Radiation-Induced Gene Expression Changes in High and Low Grade Breast Cancer Cell Types
Abstract
1. Introduction
2. Results
2.1. Gene Expression and Pathway Analysis
2.1.1. 9 Gy-Treated Immortalized Breast Cell Lines
2.1.2. 23 Gy-Treated Immortalized Breast Cell Lines
2.1.3. 9 Gy-Treated Primary Breast Cell Cultures
2.1.4. 23 Gy-Treated Primary Breast Cell Cultures
2.2. Comparison between 9Gy and 23Gy
2.3. Comparison between 9-Gy IR Treated Immortalized and Primary Cell Lines
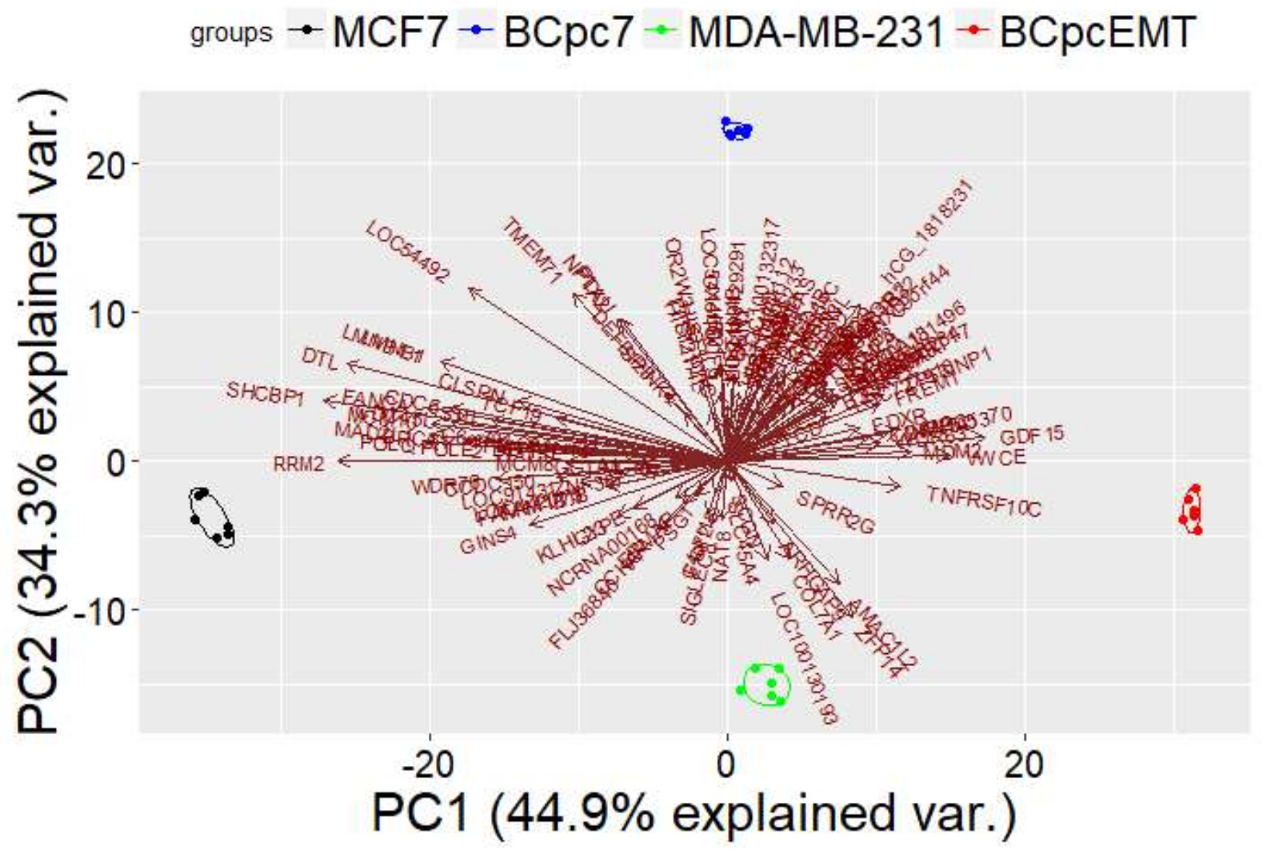
PCA Analysis
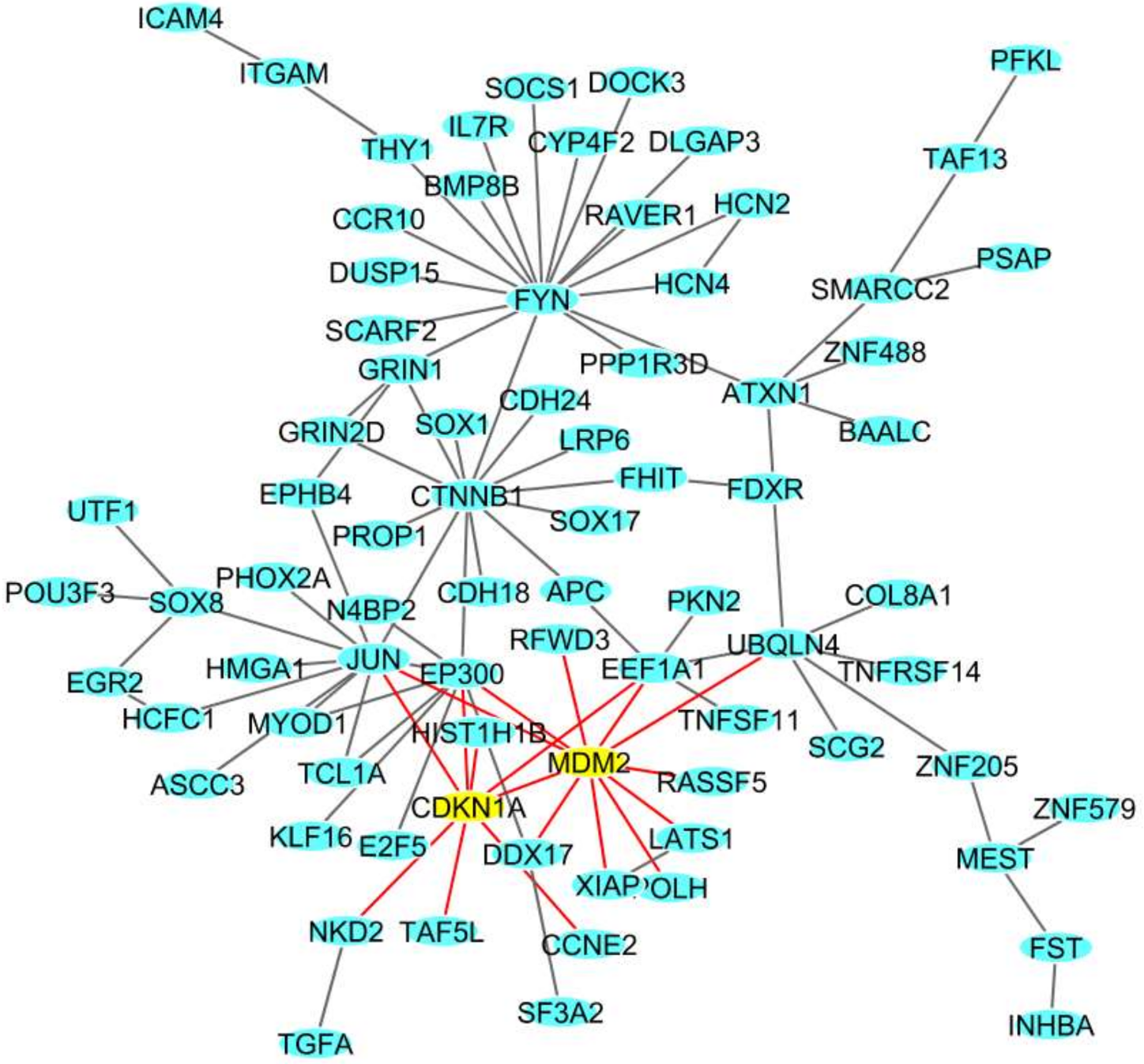
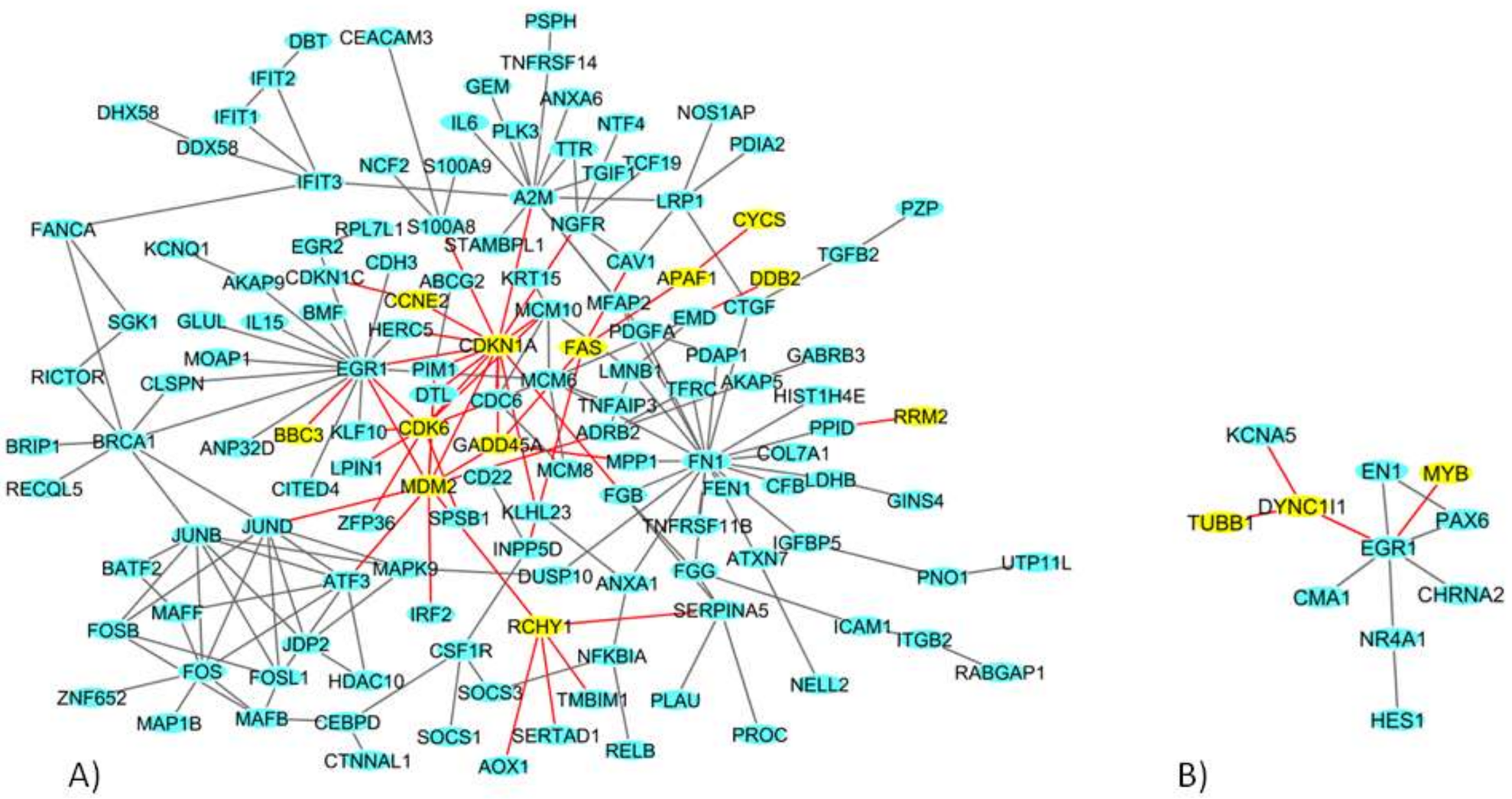
2.4. Network Analysis of 9 Gy DEGs in Immortalized and Primary Cells
3. Discussion
4. Materials and Methods
4.1. Cell Cultures and Irradiation
4.2. Gene Expression Analysis
4.3. Pathway Analysis
4.4. Network Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| RT | Radiation therapy |
| BC | Breast Cancer |
| HR | Hormone receptor |
| HER2 | Human epidermal growth factor receptor |
| IR | Ionizing radiation |
| ER | Estrogen receptor |
| PR | Progesteron receptor |
| IOERT | Intraoperative electron radiotherapy |
| DEG | Differentially expressed gene |
References
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Di Maggio, F.M.; Minafra, L.; Forte, G.I.; Cammarata, F.P.; Lio, D.; Messa, C.; Gilardi, M.C.; Bravatà, V. Portrait of inflammatory response to ionizing radiation treatment. J. Inflamm. 2015, 12, 14. [Google Scholar] [CrossRef] [PubMed]
- Formenti, S.C.; Demaria, S. Combining radiotherapy and cancer immunotherapy: A paradigm shift. J. Natl. Cancer Inst. 2013, 105, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Baskar, R.; Dai, J.; Wenlong, N.; Yeo, R.; Yeoh, K.W. Biological response of cancer cells to radiation treatment. Front. Mol. Biosci. 2014, 1, 24. [Google Scholar] [CrossRef] [PubMed]
