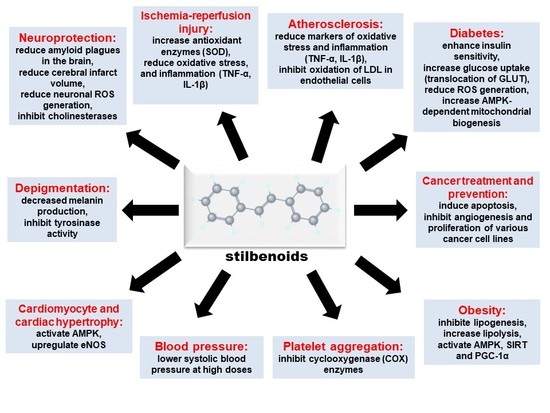
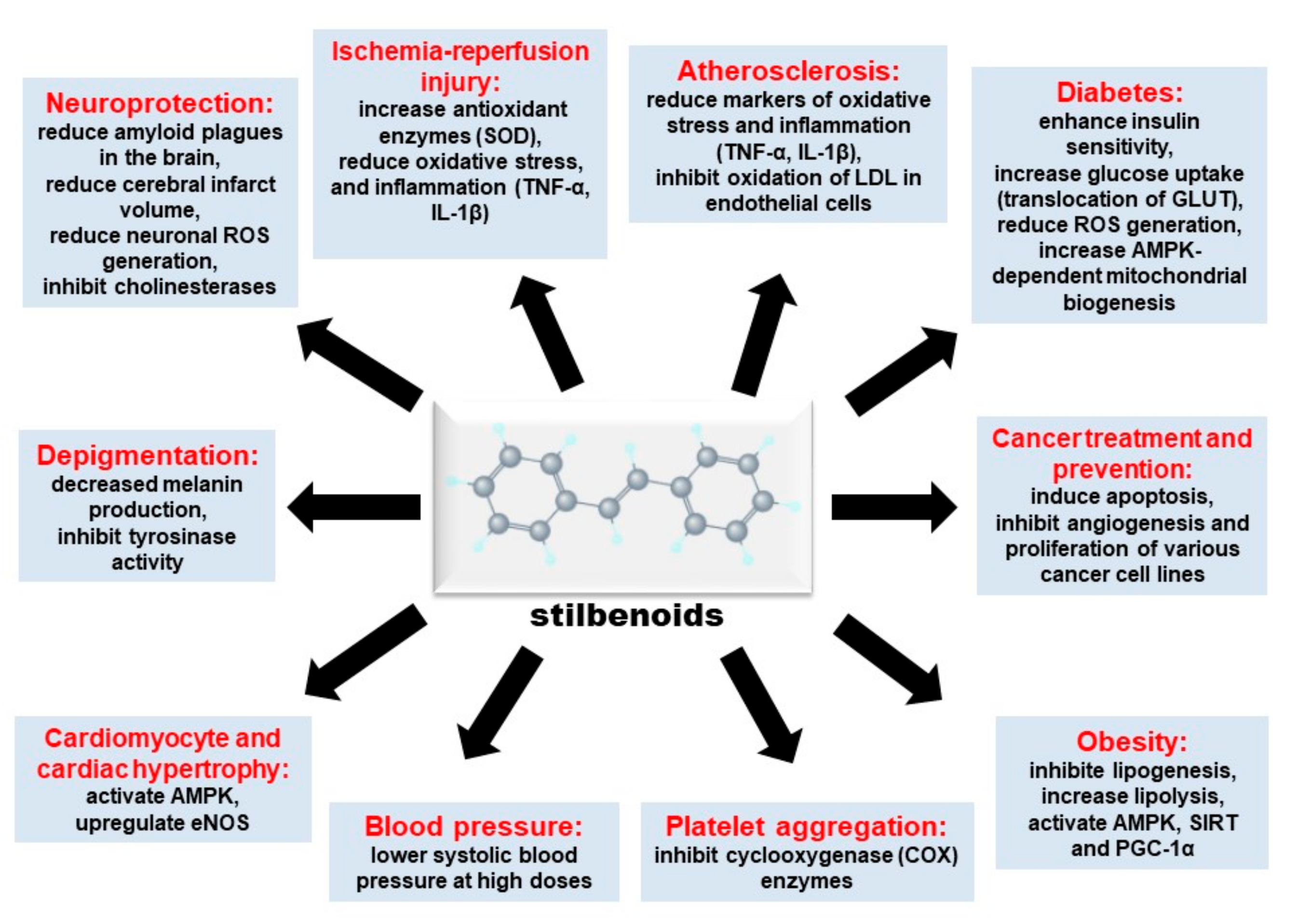
Biological Activities of Stilbenoids
Abstract
1. Introduction
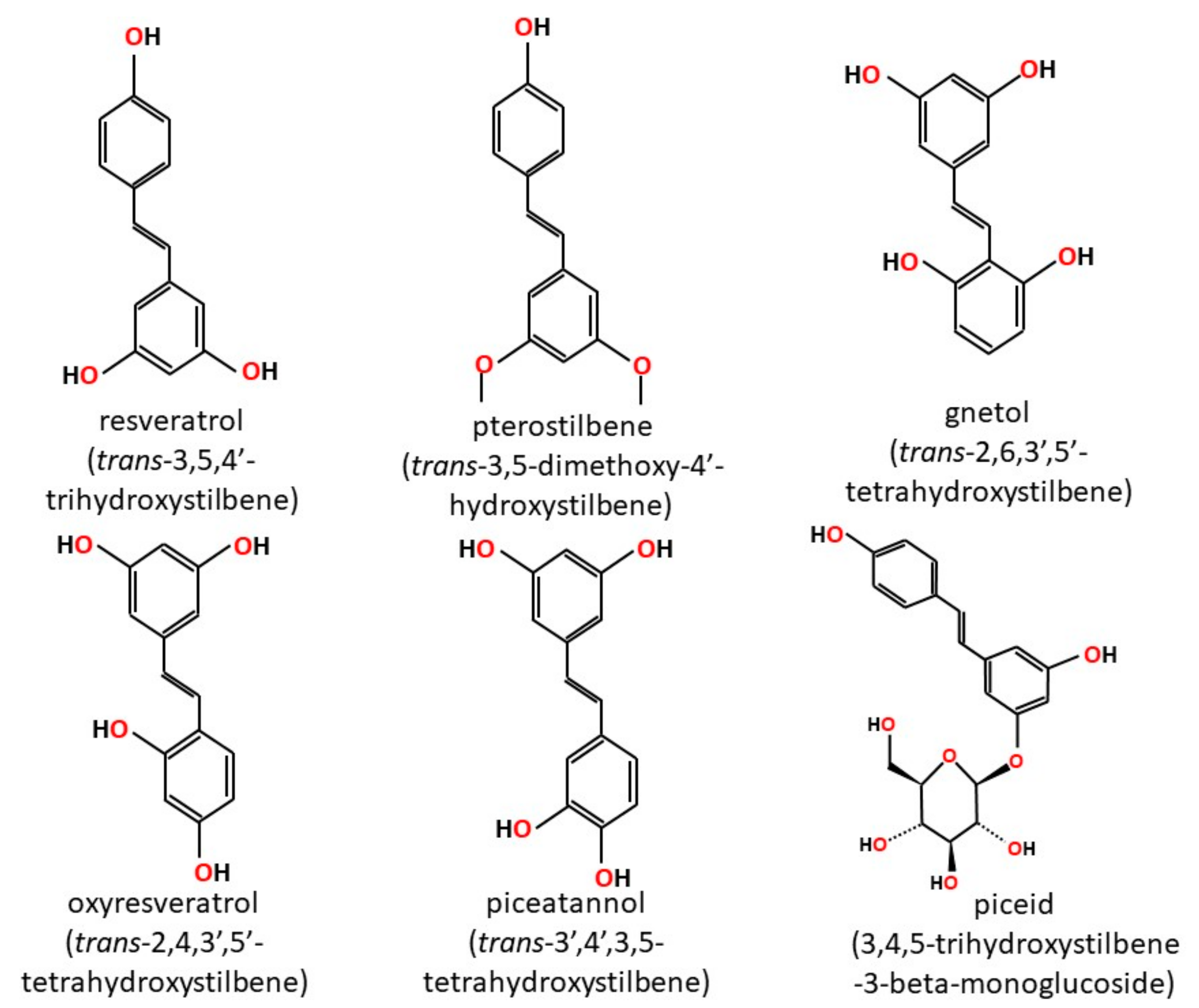
2. Stilbenoids
2.1. Bioavailability
2.2. Metabolism
2.3. Stability
2.4. Safety
3. Cardioprotective Effects of Stilbenoids
3.1. Vascular Compliance and Blood Pressure
3.2. Platelet Biology
3.3. Ischemia-Reperfusion Injury
3.4. Atherosclerosis
3.5. Cardiac Hypertrophy
3.6. Cardiovascular Human Studies
4. Other Biological Effects of Stilbenoids
4.1. Diabetes
4.2. Neurodegeneration
4.3. Obesity
4.4. Cancer Treatment and Prevention
4.5. Depigmentation
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AGE | Advanced glycated end-products |
| Akt | Protein kinase B |
| AMP | Adenosine monophosphate |
| AMPK | AMP-activated protein kinase |
| CAMKKβ | Calcium/calmodulin-dependent protein kinase kinase beta |
| cAMP | Cyclic adenosine monophosphate |
| cGMP | Cyclic guanosine monophosphate |
| CHD | Coronary heart disease |
| COX | Cyclooxygenase |
| CREB | cAMP response element-binding protein |
| CVD | Cardiovascular disease |
| CYP | Cytochrome P450 |
| ELISA | Enzyme-linked immunosorbent assay |
| eNOS | Endothelial nitric oxide synthase |
| ERK | Extracellular signal-regulated kinase |
| GLUT4 | Glucose transporter 4 |
| GSH | Glutathione |
| GSK-3β | Glycogen synthase kinase 3 beta |
| GSSH | Glutathione disulfide |
| GST | Glutathione S-transferase |
| HbA1c | Hemoglobin A1c |
| HDL | High density lipoprotein |
| HO-1 | Heme oxygenase-1 |
| HPLC | High performance liquid chromatography |
| IL | Interleukin |
| I/R | Ischemia-Reperfusion |
| LDH | Lactate dehydrogenase |
| l-DOPA | l-dihydroxyphenylalanine |
| LDL | Low density lipoprotein |
| LKB | Liver kinase B |
| MAPK | Mitogen-activated protein kinase |
| MCAO | Middle cerebral artery occlusion |
| MITF | Microphthalmia-associated transcription factor |
| MMP | Matrix metallopeptidase |
| mPTP | Membrane permeability transition pore |
| mRNA | Messenger ribonucleic acid |
| MSH | Melanocyte Stimulating Hormone |
| mTOR | Mammalian target of rapamycin |
| NAD | Nicotinamide adenine dinucleotide |
| NE | Norepinephrine |
| NFκB | Nuclear factor kappa B |
| NO | Nitric oxide |
| NOS | Nitric oxide synthase |
| O2− | Superoxide anion |
| •OH | Hydroxyl radical |
| PAI | Plasminogen activator inhibitor |
| PGC-1α | Peroxisome proliferator-activated receptor gamma coactivator-1-alpha |
| ROS | Reactive oxygen species |
| SD | Sprague-Dawley |
| SHR | Spontaneously hypertensive rat |
| SIRT1 | Sirtuin 1 |
| SOD | Superoxide dismutase |
| TGF-β | Transforming growth factor-beta |
| TNF-α | Tumor necrosis factor-alpha |
| TRYP | Tyrosinase-related protein |
| UCP | Uncoupling protein |
| UDP | Uridine diphosphate |
| WHO | World Health Organization |
| WKY | Wistar-Kyoto |
References
- Rivière, C.; Pawlus, A.D.; Mérillon, J.M. Natural stilbenoids: Distribution in the plant kingdom and chemotaxonomic interest in vitaceae. Nat. Prod. Rep. 2012, 29, 1317–1333. [Google Scholar] [CrossRef] [PubMed]
- Larronde, F.; Richard, T.; Delaunay, J.C.; Decendit, A.; Monti, J.P.; Krisa, S.; Mérillon, J.M. New stilbenoid glucosides isolated from vitis vinifera cell suspension cultures (cv. Cabernet sauvignon). Planta Medica 2005, 71, 888–890. [Google Scholar] [CrossRef] [PubMed]
- Waffo Teguo, P.; Fauconneau, B.; Deffieux, G.; Huguet, F.; Vercauteren, J.; Merillon, J.M. Isolation, identification, and antioxidant activity of three stilbene glucosides newly extracted from vitis vinifera cell cultures. J. Nat. Prod. 1998, 61, 655–657. [Google Scholar] [CrossRef] [PubMed]
- Yu, O.; Jez, J.M. Nature’s assembly line: Biosynthesis of simple phenylpropanoids and polyketides. Plant J. 2008, 54, 750–762. [Google Scholar] [CrossRef] [PubMed]
- Langcake, P.; Pryce, R.J. The production of resveratrol by vitis vinifera and other members of the vitaceae as a response to infection or injury. Physiol. Plant Pathol. 1976, 9, 77–86. [Google Scholar] [CrossRef]
- Langcake, P.; Cornford, C.A.; Pryce, R.J. Identification of pterostilbene as a phytoalexin from vitis vinifera leaves. Phytochemistry 1979, 18, 1025–1027. [Google Scholar] [CrossRef]
- López, M.; Martínez, F.; Del Valle, C.; Orte, C.; Miró, M. Analysis of phenolic constituents of biological interest in red wines by high-performance liquid chromatography. J. Chromatogr. A 2001, 922, 359–363. [Google Scholar] [CrossRef]
- Seshadri, T.R. Polyphenols of pterocarpus and dalbergia woods. Phytochemistry 1972, 11, 881–898. [Google Scholar] [CrossRef]
- Rimando, A.M.; Kalt, W.; Magee, J.B.; Dewey, J.; Ballington, J.R. Resveratrol, pterostilbene, and piceatannol in vaccinium berries. J. Agric. Food Chem. 2004, 52, 4713–4719. [Google Scholar] [CrossRef] [PubMed]
- Paul, B.; Masih, I.; Deopujari, J.; Charpentier, C. Occurrence of resveratrol and pterostilbene in age-old darakchasava, an ayurvedic medicine from India. J. Ethnopharmacol. 1999, 68, 71–76. [Google Scholar] [CrossRef]
