Recent Advances in Treatment of Coronary Artery Disease: Role of Science and Technology
Abstract
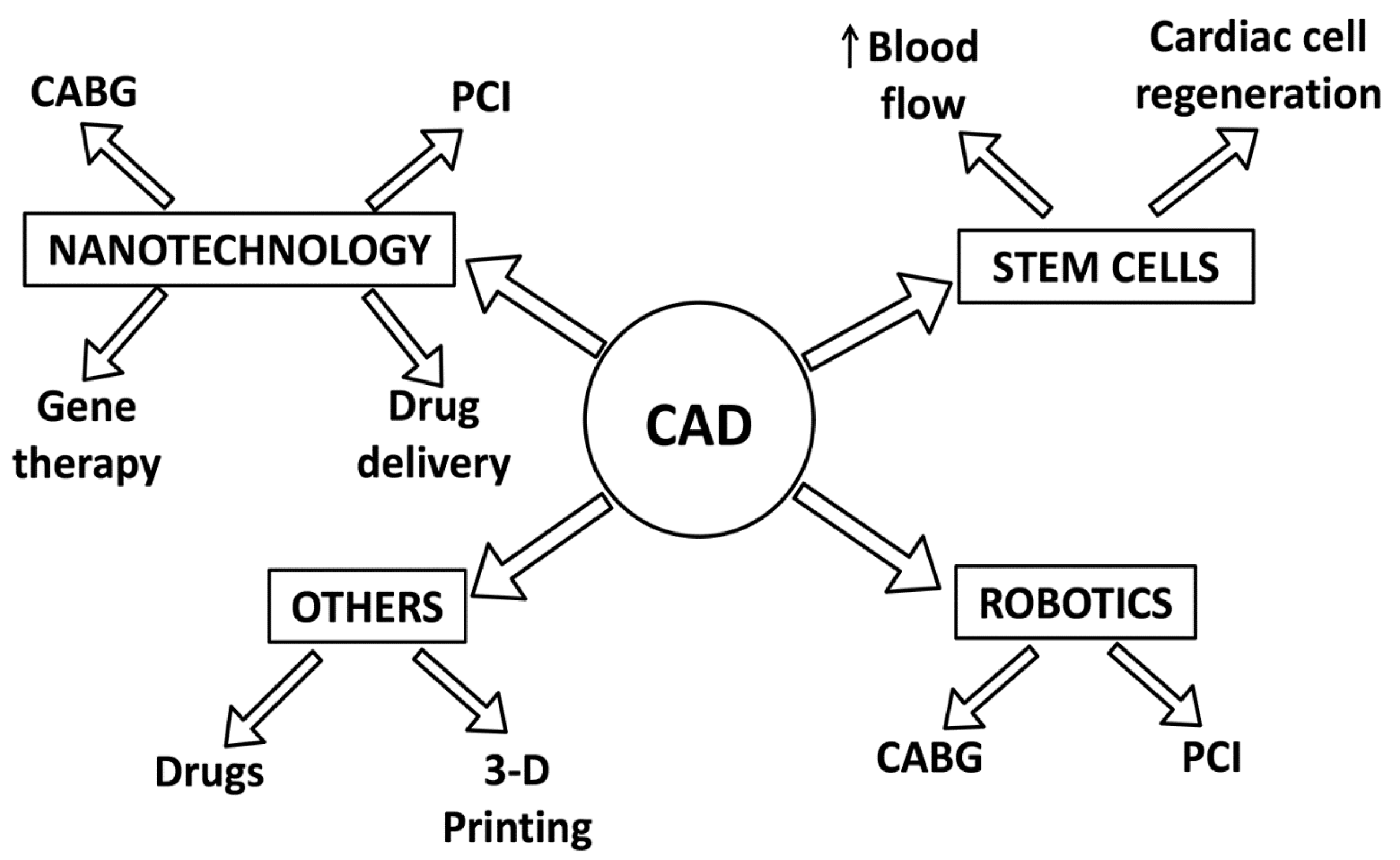
1. Introduction
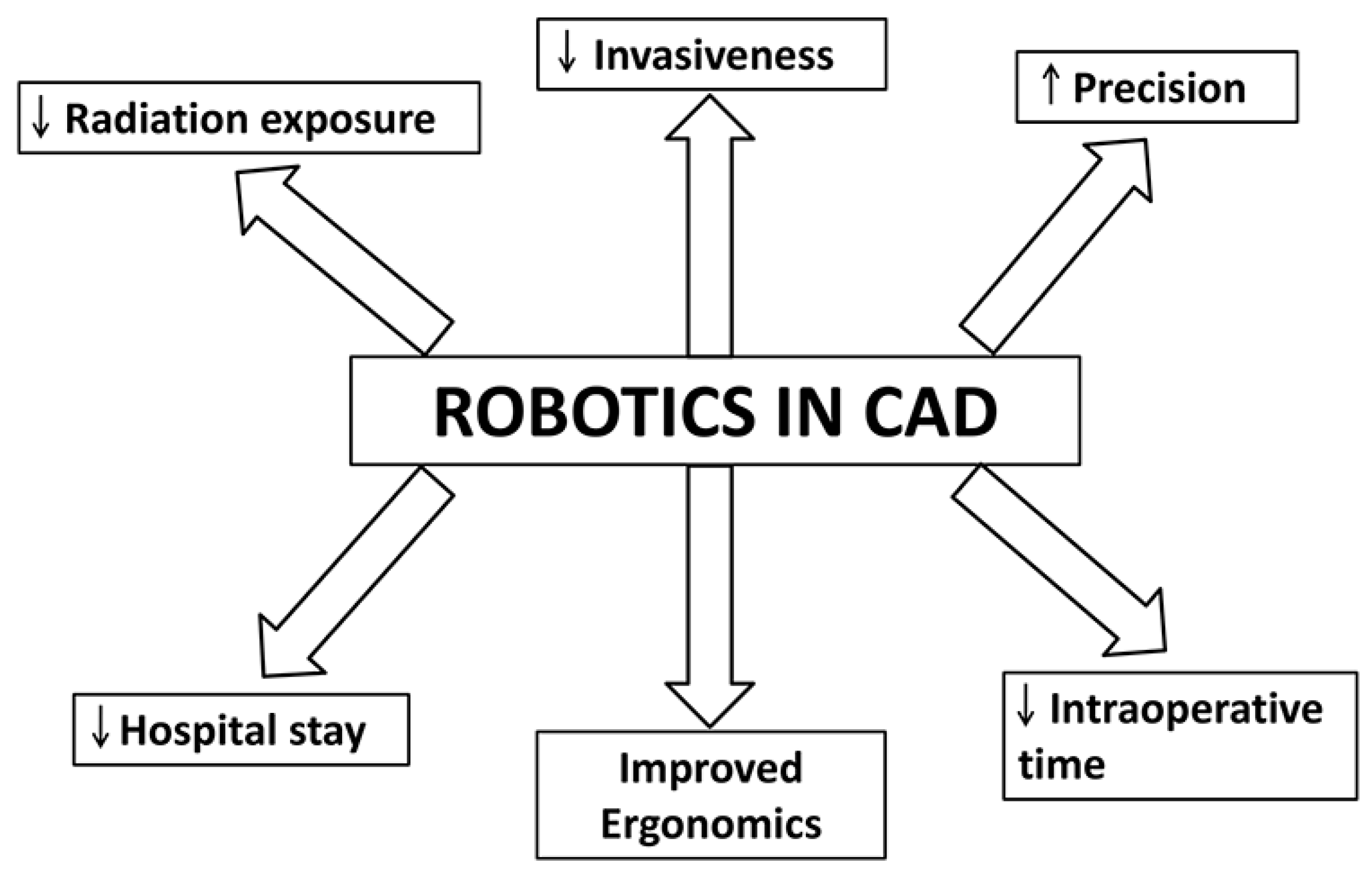
2. Robotics
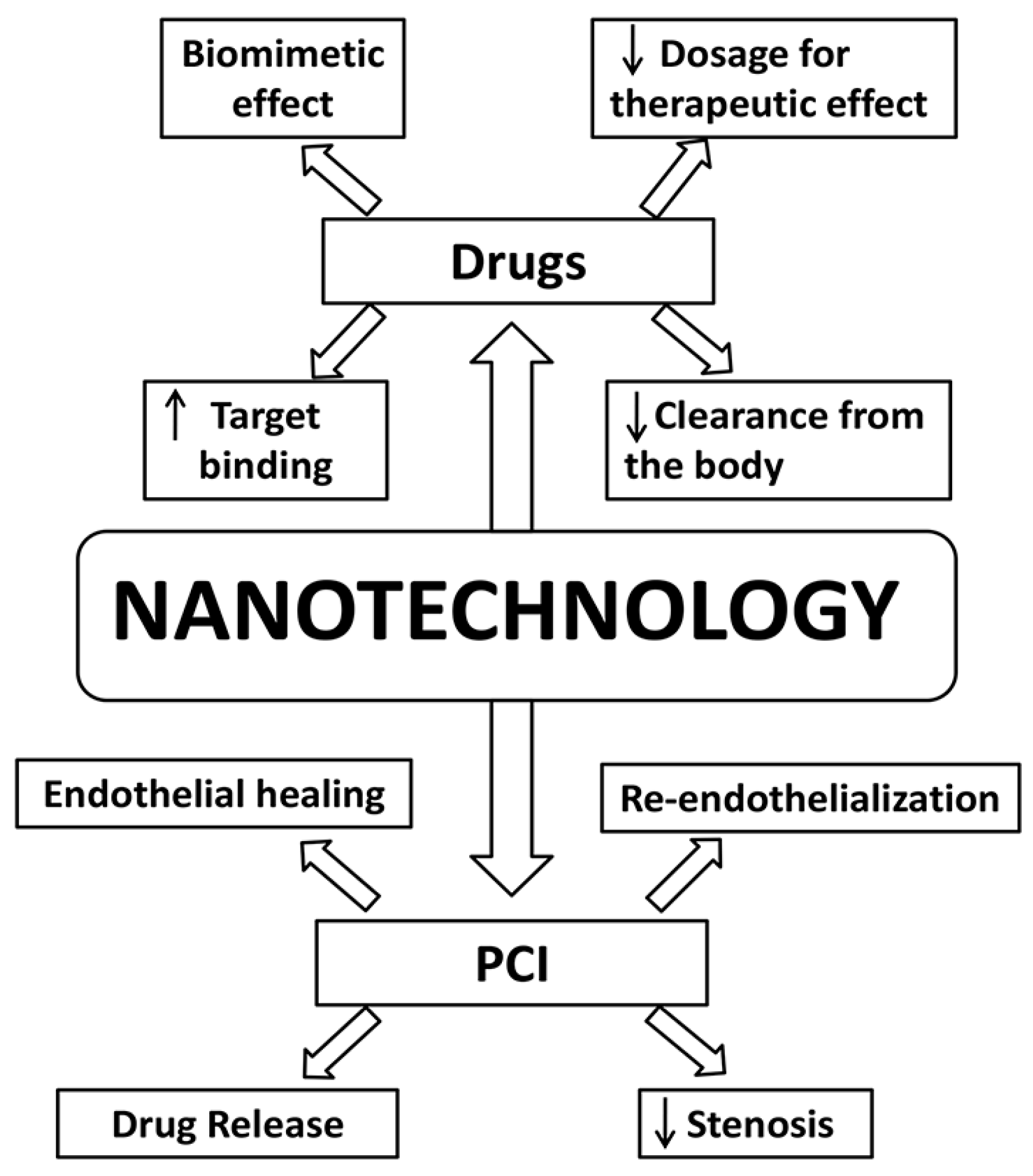
3. Nanotechnology
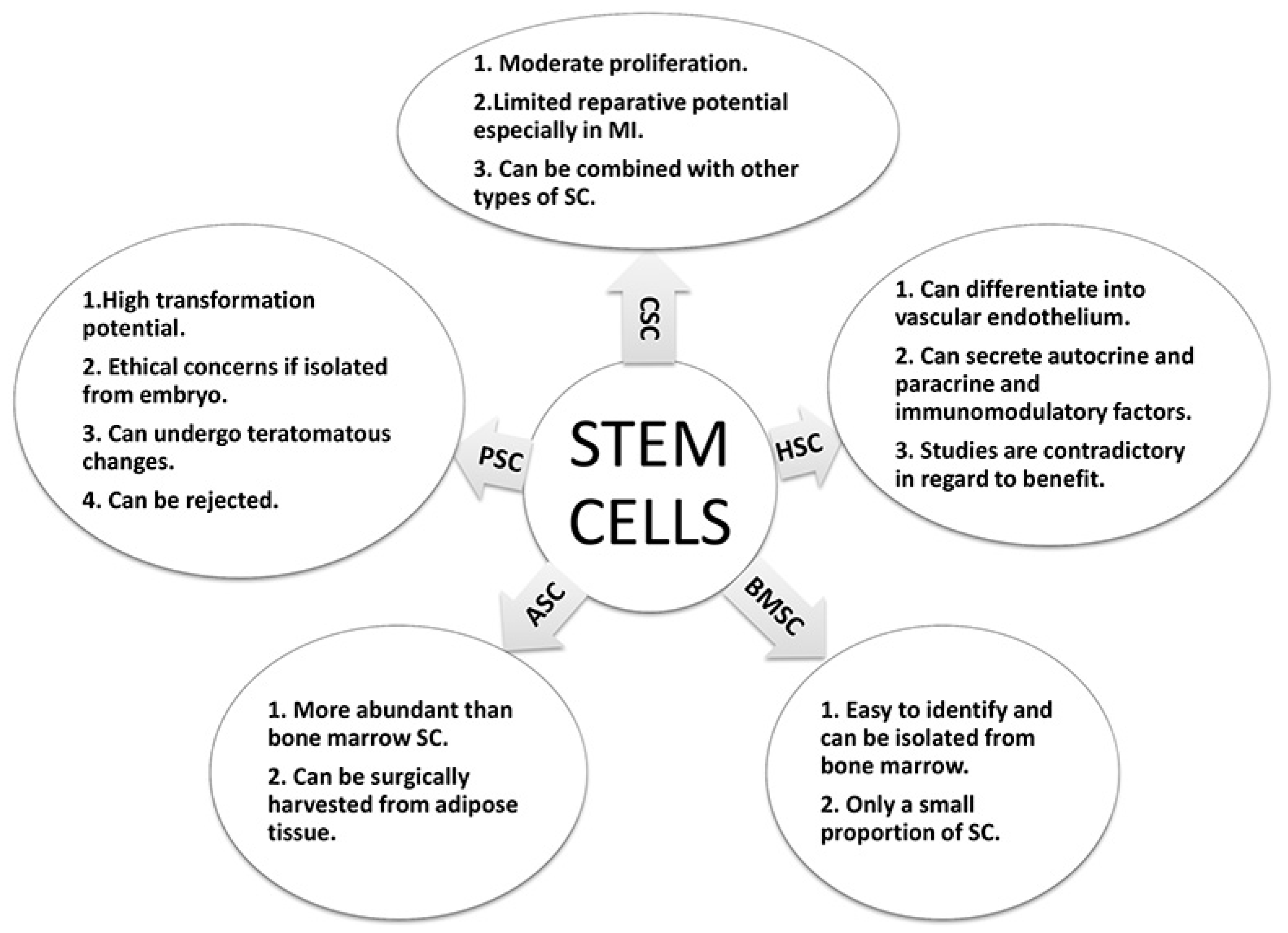
4. Stem Cells
5. Other Advancements
5.1. 3-D Printing
5.2. Drugs
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ACE | Angio-tensin-converting enzyme |
| ARNi | Angiotensin receptor-neprilysin inhibitor |
| CAD | Coronary artery disease |
| CABG | Coronary artery bypass graft |
| HDL | High-density lipoprotein |
| LCZ696 | Combination of sacubitril and valsartan |
| LVEF | Left ventricular ejection fraction |
| LDL | Low-density lipoprotein |
| MI | Acute myocardial infarction |
| PCI | Percutaneous coronary intervention |
| PCSK9 | Proprotein convertase subtilisin/kexin type 9 |
| TECAB | Totally endoscopic coronary bypass surgery |
References
- Ford, E.S.; Ajani, U.A.; Croft, J.B.; Critchley, J.A.; Labarthe, D.R.; Kottke, T.E.; Giles, W.H.; Capewell, S. Explaining the decrease in U.S. deaths from coronary disease, 1980–2000. N. Engl. J. Med. 2007, 356, 2388–2398. [Google Scholar] [CrossRef] [PubMed]
- Lozano, R.; Naghavi, M.; Foreman, K.; Lim, S.; Shibuya, K.; Aboyans, V.; Abraham, J.; Adair, T.; Aggarwal, R.; Ahn, S.Y.; et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2095–2128. [Google Scholar] [CrossRef]
- Lloyd-Jones, D.; Adams, R.J.; Brown, T.M.; Carnethon, M.; Dai, S.; de Simone, G.; Ferguson, T.B.; Ford, E.; Furie, K.; Gillespie, C.; et al. Executive summary: Heart disease and stroke statistics 2010 update: A report from the American Heart Association. Circulation 2010, 121, 948–954. [Google Scholar] [CrossRef] [PubMed]
- Nichols, M.; Townsend, N.; Scarborough, P.; Rayner, M. Cardiovascular disease in Europe 2014: Epidemiological update. Eur. Heart J. 2014, 35, 2950–2959. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, A.; Srivastava, S.; Velmurugan, M. Newer perspectives of coronary artery disease in young. World J. Cardiol. 2016, 8, 728–734. [Google Scholar] [CrossRef] [PubMed]