- Sedlmayer, F.; Reitsamer, R.; Wenz, F.; Sperk, E.; Fussl, C.; Kaiser, J.; Ziegler, I.; Zehentmayr, F.; Deutschmann, H.; Kopp, P.; et al. Intraoperative radiotherapy (IORT) as boost in breast cancer. Radiat. Oncol. 2017, 12, 23. [Google Scholar] [CrossRef] [PubMed]
- Harris, E.E.R.; Small, W.J.R. Intraoperative Radiotherapy for Breast Cancer. Front. Oncol. 2017, 7, 317. [Google Scholar] [CrossRef] [PubMed]
- Bravatà, V.; Cammarata, F.P.; Forte, G.I.; Minafra, L. “Omics” of HER2-positive breast cancer. OMICS 2013, 17, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Bravatà, V.; Stefano, A.; Cammarata, F.P.; Minafra, L.; Russo, G.; Nicolosi, S.; Pulizzi, S.; Gelfi, C.; Gilardi, M.C.; Messa, C. Genotyping analysis and ¹⁸FDG uptake in breast cancer patients: A preliminary research. J. Exp. Clin. Cancer Res. 2013, 32, 23. [Google Scholar] [CrossRef] [PubMed]
- Lacroix, M.; Leclercq, G. Relevance of breast cancer cell lines as models for breast tumours: An update. Breast Cancer Res. Treat. 2004, 83, 249–289. [Google Scholar] [CrossRef] [PubMed]
- Forte, G.I.; Minafra, L.; Bravatà, V.; Cammarata, F.P.; Lamia, D.; Pisciotta, P.; Cirrone, G.A.P.; Cuttone, G.; Gilardi, M.C.; Russo, G. Radiogenomics: The utility in patient selection. Transl. Cancer Res. 2017, 6 (Suppl. S5), S852–S874. [Google Scholar] [CrossRef]
- Minafra, L.; Bravatà, V. Cell and molecular response to IORT treatment. Transl. Cancer Res. 2014, 3, 32–47. [Google Scholar]
- Bravatà, V.; Minafra, L.; Russo, G.; Forte, G.I.; Cammarata, F.P.; Ripamonti, M.; Casarino, C.; Augello, G.; Costantini, F.; Barbieri, G.; et al. High-dose Ionizing Radiation Regulates Gene Expression Changes in the MCF7 Breast Cancer Cell Line. Anticancer Res. 2015, 35, 2577–2591. [Google Scholar] [PubMed]
- Minafra, L.; Bravatà, V.; Russo, G.; Forte, G.I.; Cammarata, F.P.; Ripamonti, M.; Candiano, G.; Cervello, M.; Giallongo, A.; Perconti, G.; et al. Gene Expression Profiling of MCF10A Breast Epithelial Cells Exposed to IOERT. Anticancer Res. 2015, 35, 3223–3234. [Google Scholar] [PubMed]
