Xyloglucan, a Plant Polymer with Barrier Protective Properties over the Mucous Membranes: An Overview
Abstract
1. Background
2. The Mucin Network and Mucosal Cell Integrity
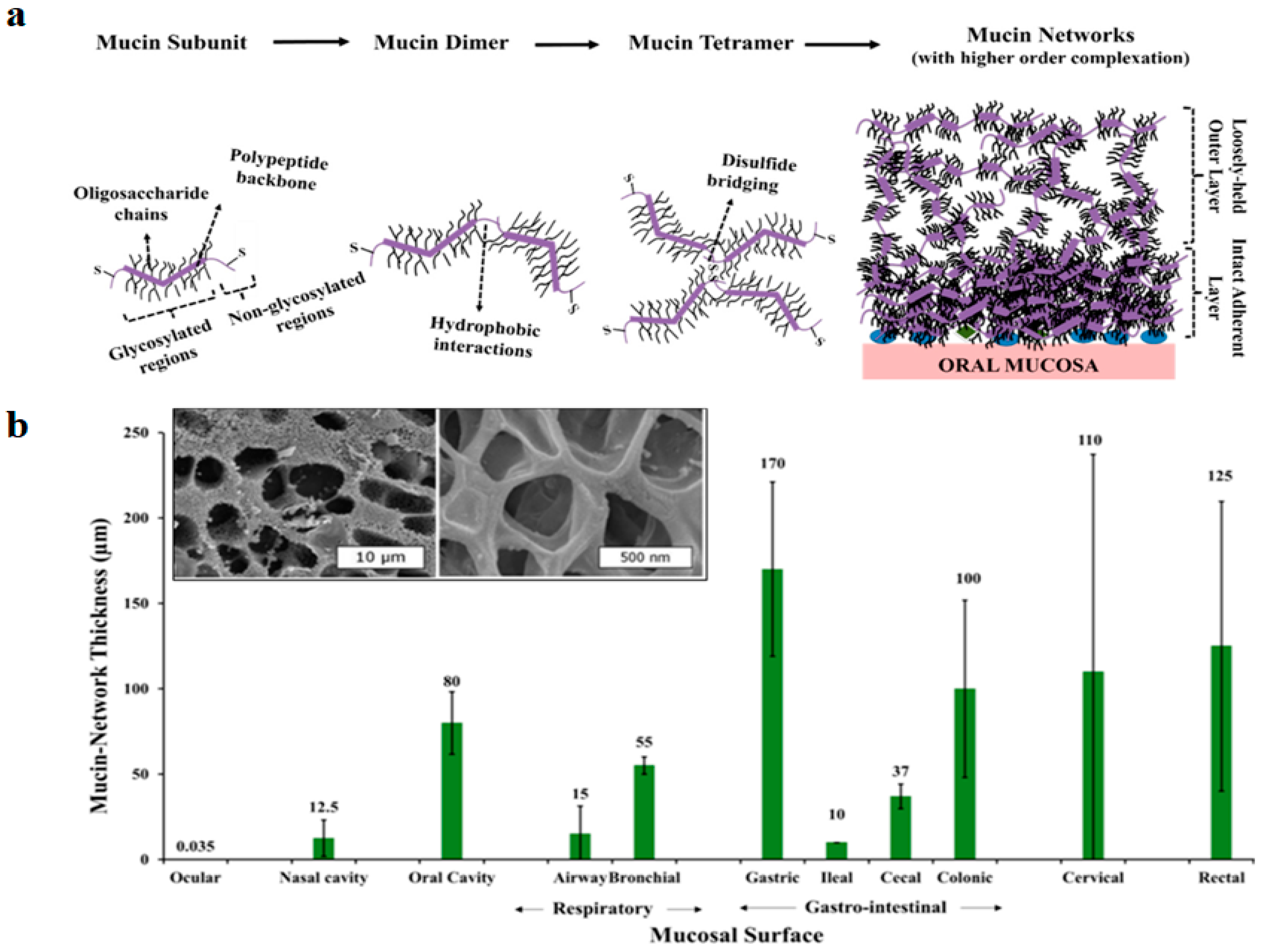
2.1. Mucin Network Thickness, Structure and Properties, Nasal and Intestinal Mucus
2.2. Glycans: Importance in Host-Microbial Interactions and Pathogenicity
2.3. Role of Tight Junctions on Mucosal Cell Integrity
2.4. Disruption of Mucosal Barrier Function as a Cause of Diseases
3. General Properties of Xyloglucan and Other Polymers
3.1. Structure
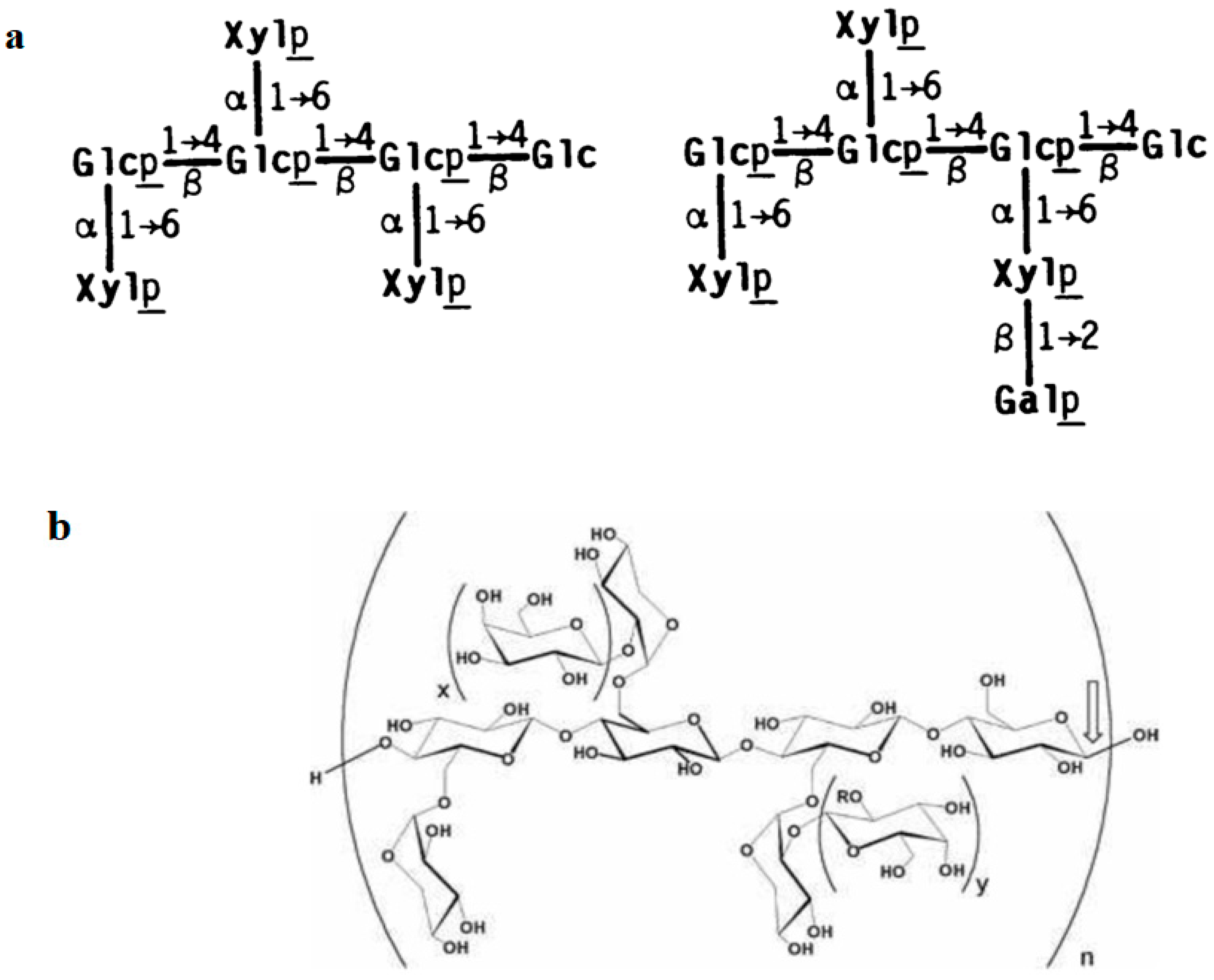
3.1.1. Structure of Xyloglucan
3.1.2. Structure of Synthetic Polymeric Mucin Analogues
3.1.3. Structure of other Mucosal Protectors: Reticulated Proteins
3.2. General Properties of Xyloglucan
3.2.1. Physicochemical Properties
3.2.2. Solubility
3.2.3. Assembly
3.2.4. Mucoadhesive Polymer
3.2.5. Viscosity
4. Barrier Properties as Novel Approaches for the Management of Bacterial Infections and Allergy
4.1. Anti-Adhesive and Anti-Invasion Properties
4.2. Inhibition of Bacterial Biofilms
4.3. Preservation of Tight Junctions (Dietary Fibres)
5. In Vitro Evidence for the Barrier Properties of Xyloglucan
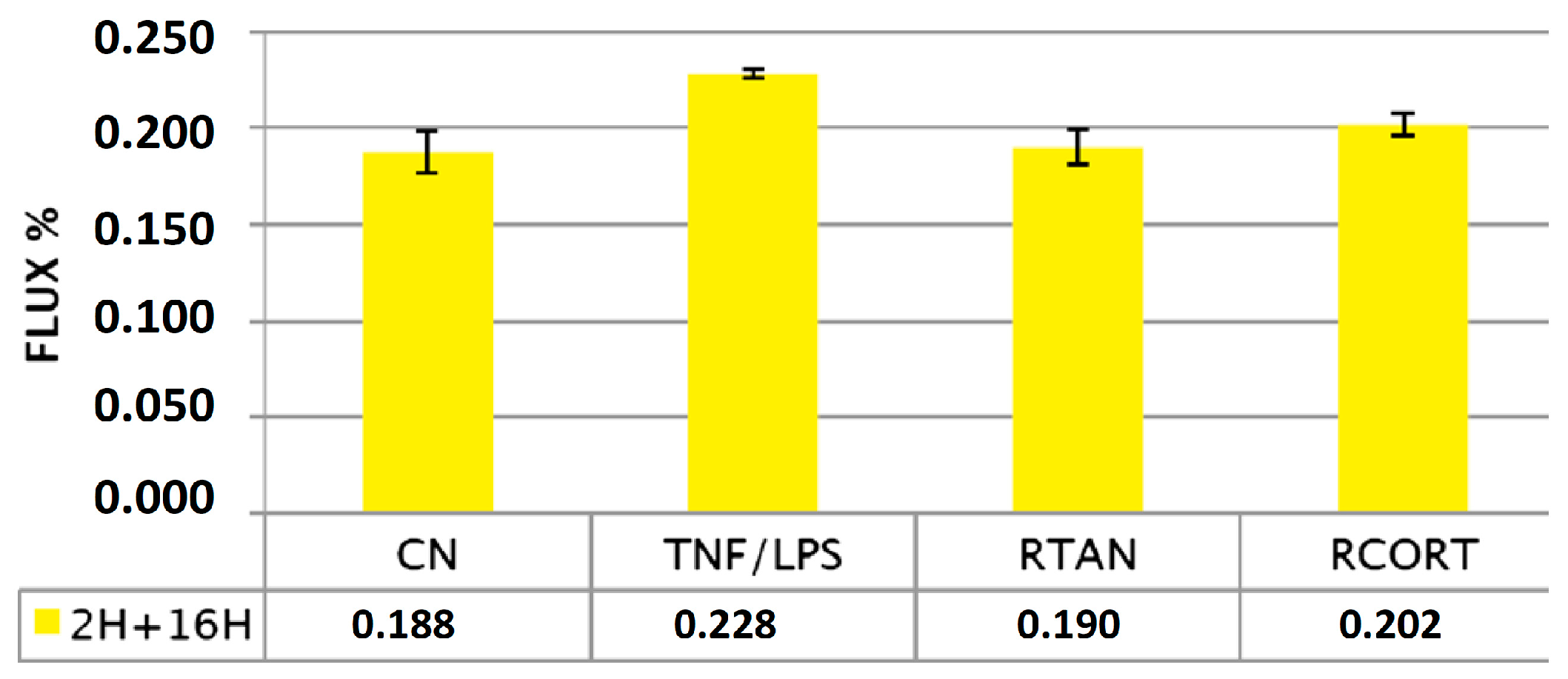
5.1. Effects of Xyloglucan to Preserve Tight Junctions and Paracellular Flux in Caco-Goblet Cells and MucilAir Cells
5.2. Anti-Adhesive Properties in Caco-Goblet Cells and Uroepithelial Cells
5.3. Anti-Invasive Properties in Caco-Goblet Cells