- Writing Group Members; Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; Das, S.R.; de Ferranti, S.; Despres, J.P.; et al. Heart disease and stroke statistics-2016 Update: A report from the American Heart Association. Circulation 2016, 133, e38. [Google Scholar] [CrossRef] [PubMed]
- Bonatti, J.; Vetrovec, G.; Riga, C.; Wazni, O.; Stadler, P. Robotic technology in cardiovascular medicine. Nat. Rev. Cardiol. 2014, 11, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Suri, R.M.; Antiel, R.M.; Burkhart, H.M.; Huebner, M.; Li, Z.; Eton, D.T.; Topilsky, T.; Sarano, M.E.; Schaff, H.V. Quality of life after early mitral valve repair using conventional and robotic approaches. Ann. Thorac. Surg. 2012, 93, 761–769. [Google Scholar] [CrossRef] [PubMed]
- Smilowitz, N.R.; Weisz, G. Robotic-assisted angioplasty: Current status and future possibilities. Curr. Cardiol. Rep. 2012, 14, 642–646. [Google Scholar] [CrossRef] [PubMed]
- Granada, J.F.; Delgado, J.A.; Uribe, M.P.; Fernandez, A.; Blanco, G.; Leon, M.B.; Weisz, G. First-in-human evaluation of a novel robotic-assisted coronary angioplasty system. JACC Cardiovasc. Interv. 2011, 4, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Weisz, G.; Metzger, D.C.; Caputo, R.P.; Delgado, J.A.; Marshall, J.J.; Vetrovec, G.W.; Reisman, M.; Waksman, R.; Granada, J.F.; Novack, V.; et al. Safety and feasibility of robotic percutaneous coronary intervention: PRECISE (Percutaneous Robotically-Enhanced Coronary Intervention) Study. J. Am. Coll. Cardiol. 2013, 61, 1596–1600. [Google Scholar] [CrossRef] [PubMed]
- Kypson, A.P. Recent trends in minimally invasive cardiac surgery. Cardiology 2007, 107, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, M.C.; Vesely, M.R.; Lee, J.D.; Lehr, E.J.; Wehman, B.; Bonaros, N.; Schachner, T.; Friedrich, G.J.; Zimrin, D.A.; Bonatti, J.O. Robotically assisted hybrid coronary revascularization: Does sequence of intervention matter? Innovations 2013, 8, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Katz, M.R.; Van Praet, F.; de Canniere, D.; Murphy, D.; Siwek, L.; Seshadri-Kreaden, U.; Friedrich, G.; Bonatti, J. Integrated coronary revascularization: Percutaneous coronary intervention plus robotic totally endoscopic coronary artery bypass. Circulation 2006, 114 (Suppl. 1), 473–476. [Google Scholar] [CrossRef] [PubMed]
- Reicher, B.; Poston, R.S.; Mehra, M.R.; Joshi, A.; Odonkor, P.; Kon, Z.; Reyes, P.A.; Zimrin, D.A. Simultaneous “hybrid” percutaneous coronary intervention and minimally invasive surgical bypass grafting: Feasibility, safety, and clinical outcomes. Am. Heart J. 2008, 155, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Fitchett, D.H.; Gupta, M.; Farkouh, M.E.; Verma, S. Cardiology Patient Page: Coronary artery revascularization in patients with diabetes mellitus. Circulation 2014, 130, e104–e106. [Google Scholar] [CrossRef] [PubMed]
- Dogan, S.; Aybek, T.; Andressen, E.; Byhahn, C.; Mierdl, S.; Westphal, K.; Matheis, G.; Moritz, A.; Wimmer-Greinecker, G. Totally endoscopic coronary artery bypass grafting on cardiopulmonary bypass with robotically enhanced telemanipulation: Report of forty-five cases. J. Thorac. Cardiovasc. Surg. 2002, 123, 1125–1131. [Google Scholar] [CrossRef] [PubMed]
- Kappert, U.; Schneider, J.; Cichon, R.; Gulielmos, V.; Tugtekin, S.M.; Nicolai, J.; Matschke, K.; Schueler, S. Development of robotic enhanced endoscopic surgery for the treatment of coronary artery disease. Circulation 2001, 104 (Suppl. 1), 102–107. [Google Scholar] [CrossRef]
- Mohr, F.W.; Falk, V.; Diegeler, A.; Walther, T.; Gummert, J.F.; Bucerius, J.; Jacobs, S.; Autschbach, R. Computer-enhanced “robotic” cardiac surgery: Experience in 148 patients. J. Thorac. Cardiovasc. Surg. 2001, 121, 842–853. [Google Scholar] [CrossRef] [PubMed]
