A Biochemical Approach to Detect Oxidative Stress in Infertile Women Undergoing Assisted Reproductive Technology Procedures
Abstract
:1. Introduction
2. Results
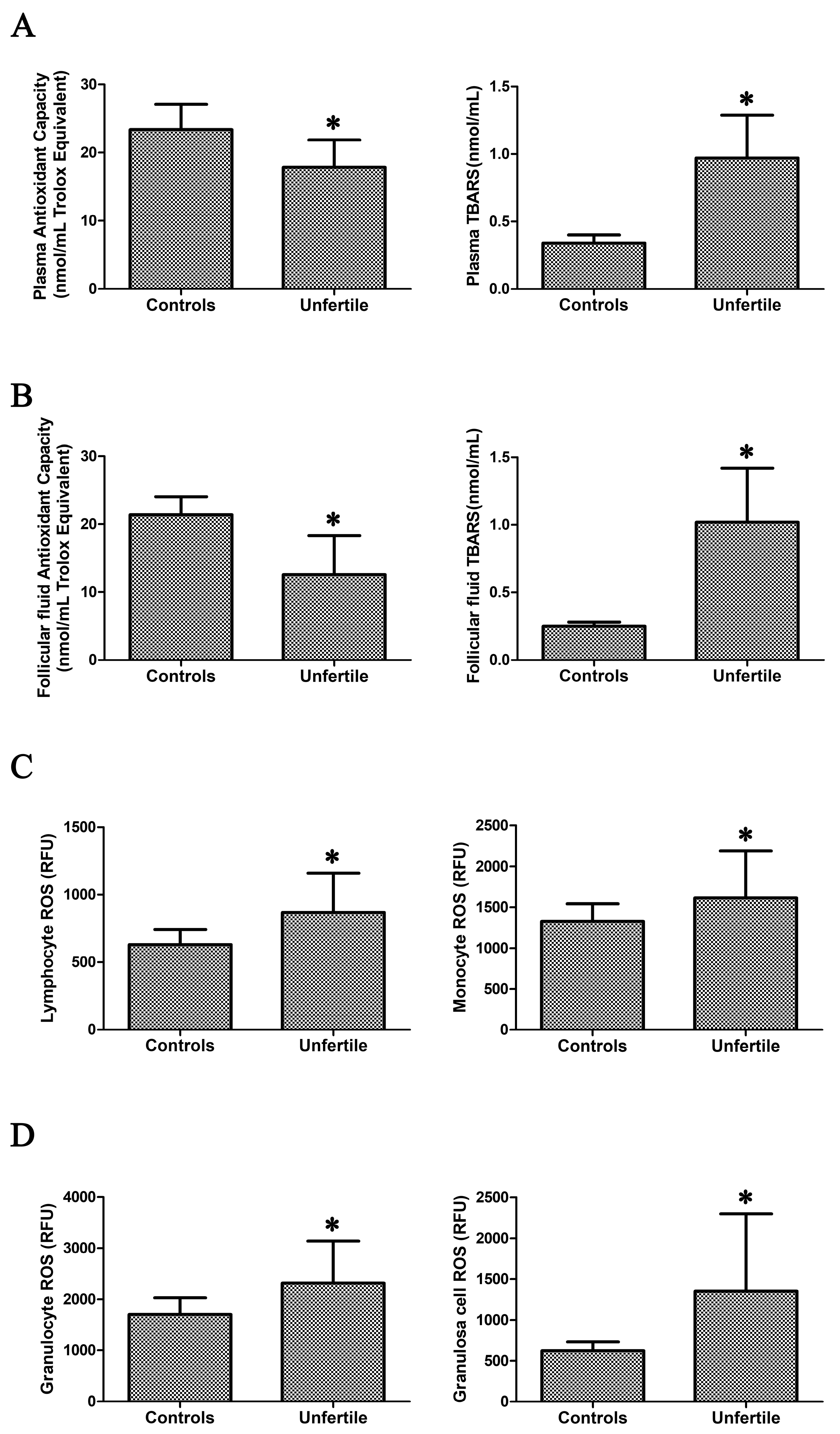
2.1. Assessment of Oxidative Stress in Blood Samples and in Granulosa Cells
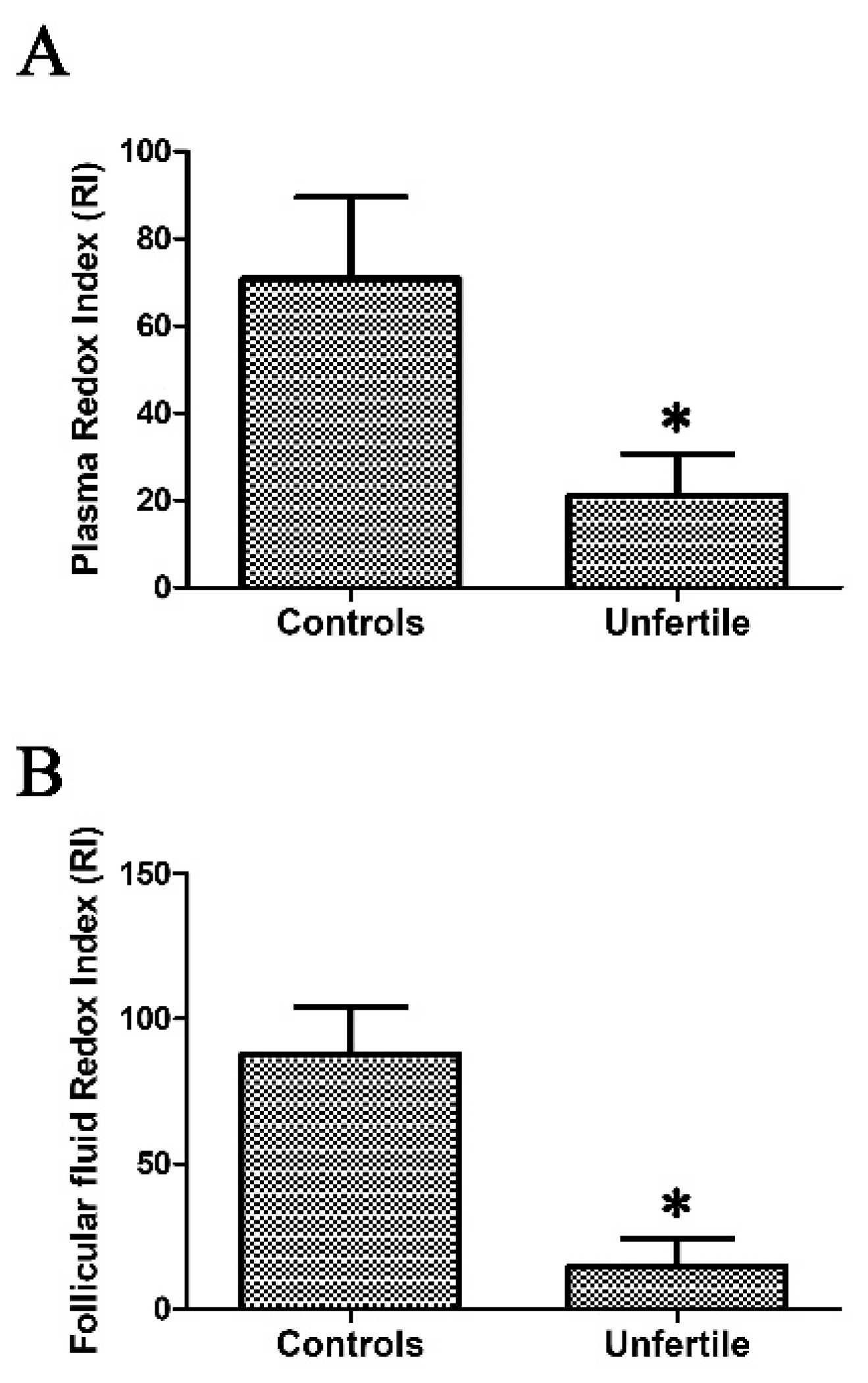
2.2. Redox Index Calculated as ORAC/TBARS Ratio as a Mirror of Oxidative Stress
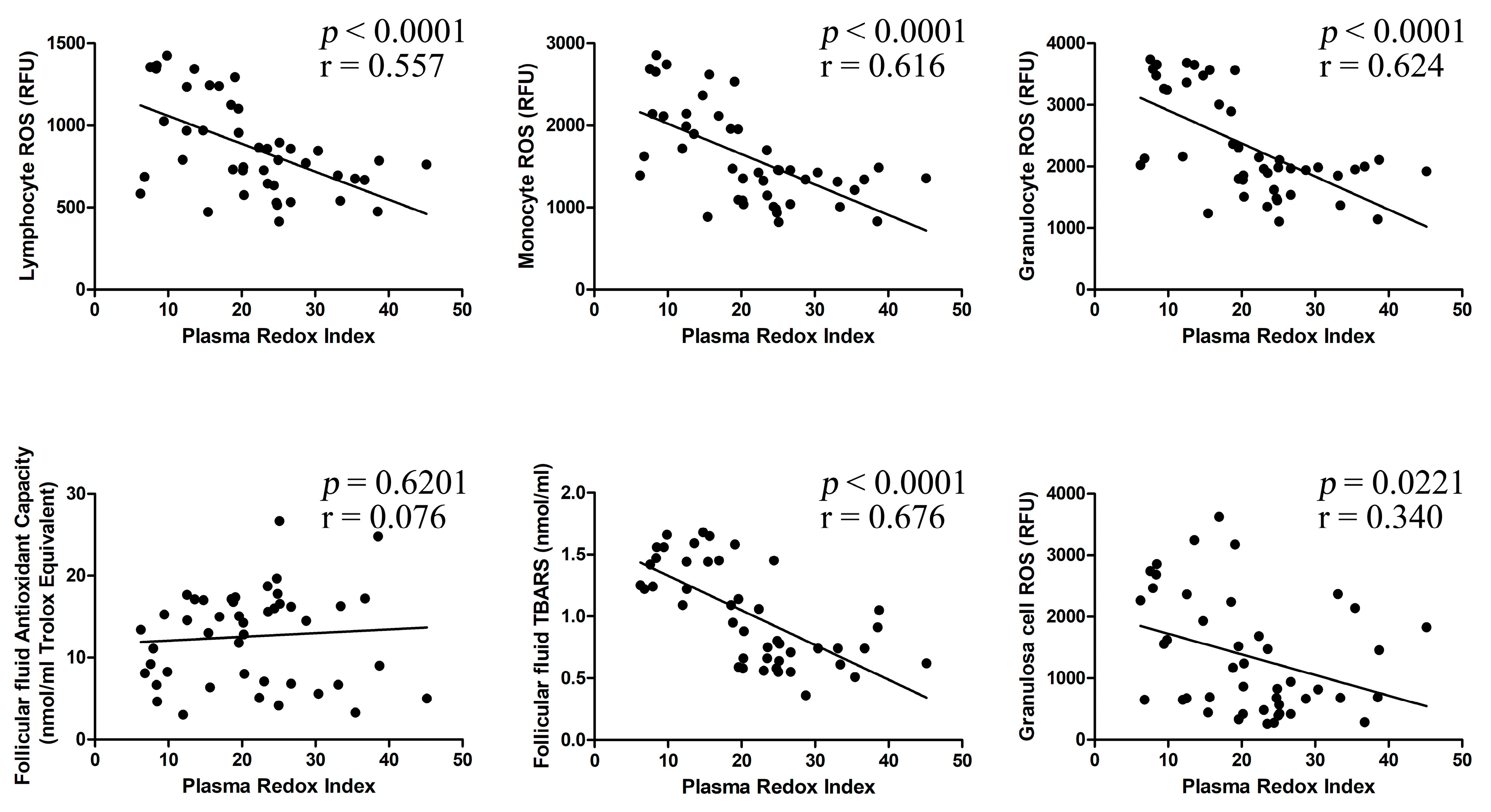
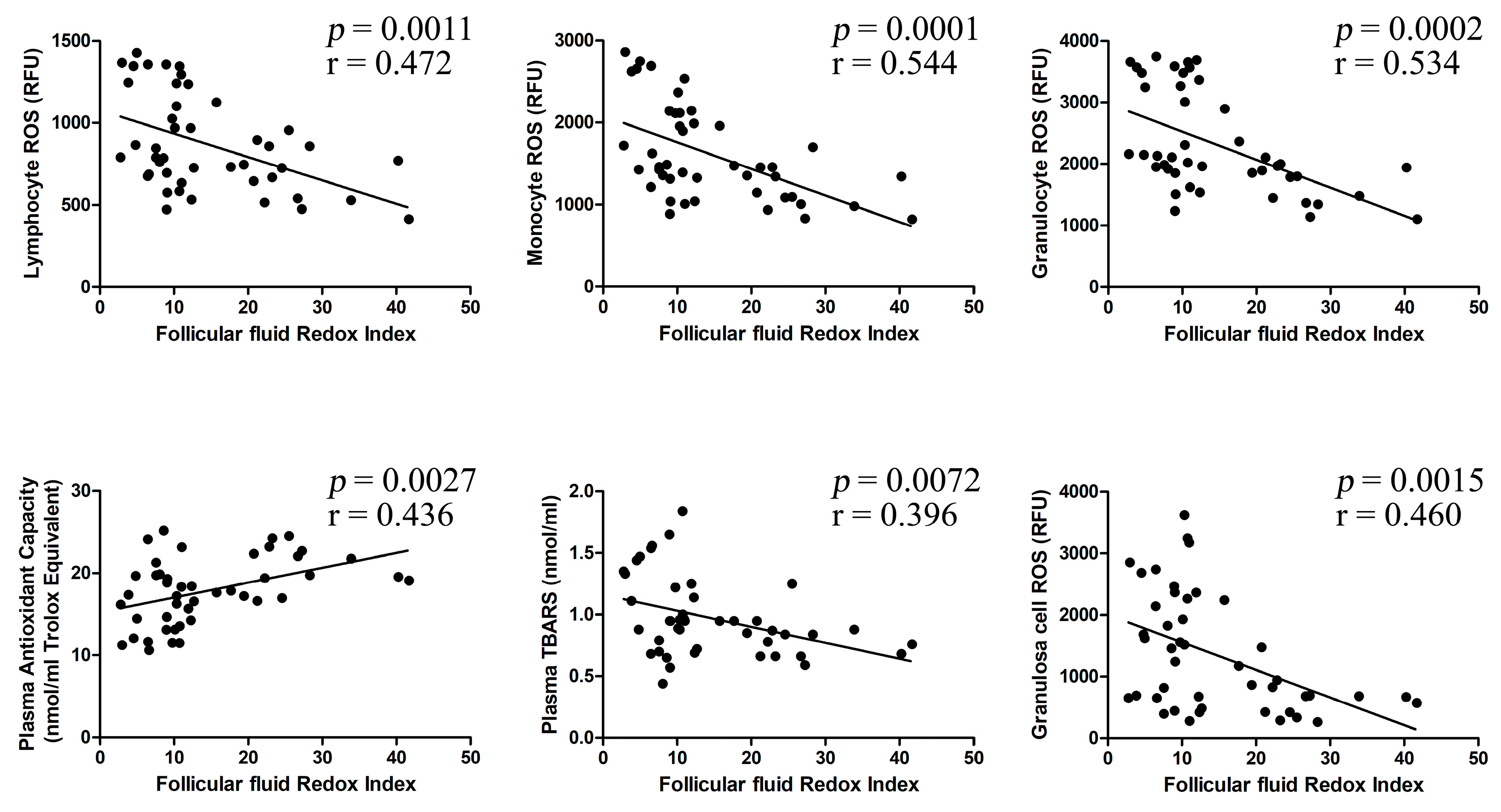
2.3. Correlation between Investigated Parameters
2.4. Systemic Oxidative Stress Parameters are Related to Assisted Reproductive Technologies (ART) Outcome
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. FF Collection and Processing
4.3. Total Antioxidant Capacity (TAC) Assay
4.4. TBARS (Thiobarbituric Acid Reactive Substances) Estimation
4.5. ROS Assessment by Flow Cytometry Analysis
4.6. ORAC/TBARS Ratio as an Index of Oxidative Stress
4.7. Measurements of Outcomes: Metaphase II
4.8. Measurements of Outcomes: Fertilization Rate (FR)
4.9. Measurements of Outcomes: Ongoing Pregnancies (at 20 Weeks of Gestation)
4.10. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schmidt, H.H.; Stocker, R.; Vollbracht, C.; Paulsen, G.; Riley, D.; Daiber, A.; Cuadrado, A. Antioxidants in Translational Medicine. Antioxid. Redox Signal. 2015, 23, 1130–1143. [Google Scholar] [CrossRef] [PubMed]
- Becatti, M.; Fiorillo, C.; Barygina, V.; Cecchi, C.; Lotti, T.; Prignano, F.; Silvestro, A.; Nassi, P.; Taddei, N. SIRT1 regulates MAPK pathways in vitiligo skin: Insight into the molecular pathways of cell survival. J. Cell Mol. Med. 2014, 18, 514–529. [Google Scholar] [CrossRef] [PubMed]
- Marrocco, I.; Altieri, F.; Peluso, I. Measurement and clinical significance of biomarkers of oxidative stress in humans. Oxid. Med. Cell. Longev. 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Devine, P.J.; Perreault, S.D.; Luderer, U. Roles of reactive oxygen species and antioxidants in ovarian toxicity. Biol. Reprod. 2012, 86. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Aponte-Mellado, A.; Premkumar, B.J.; Shaman, A.; Gupta, S. The effects of oxidative stress on female reproduction: A review. Reprod. Biol. Endocrinol. 2012, 10. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Gupta, S.; Sharma, R. Oxidative stress and its implications in female infertility—A clinician’s perspective. Reprod. Biomed. Online 2005, 11, 641–650. [Google Scholar] [CrossRef]
