Optimization of Photosynthetic Productivity in Contrasting Environments by Regulons Controlling Plant Form and Function
Abstract
1. Introduction
2. Transcription Factors Co-Optimize Productivity and Stress Tolerance
2.1. CBFs/DREBs and Abiotic Stress Tolerance
2.2. Stress-Inducible Promoters: On-off Switches Minimize Yield Penalties
2.3. To Grow or Not to Grow When the Going Gets Tough
2.4. CBFs and the Phenotype of Cool-Grown Winter Annuals
2.5. Example of a Gene Capable of Orchestrating either Reduced or Accelerated Growth under Heat/Drought
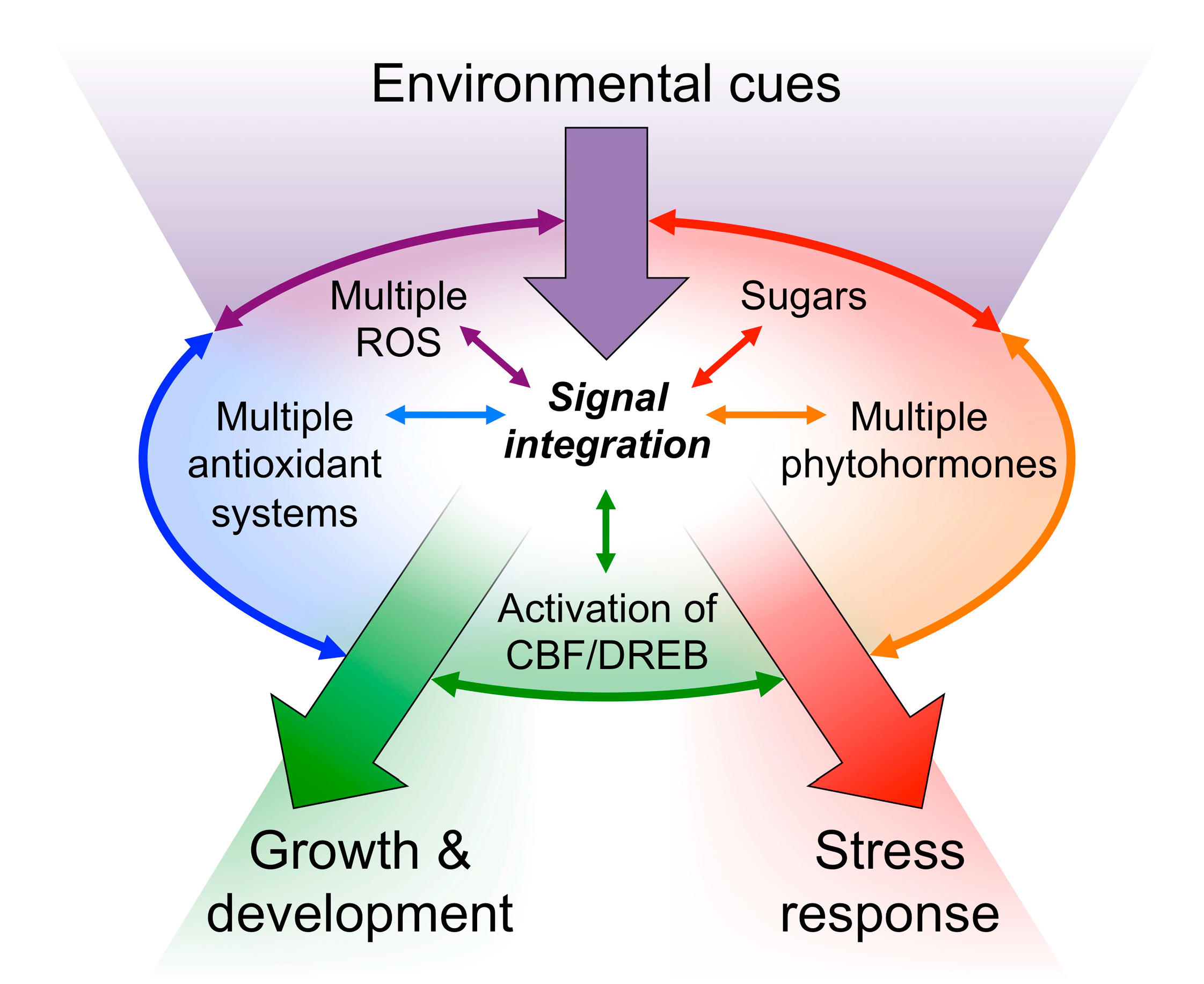
3. Redox Regulation and Photoprotection
3.1. The Chloroplast Transduces the State of the Environment into Redox Signals
3.2. Redox Signals Modulate Vascular Infrastructure
3.3. Manipulation of Thermal Energy Dissipation and Plant Growth: Photon-Capture Efficiency and Redox Regulation
4. Natural Populations Illustrate How Trade-Offs in Performance under Cool versus Hot Temperature Can Be Minimized
4.1. Arabidopsis thaliana Ecotypes from Sweden and Italy Show Differential Adaptation Patterns and CBF Expression
4.2. The Cool-Temperature Phenotype of Winter Annuals Is Reminiscent of a High-Light Phenotype
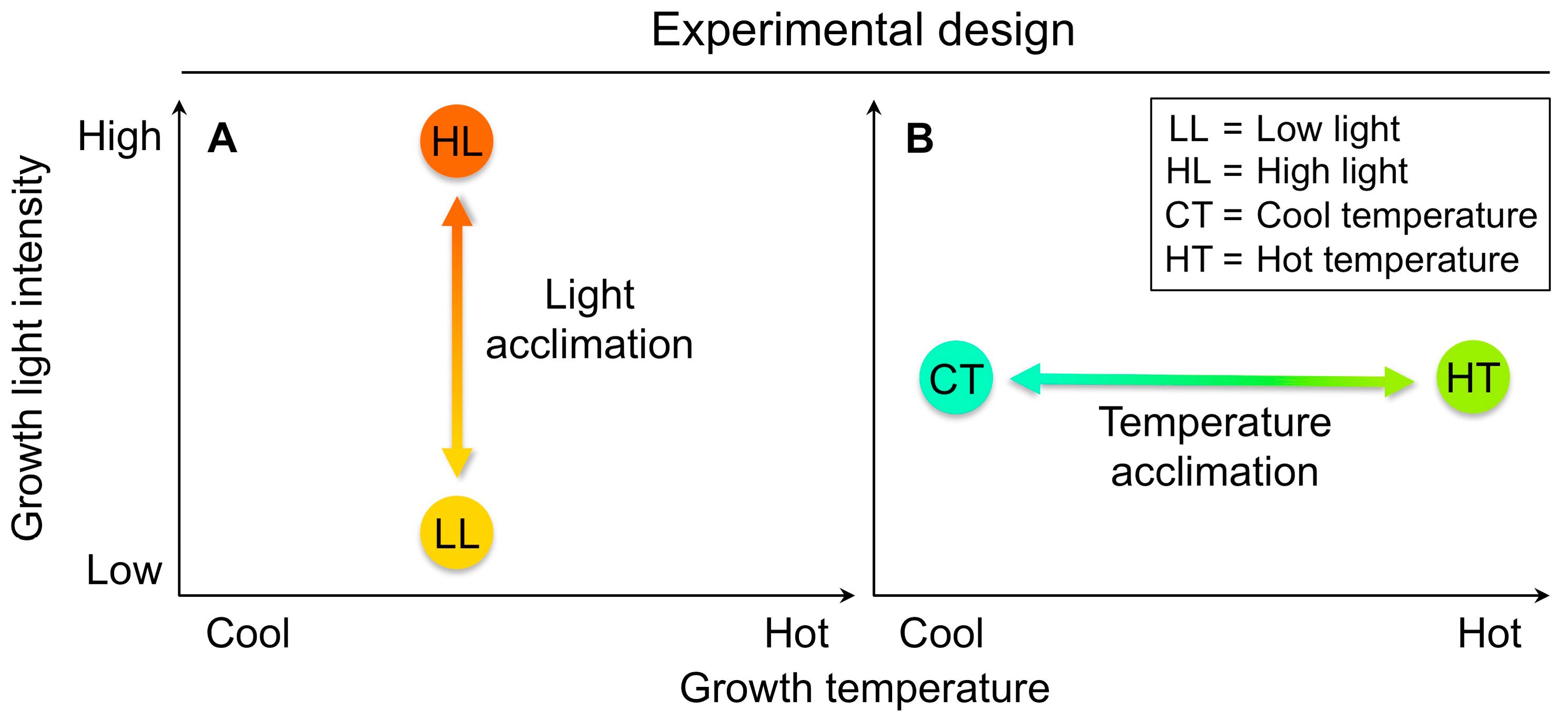
4.3. An Experimental Design Suitable to Identify On-Off Switches That Minimize Penalties
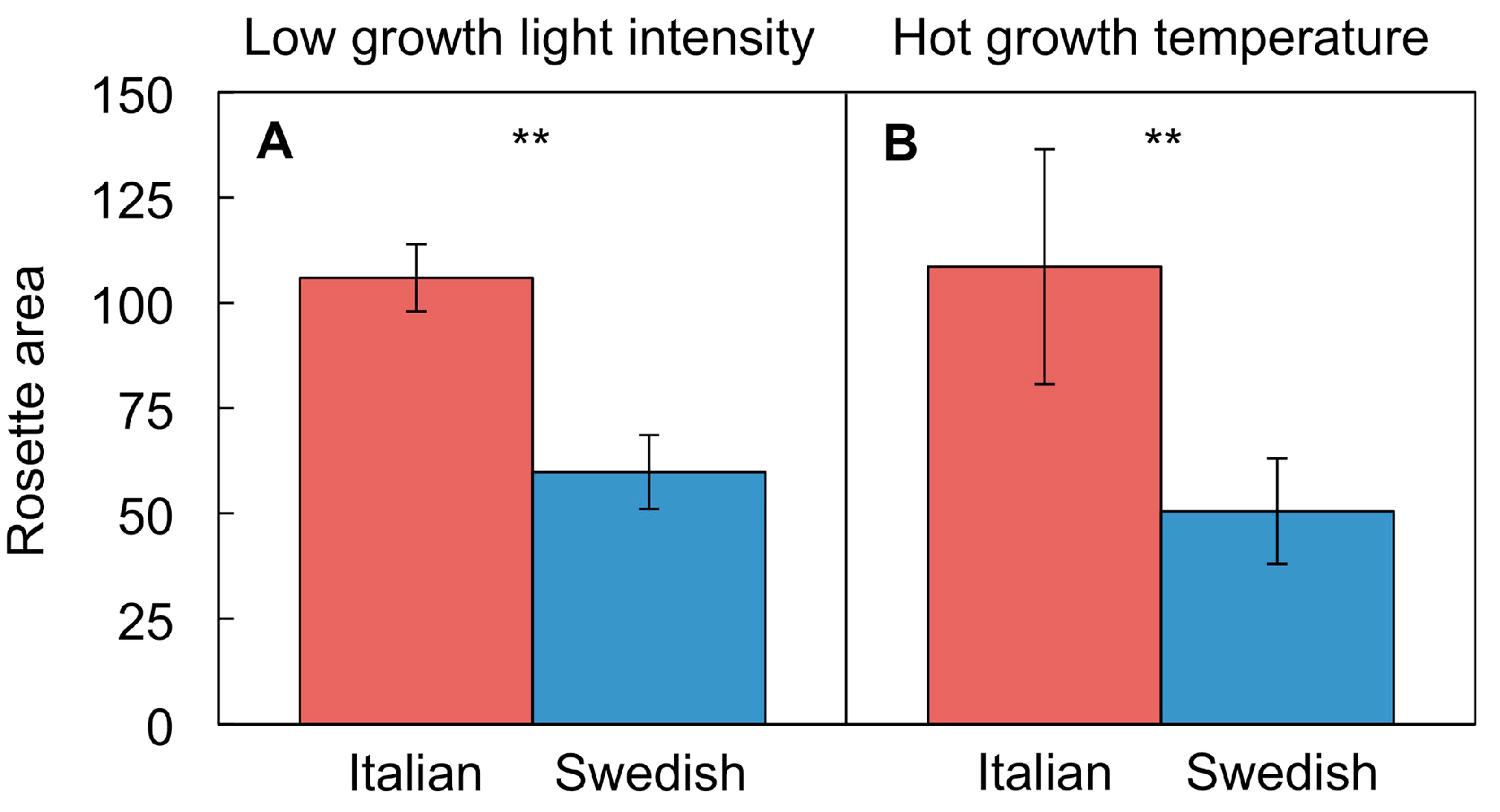
4.4. Differential Adjustment of Leaf Form and Function in Swedish and Italian Ecotypes
4.4.1. Different Growth Rates in Swedish and Italian Ecotypes
4.4.2. Differential Antioxidant Levels between Swedish and Italian Ecotypes
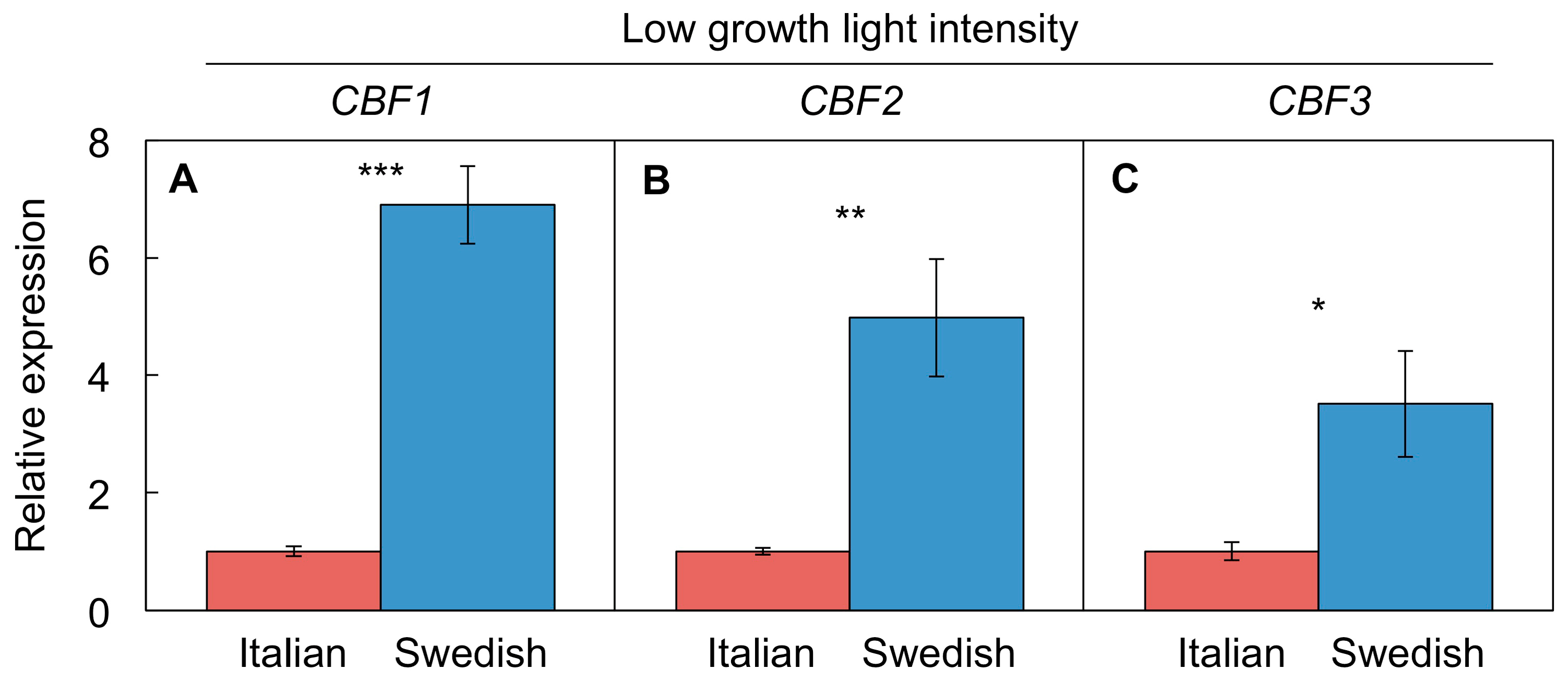
4.4.3. Differential CBF Expression between Swedish and Italian Ecotypes
4.4.4. Ability to Switch Off Other Features of the Cool-Temperature/High-Light Phenotype
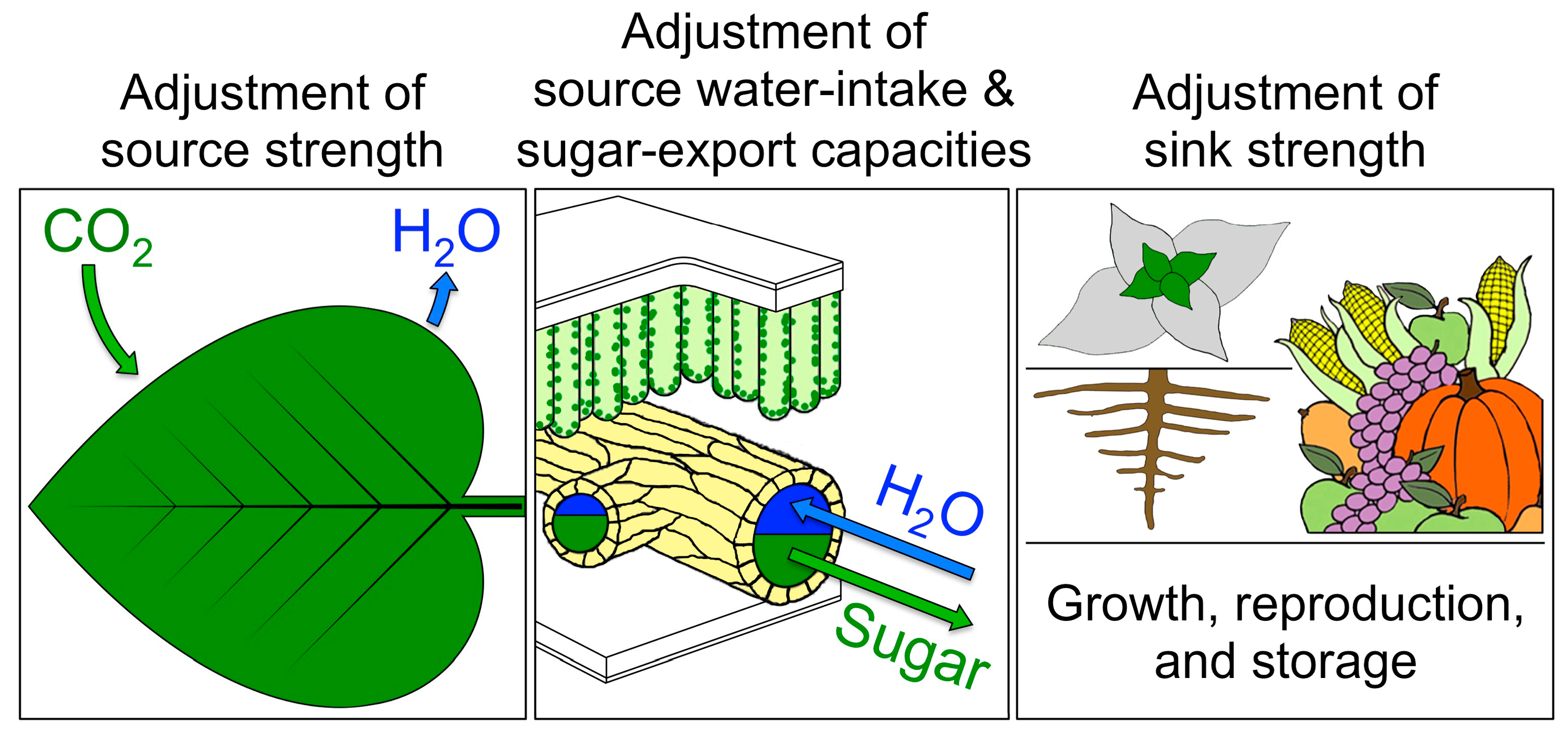
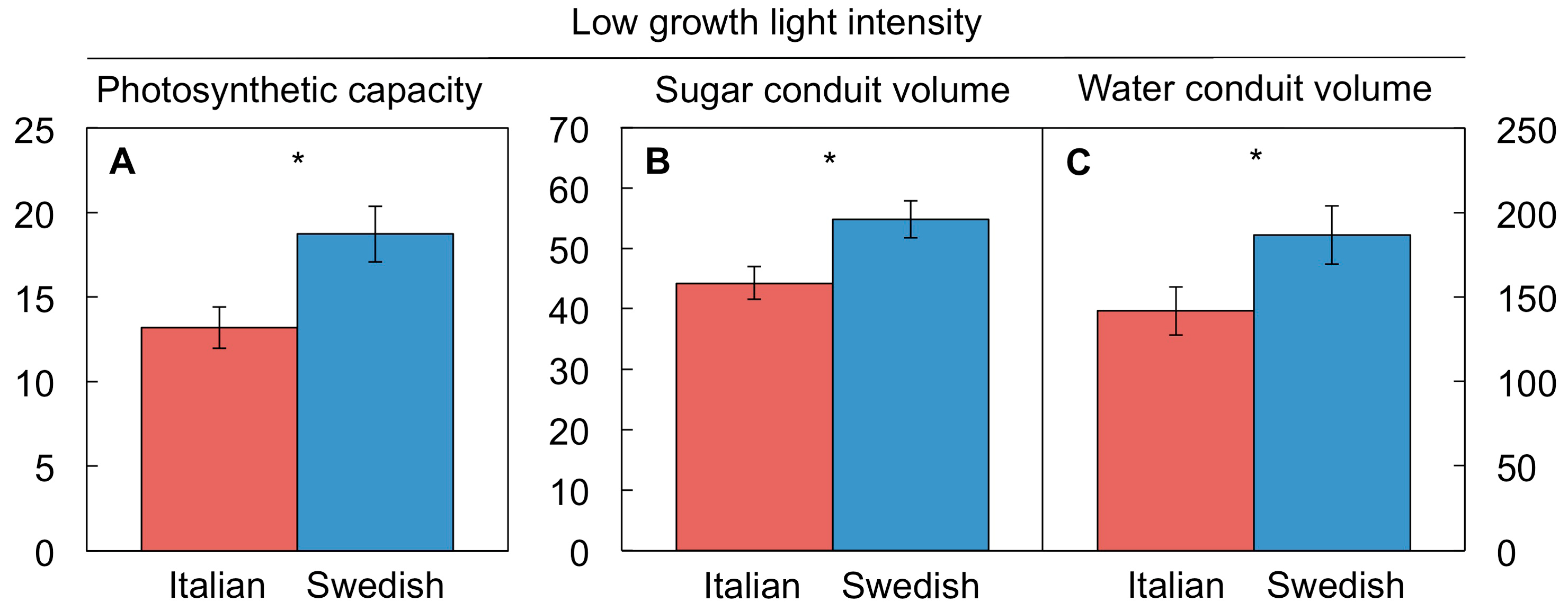
4.5. A Key Role for Vascular Adjustment: High Photosynthesis Rates Supported by Improved Sugar Removal at Cool Temperature and Improved Water Delivery at Hot Temperature
4.5.1. Coordination of Photosynthetic Productivity with Sugar and Water Transport
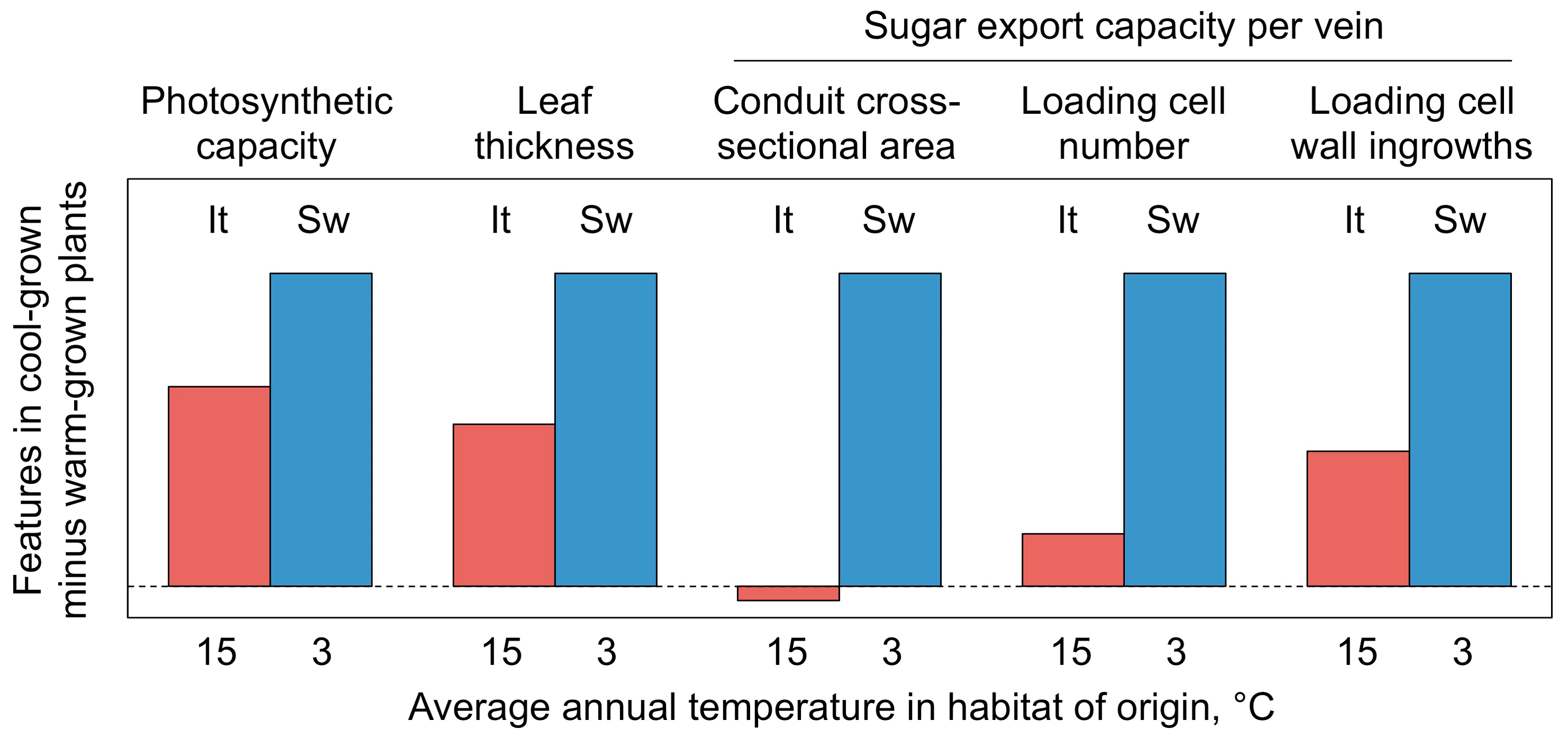
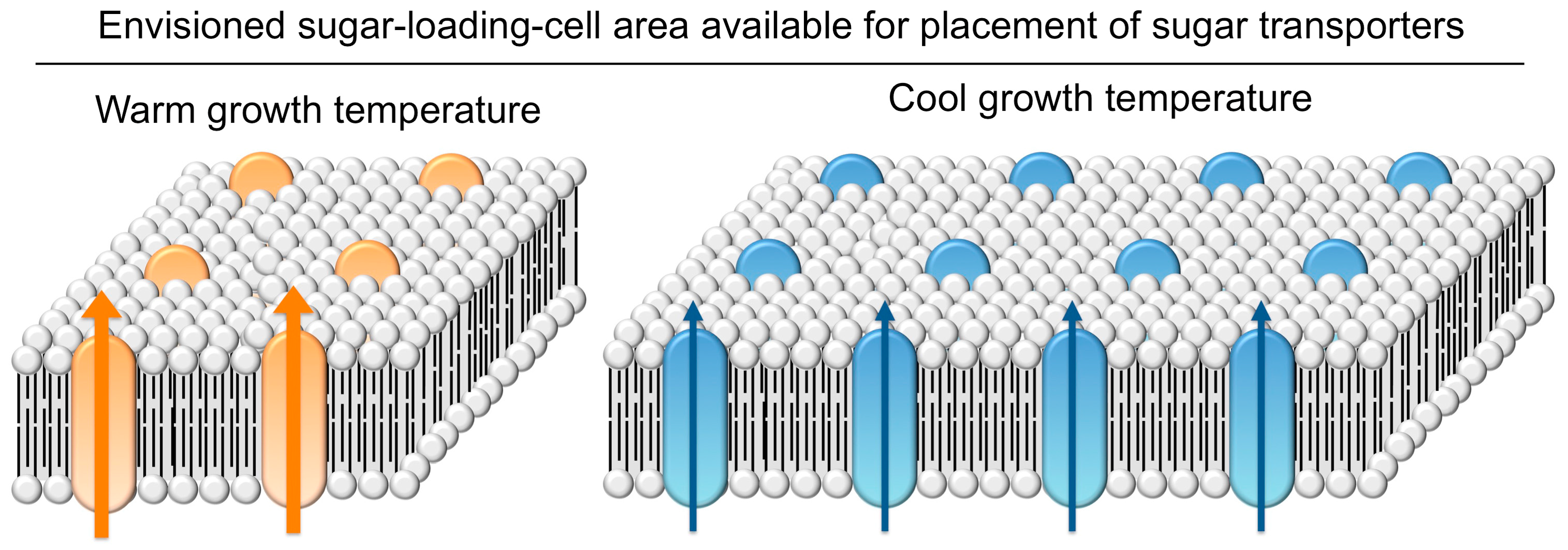
4.5.2. Upregulation of Sugar-Export Capacity under Cool Growth Temperature
4.5.3. Upregulation of Foliar Water-Import Capacity under Hot Growth Temperature
4.6. Leaf Acclimation to Light Environment Defines Sun-Loving and Shade-Loving Genotypes
4.6.1. Adjustment of Leaf Size and Thickness
4.6.2. Adjustment of Leaf Vascular Anatomy
4.7. What Is Known about the Regulators of Vascular Organization
5. From Mechanistic Insight to Crop Improvement
6. Conclusions
- To provide guidance for future germplasm screening, genomics-directed breeding, and gene editing, studies into adaptive traits should give attention to the whole-plant source–sink relationship and a range of relevant environmental conditions.
- Superior plant productivity depends on distinct plant features in different environments, and could be custom-designed for specific local contexts using environmentally induced gene regulators, such as CBF/DREB transcription factors, that coordinate multiple aspects of plant function.
- Arabidopsis thaliana remains an ideal model organism. In terms of eco-physiology, it occupies a range of native habitats, with different ecotypes manifesting distinct functionality under controlled growth conditions. On the molecular side, over a thousand sequenced A. thaliana genomes are available for transformation of selected ecotypes via precision gene editing.
Acknowledgments
Conflicts of Interest
Abbreviations
| CBF | C-repeat binding factor |
| CT | cool growth temperature |
| ΔpH | trans-thylakoid pH gradient |
| DREB | dehydration-responsive element-binding factor |
| HL | high growth light intensity |
| HT | hot growth temperature |
| LL | low growth light intensity |
| NPQ | non-photochemical quenching of chlorophyll fluorescence |
| ROS | reactive oxygen species |
References
- Long, S.P.; Ainsworth, E.A.; Leakey, A.D.B.; Nösberger, J.; Ort, D.R. Food for thought: Lower-than-expected crop yield stimulation with rising CO2 concentrations. Science 2006, 312, 1918–1921. [Google Scholar] [CrossRef] [PubMed]
- Long, S.P.; Marshall-Colon, A.; Zhu, X.-G. Meeting the global food demand of the future by engineering crop photosynthesis and yield potential. Cell 2015, 161, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Jaglo-Ottosen, K.R.; Gilmour, S.J.; Zarka, D.G.; Schabenberger, O.; Thomashow, M.F. Arabidopsis CBF1 overexpression induces COR genes and enhances freezing tolerance. Science 1998, 280, 104–106. [Google Scholar] [CrossRef] [PubMed]