- Burke, R.E.; Mira, J.G.; Datta, R.; Zava, D.T.; McGuire, W.L. Estrogen action following irradiation of human breast cancer cells. Cancer Res. 1978, 38, 2813–2817. [Google Scholar] [PubMed]
- Schmidt-Ullrich, R.K.; Valerie, K.; Chan, W.; Wazer, D.E.; Lin, P.S. Expression of oestrogen receptor and transforming growth factor-alpha in MCF7 cells after exposure to fractionated irradiation. Int. J. Radiat. Biol. 1992, 61, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Ullrich, R.K.; Valerie, K.C.; Chan, W.; McWilliams, D. Altered expression of epidermal growth factor receptor and estrogen receptor in MCF7 cells after single and repeated radiation exposures. Int. J. Radiat. Oncol. Biol. Phys. 1994, 29, 813–819. [Google Scholar] [CrossRef]
- Paulsen, G.H.U.; Strickert, T.; Marthinsen, A.B.L.; Lundgren, S. Changes in radiation sensitivity and steroid receptor content introduced by hormonal agents and ionizing radiation in breast cancer cells in vitro. Acta Oncol. 1996, 35, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.A.; Bae, S.S.; Fernandes, A.; Wu, J.; Muschel, R.J.; McKenna, W.G.; Birnbaum, M.J.; Bernhard, E.J. Selective inhibition of Ras, phosphoinositide 3 kinase, and Akt isoforms increases the radiosensitivity of human carcinoma cell lines. Cancer Res. 2005, 65, 7902–7910. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, J.; Chen, X.; Liu, T.; He, W.; Chen, Y.; Zeng, X. Molecular machinery of autophagy and its implication in cancer. Am. J. Med. Sci. 2012, 343, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Affolter, A.; Fruth, K.; Brochhausen, C.; Schmidtmann, I.; Mann, W.J.; Brieger, J. Activation of mitogen-activated protein kinase extracellular signal-related kinase in head and neck squamous cell carcinomas after irradiation as part of a rescue mechanism. Head Neck 2011, 33, 1448–1457. [Google Scholar] [CrossRef] [PubMed]