- Oyawoye, O.; Abdel-Gadir, A.; Garner, A.; Constantinovici, N.; Perrett, C.; Hardiman, P. Antioxidants and reactive oxygen species in follicular fluid of women undergoing IVF: relationship to outcome. Hum. Reprod. 2003, 18, 2270–2274. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Gupta, S.; Sharma, R.K. Role of oxidative stress in female reproduction. Reprod. Biol. Endocrinol. 2005, 3. [Google Scholar] [CrossRef] [PubMed]
- Dumesic, D.A.; Meldrum, D.R.; Katz-Jaffe, M.G.; Krisher, R.L.; Schoolcraft, W.B. Oocyte environment: Follicular fluid and cumulus cells are critical for oocyte health. Fertil. Steril. 2015, 103, 303–316. [Google Scholar] [CrossRef] [PubMed]
- Du Plessis, S.; Makker, K.; Desai, N. Impact of oxidative stress on IVF. Exp. Rev. Obstet. Gynecol. 2008, 3, 539–554. [Google Scholar] [CrossRef]
- Jozwik, M.; Wolczynski, S.; Jozwik, M.; Szamatowicz, M. Oxidative stress markers in preovulatory follicular fluid in humans. Mol. Hum. Reprod. 1999, 5, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Attaran, M.; Pasqualotto, E.; Falcone, T.; Goldberg, J.M.; Miller, K.F.; Agarwal, A.; Sharma, R.K. The effect of follicular fluid reactive oxygen species on the outcome of in vitro fertilization. Int. J. Fertil. Womens Med. 2000, 45, 314–320. [Google Scholar] [PubMed]
- Pasqualotto, E.B.; Agarwal, A.; Sharma, R.K.; Izzo, V.M.; Pinotti, J.A.; Joshi, N.J.; Rose, B.I. Effect of oxidative stress in follicular fluid on the outcome of assisted reproductive procedures. Fertil. Steril. 2004, 81, 973–976. [Google Scholar] [CrossRef] [PubMed]
- Attaran, M.; Frasor, J.; Mascha, E.; Radwanska, E.; Rawlins, R.G. The relationship of human granulosa-lutein cell proliferative index to follicular diameter and serum estradiol. Obstet. Gynecol. 1998, 91, 449–453. [Google Scholar] [CrossRef]
- Das, S.; Chattopadhyay, R.; Ghosh, S.; Ghosh, S.; Goswami, S.K.; Chakravarty, B.N.; Chaudhury, K. Reactive oxygen species level in follicular fluid—Embryo quality marker in IVF? Hum. Reprod. 2006, 21, 2403–2407. [Google Scholar] [CrossRef] [PubMed]
- Elizur, S.E.; Lebovitz, O.; Orvieto, R.; Dor, J.; Zan-Bar, T. Reactive oxygen species in follicular fluid may serve as biochemical markers to determine ovarian aging and follicular metabolic age. Gynecol. Endocrinol. 2014, 30, 705–707. [Google Scholar] [CrossRef] [PubMed]
- Shkolnik, K.; Tadmor, A.; Ben-Dor, S.; Nevo, N.; Galiani, D.; Dekel, N. Reactive oxygen species are indispensable in ovulation. Proc. Natl. Acad. Sci. USA 2011, 108, 1462–1467. [Google Scholar] [CrossRef] [PubMed]