- Bonaros, N.; Schachner, T.; Wiedemann, D.; Oehlinger, A.; Ruetzler, E.; Feuchtner, G.; Kolbitsch, C.; Velik-Salchner, C.; Friedrich, G.; Pachinger, O.; et al. Quality of life improvement after robotically assisted coronary artery bypass grafting. Cardiology 2009, 114, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Mihaljevic, T.; Jarrett, C.M.; Gillinov, A.M.; Williams, S.J.; deVilliers, P.A.; Stewart, W.J.; Svensson, L.G.; Sabik, J.F., III; Blackstone, E.H. Robotic repair of posterior mitral valve prolapse versus conventional approaches: Potential realized. J. Thorac. Cardiovasc. Surg. 2011, 141, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Schachner, T.; Bonaros, N.; Wiedemann, D.; Weidinger, F.; Feuchtner, G.; Friedrich, G.; Laufer, G.; Bonatti, J. Training surgeons to perform robotically assisted totally endoscopic coronary surgery. Ann. Thorac. Surg. 2009, 88, 523–527. [Google Scholar] [CrossRef] [PubMed]
- Cho, B.H.; Park, J.R.; Nakamura, M.T.; Odintsov, B.M.; Wallig, M.A.; Chung, B.H. Synthetic dimyristoylphosphatidylcholine liposomes assimilating into high-density lipoprotein promote regression of atherosclerotic lesions in cholesterol-fed rabbits. Exp. Biol. Med. 2010, 235, 1194–1203. [Google Scholar] [CrossRef] [PubMed]
- Winter, P.M.; Neubauer, A.M.; Caruthers, S.D.; Harris, T.D.; Robertson, J.D.; Williams, T.A.; Schmieder, A.H.; Hu, G.; Allen, J.S.; Lacy, E.K.; et al. Endothelial alpha(v)beta3 integrin-targeted fumagillin nanoparticles inhibit angiogenesis in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 2103–2109. [Google Scholar] [CrossRef] [PubMed]
- Rhee, J.W.; Wu, J.C. Advances in nanotechnology for the management of coronary artery disease. Trends Cardiovasc. Med. 2013, 23, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Myerson, J.; He, L.; Lanza, G.; Tollefsen, D.; Wickline, S. Thrombin-inhibiting perfluorocarbon nanoparticles provide a novel strategy for the treatment and magnetic resonance imaging of acute thrombosis. J. Thromb. Haemost. 2011, 9, 1292–1300. [Google Scholar] [CrossRef] [PubMed]
- Peters, D.; Kastantin, M.; Kotamraju, V.R.; Karmali, P.P.; Gujraty, K.; Tirrell, M.; Ruoslahti, E. Targeting atherosclerosis by using modular, multifunctional micelles. Proc. Natl. Acad. Sci. USA 2009, 106, 9815–9819. [Google Scholar] [CrossRef] [PubMed]
