Effect of Amelogenin Coating of a Nano-Modified Titanium Surface on Bioactivity
Abstract
:1. Introduction
2. Results
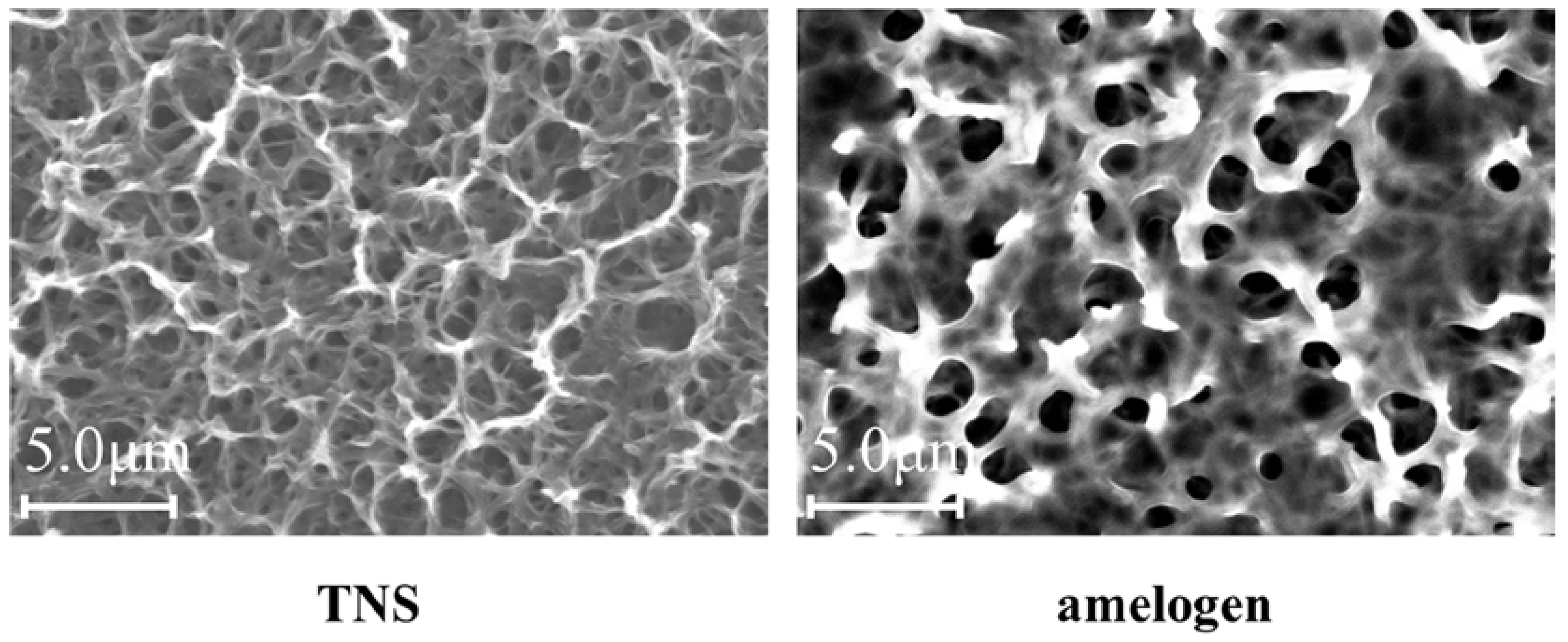
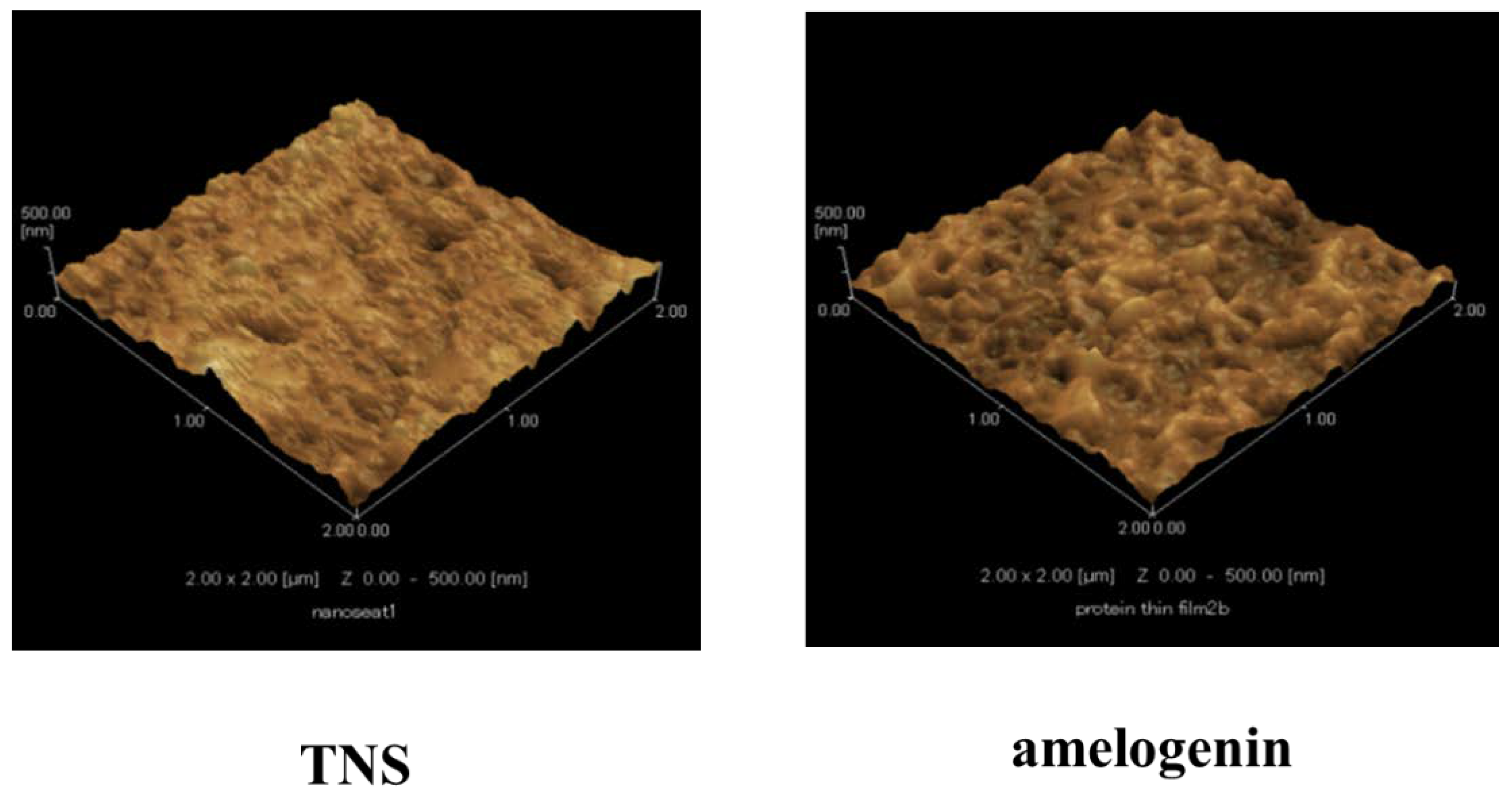
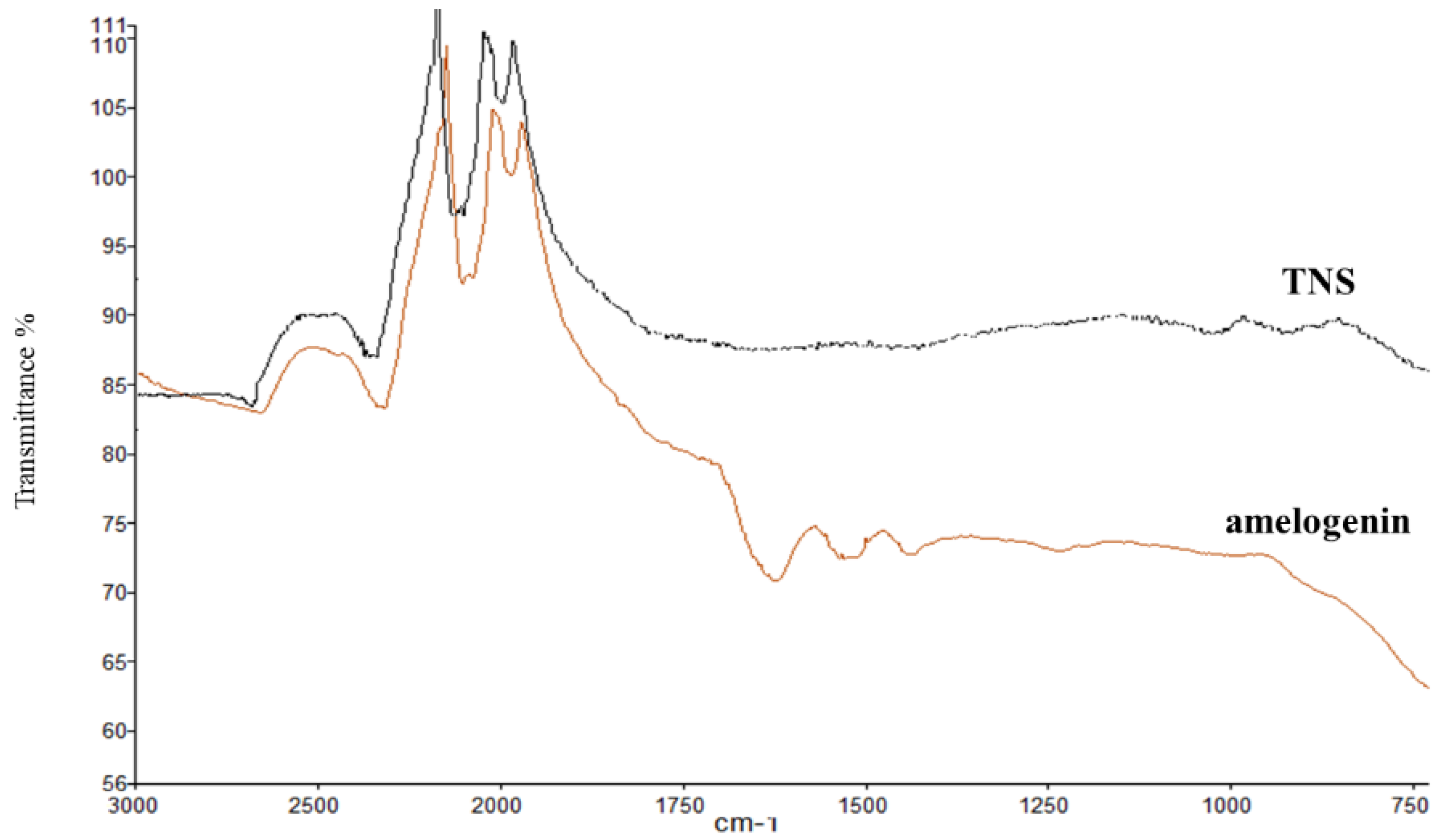
2.1. Sample Preparation
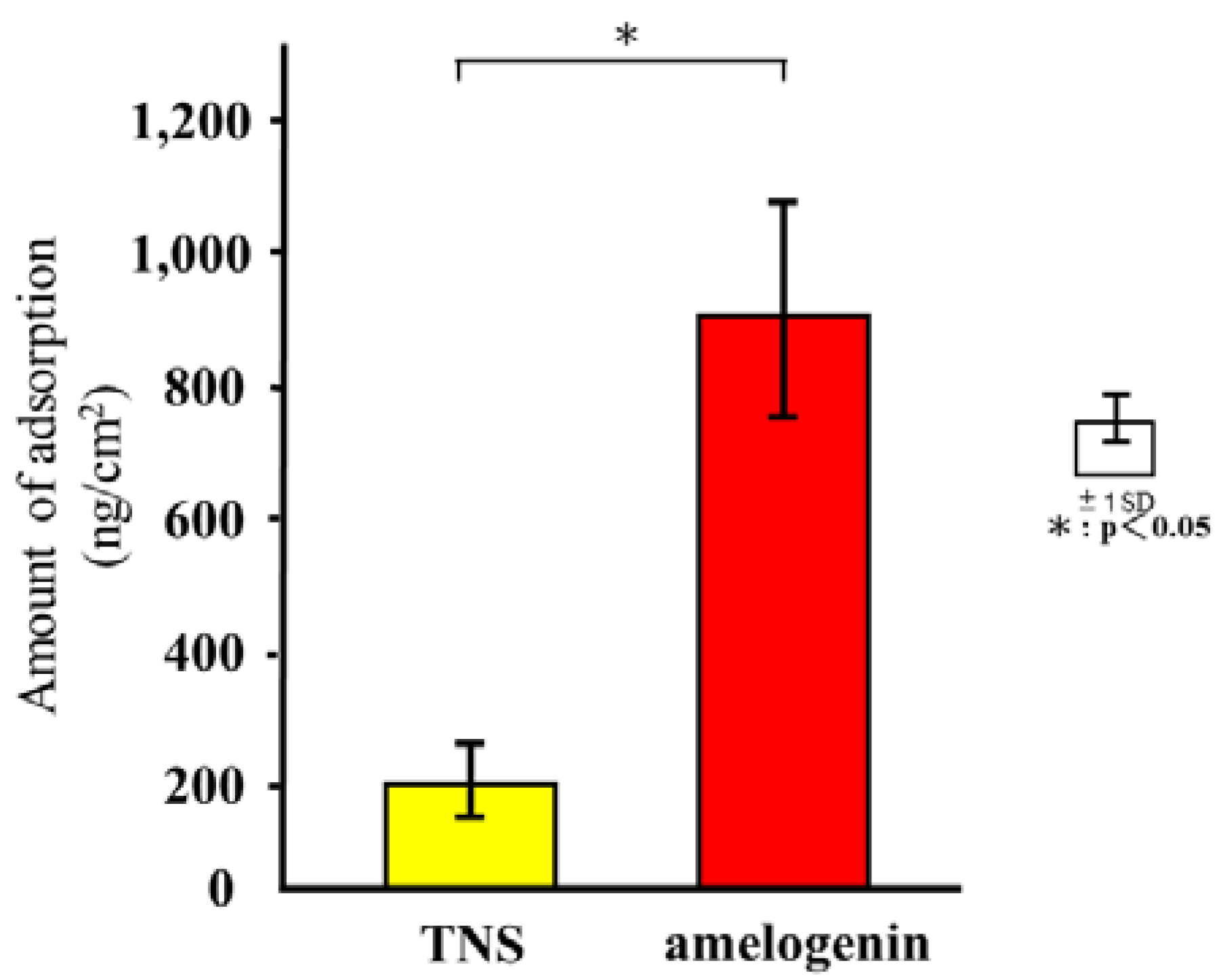
2.2. Quartz Crystal Microbalance (QCM) Measurements
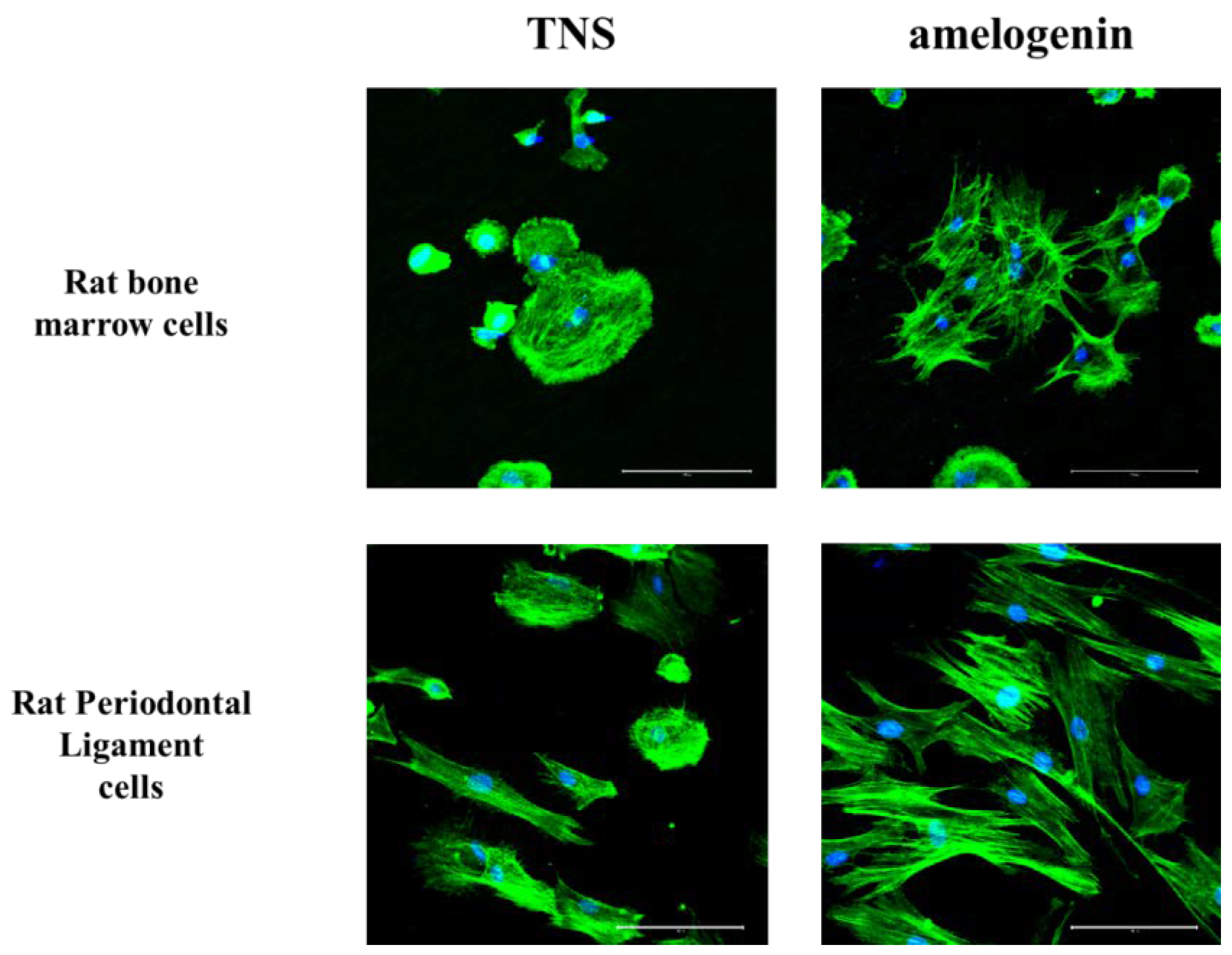
2.3. Cell Adhesion
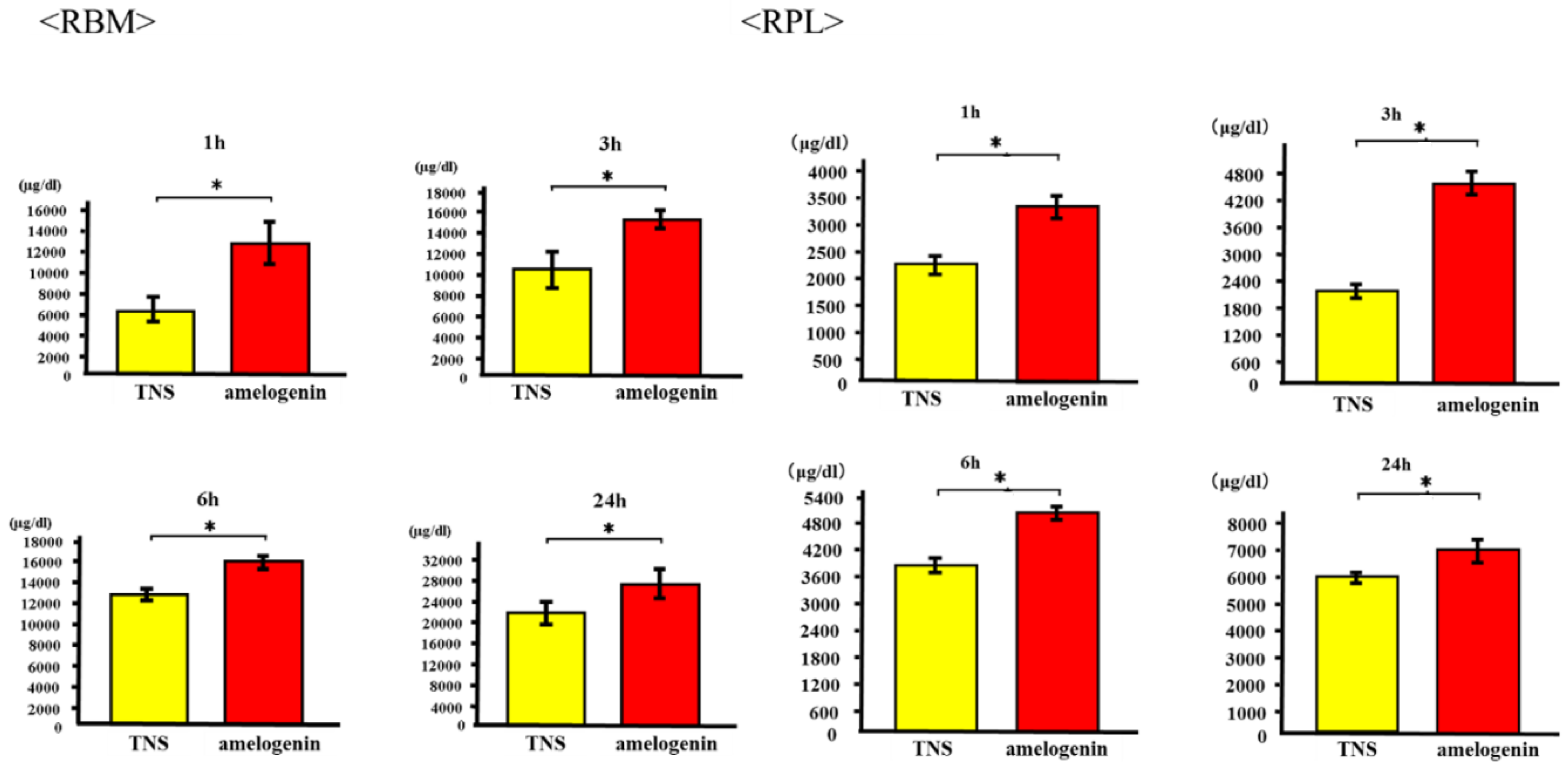
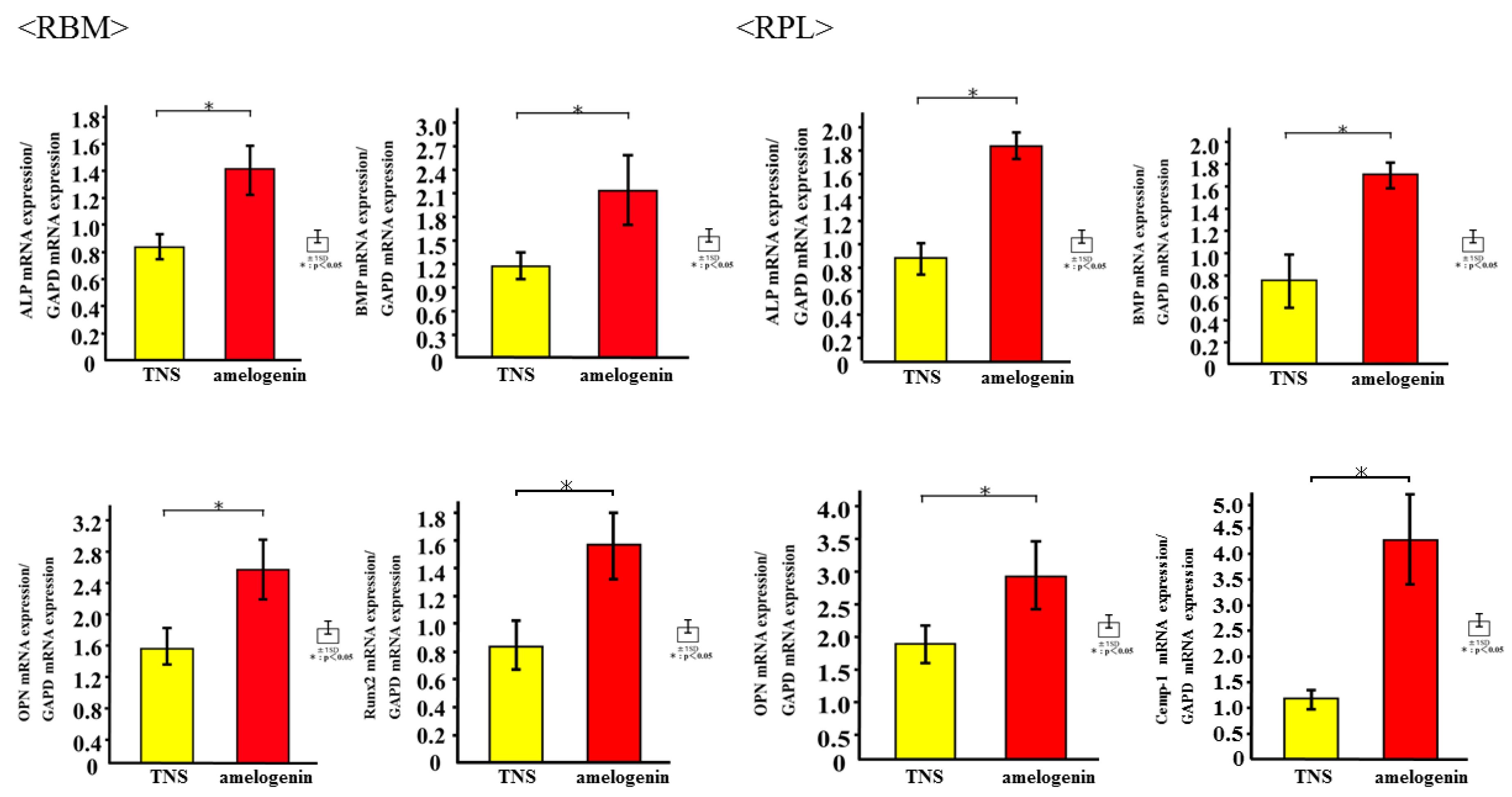
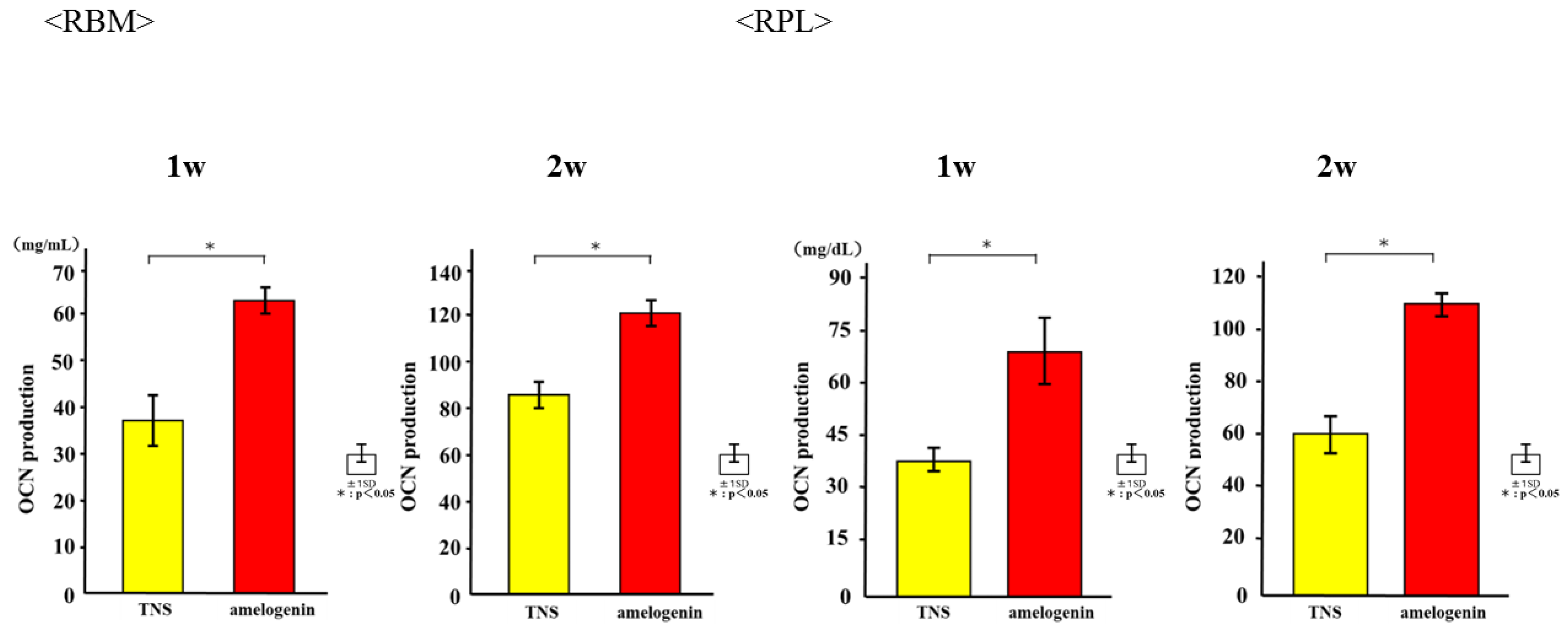
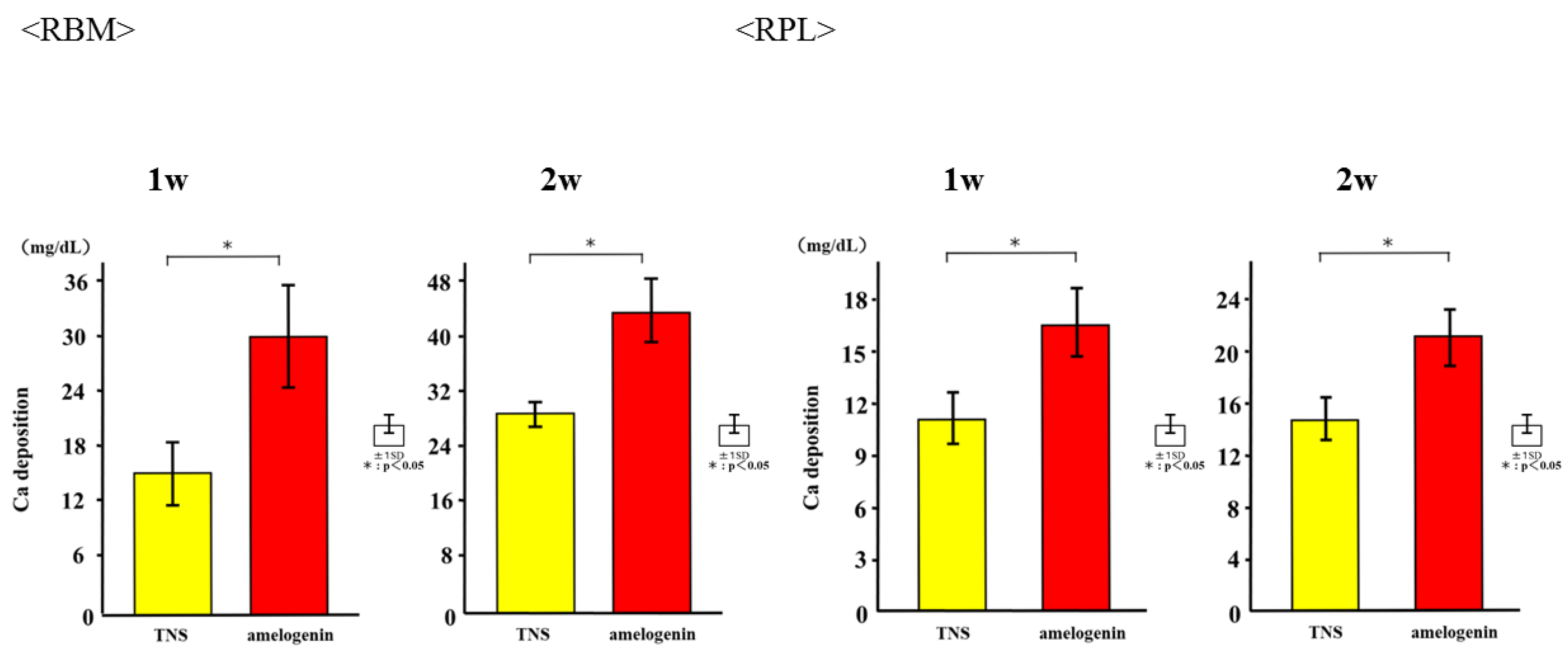
2.4. Amelogenin Induced RBM and RPL Cell Differentiation and Matrix Deposition on TNS-Modified Titanium Surfaces In Vitro
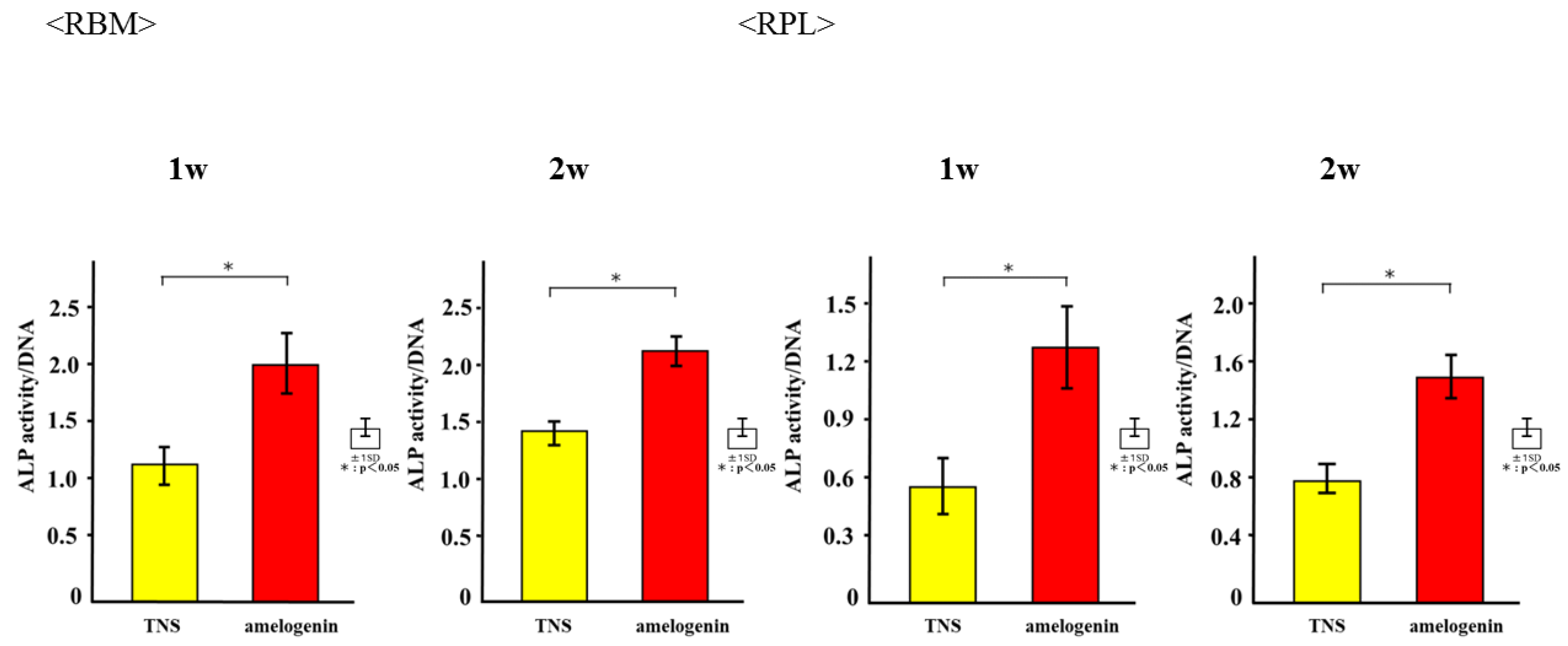
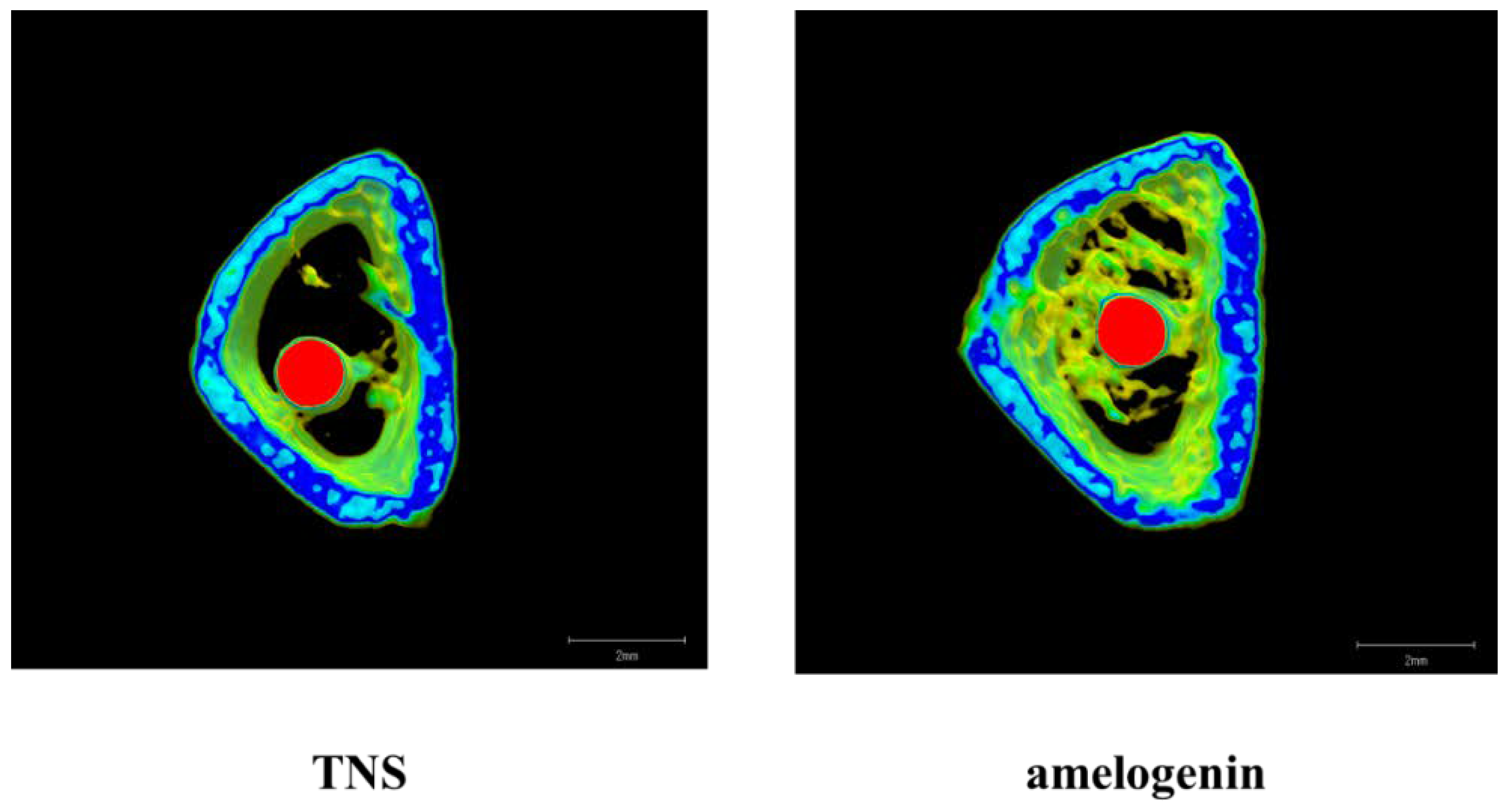
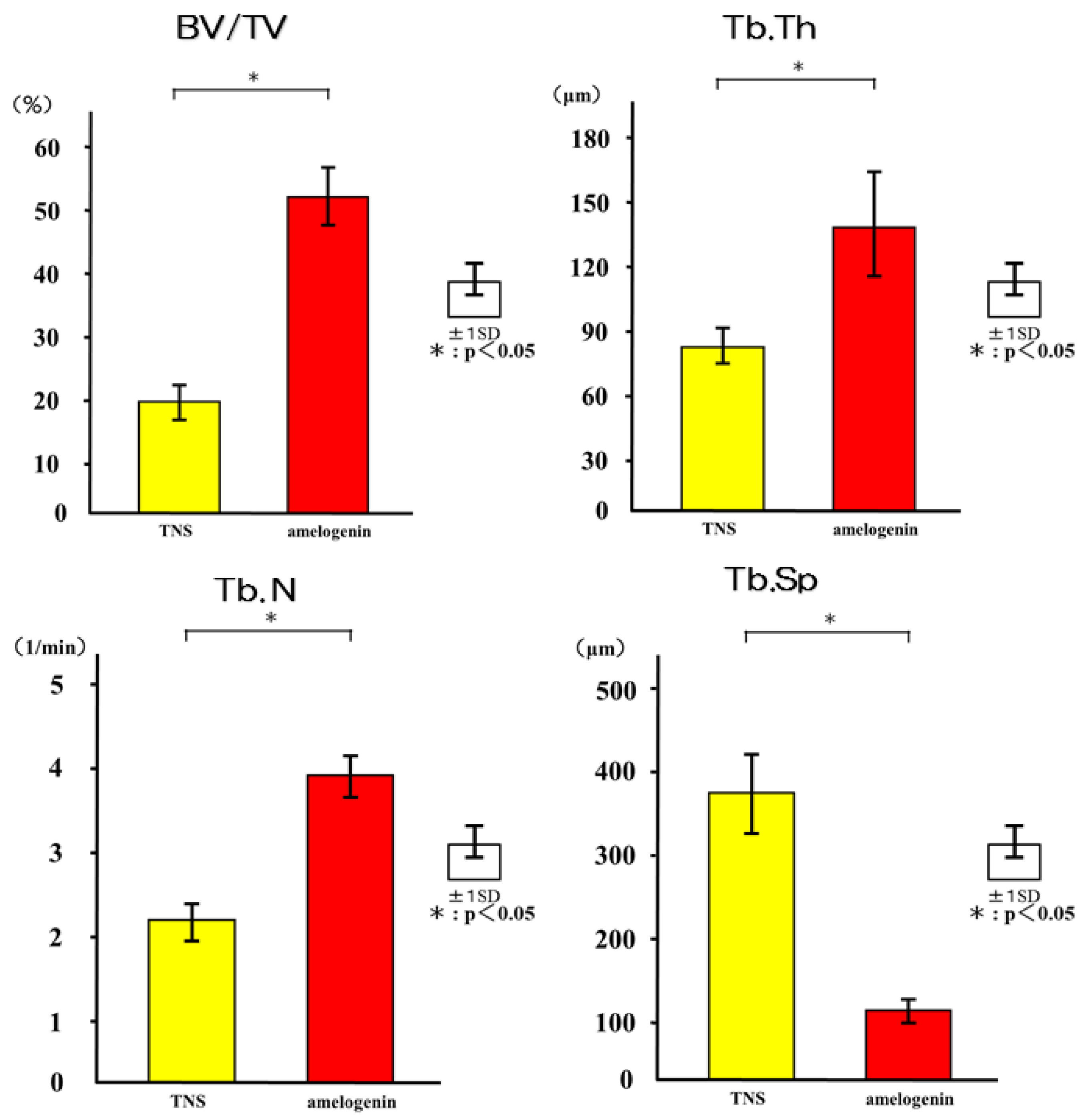
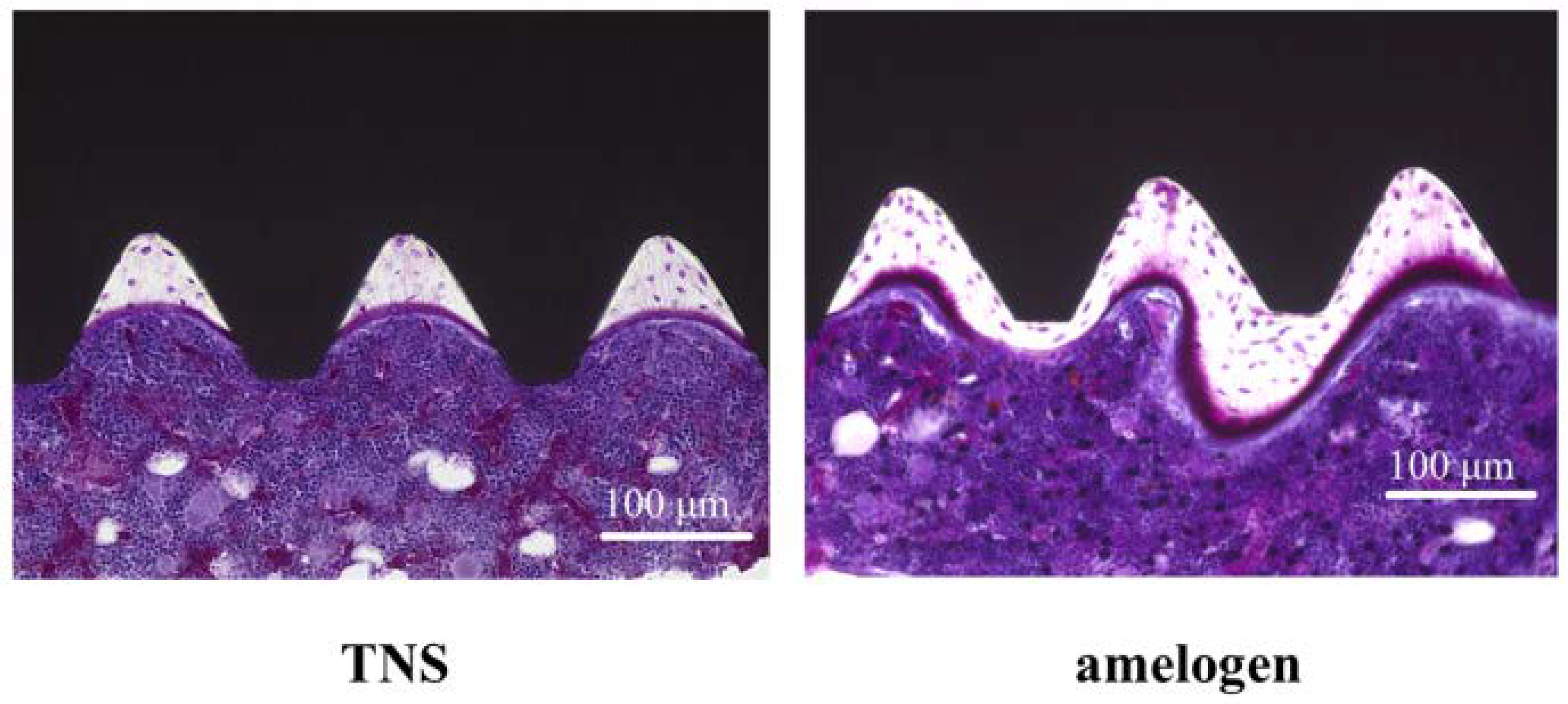
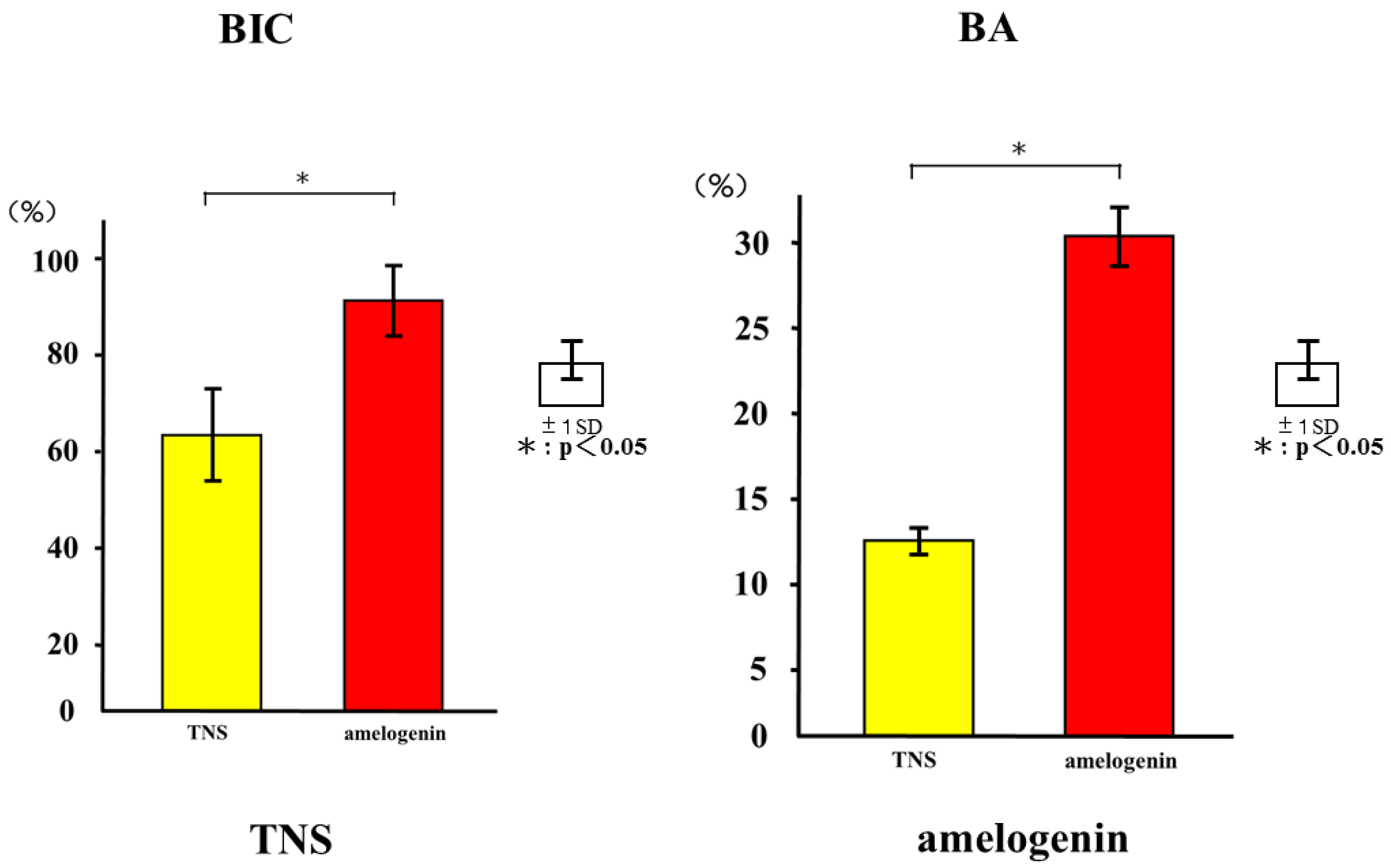
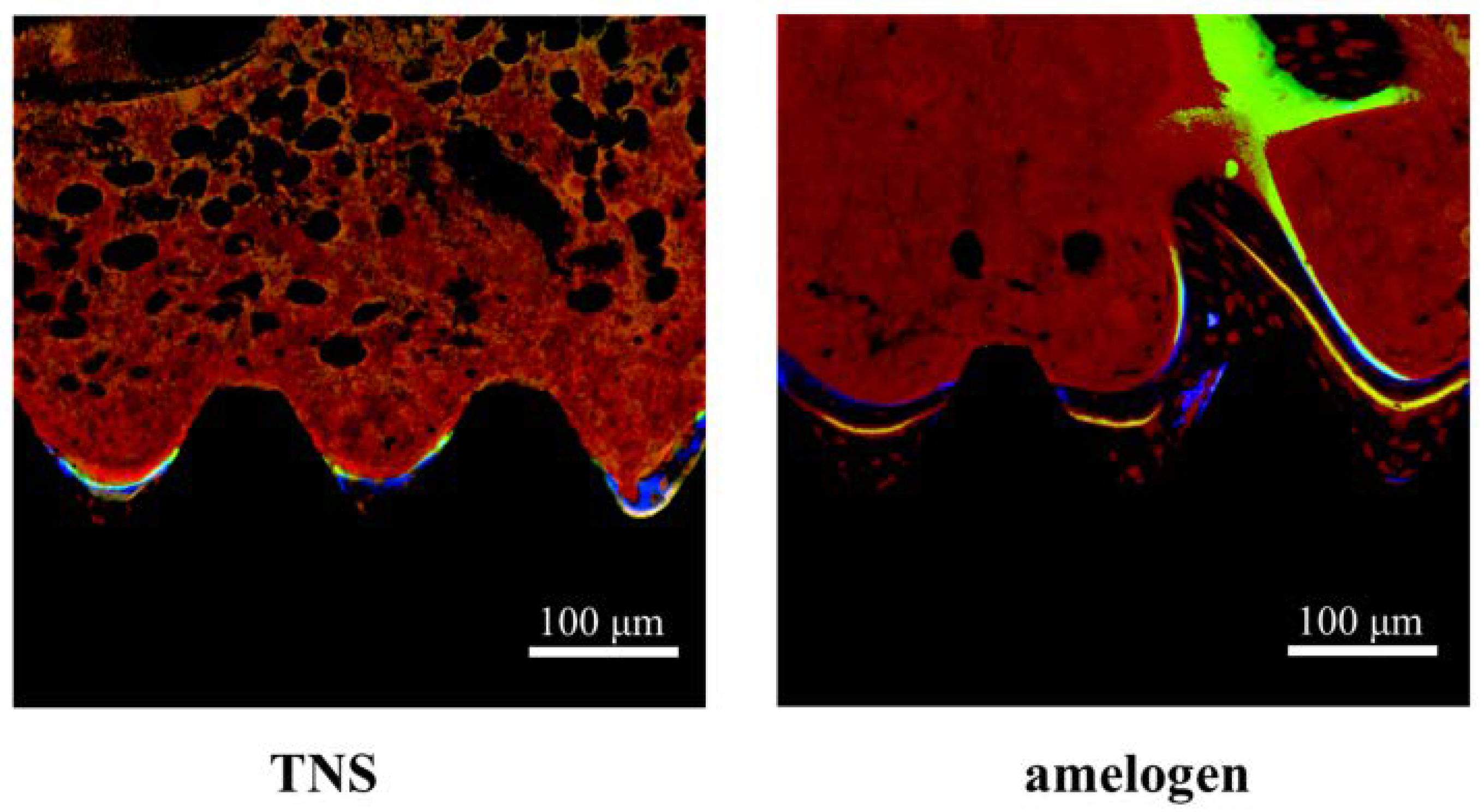
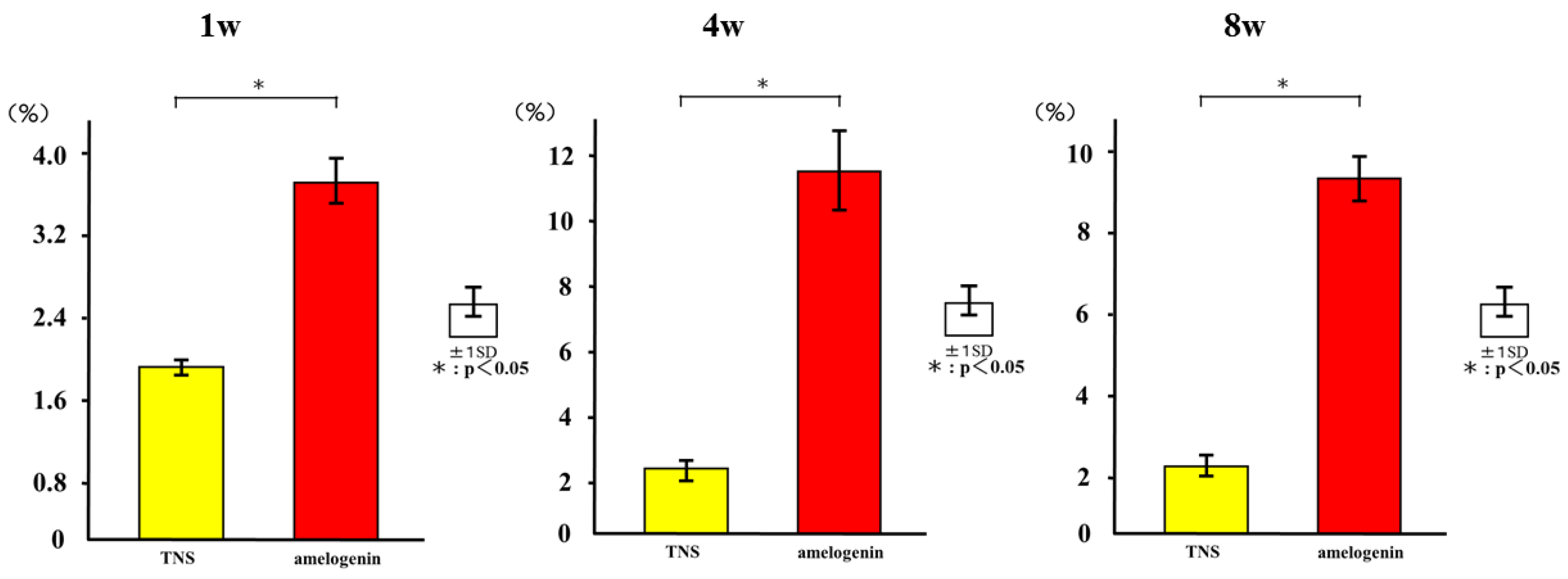
2.5. Amelogenin Induced Bone Differentiation on TNS-Modified Titanium Surfaces In Vivo
3. Discussion
4. Materials and Methods
4.1. Sample Preparation
4.2. Characterization of Materials
4.3. QCM Measurements
4.4. Cell Culture
4.5. Cell Adhesion
4.6. Amelogenin-Induced RBM and RPL Cell Differentiation and Matrix Deposition on TNS-Modified Titanium Surfaces In Vitro
4.7. Animal Model and Surgical Procedures
4.8. Sequential Fluorescence Labeling
4.9. Amelogenin-Induced Bone Differentiation on the TNS-Modified Titanium Surface In Vivo
4.10. Statistical Analyses
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| ALP | alkaline phosphatase |
| BMP | bone morphogenetic protein |
| DAPI | 4′,6-diamidino-2-phenylindole |
| EMD | enamel matrix derivative |
| FTIR | Fourier transform-infrare |
| OCN | osteocalcin |
| OPN | osteopontin |
| PBS | phosphate-buffered saline |
| QCM | Quartz crystal microbalance |
