Anti-Inflammatory and Gastroprotective Roles of Rabdosia inflexa through Downregulation of Pro-Inflammatory Cytokines and MAPK/NF-κB Signaling Pathways
Abstract
:1. Introduction
2. Results
2.1. Analysis of Total Phenolic and Flavonoid Contents of Rabdosia inflexa (RI)
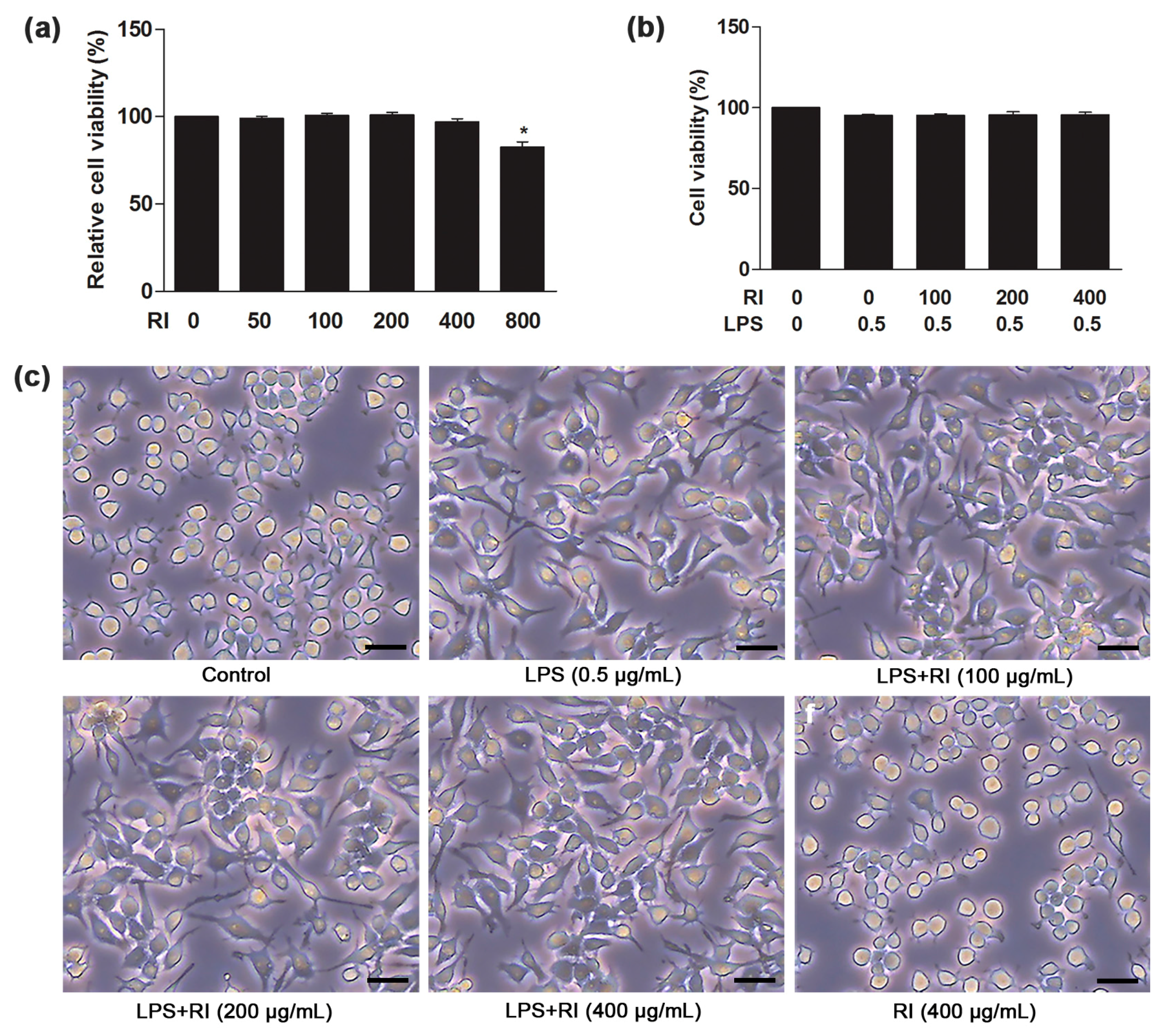
2.2. Effect of RI on Viability and Morphology of RAW 264.7 Cells
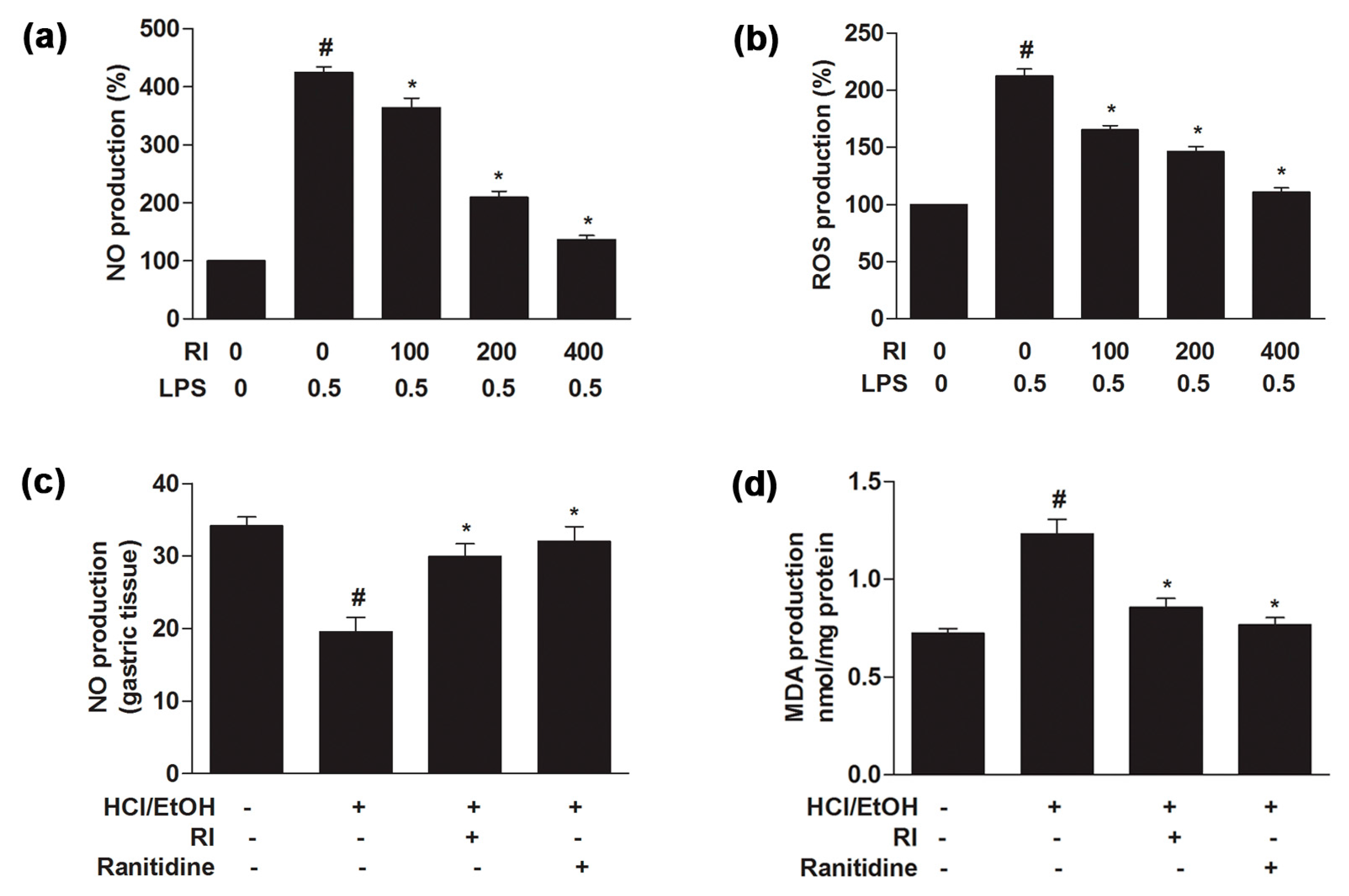
2.3. RI Attenuated the LPS-Induced NO and ROS Production in RAW 264.7 Cells
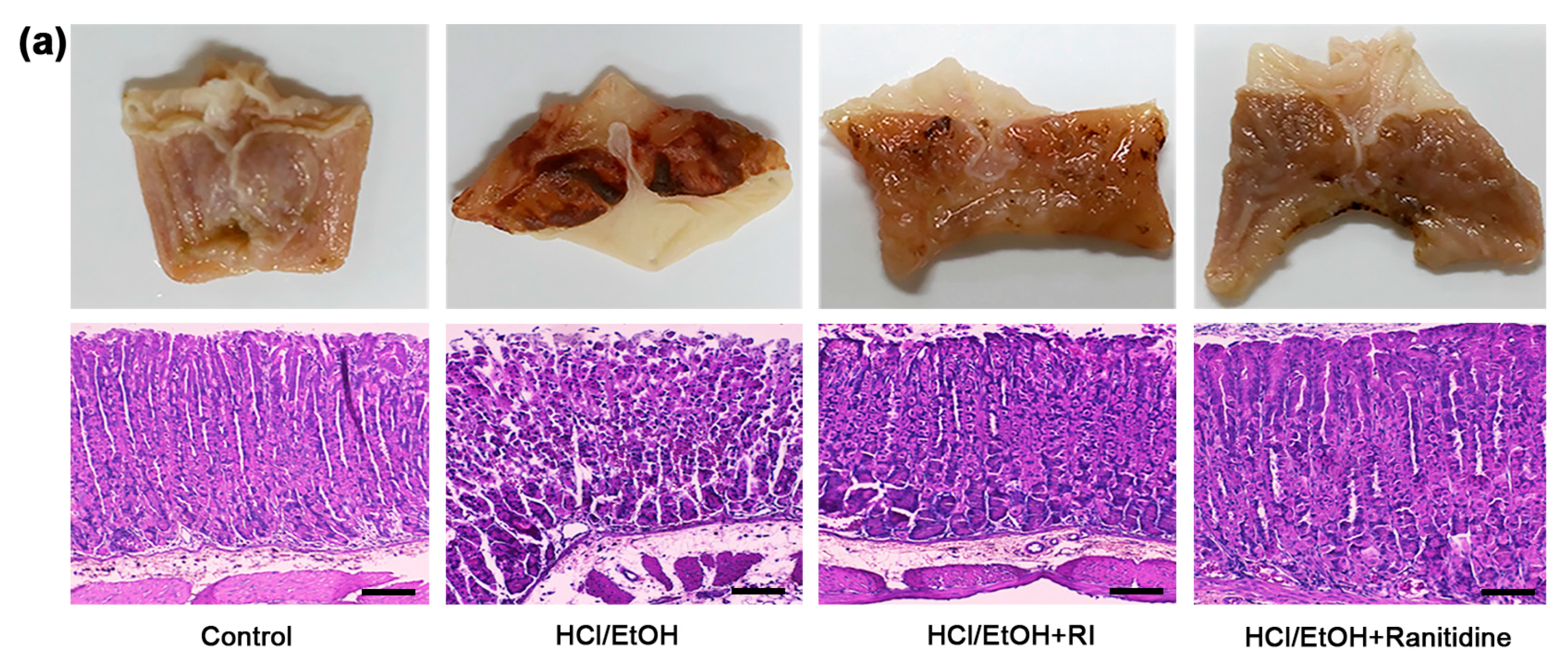
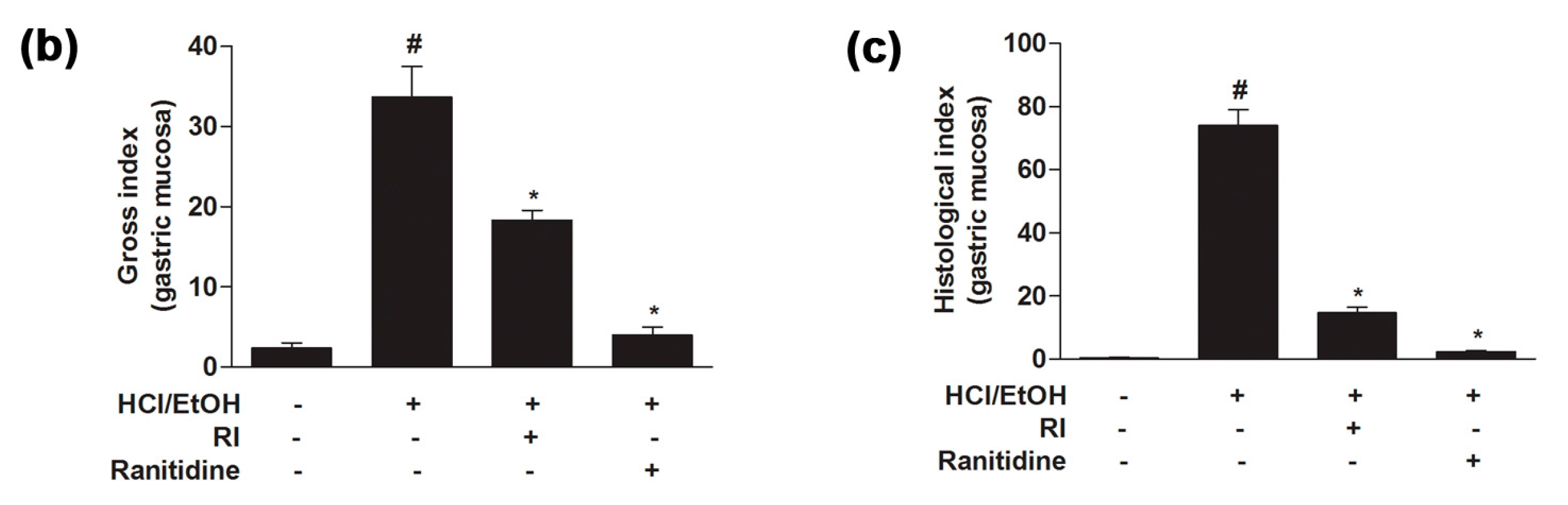
2.4. RI Improved the Gross and Histopathology of Gastric Tissue
2.5. RI Regulated the NO and MDA Production in Gastric Tissue
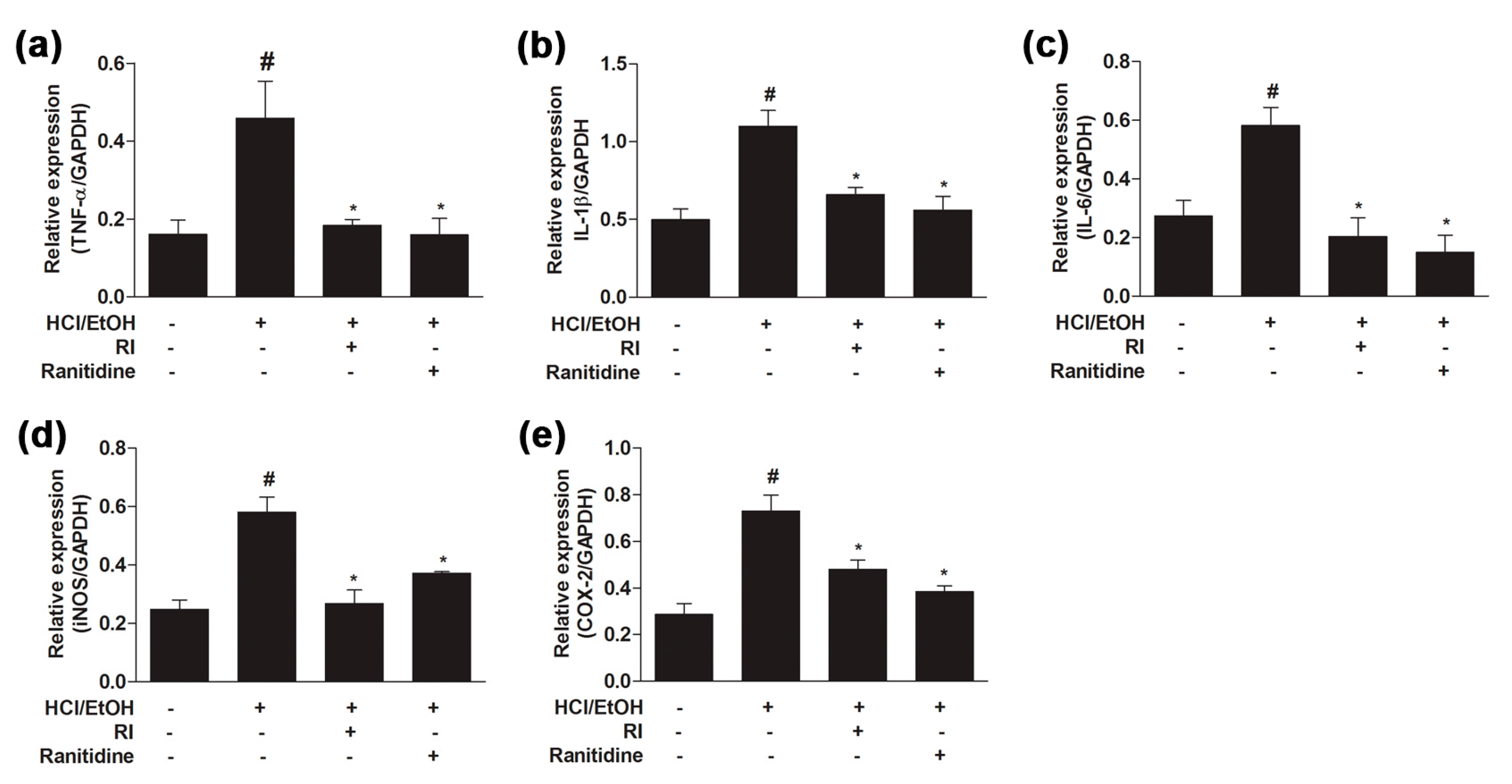
2.6. RI Suppressed the Activation Pro-Inflammatory Cytokines in Gastric Tissue
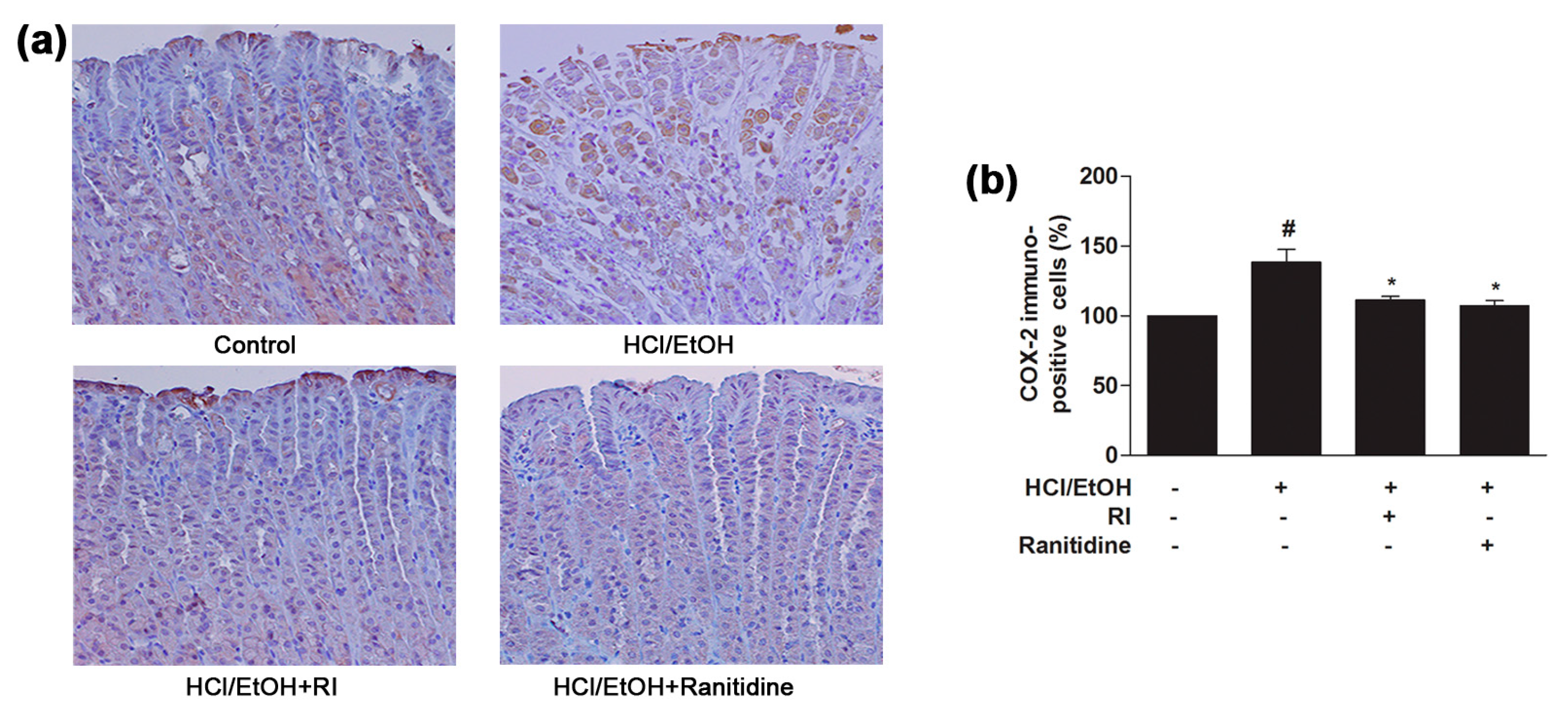
2.7. RI Inhibited the COX-2 Expression in Gastric Tissue
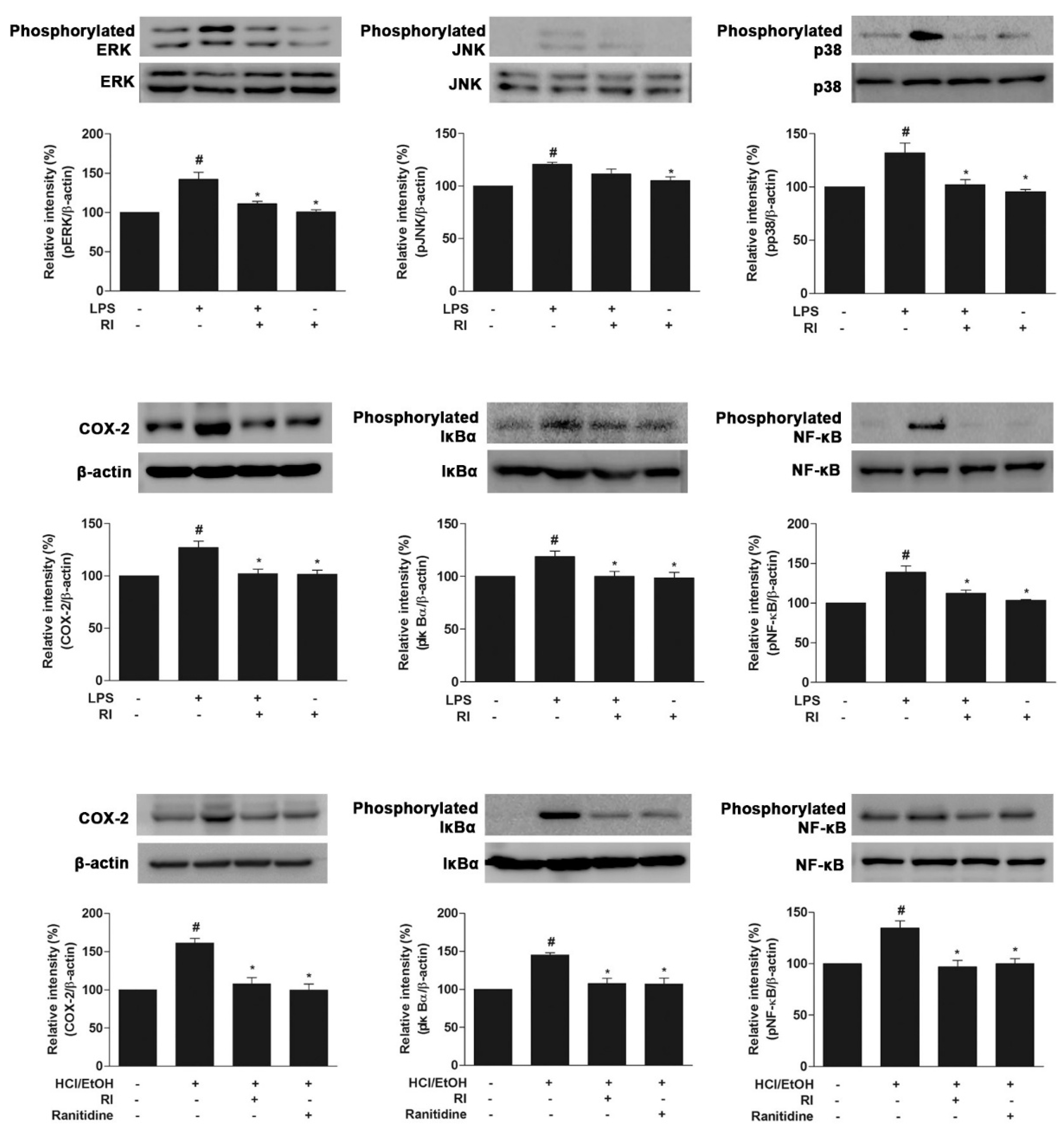
2.8. RI Blocked the MAPK Cascade, COX-2, and NF-κB Activation
3. Discussion
4. Materials and Methods
4.1. Chemicals and Antibodies
4.2. Collection and Preparation of Rabdosia Inflexa Extract
4.3. Phytochemical Analysis
4.4. RAW 264.7 Cells Culture
4.5. Cell Viability and Morphological Study
4.6. Measurement of NO and ROS in RAW 264.7 Cells
4.7. Mice Management and HCl/EtOH-Induced Gastric Ulcer Model
4.8. Gross and Histopathology of Gastric Mucosal Tissue
4.9. Analysis of Lipid Peroxidation and NO Production
4.10. RNA Extraction and Quantitative Real-Time Polymerase Chain Reaction (qPCR)
4.11. Immunohistochemical (IHC) Analysis
4.12. Western Blot Analysis
4.13. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rehm, J. The risks associated with alcohol use and alcoholism. Alcohol Res. Health 2011, 34, 135–143. [Google Scholar] [PubMed]