- Prakash, S.; Khan, M.A.; Khan, K.Z.; Zaman, A. Stilbenes of gnetum ula. Phytochemistry 1985, 24, 622–624. [Google Scholar] [CrossRef]
- Ohguchi, K.; Tanaka, T.; Iliya, I.; Ito, T.; Iinuma, M.; Matsumoto, K.; Akao, Y.; Nozawa, Y. Gnetol as a potent tyrosinase inhibitor from genus gnetum. Biosci. Biotechnol. Biochem. 2003, 67, 663–665. [Google Scholar] [CrossRef] [PubMed]
- Xiang, W.; Jiang, B.; Li, X.M.; Zhang, H.J.; Zhao, Q.S.; Li, S.H.; Sun, H.D. Constituents of gnetum montanum. Fitoterapia 2002, 73, 40–42. [Google Scholar] [CrossRef]
- Ali, Z.; Tanaka, T.; Iliya, I.; Iinuma, M.; Furusawa, M.; Ito, T.; Nakaya, K.; Murata, J.; Darnaedi, D. Phenolic constituents of gnetum klossii. J. Nat. Prod. 2003, 66, 558–560. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.S.; Wang, Y.H.; Li, R.L.; Lin, M. Stilbene dimers from the lianas of gnetum hainanense. Phytochemistry 2000, 54, 875–881. [Google Scholar] [CrossRef]
- Kato, E.; Tokunaga, Y.; Sakan, F. Stilbenoids isolated from the seeds of melinjo (gnetum gnemon l.) and their biological activity. J. Agric. Food Chem. 2009, 57, 2544–2549. [Google Scholar] [CrossRef] [PubMed]
- Nagabhushanam, K.; Thomas, S.M.; Prakash, L.; Muhammed, M. Stilbenols—A class of priviledged structures. PharmaChem 2009, 8, 2–6. [Google Scholar]
- Seyed, M.A.; Jantan, I.; Bukhari, S.N.; Vijayaraghavan, K. A comprehensive review on the chemotherapeutic potential of piceatannol for cancer treatment, with mechanistic insights. J. Agric. Food Chem. 2016, 64, 725–737. [Google Scholar] [CrossRef] [PubMed]
- Potter, G.A.; Patterson, L.H.; Wanogho, E.; Perry, P.J.; Butler, P.C.; Ijaz, T.; Ruparelia, K.C.; Lamb, J.H.; Farmer, P.B.; Stanley, L.A.; et al. The cancer preventative agent resveratrol is converted to the anticancer agent piceatannol by the cytochrome p450 enzyme cyp1b1. Br. J. Cancer 2002, 86, 774–778. [Google Scholar] [CrossRef] [PubMed]
- Piotrowska, H.; Kucinska, M.; Murias, M. Biological activity of piceatannol: Leaving the shadow of resveratrol. Mutat. Res. 2012, 750, 60–82. [Google Scholar] [CrossRef] [PubMed]
- Murias, M.; Jäger, W.; Handler, N.; Erker, T.; Horvath, Z.; Szekeres, T.; Nohl, H.; Gille, L. Antioxidant, prooxidant and cytotoxic activity of hydroxylated resveratrol analogues: Structure-activity relationship. Biochem. Pharmacol. 2005, 69, 903–912. [Google Scholar] [CrossRef] [PubMed]
- Shin, N.H.; Ryu, S.Y.; Choi, E.J.; Kang, S.H.; Chang, I.M.; Min, K.R.; Kim, Y. Oxyresveratrol as the potent inhibitor on dopa oxidase activity of mushroom tyrosinase. Biochem. Biophys. Res. Commun. 1998, 243, 801–803. [Google Scholar] [CrossRef] [PubMed]
- Likhitwitayawuid, K.; Sritularak, B.; Benchanak, K.; Lipipun, V.; Mathew, J.; Schinazi, R.F. Phenolics with antiviral activity from millettia erythrocalyx and artocarpus lakoocha. Nat. Prod. Res. 2005, 19, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Lin, H.S.; Ho, P.C.; Ng, K.Y. The impact of aqueous solubility and dose on the pharmacokinetic profiles of resveratrol. Pharm. Res. 2008, 25, 2593–2600. [Google Scholar] [CrossRef] [PubMed]
- Silva, F.; Figueiras, A.; Gallardo, E.; Nerín, C.; Domingues, F.C. Strategies to improve the solubility and stability of stilbene antioxidants: A comparative study between cyclodextrins and bile acids. Food Chem. 2014, 145, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Coimbra, M.; Isacchi, B.; van Bloois, L.; Torano, J.S.; Ket, A.; Wu, X.; Broere, F.; Metselaar, J.M.; Rijcken, C.J.; Storm, G.; et al. Improving solubility and chemical stability of natural compounds for medicinal use by incorporation into liposomes. Int. J. Pharm. 2011, 416, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Neves, A.R.; Lúcio, M.; Martins, S.; Lima, J.L.; Reis, S. Novel resveratrol nanodelivery systems based on lipid nanoparticles to enhance its oral bioavailability. Int. J. Nanomed. 2013, 8, 177–187. [Google Scholar]
- Zhang, Y.; Shang, Z.; Gao, C.; Du, M.; Xu, S.; Song, H.; Liu, T. Nanoemulsion for solubilization, stabilization, and in vitro release of pterostilbene for oral delivery. AAPS PharmSciTech 2014, 15, 1000–1008. [Google Scholar] [CrossRef] [PubMed]
- Kapetanovic, I.M.; Muzzio, M.; Huang, Z.; Thompson, T.N.; McCormick, D.L. Pharmacokinetics, oral bioavailability, and metabolic profile of resveratrol and its dimethylether analog, pterostilbene, in rats. Cancer Chemother. Pharmacol. 2011, 68, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Remsberg, C.M.; Martinez, S.E.; Akinwumi, B.C.; Anderson, H.D.; Takemoto, J.K.; Sayre, C.L.; Davies, N.M. Preclinical pharmacokinetics and pharmacodynamics and content analysis of gnetol in foodstuffs. Phytother. Res. 2015, 29, 1168–1179. [Google Scholar] [CrossRef] [PubMed]