| RBM | rat bone marrow |
| RPL | rat periodontal ligament |
| Runx2 | runt-related transcription factor 2 |
| SEM | scanning electron microscopy |
| SPM | scanning probe microscopy |
| TiO2 | titania |
| TNS | titania nano sheet |
| XPS | X-ray photoelectron spectroscopy |
References
- Moradian-Oldak, J.; Wen, H.B.; Schneider, G.B.; Stanford, C.M. Tissue engineering strategies for the future generation of dental implants. Periodontology 2000 2006, 41, 157–176. [Google Scholar] [CrossRef] [PubMed]
- Kirsch, A. The two-phase implantation method using IMZ intramobile cylinder implants. J. Oral Implantol. 1983, 11, 197–210. [Google Scholar] [PubMed]
- Skalak, R. Biomechanical considerations in osseointegrated prostheses. J. Prosthet. Dent. 1983, 49, 843–848. [Google Scholar] [CrossRef]
- Buser, D.; Warrer, K.; Karring, T. Formation of a periodontal ligament around titanium implants. J. Periodontol. 1990, 61, 597–601. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.H. Periodontal ligament formation around titanium implants using cultured periodontal ligament cells: A pilot study. Int. J. Oral Maxillofac. Implants 2000, 15, 193–196. [Google Scholar] [PubMed]
- Hasegawa, M.; Yamato, M.; Kikuchi, A.; Okano, T.; Ishikawa, I. Human periodontal ligament cell sheets can regenerate periodontal ligament tissue in an athymic rat model. Tissue Eng. 2005, 11, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Boyan, B.D.; Hummert, T.W.; Dean, D.D.; Schwartz, Z. Role of material surfaces in regulating bone and cartilage cell response. Biomaterials 1996, 17, 137–146. [Google Scholar] [CrossRef]
- Schwartz, Z.; Lohmann, C.H.; Oefinger, J.; Bonewald, L.F.; Dean, D.D.; Boyan, B.D. Implant surface characteristics modulate differentiation behavior of cells in the osteoblastic lineage. Adv. Dent. Res. 1999, 13, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, K.; Saruwatari, L.; Nakamura, H.K.; Yang, J.M.; Ogawa, T. Enhanced