- Larkindale, J.; Hall, J.D.; Knight, M.R.; Vierling, E. Heat stress phenotypes of Arabidopsis mutants implicate multiple signaling pathways in the acquisition of thermotolerance. Plant Phys. 2005, 138, 882–897. [Google Scholar] [CrossRef] [PubMed]
- Moellering, E.R.; Muthan, B.; Benning, C. Freezing tolerance in plants requires lipid remodeling at the outer chloroplast membrane. Science 2010, 330, 226–228. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.-W.; Lee, U.; Vierling, E. Arabidopsis hot mutants define multiple functions required for acclimation to high temperatures. Plant Physiol. 2003, 132, 757–767. [Google Scholar] [CrossRef] [PubMed]
- Demmig-Adams, B.; Stewart, J.J.; Adams, W.W., III. Environmental regulation of photosynthetic capacity: An integrated view. Curr. Opin. Plant Biol. 2017, 37, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Paul, M.J.; Foyer, C.H. Sink regulation of photosynthesis. J. Exp. Bot. 2001, 52, 1383–1400. [Google Scholar] [CrossRef] [PubMed]
- Ainsworth, E.A.; Bush, D.R. Carbohydrate export from the leaf: A highly regulated process and target to enhance photosynthesis and productivity. Plant Physiol. 2011, 155, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Adams, W.W., III; Cohu, C.M.; Muller, O.; Demmig-Adams, B. Foliar phloem infrastructure in support of photosynthesis. Front. Plant Sci. 2013, 4, 194. [Google Scholar] [CrossRef] [PubMed]
- Brodribb, T.J.; Feild, T.S.; Jordan, G.J. Leaf maximum photosynthetic rate and venation are linked by hydraulics. Plant Physiol. 2007, 144, 1890–1898. [Google Scholar] [CrossRef] [PubMed]
- Scoffoni, C.; Chatelet, D.S.; Pasquet-kok, J.; Rawls, M.; Donoghue, M.J.; Edwards, E.J.; Sack, L. Hyrdaulic basis for the evolution of photosynthetic productivity. Nat. Plants 2016, 2, 16072. [Google Scholar] [CrossRef] [PubMed]
- Fahlgren, N.; Gehan, M.A.; Baxter, I. Lights, camera, action: High-throughput plant phenotyping is ready for a close-up. Curr. Opin. Plant Biol. 2015, 24, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Pino, M.-T.; Skinner, J.S.; Jeknić, Z.; Hayes, P.M.; Soeldner, A.H.; Thomashow, M.F.; Chen, T.H.H. Ectopic AtCBF1 over-expression enhances freezing tolerance and induces cold acclimation-associated physiological modifications in potato. Plant Cell Environ. 2008, 31, 393–406. [Google Scholar] [CrossRef] [PubMed]
- Hüner, N.P.A.; Bode, R.; Dahal, K.; Hollis, L.; Rosso, D.; Krol, M.; Ivanov, A.G. Chloroplast redox imbalance governs phenotypic plasticity: The “grand design of photosynthesis” revisited. Front. Plant Sci. 2012, 3, 255. [Google Scholar] [CrossRef] [PubMed]
- Hüner, N.P.A.; Dahal, K.; Kurepin, L.V.; Savitch, L.; Singh, J.; Ivanov, A.G.; Kane, K.; Sarhan, F. Potential for increased photosynthetic performance and crop productivity in response to climate change: Role of CBFs and gibberellic acid. Front. Chem. 2014, 2, 18. [Google Scholar] [CrossRef] [PubMed]
- Hüner, N.P.A.; Dahal, K.; Bode, R.; Kurepin, L.V.; Ivanov, A.G. Photosynthetic acclimation, vernalization, crop productivity and ‘the grand design of photosynthesis’. J. Plant Physiol. 2016, 203, 29–43. [Google Scholar] [CrossRef] [PubMed]
- Kurepin, L.V.; Dahal, K.P.; Savitch, L.V.; Singh, J.; Bode, R.; Ivanov, A.G.; Hurry, V.; Hüner, N.P.A. Role of CBFs as integrators of chloroplast redox, phytochrome and plant hormone signaling during cold acclimation. Int. J. Mol. Sci. 2013, 14, 12729–12763. [Google Scholar] [CrossRef] [PubMed]
- Schramm, F.; Larkindale, J.; Kiehlmann, E.; Ganguli, A.; Englich, G.; Vierling, E.; von Koskull-Döring, P. A cascade of transcription factor DREB2A and heat stress transcription factor HsfA3 regulates the heat stress response of Arabidopsis. Plant J. 2008, 53, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Kidokoro, S.; Watanabe, K.; Ohori, T.; Moriwaki, T.; Maruyama, K.; Mizoi, J.; Myint Phyu Sin Htwe, N.; Fujita, Y.; Sekita, S.; Shinozaki, K.; et al. Soybean DREB1/CBF-type transcription factors function in heat and drought as well as cold stress-responsive gene expression. Plant J. 2015, 81, 505–518. [Google Scholar] [CrossRef] [PubMed]
- Lata, C.; Prasad, M. Role of DREBs in regulation of abiotic stress responses in plants. J. Exp. Bot. 2011, 62, 4731–4748. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S. The role of DREB transcription factors in abiotic stress tolerance of plants. Biotechnol. Biotechnol. Equip. 2011, 25, 2433–2442. [Google Scholar] [CrossRef]