- Tarnawski, A.S.; Ahluwalia, A.; Jones, M.K. Increased susceptibility of aging gastric mucosa to injury: The mechanisms and clinical implications. World J. Gastroenterol. 2014, 20, 4467–4482. [Google Scholar] [CrossRef] [PubMed]
- Orywal, K.; Szmitkowski, M. Alcohol dehydrogenase and aldehyde dehydrogenase in malignant neoplasms. Clin. Exp. Med. 2017, 17, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Kwiecien, S.; Jasnos, K.; Magierowski, M.; Sliwowski, Z.; Pajdo, R.; Brzozowski, B.; Mach, T.; Wojcik, D.; Brzozowski, T. Lipid peroxidation, reactive oxygen species and antioxidative factors in the pathogenesis of gastric mucosal lesions and mechanism of protection against oxidative stress—Induced gastric injury. J. Physiol. Pharmacol. 2014, 65, 613–622. [Google Scholar] [PubMed]
- Singh, P.; Vishwakarma, S.P.; Singh, R.L. Antioxidant, oxidative DNA damage protective and antimicrobial activities of the plant Trigonella foenum-graecum. J. Sci. Food Agric. 2014, 94, 2497–2504. [Google Scholar] [CrossRef] [PubMed]
- Allavena, P.; Garlanda, C.; Borrello, M.G.; Sica, A.; Mantovani, A. Pathways connecting inflammation and cancer. Curr. Opin. Genet. Dev. 2008, 18, 3–10. [Google Scholar] [CrossRef] [PubMed]
- El-Maraghy, S.A.; Rizk, S.M.; Shahin, N.N. Gastroprotective effect of crocin in ethanol-induced gastric injury in rats. Chem. Biol. Interact. 2015, 229, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Akira, S. Toll-like receptors: Lessons from knockout mice. Biochem. Soc. Trans. 2000, 28, 551–556. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Zhang, W.; Wang, T.; Jiang, H.; Zhang, Z.; Fu, Y.; Yang, Z.; Cao, Y.; Zhang, N. Geniposide plays an anti-inflammatory role via regulating TLR4 and downstream signaling pathways in lipopolysaccharide-induced mastitis in mice. Inflammation 2014, 37, 1588–1598. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.L.; Dong, C. MAP kinases in immune responses. Cell. Mol. Immunol. 2005, 2, 20–27. [Google Scholar] [PubMed]