- Zingg, D.; Riesterer, O.; Fabbro, D.; Glanzmann, C.; Bodis, S.; Pruschy, M. Differential activation of the phosphatidylinositol 3′-kinase/Akt survival pathway by ionizing radiation in tumor and primary endothelial cells. Cancer Res. 2004, 64, 5398–5406. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Toillon, R.A.; Magné, N.; Laïos, I.; Castadot, P.; Kinnaert, E.; van Houtte, P.; Desmedt, C.; Leclercq, G.; Lacroix, M. Estrogens decrease gamma-ray-induced senescence and maintain cell cycle progression in breast cancer cells independently of p53. Int. J. Radiat. Oncol. Biol. Phys. 2007, 67, 1187–1200. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Shu, Y.; Zhou, H.; Zhang, W.; Wang, H. Radiogenomics helps to achieve personalized therapy by evaluating patient responses to radiation treatment. Carcinogenesis 2015, 36, 307–317. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rappaport, N.; Fishilevich, S.; Nudel, R.; Twik, M.; Belinky, F.; Plaschkes, I.; Stein, T.I.; Cohen, D.; Oz-Levi, D.; Safran, M.; et al. Rational confederation of genes and diseases: NGS interpretation via GeneCards, MalaCards and VarElect. Biomed. Eng. Online 2017, 16 (Suppl. S1), 72. [Google Scholar] [CrossRef] [PubMed]
- Luzhna, L.; Filkowski, J.; Kovalchuk, O. High and low dose radiation effects on mammary adenocarcinoma cells—An epigenetic connection. Oncoscience 2016, 3, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Porter, J.R.; Fisher, B.E.; Baranello, L.; Liu, J.C.; Kambach, D.M.; Nie, Z.; Koh, W.S.; Luo, J.; Stommel, J.M.; Levens, D.; et al. Global Inhibition with Specific Activation: How p53 and MYC Redistribute the Transcriptome in the DNA Double-Strand Break Response. Mol. Cell. 2017, 67, 1013–1025. [Google Scholar] [CrossRef] [PubMed]
- Bravatà, V.; Minafra, L.; Cammarata, F.P.; Pisciotta, P.; Lamia, D.; Marchese, V.; Manti, L.; Cirrone, G.A.P.; Gilardi, M.C.; Cuttone, G.; et al. Gene expression profiles induced by proton and electron irradiations in breast cancer cells. Br. J. Radiol. 2018. submitted. [Google Scholar]
