The Influence of High and Low Doses of Bisphenol A (BPA) on the Enteric Nervous System of the Porcine Ileum
Abstract
1. Introduction
2. Results
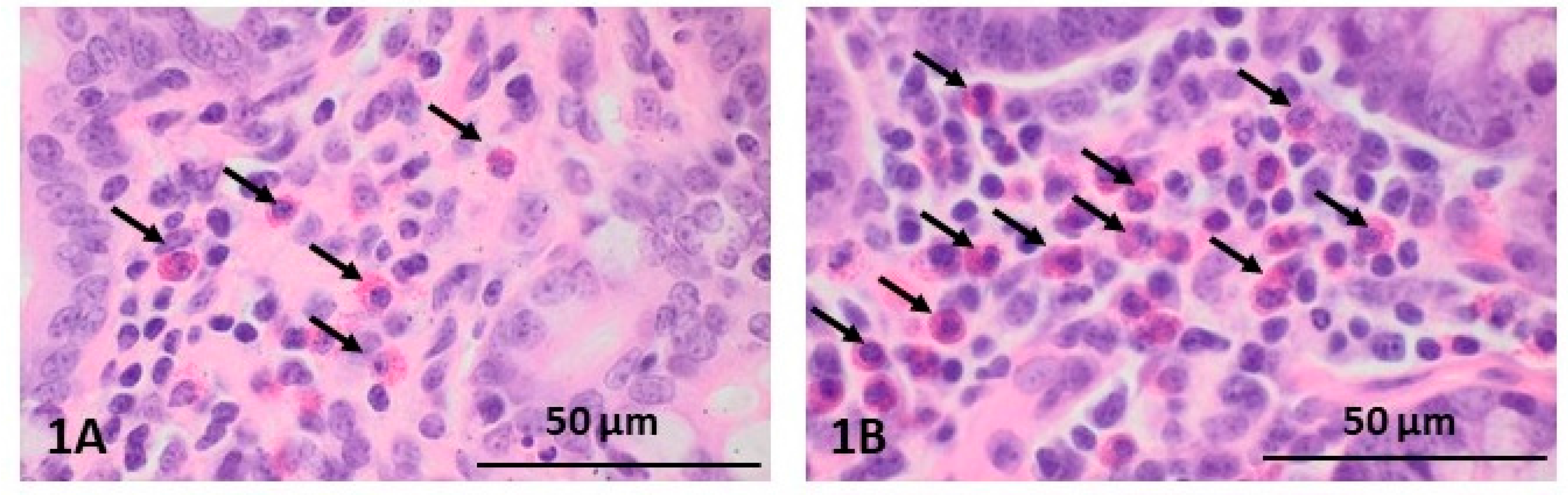
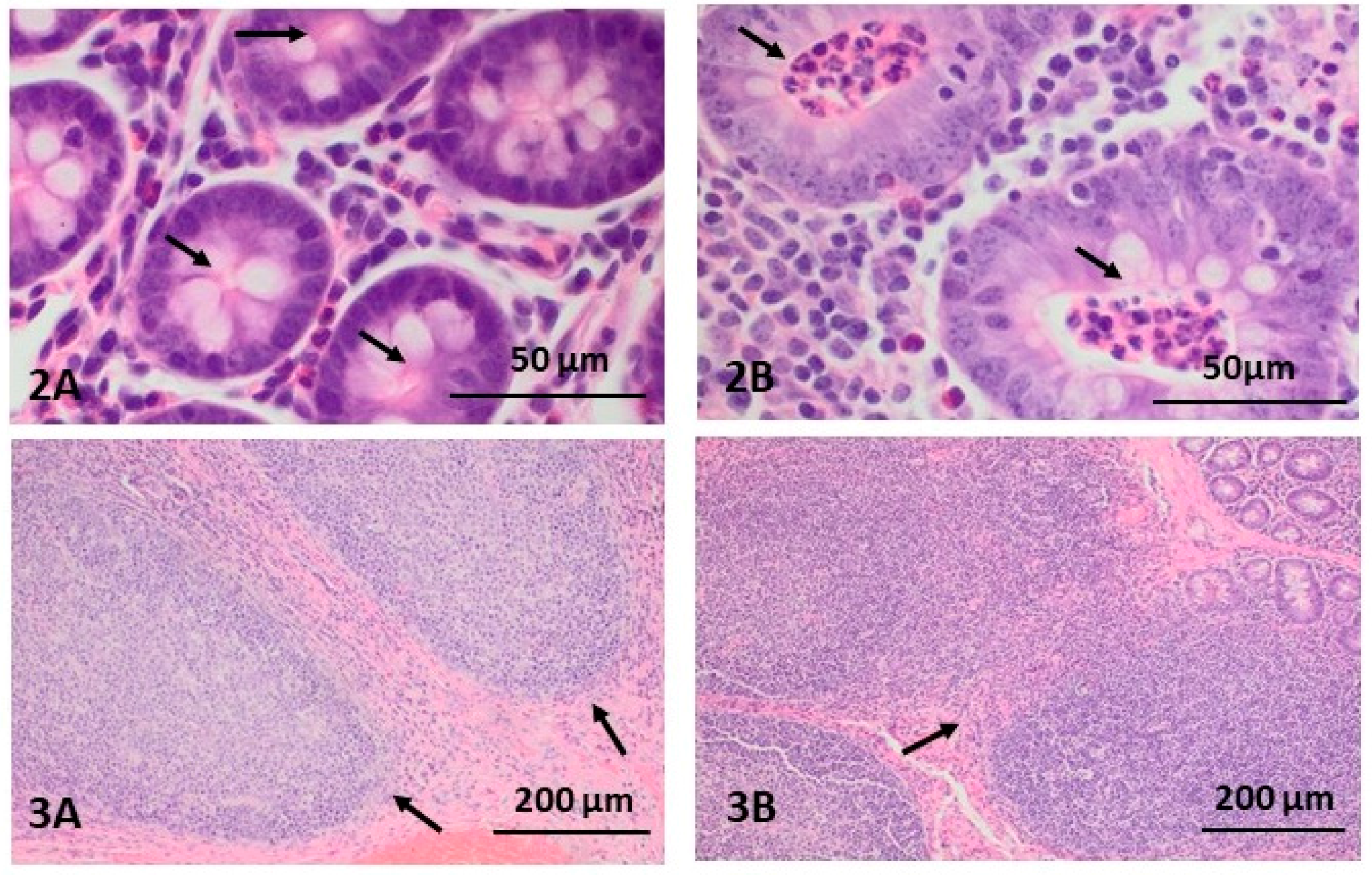
2.1. Morphological Changes
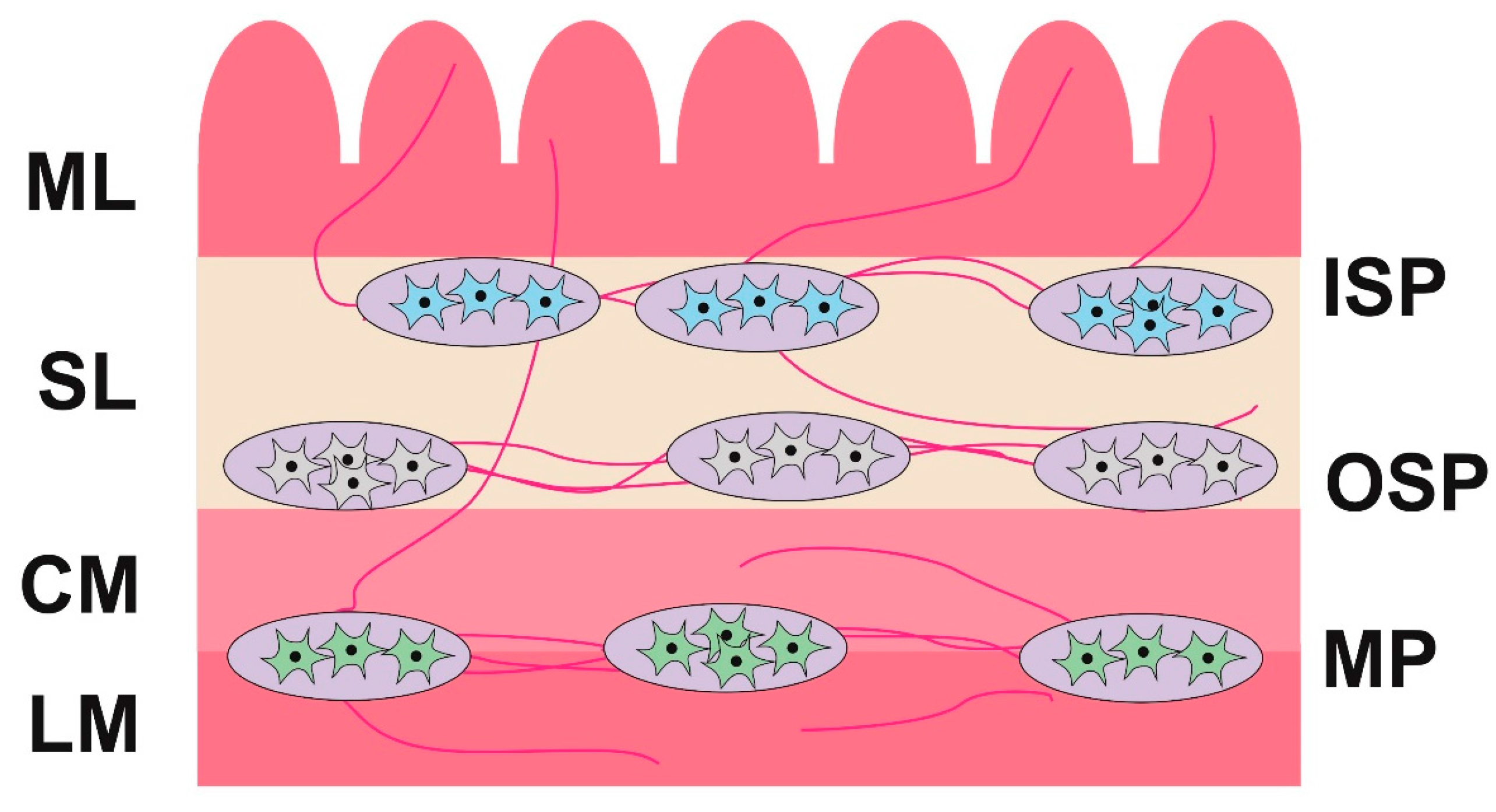
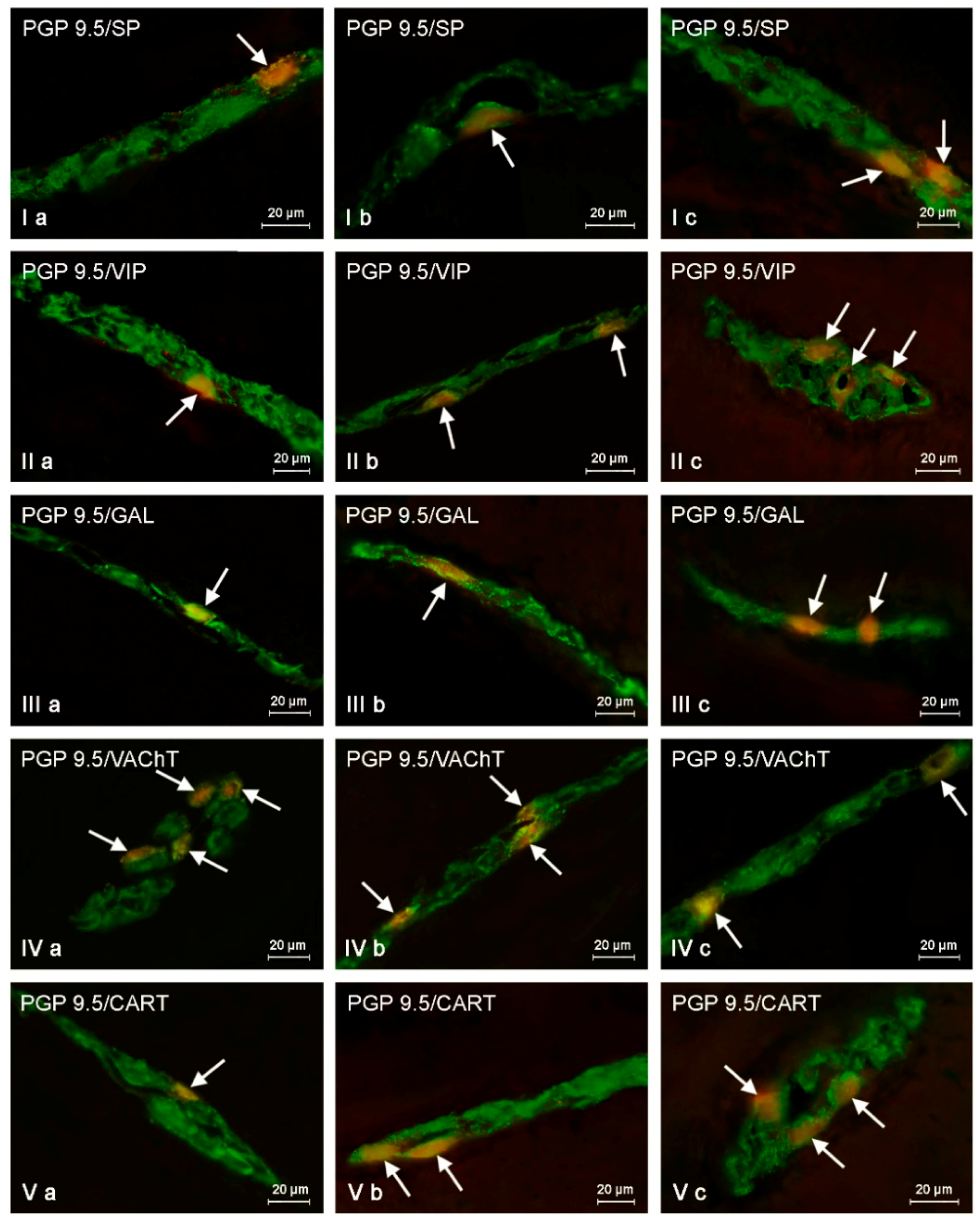
2.2. Changes in Neurochemistry in the ENS
2.2.1. Myenteric Plexus (MP)
2.2.2. Outer Submucous Plexus (OSP)
2.2.3. Inner Submucous Plexus (ISP)
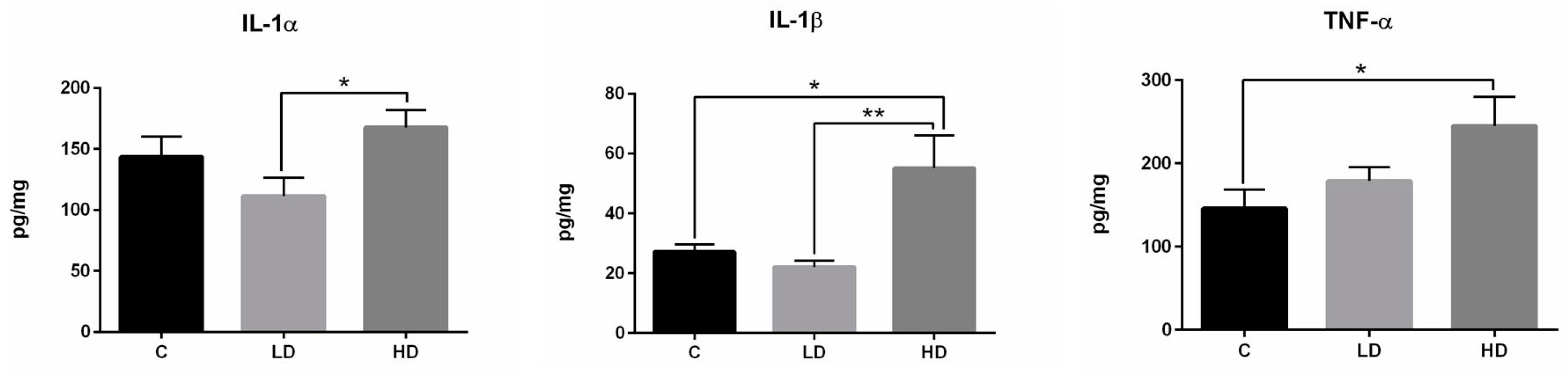
2.3. Changes in Cytokine Levels
3. Discussion
4. Materials and Methods
4.1. Experimental Animals and Tissue Collection
4.2. Determination of Neurochemical Characteristics of Enteric Neurons
4.3. Determination of Interleukin Levels by ELISA
4.4. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Suzuki, K.; Ishikawa, K.; Sugiyama, K.; Furuta, H.; Nishimura, F. Content and release of bisphenol A from polycarbonate dental products. Dent. Mater. J. 2000, 19, 389–395. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Vandenberg, L.N.; Hauser, R.; Marcus, M.; Olea, N.; Welshons, W.V. Human exposure to bisphenol A (BPA). Reprod. Toxicol. 2007, 24, 139–177. [Google Scholar] [CrossRef] [PubMed]
- Ikezuki, Y.; Tsutsumi, O.; Takai, Y.; Kamei, Y.; Taketani, Y. Determination of bisphenol A concentrations in human biological fluids reveals significant early prenatal exposure. Hum. Reprod. 2002, 17, 2839–2841. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Pierik, F.H.; Hauser, R.; Duty, S.; Angerer, J.; Park, M.M.; Burdorf, A.; Hofman, A.; Jaddoe, V.W.; Mackenbach, J.P.; et al. Urinary metabolite concentrations of organophosphorous pesticides, bisphenol A, and phthalates among pregnant women in Rotterdam, The Netherlands: The Generation R study. Environ. Res. 2008, 108, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Vandenberg, L.N.; Chahoud, I.; Heindel, J.J.; Padmanabhan, V.; Paumgartten, F.J.; Schoenfelder, G. Urinary, circulating, and tissue biomonitoring studies indicate widespread exposure to bisphenol A. Environ Health Perspect. 2010, 118, 1055–1070. [Google Scholar] [CrossRef] [PubMed]
- MacLusky, N.J.; Hajszan, T.; Leranth, C. The environmental estrogen bisphenol A inhibits estradiol-induced hippocampal synaptogenesis. Environ. Health Perspect. 2005, 113, 675–679. [Google Scholar] [CrossRef] [PubMed]
- Braniste, V.; Audebert, M.; Zalko, D.; Houdeau, E. Bisphenol A in the gut: Another break in the wall? In Multi-System Endocrine Disruption, 2nd ed.; Bourguignon, J.P., Jégou, B., Kerdelhué, B., Toppari, J., Christen, Y., Eds.; Springer-Verlag: Berlin/Heidelberg, Germany; New York, NY, USA, 2011; pp. 127–144. ISBN 978-3-642-22774-5. [Google Scholar]
- Savabieasfahani, M.; Kannan, K.; Astapova, O.; Evans, N.P.; Padmanabhan, V. Developmental programming: Differential effects of prenatal exposure to bisphenol-A or methoxychlor on reproductive function. Endocrinology 2006, 147, 5956–5966. [Google Scholar] [CrossRef] [PubMed]
- Hugo, E.R.; Brandebourg, T.D.; Woo, J.G.; Loftus, J.; Alexander, J.W.; Ben-Jonathan, N. Bisphenol A at environmentally relevant doses inhibits adiponectin release from human adipose tissue explants and adipocytes. Environ. Health. Perspect. 2008, 116, 1642–1647. [Google Scholar] [CrossRef] [PubMed]