- Siristatidis, C.; Askoxylaki, M.; Varounis, C.; Kassanos, D.; Chrelias, C. E-selectin, resistin and reactive oxygen species levels in GnRH -agonist and -antagonist protocols in IVF/ICSI: A prospective cohort study. J. Assist. Reprod. Genet. 2015, 32, 959–967. [Google Scholar] [CrossRef] [PubMed]
- Appasamy, M.; Jauniaux, E.; Serhal, P.; Al-Qahtani, A.; Groome, N.P.; Muttukrishna, S. Evaluation of the relationship between follicular fluid oxidative stress, ovarian hormones, and response to gonadotropin stimulation. Fertil. Steril. 2008, 89, 912–921. [Google Scholar] [CrossRef] [PubMed]
- Al-Fadhli, R.; Sylvestre, C.; Buckett, W.; Tan, S.L.; Tulandi, T. A randomized trial of superovulation with two different doses of letrozole. Fertil. Steril. 2006, 85, 161–164. [Google Scholar] [CrossRef] [PubMed]
- Jana, S.K.; K, N.B.; Chattopadhyay, R.; Chakravarty, B.; Chaudhury, K. Upper control limit of reactive oxygen species in follicular fluid beyond which viable embryo formation is not favorable. Reprod. Toxicol. 2010, 29, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Wiener-Megnazi, Z.; Vardi, L.; Lissak, A.; Shnizer, S.; Reznick, A.Z.; Ishai, D.; Lahav-Baratz, S.; Shiloh, H.; Koifman, M.; Dirnfeld, M. Oxidative stress indices in follicular fluid as measured by the thermochemiluminescence assay correlate with outcome parameters in in vitro fertilization. Fertil. Steril. 2004, 82, 1171–1176. [Google Scholar] [CrossRef] [PubMed]
- Seino, T.; Saito, H.; Kaneko, T.; Takahashi, T.; Kawachiya, S.; Kurachi, H. Eight-hydroxy-2′-deoxyguanosine in granulosa cells is correlated with the quality of oocytes and embryos in an in vitro fertilization-embryo transfer program. Fertil. Steril. 2002, 77, 1184–1190. [Google Scholar] [CrossRef]
- Tamura, H.; Takasaki, A.; Miwa, I.; Taniguchi, K.; Maekawa, R.; Asada, H.; Taketani, T.; Matsuoka, A.; Yamagata, Y.; Shimamura, K.; et al. Oxidative stress impairs oocyte quality and melatonin protects oocytes from free radical damage and improves fertilization rate. J. Pineal. Res. 2008, 44, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Chattopadhyay, R.; Chakravarty, B.; Chaudhury, K. Markers of oxidative stress in follicular fluid of women with endometriosis and tubal infertility undergoing IVF. Reprod. Toxicol. 