intrinsic biomechanical properties of osteoblastic mineralized tissue on roughened titanium surface. J. Biomed. Mater. Res. A 2005, 72, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, T.; Ozawa, S.; Shih, J.H.; Ryu, K.H.; Sukotjo, C.; Yang, J.M.; Nishimura, I. Biomechanical evaluation of osseous implants having different surface topographies in rats. J. Dent. Res. 2000, 79, 1857–1863. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, T.; Nishimura, I. Different bone integration profiles of turned and acid-etched implants associated with modulated expression of extracellular matrix genes. Int. J. Oral Maxillofac. Implants 2003, 18, 200–210. [Google Scholar] [PubMed]
- Liu, X.; Chu, P.K.; Ding, C. Surface modification of titanium, titanium alloys, and related materials for biomedical applications. Mater. Sci. Eng. R Rep. 2004, 47, 49–121. [Google Scholar] [CrossRef]
- Martin, J.Y.; Schwartz, Z.; Hummert, T.W.; Schraub, D.M.; Simpson, J.; Lankford, J., Jr.; Dean, D.D.; Cochran, D.L.; Boyan, B.D. Effect of titanium surface roughness on proliferation, differentiation, and protein synthesis of human osteoblast-like cells (MG63). J. Biomed. Mater. Res. 1995, 29, 389–401. [Google Scholar] [CrossRef] [PubMed]
- Hamlet, S.; Alfarsi, M.; George, R.; Ivanovski, S. The effect of hydrophilic titanium surface modification on macrophage inflammatory cytokine gene expression. Clin. Oral Implants Res. 2012, 23, 584–590. [Google Scholar] [CrossRef] [PubMed]
- Cooper, L.F.; Masuda, T.; Yliheikkilä, P.K.; Felton, D.A. Generalizations regarding the process and phenomenon of osseointegration. Part II. In vitro studies. Int. J. Oral Maxillofac. Implants 1997, 13, 163–174. [Google Scholar]
- Setzer, B.; Bächle, M.; Metzger, M.C.; Kohal, R.J. The gene-expression and phenotypic response of hFOB 1.19 osteoblasts to surface-modified titanium and zirconia. Biomaterials 2009, 30, 979–990. [Google Scholar] [CrossRef] [PubMed]