5.4. Wound Healing on Confluent Conjunctival Cells
5.5. Effects on Keratinocytes and Fibroblasts
6. Animal Studies with Xyloglucan and other Mucosal Protectors
6.1. Xyloglucan in a Model of Intestinal Injuries
6.2. Xyloglucan in a Model of Dry Eye
6.3. Xyloglucan in a Model of Corneal Lesion in Rabbits
6.4. Reticulated Protein, Hibiscus and Propolis to Reduce Intestinal E. Coli Load
7. Clinical Studies with Xyloglucan in Gastroenteritis
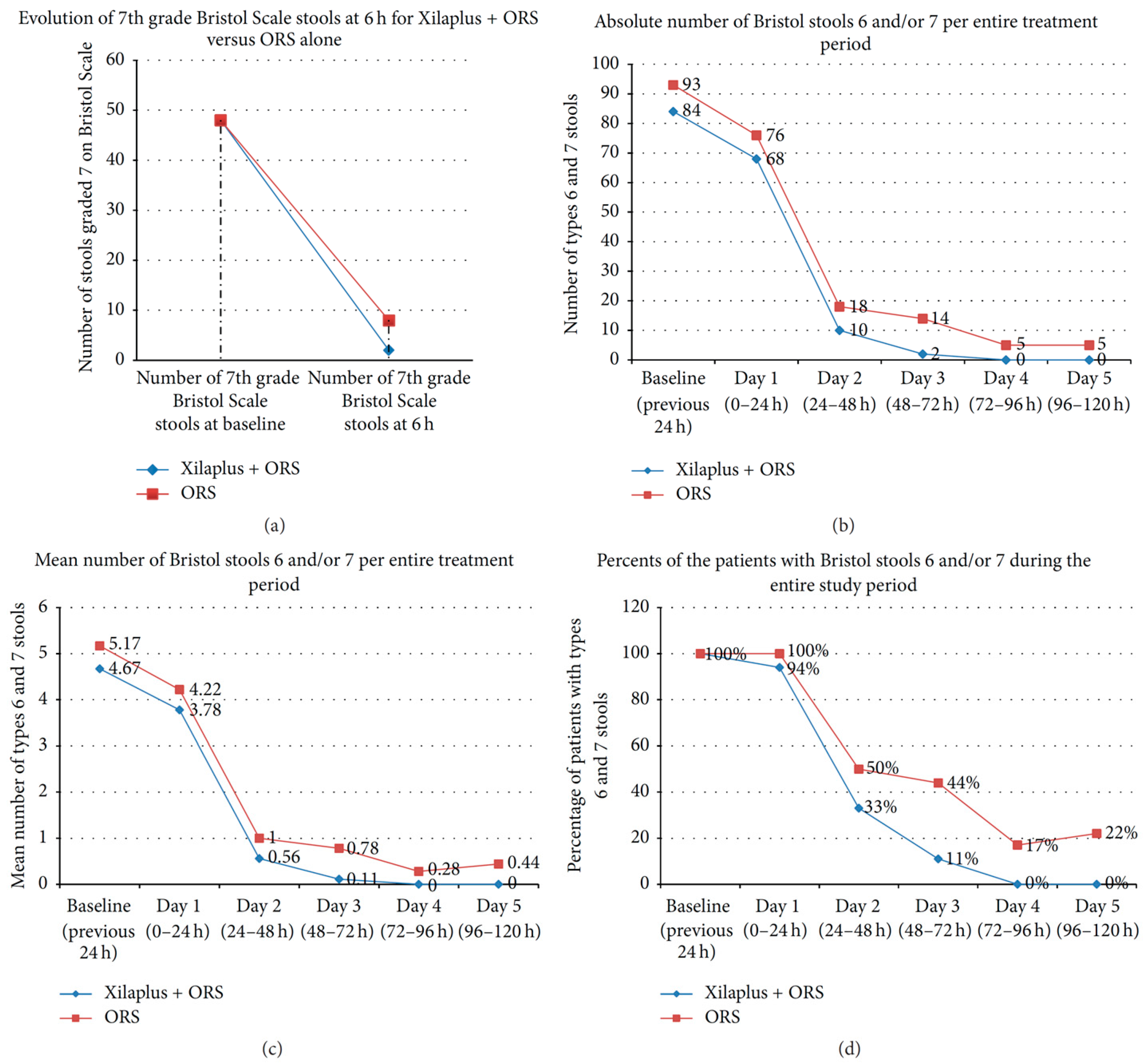
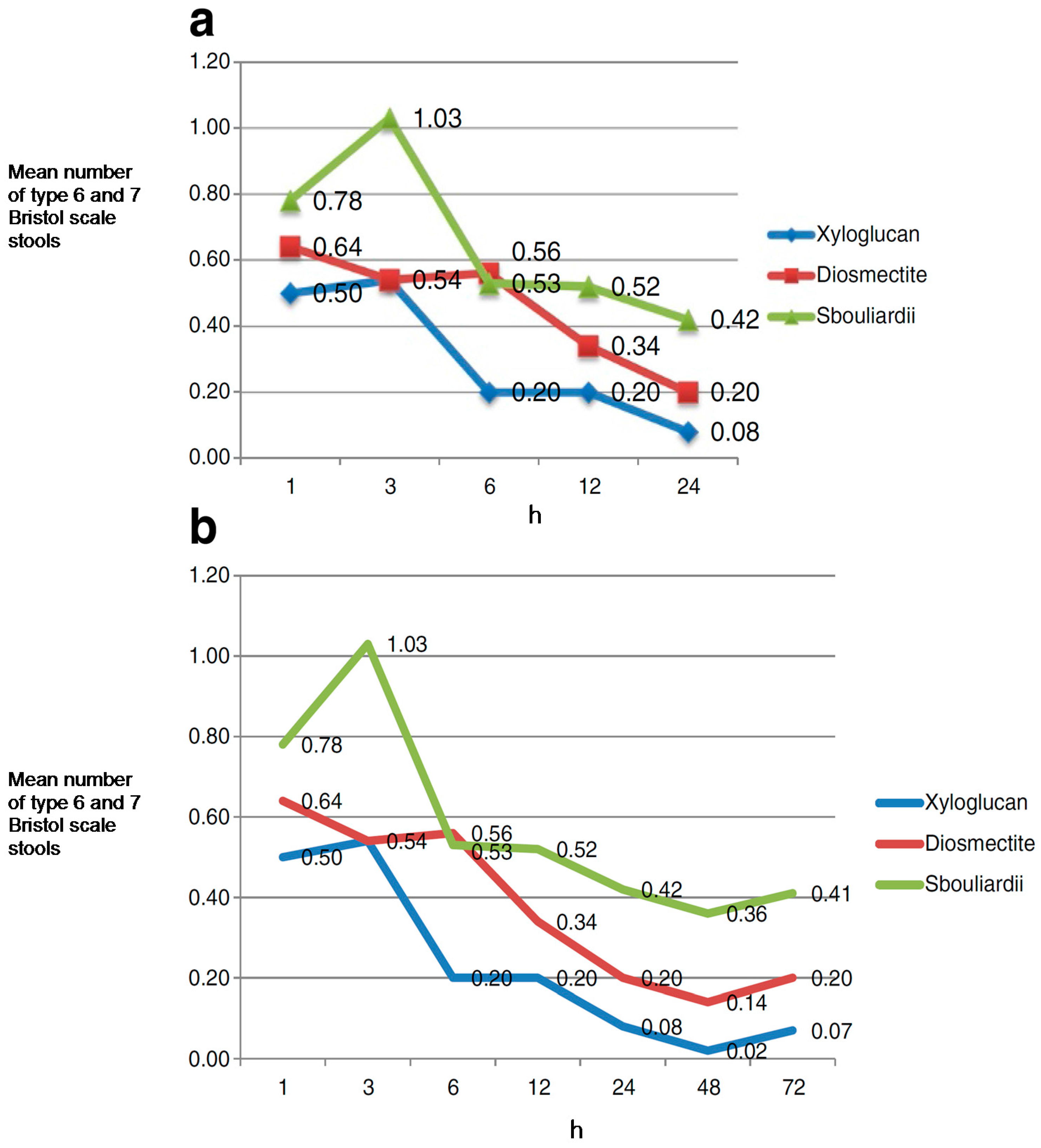
7.1. Gastroenteritis in Children
7.2. Gastroenteritis in Adults
8. Clinical Studies with Mucosal Protectors in Irritable Bowel Syndrome
9. Clinical Studies with Mucosal Protectors in Urinary Tract Infections
9.1. Early Treatment of Patients with Symptoms of Urinary Tract Infections
9.2. Prevention of Uncomplicated Cystitis Recurrences
10. Clinical Studies with Xyloglucan in Nasal Disorders
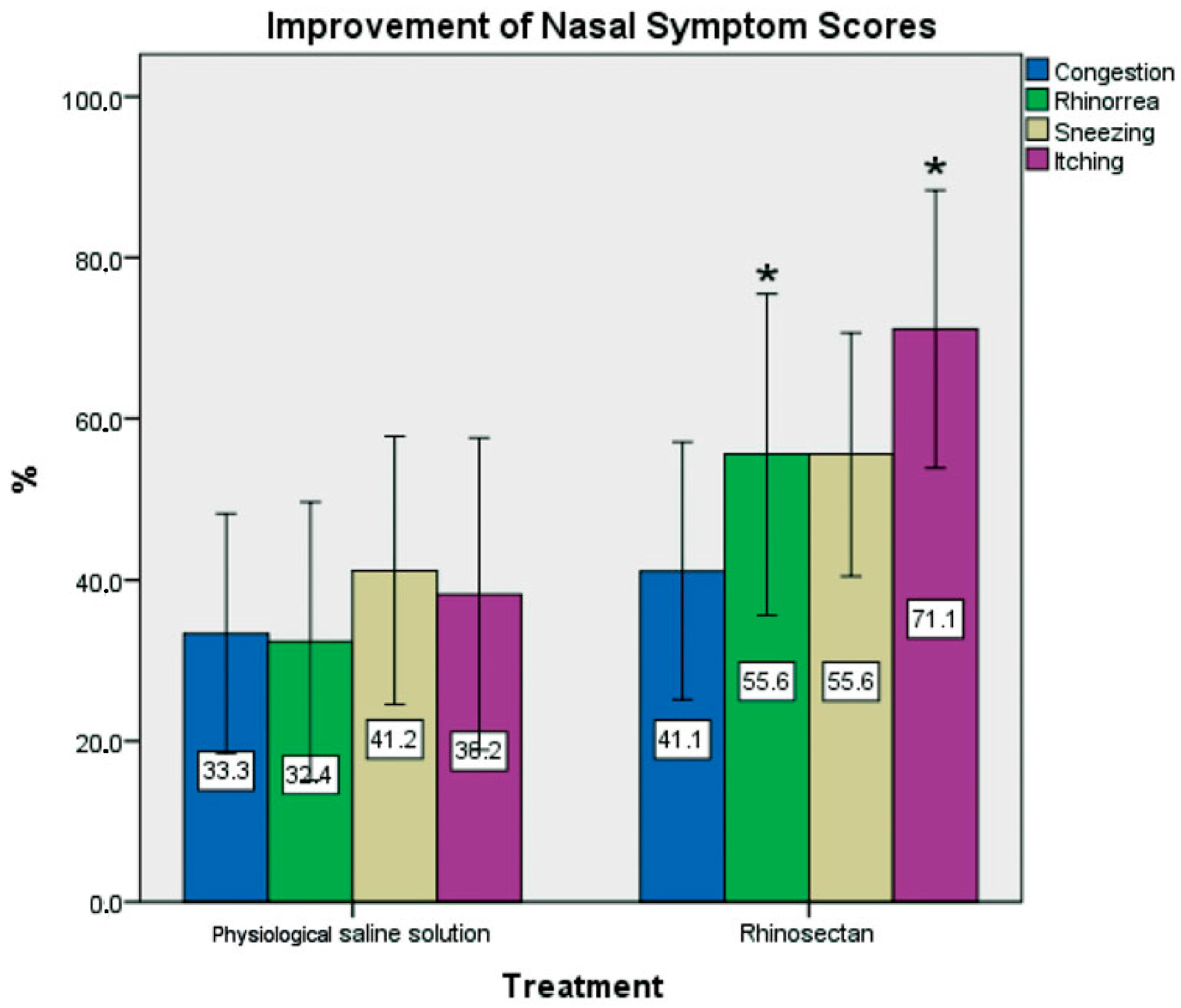
Xyloglucan-Based Nasal Spray in Rhinosinusitis
11. Clinical Studies with Xyloglucan in Ophthalmology
Xyloglucan for the Treatment of Dry Eye Syndrome
12. Clinical Studies with Xyloglucan in Infantile Colic
13. Concluding Remarks and Future Perspectives
Conflicts of Interest
References
- De Servi, B.; Ranzini, F.; Piqué, N. Protective barrier properties of Rhinosectan® spray (containing xyloglucan) on an organotypic 3D airway tissue model (MucilAir): Results of an vitro study. Allergy Asthma Clin. Immunol. 2017, 13, 37. [Google Scholar] [CrossRef] [PubMed]