- Maffini, M.V.; Rubin, B.S.; Sonnenschein, C.; Soto, A.M. Endocrine disruptors and reproductive health: The case of bisphenol-A. Mol. Cell. Endocrinol. 2006, 254–255, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Aloisi, A.M.; Della Seta, D.; Rendo, C.; Ceccarelli, I.; Scaramuzzino, A.; Farabollini, F. Exposure to the estrogenic pollutant bisphenol A affects pain behavior induced by subcutaneous formalin injection in male and female rats. Brain Res. 2002, 937, 1–7. [Google Scholar] [CrossRef]
- Wang, W.; Wang, J.; Wang, Q.; Wu, W.; Huan, F.; Xiao, H. Bisphenol A modulates calcium currents and intracellular calcium concentration in rat dorsal root ganglion neurons. J. Membr. Biol. 2013, 246, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Itoh, K.; Dai, H.; Han, L.; Wang, X.; Kato, S.; Sugimoto, T.; Fushiki, S. Prenatal and lactational exposure to low-doses of bisphenol A alters adult mice behavior. Brain Dev. 2012, 34, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Viberg, H.; Fredriksson, A.; Buratovic, S.; Eriksson, P. Dose-dependent behavioral disturbances after a single neonatal Bisphenol A dose. Toxicology 2011, 290, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Tai, F.; Song, Z.; Wu, R.; Zhang, X.; He, F. Pubertal exposure to bisphenol A disrupts behavior in adult C57BL/6J mice. Environ. Toxicol. Pharmacol. 2011, 31, 88–99. [Google Scholar] [CrossRef] [PubMed]
- Hajszan, T.; Leranth, C. Bisphenol A interferes with synaptic remodeling. Front. Neuroendocrinol. 2010, 31, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Viberg, H.; Lee, I. A single exposure to bisphenol A alters the levels of important neuroproteins in adult male and female mice. Neurotoxicology 2012, 33, 1390–1395. [Google Scholar] [CrossRef] [PubMed]
- Yokosuka, M.; Ohtani-Kaneko, R.; Yamashita, K.; Muraoka, D.; Kuroda, Y.; Watanabe, C. Estrogen and environmental estrogenic chemicals exert developmental effects on rat hypothalamic neurons and glias. Toxicol. Vitro 2008, 22, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Seki, S.; Aoki, M.; Hosokawa, T.; Saito, T.; Masuma, R.; Komori, M.; Kurasaki, M. Bisphenol-A suppresses neurite extension due to inhibition of phosphorylation of mitogen-activated protein kinase in PC12 cells. Chem. Biol. Interact. 2011, 194, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Sawai, C.; Anderson, K.; Walser-Kuntz, D. Effect of bisphenol A on murine immune function: Modulation of interferon-gamma, IgG2a, and disease symptoms in NZB X NZW F1 mice. Environ. Health Perspect. 2003, 111, 1883–1887. [Google Scholar] [CrossRef] [PubMed]
- Sakazaki, H.; Ueno, H.; Nakamuro, K. Estrogen receptor alpha in mouse splenic lymphocytes: Possible involvement in immunity. Toxicol. Lett. 2002, 133, 221–229. [Google Scholar] [CrossRef]
- Segura, J.J.; Jiménez-Rubio, A.; Pulgar, R.; Olea, N.; Guerrero, J.M.; Calvo, J.R. In vitro effect of the resin component bisphenol A on substrate adherence capacity of macrophages. J. Endod. 1999, 25, 341–344. [Google Scholar] [CrossRef]
- Rogers, J.A.; Metz, L.; Yong, V.W. Review: Endocrine disrupting chemicals and immune responses: A focus on bisphenol-A and its potential mechanisms. Mol. Immunol. 2013, 53, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; An, B.S.; Yang, H.; Jeung, E.B. Effects of octylphenol and bisphenol A on the expression of calcium transport genes in the mouse duodenum and kidney during pregnancy. Toxicology 2013, 303, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Harnish, D.C.; Albert, L.M.; Leathurby, Y.; Eckert, A.M.; Ciarletta, A.; Kasaian, M.; Keith, J.C., Jr. Beneficial effects of estrogen treatment in the HLA-B27 transgenic rat model of inflammatory bowel disease. Am. J. Physiol. Gastrointest. Liver Physiol. 2004, 286, G118–G125. [Google Scholar] [CrossRef] [PubMed]