- Fredman, G.; Kamaly, N.; Spolitu, S.; Milton, J.; Ghorpade, D.; Chiasson, R.; Kuriakose, G.; Perretti, M.; Farokhzad, O.; Tabas, I. Targeted nanoparticles containing the proresolving peptide Ac2-26 protect against advanced atherosclerosis in hypercholesterolemic mice. Sci. Transl. Med. 2015, 7. [Google Scholar] [CrossRef] [PubMed]
- Leuschner, F.; Dutta, P.; Gorbatov, R.; Novobrantseva, T.I.; Donahoe, J.S.; Courties, G.; Lee, K.M.; Kim, J.I.; Markmann, J.F.; Marinelli, B.; et al. Therapeutic siRNA silencing in inflammatory monocytes in mice. Nat. Biotechnol. 2011, 29, 1005–1010. [Google Scholar] [CrossRef] [PubMed]
- Lobatto, M.E.; Fayad, Z.A.; Silvera, S.; Vucic, E.; Calcagno, C.; Mani, V.; Dickson, S.D.; Nicolay, K.; Banciu, M.; Schiffelers, R.M.; et al. Multimodal clinical imaging to longitudinally assess a nanomedical anti-inflammatory treatment in experimental atherosclerosis. Mol. Pharm. 2010, 7, 2020–2029. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Lobatto, M.E.; Hassing, L.; van der Staay, S.; van Rijs, S.M.; Calcagno, C.; Braza, M.S.; Baxter, S.; Fay, F.; Sanchez-Gaytan, B.L.; et al. Inhibiting macrophage proliferation suppresses atherosclerotic plaque inflammation. Sci. Adv. 2015, 1. [Google Scholar] [CrossRef] [PubMed]
- Reddy, M.K.; Vasir, J.K.; Sahoo, S.K.; Jain, T.K.; Yallapu, M.M.; Labhasetwar, V. Inhibition of apoptosis through localized delivery of rapamycin-loaded nanoparticles prevented neointimal hyperplasia and reendothelialized injured artery. Circ. Cardiovasc. Interv. 2008, 1, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Amin, D.; Messersmith, P.B.; Anthony, J.E.; Prud’homme, R.K. Polymer directed self-assembly of pH-responsive antioxidant nanoparticles. Langmuir 2015, 31, 3612–3620. [Google Scholar] [CrossRef] [PubMed]
- Costa, J.R., Jr.; Abizaid, A.; Costa, R.; Feres, F.; Tanajura, L.F.; Abizaid, A.; Maldonado, G.; Staico, R.; Siqueira, D.; Sousa, A.G.; et al. 1-year results of the hydroxyapatite polymer-free sirolimus-eluting stent for the treatment of single de novo coronary lesions: The VESTASYNC I trial. JACC Cardiovasc. Interv. 2009, 2, 422–427. [Google Scholar] [CrossRef] [PubMed]
