Miniaturization of the Clonogenic Assay Using Confluence Measurement
Abstract
1. Introduction
2. Results
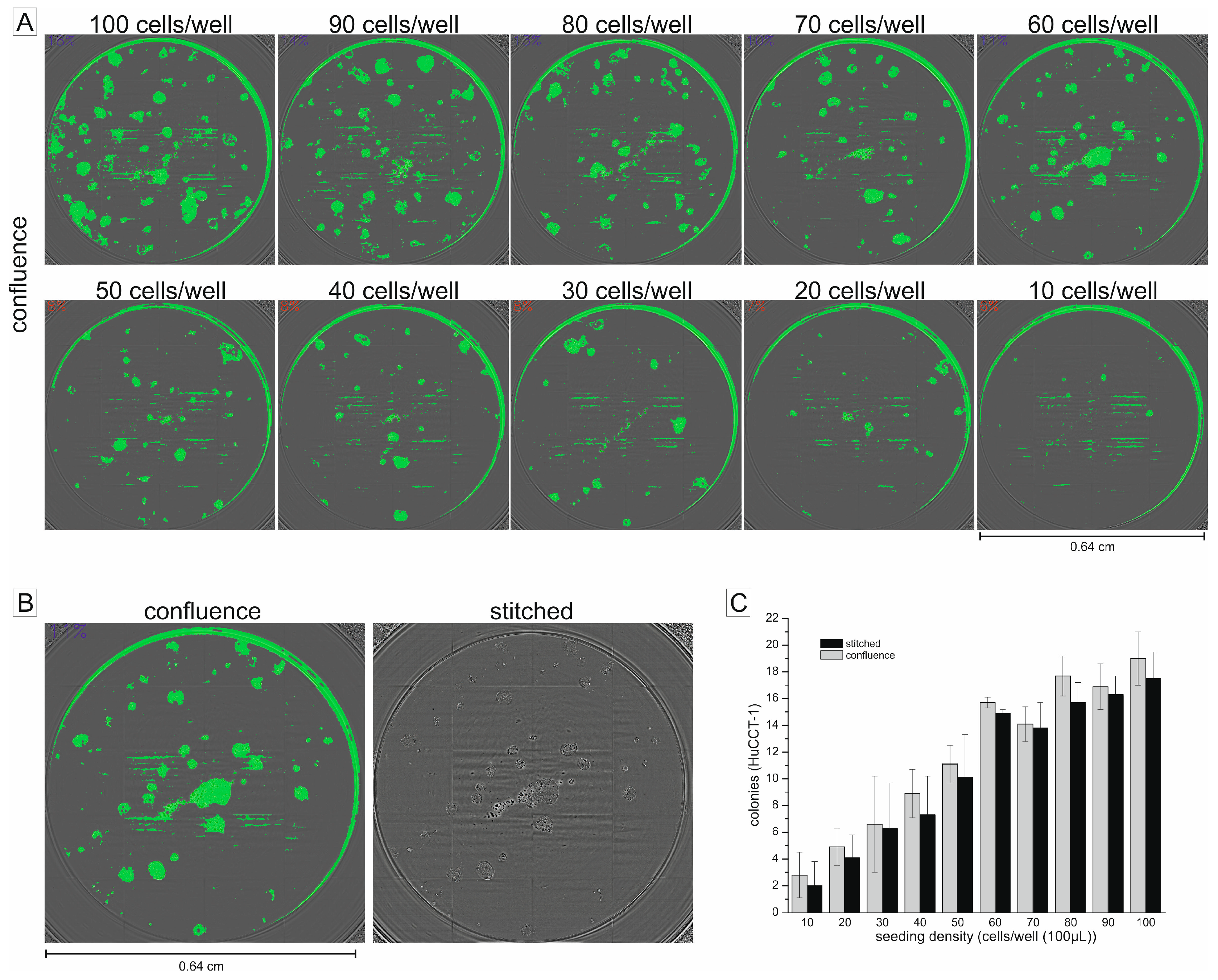
2.1. Using the Confluence Measurement to Translate the Clonogenic Assay in a 96-Well Microplate Format
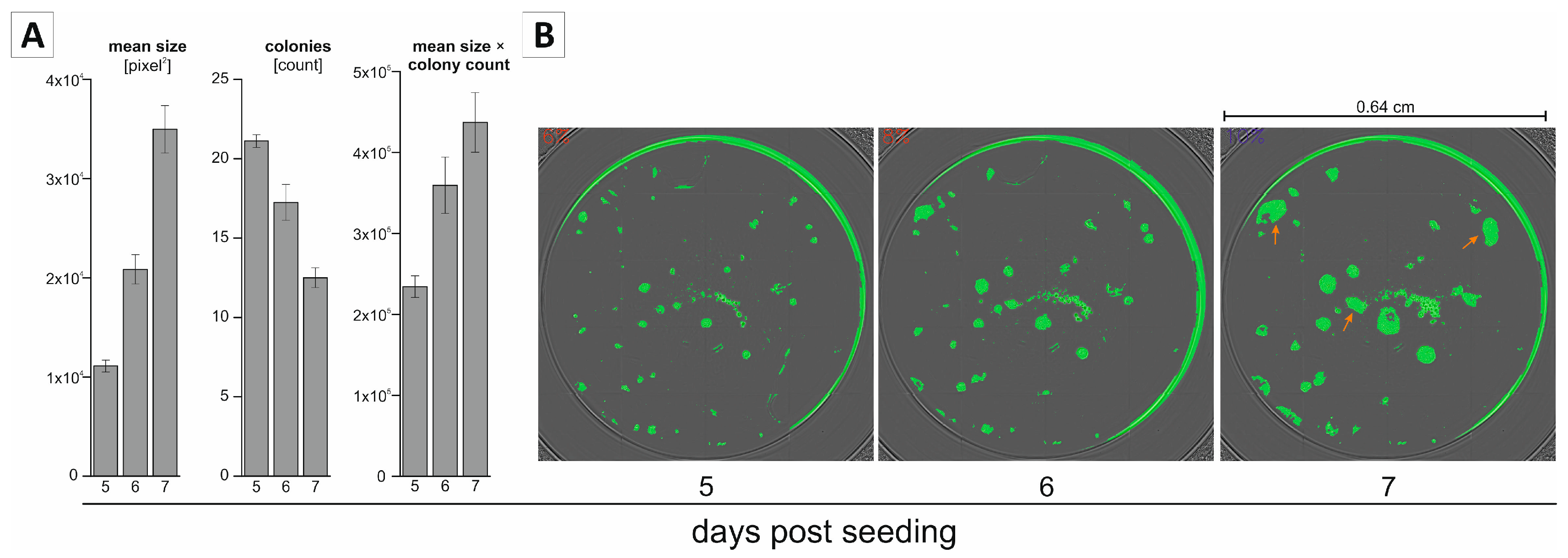
2.2. Non-Endpoint Analysis of Clonogenic Growth in the 96-Well Format
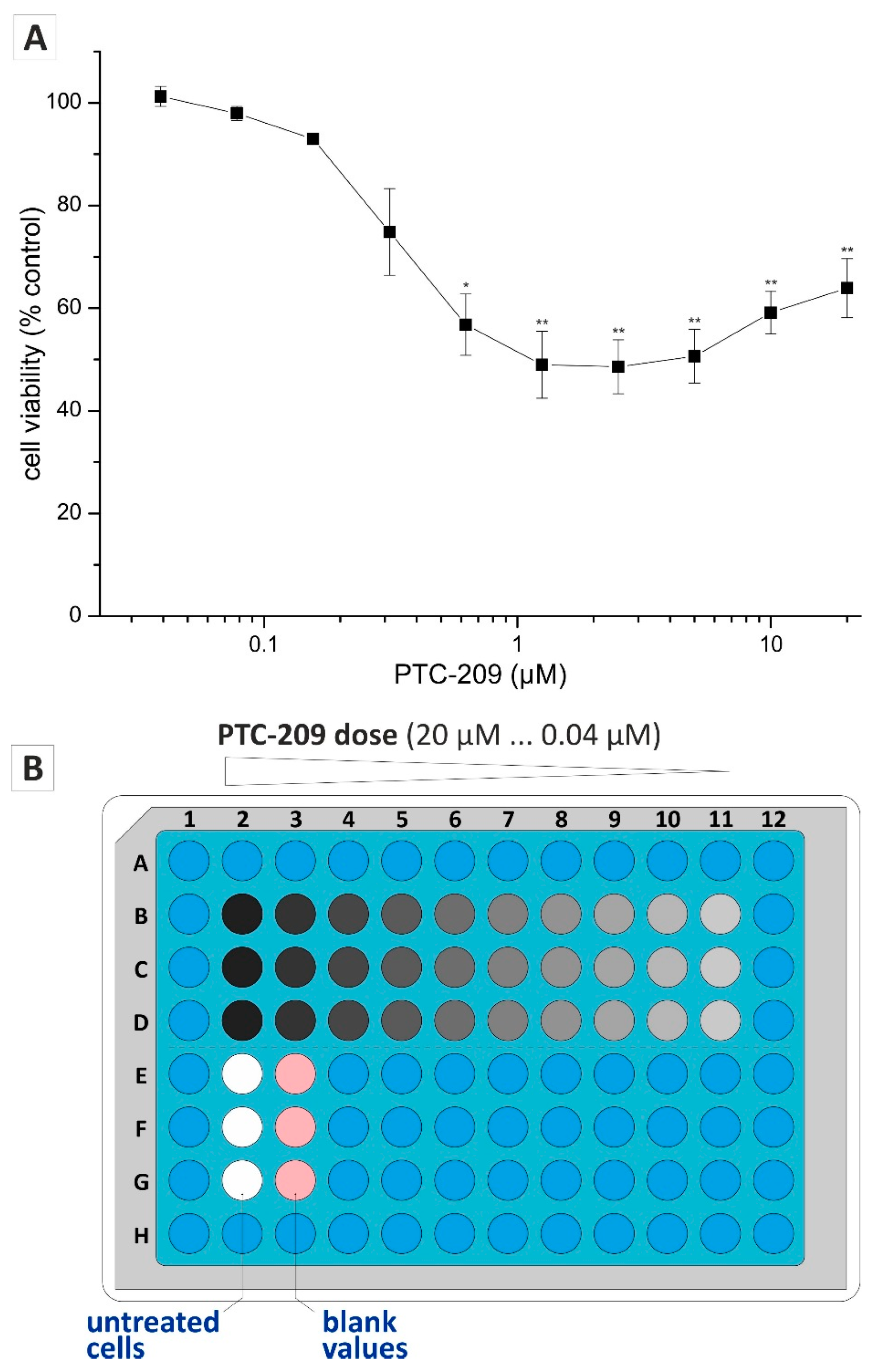
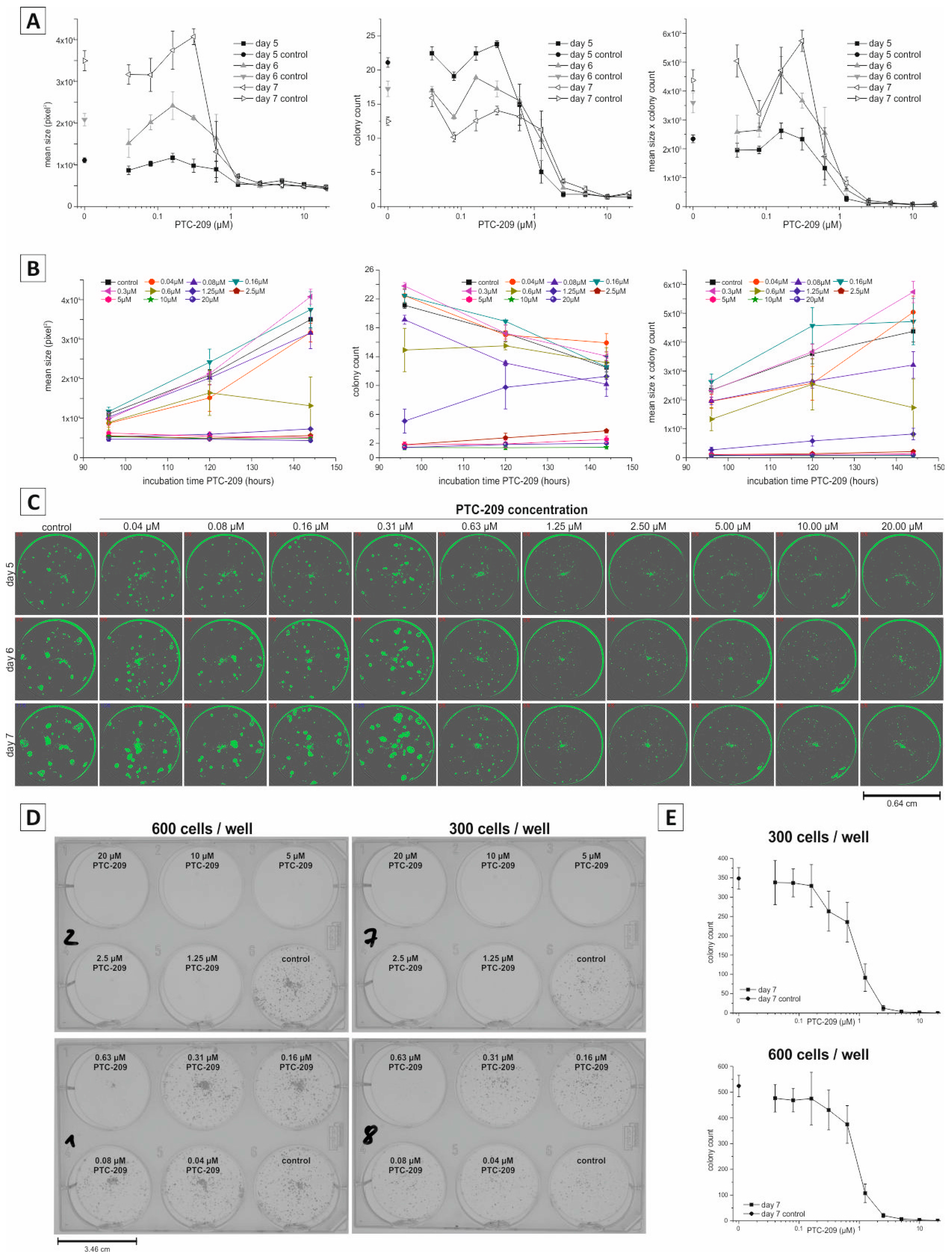
2.3. Application of Non-Endpoint Measurement—Dose-Dependent Effect of PTC-209 on Clonogenic Growth
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Substances
4.2. Clonogenic Assay
4.3. Drug Cytotoxicity
4.4. Statistics
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Puck, T.T.; Marcus, P.I. Action of x-rays on mammalian cells. J. Exp. Med. 1956, 103, 653–666. [Google Scholar] [CrossRef] [PubMed]
