Gong, J.; Tsao, R.; Zhou, T.; Yu, H.; Poppe, C.; Johnson, R.; Du, Z. Antimicrobial activity of essential oils and structurally related synthetic food additives towards selected pathogenic and beneficial gut bacteria. J. Appl. Microbiol. 2006, 100, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Solórzano-Santos, F.; Miranda-Novales, M.G. Essential oils from aromatic herbs as antimicrobial agents. Curr. Opin. Biotechnol. 2012, 23, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Nuryastuti, T.; van der Mei, H.C.; Busscher, H.J.; Iravati, S.; Aman, A.T.; Krom, B.P. Effect of cinnamon oil on icaA expression and biofilm formation by staphylococcus epidermidis. Appl. Environ. Microbiol. 2009, 75, 6850–6855. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.T.; Chen, P.F.; Chang, S.C. Antibacterial activity of leaf essential oils and their constituents from Cinnamomum osmophloeum. J. Ethnopharmacol. 2001, 77, 123–127. [Google Scholar] [CrossRef]
- Shreaz, S.; Wani, W.A.; Behbehani, J.M.; Raja, V.; Irshad, M.; Karched, M.; Ali, I.; Siddiqi, W.A.; Hun, L.T. Cinnamaldehyde and its derivatives, a novel class of antifungal agents. Fitoterapia 2016, 112, 116–131. [Google Scholar] [CrossRef] [PubMed]
- Mendes, S.J.; Sousa, FI.; Pereira, D.M.; Ferro, T.A.; Pereira, I.C.; Silva, B.L.; Pinheiro, A.J.; Mouchrek, A.Q.; Monteiro-Neto, V.; Costa, S.K.; et al. Cinnamaldehyde modulates LPS-induced systemic inflammatory response syndrome through TRPA1-dependent and independent mechanisms. Int. Immunopharmacol. 2016, 34, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Gowder, S.J.T. Safety assessment of food flavor-cinnamaldehyde. Biosafety 2014, 3, e147. [Google Scholar] [CrossRef]
- Jenner, P.M.; Hagan, E.C.; Taylor, J.M.; Cook, E.L.; Fitzhugh, O.G. Food flavorings and compounds of related structure. I. Acute oral toxicity. Food Cosmet. Toxicol. 1964, 2, 327–343. [Google Scholar] [CrossRef]
- Wang, L.; Hou, Y.; Yi, D.; Ding, B.; Zhao, D.; Wang, Z.; Zhu, H.; Liu, Y.; Gong, J.; Assaad, H.; et al. Beneficial roles of dietary oleum cinnamomi in alleviating intestinal injury. Front. Biosci. (Landmark Ed.) 2015, 20, 814–828. [Google Scholar] [PubMed]
- Ogawa, K.; Ito, M. Appetite-enhancing Effects of trans-cinnamaldehyde, benzylacetone and 1-phenyl-2-butanone by inhalation. Planta. Med. 2016, 82, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Kang, L.; Zhang, D.; Ma, C.; Zhang, J.; Jia, K.; Liu, J.; Wang, R.; Kong, L. Cinnamaldehyde and allopurinol reduce fructose-induced cardiac inflammation and fibrosis by attenuating CD36-mediated TLR4/6-IRAK4/1 signaling to suppress NLRP3 inflammasome activation. Sci. Rep. 2016, 6, 27460. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Wang, L.; Zhang, W.; Yang, Z.; Ding, B.; Zhu, H.; Liu, Y.; Qiu, Y.; Yin, Y.; Wu, G. Protective effects of N-acetylcysteine on intestinal functions of piglets challenged with lipopolysaccharide. Amino Acids 2012, 43, 1233–1242. [Google Scholar] [CrossRef] [PubMed]