- Ryan, S.; McNicholas, W.T.; Taylor, C.T. A critical role for p38 map kinase in NF-kappaB signaling during intermittent hypoxia/reoxygenation. Biochem. Biophys. Res. Commun. 2007, 355, 728–733. [Google Scholar] [CrossRef] [PubMed]
- Dambrova, M.; Zvejniece, L.; Skapare, E.; Vilskersts, R.; Svalbe, B.; Baumane, L.; Muceniece, R.; Liepinsh, E. The anti-inflammatory and antinociceptive effects of NF-kappaB inhibitory guanidine derivative ME10092. Int. Immunopharmacol. 2010, 10, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, H.; Itokawa, H.; Ikuta, A. [Studies on the constituents of the flower of Camellia japonica (2)]. Yakugaku Zasshi 1984, 104, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Fujita, T.; Takeda, Y.; Yuasa, E.; Okamura, A.; Shingu, T.; Yokoi, T. Structure of Inflexinol, a New Cyto-Toxic Diterpene from Rabdosia-Inflexa. Phytochemistry 1982, 21, 903–905. [Google Scholar] [CrossRef]
- Kubo, I.; Nakanishi, K.; Kamikawa, T.; Isobe, T.; Kubota, T. Structure of Inflexin. Chem. Lett. 1977, 6, 99–102. [Google Scholar] [CrossRef]
- Ahn, D.W.; Ryu, J.K.; Kim, J.; Kim, Y.T.; Yoon, Y.B.; Lee, K.; Hong, J.T. Inflexinol reduces severity of acute pancreatitis by inhibiting nuclear factor-kappaB activation in cerulein-induced pancreatitis. Pancreas 2013, 42, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Salama, S.M.; Gwaram, N.S.; AlRashdi, A.S.; Khalifa, S.A.; Abdulla, M.A.; Ali, H.M.; El-Seedi, H.R. A Zinc Morpholine Complex Prevents HCl/Ethanol-Induced Gastric Ulcers in a Rat Model. Sci. Rep. 2016, 6, 29646. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.R.; Xiao, M.S.; Zhao, J.J.; Li, Z.H.; Xing, B.S.; Li, X.D.; Kong, M.Z.; Li, L.Y.; Zhang, Q.; Liu, Y.W.; et al. An Overview of Plant Phenolic Compounds and Their Importance in Human Nutrition and Management of Type 2 Diabetes. Molecules 2016, 21, 1374. [Google Scholar] [CrossRef] [PubMed]