- Houdeau, E.; Moriez, R.; Leveque, M.; Salvador-Cartier, C.; Waget, A.; Leng, L.; Bueno, L.; Bucala, R.; Fioramonti, J. Sex steroid regulation of macrophage migration inhibitory factor in normal and inflamed colon in the female rat. Gastroenterology 2007, 132, 982–993. [Google Scholar] [CrossRef] [PubMed]
- Gonkowski, S. Substance P as a neuronal factor in the enteric nervous system of the porcine descending colon in physiological conditions and during selected pathogenic processes. Biofactors 2013, 39, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Makowska, K.; Gonkowski, S.; Zielonka, L.; Dabrowski, M.; Calka, J. T2 Toxin-Induced Changes in Cocaine- and Amphetamine-Regulated Transcript (CART)-Like Immunoreactivity in the Enteric Nervous System Within Selected Fragments of the Porcine Digestive Tract. Neurotox. Res. 2017, 3, 136–147. [Google Scholar] [CrossRef] [PubMed]
- Rogala, D.; Kulik-Kupka, K.; Spychala, A.; Sniezek, E.; Janicka, A.; Moskalenko, O. Bisfenol A—Niebezpieczny związek ukryty w tworzywach sztucznych. Probl. Hig. Epidemiol. 2016, 97, 213–219. [Google Scholar]
- Vandenberg, L.N. Exposure to bisphenol A in Canada: Invoking the precautionary principle. CMAJ 2011, 183, 1265–1270. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, T.P.; Eaglstein, W.H.; Davis, S.C.; Mertz, P. The pig as a model for human wound healing. Wound Repair Regen. 2001, 9, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.C.; Swindle, M.M. Preparation of swine for the laboratory. ILAR J. 2006, 47, 358–363. [Google Scholar] [CrossRef] [PubMed]
- Litten-Brown, J.C.; Corson, A.M.; Clarke, L. Porcine models for the metabolic syndrome, digestive and bone disorders: A general overview. Animal 2010, 4, 899–920. [Google Scholar] [CrossRef] [PubMed]
- Verma, N.; Rettenmeier, A.W.; Schmitz-Spanke, S. Recent advances in the use of Sus scrofa (pig) as a model system for proteomic studies. Proteomics 2011, 11, 776–793. [Google Scholar] [CrossRef] [PubMed]
- Nezami, B.G.; Srinivasan, S. Enteric Nervous System in the Small Intestine: Pathophysiology and Clinical Implications. Curr. Gastroenterol. Rep. 2010, 12, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.R.; Timmermans, J.P. Lessons from the porcine enteric nervous system. Neurogastroenterol. Motil. 2004, 1, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Wojtkiewicz, J.; Rytel, L.; Makowska, K.; Gonkowski, S. Co-localization of zinc transporter 3 (ZnT3) with sensory neuromediators and/or neuromodulators in the enteric nervous system of the porcine esophagus. Biometals 2017, 30, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Wojtkiewicz, J.; Makowska, K.; Bejer-Olenska, E.; Gonkowski, S. Zinc transporter 3 (Znt3) as an active substance in the enteric nervous system of the porcine esophagus. J. Mol. Neurosci. 2017, 61, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Kaminska, B.; Landowski, P.; Majewski, M.; Gonkowski, S.; Dobosz, M.; Ismail, H.; Szlagatys-Sidorkiewicz, A.; Korzon, M.; Krzykowski, G. Enteric nervous system in patients after colectomy with drug-resistant ulcerative colitis. In Recent Advances in the Knowledge of Children Bowel Diseases, 2nd ed.; Korzon, M., Walkowiak, J., Kaminska, B., Eds.; A. Marszałek Press: Toruń, Poland, 2006; pp. 105–113. [Google Scholar]