2013, 42, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Da Broi, M.G.; De Albuquerque, F.O.; De Andrade, A.Z.; Cardoso, R.L.; Jordão Junior, A.A.; Navarro, P.A. Increased concentration of 8-hydroxy-2′-deoxyguanosine in follicular fluid of infertile women with endometriosis. Cell Tissue Res. 2016, 366, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Adeldust, H.; Zeinoaldini, S.; Kohram, H.; Amiri Roudbar, M.; Daliri Joupari, M. In vitro maturation of ovine oocyte in a modified granulosa cells co-culture system and α-tocopherol supplementation: Effects on nuclear maturation and cleavage. J. Anim. Sci. Technol. 2015, 57, 27. [Google Scholar] [CrossRef] [PubMed]
- Siristatidis, C.; Vogiatzi, P.; Varounis, C.; Askoxylaki, M.; Chrelias, C.; Papantoniou, N. The effect of reactive oxygen species on embryo quality in IVF. In Vivo 2016, 30, 149–153. [Google Scholar] [PubMed]
- Fujimoto, V.Y.; Bloom, M.S.; Huddleston, H.G.; Shelley, W.B.; Ocque, A.J.; Browne, R.W. Correlations of follicular fluid oxidative stress biomarkers and enzyme activities with embryo morphology parameters during in vitro fertilization. Fertil. Steril. 2011, 96, 1357–1361. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, A.; Shrivastav, T.G.; Chaube, S.K. An increase of granulosa cell apoptosis mediates aqueous neem (Azadirachta indica) leaf extract-induced oocyte apoptosis in rat. Int. J. Appl. Basic Med. Res. 2013, 3, 27–36. [Google Scholar] [PubMed]
- Yang, H.; Xie, Y.; Yang, D.; Ren, D. Oxidative stress-induced apoptosis in granulosa cells involves JNK, p53 and Puma. Oncotarget 2017, 8, 25310–25322. [Google Scholar] [CrossRef] [PubMed]
- Saeed-Zidane, M.; Linden, L.; Salilew-Wondim, D.; Held, E.; Neuhoff, C.; Tholen, E.; Hoelker, M.; Schellander, K.; Tesfaye, D. Cellular and exosome mediated molecular defense mechanism in bovine granulosa cells exposed to oxidative stress. PLoS ONE 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Cortes-Wanstreet, M.M.; Giedzinski, E.; Limoli, C.L.; Luderer, U. Overexpression of glutamate-cysteine ligase protects human COV434 granulosa tumour cells against oxidative and gamma-radiation-induced cell death. Mutagenesis 2009, 24, 211–224. [Google Scholar] [CrossRef] [PubMed]
- Szczepańska, M.; Koźlik, J.; Skrzypczak, J.; Mikołajczyk, M. Oxidative stress may be a piece in the endometriosis puzzle. Fertil. Steril. 2003, 79, 1288–1293. [Google Scholar] [CrossRef]