- Franken, N.A.; Rodermond, H.M.; Stap, J.; Haveman, J.; van Bree, C. Clonogenic assay of cells in vitro. Nat. Protoc. 2006, 1, 2315–2319. [Google Scholar] [CrossRef] [PubMed]
- Munshi, A.; Hobbs, M.; Meyn, R.E. Clonogenic cell survival assay. Methods Mol. Med. 2005, 110, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Buch, K.; Peters, T.; Nawroth, T.; Sanger, M.; Schmidberger, H.; Langguth, P. Determination of cell survival after irradiation via clonogenic assay versus multiple MTT Assay—A comparative study. Radiat. Oncol. 2012, 7, 1. [Google Scholar] [CrossRef] [PubMed]
- Buick, R.N.; MacKillop, W.J. Measurement of self-renewal in culture of clonogenic cells from human ovarian carcinoma. Br. J. Cancer 1981, 44, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Mayr, C.; Wagner, A.; Loeffelberger, M.; Bruckner, D.; Jakab, M.; Berr, F.; di Fazio, P.; Ocker, M.; Neureiter, D.; Pichler, M.; et al. The BMI1 inhibitor PTC-209 is a potential compound to halt cellular growth in biliary tract cancer cells. Oncotarget 2016, 7, 745–758. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.L.; Si, L.B.; Zeng, A.; Long, F.; Qi, Z.; Zhao, R.; Bai, M. MicroRNA-21 antisense oligonucleotide improves the sensitivity of A375 human melanoma cell to Cisplatin: an in vitro study. J. Cell. Biochem. 2017. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; He, M.; Yin, Y.; Yan, T.; Cheng, W.; Huang, Z.; Zhang, L.; Zhang, H.; Liu, P.; Zhu, W.; et al. miR-1296-5p decreases ERBB2 expression to inhibit the cell proliferation in ERBB2-positive breast cancer. Cancer Cell. Int. 2017, 17, 95. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Yoshida, T.; Okabe, M.; Zhou, K.; Wang, F.; Soko, C.; Saito, S.; Nikaido, T. Isolation of Stem-Like Cancer Cells in Primary Endometrial Cancer Using Cell Surface Markers CD133 and CXCR4. Transl. Oncol. 2017, 10, 976–987. [Google Scholar] [CrossRef] [PubMed]
- Schrag, D.; Garewal, H.S.; Burstein, H.J.; Samson, D.J.; von Hoff, D.D.; Somerfield, M.R.; ASCO Working Group on Chemotherapy Sensitivity; Resistance Assays. American Society of Clinical Oncology Technology Assessment: Chemotherapy sensitivity and resistance assays. J. Clin. Oncol. 2004, 22, 3631–3638. [Google Scholar] [CrossRef] [PubMed]
- Chumsri, S.; Phatak, P.; Edelman, M.J.; Khakpour, N.; Hamburger, A.W.; Burger, A.M. Cancer stem cells and individualized therapy. Cancer Genomics Proteomics 2007, 4, 165–174. [Google Scholar] [PubMed]
- Suraneni, M.V.; Badeaux, M.D. Tumor-Initiating Cells, Cancer Metastasis and Therapeutic Implications. In Madame Curie Bioscience Database; Landes Bioscience: Austin, TX, USA, 2000–2013. Available online: https://www.ncbi.nlm.nih.gov/books/NBK169227/ (accessed on 4 December 2017).