- Oponowicz, A.; Kozłowska, A.; Gonkowski, S.; Godlewski, J.; Majewski, M. Changes in the distribution of cocaine- and amphetamine-regulated transcript-containing neural structures in the human colon affected by the neoplastic process. Int. J. Mol. Sci. 2018, 19, 414. [Google Scholar] [CrossRef] [PubMed]
- Bulc, M.; Gonkowski, S.; Całka, J. Expression of Cocaine and Amphetamine Regulated Transcript (CART) in the Porcine Intramural Neurons of Stomach in the Course of Experimentally Induced Diabetes Mellitus. J. Mol. Neurosci. 2015, 57, 376–385. [Google Scholar] [CrossRef] [PubMed]
- Furness, J.B. Types of neurons in the enteric nervous system. J. Auton. Nerv. Syst. 2000, 81, 87–96. [Google Scholar] [CrossRef]
- Crowe, R.; Kamm, M.A.; Burnstock, G.; Leonard-Jones, J.E. Peptide-containing neurons in different region of the submucous plexus of human sigmoid colon. Gastroenterology 1992, 102, 461–467. [Google Scholar] [CrossRef]
- Furness, J.B.; Callaghan, B.P.; Rivera, L.R.; Cho, H.J. The enteric nervous system and gastrointestinal innervation: Integrated local and central control. Adv. Exp. Med. Biol. 2014, 817, 39–71. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Itoh, K.; Sugimoto, T.; Fushiki, S. Prenatal exposure to bisphenol A affects adult murine neocortical structure. Neurosci. Lett. 2007, 420, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Wolstenholme, J.T.; Rissman, E.F.; Connelly, J.J. The role of bisphenol A in shaping the brain, epigenome and behavior. Horm. Behav. 2011, 59, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Kloosterman, W.P.; Plasterk, R.H. The diverse functions of microRNAs in animal development and disease. Dev. Cell 2006, 11, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Luo, Z.M.; Guo, X.M.; Su, D.F.; Liu, X. An updated role of microRNA-124 in central nervous system disorders: A review. Front. Cell. Neurosci. 2015, 9, 193. [Google Scholar] [CrossRef] [PubMed]
- Sempere, L.F.; Freemantle, S.; Pitha-Rowe, I.; Moss, E.; Dmitrovsky, E.; Ambros, V. Expression profiling of mammalian microRNAs uncovers a subset of brain-expressed microRNAs with possible roles in murine and human neuronal differentiation. Genome Biol. 2004, 5, R13. [Google Scholar] [CrossRef] [PubMed]
- Bak, M.; Silahtaroglu, A.; Møller, M.; Christensen, M.; Rath, M.F.; Skryabin, B.; Tommerup, N.; Kauppinen, S. MicroRNA expression in the adult mouse central nervous system. RNA 2008, 14, 432–444. [Google Scholar] [CrossRef] [PubMed]
- Derghal, A.; Djelloul, M.; Trouslard, J.; Mounien, L. An emerging role of micro-RNA in the effect of the endocrine disruptors. Front. Neurosci. 2016, 10, 318. [Google Scholar] [CrossRef] [PubMed]
- Inadera, H. Neurological effects of bisphenol A and its analogues. Int. J. Med. Sci. 2015, 12, 926–936. [Google Scholar] [CrossRef] [PubMed]
- Tilghman, S.L.; Bratton, M.R.; Segar, H.C.; Martin, E.C.; Rhodes, L.V.; Li, M.; McLachlan, J.A.; Wiese, T.E.; Nephew, K.P.; Burow, M.E. Endocrine disruptor regulation of microRNA expression in breast carcinoma cells. PLoS ONE 2012, 7, e32754. [Google Scholar] [CrossRef] [PubMed]
- Sicard, F.; Gayral, M.; Lulka, H.; Buscail, L.; Cordelier, P. Targeting miR-21 for the therapy of pancreatic cancer. Mol. Ther. J. Am. Soc. Gene Ther. 2013, 21, 986–994. [Google Scholar] [CrossRef] [PubMed]
- Veiga-Lopez, A.; Luense, L.J.; Christenson, L.K.; Padmanabhan, V. Developmental programming: Gestational bisphenol-A treatment alters trajectory of fetal ovarian gene expression. Endocrinology 2013, 154, 1873–1884. [Google Scholar] [CrossRef] [PubMed]