- Akhtar, M.; Jaiswal, A.; Taj, G.; Jaiswal, J.P.; Qureshi, M.I.; Singh, N.K. DREB1/CBF transcription factors: Their structure, function and role in abiotic stress tolerance in plants. J. Genet. 2012, 91, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, P.K.; Gupta, K.; Lopato, S.; Agarwal, P. Dehydration responsive element binding transcription factors and their applications for the engineering of stress tolerance. J. Exp. Bot. 2017, 68, 2135–2148. [Google Scholar] [CrossRef] [PubMed]
- Thomashow, M.F. Molecular basis of plant cold acclimation: Insights gained from studying the CBF cold response pathway. Plant Physiol. 2010, 154, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Hwang, J.E.; Lim, C.J.; Kim, D.Y.; Lee, S.Y.; Lim, C.O. Arabidopsis DREB2C functions as a transcriptional activator of HsfA3 during the heat stress response. Biochem. Biophys. Res. Commun. 2010, 401, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Haake, V.; Cook, D.; Riechmann, J.; Pineda, O.; Thomashow, M.F.; Zhang, J.Z. Transcription factor CBF4 is a regulator of drought adaptation in Arabidopsis. Plant Physiol. 2002, 130, 639–648. [Google Scholar] [CrossRef] [PubMed]
- Shavrukov, Y.; Baho, M.; Lopato, S.; Langridge, P. The TaDREB3 transgene transferred by conventional crossings to different genetic backgrounds of bread wheat improves drought tolerance. Plant Biotechnol. J. 2016, 14, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Ausin, I.; Seleznev, A.; Méndez-Vigo, B.; Picó, F.X.; Sureshkumar, S.; Sundaramoorthi, V.; Bulach, D.; Powell, D.; Seemann, T.; et al. Natural variation identifies ICARUS1, a universal gene required for cell proliferation and growth at high temperatures in Arabidopsis thaliana. PLoS Genet. 2015, 11, e1005085. [Google Scholar] [CrossRef] [PubMed]
- Wingler, A. Comparison of signaling interactions determining annual and perennial plant growth in response to low temperature. Front. Plant Sci. 2015, 12, 794. [Google Scholar] [CrossRef] [PubMed]
- Kurbidaeva, A.; Novokreshchenova, M.; Ezhova, T. ICE genes in Arabidopsis thaliana: Clinal variation in DNA polymorphism and sequence diversification. Biol. Plant. 2015, 59, 245–252. [Google Scholar] [CrossRef]
- Alonso-Blanco, C.; Gomez-Mena, C.; Llorente, F.; Koornneef, M.; Salinas, J.; Martínez-Zapater, J.M. Genetic and molecular analyses of natural variation indicate CBF2 as a candidate gene for underlying freezing tolerance quantitative trait locus in Arabidopsis. Plant Physiol. 2005, 139, 1304–1312. [Google Scholar] [CrossRef] [PubMed]
- Gery, C.; Zuther, E.; Schulz, E.; Legoupi, J.; Chauveau, A.; McKhann, H.; Hincha, D.K.; Téoulé, E. Natural variation in the freezing tolerance of Arabidopsis thaliana: Effects of RNAi-induced CBF depletion and QTL localization vary among accessions. Plant Sci. 2011, 180, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Zhang, H.; Sun, T.; Shi, Y.; Wang, J.; Zhang, B.; Wang, Z.; Zhou, Y.; Gu, H. Natural variation of C-repeat-binding factor (CBFs) genes is a major cause of divergence in freezing tolerance among a group of Arabidopsis thaliana populations along the Yangtze River in China. New Phytol. 2013, 199, 1069–1080. [Google Scholar] [CrossRef] [PubMed]
- Oakley, C.G.; Ågren, J.; Atchison, R.A.; Schmeske, D.W. QTL mapping of freezing tolerance: Links to fitness and adaptive trade-offs. Mol. Ecol. 2014, 23, 4304–4315. [Google Scholar] [CrossRef] [PubMed]
- Hannah, M.A.; Wiese, D.; Freund, S.; Fiehn, O.; Heyer, A.G.; Hincha, D.K. Natural genetic variation of freezing tolerance in Arabidopsis. Plant Physiol. 2006, 142, 98–112. [Google Scholar] [CrossRef] [PubMed]
- Le, M.Q.; Engelsberger, W.R.; Hincha, D.K. Natural genetic variation in acclimation capacity at sub-zero temperatures after cold acclimation at 4 °C in different Arabidopsis thaliana accessions. Cryobiology 2008, 57, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Le, M.Q.; Pagter, M.; Hincha, D.K. Global changes in gene expression, assayed by microarray hybridization and quantitative RT-PCR, during acclimation of three Arabidopsis thaliana accessions to sub-zero temperatures after cold acclimation. Plant Mol. Biol. 2015, 87, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Usadel, B.; Bläsing, O.E.; Gibon, Y.; Poree, F.; Höhne, M.; Günter, M.; Trethewey, R.; Kamlage, B.; Poorter, H.; Stitt, M. Multilevel genomic analysis of the response of transcripts, enzyme activities and metabolites in Arabidopsis rosettes to a progressive decrease of temperature in the non-freezing range. Plant Cell Environ. 2008, 31, 518–547. [Google Scholar] [CrossRef] [PubMed]