- Mierziak, J.; Kostyn, K.; Kulma, A. Flavonoids as Important Molecules of Plant Interactions with the Environment. Molecules 2014, 19, 16240–16265. [Google Scholar] [CrossRef] [PubMed]
- Dzoyem, J.P.; Eloff, J.N. Anti-inflammatory, anticholinesterase and antioxidant activity of leaf extracts of twelve plants used traditionally to alleviate pain and inflammation in South Africa. J. Ethnopharmacol. 2015, 160, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Kadayat, T.; Kim, D.E.; Lee, E.S.; Park, P.H. TI-I-174, a Synthetic Chalcone Derivative, Suppresses Nitric Oxide Production in Murine Macrophages via Heme Oxygenase-1 Induction and Inhibition of AP-1. Biomol. Ther. 2014, 22, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Alli, R.; Vogel, P.; Geiger, T.L. IL-10 modulates DSS-induced colitis through a macrophage-ROS-NO axis. Mucosal Immunol. 2014, 7, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.S.; Jeong, D.; Yi, Y.S.; Lee, B.H.; Kim, T.W.; Htwe, K.M.; Kim, Y.D.; Yoon, K.D.; Hong, S.; Lee, W.S.; et al. Myrsine seguinii ethanolic extract and its active component quercetin inhibit macrophage activation and peritonitis induced by LPS by targeting to Syk/Src/IRAK-1. J. Ethnopharmacol. 2014, 151, 1165–1174. [Google Scholar] [CrossRef] [PubMed]
- Xie, M.; Chen, H.; Nie, S.; Tong, W.; Yin, J.; Xie, M. Gastroprotective effect of gamma-aminobutyric acid against ethanol-induced gastric mucosal injury. Chem. Biol. Interact. 2017, 272, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Oh, T.Y.; Kim, Y.K.; Baik, J.H.; So, S.; Hahm, K.B.; Surh, Y.J. Protective effects of green tea polyphenol extracts against ethanol-induced gastric mucosal damages in rats: Stress-responsive transcription factors and MAP kinases as potential targets. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2005, 579, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Calatayud, S.; Barrachina, D.; Esplugues, J.V. Nitric oxide: Relation to integrity, injury, and healing of the gastric mucosa. Microsc. Res. Tech. 2001, 53, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhao, X.; Sun, P.; Qian, J.; Shi, Y.; Wang, R. Preventive effect of Gardenia jasminoides on HCl/ethanol induced gastric injury in mice. J. Pharmacol. Sci. 2017, 133, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.; Yang, Y.; Kwak, Y.S.; Song, G.G.; Kim, M.Y.; Rhee, M.H.; Cho, J.Y. Ginsenoside Rc from Panax ginseng exerts anti-inflammatory activity by targeting TANK-binding kinase 1/interferon regulatory factor-3 and p38/ATF-2. J. Ginseng Res. 2017, 41, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Park, J.G.; Kim, S.C.; Kim, Y.H.; Yang, W.S.; Kim, Y.; Hong, S.; Kim, K.H.; Yoo, B.C.; Kim, S.H.; Kim, J.H.; et al. Anti-Inflammatory and Antinociceptive Activities of Anthraquinone-2-Carboxylic Acid. Mediat. Inflamm. 2016, 2016, 1903849. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Tian, X.; Gou, L.; Fu, X.; Li, S.; Lan, N.; Yin, X. Protective effect of l-citrulline against ethanol-induced gastric ulcer in rats. Environ. Toxicol. Pharmacol. 2012, 34, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Boligon, A.A.; de Freitas, R.B.; de Brum, T.F.; Waczuk, E.P.; Klimaczewski, C.V.; de Avila, D.S.; Athayde, M.L.; de Freitas Bauermann, L. Antiulcerogenic activity of Scutia buxifolia on gastric ulcers induced by ethanol in rats. Acta Pharm. Sin. B 2014, 4, 358–367. [Google Scholar] [CrossRef] [PubMed]
- Park, S.W.; Oh, T.Y.; Kim, Y.S.; Sim, H.; Park, S.J.; Jang, E.J.; Park, J.S.; Baik, H.W.; Hahm, K.B. Artemisia asiatica extracts protect against ethanol-induced injury in gastric mucosa of rats. J. Gastroenterol. Hepatol. 2008, 23, 976–984. [Google Scholar] [CrossRef] [PubMed]