- Schratt, G.M.; Tuebing, F.; Nigh, E.A.; Kane, C.G.; Sabatini, M.E.; Kiebler, M.; Greenberg, M.E. A brain-specific microRNA regulates dendritic spine development. Nature 2006, 439, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Giraldez, A.J.; Cinalli, R.M.; Glasner, M.E.; Enright, A.J.; Thomson, J.M.; Baskerville, S.; Hammond, S.M.; Bartel, D.P.; Schier, A.F. MicroRNAs regulate brain morphogenesis in zebrafish. Science 2005, 308, 833–838. [Google Scholar] [CrossRef] [PubMed]
- Arciszewski, M.B.; Sand, E.; Ekblad, E. Vasoactive intestinal peptide rescues cultured rat myenteric neurons from lipopolysaccharide induced cell death. Regul. Pept. 2008, 146, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Cordero-Llana, O.; Rinaldi, F.; Brennan, P.A.; Wynick, D.; Caldwell, M.A. Galanin promotes neuronal differentiation from neural progenitor cells in vitro and contributes to the generation of new olfactory neurons in the adult mouse brain. Exp. Neurol. 2014, 256, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Holmes, F.E.; Mahoney, S.A.; Wynick, D. Use of genetically engineered transgenic mice to investigate the role of galanin in the peripheral nervous system after injury. Neuropeptides 2005, 39, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; MacTavish, D.; Kar, S.; Jhamandas, J.H. Galanin attenuates beta-amyloid (Abeta) toxicity in rat cholinergic basal forebrain neurons. Neurobiol. Dis. 2006, 21, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Schouten, J.W. Neuroprotection in traumatic brain injury: A complex struggle against the biology of nature. Curr. Opin. Crit. Care 2007, 13, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Lundstrom, L.; Elmquist, A.; Bartfai, T.; Langel, U. Galanin and its receptors in neurological disorders. Neuromolecular. Med. 2005, 7, 157–180. [Google Scholar] [CrossRef]
- Lallemend, F.; Lefebvre, P.P.; Hans, G.; Rigo, J.M.; Van de Water, T.R.; Moonen, G.; Malgrange, B. Substance P protects spiral ganglion neurons from apoptosis via PKC-Ca2+-MAPK/ERK pathways. J. Neurochem. 2003, 87, 508–521. [Google Scholar] [CrossRef] [PubMed]
- Amadoro, G.; Pieri, M.; Ciotti, M.T.; Carunchio, I.; Canu, N.; Calissano, P.; Zona, C.; Severini, C. Substance P provides neuroprotection in cerebellar granule cells through Akt and MAPK/Erk activation: Evidence for the involvement of the delayed rectifier potassium current. Neuropharmacology 2007, 52, 1366–1377. [Google Scholar] [CrossRef] [PubMed]
- Mao, P.; Meshul, C.K.; Thuillier, P.; Reddy, P.H. Neurotransmitter CART as a New Therapeutic Candidate for Parkinson’s Disease. Pharmaceuticals (Basel) 2013, 6, 108–123. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, S.; Maeda, T.; Kume, T.; Kochiyama, H.; Akaike, A.; Shimohama, S.; Kimura, J. Nicotine protects cultured cortical neurons against glutamate-induced cytotoxicity via α7-neuronal receptors and neuronal CNS receptors. Brain Res. 1997, 765, 135–140. [Google Scholar] [CrossRef]
- Nakamizo, T.; Kawamata, J.; Yamashita, H.; Kanki, R.; Kihara, T.; Sawada, H.; Akaike, A.; Shimohama, S. Stimulation of nicotinic acetylcholine receptor protects motor neurons. Biochem. Biophys. Res. Commun. 2005, 330, 1285–1289. [Google Scholar] [CrossRef] [PubMed]