- Medina, J.; Catalá, R.; Salinas, J. The CBFs: Three arabidopsis transcription factors to cold acclimate. Plant Sci. 2011, 180, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Zuther, E.; Schulz, E.; Childs, L.H.; Hincha, D.K. Clinal variation in the non-acclimated and cold-acclimated freezing tolerance of Arabidopsis thaliana accessions. Plant Cell Environ. 2012, 35, 1860–1878. [Google Scholar] [CrossRef] [PubMed]
- Gilmour, S.J.; Fowler, S.G.; Thomashow, M.F. Arabidopsis transcriptional activators CBF1, CBF2, and CBF3 have matching functional activities. Plant Mol. Biol. 2004, 54, 767–781. [Google Scholar] [CrossRef] [PubMed]
- Savitch, L.V.; Allard, G.; Seki, M.; Robert, L.S.; Tinker, N.A.; Huner, N.P.A.; Shinozaki, K.; Singh, J. The effect of overexpression of two Brassica CBF/DREB1-like transcription factors on photosynthetic capacity and freezing tolerance in Brassica napus. Plant Cell Physiol. 2005, 46, 1525–1539. [Google Scholar] [CrossRef] [PubMed]
- Bihani, P.; Char, B.; Bhargava, S. Transgenic expression of sorghum DREB2 in rice improves tolerance and yield under water limitation. J. Agric. Sci. 2011, 149, 95–101. [Google Scholar] [CrossRef]
- Morran, S.; Eini, O.; Pyvovarenko, T.; Parent, B.; Singh, R.; Ismagul, A.; Eliby, S.; Shirley, N.; Langridge, P.; Lopato, S. Improvement of stress tolerance of wheat and barley by modulation of expression of DREB/CBF factors. Plant Biotechnol. J. 2011, 9, 230–249. [Google Scholar] [CrossRef] [PubMed]
- Kovalchuk, N.; Jia, W.; Eini, O.; Morran, S.; Pyvovarenko, T.; Fletcher, S.; Bazanova, N.; Harris, J.; Beck-Oldach, K.; Shavrukov, Y.; et al. Optimization of TaDREB3 gene expression in transgenic barley using cold-inducible promoters. Plant Biotechnol. J. 2013, 11, 659–670. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Noctor, G. Redox regulation in photosynthetic organisms: Signaling, acclimation, and practical implications. Antioxid. Redox Signal. 2009, 11, 861–905. [Google Scholar] [CrossRef] [PubMed]
- Schippers, J.H.M.; Foyer, C.H.; van Dongen, J.T. Redox regulation in shoot growth, SAM maintenance and flowering. Curr. Opin. Plant Biol. 2016, 29, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Tognetti, V.B.; Bielach, A.; Hrtyan, M. Redox regulation at the site of primary growth: Auxin, cytokinin and ROS crosstalk. Plant Cell Environ. 2017, 40, 2586–2605. [Google Scholar] [CrossRef] [PubMed]
- Demmig-Adams, B.; Cohu, C.M.; Amiard, V.; van Zadelhoff, G.; Veldink, G.A.; Muller, O.; Adams, W.W., III. Emerging trade-offs—Impact of photoprotectants (PsbS, xanthophylls, and vitamin E) on oxylipins as regulators of development and defense. New Phytol. 2013, 197, 720–729. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, A.; Redij, T.; Sharma, B.; Suprasanna, P. Interaction between hormone and redox signaling in plants: Divergent pathways and convergent roles. In Mechanism of Plant Hormone Signaling under Stress; Pandey, G.K., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2017; Part II; pp. 1–22. [Google Scholar] [CrossRef]
- Schmidt, R.; Kunkowska, A.B.; Schippers, J.H.M. Role of reactive oxygen species during cell expansion in leaves. Plant Physiol. 2016, 172, 2098–2106. [Google Scholar] [CrossRef] [PubMed]
- Mittler, R. ROS are good. Trends Plant Sci. 2017, 22, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Dolferus, R. To grow or not to grow: A stressful decision for plants. Plant Sci. 2014, 229, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Sewelam, N.; Kazan, K.; Schenk, P.M. Global plant stress signaling: Reactive oxygen species at the cross-road. Front. Plant Sci. 2016, 7, 187. [Google Scholar] [CrossRef] [PubMed]
- Monson, R.K.; Sparks, J.P.; Rosenstiel, T.N.; Scott-Denton, L.E.; Huxman, T.E.; Harley, P.C.; Turnipseed, A.A.; Burns, S.P.; Backlund, B.; Hu, J. Climatic influences on net ecosystem CO2 exchange during the transition from wintertime carbon source to springtime carbon sink in a high-elevation, subalpine forest. Oecologia 2005, 146, 130–147. [Google Scholar] [CrossRef] [PubMed]
- Zarter, C.R.; Demmig-Adams, B.; Ebbert, V.; Adamska, I.; Adams, W.W., III. Photosynthetic capacity and light harvesting efficiency during the winter-to-spring transition in subalpine conifers. New Phytol. 2006, 172, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Adams, W.W., III; Demmig-Adams, B. Lessons from nature: A personal perspective. In Non-Photochemical Quenching and Energy Dissipation in Plants, Algae and Cyanobacteria, Advances in Photosynthesis and Respiration; Demmig-Adams, B., Garab, G., Adams, W.W., III, Govindjee, Eds.; Springer: Dordrecht, The Nertherlands, 2014; Volume 40, pp. 45–72. [Google Scholar] [CrossRef]