- Marier, J.F.; Vachon, P.; Gritsas, A.; Zhang, J.; Moreau, J.P.; Ducharme, M.P. Metabolism and disposition of resveratrol in rats: Extent of absorption, glucuronidation, and enterohepatic recirculation evidenced by a linked-rat model. J. Pharmacol. Exp. Ther. 2002, 302, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Remsberg, C.M.; Yáñez, J.A.; Ohgami, Y.; Vega-Villa, K.R.; Rimando, A.M.; Davies, N.M. Pharmacometrics of pterostilbene: Preclinical pharmacokinetics and metabolism, anticancer, antiinflammatory, antioxidant and analgesic activity. Phytother. Res. 2008, 22, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Roupe, K.A.; Yáñez, J.A.; Teng, X.W.; Davies, N.M. Pharmacokinetics of selected stilbenes: Rhapontigenin, piceatannol and pinosylvin in rats. J. Pharm. Pharmacol. 2006, 58, 1443–1450. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.S.; Tringali, C.; Spatafora, C.; Wu, C.; Ho, P.C. A simple and sensitive hplc-uv method for the quantification of piceatannol analog trans-3,5,3′,4′-tetramethoxystilbene in rat plasma and its application for a pre-clinical pharmacokinetic study. J. Pharm. Biomed. Anal. 2010, 51, 679–684. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Yeo, S.C.M.; Elhennawy, M.G.A.A.; Lin, H.-S. Oxyresveratrol: A bioavailable dietary polyphenol. J. Funct. Foods 2016, 22, 122–131. [Google Scholar] [CrossRef]
- Dellinger, R.W.; Garcia, A.M.; Meyskens, F.L. Differences in the glucuronidation of resveratrol and pterostilbene: Altered enzyme specificity and potential gender differences. Drug Metab. Pharmacokinet. 2014, 29, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Walle, T. Bioavailability of resveratrol. Ann. N. Y. Acad. Sci. 2011, 1215, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Andlauer, W.; Kolb, J.; Siebert, K.; Fürst, P. Assessment of resveratrol bioavailability in the perfused small intestine of the rat. Drugs Exp. Clin. Res. 2000, 26, 47–55. [Google Scholar] [PubMed]
- Kuhnle, G.; Spencer, J.P.; Chowrimootoo, G.; Schroeter, H.; Debnam, E.S.; Srai, S.K.; Rice-Evans, C.; Hahn, U. Resveratrol is absorbed in the small intestine as resveratrol glucuronide. Biochem. Biophys. Res. Commun. 2000, 272, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Boocock, D.J.; Patel, K.R.; Faust, G.E.; Normolle, D.P.; Marczylo, T.H.; Crowell, J.A.; Brenner, D.E.; Booth, T.D.; Gescher, A.; Steward, W.P. Quantitation of trans-resveratrol and detection of its metabolites in human plasma and urine by high performance liquid chromatography. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2007, 848, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Walle, T.; Hsieh, F.; DeLegge, M.H.; Oatis, J.E.; Walle, U.K. High absorption but very low bioavailability of oral resveratrol in humans. Drug Metab. Dispos. 2004, 32, 1377–1382. [Google Scholar] [CrossRef] [PubMed]
- Calamini, B.; Ratia, K.; Malkowski, M.G.; Cuendet, M.; Pezzuto, J.M.; Santarsiero, B.D.; Mesecar, A.D. Pleiotropic mechanisms facilitated by resveratrol and its metabolites. Biochem. J. 2010, 429, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.G.; Monteiro, J.; Marques, R.R.; Silva, A.M.; Martínez, C.; Canle, M.; Faria, J.L. Photochemical and photocatalytic degradation of trans-resveratrol. Photochem. Photobiol. Sci. 2013, 12, 638–644. [Google Scholar] [CrossRef] [PubMed]
- Rius, C.; Abu-Taha, M.; Hermenegildo, C.; Piqueras, L.; Cerda-Nicolas, J.M.; Issekutz, A.C.; Estañ, L.; Cortijo, J.; Morcillo, E.J.; Orallo, F.; et al. Trans- but not cis-resveratrol impairs angiotensin-ii-mediated vascular inflammation through inhibition of nf-κb activation and peroxisome proliferator-activated receptor-gamma upregulation. J. Immunol. 2010, 185, 3718–3727. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.C.; Lee, C.H.; Song, T.Y. Evaluation of resveratrol oxidation in vitro and the crucial role of bicarbonate ions. Biosci. Biotechnol. Biochem. 2010, 74, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Delmas, D.; Aires, V.; Limagne, E.; Dutartre, P.; Mazué, F.; Ghiringhelli, F.; Latruffe, N. Transport, stability, and biological activity of resveratrol. Ann. N. Y. Acad. Sci. 2011, 1215, 48–59. [Google Scholar] [CrossRef] [PubMed]
- Pantusa, M.; Bartucci, R.; Rizzuti, B. Stability of trans-resveratrol associated with transport proteins. J. Agric. Food Chem. 2014, 62, 4384–4391. [Google Scholar] [CrossRef] [PubMed]
- Cottart, C.H.; Nivet-Antoine, V.; Laguillier-Morizot, C.; Beaudeux, J.L. Resveratrol bioavailability and toxicity in humans. Mol. Nutr. Food Res. 2010, 54, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Riche, D.M.; McEwen, C.L.; Riche, K.D.; Sherman, J.J.; Wofford, M.R.; Deschamp, D.; Griswold, M. Analysis of safety from a human clinical trial with pterostilbene. J. Toxicol. 2013, 2013, 463595. [Google Scholar] [CrossRef] [PubMed]
- The who monica project. A worldwide monitoring system for cardiovascular diseases: Cardiovascular mortality and risk factors in selected communities. In World Health Statistics Annual; World Health Organization: Geneva, Switzerland, 1989; pp. 27–149.