- Thompson, S.A.; Smith, O.; Linn, D.M.; Linn, C.L. Acetylcholine neuroprotection against glutamate-induced excitotoxicity in adult pig retinal ganglion cells is partially mediated through alpha4 nAChRs. Exp. Eye Res. 2006, 83, 1135–1145. [Google Scholar] [CrossRef] [PubMed]
- The, F.; Cailotto, C.; van der Vliet, J.; de Jonge, W.J.; Bennink, R.J.; Buijs, R.M.; Boeckxstaens, G.E. Central activation of the cholinergic anti-inflammatory pathway reduces surgical inflammation in experimental post-operative ileus. Br. J. Pharmacol. 2011, 163, 1007–1016. [Google Scholar] [CrossRef] [PubMed]
- Kolgazi, M.; Uslu, U.; Yuksel, M.; Velioglu-Ogunc, A.; Ercan, F.; Alican, I. The role of cholinergic anti-inflammatory pathway in acetic acid-induced colonic inflammation in the rat. Chem. Biol. Interact. 2013, 205, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Pepeu, G.; Grazia Giovannini, M. The fate of the brain cholinergic neurons in neurodegenerative diseases. Brain Res. 2017, 1670, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Pidsudko, Z.; Wasowicz, K.; Kaleczyc, J.; Majewski, M.; Lakomy, M. Proliferative enteropathy (PPE)-induced changes in the expression of DBH, VAChT and NOS in the neurons of intramural ganglia of the porcine ileum. Vet. Med. 2008, 53, 533–542. [Google Scholar] [CrossRef]
- Kaleczyc, J.; Klimczuk, M.; Franke-Radowiecka, A.; Sienkiewicz, W.; Majewski, M.; Łakomy, M. The distribution and chemical coding of intramural neurons supplying the porcine stomach—The study on normal pigs and on animals suffering from swine dysentery. Anat. Histol. Embryol. 2007, 36, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Park, J.; Bui, P.T.C.; Choi, K.; Gye, M.C.; Hong, Y.C.; Kim, J.H.; Lee, Y.J. Bisphenol A induces COX-2 through the mitogen-activated protein kinase pathway and is associated with levels of inflammation-related markers in elderly populations. Environ. Res. 2017, 158, 490–498. [Google Scholar] [CrossRef] [PubMed]
- Sugita-Konishi, Y.; Shimura, S.; Nishikawa, T.; Sunaga, F.; Naito, H.; Suzuki, Y. Effect of Bisphenol A on non-specific immunodefenses against non-pathogenic Escherichia coli. Toxicol. Lett. 2003, 136, 217–227. [Google Scholar] [CrossRef]
- Mantyh, C.R.; Vigna, S.R.; Maggio, J.E.; Mantyh, P.W.; Bollinger, R.R.; Pappas, T.N. Substance P binding sites on intestinal lymphoid aggregates and blood vessels in inflammatory bowel disease correspond to authentic NK-1 receptors. Neurosci. Lett. 1994, 178, 255–259. [Google Scholar] [CrossRef]
- Vota, D.; Aguero, M.; Grasso, E.; Hauk, V.; Gallino, L.; Soczewski, E.; Pérez Leirós, C.; Ramhorst, R. Progesterone and VIP cross-talk enhances phagocytosis and anti-inflammatory profile in trophoblast-derived cells. Mol. Cell. Endocrinol. 2017, 443, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Koller, A.; Bianchini, R.; Schlager, S.; Münz, C.; Kofler, B.; Wiesmayr, S. The neuropeptide galanin modulates natural killer cell function. Neuropeptides 2017, 64, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Ganea, D.; Peng, X.; Jonakait, G.M. Galanin down-regulates microglial tumor necrosis factor-alpha production by a post-transcriptional mechanism. J. Neuroimmunol. 2003, 134, 52–60. [Google Scholar] [CrossRef]
- Lang, R.; Kofler, B. The galanin peptide family in inflammation. Neuropeptides 2011, 45, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Pant, J.; Ranjan, P.; Deshpande, S.B. Bisphenol A decreases atrial contractility involving NO-dependent G-cyclase signaling pathway. J. Appl. Toxicol. 2011, 31, 698–702. [Google Scholar] [CrossRef] [PubMed]