- Bravatà, V.; Minafra, L.; Forte, G.I.; Cammarata, F.P.; Russo, G.; Di Maggio, F.M.; Augello, G.; Lio, D.; Gilardi, M.C. Cytokine profile of breast cell lines after different radiation doses. Int. J. Radiat. Biol. 2017, 93, 1217–1226. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lu, W.; Chen, D.; Boohaker, R.J.; Zhai, L.; Padmalayam, I.; Wennerberg, K.; Xu, B.; Zhang, W. KIFC1 is a novel potential therapeutic target for breast cancer. Cancer Biol. Ther. 2015, 16, 1316–1322. [Google Scholar] [CrossRef] [PubMed]
- Young, A.R.; Narita, M. SASP reflects senescence. EMBO Rep. 2009, 10, 228–230. [Google Scholar] [CrossRef] [PubMed]
- Minafra, L.; Bravatà, V.; Cammarata, F.P.; Di Maggio, F.M.; Forte, G.I. SASPects of Radiation Induced Senescence. Ann. Radiat. Ther. Oncol. 2017, 1, 1006. Available online: http://www.remedypublications.com/annals-of-radiation-therapy-and-oncology/articles/pdfs_folder/arto-v1-id1006.pdf (accessed on 4 April 2018).
- Camphausen, K.; Purow, B.; Sproull, M.; Scott, T.; Ozawa, T.; Deen, D.F.; Tofilon, P.J. Orthotopic growth of human glioma cells quantitatively and qualitatively influences radiation-induced changes in gene expression. Cancer Res. 2005, 65, 10389–10393. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Khodarev, N.N.; Park, J.O.; Yu, J.; Gupta, N.; Nodzenski, E.; Roizman, B.; Weichselbaum, R.R. Dose-dependent and independent temporal patterns of gene responses to ionizing radiation in normal and tumor cells and tumor xenografts. Proc. Natl. Acad. Sci. USA 2001, 98, 12665–12670. [Google Scholar] [CrossRef] [PubMed]
- Amundson, S.A.; Do, K.T.; Vinikoor, L.C.; Lee, R.A.; Koch-Paiz, C.A.; Ahn, J.; Reimers, M.; Chen, Y.; Scudiero, D.A.; Weinstein, J.N.; et al. Integrating global gene expression and radiation survival parameters across the 60 cell lines of the National Cancer Institute Anticancer Drug Screen. Cancer Res. 2008, 68, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Lü, X.; de la Peña, L.; Barker, C.; Camphausen, K.; Tofilon, P.J. Radiation-induced changes in gene expression involve recruitment of existing messenger RNAs to and away from polysomes. Cancer Res. 2006, 66, 1052–1061. [Google Scholar] [CrossRef] [PubMed]