- St Leger, A.S.; Cochrane, A.L.; Moore, F. Factors associated with cardiac mortality in developed countries with particular reference to the consumption of wine. Lancet 1979, 1, 1017–1020. [Google Scholar] [CrossRef]
- Renaud, S.; de Lorgeril, M. Wine, alcohol, platelets, and the french paradox for coronary heart disease. Lancet 1992, 339, 1523–1526. [Google Scholar] [CrossRef]
- Zordoky, B.N.; Robertson, I.M.; Dyck, J.R. Preclinical and clinical evidence for the role of resveratrol in the treatment of cardiovascular diseases. Biochim. Biophys. Acta 2015, 1852, 1155–1177. [Google Scholar] [CrossRef] [PubMed]
- Behbahani, J.; Thandapilly, S.J.; Louis, X.L.; Huang, Y.; Shao, Z.; Kopilas, M.A.; Wojciechowski, P.; Netticadan, T.; Anderson, H.D. Resveratrol and small artery compliance and remodeling in the spontaneously hypertensive rat. Am. J. Hypertens. 2010, 23, 1273–1278. [Google Scholar] [CrossRef] [PubMed]
- Thandapilly, S.J.; Wojciechowski, P.; Behbahani, J.; Louis, X.L.; Yu, L.; Juric, D.; Kopilas, M.A.; Anderson, H.D.; Netticadan, T. Resveratrol prevents the development of pathological cardiac hypertrophy and contractile dysfunction in the shr without lowering blood pressure. Am. J. Hypertens. 2010, 23, 192–196. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Dai, Y.; Yan, S.; Shi, Y.; Li, J.; Liu, J.; Cha, L.; Mu, J. Resveratrol lowers blood pressure in spontaneously hypertensive rats via calcium-dependent endothelial no production. Clin. Exp. Hypertens. 2016, 38, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Thandapilly, S.J.; Louis, X.L.; Behbahani, J.; Movahed, A.; Yu, L.; Fandrich, R.; Zhang, S.; Kardami, E.; Anderson, H.D.; Netticadan, T. Reduced hemodynamic load aids low-dose resveratrol in reversing cardiovascular defects in hypertensive rats. Hypertens. Res. 2013, 36, 866–872. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ma, W.; Zhang, P.; He, S.; Huang, D. Effect of resveratrol on blood pressure: A meta-analysis of randomized controlled trials. Clin. Nutr. 2015, 34, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Riche, D.M.; Riche, K.D.; Blackshear, C.T.; McEwen, C.L.; Sherman, J.J.; Wofford, M.R.; Griswold, M.E. Pterostilbene on metabolic parameters: A randomized, double-blind, and placebo-controlled trial. Evid. Based Complement. Altern. Med. 2014, 2014, 459165. [Google Scholar] [CrossRef] [PubMed]
- Freynhofer, M.K.; Bruno, V.; Wojta, J.; Huber, K. The role of platelets in athero-thrombotic events. Curr. Pharm. Des. 2012, 18, 5197–5214. [Google Scholar] [CrossRef] [PubMed]
- Ling, G.; Ovbiagele, B. Oral antiplatelet therapy in the secondary prevention of atherothrombotic events. Am. J. Cardiovasc. Drugs 2009, 9, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.V.; Dusse, L.M.; Vieira, L.M.; Carvalho, M.D. Platelet antiaggregants in primary and secondary prevention of atherothrombotic events. Arq. Bras. Cardiol. 2013, 100, e78–e84. [Google Scholar] [PubMed]
- Olas, B.; Wachowicz, B.; Saluk-Juszczak, J.; Zieliński, T. Effect of resveratrol, a natural polyphenolic compound, on platelet activation induced by endotoxin or thrombin. Thromb. Res. 2002, 107, 141–145. [Google Scholar] [CrossRef]
- Olas, B.; Wachowicz, B.; Holmsen, H.; Fukami, M.H. Resveratrol inhibits polyphosphoinositide metabolism in activated platelets. Biochim. Biophys. Acta 2005, 1714, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Stef, G.; Csiszar, A.; Lerea, K.; Ungvari, Z.; Veress, G. Resveratrol inhibits aggregation of platelets from high-risk cardiac patients with aspirin resistance. J. Cardiovasc. Pharmacol. 2006, 48, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Zbikowska, H.M.; Olas, B.; Wachowicz, B.; Krajewski, T. Response of blood platelets to resveratrol. Platelets 1999, 10, 247–252. [Google Scholar] [PubMed]
- Shen, M.Y.; Hsiao, G.; Liu, C.L.; Fong, T.H.; Lin, K.H.; Chou, D.S.; Sheu, J.R. Inhibitory mechanisms of resveratrol in platelet activation: Pivotal roles of p38 mapk and no/cyclic gmp. Br. J. Haematol. 2007, 139, 475–485. [Google Scholar] [CrossRef] [PubMed]
- Messina, F.; Guglielmini, G.; Curini, M.; Orsini, S.; Gresele, P.; Marcotullio, M.C. Effect of substituted stilbenes on platelet function. Fitoterapia 2015, 105, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Kloypan, C.; Jeenapongsa, R.; Sri-in, P.; Chanta, S.; Dokpuang, D.; Tip-pyang, S.; Surapinit, N. Stilbenoids from gnetum macrostachyum attenuate human platelet aggregation and adhesion. Phytother. Res. 2012, 26, 1564–1568. [Google Scholar] [CrossRef] [PubMed]
- Ray, P.S.; Maulik, G.; Cordis, G.A.; Bertelli, A.A.; Bertelli, A.; Das, D.K. The red wine antioxidant resveratrol protects isolated rat hearts from ischemia reperfusion injury. Free Radic. Biol. Med. 1999, 27, 160–169. [Google Scholar] [CrossRef]
- Shen, M.; Wu, R.X.; Zhao, L.; Li, J.; Guo, H.T.; Fan, R.; Cui, Y.; Wang, Y.M.; Yue, S.Q.; Pei, J.M. Resveratrol attenuates ischemia/reperfusion injury in neonatal cardiomyocytes and its underlying mechanism. PLoS ONE 2012, 7, e51223. [Google Scholar] [CrossRef] [PubMed]
- Xi, J.; Wang, H.; Mueller, R.A.; Norfleet, E.A.; Xu, Z. Mechanism for resveratrol-induced cardioprotection against reperfusion injury involves glycogen synthase kinase 3beta and mitochondrial permeability transition pore. Eur. J. Pharmacol. 2009, 604, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.T.; Kwon, D.Y.; Park, O.J.; Kim, M.S. Resveratrol protects ros-induced cell death by activating ampk in h9c2 cardiac muscle cells. Genes Nutr. 2008, 2, 323–326. [Google Scholar] [CrossRef] [PubMed]
- Bradamante, S.; Barenghi, L.; Piccinini, F.; Bertelli, A.A.; De Jonge, R.; Beemster, P.; De Jong, J.W. Resveratrol provides late-phase cardioprotection by means of a nitric oxide- and adenosine-mediated mechanism. Eur. J. Pharmacol. 2003, 465, 115–123. [Google Scholar] [CrossRef]
- Hattori, R.; Otani, H.; Maulik, N.; Das, D.K. Pharmacological preconditioning with resveratrol: Role of nitric oxide. Am. J. Physiol. Heart Circ. Physiol. 2002, 282, H1988–H1995. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.; Jia, G.L.; Wang, Y.M.; Ma, H. Cardioprotective effect of resvaratrol pretreatment on myocardial ischemia-reperfusion induced injury in rats. Vascul. Pharmacol. 2006, 45, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Zhang, L.; Li, F.; Hu, C.P.; Zhang, Z. Restoration of sirt1 function by pterostilbene attenuates hypoxia-reoxygenation injury in cardiomyocytes. Eur. J. Pharmacol. 2016, 776, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Tang, B.L. Sirt1 and the mitochondria. Mol. Cells 2016, 39, 87–95. [Google Scholar] [PubMed]
- Yu, Z.; Wang, S.; Zhang, X.; Li, Y.; Zhao, Q.; Liu, T. Pterostilbene protects against myocardial ischemia/reperfusion injury via suppressing oxidative/nitrative stress and inflammatory response. Int. Immunopharmacol. 2017, 43, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Lu, S.; Zhong, J.; Huang, K.; Zhang, S. Protective effects of pterostilbene against myocardial ischemia/reperfusion injury in rats. Inflammation 2017, 40, 578. [Google Scholar] [CrossRef] [PubMed]
- Lv, M.; Liu, K.; Fu, S.; Li, Z.; Yu, X. Pterostilbene attenuates the inflammatory reaction induced by ischemia/reperfusion in rat heart. Mol. Med. Rep. 2015, 11, 724–728. [Google Scholar] [CrossRef] [PubMed]
- Hung, L.M.; Chen, J.K.; Lee, R.S.; Liang, H.C.; Su, M.J. Beneficial effects of astringinin, a resveratrol analogue, on the ischemia and reperfusion damage in rat heart. Free Radic. Biol. Med. 2001, 30, 877–883. [Google Scholar] [CrossRef]
- Schwenke, D.C.; Carew, T.E. Initiation of atherosclerotic lesions in cholesterol-fed rabbits. Ii. Selective retention of ldl vs. Selective increases in ldl permeability in susceptible sites of arteries. Arteriosclerosis 1989, 9, 908–918. [Google Scholar] [CrossRef] [PubMed]
- Folcik, V.A.; Nivar-Aristy, R.A.; Krajewski, L.P.; Cathcart, M.K. Lipoxygenase contributes to the oxidation of lipids in human atherosclerotic plaques. J. Clin. Investig. 1995, 96, 504–510. [Google Scholar] [CrossRef] [PubMed]
- Staprãns, I.; Rapp, J.H.; Pan, X.M.; Hardman, D.A.; Feingold, K.R. Oxidized lipids in the diet accelerate the development of fatty streaks in cholesterol-fed rabbits. Arterioscler. Thromb. Vasc. Biol. 1996, 16, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Miyagi, Y.; Miwa, K.; Inoue, H. Inhibition of human low-density lipoprotein oxidation by flavonoids in red wine and grape juice. Am. J. Cardiol. 1997, 80, 1627–1631. [Google Scholar] [CrossRef]