- Oshima, T.; Miwa, H. Gastrointestinal mucosal barrier function and diseases. J. Gastroenterol. 2016, 51, 768–778. [Google Scholar] [CrossRef] [PubMed]
- Andersson, M.; Greiff, L.; Ojeda, P.; Wollmer, P. Barrier-enforcing measures as treatment principle in allergic rhinitis: A systematic review. Curr. Med. Res. Opin. 2014, 30, 1131–1137. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Al-Sadi, R.; Said, H.M.; Ma, T.Y. Lipopolysaccharide causes an increase in intestinal tight junction permeability in vitro and in vivo by inducing enterocyte membrane expression and localization of TLR-4 and CD14. Am. J. Pathol. 2013, 182, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Mattila, P.; Joenväärä, S.; Renkonen, J.; Toppila-Salmi, S.; Renkonen, R. Allergy as an epithelial barrier disease. Clin. Transl. Allergy 2011, 1, 5. [Google Scholar] [CrossRef] [PubMed]
- Eutamene, H.; Beaufrand, C.; Harkat, C.; Theodorou, V. The role of mucoprotectants in the management of gastrointestinal disorders. Expert Rev. Gastroenterol. Hepatol. 2018, 12, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Gao, R.; Kong, C.; Huang, L.; Li, H.; Qu, X.; Lan, P.; Wang, J.; Qin, H. Mucosa-associated microbiota signature in colorectal cancer. Eur. J. Clin. Microbiol. Infect. Dis. 2017, 36, 2073–2083. [Google Scholar] [CrossRef] [PubMed]
- Fulbright, L.E.; Ellermann, M.; Arthur, J.C. The microbiome and the hallmarks of cancer. PLoS Pathog 2017, 13, e1006480. [Google Scholar] [CrossRef] [PubMed]