- Mei, X.; Xu, D.; Xu, S.; Zheng, Y.; Xu, S. Novel role of Zn(II)-curcumin in enhancing cell proliferation and adjusting proinflammatory cytokine-mediated oxidative damage of ethanol-induced acute gastric ulcers. Chem. Biol. Interact. 2012, 197, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Akanda, M.R.; Park, B.Y. Involvement of MAPK/NF-kappaB signal transduction pathways: Camellia japonica mitigates inflammation and gastric ulcer. Biomed. Pharmacother. 2017, 95, 1139–1146. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Huang, H.; Niu, X.; Fan, T.; Mu, Q.; Li, H. Protective effect of tetrahydrocoptisine against ethanol-induced gastric ulcer in mice. Toxicol. Appl. Pharmacol. 2013, 272, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Ci, X.; Ren, R.; Xu, K.; Li, H.; Yu, Q.; Song, Y.; Wang, D.; Li, R.; Deng, X. Schisantherin A exhibits anti-inflammatory properties by down-regulating NF-kappaB and MAPK signaling pathways in lipopolysaccharide-treated RAW 264.7 cells. Inflammation 2010, 33, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.G.; Yu, Y.; Weiss, R.M.; Felder, R.B. Endoplasmic reticulum stress increases brain MAPK signaling, inflammation and renin-angiotensin system activity and sympathetic nerve activity in heart failure. Am. J. Physiol. Heart Circ. Physiol. 2016, 311, H871–H880. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Han, J.; Yang, S.; Hu, Y.; Liu, H.; Zhao, F. 11-O-acetylcyathatriol inhibits MAPK/p38-mediated inflammation in LPS-activated RAW 264.7 macrophages and has a protective effect on ethanol-induced gastric injury. Mol. Med. Rep. 2016, 14, 874–880. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Zhou, Y.; Shen, P. NF-kappaB and its regulation on the immune system. Cell. Mol. Immunol. 2004, 1, 343–350. [Google Scholar] [PubMed]
- Kang, J.W.; Yun, N.; Han, H.J.; Kim, J.Y.; Kim, J.Y.; Lee, S.M. Protective Effect of Flos Lonicerae against Experimental Gastric Ulcers in Rats: Mechanisms of Antioxidant and Anti-Inflammatory Action. Evid. Based Complement. Altern. Med. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Hinz, M.; Scheidereit, C. The IkappaB kinase complex in NF-kappaB regulation and beyond. EMBO Rep. 2014, 15, 46–61. [Google Scholar] [CrossRef] [PubMed]