- Rajender, G.; Narayanan, N.G. Liquid chromatography-tandem mass spectrometry method for determination of Sirolimus coated drug eluting nano porous carbon stents. Biomed. Chromatogr. 2010, 24, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Tsukie, N.; Nakano, K.; Matoba, T.; Masuda, S.; Iwata, E.; Miyagawa, M.; Zhao, G.; Meng, W.; Kishimoto, J.; Sunagawa, K.; et al. Pitavastatin-incorporated nanoparticle-eluting stents attenuate in-stent stenosis without delayed endothelial healing effects in a porcine coronary artery model. J. Atheroscler. Thromb. 2013, 20, 32–45. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Liong, M.; Li, Z.; Zink, J.I.; Tamanoi, F. Biocompatibility, biodistribution, and drug-delivery efficiency of mesoporous silica nanoparticles for cancer therapy in animals. Small 2010, 6, 1794–1805. [Google Scholar] [CrossRef] [PubMed]
- Acharya, G.; Lee, C.H.; Lee, Y. Optimization of cardiovascular stent against restenosis: Factorial design-based statistical analysis of polymer coating conditions. PLoS ONE 2012, 7, e43100. [Google Scholar] [CrossRef] [PubMed]
- Danenberg, H.D.; Golomb, G.; Groothuis, A.; Gao, J.; Epstein, H.; Swaminathan, R.V.; Seifert, P.; Edelman, E.R. Liposomal alendronate inhibits systemic innate immunity and reduces in-stent neointimal hyperplasia in rabbits. Circulation 2003, 108, 2798–2804. [Google Scholar] [CrossRef] [PubMed]
- Kolodgie, F.D.; John, M.; Khurana, C.; Farb, A.; Wilson, P.S.; Acampado, E.; Desai, N.; Soon-Shiong, P.; Virmani, R. Sustained reduction of in-stent neointimal growth with the use of a novel systemic nanoparticle paclitaxel. Circulation 2002, 106, 1195–1198. [Google Scholar] [CrossRef] [PubMed]