- Asano, S.; Tune, J.D.; Dick, G.M. Bisphenol A activates Maxi-K (K(Ca)1.1) channels in coronary smooth muscle. Br. J. Pharmacol. 2010, 160, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, K.; Tarafder, P.; Paul, G. Bisphenol A inhibits duodenal movement ex vivo of rat through nitric oxide-mediated soluble guanylyl cyclase and α-adrenergic signaling pathways. J. Appl. Toxicol. 2016, 36, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Van Geldre, L.A.; Lefebvre, R.A. Interaction of NO and VIP in gastrointestinal smooth muscle relaxation. Curr. Pharm. Des. 2004, 10, 2483–2497. [Google Scholar] [CrossRef] [PubMed]
- Ekblad, E.; Kuhar, M.; Wierup, N.; Sundler, F. Cocaine- and amphetamine-regulated transcript: Distribution and function in rat gastrointestinal tract. Neurogastroenterol. Motil. 2003, 15, 545–557. [Google Scholar] [CrossRef] [PubMed]
- Sternini, C.; Anselmi, L.; Guerrini, S.; Cervio, E.; Pham, T.; Balestra, B.; Vicini, R.; Baiardi, P.; D’agostino, G.L.; Tonini, M. Role of galanin receptor 1 in peristaltic activity in the guinea pig ileum. Neuroscience 2004, 125, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Lordal, M.; Johansson, C.; Hellström, C. Myoelectric pattern and effects on small bowel transit induced by the tachykinins neurokinin A, neurokinin B, substance P and neuropeptide K in the rat. Neurogastroenterol. Motil. 1993, 5, 33–40. [Google Scholar] [CrossRef]
- Roza, C.; Reeh, P.W. Substance P, calcitonin gene related peptide and PGE2 co-released from the mouse colon: A new model to study nociceptive and inflammatory responses in viscera, in vitro. Pain 2001, 93, 213–219. [Google Scholar] [CrossRef]
- Makowska, K.; Obremski, K.; Zielonka, L.; Gonkowski, S. The Influence of Low Doses of Zearalenone and T-2 Toxin on Calcitonin Gene Related Peptide-Like Immunoreactive (CGRP-LI) Neurons in the ENS of the Porcine Descending Colon. Toxins (Basel) 2017, 10, 98. [Google Scholar] [CrossRef] [PubMed]
- Chiocchetti, R.; Grandis, A.; Bombardi, C.; Lucchi, M.L.; Dal Lago, D.T.; Bortolami, R.; Furness, J.B. Extrinsic and intrinsic sources of calcitonin gene-related peptide immunoreactivity in the lamb ileum: A morphometric and neurochemical investigation. Cell Tissue Res. 2006, 323, 183–196. [Google Scholar] [CrossRef] [PubMed]
- Ishido, M.; Morita, M.; Oka, S.; Masuo, Y. Alteration of gene expression of G protein-coupled receptors in endocrine disruptors-caused hyperactive rats. Regul. Pept. 2005, 126, 145–153. [Google Scholar] [CrossRef] [PubMed]
- EFSA J. Opinion of the Scientific Panel on food additives, flavorings, processing aids and materials in contact with food on a request from the Commission to 2,2-bis(4-hydroxyphenyl) propane (Bisphenol A). EFSA J. 2006, 428, 1–75. [Google Scholar]
- Grob, K.; Gürtler, R.; Husøy, T.; Mennes, W.; Milana, M.R.; Penninks, A.; Roland, F.; Silano, V.; Smith, A.; Poças, M.D.F.T.; et al. Scientific Opinion on the risks to public health related to the presence of bisphenol A (BPA) in foodstuffs: Executive summary. EFSA J. 2015, 13, 3978–4599. [Google Scholar]
- Wojtkiewicz, J.; Rowniak, M.; Crayton, R.; Gonkowski, S.; Robak, A.; Zalecki, M.; Majewski, M.; Klimaschewski, L. Axotomy-induced changes in the chemical coding pattern of colon projecting calbindin-positive neurons in the inferior mesenteric ganglia of the pig. J. Mol. Neurosci. 2013, 5, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Obremski, K.; Podlasz, P.; Zmigrodzka, M.; Winnicka, A.; Wozny, M.; Brzuzan, P.; Jakimiuk, E.; Wojtacha, P.; Gajecka, M.; Zielonka, L.; Gajecki, M. The effect of T-2 toxin on percentages of CD4+, CD8+, CD4+ CD8+ and CD21+ lymphocytes, and mRNA expression levels of selected cytokines in porcine ileal Peyer’s patches. Pol. J. Vet. Sci. 2013, 16, 341–349. [Google Scholar] [CrossRef] [PubMed]