- Gevers, D.; Kugathasan, S.; Denson, L.A.; Vázquez-Baeza, Y.; van Treuren, W; Ren, B.; Schwager, E.; Knights, D.; Song, S.J.; Yassour, M.; et al. The treatment-naive microbiome in new-onset Crohn’s disease. Cell Host Microbe 2014, 15, 382–392. [Google Scholar] [CrossRef] [PubMed]
- Moser, G.; Fournier, C.; Peter, J. Intestinal microbiome-gut-brain axis and irritable bowel syndrome. Wien. Med. Wochenschr. 2017. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Jaber, V.; Lukiw, W.J. Secretory Products of the Human GI Tract Microbiome and Their Potential Impact on Alzheimer’s Disease (AD): Detection of Lipopolysaccharide (LPS) in AD Hippocampus. Front. Cell Infect. Microbiol. 2017, 7, 318. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.; Kang, Y. Gut microbiota and chronic kidney disease: Implications for novel mechanistic insights and therapeutic strategies. Int. Urol. Nephrol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Steelant, B.; Farré, R.; Wawrzyniak, P.; Belmans, J.; Dekimpe, E.; Vanheel, H.; van Gerven, L.; Krohn, I.K.; Bullens, D.M.A.; Ceuppens, J.L.; et al. Impaired barrier function in patients with house dust mite-induced allergic rhinitis is accompanied by decreased occludin and zonula occludens-1 expression. J. Allergy Clin. Immunol. 2016, 137, 1043–1053.e5. [Google Scholar] [CrossRef] [PubMed]