- Margolis, J.; McDonald, J.; Heuser, R.; Klinke, P.; Waksman, R.; Virmani, R.; Desai, N.; Hilton, D. Systemic nanoparticle paclitaxel (nab-paclitaxel) for in-stent restenosis I (SNAPIST-I): A first-in-human safety and dose-finding study. Clin. Cardiol. 2007, 30, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Stinchcombe, T.E. Nanoparticle albumin-bound paclitaxel: A novel Cremphor-EL-free formulation of paclitaxel. Nanomedicine 2007, 2, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Joo, J.R.; Nam, H.Y.; Nam, S.H.; Baek, I.; Park, J.S. Thermal process for enhancing mechanical strength of PLGA nanoparticle layers on coronary stents. Bull. Korean Chem. Soc. 2009, 30, 1985–1988. [Google Scholar]
- Chan, J.M.; Rhee, J.W.; Drum, C.L.; Bronson, R.T.; Golomb, G.; Langer, R.; Farokhzad, O.C. In vivo prevention of arterial restenosis with paclitaxel-encapsulated targeted lipid-polymeric nanoparticles. Proc. Natl. Acad. Sci. USA 2011, 108, 19347–19352. [Google Scholar] [CrossRef] [PubMed]
- Chorny, M.; Fishbein, I.; Yellen, B.B.; Alferiev, I.S.; Bakay, M.; Ganta, S.; Adamo, R.; Amiji, M.; Friedman, G.; Levy, R.J. Targeting stents with local delivery of paclitaxel-loaded magnetic nanoparticles using uniform fields. Proc. Natl. Acad. Sci. USA 2010, 107, 8346–8351. [Google Scholar] [CrossRef] [PubMed]
- Joner, M.; Morimoto, K.; Kasukawa, H.; Steigerwald, K.; Merl, S.; Nakazawa, G.; John, M.C.; Finn, A.V.; Acampado, E.; Kolodgie, F.D.; et al. Site-specific targeting of nanoparticle prednisolone reduces in-stent restenosis in a rabbit model of established atheroma. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1960–1966. [Google Scholar] [CrossRef] [PubMed]
- Lanza, G.M.; Yu, X.; Winter, P.M.; Abendschein, D.R.; Karukstis, K.K.; Scott, M.J.; Chinen, L.K.; Fuhrhop, R.W.; Scherrer, D.E.; Wickline, S.A. Targeted antiproliferative drug delivery to vascular smooth muscle cells with a magnetic resonance imaging nanoparticle contrast agent: Implications for rational therapy of restenosis. Circulation 2002, 106, 2842–2847. [Google Scholar] [CrossRef] [PubMed]