- Cohu, C.M.; Muller, O.; Stewart, J.J.; Demmig-Adams, B.; Adams, W.W., III. Association between minor loading vein architecture and light- and CO2-saturated rates of photosynthetic oxygen evolution among Arabidopsis thaliana ecotypes from different latitudes. Front. Plant Sci. 2013, 4, 264. [Google Scholar] [CrossRef] [PubMed]
- Soriano, M.A.; Orgaz, F.; Villalobos, F.J.; Fereres, E. Efficiency of water use of early plantings of sunflower. Eur. J. Agron. 2004, 21, 465–476. [Google Scholar] [CrossRef]
- Anastasi, U.; Cammarata, M.; Abbate, V. Yield potential and oil quality of sunflower (oleic and standard) grown between autumn and summer. Ital. J. Agron. 2000, 4, 23–36. [Google Scholar]
- Lovell, J.T.; Juenger, T.E.; Michaels, S.D.; Lasky, J.R.; Platt, A.; Richards, J.H.; Yu, X.; Easlon, H.M.; Sen, S.; McKay, J.K. Pleiotropy of FRIGIDA enhances the potential for multivariate adaptation. Proc. R. Soc. B. 2013, 280, 20131043. [Google Scholar] [CrossRef] [PubMed]
- Grillo, M.A.; Li, C.; Hammond, M.; Wang, L.; Schemske, D.W. Genetic architecture of flowering time differentiation between locally adapted populations of Arabidopsis thaliana. New Phytol. 2013, 197, 1321–1331. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.R.; Ahn, T.K.; Avenson, T.J.; Ballotari, M.; Cruz, J.A.; Kramer, D.M.; Bassi, R.; Fleming, G.R.; Keasling, J.D.; Niyogi, K.K. Lutein accumulation in the absence of zeaxanthin restores nonphotochemical quenching in the Arabidopsis thaliana npq1 mutant. Plant Cell 2009, 21, 1798–1812. [Google Scholar] [CrossRef] [PubMed]
- Demmig-Adams, B.; Stewart, J.J.; Burch, T.A.; Adams, W.W., III. Insights from placing photosynthetic light harvesting into context. J. Phys. Chem. Lett. 2014, 5, 2880–2889. [Google Scholar] [CrossRef] [PubMed]
- Demmig-Adams, B.; Cohu, C.M.; Muller, O.; Adams, W.W., III. Modulation of photosynthetic energy conversion efficiency in nature: From seconds to seasons. Photosynth. Res. 2012, 113, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Russin, W.A.; Evert, R.F.; Vanderveer, P.J.; Sharkey, T.D.; Briggs, S.P. Modification of a specific class of plasmodesmata and loss of sucrose export ability in the sucrose export defective1 maize mutant. Plant Cell 1996, 8, 645–658. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hofius, D.; Hajirezael, M.R.; Geiger, M.; Tschiersch, H.; Melzer, M.; Sonnewald, U. RNAi-mediated tocopherol deficiency impairs photoassimilate export in transgenic potato plants. Plant Physiol. 2004, 135, 1256–1268. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Song, W.; Sage, T.L.; DellaPenna, D. Tocopherols play a crucial role in low-temperature adaptation and phloem loading in Arabidopsis. Plant Cell 2006, 18, 2710–2732. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Song, W.; Sage, T.L.; DellaPenna, D. Role of callose synthases in transfer cell wall development of Arabidopsis tocopherol deficient mutants. Front. Plant Sci. 2014, 5, 46. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.J.; Polutchko, S.K.; Adams, W.W., III; Cohu, C.M.; Wenzl, C.A.; Demmig-Adams, B. Light, temperature, and tocopherol status influence foliar vascular anatomy and leaf function in Arabidopsis thaliana. Physiol. Plant. 2017, 160, 98–110. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.J.; Adams, W.W., III; Cohu, C.M.; Demmig-Adams, B. Tocopherols modulate leaf vein arrangement and composition without impacting photosynthesis. Photosynthetica 2018. [Google Scholar] [CrossRef]
- Hacke, U.G.; Sperry, J.S. Functional and ecological xylem anatomy. Perspect. Plant Ecol. Evol. Syst. 2001, 4, 97–115. [Google Scholar] [CrossRef]
- Fichot, R.; Chamaillard, S.; Depardieu, C.; Le Thiec, D.; Cochard, H.; Barigah, T.S.; Brignolas, F. Hydraulic efficiency and coordination with xylem resistance to cavitation, leaf function, and growth performance among eight unrelated Populus deltoids × Populus nigra hybrids. J. Exp. Bot. 2011, 62, 2093–2106. [Google Scholar] [CrossRef] [PubMed]
- Adams, W.W., III; Stewart, J.J.; Cohu, C.M.; Muller, O.; Demmig-Adams, B. Habitat temperature and precipitation of Arabidopsis thaliana ecotypes determine the response of foliar vasculature, photosynthesis, and transpiration to growth temperature. Front. Plant Sci. 2016, 7, 1026. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.J.; Demmig-Adams, B.; Cohu, C.M.; Wenzl, C.A.; Muller, O.; Adams, W.W., III. Growth temperature impact on leaf form and function in Arabidopsis thaliana ecotypes from northern and southern Europe. Plant Cell Environ. 2016, 39, 1549–1558. [Google Scholar] [CrossRef] [PubMed]