- Salmon, S.E.; Hamburger, A.W.; Soehnlen, B.; Durie, B.G.; Alberts, D.S.; Moon, T.E. Quantitation of differential sensitivity of human-tumor stem cells to anticancer drugs. N. Engl. J. Med. 1978, 298, 1321–1327. [Google Scholar] [CrossRef] [PubMed]
- Kreso, A.; Dick, J.E. Evolution of the cancer stem cell model. Cell Stem Cell 2014, 14, 275–291. [Google Scholar] [CrossRef] [PubMed]
- Franco, S.S.; Szczesna, K.; Iliou, M.S.; Al-Qahtani, M.; Mobasheri, A.; Kobolak, J.; Dinnyes, A. In vitro models of cancer stem cells and clinical applications. BMC Cancer 2016, 16, 738. [Google Scholar] [CrossRef] [PubMed]
- Rafehi, H.; Orlowski, C.; Georgiadis, G.T.; Ververis, K.; El-Osta, A.; Karagiannis, T.C. Clonogenic assay: Adherent cells. J. Vis. Exp. 2011. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Qiu, Q.; Zhang, X.; Jiang, Z.; Leng, Q.; Liu, Z.; Stass, S.A.; Jiang, F. Aldehyde dehydrogenase 1 A1-positive cell population is enriched in tumor-initiating cells and associated with progression of bladder cancer. Cancer Epidemiol. Biomarkers Prev. 2010, 19, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Charafe-Jauffret, E.; Ginestier, C.; Bertucci, F.; Cabaud, O.; Wicinski, J.; Finetti, P.; Josselin, E.; Adelaide, J.; Nguyen, T.T.; Monville, F.; et al. ALDH1-positive cancer stem cells predict engraftment of primary breast tumors and are governed by a common stem cell program. Cancer Res. 2013, 73, 7290–7300. [Google Scholar] [CrossRef] [PubMed]
- Douville, J.; Beaulieu, R.; Balicki, D. ALDH1 as a functional marker of cancer stem and progenitor cells. Stem Cells Dev. 2009, 18, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Weiswald, L.B.; Bellet, D.; Dangles-Marie, V. Spherical cancer models in tumor biology. Neoplasia 2015, 17, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Miyagiwa, M.; Ichida, T.; Tokiwa, T.; Sato, J.; Sasaki, H. A new human cholangiocellular carcinoma cell line (HuCC-T1) producing carbohydrate antigen 19/9 in serum-free medium. In Vitro Cell Dev. Biol. 1989, 25, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Wachter, J.; Neureiter, D.; Alinger, B.; Pichler, M.; Fuereder, J.; Oberdanner, C.; di Fazio, P.; Ocker, M.; Berr, F.; Kiesslich, T. Influence of five potential anticancer drugs on wnt pathway and cell survival in human biliary tract cancer cells. Int. J. Biol. Sci. 2012, 8, 15–29. [Google Scholar] [CrossRef] [PubMed][Green Version]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mayr, C.; Beyreis, M.; Dobias, H.; Gaisberger, M.; Pichler, M.; Ritter, M.; Jakab, M.; Neureiter, D.; Kiesslich, T. Miniaturization of the Clonogenic Assay Using Confluence Measurement. Int. J. Mol. Sci. 2018, 19, 724. https://doi.org/10.3390/ijms19030724
Mayr C, Beyreis M, Dobias H, Gaisberger M, Pichler M, Ritter M, Jakab M, Neureiter D, Kiesslich T. Miniaturization of the Clonogenic Assay Using Confluence Measurement. International Journal of Molecular Sciences. 2018; 19(3):724. https://doi.org/10.3390/ijms19030724
Chicago/Turabian StyleMayr, Christian, Marlena Beyreis, Heidemarie Dobias, Martin Gaisberger, Martin Pichler, Markus Ritter, Martin Jakab, Daniel Neureiter, and Tobias Kiesslich. 2018. "Miniaturization of the Clonogenic Assay Using Confluence Measurement" International Journal of Molecular Sciences 19, no. 3: 724. https://doi.org/10.3390/ijms19030724
APA StyleMayr, C., Beyreis, M., Dobias, H., Gaisberger, M., Pichler, M., Ritter, M., Jakab, M., Neureiter, D., & Kiesslich, T. (2018). Miniaturization of the Clonogenic Assay Using Confluence Measurement. International Journal of Molecular Sciences, 19(3), 724. https://doi.org/10.3390/ijms19030724