- Jeurissen, S.H.; Lewis, F.; van der Klis, J.D.; Mroz, Z.; Rebel, J.M.; ter Huurne, A.A. Parameters and techniques to determine intestinal health of poultry as constituted by immunity, integrity and functionality. Curr. Issues Intest. Microbiol. 2002, 3, 1–14. [Google Scholar] [PubMed]
- Fasina, Y.O.; Moran, E.T.; Ashwell, C.M.; Conner, D.E. Effect of dietary gelatin supplementation on the expression of selected enterocyte genes, intestinal development and early chick performance. Int. J. Poult. Sci. 2007, 6, 944–951. [Google Scholar] [CrossRef]
- Tan, B.; Yin, Y.; Kong, X.; Li, P.; Li, X.; Gao, H.; Li, X.; Huang, R.; Wu, G. l-Arginine stimulates proliferation and prevents endotoxin-induced death of intestinal cells. Amino Acids 2010, 38, 1227–1235. [Google Scholar] [CrossRef] [PubMed]
- Ott, S.J.; Musfeldt, M.; Ullmann, U.; Hampe, J.; Schreiber, S. Quantification of intestinal bacterial populations by real-time PCR with a universal primer set and minor groove binder probes: A global approach to the enteric flora. J. Clin. Microbiol. 2004, 42, 2566–2572. [Google Scholar] [CrossRef] [PubMed]
- Nieto-Bobadilla, M.S.; Siepmann, F.; Djouina, M.; Dubuquoy, L.; Tesse, N.; Willart, J.F.; Dubreuil, L.; Siepmann, J.; Neut, C. Controlled delivery of a new broad spectrum antibacterial agent against colitis: In vitro and in vivo performance. Eur. J. Pharm. Biopharm. 2015, 96, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.J.; Kovacs-Nolan, J.; Yang, C.; Archbold, T.; Fan, M.Z.; Mine, Y. l-cysteine supplementation attenuates local inflammation and restores gut homeostasis in a porcine model of colitis. Biochim. Biophys. Acta. 2009, 1790, 1161–1169. [Google Scholar] [CrossRef] [PubMed]
- West, A.B.; Isaac, C.A.; Carboni, J.M.; Morrow, J.S.; Mooseker, M.S.; Barwick, K.W. Localization of villin, a cytoskeletal protein specific to microvilli, in human ileum and colon and incolonic neoplasms. Gastroenterology 1988, 94, 343–352. [Google Scholar] [CrossRef]
- Wang, Y.; Srinivasan, K.; Siddiqui, M.R.; George, S.P.; Tomar, A.; Khurana, S. A novel role for villin in intestinal epithelial cell survival and homeostasis. J. Biol. Chem. 2008, 283, 9454–9464. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Wang, L.; Yi, D.; Ding, B.; Yang, Z.; Li, J.; Chen, X.; Qiu, Y.; Wu, G. N-acetylcysteine reduces inflammation in the small intestine by regulating redox, EGF and TLR4 signaling. Amino Acids 2013, 45, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Helmrath, M.A.; Shin, C.E.; Erwin, C.R. Intestinal adaptation is enhanced by epidermal growth factor independent of increased ileal epidermal growth factor receptor expression. J. Pediatr. Surg. 1998, 33, 980–985. [Google Scholar]
- Monteiro, A.C.; Parkos, C.A. Intracellular mediators of JAM-A-dependent epithelial barrier function. Ann. N. Y. Acad. Sci. 2012, 1257, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Jobgen, W.S.; Fried, S.K.; Fu, W.J.; Meininger, C.J.; Wu, G. Regulatory role for the arginine-nitric oxide pathway in metabolism of energy substrates. J. Nutr. Biochem. 2006, 17, 571–588. [Google Scholar] [CrossRef] [PubMed]