- Vuorinen, A.L.; Kelloniemi, J.; Valkonen, J.P.T. Why do viruses need phloem for systemic invasion of plants? Plant Sci. 2011, 181, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Hipper, C.; Brault, V.; Ziegler-Graff, V.; Revers, F. Viral and cellular factors involved in phloem transport of plant viruses. Front. Plant Sci. 2013, 4, 154. [Google Scholar] [CrossRef] [PubMed]
- Yadeta, K.A.; Thomma, P.H.J. The xylem as battleground for plant hosts and vascular wilt pathogens. Front. Plant Sci. 2013, 4, 97. [Google Scholar] [CrossRef] [PubMed]
- Li, X.P.; Gilmore, A.M.; Caffarri, S.; Bassi, R.; Golan, T.; Kramer, D.; Niyogi, K.K. Regulation of photosynthetic light harvesting involves intrathylakoid lumen pH sensing by the PsbS protein. J. Biol. Chem. 2004, 279, 22866–22874. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.P.; Ruban, A.V. Restoration of rapidly reversible photoprotective energy dissipation in the absence of PsbS protein by enhanced ΔpH. J. Biol. Chem. 2012, 286, 19973–19981. [Google Scholar] [CrossRef] [PubMed]
- Strand, D.D.; Kramer, D.M. Control of non-photochemical exciton quenching by the proton circuit of photosynthesis. In Non-Photochemical Quenching and Energy Dissipation in Plants, Algae and Cyanobacteria, Advances in Photosynthesis and Respiration; Demmig-Adams, B., Garab, G., Adams, W.W., III, Govindjee, Eds.; Springer: Dordrecht, The Nertherlands, 2014; Volume 40, pp. 387–408. [Google Scholar]
- Armbruster, U.; Leonelli, L.; Correa Galvis, V.; Strand, D.; Quinn, E.H.; Jonikas, M.C.; Niyogi, K.K. Regulation and levels of the thylakoid K+/H+ antiporter KEA3 shape the dynamic response of photosynthesis in fluctuating light. Plant Cell Physiol. 2016, 57, 1557–1567. [Google Scholar] [CrossRef] [PubMed]
- Krondijk, J.; Glowacka, K.; Leonelli, L.; Gabilly, S.T.; Iwai, M.; Niyogi, K.K.; Long, S.P. Improving photosynthesis by accelerating recovery from photoprotection. Science 2016, 354, 857–861. [Google Scholar] [CrossRef] [PubMed]
- Esteban, R.; Olano, J.M.; Castresana, J.; Fernández-Marín, B.; Hernández, A.; Becerril, J.M.; García-Plazaola, J.I. Distribution and evolutionary trends of photoprotective isoprenoids (xanthophylls and tocopherols) within the plant kingdom. Physiol. Plant. 2009, 135, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.J.; Polutchko, S.K.; Adams, W.W., III; Demmig-Adams, B. Acclimation of Swedish and Italian ecotypes of Arabidopsis thaliana to light intensity. Photosynth. Res. 2017, 134, 215–229. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.J.; Adams, W.W., III; Cohu, C.M.; Polutchko, S.K.; Lombardi, E.M.; Demmig-Adams, B. Differences in light-harvesting, acclimation to growth light environment, and leaf structural development between Swedish and Italian ecotypes of Arabidopsis thaliana. Planta 2015, 242, 1277–1290. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.J.; Adams, W.W., III; Demmig-Adams, B. Photoprotection kinetics of Italian and Swedish ecotypes of Arabidopsis thaliana. Unpublished work. 2018. [Google Scholar]
- Baker, C.R.; Stewart, J.J.; Niyogi, K.K.; Adams, W.W., III; Demmig-Adams, B. Transcriptome analysis of Italian and Swedish Arabidopsis thaliana ecotypes acclimated to contrasting environmental conditions. Manuscript in preparation.
- Ågren, J.; Schemske, D.W. Reciprocal transplants demonstrate strong adaptive differentiation of the model organism Arabidopsis thaliana in its native range. New Phytol. 2012, 194, 1112–1122. [Google Scholar] [CrossRef] [PubMed]
- Novillo, F.; Alonso, J.M.; Ecker, J.R.; Salinas, J. CBF2/DREB1C is a negative regulator of CBF1/DREB1B and CBF3/DREB1A expression and plays a central role in stress tolerance in Arabidopsis. Proc. Natl. Acad. Sci. USA 2004, 101, 3985–3990. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Zhang, Z.; Xie, S.; Si, T.; Li, Y.; Shu, J.-K. Mutational evidence for the critical role of CBF transcription factors in cold acclimation in Arabidopsis. Plant Physiol. 2016, 171, 2744–2759. [Google Scholar] [CrossRef] [PubMed]
- Gehan, M.A.; Park, S.; Gilmour, S.J.; An, C.; Lee, C.-M.; Thomashow, M.F. Natural variation in the C-repeat binding factor cold response pathway correlates with local adaptation of Arabidopsis ecotypes. Plant J. 2015, 84, 682–693. [Google Scholar] [CrossRef] [PubMed]
- Monroe, J.G.; McGovern, C.; Lasky, J.R.; Grogan, K.; Beck, J.; McKay, J.K. Adaptation to warmer climates by parallel functional evolution of CBF genes in Arabidopsis thaliana. Mol. Ecol. 2016, 25, 3632–3644. [Google Scholar] [CrossRef] [PubMed]