- Steelant, B.; Seys, S.F.; Boeckxstaens, G.; Akdis, C.A.; Ceupeens, J.L.; Hellings, P.W. Restoring airway epitelial barrier dysfunction: A new therapeutic challenge in allergic airway disease. Rhinology 2016, 54, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Allegrini, A.; Pavone, D.; Carluccio, F. A randomized controlled trial comparing a xyloglucan-based nasal spray with saline in adults with symptoms of rhinosinusitis. Curr. Med. Res. Opin. 2017, 19, 1–9. [Google Scholar] [CrossRef] [PubMed]
- De Servi, B.; Ranzini, F.; Piqué, N. Effect of Utipro(®) (containing gelatin-xyloglucan) against Escherichia coli invasion of intestinal epithelial cells: Results of an in vitro study. Future Microbiol. 2016, 11, 651–658. [Google Scholar] [CrossRef] [PubMed]
- Fraile, B.; Alcover, J.; Royuela, M.; Rodríguez, D.; Chaves, C.; Palacios, R.; Piqué, N. Xyloglucan, hibiscus and propolis for the prevention of urinary tract infections: Results of in vitro studies. Future Microbiol. 2017, 12, 721–731. [Google Scholar] [CrossRef] [PubMed]
- Brozek, J.L.; Bousquet, J.; Baena-Cagnani, C.E.; Bonini, S.; Canonica, G.W.; Casale, T.B.; van Wijk, R.G.; Ohta, K.; Zuberbier, T.; Schünemann, H.J. Global Allergy and Asthma European Network; Grading of Recommendations Assessment, Development and Evaluation Working Group. Allergic Rhinitis and its Impact on Asthma (ARIA) guidelines: 2010 revision. J. Allergy Clin. Immunol. 2010, 126, 466–476. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Antimicrobial Resistance: Global Report on Surveillance. 2014. Available online: http://apps.who.int/iris/bitstream/10665/112642/1/9789241564748_eng.pdf (accessed on 30 December 2017).