- Ou, H.C.; Chou, F.P.; Sheen, H.M.; Lin, T.M.; Yang, C.H.; Huey-Herng Sheu, W. Resveratrol, a polyphenolic compound in red wine, protects against oxidized ldl-induced cytotoxicity in endothelial cells. Clin. Chim. Acta 2006, 364, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Voloshyna, I.; Hai, O.; Littlefield, M.J.; Carsons, S.; Reiss, A.B. Resveratrol mediates anti-atherogenic effects on cholesterol flux in human macrophages and endothelium via pparγ and adenosine. Eur. J. Pharmacol. 2013, 698, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Zang, M.; Xu, S.; Maitland-Toolan, K.A.; Zuccollo, A.; Hou, X.; Jiang, B.; Wierzbicki, M.; Verbeuren, T.J.; Cohen, R.A. Polyphenols stimulate amp-activated protein kinase, lower lipids, and inhibit accelerated atherosclerosis in diabetic ldl receptor-deficient mice. Diabetes 2006, 55, 2180–2191. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Zorita, S.; Tréguer, K.; Mercader, J.; Carpéné, C. Resveratrol directly affects in vitro lipolysis and glucose transport in human fat cells. J. Physiol. Biochem. 2013, 69, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.C.; Chen, T.G.; Tai, Y.T.; Chen, T.L.; Chiu, W.T.; Chen, R.M. Resveratrol attenuates oxidized ldl-evoked lox-1 signaling and consequently protects against apoptotic insults to cerebrovascular endothelial cells. J. Cereb. Blood Flow Metab. 2011, 31, 842–854. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.L.; Chang, H.C.; Chen, T.L.; Chang, J.H.; Chiu, W.T.; Lin, J.W.; Chen, R.M. Resveratrol protects against oxidized ldl-induced breakage of the blood-brain barrier by lessening disruption of tight junctions and apoptotic insults to mouse cerebrovascular endothelial cells. J. Nutr. 2010, 140, 2187–2192. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhou, G.; Song, W.; Tan, X.; Guo, Y.; Zhou, B.; Jing, H.; Zhao, S.; Chen, L. Pterostilbene protects vascular endothelial cells against oxidized low-density lipoprotein-induced apoptosis in vitro and in vivo. Apoptosis 2012, 17, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Cui, L.; Zhou, G.; Jing, H.; Guo, Y.; Sun, W. Pterostilbene, a natural small-molecular compound, promotes cytoprotective macroautophagy in vascular endothelial cells. J. Nutr. Biochem. 2013, 24, 903–911. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. Pterostilbene, a novel natural plant conduct, inhibits high fat-induced atherosclerosis inflammation via nf-κb signaling pathway in toll-like receptor 5 (tlr5) deficient mice. Biomed. Pharmacother. 2016, 81, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Park, E.S.; Lim, Y.; Hong, J.T.; Yoo, H.S.; Lee, C.K.; Pyo, M.Y.; Yun, Y.P. Pterostilbene, a natural dimethylated analog of resveratrol, inhibits rat aortic vascular smooth muscle cell proliferation by blocking akt-dependent pathway. Vascul. Pharmacol. 2010, 53, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Llarena, M.; Andrade, F.; Hasnaoui, M.; Portillo, M.P.; Pérez-Matute, P.; Arbones-Mainar, J.M.; Hijona, E.; Villanueva-Millán, M.J.; Aguirre, L.; Carpéné, C.; et al. Potential renoprotective effects of piceatannol in ameliorating the early-stage nephropathy associated with obesity in obese zucker rats. J. Physiol. Biochem. 2016, 72, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Uchida-Maruki, H.; Inagaki, H.; Ito, R.; Kurita, I.; Sai, M.; Ito, T. Piceatannol lowers the blood glucose level in diabetic mice. Biol. Pharm. Bull. 2015, 38, 629–633. [Google Scholar] [CrossRef] [PubMed]
- Chan, A.Y.; Dolinsky, V.W.; Soltys, C.L.; Viollet, B.; Baksh, S.; Light, P.E.; Dyck, J.R. Resveratrol inhibits cardiac hypertrophy via amp-activated protein kinase and akt. J. Biol. Chem. 2008, 283, 24194–24201. [Google Scholar] [CrossRef] [PubMed]
- Juric, D.; Wojciechowski, P.; Das, D.K.; Netticadan, T. Prevention of concentric hypertrophy and diastolic impairment in aortic-banded rats treated with resveratrol. Am. J. Physiol. Heart Circ. Physiol. 2007, 292, H2138–H2143. [Google Scholar] [CrossRef] [PubMed]
- Thandapilly, S.J.; Louis, X.L.; Yang, T.; Stringer, D.M.; Yu, L.; Zhang, S.; Wigle, J.; Kardami, E.; Zahradka, P.; Taylor, C.; et al. Resveratrol prevents norepinephrine induced hypertrophy in adult rat cardiomyocytes, by activating no-ampk pathway. Eur. J. Pharmacol. 2011, 668, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Wojciechowski, P.; Juric, D.; Louis, X.L.; Thandapilly, S.J.; Yu, L.; Taylor, C.; Netticadan, T. Resveratrol arrests and regresses the development of pressure overload- but not volume overload-induced cardiac hypertrophy in rats. J. Nutr. 2010, 140, 962–968. [Google Scholar] [CrossRef] [PubMed]
- Gealekman, O.; Abassi, Z.; Rubinstein, I.; Winaver, J.; Binah, O. Role of myocardial inducible nitric oxide synthase in contractile dysfunction and beta-adrenergic hyporesponsiveness in rats with experimental volume-overload heart failure. Circulation 2002, 105, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Dolinsky, V.W.; Chakrabarti, S.; Pereira, T.J.; Oka, T.; Levasseur, J.; Beker, D.; Zordoky, B.N.; Morton, J.S.; Nagendran, J.; Lopaschuk, G.D.; et al. Resveratrol prevents hypertension and cardiac hypertrophy in hypertensive rats and mice. Biochim. Biophys. Acta 2013, 1832, 1723–1733. [Google Scholar] [CrossRef] [PubMed]
- Thandapilly, S.J.; LeMaistre, J.L.; Louis, X.L.; Anderson, C.M.; Netticadan, T.; Anderson, H.D. Vascular and cardiac effects of grape powder in the spontaneously hypertensive rat. Am. J. Hypertens. 2012, 25, 1070–1076. [Google Scholar] [CrossRef] [PubMed]
- Akinwumi, B.C.; Raj, P.; Lee, D.I.; Acosta, C.; Yu, L.; Thomas, S.M.; Nagabhushanam, K.; Majeed, M.; Davies, N.M.; Netticadan, T.; et al. Disparate effects of stilbenoid polyphenols on hypertrophic cardiomyocytes in vitro vs. In the spontaneously hypertensive heart failure rat. Molecules 2017, 22, 204. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, J.; Conte, C.; Fontana, L.; Mittendorfer, B.; Imai, S.; Schechtman, K.B.; Gu, C.; Kunz, I.; Rossi Fanelli, F.; Patterson, B.W.; et al. Resveratrol supplementation does not improve metabolic function in nonobese women with normal glucose tolerance. Cell Metab. 2012, 16, 658–664. [Google Scholar] [CrossRef] [PubMed]
- Juan, M.E.; Vinardell, M.P.; Planas, J.M. The daily oral administration of high doses of trans-resveratrol to rats for 28 days is not harmful. J. Nutr. 2002, 132, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Tomé-Carneiro, J.; Gonzálvez, M.; Larrosa, M.; García-Almagro, F.J.; Avilés-Plaza, F.; Parra, S.; Yáñez-Gascón, M.J.; Ruiz-Ros, J.A.; García-Conesa, M.T.; Tomás-Barberán, F.A.; et al. Consumption of a grape extract supplement containing resveratrol decreases oxidized ldl and apob in patients undergoing primary prevention of cardiovascular disease: A triple-blind, 6-month follow-up, placebo-controlled, randomized trial. Mol. Nutr. Food Res. 2012, 56, 810–821. [Google Scholar] [CrossRef] [PubMed]
- Tomé-Carneiro, J.; Gonzálvez, M.; Larrosa, M.; Yáñez-Gascón, M.J.; García-Almagro, F.J.; Ruiz-Ros, J.A.; García-Conesa, M.T.; Tomás-Barberán, F.A.; Espín, J.C. One-year consumption of a grape nutraceutical containing resveratrol improves the inflammatory and fibrinolytic status of patients in primary prevention of cardiovascular disease. Am. J. Cardiol. 2012, 110, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Tomé-Carneiro, J.; Gonzálvez, M.; Larrosa, M.; Yáñez-Gascón, M.J.; García-Almagro, F.J.; Ruiz-Ros, J.A.; Tomás-Barberán, F.A.; García-Conesa, M.T.; Espín, J.C. Grape resveratrol increases serum adiponectin and downregulates inflammatory genes in peripheral blood mononuclear cells: A triple-blind, placebo-controlled, one-year clinical trial in patients with stable coronary artery disease. Cardiovasc. Drugs Ther. 2013, 27, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Rimando, A.M.; Nagmani, R.; Feller, D.R.; Yokoyama, W. Pterostilbene, a new agonist for the peroxisome proliferator-activated receptor alpha-isoform, lowers plasma lipoproteins and cholesterol in hypercholesterolemic hamsters. J. Agric. Food Chem. 2005, 53, 3403–3407. [Google Scholar] [CrossRef] [PubMed]
- Miura, D.; Miura, Y.; Yagasaki, K. Hypolipidemic action of dietary resveratrol, a phytoalexin in grapes and red wine, in hepatoma-bearing rats. Life Sci. 2003, 73, 1393–1400. [Google Scholar] [CrossRef]
- Sahebkar, A. Effects of resveratrol supplementation on plasma lipids: A systematic review and meta-analysis of randomized controlled trials. Nutr. Rev. 2013, 71, 822–835. [Google Scholar] [CrossRef] [PubMed]
- Szkudelski, T.; Szkudelska, K. Resveratrol and diabetes: From animal to human studies. Biochim. Biophys. Acta 2015, 1852, 1145–1154. [Google Scholar] [CrossRef] [PubMed]
- Penumathsa, S.V.; Thirunavukkarasu, M.; Zhan, L.; Maulik, G.; Menon, V.P.; Bagchi, D.; Maulik, N. Resveratrol enhances glut-4 translocation to the caveolar lipid raft fractions through ampk/akt/enos signalling pathway in diabetic myocardium. J. Cell Mol. Med. 2008, 12, 2350–2361. [Google Scholar] [CrossRef] [PubMed]