- Askoxylaki, M.; Siristatidis, C.; Chrelias, C.; Vogiatzi, P.; Creatsa, M.; Salamalekis, G.; Vrantza, T.; Vrachnis, N.; Kassanos, D. Reactive oxygen species in the follicular fluid of subfertile women undergoing in vitro fertilization: A short narrative review. J. Endocrinol. Investig. 2013, 36, 1117–1120. [Google Scholar] [PubMed]
- Borowiecka, M.; Wojsiat, J.; Polac, I.; Radwan, M.; Radwan, P.; Zbikowska, H.M. Oxidative stress markers in follicular fluid of women undergoing in vitro fertilization and embryo transfer. Syst. Biol. Reprod. Med. 2012, 58, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Velthut, A.; Zilmer, M.; Zilmer, K.; Kaart, T.; Karro, H.; Salumets, A. Elevated blood plasma antioxidant status is favourable for achieving IVF/ICSI pregnancy. Reprod. Biomed. Online 2013, 26, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Luderer, U. Ovarian toxicity from reactive oxygen species. Vitam. Horm. 2014, 94, 99–127. [Google Scholar] [PubMed]
- Prasad, S.; Tiwari, M.; Pandey, A.N.; Shrivastav, T.G.; Chaube, S.K. Impact of stress on oocyte quality and reproductive outcome. J. Biomed. Sci. 2016, 23. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, A.; Roscino, M.T.; Binetti, F.; Sciorsci, R.L. Roles of reactive oxygen species in female reproduction. Reprod. Domest. Anim. 2012, 47, 344–352. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Igarashi, H.; Amita, M.; Hara, S.; Matsuo, K.; Kurachi, H. Molecular mechanism of poor embryo development in postovulatory aged oocytes: Mini review. J. Obstet. Gynaecol. Res. 2013, 39, 1431–1439. [Google Scholar] [CrossRef] [PubMed]
- Berker, B.; Kaya, C.; Aytac, R.; Satiroglu, H. Homocysteine concentrations in follicular fluid are associated with poor oocyte and embryo qualities in polycystic ovary syndrome patients undergoing assisted reproduction. Hum. Reprod. 2009, 24, 2293–2302. [Google Scholar] [CrossRef] [PubMed]
- Barygina, V.V.; Becatti, M.; Soldi, G.; Prignano, F.; Lotti, T.; Nassi, P.; Wright, D.; Taddei, N.; Fiorillo, C. Altered redox status in the blood of psoriatic patients: Involvement of NADPH oxidase and role of anti-TNF-α therapy. Redox Rep. 2013, 18, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Becatti, M.; Emmi, G.; Silvestri, E.; Bruschi, G.; Ciucciarelli, L.; Squatrito, D.; Vaglio, A.; Taddei, N.; Abbate, R.; Emmi, L.; et al. Neutrophil activation promotes fibrinogen oxidation and thrombus formation in behçet disease. Circulation 2016, 133, 302–311. [Google Scholar] [PubMed]