- World Health Organization. Global Priority List of Antibiotic-Resistant Bacteria to Guide Research, Discovery, and Development of New Antibiotics. 2017. Available online: http://www.who.int/medicines/publications/global-priority-list-antibiotic-resistant-bacteria/en/ (accessed on 30 December 2017).
- Costelloe, C.; Metcalfe, C.; Lovering, A.; Mant, D.; Hay, A.D. Effect of antibiotic prescribing in primary care on antimicrobial resistance in individual patients: Systematic review and meta-analysis. BMJ 2010, 340, c2096. [Google Scholar] [CrossRef] [PubMed]
- Burgalassi, S.; Raimondi, L.; Pirisino, R.; Banchelli, G.; Boldrini, E.; Saettone, M.F. Effect of xyloglucan (tamarind seed polysaccharide) on conjunctival cell adhesion to laminin and on corneal epithelium wound healing. Eur. J. Ophthalmol. 2000, 10, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Gnessi, L.; Bacarea, V.; Marusteri, M.; Piqué, N. Xyloglucan for the treatment of acute diarrhea: Results of a randomized, controlled, open-label, parallel group, multicentre, national clinical trial. BMC Gastroenterol. 2015, 15, 153. [Google Scholar] [CrossRef] [PubMed]
- Plesea Condratovici, C.; Bacarea, V.; Piqué, N. Xyloglucan for the treatment of acute gastroenteritis in children: Results of a randomized controlled, clinical trial. Gastroenterol. Res. Pract. 2016, 2016, 6874207. [Google Scholar] [CrossRef] [PubMed]
- Guglietta, A. Recurrent urinary tract infections in women: Risk factors, etiology, pathogenesis and prophylaxis. Future Microbiol. 2017, 12, 239–246. [Google Scholar] [CrossRef] [PubMed]
- García-Larrosa, A.; Alexe, O. Efficacy and safety of a medical device versus placebo in the early treatment of patients with symptoms of urinary tract infection: A randomized controlled trial. Clin. Microbiol. 2016, 5, 233. [Google Scholar] [CrossRef]
- Salvatorelli, N.; García-Larrosa, A.; Allegrini, A.; Pavone, D. A New Approach to the Treatment of Uncomplicated Cystitis: Results of a Randomized Placebo-Controlled Clinical Trial. Urol. Int. 2016, 97, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Rolando, M.; Valente, C. Establishing the tolerability and performance of tamarind seed polysaccharide (TSP) in treating dry eye syndrome: Results of a clinical study. BMC Ophthalmol. 2007, 7, 5. [Google Scholar] [CrossRef] [PubMed]
- Authimoolam, S.P.; Dziubla, T.D. Biopolymeric mucin and synthetic polymer analogs: Their structure, function and role in biomedical applications. Polymers 2016, 8, 71. [Google Scholar] [CrossRef]
- Rogers, D.F. Airway goblet cells: Responsive and adaptable front-line defenders. Eur. Respir. J. 1994, 7, 1690–1706. [Google Scholar] [CrossRef] [PubMed]
- Takiishi, T.; Fenero, C.I.M.; Câmara, N.O.S. Intestinal barrier and gut microbiota: Shaping our immune responses throughout life. Tissue Barriers 2017, e1373208. [Google Scholar] [CrossRef] [PubMed]
- Yuksel, H.; Turkeli, A. Airway epithelial barrier dysfunction in the pathogenesis and prognosis of respiratory tract diseases in childhood and adulthood. Tissue Barriers 2017, e1367458. [Google Scholar] [CrossRef] [PubMed]
- Hooper, L.V.; Gordon, J.I. Glycans as legislators of host-microbial interactions: Spanning the spectrum from symbiosis to pathogenicity. Glycobiology 2001, 11, 1R–10R. [Google Scholar] [CrossRef] [PubMed]
- Ruch, T.R.; Engel, J.N. Targeting the Mucosal Barrier: How Pathogens Modulate the Cellular Polarity Network. Cold Spring Harb. Perspect. Biol. 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Fries, W.; Belvedere, A.; Vetrano, S. Sealing the broken barrier in IBD: Intestinal permeability, epithelial cells and junctions. Curr. Drug Targets 2013, 14, 1460–1470. [Google Scholar] [CrossRef] [PubMed]
- Grover, M. Role of gut pathogens in development of irritable bowel syndrome. Indian J. Med. Res. 2014, 139, 11–18. [Google Scholar] [PubMed]
- Kojima, T.; Go, M.; Takano, K.; Kurose, M.; Ohkuni, T.; Koizumi, J.; Kamekura, R.; Ogasawara, N.; Masaki, T.; Fuchimoto, J.; et al. Regulation of tight junctions in upper airway epithelium. Biomed. Res. Int 2013, 2013, 947072. [Google Scholar] [CrossRef] [PubMed]
- Soyka, M.B.; Wawrzyniak, P.; Eiwegger, T.; Holzmann, D.; Treis, A.; Wanke, K.; Jeannette, I.K.; Akdis, C.A. Defective epithelial barrier in chronic rhinosinusitis: The regulation of tight junctions by IFN-γ and IL-4. J. Allergy Clin. Immunol. 2012, 130, 1087–1096.e10. [Google Scholar] [CrossRef] [PubMed]
- Fukuoka, A.; Matsushita, K.; Morikawa, T.; Takano, H.; Yoshimoto, T. Diesel exhaust particles exacerbate allergic rhinitis in mice by disrupting the nasal epithelial barrier. Clin. Exp. Allergy 2016, 46, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Pauly, M.; Keegstra, K. Biosynthesis of the Plant Cell Wall Matrix Polysaccharide Xyloglucan. Annu. Rev. Plant. Biol. 2016, 67, 235–259. [Google Scholar] [CrossRef] [PubMed]