- Khazaei, M.; Karimi, J.; Sheikh, N.; Goodarzi, M.T.; Saidijam, M.; Khodadadi, I.; Moridi, H. Effects of resveratrol on receptor for advanced glycation end products (rage) expression and oxidative stress in the liver of rats with type 2 diabetes. Phytother. Res. 2016, 30, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Moridi, H.; Karimi, J.; Sheikh, N.; Goodarzi, M.T.; Saidijam, M.; Yadegarazari, R.; Khazaei, M.; Khodadadi, I.; Tavilani, H.; Piri, H.; et al. Resveratrol-dependent down-regulation of receptor for advanced glycation end-products and oxidative stress in kidney of rats with diabetes. Int. J. Endocrinol. Metab. 2015, 13, e23542. [Google Scholar] [CrossRef] [PubMed]
- Soufi, F.G.; Mohammad-Nejad, D.; Ahmadieh, H. Resveratrol improves diabetic retinopathy possibly through oxidative stress—Nuclear factor κb—Apoptosis pathway. Pharmacol. Rep. 2012, 64, 1505–1514. [Google Scholar] [CrossRef]
- Um, J.H.; Park, S.J.; Kang, H.; Yang, S.; Foretz, M.; McBurney, M.W.; Kim, M.K.; Viollet, B.; Chung, J.H. Amp-activated protein kinase-deficient mice are resistant to the metabolic effects of resveratrol. Diabetes 2010, 59, 554–563. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, J.K.; Thomas, S.; Nanjan, M.J. Resveratrol supplementation improves glycemic control in type 2 diabetes mellitus. Nutr. Res. 2012, 32, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Movahed, A.; Nabipour, I.; Lieben Louis, X.; Thandapilly, S.J.; Yu, L.; Kalantarhormozi, M.; Rekabpour, S.J.; Netticadan, T. Antihyperglycemic effects of short term resveratrol supplementation in type 2 diabetic patients. Evid.-Based Complement. Alternat. Med. 2013, 2013, 851267. [Google Scholar] [CrossRef] [PubMed]
- Thazhath, S.S.; Wu, T.; Bound, M.J.; Checklin, H.L.; Standfield, S.; Jones, K.L.; Horowitz, M.; Rayner, C.K. Administration of resveratrol for 5 wk has no effect on glucagon-like peptide 1 secretion, gastric emptying, or glycemic control in type 2 diabetes: A randomized controlled trial. Am. J. Clin. Nutr. 2016, 103, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Bo, S.; Ponzo, V.; Ciccone, G.; Evangelista, A.; Saba, F.; Goitre, I.; Procopio, M.; Pagano, G.F.; Cassader, M.; Gambino, R. Six months of resveratrol supplementation has no measurable effect in type 2 diabetic patients. A randomized, double blind, placebo-controlled trial. Pharmacol. Res. 2016, 111, 896–905. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Zorita, S.; Fernández-Quintela, A.; Aguirre, L.; Macarulla, M.T.; Rimando, A.M.; Portillo, M.P. Pterostilbene improves glycaemic control in rats fed an obesogenic diet: Involvement of skeletal muscle and liver. Food Funct. 2015, 6, 1968–1976. [Google Scholar] [CrossRef] [PubMed]
- Bhakkiyalakshmi, E.; Shalini, D.; Sekar, T.V.; Rajaguru, P.; Paulmurugan, R.; Ramkumar, K.M. Therapeutic potential of pterostilbene against pancreatic beta-cell apoptosis mediated through nrf2. Br. J. Pharmacol. 2014, 171, 1747–1757. [Google Scholar] [CrossRef] [PubMed]
- Manickam, M.; Ramanathan, M.; Jahromi, M.A.; Chansouria, J.P.; Ray, A.B. Antihyperglycemic activity of phenolics from pterocarpus marsupium. J. Nat. Prod. 1997, 60, 609–610. [Google Scholar] [CrossRef] [PubMed]
- Grover, J.K.; Vats, V.; Yadav, S.S. Pterocarpus marsupium extract (vijayasar) prevented the alteration in metabolic patterns induced in the normal rat by feeding an adequate diet containing fructose as sole carbohydrate. Diabetes Obes. Metab. 2005, 7, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Nemes-Nagy, E.; Szocs-Molnár, T.; Dunca, I.; Balogh-Sămărghiţan, V.; Hobai, S.; Morar, R.; Pusta, D.L.; Crăciun, E.C. Effect of a dietary supplement containing blueberry and sea buckthorn concentrate on antioxidant capacity in type 1 diabetic children. Acta Physiol. Hung. 2008, 95, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Habanova, M.; Haban, M.; Kobidova, R.; Schwarzova, M.; Gazo, J. Analysis of biologically active substances in bilberry (Vaccinium myrtillus l.) in selected natural localities of Slovak Republic. J. Cent. Eur. Agric. 2013, 14, 1210–1219. [Google Scholar] [CrossRef]
- Yang, Y.; Fan, C.; Wang, B.; Ma, Z.; Wang, D.; Gong, B.; Di, S.; Jiang, S.; Li, Y.; Li, T.; et al. Pterostilbene attenuates high glucose-induced oxidative injury in hippocampal neuronal cells by activating nuclear factor erythroid 2-related factor 2. Biochim. Biophys. Acta 2017, 1863, 827–837. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Liu, H.; Yue, L.; Li, X.; Zhao, L.; Yang, X.; Wang, X.; Yang, Y.; Qu, Y. Neuroprotective effects of pterostilbene against oxidative stress injury: Involvement of nuclear factor erythroid 2-related factor 2 pathway. Brain Res. 2016, 1643, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Su, Q.; Pu, H.; Hu, C. Neuroprotection by combination of resveratrol and enriched environment against ischemic brain injury in rats. Neurol. Res. 2016, 38, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Gao, H.; Zhang, W.; Wang, Y. Resveratrol alleviates nerve injury after cerebral ischemia and reperfusion in mice by inhibiting inflammation and apoptosis. Int. J. Clin. Exp. Med. 2015, 8, 3219–3226. [Google Scholar] [PubMed]
- Ruszkiewicz, J.; Albrecht, J. Changes in the mitochondrial antioxidant systems in neurodegenerative diseases and acute brain disorders. Neurochem. Int. 2015, 88, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.T.; Ko, M.C.; Chen, B.Y.; Huang, J.C.; Hsieh, C.W.; Lee, M.C.; Chiou, R.Y.; Wung, B.S.; Peng, C.H.; Yang, Y.L. Neuroprotective effects of resveratrol on mptp-induced neuron loss mediated by free radical scavenging. J. Agric. Food Chem. 2008, 56, 6910–6913. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Shi, J.S.; Zhou, H.; Wilson, B.; Hong, J.S.; Gao, H.M. Resveratrol protects dopamine neurons against lipopolysaccharide-induced neurotoxicity through its anti-inflammatory actions. Mol. Pharmacol. 2010, 78, 466–477. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Chen, W.D.; Wang, Y.D. B-amyloid: The key peptide in the pathogenesis of alzheimer’s disease. Front. Pharmacol. 2015, 6, 221. [Google Scholar] [CrossRef] [PubMed]
- Butterfield, D.A.; Lauderback, C.M. Lipid peroxidation and protein oxidation in alzheimer’s disease brain: Potential causes and consequences involving amyloid beta-peptide-associated free radical oxidative stress. Free Radic. Biol. Med. 2002, 32, 1050–1060. [Google Scholar] [CrossRef]
- Marambaud, P.; Zhao, H.; Davies, P. Resveratrol promotes clearance of alzheimer’s disease amyloid-beta peptides. J. Biol. Chem. 2005, 280, 37377–37382. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Fan, C.; Chen, N.; Huang, J.; Yang, Q. Resveratrol pretreatment attenuates cerebral ischemic injury by upregulating expression of transcription factor nrf2 and ho-1 in rats. Neurochem. Res. 2011, 36, 2352–2362. [Google Scholar] [CrossRef] [PubMed]
- Simão, F.; Matté, A.; Pagnussat, A.S.; Netto, C.A.; Salbego, C.G. Resveratrol prevents ca1 neurons against ischemic injury by parallel modulation of both gsk-3β and creb through pi3-k/akt pathways. Eur. J. Neurosci. 2012, 36, 2899–2905. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Sun, Z.; Liu, Y.; Jia, Y.; Zhang, B.; Zhang, J. Resveratrol improves cognition and reduces oxidative stress in rats with vascular dementia. Neural. Regen. Res. 2013, 8, 2050–2059. [Google Scholar] [PubMed]
- Naik, B.; Nirwane, A.; Majumdar, A. Pterostilbene ameliorates intracerebroventricular streptozotocin induced memory decline in rats. Cogn. Neurodyn. 2017, 11, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Ban, J.Y.; Jeon, S.Y.; Nguyen, T.T.; Bae, K.; Song, K.S.; Seong, Y.H. Neuroprotective effect of oxyresveratrol from smilacis chinae rhizome on amyloid beta protein (25–35)-induced neurotoxicity in cultured rat cortical neurons. Biol. Pharm. Bull. 2006, 29, 2419–2424. [Google Scholar] [CrossRef] [PubMed]
- Andrabi, S.A.; Spina, M.G.; Lorenz, P.; Ebmeyer, U.; Wolf, G.; Horn, T.F. Oxyresveratrol (trans-2,3′,4,5′-tetrahydroxystilbene) is neuroprotective and inhibits the apoptotic cell death in transient cerebral ischemia. Brain Res. 2004, 1017, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Sermboonpaisarn, T.; Sawasdee, P. Potent and selective butyrylcholinesterase inhibitors from ficus foveolata. Fitoterapia 2012, 83, 780–784. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.S.; Thomas, R.G.; Craft, S.; van Dyck, C.H.; Mintzer, J.; Reynolds, B.A.; Brewer, J.B.; Rissman, R.A.; Raman, R.; Aisen, P.S.; et al. A randomized, double-blind, placebo-controlled trial of resveratrol for alzheimer disease. Neurology 2015, 85, 1383–1391. [Google Scholar] [CrossRef] [PubMed]