- Godino, R.F.; Fernández, A.I. New polymorphism of the influenza virus resistance Mx1 gene in Iberian domestic pigs. Postdoc. J. 2016, 4, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Loonen, L.M.; Stolte, E.H.; Jaklofsky, M.T.; Meijerink, M.; Dekker, J.; van Baarlen, P.; Wells, J.M. REG3γ-deficient mice have altered mucus distribution and increased mucosal inflammatory responses to the microbiota and enteric pathogens in the ileum. Mucosal Immunol. 2014, 7, 939–947. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Wang, L.; Ding, B.; Liu, Y.; Zhu, H.; Liu, J.; Li, Y.; Wu, X.; Yin, Y.; Wu, G. Dietary α-ketoglutarate supplementation ameliorates intestinal injury in lipopolysaccharide-challenged piglets. Amino Acids 2010, 39, 555–564. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, L.; Li, D.; Yin, Y.; Wang, X.; Li, P.; Dangott, L.J.; Hu, W.; Wu, G. Intrauterine growth restriction affects the proteomes of the small intestine, liver and skeletal muscle in newborn pigs. J. Nutr. 2008, 138, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Tako, E.; Glahn, R.P.; Welch, R.M.; Lei, X.; Yasuda, K.; Miller, D.D. Dietary inulin affects the expression of intestinal enterocyte iron transporters, receptors and storage protein and alters the microbiota in the pig intestine. Br. J. Nutr. 2008, 99, 472–480. [Google Scholar] [CrossRef] [PubMed]
- Tuomisto, S.; Karhunen, P.J.; Pessi, T. Time-dependent post mortem changes in the composition of intestinal bacteria using real-time quantitative PCR. Gut Pathog. 2013, 5, 35. [Google Scholar] [CrossRef] [PubMed]
- Kang, P.; Xiao, H.; Hou, Y.; Ding, B.; Liu, Y.; Zhu, H.; Hu, Q.; Hu, Y.; Yin, Y. Effects of Astragalus polysaccharides, Achyranthes bidentata polysaccharides, and Acantbepanax senticosus paponin on the performance and immunity in weaned pigs. Asian-Aust. J. Anim. Sci. 2010, 23, 750–756. [Google Scholar] [CrossRef]
- Kang, P.; Zhang, L.; Hou, Y.; Ding, B.; Yi, D.; Wang, L.; Zhu, H.; Liu, Y.; Yin, Y.; Wu, G. Effects of l-proline on the growth performance, and blood parameters in weaned lipopolysaccharide (LPS)-challenged pigs. Asian-Aust. J. Anim. Sci. 2014, 27, 1150–1156. [Google Scholar] [CrossRef] [PubMed]
- Uni, Z.; Ganot, S.; Sklan, D. Posthatch development of mucosal function in the broiler small intestine. Poult. Sci. 1998, 77, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.S.; DeMouchelle, E.; Koniuchi, D. A simple fluorimetric method for the determination of RNA and DNA in tissue. J. Lab. Clin. Med. 1972, 80, 598–601. [Google Scholar] [PubMed]
- Munro, H.N.; Fleck, A. Analysis of Tissues and Body Fluids for Nitrogenous Constituents. In Mammalian Protein Metabolism; Academic Press: New York, NY, USA, 1969; pp. 465–483. ISBN 978-1-4832-3211-9. [Google Scholar]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L. Protein measurement with the folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [PubMed]