- Kasuga, T.; Hiramatsu, M.; Hoson, A.; Sekino, T.; Niihara, K. Titania nanotubes prepared by chemical processing. Adv. Mater. 1999, 11, 1307–1311. [Google Scholar] [CrossRef]
- Komasa, S.; Taguchi, Y.; Nishida, H.; Tanaka, M.; Kawazoe, T. Bioactivity of nanostructure on titanium surface modified by chemical processing at room temperature. J. Prosthodont. Res. 2012, 56, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Fujino, T.; Taguchi, Y.; Komasa, S.; Sekino, T.; Tanaka, M. Cell differentiation on nanoscale features of a titanium surface: Effects of deposition time in NaOH solution. J. Hard Tissue Biol. 2014, 23, 63–70. [Google Scholar] [CrossRef]
- Xing, H.; Komasa, S.; Taguchi, Y.; Sekino, T.; Okazaki, J. Osteogenic activity of titanium surfaces with nanonetwork structures. Int. J. Nanomed. 2014, 9, 1741–1755. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Komasa, S.; Mashimo, C.; Sekino, T.; Okazaki, J. Effect of ultraviolet treatment on bacterial attachment and osteogenic activity to alkali-treated titanium with nanonetwork structures. Int. J. Nanomed. 2017, 12, 4633–4646. [Google Scholar] [CrossRef] [PubMed]
- Nakano, Y.; Komasa, S.; Taguchi, Y.; Sekino, T.; Okazaki, J. Rat endothelial cell attachment, behavior and gene expression on NaOH-treated titanium surfaces. J. Oral Tissue Eng. 2013, 11, 189–200. [Google Scholar]
- Hara, Y.; Komasa, S.; Yoshimine, S.; Nishizaki, H.; Okazaki, J. Effect of nano modified titanium surface on adsorption of rat periodontal ligament cells. J. Osaka Dent. Univ. 2018, in press. [Google Scholar]
- Hoang, A.M.; Klebe, R.J.; Steffensen, B.; Ryu, O.H.; Simmer, J.P.; Cochran, D.L. Amelogenin is a cell adhesion protein. J. Dent. Res. 2002, 81, 497–500. [Google Scholar] [CrossRef] [PubMed]