- Sofi, F.; Whittaker, A.; Cesari, F.; Gori, A.M.; Fiorillo, C.; Becatti, M.; Marotti, I.; Dinelli, G.; Casini, A.; Abbate, R.; et al. Characterization of Khorasan wheat (Kamut) and impact of a replacement diet on cardiovascular risk factors: Cross-over dietary intervention study. Eur. J. Clin. Nutr. 2013, 67, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Barygina, V.; Becatti, M.; Lotti, T.; Moretti, S.; Taddei, N.; Fiorillo, C. Treatment with low-dose cytokines reduces oxidative-mediated injury in perilesional keratinocytes from vitiligo skin. J. Dermatol. Sci. 2015, 79, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Prieto, L.; Quesada, J.F.; Cambero, O.; Pacheco, A.; Pellicer, A.; Codoceo, R.; Garcia-Velasco, J.A. Analysis of follicular fluid and serum markers of oxidative stress in women with infertility related to endometriosis. Fertil. Steril. 2012, 98, 126–130. [Google Scholar] [CrossRef] [PubMed]
- Freitas, C.; Neto, A.C.; Matos, L.; Silva, E.; Ribeiro, Â.; Silva-Carvalho, J.L.; Almeida, H. Follicular fluid redox involvement for ovarian follicle growth. J. Ovarian Res. 2017, 12, 44. [Google Scholar] [CrossRef] [PubMed]
- Becatti, M.; Mannucci, A.; Barygina, V.; Mascherini, G.; Emmi, G.; Silvestri, E.; Wright, D.; Taddei, N.; Galanti, G.; Fiorillo, C. Redox status alterations during the competitive season in élite soccer players: Focus on peripheral leukocyte-derived ROS. Intern Emerg. Med. 2017, 12, 777–788. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; He, G.; Chen, M.; Zuo, T.; Xu, W.; Liu, X. The role of antioxidant enzymes in the ovaries. Oxid. Med. Cell Longev. 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Eruslanov, E.; Kusmartsev, S. Identification of ROS using oxidized DCFDA and flow-cytometry. Methods Mol. Biol. 2010, 594, 57–72. [Google Scholar] [PubMed]
- Whittaker, A.; Sofi, F.; Luisi, M.L.; Rafanelli, E.; Fiorillo, C.; Becatti, M.; Abbate, R.; Casini, A.; Gensini, G.F.; Benedettelli, S. An organic khorasan wheat-based replacement diet improves risk profile of patients with acute coronary syndrome: A randomized crossover trial. Nutrients 2015, 7, 3401–3415. [Google Scholar] [CrossRef] [PubMed]
| Redox Parameters | Plasma ORAC | Plasma TBARS | Lymphocyte ROS | Monocyte ROS | Granulocyte ROS | FF ORAC | FF TBARS | Granulosa Cell ROS |
|---|---|---|---|---|---|---|---|---|
| Plasma Redox Index (RI) | r = 0.838 | r = −0.905 | r = −0.640 | r = −0.669 | r = −0.724 | r = 0.083 | r = −0.748 | r = −0.400 |
| p < 0.001 | p < 0.001 | p < 0.001 | p < 0.001 | p < 0.001 | p = 0.639 | p < 0.001 | p = 0.019 | |