- Essmann, F.; Engels, I.H.; Totzke, G.; Schulze-Osthoff, K.; Jänicke, R.U. Apoptosis resistance of MCF7 breast carcinoma cells to ionizing radiation is independent of p53 and cell cycle control but caused by the lack of caspase-3 and a caffeine-inhibitable event. Cancer Res. 2004, 64, 7065–7072. [Google Scholar] [CrossRef] [PubMed]
- Neve, R.M.; Chin, K.; Fridlyand, J.; Yeh, J.; Baehner, F.L.; Fevr, T.; Clark, L.; Bayani, N.; Coppe, J.P.; Tong, F.; et al. A collection of breast cancer cell lines for the study of functionally distinct cancer subtypes. Cancer Cell 2006, 10, 515–527. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Cheng, H.; Bai, Z.; Li, J. Breast Cancer Cell Line Classification and Its Relevance with Breast Tumor Subtyping. J. Cancer 2017, 8, 3131–3141. [Google Scholar] [CrossRef] [PubMed]
- Minafra, L.; Norata, R.; Bravatà, V.; Viola, M.; Lupo, C.; Gelfi, C.; Messa, C. Unmasking epithelial-mesenchymal transition in a breast cancer primary culture: A study report. BMC Res. Notes 2012, 5, 343. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Minafra, L.; Bravatà, V.; Forte, G.I.; Cammarata, F.P.; Gilardi, M.C.; Messa, C. Gene expression profiling of epithelial-mesenchymal transition in primary breast cancer cell culture. Anticancer Res. 2014, 34, 2173–2183. [Google Scholar] [PubMed]
- Reis-Filho, J.S.; Pusztai, L. Gene expression profiling in breast cancer: Classification, prognostication, and prediction. Lancet 2011, 378, 1812–1823. [Google Scholar] [CrossRef]
- Langen, B.; Rudqvist, N.; Parris, T.Z.; Schüler, E.; Spetz, J.; Helou, K.; Forssell-Aronsson, E. Transcriptional response in normal mouse tissues after i.v. (211)At administration—Response related to absorbed dose, dose rate, and time. EJNMMI Res. 2015, 5, 1. [Google Scholar] [CrossRef] [PubMed]