- Fukumoto, S.; Kiba, T.; Hall, B.; Iehara, N.; Nakamura, T.; Longenecker, G.; Krebsbach, P.H.; Nanci, A.; Kulkarni, A.B.; Yamada, Y. Ameloblastin is a cell adhesion molecule required for maintaining the differentiation state of ameloblasts. J. Cell Biol. 2004, 167, 973–983. [Google Scholar] [CrossRef] [PubMed]
- Gibson, C.W. The amelogenin “enamel proteins” and cells in the periodontium. Crit. Rev. Eukaryot. Gene Expr. 2008, 18, 345–360. [Google Scholar] [CrossRef] [PubMed]
- Zetterström, O.; Andersson, C.; Eriksson, L.; Fredriksson, A.; Friskopp, J.; Heden, G.; Jansson, B.; Lundgren, T.; Nilveus, R.; Olsson, A.; et al. Clinical safety of enamel matrix derivative (EMDOGAIN) in the treatment of periodontal defects. J. Clin. Periodontol. 1997, 24, 697–704. [Google Scholar] [CrossRef] [PubMed]
- Sculean, A.; Chiantella, G.C.; Windisch, P.; Donos, N. Clinical and histologic evaluation of human lntrabony defects treated with an enamel matrix protein derivative (Emdogain). Int. J. Periodontics Restor. Dent. 2000, 20, 374–381. [Google Scholar]
- Esposito, M.; Grusovin, M.G.; Coulthard, P.; Worthington, H.V. Enamel matrix derivative (Emdogain) for periodontal tissue regeneration in intrabony defects. Cochrane Database Syst. Rev. 2005, 4, CD003875. [Google Scholar]
- Gestrelius, S.; Lyngstadaas, S.P.; Hammarström, L. Emdogain–periodontal regeneration based on biomimicry. Clin. Oral Investig. 2000, 4, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Bosshardt, D.D. Biological mediators and periodontal regeneration: A review of enamel matrix proteins at the cellular and molecular levels. J. Clin. Periodontol. 2008, 35, 87–105. [Google Scholar] [CrossRef] [PubMed]
- Kusumoto, T.; Yin, D.; Zhang, H.; Chen, L.; Nishizaki, H.; Komasa, Y.; Okazaki, J.; Komasa, S. Evaluation of the osteointegration of a novel alkali-treated implant system in vivo. J. Hard Tissue Biol. 2017, 26, 355–360. [Google Scholar] [CrossRef]