- Witte, A.V.; Kerti, L.; Margulies, D.S.; Flöel, A. Effects of resveratrol on memory performance, hippocampal functional connectivity, and glucose metabolism in healthy older adults. J. Neurosci. 2014, 34, 7862–7870. [Google Scholar] [CrossRef] [PubMed]
- Evans, H.M.; Howe, P.R.; Wong, R.H. Effects of resveratrol on cognitive performance, mood and cerebrovascular function in post-menopausal women; a 14-week randomised placebo-controlled intervention trial. Nutrients 2017, 9, 27. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.H.; Raederstorff, D.; Howe, P.R. Acute resveratrol consumption improves neurovascular coupling capacity in adults with type 2 diabetes mellitus. Nutrients 2016, 8, 425. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.H.; Nealon, R.S.; Scholey, A.; Howe, P.R. Low dose resveratrol improves cerebrovascular function in type 2 diabetes mellitus. Nutr. Metab. Cardiovasc. Dis. 2016, 26, 393–399. [Google Scholar] [CrossRef] [PubMed]
- WHO. Obesity: Preventing and Managing the World Epidemic; World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- Baile, C.A.; Yang, J.Y.; Rayalam, S.; Hartzell, D.L.; Lai, C.Y.; Andersen, C.; Della-Fera, M.A. Effect of resveratrol on fat mobilization. Ann. N. Y. Acad. Sci. 2011, 1215, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Baur, J.A.; Pearson, K.J.; Price, N.L.; Jamieson, H.A.; Lerin, C.; Kalra, A.; Prabhu, V.V.; Allard, J.S.; Lopez-Lluch, G.; Lewis, K.; et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature 2006, 444, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Timmers, S.; Konings, E.; Bilet, L.; Houtkooper, R.H.; van de Weijer, T.; Goossens, G.H.; Hoeks, J.; van der Krieken, S.; Ryu, D.; Kersten, S.; et al. Calorie restriction-like effects of 30 days of resveratrol supplementation on energy metabolism and metabolic profile in obese humans. Cell Metab. 2011, 14, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Pearson, K.J.; Baur, J.A.; Lewis, K.N.; Peshkin, L.; Price, N.L.; Labinskyy, N.; Swindell, W.R.; Kamara, D.; Minor, R.K.; Perez, E.; et al. Resveratrol delays age-related deterioration and mimics transcriptional aspects of dietary restriction without extending life span. Cell Metab. 2008, 8, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Dal-Pan, A.; Blanc, S.; Aujard, F. Resveratrol suppresses body mass gain in a seasonal non-human primate model of obesity. BMC Physiol. 2010, 10, 11. [Google Scholar] [CrossRef] [PubMed]
- Dal-Pan, A.; Terrien, J.; Pifferi, F.; Botalla, R.; Hardy, I.; Marchal, J.; Zahariev, A.; Chery, I.; Zizzari, P.; Perret, M.; et al. Caloric restriction or resveratrol supplementation and ageing in a non-human primate: First-year outcome of the restrikal study in microcebus murinus. Age (Dordr) 2011, 33, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Howitz, K.T.; Bitterman, K.J.; Cohen, H.Y.; Lamming, D.W.; Lavu, S.; Wood, J.G.; Zipkin, R.E.; Chung, P.; Kisielewski, A.; Zhang, L.L.; et al. Small molecule activators of sirtuins extend saccharomyces cerevisiae lifespan. Nature 2003, 425, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Lagouge, M.; Argmann, C.; Gerhart-Hines, Z.; Meziane, H.; Lerin, C.; Daussin, F.; Messadeq, N.; Milne, J.; Lambert, P.; Elliott, P.; et al. Resveratrol improves mitochondrial function and protects against metabolic disease by activating sirt1 and pgc-1alpha. Cell 2006, 127, 1109–1122. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Zorita, S.; Fernández-Quintela, A.; Lasa, A.; Aguirre, L.; Rimando, A.M.; Portillo, M.P. Pterostilbene, a dimethyl ether derivative of resveratrol, reduces fat accumulation in rats fed an obesogenic diet. J. Agric. Food Chem. 2014, 62, 8731. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, L.; Milton-Laskibar, I.; Hijona, E.; Bujanda, L.; Rimando, A.M.; Portillo, M.P. Effects of pterostilbene in brown adipose tissue from obese rats. J. Physiol. Biochem. 2017, 73, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Nagao, K.; Jinnouchi, T.; Kai, S.; Yanagita, T. Pterostilbene, a dimethylated analog of resveratrol, promotes energy metabolism in obese rats. J. Nutr. Biochem. 2017, 43, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Christenson, J.; Whitby, S.J.; Mellor, D.; Thomas, J.; McKune, A.; Roach, P.D.; Naumovski, N. The effects of resveratrol supplementation in overweight and obese humans: A systematic review of randomized trials. Metab. Syndr. Relat. Disord. 2016, 14, 323–333. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, M.M.; Vestergaard, P.F.; Clasen, B.F.; Radko, Y.; Christensen, L.P.; Stødkilde-Jørgensen, H.; Møller, N.; Jessen, N.; Pedersen, S.B.; Jørgensen, J.O. High-dose resveratrol supplementation in obese men: An investigator-initiated, randomized, placebo-controlled clinical trial of substrate metabolism, insulin sensitivity, and body composition. Diabetes 2013, 62, 1186–1195. [Google Scholar] [CrossRef] [PubMed]
- Chachay, V.S.; Macdonald, G.A.; Martin, J.H.; Whitehead, J.P.; O’Moore-Sullivan, T.M.; Lee, P.; Franklin, M.; Klein, K.; Taylor, P.J.; Ferguson, M.; et al. Resveratrol does not benefit patients with nonalcoholic fatty liver disease. Clin. Gastroenterol. Hepatol. 2014, 12, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Méndez-del Villar, M.; González-Ortiz, M.; Martínez-Abundis, E.; Pérez-Rubio, K.G.; Lizárraga-Valdez, R. Effect of resveratrol administration on metabolic syndrome, insulin sensitivity, and insulin secretion. Metab. Syndr. Relat. Disord. 2014, 12, 497–501. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Quintela, A.; Carpéné, C.; Fernández, M.; Aguirre, L.; Milton-Laskibar, I.; Contreras, J.; Portillo, M.P. Anti-obesity effects of resveratrol: Comparison between animal models and humans. J. Physiol. Biochem. 2016, 73, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Chang, T.K.; Lee, W.B.; Ko, H.H. Trans-resveratrol modulates the catalytic activity and mrna expression of the procarcinogen-activating human cytochrome p450 1b1. Can. J. Physiol. Pharmacol. 2000, 78, 874–881. [Google Scholar] [CrossRef] [PubMed]
- Chun, Y.J.; Oh, Y.K.; Kim, B.J.; Kim, D.; Kim, S.S.; Choi, H.K.; Kim, M.Y. Potent inhibition of human cytochrome p450 1b1 by tetramethoxystilbene. Toxicol. Lett. 2009, 189, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Signorelli, P.; Ghidoni, R. Resveratrol as an anticancer nutrient: Molecular basis, open questions and promises. J. Nutr. Biochem. 2005, 16, 449–466. [Google Scholar] [CrossRef] [PubMed]
- Szaefer, H.; Cichocki, M.; Brauze, D.; Baer-Dubowska, W. Alteration in phase i and ii enzyme activities and polycyclic aromatic hydrocarbons-dna adduct formation by plant phenolics in mouse epidermis. Nutr. Cancer 2004, 48, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Whitlock, N.C.; Baek, S.J. The anticancer effects of resveratrol: Modulation of transcription factors. Nutr. Cancer 2012, 64, 493–502. [Google Scholar] [CrossRef] [PubMed]
- Carter, L.G.; D’Orazio, J.A.; Pearson, K.J. Resveratrol and cancer: Focus on in vivo evidence. Endocr.-Relat. Cancer 2014, 21, R209–R225. [Google Scholar] [CrossRef] [PubMed]
- McCormack, D.; McFadden, D. Pterostilbene and cancer: Current review. J. Surg. Res. 2012, 173, e53–e61. [Google Scholar] [CrossRef] [PubMed]
- Kosuru, R.; Rai, U.; Prakash, S.; Singh, A.; Singh, S. Promising therapeutic potential of pterostilbene and its mechanistic insight based on preclinical evidence. Eur. J. Pharmacol. 2016, 789, 229–243. [Google Scholar] [CrossRef] [PubMed]
- Pavan, A.R.; Silva, G.D.; Jornada, D.H.; Chiba, D.E.; Fernandes, G.F.; Man Chin, C.; Dos Santos, J.L. Unraveling the anticancer effect of curcumin and resveratrol. Nutrients 2016, 8, 628. [Google Scholar] [CrossRef] [PubMed]
- Nutakul, W.; Sobers, H.S.; Qiu, P.; Dong, P.; Decker, E.A.; McClements, D.J.; Xiao, H. Inhibitory effects of resveratrol and pterostilbene on human colon cancer cells: A side-by-side comparison. J. Agric. Food Chem. 2011, 59, 10964–10970. [Google Scholar] [CrossRef] [PubMed]
- Hagiwara, K.; Kosaka, N.; Yoshioka, Y.; Takahashi, R.U.; Takeshita, F.; Ochiya, T. Stilbene derivatives promote ago2-dependent tumour-suppressive microrna activity. Sci. Rep. 2012, 2, 314. [Google Scholar] [CrossRef] [PubMed]
- Kita, Y.; Miura, Y.; Yagasaki, K. Antiproliferative and anti-invasive effect of piceatannol, a polyphenol present in grapes and wine, against hepatoma ah109a cells. J. Biomed. Biotechnol. 2012, 2012, 672416. [Google Scholar] [CrossRef] [PubMed]
- Jayasooriya, R.G.; Lee, Y.G.; Kang, C.H.; Lee, K.T.; Choi, Y.H.; Park, S.Y.; Hwang, J.K.; Kim, G.Y. Piceatannol inhibits mmp-9-dependent invasion of tumor necrosis factor-α-stimulated du145 cells by suppressing the akt-mediated nuclear factor-κb pathway. Oncol. Lett. 2013, 5, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Ko, H.S.; Lee, H.J.; Kim, S.H.; Lee, E.O. Piceatannol suppresses breast cancer cell invasion through the inhibition of mmp-9: Involvement of pi3k/akt and nf-κb pathways. J. Agric. Food Chem. 2012, 60, 4083–4089. [Google Scholar] [CrossRef] [PubMed]