- Baselet, B.; Belmans, N.; Coninx, E.; Lowe, D.; Janssen, A.; Michaux, A.; Tabury, K.; Raj, K.; Quintens, R.; Benotmane, M.A.; et al. Functional Gene Analysis Reveals Cell Cycle Changes and Inflammation in Endothelial Cells Irradiated with a Single X-ray Dose. Front. Pharmacol. 2017, 8, 213. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.A.; Chen, Y.J.; Chen, J.J. Testing for differentially expressed genes with microarray data. Nucleic Acids Res. 2003, 31, e52. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.; Domrachev, M.; Lash, A.E. Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res. 2002, 30, 207–210. [Google Scholar] [CrossRef] [PubMed]
- Fabregat, A.; Jupe, S.; Matthews, L.; Sidiropoulos, K.; Gillespie, M.; Garapati, P.; Haw, R.; Jassal, B.; Korninger, F.; May, B.; et al. The Reactome Pathway Knowledgebase. Nucleic Acids Res. 2018, 46, D649–D655. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, D. BioCarta. Biotech Softw. Internet Rep. 2001, 2, 117–120. [Google Scholar] [CrossRef]
- Ogata, H.; Goto, S.; Sato, K.; Fujibuchi, W.; Bono, H.; Kanehisa, M. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 1999, 27, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Cava, C.; Colaprico, A.; Bertoli, G.; Bontempi, G.; Mauri, G.; Castiglioni, I. How interacting pathways are regulated by miRNAs in breast cancer subtypes. BMC Bioinform. 2016, 17 (Suppl. S12), 348. [Google Scholar] [CrossRef] [PubMed]
- Cava, C.; Bertoli, G.; Castiglioni, I. Integrating genetics and epigenetics in breast cancer: Biological insights, experimental, computational methods and therapeutic potential. BMC Syst. Biol. 2015, 9, 62. [Google Scholar] [CrossRef] [PubMed]
- Lex, A.; Gehlenborg, N.; Strobelt, H.; Vuillemot, R.; Pfister, H. UpSet: Visualization of Intersecting Sets. IEEE Trans. Vis. Comput. Graph. 2014, 20, 1983–1992. [Google Scholar] [CrossRef] [PubMed]
- GGBIPLOT. Available online: http://github.com/vqv/ggbiplot (accessed on 1 February 2018).