- Xu, L.C.; Siedlecki, C.A. Effects of surface wettability and contact time on protein adhesion to biomaterial surfaces. Biomaterials 2007, 28, 3273–3283. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Chu, P.K.; Zhang, Y.; Wu, Z. Antibacterial coatings on titanium implants. J. Biomed. Mater. Res. B Appl. Biomater. 2009, 91, 470–480. [Google Scholar] [CrossRef] [PubMed]
- Tashiro, Y.; Komasa, S.; Miyake, A.; Nishizaki, H.; Okazaki, J. Analysis of titania nanosheet adsorption behavior using a quartz crystal microbalance sensor. Adv. Mater. Sci. Eng. 2018. [Google Scholar] [CrossRef]
- Dowling, D.P.; Miller, I.S.; Ardhaoui, M.; Gallagher, W.M. Effect of surface wettability and topography on the adhesion of osteosarcoma cells on plasma-modified polystyrene. J. Biomater. Appl. 2011, 26, 327–347. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Igarashi, T.; Okumori, N.; Igarashi, T.; Maetani, T.; Liu, B.; Yoshinari, M. Influence of surface wettability on competitive protein adsorption and initial attachment of osteoblasts. Biomed. Mater. 2009, 4, 045002. [Google Scholar] [CrossRef] [PubMed]
- Du, C.; Schneider, G.B.; Zaharias, R.; Abbott, C.; Seabold, D.; Stanford, C.; Moradian-Oldak, J. Apatite/amelogenin coating on titanium promotes osteogenic gene expression. J. Dent. Res. 2005, 84, 1070–1074. [Google Scholar] [CrossRef] [PubMed]
- Kodama, H.A.; Amagai, Y.; Sudo, H.; Kasai, S.; Yamamoto, S. Establishment of a clonal osteogenic cell line from newborn mouse calvaria. Jpn. J. Oral Biol. 1981, 23, 899–901. [Google Scholar] [CrossRef]
- Ayukawa, Y.; Takeshita, F.; Inoue, T.; Yoshinari, M.; Shimono, M.; Suetsugu, T.; Tanaka, T. An immunoelectron microscopic localization of noncollagenous bone proteins (osteocalcin and osteopontin) at the bone–titanium interface of rat tibiae. J. Biomed. Mater. Res. 1998, 41, 111–119. [Google Scholar] [CrossRef]