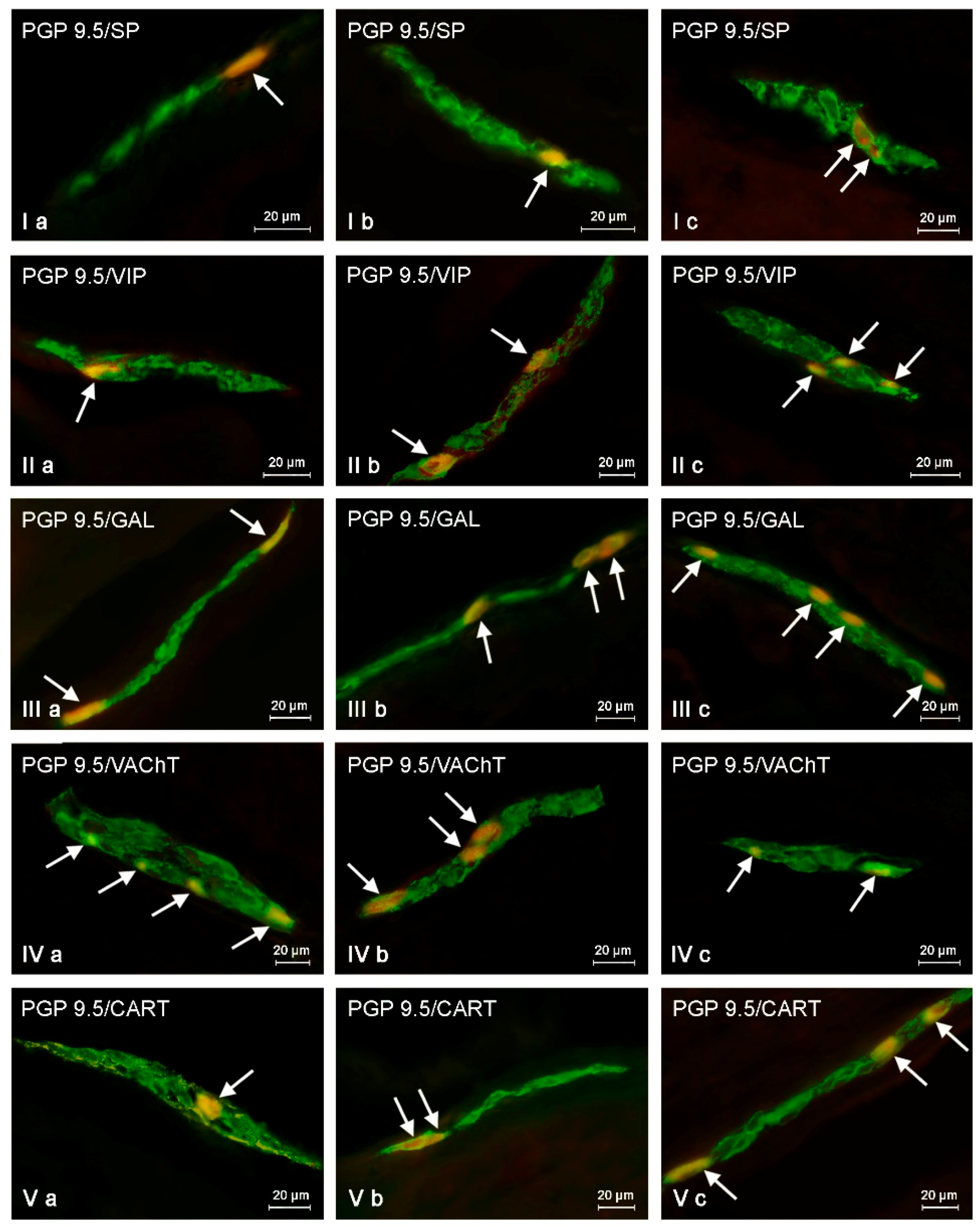
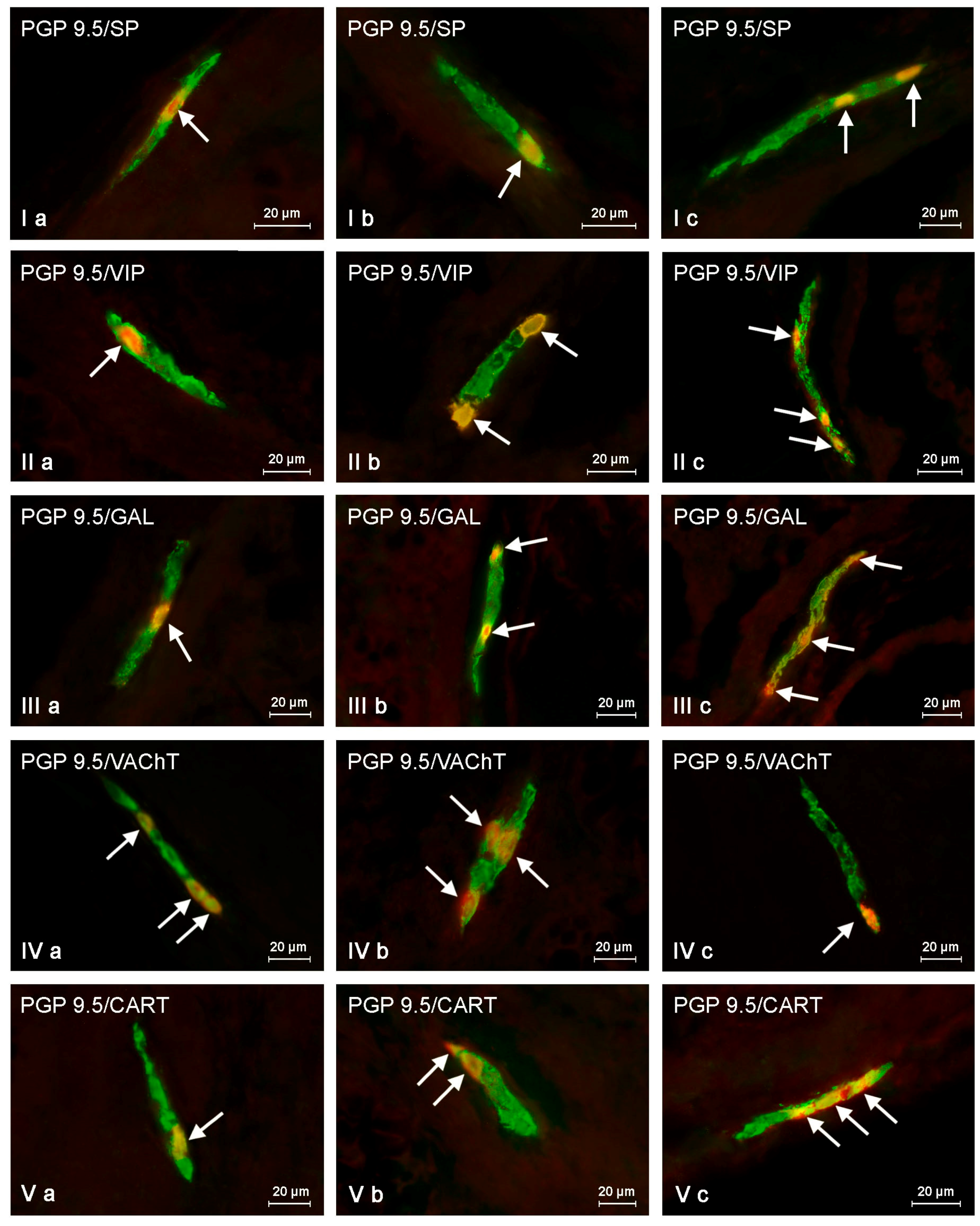
| Part of ENS | Animal Group | Active Neuronal Substances Studied During the Experiment | ||||
|---|---|---|---|---|---|---|
| VAChT | VIP | SP | GAL | CART | ||
| Myenteric plexus | C group | 18.35 ± 0.21 | 15.05 ± 0.17 | 11.01 ± 0.16 | 7.91 ± 0.04 | 7.58 ± 0.39 |
| LD group | 17.54 ± 0.24 * | 15.21 ± 0.35 | 11.03 ± 0.47 | 8.06 ± 0.32 | 10.70 ± 0.40 *** | |
| HD group | 13,67 ± 0.18 *** | 17.51 ± 0.04 *** | 15.15 ± 0.28 *** | 13.40 ± 0.39 *** | 17.09 ± 0.52 *** | |
| Outer submucous plexus | C group | 19.93 ± 0.10 | 10.57 ± 0.39 | 9.76 ± 0.12 | 9.60 ± 0.41 | 4.47 ± 0.11 |
| LD group | 18.24 ± 0.23 *** | 12.61 ± 0.11 *** | 10.39 ± 0.32 | 12.92 ± 0.42 *** | 5.29 ± 0.36 | |
| HD group | 14.70 ± 0.12 *** | 16.00 ± 0.08 *** | 17.73 ± 0.33 *** | 18.08 ± 0.35 *** | 9.65 ± 0.20 *** | |
| Inner submucous plexus | C group | 26.42 ± 0.38 | 7.64 ± 0.17 | 9.32 ± 0.37 | 14.14 ± 0.13 | 3.80 ± 0.16 |
| LD group | 25.21 ± 0.23 * | 10.85 ± 0.34 *** | 10.32 ± 0.31 | 18.17 ± 0.08 *** | 5.30 ± 0.24 *** | |
| HD group | 20.70 ± 0.11 *** | 17.80 ± 0.28 *** | 15.83 ± 0.42 *** | 22.72 ± 0.30 *** | 8.02 ± 0.04 *** | |
| Primary Antibody | ||||
| Antisera | Code | Host Species | Dilution | Supplier |
| PGP9.5 | 7863-2004 | Mouse | 1:1000 | Bio-Rad, Hercules, CA, USA |
| CART | H-003-61 | Rabbit | 1:16,000 | Phoenix Pharmaceuticals, Inc., Burlingame, CA, USA |
| GAL | AB2233 | Guinea Pig | 1:2000 | EMD Millipore, Burlington, MA, USA |
| SP | 8450-0505 | Rat | 1:1000 | Bio-Rad |
| VAChT | H-V007 | Rabbit | 1:2000 | Phoenix Pharmaceuticals |
| VIP | 9535-0204 | Mouse | 1:2000 | Bio-Rad |
| Secondary antibodies | ||||
| Reagent | Dilution | Supplier | ||
| AF 488 donkey anti-mouse IgG (H + L) | 1:1000 | Thermo Fisher Scientific, Waltham, MA, USA | ||
| AF 546 goat anti-guinea pig IgG (H + L) | 1:1000 | Thermo Fisher Scientific | ||
| AF 546 goat anti-rabbit IgG (H + L) | 1:1000 | Thermo Fisher Scientific | ||
| AF 546 goat anti-rat IgG (H + L) | 1:1000 | Thermo Fisher Scientific | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szymanska, K.; Makowska, K.; Gonkowski, S. The Influence of High and Low Doses of Bisphenol A (BPA) on the Enteric Nervous System of the Porcine Ileum. Int. J. Mol. Sci. 2018, 19, 917. https://doi.org/10.3390/ijms19030917
Szymanska K, Makowska K, Gonkowski S. The Influence of High and Low Doses of Bisphenol A (BPA) on the Enteric Nervous System of the Porcine Ileum. International Journal of Molecular Sciences. 2018; 19(3):917. https://doi.org/10.3390/ijms19030917
Chicago/Turabian StyleSzymanska, Kamila, Krystyna Makowska, and Slawomir Gonkowski. 2018. "The Influence of High and Low Doses of Bisphenol A (BPA) on the Enteric Nervous System of the Porcine Ileum" International Journal of Molecular Sciences 19, no. 3: 917. https://doi.org/10.3390/ijms19030917
APA StyleSzymanska, K., Makowska, K., & Gonkowski, S. (2018). The Influence of High and Low Doses of Bisphenol A (BPA) on the Enteric Nervous System of the Porcine Ileum. International Journal of Molecular Sciences, 19(3), 917. https://doi.org/10.3390/ijms19030917