| FF Redox Index (RI) | r = 0.474 | r = −0.506 | r = −0.431 | r = −0.520 | r = −0.489 | r = 0.761 | r = −0.520 | r = −0.528 |
| p = 0.005 | p = 0.002 | p = 0.011 | p = 0.002 | p = 0.003 | p < 0.001 | p = 0.002 | p = 0.001 | |
| Lymphocyte ROS | r = −0.618 | r = 0.581 | - | r = 0.925 | r = 0.914 | r = −0.150 | r = 0.664 | r = 0.645 |
| p < 0.001 | p < 0.001 | - | p < 0.001 | p < 0.001 | p = 0.397 | p < 0.001 | p < 0.001 | |
| Monocyte ROS | r = −0.695 | r = 0.621 | r = 0.925 | - | r = 0.937 | r = −0.211 | r = 0.761 | r = 0.619 |
| p < 0.001 | p < 0.001 | p < 0.001 | - | p < 0.001 | p = 0.231 | p < 0.001 | p < 0.001 | |
| Granulocyte ROS | r = −0.749 | r = 0.645 | r = 0.914 | r = 0.937 | - | r = −0.134 | r = 0.759 | r = 0.680 |
| p < 0.001 | p < 0.001 | p < 0.001 | p < 0.001 | - | p = 0.451 | p < 0.001 | p < 0.001 | |
| FF ORAC | r = 0.124 | r = −0.238 | r = −0.150 | r = −0.211 | r = −0.134 | - | r = 0.027 | r = −0.159 |
| p = 0.485 | p = 0.175 | p = 0.397 | p = 0.231 | p = 0.451 | - | p = 0.880 | p = 0.368 | |
| FF TBARS | r = −0.728 | r = 0.682 | r = 0.664 | r = 0.761 | r = 0.759 | r = 0.027 | - | r = 0.557 |
| p < 0.001 | p < 0.001 | p < 0.001 | p < 0.001 | p < 0.001 | p = 0.880 | - | p = 0.001 | |
| Granulosa cell ROS | r = −0.529 | r = 0.394 | r = 0.645 | r = 0.619 | r = 0.680 | r = −0.159 | r = 0.557 | - |
| p = 0.001 | p = 0.021 | p < 0.001 | p < 0.001 | p < 0.001 | p = 0.368 | p = 0.001 | - |
| Redox Parameters | Metaphase II (%) (n = 45) | Fertilization Rate (%) (n = 45) | Ongoing Pregnancies (n = 12) |
|---|---|---|---|
| Plasma TBARS | r = −0.540; p = 0.001 | r = −0.685; p < 0.001 | r = −0.471; p = 0.005 |
| Plasma ORAC | r = 0.524; p = 0.001 | r = 0.469; p = 0.005 | r = 0.504; p = 0.002 |
| Plasma Redox Index (RI) | r = 0.463; p = 0.006 | r = 0.520; p = 0.002 | r = 0.549; p = 0.001 |
| Lymphocyte ROS | r = −0.542; p = 0.001 | r = −0.312; p = 0.072 | r = −0.384; p = 0.025 |
| Monocyte ROS | r = −0.494; p = 0.003 | r = −0.367; p = 0.033 | r = −0.433; p = 0.010 |
| Granulocyte ROS | r = −0.572; p < 0.001 | r = −0.333; p = 0.054 | r = −0.465; p = 0.006 |
| FF TBARS | r = −0.451; p = 0.007 | r = −0.476; p = 0.004 | r = −0.351; p = 0.042 |
| FF ORAC | r = −0.212; p = 0.229 | r = 0.316; p = 0.069 | r = 0.404; p = 0.018 |
| FF Redox Index (RI) | r = 0.459; p = 0.006 | r = 0.413; p = 0.015 | r = 0.518; p = 0.002 |
| Granulosa cell ROS | r = −0.654; p < 0.001 | r = −0.268; p = 0.126 | r = −0.357; p = 0.038 |