- Colaprico, A.; Cava, C.; Bertoli, G.; Bontempi, G.; Castiglioni, I. Integrative analysis with Monte Carlo Cross-Validation reveals miRNAs regulating pathways cross-talk in aggressive breast cancer. BioMed Res. Int. 2015, 2015, 831314. [Google Scholar] [CrossRef] [PubMed]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef] [PubMed]
- Georgakilas, A.G.; Pavlopoulou, A.; Louka, M.; Nikitaki, Z.; Vorgias, C.E.; Bagos, P.G.; Michalopoulos, I. Emerging molecular networks common in ionizing radiation, immune and inflammatory responses by employing bioinformatics approaches. Cancer Lett. 2015, 368, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Cava, C.; Bertoli, G.; Colaprico, A.; Olsen, C.; Bontempi, G.; Castiglioni, I. Integration of multiple networks and pathways identifies cancer driver genes in pan-cancer analysis. BMC Genomics 2018, 19. [Google Scholar] [CrossRef] [PubMed]


| 9 Gy | 23 Gy | 9 ∩ 23 Gy | |||
|---|---|---|---|---|---|
| N° DEGs | N° PATHs | N° DEGs | N° PATHs | N° PATHs | |
| TOTAL MCF10A | 560 | 10 | 582 | 5 | 2 |
| TOTAL MCF7 | 777 | 19 | 505 | 37 | 7 |
| TOTAL MDA-MB-231 | 416 | 14 | 627 | 14 | 14 |
| MCF10A ∩ MCF7 | 53 | 0 | 36 | 3 | |
| MCF10A ∩ MDA-MB-231 | 20 | 0 | 25 | 0 | |
| MCF7 ∩ MDA-MB-231 | 32 | 0 | 47 | 12 | |
| MCF10A ∩ MCF7 ∩ MDA-MB-231 | 7 | 0 | 7 | 0 | |
| MCF10A-specific | 480 | 10 | 514 | 2 | |
| MCF7-specific | 685 | 19 | 415 | 22 | |
| MDA-MB-231-specific | 357 | 14 | 548 | 2 | |
| TOTAL HMEC | 435 | 17 | 658 | 54 | 16 |
| TOTAL BCpc7 | 527 | 68 | 621 | 62 | 58 |
| TOTAL BCpcEMT | 687 | 43 | 753 | 35 | 33 |
| HMEC ∩ BCpc7 | 23 | 1 | 68 | 21 | |
| HMEC ∩ BCpcEMT | 73 | 0 | 79 | 0 | |
| BCpc7 ∩ BCpcEMT | 194 | 27 | 101 | 2 | |
| HMEC ∩ BCpc7 ∩ BCpcEMT | 84 | 15 | 203 | 33 | |
| HMEC-specific | 255 | 1 | 308 | 0 | |
| BCpc7-specific | 226 | 25 | 249 | 6 | |
| BCpcEMT-specific | 336 | 1 | 370 | 0 | |
| MCF10A ∩ HMEC | 11 | 1 | |||
| MCF7 ∩ BCpc7 | 64 | 6 | |||
| MDA-MB-231 ∩ BCpcEMT | 59 | 9 | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bravatà, V.; Cava, C.; Minafra, L.; Cammarata, F.P.; Russo, G.; Gilardi, M.C.; Castiglioni, I.; Forte, G.I. Radiation-Induced Gene Expression Changes in High and Low Grade Breast Cancer Cell Types. Int. J. Mol. Sci. 2018, 19, 1084. https://doi.org/10.3390/ijms19041084
Bravatà V, Cava C, Minafra L, Cammarata FP, Russo G, Gilardi MC, Castiglioni I, Forte GI. Radiation-Induced Gene Expression Changes in High and Low Grade Breast Cancer Cell Types. International Journal of Molecular Sciences. 2018; 19(4):1084. https://doi.org/10.3390/ijms19041084
Chicago/Turabian StyleBravatà, Valentina, Claudia Cava, Luigi Minafra, Francesco Paolo Cammarata, Giorgio Russo, Maria Carla Gilardi, Isabella Castiglioni, and Giusi Irma Forte. 2018. "Radiation-Induced Gene Expression Changes in High and Low Grade Breast Cancer Cell Types" International Journal of Molecular Sciences 19, no. 4: 1084. https://doi.org/10.3390/ijms19041084
APA StyleBravatà, V., Cava, C., Minafra, L., Cammarata, F. P., Russo, G., Gilardi, M. C., Castiglioni, I., & Forte, G. I. (2018). Radiation-Induced Gene Expression Changes in High and Low Grade Breast Cancer Cell Types. International Journal of Molecular Sciences, 19(4), 1084. https://doi.org/10.3390/ijms19041084