- Urakawa, H.; Mimura, M.; Kajiwara, K. Diversity and versatility of plant seed xyloglucan. Trends Glycosci. Glycotechnol. 2002, 14, 355–376. [Google Scholar] [CrossRef][Green Version]
- Mahajan, H.S.; Tyagi, V.K.; Patil, R.R.; Dusunge, S.B. Thiolated xyloglucan: Synthesis, characterization and evaluation as mucoadhesive in situ gelling agent. Carbohydr. Polym. 2013, 91, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, H.S.; Tyagi, V.; Lohiya, G.; Nerkar, P. Thermally reversible xyloglucan gels as vehicles for nasal drug delivery. Drug Deliv. 2012, 19, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, S.; Suisha, F.; Kawasaki, N.; Shirakawa, M.; Yamatoya, K.; Attwood, D. Thermally reversible xyloglucan gels as vehicles for rectal drug delivery. J. Control. Release 1998, 56, 75–83. [Google Scholar] [CrossRef]
- Kulkarni, A.D.; Josh, A.A.; Patil, C.L.; Amale, P.D.; Patel, H.M.; Surana, S.J.; Belgamwar, V.S.; Chaudhari, K.S.; Pardeshi, C.H. Xyloglucan: A functional biomacromolecule for drug delivery applications. Int. J. Biol. Macromol. 2017, 104, 799–812. [Google Scholar] [CrossRef] [PubMed]
- Bashir, A.; Sharma, P.K.; Warsi, M.H. Extraction and characterization of xyloglucan (tamarind seed polysaccharide) as pharmaceutical excipient. WJPPS 2016, 6, 2209–2220. [Google Scholar] [CrossRef]
- McDougall, G.J.; Fry, S.C. Structure-Activity relationships for xyloglucan oligosaccharides with antiauxin activity. Plant. Physiol. 1989, 89, 883–887. [Google Scholar] [CrossRef] [PubMed]
- Alexea, O.; Bacarea, V.; Piqué, N. The combination of oligo- and poly-saccharides and reticulated protein for the control of symptoms of patients with irritable bowel syndrome: Results of a randomized, placebo-controlled, double-blind, parallel group, multicentre clinical trial. United Eur. Gastroenterol. J. 2016, 4, 455–465. [Google Scholar] [CrossRef] [PubMed]
- Allegrini, A.; Costantini, M. Gelatine tannate for the treatment of acute diarrhoea in adults. J. Gastroint. Dig. Syst. 2012, 2, 3. [Google Scholar] [CrossRef]
- Esteban Carretero, J.; Durbán Reguera, F.; López-Argüeta Alvarez, S.; López Montes, J. A comparative analysis of response to vs. ORS + gelatin tannate pediatric patients with acute diarrhea. Rev. Espan. Enferm. Dig. 2009, 101, 41–48. [Google Scholar]
- Manchanda, R.; Arora, S.C.; Manchanda, R. Tamarind seed polysaccharide and its modifications-versatile pharmaceutical excipients-A review. Int. J. PharmTech Res. 2014, 6, 412–420. [Google Scholar]
- Satle, A.; Agrawal, S. Solubility enhancement potential of tamarind seed polysaccharide as a solubilizer. Int. J. Curr. Pharm. Res. 2012, 4, 67–73. [Google Scholar]
- Sahoo, R.; Sahoo, S. Natural polymer of tamarind seed: A prospective carrier for ocular drug delivery. Eur. Sci. J. 2015, 11, 267–275. [Google Scholar]
- Picout, D.R.; Ross-Murphy, S.B.; Errington, N.; Harding, S.E. Pressure cell assisted solubilization of xyloglucans: Tamarind seed polysaccharide and detarium gum. Biomacromolecules 2003, 4, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Kozioł, A.; Cybulska, J.; Pieczywek, P.M.; Zdunek, A. Evaluation of Structure and Assembly of Xyloglucan from Tamarind Seed (Tamarindus indica L.) with Atomic Force Microscopy. Food Biophys. 2015, 10, 396–402. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Pal, K.; Anis, A.; Pramanik, K.; Prabhakar, B. Polymers in mucoadhesive drug-delivery systems: A brief note. Des. Monomer. Polym. 2009, 12, 483–495. [Google Scholar] [CrossRef]
- Kumar, A.; Garg, T.; Sarma, G.S.; Rath, G.; Goyal, A.K. Optimization of combinational intranasal drug delivery system for the management of migraine by using statistical design. Eur. J. Pharm. Sci. 2015, 70, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Madgulkar, A.R.; Bhalekar, M.R.; Asgaonkar, K.D.; Dikpati, A.A. Synthesis and characterization of a novel mucoadhesive derivative of xyloglucan. Carbohydr. Polym. 2016, 135, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, H.S.; Deshmukh, S.R. Development and evaluation of gel-forming ocular films based on xyloglucan. Carbohydr. Polym. 2015, 122, 243–247. [Google Scholar] [CrossRef] [PubMed]
- York, W.S.; van Halbeek, H.; Darvill, A.G.; Albersheim, P. Structural analysis of xyloglucan oligosaccharides by 1H-n.m.r. spectroscopy and fast-atom-bombardment mass spectrometry. Carbohydr. Res. 1990, 200, 9–31. [Google Scholar] [CrossRef]
- Arruda, I.R.S.; Albuquerque, P.B.S.; Santos, G.R.C.; Silva, A.G.; Mourão, P.A.S.; Correia, M.T.S.; Vicente, A.A.; Carneiro-da-Cunha, M.G. Structure and rheological properties of a xyloglucan extracted from Hymenaea courbaril var. courbaril seeds. Int. J. Biol. Macromol. 2015, 73, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Gidley, M.J.; Lillford, P.J.; Rowlands, D.W.; Lang, P.; Dentini, M.; Crescenzi, V.; Edwards, M.; Fanutti, C.; Reid, J.S. Structure and solution properties of tamarind-seed polysaccharide. Carbohydr. Res. 1991, 214, 299–314. [Google Scholar] [CrossRef]