| Metaphase II (%) | - | r = 0.378; p = 0.027 | r = 0.452; p = 0.007 |
| Fertilization Rate (%) | r = 0.378; p = 0.027 | - | r = 0.368; p = 0.032 |
| Ongoing pregnancies | r = 0.452; p = 0.007 | r = 0.368; p = 0.032 | - |
| Parameters | PATIENTS (n = 45) | CONTROLS (n = 45) | |
|---|---|---|---|
| Mean ± SD | Mean ± SD | p-value | |
| Age (years) | 35.0 ± 3.3 | 34.5 ± 4.5 | not significant |
| BMI (Kg/m2) | 22.8 ± 4.1 | 21.0 ± 1.5 | 0.0069 |
| Duration of infertility (years) | 3.2 ± 2.0 | 1.8 ± 0.8 | <0.0001 |
| FSH (mIU/mL) | 8.3 ± 3.8 | 6.0 ± 2.2 | 0.0007 |
| LH (mIU/mL) | 5.9 ± 3.1 | 4.4 ± 2.1 | 0.0086 |
| Estradiol (pg/mL) | 57.1 ± 24.8 | 48.5 ± 3.1 | 0.0233 |
| AMH (ng/mL) | 2.4 ± 2.0 | 4.2 ± 3.6 | 0.0043 |
| Smoking habits (%) | 26.7 | 28.0 | not significant |
| GnRH Antagonist (%) | 100 | 100 | not significant |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Becatti, M.; Fucci, R.; Mannucci, A.; Barygina, V.; Mugnaini, M.; Criscuoli, L.; Giachini, C.; Bertocci, F.; Picone, R.; Emmi, G.; et al. A Biochemical Approach to Detect Oxidative Stress in Infertile Women Undergoing Assisted Reproductive Technology Procedures. Int. J. Mol. Sci. 2018, 19, 592. https://doi.org/10.3390/ijms19020592
Becatti M, Fucci R, Mannucci A, Barygina V, Mugnaini M, Criscuoli L, Giachini C, Bertocci F, Picone R, Emmi G, et al. A Biochemical Approach to Detect Oxidative Stress in Infertile Women Undergoing Assisted Reproductive Technology Procedures. International Journal of Molecular Sciences. 2018; 19(2):592. https://doi.org/10.3390/ijms19020592
Chicago/Turabian StyleBecatti, Matteo, Rossella Fucci, Amanda Mannucci, Victoria Barygina, Marco Mugnaini, Luciana Criscuoli, Claudia Giachini, Francesco Bertocci, Rita Picone, Giacomo Emmi, and et al. 2018. "A Biochemical Approach to Detect Oxidative Stress in Infertile Women Undergoing Assisted Reproductive Technology Procedures" International Journal of Molecular Sciences 19, no. 2: 592. https://doi.org/10.3390/ijms19020592
APA StyleBecatti, M., Fucci, R., Mannucci, A., Barygina, V., Mugnaini, M., Criscuoli, L., Giachini, C., Bertocci, F., Picone, R., Emmi, G., Evangelisti, P., Rizzello, F., Cozzi, C., Taddei, N., Fiorillo, C., & Coccia, M. E. (2018). A Biochemical Approach to Detect Oxidative Stress in Infertile Women Undergoing Assisted Reproductive Technology Procedures. International Journal of Molecular Sciences, 19(2), 592. https://doi.org/10.3390/ijms19020592