- Akanda, M.; Tae, H.-J.; Kim, I.-S.; Ahn, D.; Tian, W.; Islam, A.; Nam, H.-H.; Choo, B.-K.; Park, B.-Y. Hepatoprotective Role of Hydrangea macrophylla against Sodium Arsenite-Induced Mitochondrial-Dependent Oxidative Stress via the Inhibition of MAPK/Caspase-3 Pathways. Int. J. Mol. Sci. 2017, 18, 1482. [Google Scholar] [CrossRef] [PubMed]
- Jing, L.; Ma, H.; Fan, P.; Gao, R.; Jia, Z. Antioxidant potential, total phenolic and total flavonoid contents of Rhododendron anthopogonoides and its protective effect on hypoxia-induced injury in PC12 cells. BMC Complement. Altern. Med. 2015, 15, 287. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yu, T.; Lee, Y.G.; Yang, W.S.; Oh, J.; Jeong, D.; Lee, S.; Kim, T.W.; Park, Y.C.; Sung, G.H.; et al. Methanol extract of Hopea odorata suppresses inflammatory responses via the direct inhibition of multiple kinases. J. Ethnopharmacol. 2013, 145, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Nam, S.Y.; Kim, N.; Lee, C.S.; Choi, K.D.; Lee, H.S.; Jung, H.C.; Song, I.S. Gastric mucosal protection via enhancement of MUC5AC and MUC6 by geranylgeranylacetone. Dig. Dis. Sci. 2005, 50, 2110–2120. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yu, T.; Jang, H.J.; Byeon, S.E.; Song, S.Y.; Lee, B.H.; Rhee, M.H.; Kim, T.W.; Lee, J.; Hong, S.; et al. In vitro and in vivo anti-inflammatory activities of Polygonum hydropiper methanol extract. J. Ethnopharmacol. 2012, 139, 616–625. [Google Scholar] [CrossRef] [PubMed]
| Plant Extract | Total Phenolic (mg GAE/g Extract) | Total Flavonoid (mg RU/g Extract) | Total Yield (%) |
|---|---|---|---|
| RI | 143.288 ± 1.68 | 256.301 ± 1.40 | 27.13 |
| Gene | Primers Sequence (5′–3′) | Genebank Accession No. |
|---|---|---|