- Castillo, M.; Martín-Orúe, S.M.; Manzanilla, E.G.; Badiola, I.; Martín, M.; Gasa, J. Quantification of total bacteria, enterobacteria and lactobacilli populations in pig digesta by real-time PCR. Vet. Microbiol. 2006, 114, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Yi, D.; Hou, Y.; Wang, L.; Long, M.; Hu, S.; Mei, H.; Yan, L.; Hu, C.A.; Wu, G. N-acetylcysteine stimulates protein synthesis in enterocytes independently of glutathione synthesis. Amino Acids 2016, 48, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Meurens, F.; Berri, M.; Auray, G.; Melo, S.; Levast, B.; Virlogeux-Payant, I.; Chevaleyre, C.; Gerdts, V.; Salmon, H. Early immune response following Salmonella enterica subspecies enterica serovar Typhimurium infection in porcine jejunal gut loops. Vet. Res. 2009, 40, 5. [Google Scholar] [CrossRef] [PubMed]
- Nygard, A.; Jørgensen, C.B.; Cirera, S.; Fredholm, M. Selection of reference genes for gene expression studies in pig tissues using SYBR green qPCR. BMC Mol. Biol. 2007, 8, 67. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−∆∆Ct method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Yi, D.; Hou, Y.; Wang, L.; Ding, B.; Yang, Z.; Li, J.; Long, M.; Liu, Y.; Wu, G. Dietary N-acetylcysteine supplementation alleviates liver injury in lipopolysaccharide-challenged piglets. Br. J. Nutr. 2014, 111, 46–54. [Google Scholar] [CrossRef] [PubMed]
| Item | Control Group | OCM Group | p-Value |
|---|---|---|---|
| ADG (g/d) | 260 ± 60.4 | 308 ± 60.3 | 0.138 |
| ADFI (g/d) | 391 ± 53.9 b | 444 ± 33.9 a | 0.035 |
| F/G | 1.54 ± 0.23 | 1.42 ± 0.21 | 0.308 |
| Diarrhoea incidence (%) | 9.52 ± 1.29 a | 5.95 ± 1.10 b | <0.001 |
| Item | Control Group | OCM Group | p-Value |
|---|---|---|---|
| Insulin (μU/mL) | 5.24 ± 1.07 b | 8.16 ± 1.30 a | 0.038 |
| Cortisol (ng/mL) | 49.3 ± 11.0 | 42.8 ± 7.96 | 0.499 |
| IGF-1 (ng/mL) | 235 ± 57.4 | 242 ± 57.9 | 0.193 |
| PGE2 (pg/mL) | 29.3 ± 3.09 | 29.0 ± 2.76 | 0.813 |
| Item | Control Group | OCM Group | p-Value |
|---|---|---|---|
| Villus height (µm) | |||
| Duodenum | 246 ± 33.1 | 249 ± 21.1 | 0.834 |
| Jejunum | 241 ± 37.3 | 258 ± 18.9 | 0.281 |
| Ileum | 227 ± 44.2 | 249 ± 15.4 | 0.090 |
| Crypt depth (µm) | |||
| Duodenum | 83.0 ± 13.5 | 93.5 ± 14.4 | 0.302 |
| Jejunum | 88.4 ± 10.9 | 94.9 ± 12.4 | 0.283 |
| Ileum | 80.2 ± 14.8 | 91.9 ± 10.8 | 0.062 |
| Villus height/crypt depth | |||
| Duodenum | 2.98 ± 0.18 | 2.66 ± 0.23 | 0.325 |
| Jejunum | 2.75 ± 0.31 | 2.75 ± 0.39 | 0.906 |
| Ileum | 2.85 ± 0.40 | 2.74 ± 0.36 | 0.634 |
| Villus width (µm) | |||
| Duodenum | 121 ± 12.2 b | 147 ± 8.61 a | <0.001 |
| Jejunum | 125 ± 12.4 b | 149 ± 8.92 a | <0.001 |
| Ileum | 104 ± 10.9 | 116 ± 22.1 | 0.216 |
| Villous surface area (µm2) | |||
| Duodenum | 20803 ± 3305 b | 25544 ± 2114 a | 0.004 |
| Jejunum | 20029 ± 3086 b | 25729 ± 1617 a | <0.001 |
| Ileum | 15419 ± 3252 | 18593 ± 3644 | 0.087 |
| Item | Control Group | OCM Group | p-Value |
|---|---|---|---|
| DNA (mg/g protein) | |||
| Duodenum | 2.49 ± 0.42 | 2.32 ± 0.33 | 0.380 |