- Ågren, J.; Oakley, C.G.; McKay, J.K.; Lovell, J.T.; Schemske, D.W. Genetic mapping of adaptation reveals fitness tradeoffs in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2013, 110, 21077–21082. [Google Scholar] [CrossRef] [PubMed]
- Cohu, C.M.; Lombardi, E.; Adams, W.W., III; Demmig-Adams, B. Increased nutritional quality of plants for long-term spaceflight missions through choice of plant variety and manipulation of growth conditions. Acta Astronaut. 2014, 94, 799–806. [Google Scholar] [CrossRef]
- Anderson, J.M.; Chow, W.S.; Park, Y.-I. The grand design of photosynthesis: Acclimation of the photosynthetic apparatus to environmental cues. Photosynth. Res. 1995, 46, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Cohu, C.M.; Muller, O.; Adams, W.W., III; Demmig-Adams, B. Leaf anatomical and photosynthetic acclimation to cool temperature and high light in two winter versus two summer annuals. Physiol. Plant. 2014, 152, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Oakley, C.G.; Savage, L.; Lotz, S.; Larson, G.R.; Thomashow, M.F.; Kramer, D.M.; Schemske, D.W. Genetic basis of photosynthetic responses to cold in two locally adapted populations of Arabidopsis thaliana. J. Exp. Bot. 2018, 69, 699–709. [Google Scholar] [CrossRef] [PubMed]
- Polutchko, S.K.; Stewart, J.J.; Demmig-Adams, B.; Adams, W.W., III. Evaluating the link between photosynthetic capacity and leaf vascular organization with principal component analysis. Photosynthetica 2018. [Google Scholar] [CrossRef]
- Krapp, A.; Hofmann, B.; Schäfer, C.; Stitt, M. Regulation of the expression of rbcS and other photosynthetic genes by carbohydrates: A mechanism for the “sink regulation” of photosynthesis? Plant J. 1993, 3, 817–828. [Google Scholar] [CrossRef]
- Krapp, A.; Stitt, M. An evaluation of direct and indirect mechanisms for the “sink-regulation” of photosynthesis in spinach: Changes in gas exchange, carbohydrates, metabolites, enzyme activities and steady-state transcript levels after cold-girdling source leaves. Planta 1995, 195, 313–323. [Google Scholar] [CrossRef]
- Adams, W.W., III; Cohu, C.M.; Amiard, V.; Demmig-Adams, B. Associations between the acclimation of phloem-cell wall ingrowths in minor veins and maximal photosynthesis rate. Front. Plant Sci. 2014, 5, 24. [Google Scholar] [CrossRef] [PubMed]
- Haritatos, E.; Medville, R.; Turgeon, R. Minor vein structure and sugar transport in Arabidopsis thaliana. Planta 2000, 211, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Cohu, C.M.; Muller, O.; Demmig-Adams, B.; Adams, W.W., III. Minor loading vein acclimation for three Arabidopsis thaliana ecotypes in response to growth under different temperature and light regimes. Front. Plant Sci. 2013, 4, 240. [Google Scholar] [CrossRef] [PubMed]
- Sperry, J.S.; Venturas, M.D.; Anderegg, W.R.L.; Mencucinni, M.; Mackay, D.S.; Wang, Y.; Love, D.M. Predicting stomatal responses to the environment from the optimization of photosynthetic gain and hydraulic cost. Plant Cell Environ. 2017, 40, 816–830. [Google Scholar] [CrossRef] [PubMed]
- Lim, C.J.; Hwang, J.E.; Chen, H.; Hong, J.K.; Yang, K.A.; Choi, M.S.; Lee, K.O.; Chung, W.S.; Lee, S.Y.; Lim, C.O. Over-expression of the Arabidopsis DRE/CRT-binding transcription factor DREB2C enhances thermotolerance. Biochem. Biophys. Res. Commun. 2007, 362, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Je, J.; Song, C.; Hwang, J.E.; Lim, C.O. A proximal promoter region of Arabidopsis DREB2C confers tissue-specific expression under heat stress. J. Integr. Plant Biol. 2012, 54, 640–651. [Google Scholar] [CrossRef] [PubMed]
- Blackman, C.J.; Brodribb, T.J.; Jordan, G.J. Leaf hydraulic vulnerability is related to conduit dimensions and drought resistance across a diverse range of woody angiosperms. New Phytol. 2010, 188, 1113–1123. [Google Scholar] [CrossRef] [PubMed]
- Pfautch, S.; Harbusch, M.; Wesolowski, A.; Smith, R.; Macfarlane, C.; Tjoelker, M.G.; Reich, P.B.; Adams, M.A. Climate determines vascular traits in the ecologically diverse genus Eucalyptus. Ecol. Lett. 2016, 19, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Givnish, T.J. Adaptation to sun and shade: A whole-plant perspective. Aust. J. Plant Physiol. 1988, 15, 63–92. [Google Scholar] [CrossRef]