- Videira, I.F.; Moura, D.F.; Magina, S. Mechanisms regulating melanogenesis. An. Bras. Dermatol. 2013, 88, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Cestari, T.F.; Dantas, L.P.; Boza, J.C. Acquired hyperpigmentations. An. Bras. Dermatol. 2014, 89, 11–25. [Google Scholar] [CrossRef] [PubMed]
- Brożyna, A.A.; Jóźwicki, W.; Carlson, J.A.; Slominski, A.T. Melanogenesis affects overall and disease-free survival in patients with stage iii and iv melanoma. Hum. Pathol. 2013, 44, 2071–2074. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Kim, T.K.; Brożyna, A.A.; Janjetovic, Z.; Brooks, D.L.; Schwab, L.P.; Skobowiat, C.; Jóźwicki, W.; Seagroves, T.N. The role of melanogenesis in regulation of melanoma behavior: Melanogenesis leads to stimulation of hif-1α expression and hif-dependent attendant pathways. Arch. Biochem. Biophys. 2014, 563, 79–93. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Paus, R.; Mihm, M.C. Inhibition of melanogenesis as an adjuvant strategy in the treatment of melanotic melanomas: Selective review and hypothesis. Anticancer. Res. 1998, 18, 3709–3715. [Google Scholar] [PubMed]
- Lee, T.H.; Seo, J.O.; Baek, S.H.; Kim, S.Y. Inhibitory effects of resveratrol on melanin synthesis in ultraviolet b-induced pigmentation in guinea pig skin. Biomol. Ther. 2014, 22, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.S.; Hyun, C.G.; Lee, N.H.; Park, S.S.; Shin, D.B. Comparative depigmentation effects of resveratrol and its two methyl analogues in α-melanocyte stimulating hormone-triggered b16/f10 murine melanoma cells. Prev. Nutr. Food. Sci. 2016, 21, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.M.; Yun, J.; Lee, C.K.; Lee, H.; Min, K.R.; Kim, Y. Oxyresveratrol and hydroxystilbene compounds. Inhibitory effect on tyrosinase and mechanism of action. J. Biol. Chem. 2002, 277, 16340–16344. [Google Scholar] [CrossRef] [PubMed]
- Yokozawa, T.; Kim, Y.J. Piceatannol inhibits melanogenesis by its antioxidative actions. Biol. Pharm. Bull. 2007, 30, 2007–2011. [Google Scholar] [CrossRef] [PubMed]
| Stilbenoid | Oral Dose (mg/kg) | Half Life (h) | Oral Bioavailability (Rats) (%) |
|---|---|---|---|
| resveratrol | 50 | 1.48 [31] | 29.8 [31] |
| pterostilbene | 20 | 1.73 [32] | 80 [29] |
| gnetol | 100 | 4.2 [30] | 6.59 [30] |
| piceatannol | 10 | 4.23 [33] | 50.7 [34] |
| oxyresveratrol | 24.4 | – | 9.13 [35] |
| Study | Model | Stilbenoid | Dose | Result |
|---|---|---|---|---|
| Cardiovascular/Blood Pressure | ||||
| Behbahani | WKY rat | resveratrol | 2.5 mg/kg per day (for 10 weeks) | increased compliance and reduced wall stiffness in mesenteric small arteries |
| Thandapilly et al. [55] | SHR | resveratrol | 2.5 mg/kg per day (for 10 weeks) | prevention of developed concentric hypertrophy, systolic/diastolic dysfunction; no effect on blood pressure (BP) |
| Li et al. [56] | SHR | resveratrol | 200 mg/kg (for 4 weeks) | increased endothelial NO production; reduced BP |
| Riche et al. [59] | human | pterostilbene | 125 mg twice daily (for 6–8 weeks) | increased LDL cholesterol and reduced BP |
| Tome-Carneiro et al. [110] | human, stable coronary artery disease | grape extract + resveratrol | 8.1 mg/day (6 months); then 16.2 mg/day (6 months) | increased anti-inflammatory serum adiponectin, decreased thrombogenic PAI-1 |
| Platelet biology | ||||
| Olas et al. [63] | in vitro | resveratrol | 25–100 μg/mL | inhibition of adhesion of platelets to fibrinogen/collagen |
| I/R Injury | ||||
| Yu et al. [79] | SD rat (30 min ischemia; 3 h reperfusion) | pterostilbene | 10 mg/kg | reduced superoxide generation, MDA; increased SOD; reduced myocardial infarction and apoptosis |
| Hung et al. [82] | SD rat | piceatannol | 2.5 × 10−4 g/kg | reduced incidence and duration of ventricular tachycardia, ventricular fibrillation; prevention of mortality, increased NO and decreased LDH levels |
| Diabetes | ||||
| Um et al. [120] | AMPK subunit (α1/α2) deficient mice | resveratrol | 400 mg/kg per day (12 weeks) | AMPK dependent: increased insulin sensitivity, glucose tolerance and mitochondrial biogenesis |
| Bhatt et al. [121] | humans (with type II diabetis mellitus) | resveratrol | 250 mg/day for 3 months | improved HbA1c, systolic BP, total cholesterol and total protein |
| Gomez-Zorita et al. [125] | diabetic rat (induced by obesogenic diet) | pterostilbene | 15–30 mg/day for 6 weeks | improved glycaemic control due to increased hepatic glucokinase activity and skeletal muscle glucose uptake |
| Nemes-Nagy et al. [129] | human (children with T1DM) | blueberry and sea buckthorn concentrate | 3 × 1 comprimates per day for 2 months | increased SOD activity, decreased levels of glycated hemoglobin and increased C peptide concentration |
| Neurodegeneration | ||||
| Ren et al. [141] | SD rat | resveratrol | 15–30 mg/kg for 7 days | Reduced cerebral infarct volume, decreased MDA levels, restored SOD activity, increased Nrf2 and HO-1 and reduced caspase-3 expression |
| Ma et al. [143] | rat model (vascular dementia) | resveratrol | 25 mg/kg per day | decreased malonyldialdehyde levels; increased SOD activity and glutathione levels; improved learning and memory ability |
| Naik et al. [144] | SD rat (streptozotocin-induced memory deficit) | pterostilbene | 10–50 mg/kg per day for 13 days | improved memory and cognition; improved brain antioxidants [catalase, SOD, glutathione (GSH)]; improved cholinergic transmission. |
| Ban et al. [145] | SD rat | oxyresveratrol | 10 μM | inhibition of Aβ-induced neuronal cell death, elevation of cytosolic [Ca] and ROS generation |
| Evans et al. [150] | human (postmenopausal women) | resveratrol | 75 mg twice per day for 14 weeks | improved memory, mood and overall cognitive performance |
| Obesity | ||||
| Timmers et al. [156] | Human (obese) | resveratrol | 150 mg/day for 30 days | reduced sleeping and resting metabolic rate; in muscle, activated AMPK, increased SIRT1 and PGC-1α protein levels; decreased systolic BP and improved HOMA index |
| Aguirre et al. [163] | Zucker (fa/fa) rat model | pterostilbene | 15–30 mg/kg per day for 6 weeks | increased thermogenic and oxidative capacity of brown adipose tissue |
| Cancer | ||||
| Nutakul et al. [179] | human colon cancer cells | resveratrol and pterostilbene | 0–100 μM for between 30 min to 10 days | pterostilbene: more potent inhibitor of colony formation, stronger apoptosis-inducing effects, and 2–4-fold higher intracellular pterostilbene levels than resveratrol |
| Jayasooriya et al. [178] | human prostate cancer cells | piceatannol | 0–40 μM for 24 h | inhibition of TNF-α-induced invasion of cancer cells through suppression of MMP-9 activation via the Akt-mediated NF-ĸB pathway |
| Remsberg et al. [30] | SD rats | gnetol | 10–100 mg/kg per day, for 0–72 h | reductions in cell viability in cancer cell lines (i.e., colorectal cancer); activities in COX-1, COX-2, histone deacetylase and decreased inflammation |
| Pigmentation | ||||
| Lee et al. [189] | male guinea pig model | resveratrol | dissolved in ethanol/propylene glycol (3:7, v/v) | reduced expression of melanogenesis-related proteins; decreased hyperpigmentation in ultraviolet B-stimulated skin |
| Yoon et al. [190] | B16/F10 murine melanoma cells | pterostilbene and resveratrol trimethyl ether (RTE) | 10 μM for 48 h | inhibition of α-MSH-induced melanin synthesis, stronger downregulation of tyrosinase protein expression and α-MSH stimulated protein than RTE |
| Yokozawa et al. [192] | B16/F10 melanoma cells | piceatannol | 0–400 μM for 24 h | greater antityrosinase activity than kojic and resveratrol; down-regulation of melanin content, suppressed ROS generation and enhanced GSH/GSSG ratio |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akinwumi, B.C.; Bordun, K.-A.M.; Anderson, H.D. Biological Activities of Stilbenoids. Int. J. Mol. Sci. 2018, 19, 792. https://doi.org/10.3390/ijms19030792
Akinwumi BC, Bordun K-AM, Anderson HD. Biological Activities of Stilbenoids. International Journal of Molecular Sciences. 2018; 19(3):792. https://doi.org/10.3390/ijms19030792
Chicago/Turabian StyleAkinwumi, Bolanle C., Kimberly-Ann M. Bordun, and Hope D. Anderson. 2018. "Biological Activities of Stilbenoids" International Journal of Molecular Sciences 19, no. 3: 792. https://doi.org/10.3390/ijms19030792
APA StyleAkinwumi, B. C., Bordun, K.-A. M., & Anderson, H. D. (2018). Biological Activities of Stilbenoids. International Journal of Molecular Sciences, 19(3), 792. https://doi.org/10.3390/ijms19030792