- Guo, J.; Padilla, R.J.; Ambrose, W.; De Kok, I.J.; Cooper, L.F. The effect of hydrofluoric acid treatment of TiO2 grit blasted titanium implants on adherent osteoblast gene expression in vitro and in vivo. Biomaterials 2007, 28, 5418–5425. [Google Scholar] [CrossRef] [PubMed]
- Isa, Z.M.; Schneider, G.B.; Zaharias, R.; Seabold, D.; Stanford, C.M. Effects of fluoride-modified titanium surfaces on osteoblast proliferation and gene expression. Int. J. Oral Maxillofac. Implants 2006, 21, 203–211. [Google Scholar] [PubMed]
- Marinucci, L.; Balloni, S.; Becchetti, E.; Belcastro, S.; Guerra, M.; Calvitti, M.; Lull, C.; Calvi, E.M.; Locci, P. Effect of titanium surface roughness on human osteoblast proliferation and gene expression in vitro. Int. J. Oral Maxillofac. Implants 2006, 21, 719–725. [Google Scholar] [PubMed]
- Kawana, F.; Sawae, Y.; Sahara, T.; Tanaka, S.; Debari, K.; Shimizu, M.; Sasaki, T. Porcine enamel matrix derivative enhances trabecular bone regeneration during wound healing of injured rat femur. Anat. Rec. 2001, 264, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Shimizu-Ishiura, M.; Tanaka, S.; Lee, W.S.; Debari, K.; Sasaki, T. Effects of enamel matrix derivative to titanium implantation in rat femurs. J. Biomed. Mater. Res. 2002, 60, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Slavkin, H.C.; Bessem, C.; Fincham, A.G.; Bringas, P., Jr.; Santos, V.; Snead, M.L.; Zeichner-David, M. Human and mouse cementum proteins immunologically related to enamel proteins. Biochim. Biophys. Acta 1989, 991, 12–18. [Google Scholar] [CrossRef]
- Viswanathan, H.L.; Berry, J.E.; Foster, B.L.; Gibson, C.W.; Li, Y.; Kulkarni, W.B.; Snead, M.L.; Somerman, M.J. Amelogenin: A potential regulator of cementum-associated genes. J. Periodontol. 2003, 74, 1423–1431. [Google Scholar] [CrossRef] [PubMed]
- Hammarstrom, L. The role of enamel matrix proteins in the development of cementum and periodontal tissues. In Ciba Foundation Symposium 205-Dental Enamel; John Wiley & Sons, Ltd.: Somerset, NJ, USA, 2008; Volume 246, pp. 246–260. [Google Scholar]