| Jejunum | 2.52 ± 0.49 | 2.30 ± 0.57 | 0.605 |
| Ileum | 1.84 ± 0.33 b | 3.61 ± 0.74 a | <0.001 |
| RNA/DNA ratio | |||
| Duodenum | 0.17 ± 0.04 | 0.16 ± 0.03 | 0.323 |
| Jejunum | 0.16 ± 0.02 | 0.17 ± 0.04 | 0.890 |
| Ileum | 0.15 ± 0.03 b | 0.26 ± 0.02a | <0.001 |
| Protein/DNA ratio | |||
| Duodenum | 417 ± 71.5 | 422 ± 96.4 | 0.918 |
| Jejunum | 413 ± 89.2 | 435 ± 108 | 0.390 |
| Ileum | 533 ± 71.9 | 555 ± 65.2 | 0.542 |
| Genes | Forward | Reverse |
|---|---|---|
| Bax | TTTCTGACGGCAACTTCAACTG | AGCCACAAAGATGGTCACTGTCT |
| Bcl-xL | GAAACCCCTAGTGCCATCAA | GGGACGTCAGGTCACTGAAT |
| Villin | TATTATTGGTGTTCGTGCTA | TCTGGAGGAATAGGATACTAA |
| JAM-A | AATCAGTGTTCCCTCCTCTGCTAC | ACGGTTGCTCTTGGGCTCT |
| mTOR | TTGTTGCCCCCTATTGTGAAG | CCTTTCGAGATGGCAATGGA |
| P70S6K | GGAAACAAGTGGAATAGAGCAGATG | TTGGAAGTGGTGCAGAAGCTT |
| 4EBP1 | CCGGAAGTTCCTAATGGAGTGT | GGTTCTGGCTGGCATCTGT |
| EGFR | GGCCTCCATGCTTTTGAGAA | GACGCTATGTCCAGGCCAA |
| Ras | AAGAGCGACCTCACCACCA | GCGTTCTTGGCACTCGTCT |
| Erk1/2 | AAGCTCTTGAAGACGCAGCAC | CAGCAGGTTGGAAGGTTTGAG |
| AMPK | CGACGTGGAGCTGTACTGCTT | CATAGGTCAGGCAGAACTTGC |
| UCP2 | AGGGTCCCCGAGCCTTCT | CAGCTGCTCATAGGTGACAAACA |
| OASL | GGCACCCCTGTTTTCCTCT | AGCACCGCTTTTGGATGG |
| MX1 | AGTGCGGCTGTTTACCAAG | TTCACAAACCCTGGCAACTC |
| MX2 | CGCATTCTTTCACTCGCATC | CCTCAACCCACCAACTCACA |
| REG3G | CTGTCTCAGGTCCAAGGTGAAG | CAAGGCATAGCAGTAGGAAGCA |
| Enterobacteriaceae family | CATTGACGTTACCCGCAGAAGAAGC | CTCTACGAGACTCAAGCTTGC |
| Enterococcus genus | CCCTTATTGTTAGTTGCCATCATT | ACTCGTTGTACTTCCCATTGT |
| Clostridium coccoides | AATGACGGTACCTGACTAA | CTTTGAGTTTCATTCTTGCGAA |
| Lactobacillus genus | AGCAGTAGGGAATCTTCCA | CACCGCTACACATGGAG |
| Bifidobacterium genus | TCGCGTC(C/T)GGTGTGAAAG | CCACATCCAGC(A/G)TCCAC |
| Total eubacteria (16S rRNA) | CGGTCCAGACTCCTACGGG | TTACCGCGGCTGCTGGCAC |
| RPL4 | GAGAAACCGTCGCCGAAT | GCCCACCAGGAGCAAGTT |
| GAPDH | CGTCCCTGAGACACGATGGT | CCCGATGCGGCCAAAT |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yi, D.; Fang, Q.; Hou, Y.; Wang, L.; Xu, H.; Wu, T.; Gong, J.; Wu, G. Dietary Supplementation with Oleum Cinnamomi Improves Intestinal Functions in Piglets. Int. J. Mol. Sci. 2018, 19, 1284. https://doi.org/10.3390/ijms19051284
Yi D, Fang Q, Hou Y, Wang L, Xu H, Wu T, Gong J, Wu G. Dietary Supplementation with Oleum Cinnamomi Improves Intestinal Functions in Piglets. International Journal of Molecular Sciences. 2018; 19(5):1284. https://doi.org/10.3390/ijms19051284
Chicago/Turabian StyleYi, Dan, Qiuhong Fang, Yongqing Hou, Lei Wang, Haiwang Xu, Tao Wu, Joshua Gong, and Guoyao Wu. 2018. "Dietary Supplementation with Oleum Cinnamomi Improves Intestinal Functions in Piglets" International Journal of Molecular Sciences 19, no. 5: 1284. https://doi.org/10.3390/ijms19051284
APA StyleYi, D., Fang, Q., Hou, Y., Wang, L., Xu, H., Wu, T., Gong, J., & Wu, G. (2018). Dietary Supplementation with Oleum Cinnamomi Improves Intestinal Functions in Piglets. International Journal of Molecular Sciences, 19(5), 1284. https://doi.org/10.3390/ijms19051284