- Mendes, F.R.S.; Bastos, M.S.R.; Mendes, L.G.; Silva, A.R.A.; Sousa, F.D.; Monteiro-Moreira, A.C.O.; Cheng, H.N.; Biswas, A.; Moreira, R.A. Preparation and evaluation of hemicellulose films and their blends. Food Hydrocoll. 2017, 70, 181–190. [Google Scholar] [CrossRef]
- Cozens, D.; Read, R.C. Anti-adhesion methods as novel therapeutics for bacterial infections. Expert Rev. Anti-Infect. Ther. 2012, 10, 1457–1468. [Google Scholar] [CrossRef] [PubMed]
- Sattin, S.; Bernardi, A. Glycoconjugates and Glycomimetics as Microbial Anti-Adhesives. Trends Biotechnol. 2016, 34, 483–495. [Google Scholar] [CrossRef] [PubMed]
- Rendueles, O.; Kaplan, J.B.; Ghigo, J.M. Antibiofilm polysaccharides. Environ. Microbiol. 2013, 15, 334–346. [Google Scholar] [CrossRef] [PubMed]
- Blumberg, J.B.; Basu, A.; Krueger, C.G.; Lila, M.A.; Neto, C.C.; Novotny, J.A.; Reed, J.D.; Rodriguez-Mateos, A.; Toner, C.D. Impact of Cranberries on Gut Microbiota and Cardiometabolic Health: Proceedings of the Cranberry Health Research Conference 2015. Adv. Nutr. 2016, 7, 759S–770S. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Kim, C.Y.; Kaur, A.; Lamothe, L.; Shaikh, M.; Keshavarzianc, A.; Hamaker, B.R. Dietary fibre-based SCFA mixtures promote both protection and repair of intestinal epithelial barrier function in a Caco-2 cell model. Food Funct. 2017, 8, 1166–1173. [Google Scholar] [CrossRef] [PubMed]
- Ojeda, P.; Piqué, N.; Alonso, A.; Delgado, J.; Feo, F.; Igea, J.M.; Navarro, A.; Olaguibel, J.M.; Subiza, J.; Nieto, C.; et al. A topical microemulsion for the prevention of allergic rhinitis symptoms: Results of a randomized, controlled, double-blind, parallel group, multicentre, multinational clinical trial (Nares study). Allergy Asthma Clin. Immunol. 2013, 9, 32. [Google Scholar] [CrossRef] [PubMed]
- Katouli, M. Population structure of gut Escherichia coli and its role in development of extra-intestinal infections. Iran. J. Microbiol. 2010, 2, 59–72. [Google Scholar] [PubMed]
- Nie, W.; Deters, A.M. Tamarind Seed Xyloglucans Promote Proliferation and Migration of Human Skin Cells through Internalization via Stimulation of Proproliferative Signal Transduction Pathways. Dermatol. Res. Pract. 2013, 2013, 359756. [Google Scholar] [CrossRef] [PubMed]
- Bueno, L.; Theodorou, V.; Sekkal, S. Xyloglucan: A new agent to protect the intestinal mucosa and to prevent bacterially mediated alteration of tight junction permeability. In Proceedings of the 22nd United European Gastroenterology (UEG) week, Vienna, Austria, 18–22 October 2014. Abstract P1675. [Google Scholar]
- Burgalassi, S.; Panichi, L.; Chetoni, P.; Saettone, M.F.; Boldrini, E. Development of a simple dry eye model in the albino rabbit and evaluation of some tear substitutes. Ophthalmic Res. 1999, 31, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Olier, M.; Sekkal, S.; Harkat, C.; Eutamene, H.; Theodorou, V. Evaluation of reticulated gelatin-hibiscus propolis against intestinal comensal species commonly associated with urinary tract infections. Future Microbiol. 2017, 12, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Vicario, M.; González-Castro, A.M.; Martínez, C.; Lobo, B.; Pigrau, M.; Guilarte, M.; de Torres, I.; Mosquera, J.L.; Fortea, M.; Sevillano-Aguilera, C.; et al. Increased humoral immunity in the jejunum of diarrhoea-predominant irritable bowel syndrome associated with clinical manifestations. Gut 2015, 64, 1379–1388. [Google Scholar] [CrossRef] [PubMed]
- Vandenplas, Y.; Bacarea, A.; Marusteri, M.; Bacarea, V.; Constantin, M.; Manaloche, M. Efficacy and safety of APT198K for the treatment of infantile colic: A pilot. J. Comp. Eff. Res. 2017, 6, 137–144. [Google Scholar] [CrossRef]
- Kezic, S.; Novak, N.; Jakasa, I.; Jungersted, J.M.; Simon, M.; Brandner, J.M.; Middelkamp-Hub, M.A.; Weidinger, S. Skin barrier in atopic dermatitis. Front Biosci. (Landmark Ed) 2014, 19, 542–556. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piqué, N.; Gómez-Guillén, M.D.C.; Montero, M.P. Xyloglucan, a Plant Polymer with Barrier Protective Properties over the Mucous Membranes: An Overview. Int. J. Mol. Sci. 2018, 19, 673. https://doi.org/10.3390/ijms19030673
Piqué N, Gómez-Guillén MDC, Montero MP. Xyloglucan, a Plant Polymer with Barrier Protective Properties over the Mucous Membranes: An Overview. International Journal of Molecular Sciences. 2018; 19(3):673. https://doi.org/10.3390/ijms19030673
Chicago/Turabian StylePiqué, Núria, María Del Carmen Gómez-Guillén, and María Pilar Montero. 2018. "Xyloglucan, a Plant Polymer with Barrier Protective Properties over the Mucous Membranes: An Overview" International Journal of Molecular Sciences 19, no. 3: 673. https://doi.org/10.3390/ijms19030673
APA StylePiqué, N., Gómez-Guillén, M. D. C., & Montero, M. P. (2018). Xyloglucan, a Plant Polymer with Barrier Protective Properties over the Mucous Membranes: An Overview. International Journal of Molecular Sciences, 19(3), 673. https://doi.org/10.3390/ijms19030673