- Brunette, D.M.; Chehroudi, B. The effects of the surface topography of micromachined titanium substrata on cell behavior in vitro and in vivo. J. Biomech. Eng. 1999, 121, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Sikavitsas, V.I.; van den Dolder, J.; Bancroft, G.N.; Jansen, J.A.; Mikos, A.G. Influence of the in vitro culture period on the in vivo performance of cell/titanium bone tissue-engineered constructs using a rat cranial critical size defect model. J. Biomed. Mater. Res. 2003, 67, 944–951. [Google Scholar] [CrossRef] [PubMed]
- Ferris, D.M.; Moodie, G.D.; Dimond, P.M.; Giorani, C.W.; Ehrlich, M.G.; Valentini, R.F. RGD-coated titanium implants stimulate increased bone formation in vivo. Biomaterials 1999, 20, 2323–2331. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Terada, C.; Komasa, S.; Kusumoto, T.; Kawazoe, T.; Okazaki, J. Effect of Amelogenin Coating of a Nano-Modified Titanium Surface on Bioactivity. Int. J. Mol. Sci. 2018, 19, 1274. https://doi.org/10.3390/ijms19051274
Terada C, Komasa S, Kusumoto T, Kawazoe T, Okazaki J. Effect of Amelogenin Coating of a Nano-Modified Titanium Surface on Bioactivity. International Journal of Molecular Sciences. 2018; 19(5):1274. https://doi.org/10.3390/ijms19051274
Chicago/Turabian StyleTerada, Chisato, Satoshi Komasa, Tetsuji Kusumoto, Takayoshi Kawazoe, and Joji Okazaki. 2018. "Effect of Amelogenin Coating of a Nano-Modified Titanium Surface on Bioactivity" International Journal of Molecular Sciences 19, no. 5: 1274. https://doi.org/10.3390/ijms19051274
APA StyleTerada, C., Komasa, S., Kusumoto, T., Kawazoe, T., & Okazaki, J. (2018). Effect of Amelogenin Coating of a Nano-Modified Titanium Surface on Bioactivity. International Journal of Molecular Sciences, 19(5), 1274. https://doi.org/10.3390/ijms19051274