- Goldstein, G.; Santiago, L.S.; Campanello, P.I.; Avalos, G.; Zhang, Y.-J.; Villagra, M. Facing shortage or excessive light: How tropical and subtropical trees adjust their photosynthetic behavior and life history traits to a dynamic forest environment. In Tropical Tree Physiology: Adaptations and Responses in a Changing Environment, Tree Physiology; Goldstein, G., Santiago, L.S., Eds.; Springer: Dordrecht, The Netherlands, 2016; Volume 6, pp. 319–336. [Google Scholar]
- Evans, J.R.; Poorter, H. Photosynthetic acclimation of plants to growth irradiance: The relative importance of specific leaf area and nitrogen partitioning in maximizing carbon gain. Plant Cell Environ. 2001, 24, 755–767. [Google Scholar] [CrossRef]
- Niinemets, U. The controversy over traits conferring shade-tolerance in trees: Ontogenetic changes revisited. J. Ecol. 2006, 94, 464–470. [Google Scholar] [CrossRef]
- Killi, D.; Bussotti, F.; Raschi, A.; Haworth, M. Adaptation to high temperature mitigates the impact of water deficit during combined heat and drought stress in C3 sunflower and C4 maize varieties with contrasting drought tolerance. Physiol. Plant. 2017, 159, 130–147. [Google Scholar] [CrossRef] [PubMed]
- Scarpella, E.; Barkoulas, M.; Tsiantis, M. Control of leaf and vein development by auxin. Cold Spring Harb. Perspect. Biol. 2010, 2, a001511. [Google Scholar] [CrossRef] [PubMed]
- Verna, C.; Sawchuk, M.G.; Linh, N.M.; Scarpella, E. Control of vein network topology by auxin transport. BMC Biol. 2015, 13, 94. [Google Scholar] [CrossRef] [PubMed]
- McAdam, S.A.M.; Eléouët, M.P.; Best, M.; Brodribb, T.J.; Murphy, M.C.; Cook, S.D.; Dalmais, M.; Dimitriou, T.; Gélinas-Marion, A.; Gill, W.M.; et al. Linking auxin with photosynthetic rate via leaf venation. Plant Physiol. 2017, 175, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Dai, M.; Yao, J.; Xiao, B.; Li, X.; Zhang, Q.; Xiong, L. Overexpressing a NAM, ATAF, and CUC (NAC) transcription factor enhances drought resistance and salt tolerance in rice. Proc. Natl. Acad. Sci. USA 2006, 103, 12987–12992. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Chen, X.; Hong, Y.-Y.; Wang, Y.; Xu, P.; Ke, S.-D.; Liu, H.-Y.; Shu, J.-K.; Oliver, D.J.; Xiang, C.-B. Activated expression of an Arabidopsis HD-START protein confers drought tolerance with improved root system and reduced stomatal density. Plant Cell 2008, 20, 1134–1151. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.E.; Repetti, P.P.; Adams, T.R.; Creelman, R.A.; Wu, J.; Warner, D.C.; Anstrom, D.C.; Bensen, R.J.; Castiglioni, P.P.; Donnarummo, M.G.; et al. Plant nuclear factor Y (NF-Y) B subunits confer drought tolerance and lead to improved corn yields on water-limited acres. Proc. Natl. Acad. Sci. USA 2007, 104, 16450–16455. [Google Scholar] [CrossRef] [PubMed]
- Karaba, A.; Dixit, S.; Greco, R.; Aharoni, A.; Trijatmiko, K.R.; Marsch-Martinez, N.; Krishnan, A.; Nataraja, K.N.; Udayakumar, M.; Pereira, A. Improvement of water use efficiency in rice by expression of HARDY, an Arabidopsis drought and salt tolerance gene. Proc. Natl. Acad. Sci. USA 2007, 104, 15270–15275. [Google Scholar] [CrossRef] [PubMed]
- Bartels, D.; Phillips, J. Drought stress tolerance. In Genetic Modification of Plants, Biotechnology in Agriculture and Forestry; Kempken, F., Jung, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; Volume 64, pp. 139–157. [Google Scholar]
| Water Conduit Number per Vein * | Individual Water Conduit Cross-Sectional Area, µm2 ** | |
|---|---|---|
| Col-0 wild-type | 8.0 ± 0.5 | 23.8 ± 1.7 |
| Tocopherol-deficient vte1 mutant | 10.5 ± 0.4 | 20.5 ± 0.6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Demmig-Adams, B.; Stewart, J.J.; Baker, C.R.; Adams, W.W. Optimization of Photosynthetic Productivity in Contrasting Environments by Regulons Controlling Plant Form and Function. Int. J. Mol. Sci. 2018, 19, 872. https://doi.org/10.3390/ijms19030872
Demmig-Adams B, Stewart JJ, Baker CR, Adams WW. Optimization of Photosynthetic Productivity in Contrasting Environments by Regulons Controlling Plant Form and Function. International Journal of Molecular Sciences. 2018; 19(3):872. https://doi.org/10.3390/ijms19030872
Chicago/Turabian StyleDemmig-Adams, Barbara, Jared J. Stewart, Christopher R. Baker, and William W. Adams. 2018. "Optimization of Photosynthetic Productivity in Contrasting Environments by Regulons Controlling Plant Form and Function" International Journal of Molecular Sciences 19, no. 3: 872. https://doi.org/10.3390/ijms19030872
APA StyleDemmig-Adams, B., Stewart, J. J., Baker, C. R., & Adams, W. W. (2018). Optimization of Photosynthetic Productivity in Contrasting Environments by Regulons Controlling Plant Form and Function. International Journal of Molecular Sciences, 19(3), 872. https://doi.org/10.3390/ijms19030872