| TNF-α | TTGACCTCAGCGCTGAGTTG | NM_013693 |
| CCTGTAGCCCACGTCGTAGC | ||
| IL-1β | CAGGATGAGGACATGAGCACC | XM_006498795 |
| CTCTGCAGACTCAAACTCCAC | ||
| IL-6 | GTACTCCAGAAGACCAGAGG | NM_001314054 |
| TGCTGGTGACAACCACGGCC | ||
| iNOS | CCCTTCCGAAGTTTCTGGCAGCAGC | XM_006532446 |
| GGCTGTCAGAGCCTCGTGGCTTTGG | ||
| COX-2 | CACTACATCCTGACCCACTT | NM_011198 |
| ATGCTCCTGCTTGAGTATGT | ||
| GAPDH | CACTCACGGCAAATTCAACGGCAC | XM_017321385 |
| GACTCCACGACATACTCAGCAC |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akanda, M.R.; Kim, I.-S.; Ahn, D.; Tae, H.-J.; Nam, H.-H.; Choo, B.-K.; Kim, K.; Park, B.-Y. Anti-Inflammatory and Gastroprotective Roles of Rabdosia inflexa through Downregulation of Pro-Inflammatory Cytokines and MAPK/NF-κB Signaling Pathways. Int. J. Mol. Sci. 2018, 19, 584. https://doi.org/10.3390/ijms19020584
Akanda MR, Kim I-S, Ahn D, Tae H-J, Nam H-H, Choo B-K, Kim K, Park B-Y. Anti-Inflammatory and Gastroprotective Roles of Rabdosia inflexa through Downregulation of Pro-Inflammatory Cytokines and MAPK/NF-κB Signaling Pathways. International Journal of Molecular Sciences. 2018; 19(2):584. https://doi.org/10.3390/ijms19020584
Chicago/Turabian StyleAkanda, Md Rashedunnabi, In-Shik Kim, Dongchoon Ahn, Hyun-Jin Tae, Hyeon-Hwa Nam, Byung-Kil Choo, Kyunghwa Kim, and Byung-Yong Park. 2018. "Anti-Inflammatory and Gastroprotective Roles of Rabdosia inflexa through Downregulation of Pro-Inflammatory Cytokines and MAPK/NF-κB Signaling Pathways" International Journal of Molecular Sciences 19, no. 2: 584. https://doi.org/10.3390/ijms19020584
APA StyleAkanda, M. R., Kim, I.-S., Ahn, D., Tae, H.-J., Nam, H.-H., Choo, B.-K., Kim, K., & Park, B.-Y. (2018). Anti-Inflammatory and Gastroprotective Roles of Rabdosia inflexa through Downregulation of Pro-Inflammatory Cytokines and MAPK/NF-κB Signaling Pathways. International Journal of Molecular Sciences, 19(2), 584. https://doi.org/10.3390/ijms19020584