- Bakhshi, R.; Darbyshire, A.; Evans, J.E.; You, Z.; Lu, J.; Seifalian, A.M. Polymeric coating of surface modified nitinol stent with POSS-nanocomposite polymer. Colloids Surf. B Biointerfaces 2011, 86, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Ceylan, H.; Tekinay, A.B.; Guler, M.O. Selective adhesion and growth of vascular endothelial cells on bioactive peptide nanofiber functionalized stainless steel surface. Biomaterials 2011, 32, 8797–8805. [Google Scholar] [CrossRef] [PubMed]
- Kushwaha, M.; Anderson, J.M.; Bosworth, C.A.; Andukuri, A.; Minor, W.P.; Lancaster, J.R., Jr.; Anderson, P.G.; Brott, B.C.; Jun, H.W. A nitric oxide releasing, self assembled peptide amphiphile matrix that mimics native endothelium for coating implantable cardiovascular devices. Biomaterials 2010, 31, 1502–1508. [Google Scholar] [CrossRef] [PubMed]
- Polyak, B.; Fishbein, I.; Chorny, M.; Alferiev, I.; Williams, D.; Yellen, B.; Friedman, G.; Levy, R.J. High field gradient targeting of magnetic nanoparticle-loaded endothelial cells to the surfaces of steel stents. Proc. Natl. Acad. Sci. USA 2008, 105, 698–703. [Google Scholar] [CrossRef] [PubMed]
- Hashi, C.K.; Zhu, Y.; Yang, G.Y.; Young, W.L.; Hsiao, B.S.; Wang, K.; Chu, B.; Li, S. Antithrombogenic property of bone marrow mesenchymal stem cells in nanofibrous vascular grafts. Proc. Natl. Acad. Sci. USA 2007, 104, 11915–11920. [Google Scholar] [CrossRef] [PubMed]
- Stankus, J.J.; Soletti, L.; Fujimoto, K.; Hong, Y.; Vorp, D.A.; Wagner, W.R. Fabrication of cell microintegrated blood vessel constructs through electrohydrodynamic atomization. Biomaterials 2007, 28, 2738–2746. [Google Scholar] [CrossRef] [PubMed]
- Yin, R.X.; Yang, D.Z.; Wu, J.Z. Nanoparticle drug- and gene-eluting stents for the prevention and treatment of coronary restenosis. Theranostics 2014, 4, 175–200. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Zheng, Y.; Yin, T.; Tang, C.; Yu, Q.; Wang, G. Coronary drug-eluting stents: From design optimization to newer strategies. J. Biomed. Mater. Res. A 2014, 102, 1625–1640. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.G.; Lee, Y.; Park, T.G. Controlled gene-eluting metal stent fabricated by bio-inspired surface modification with hyaluronic acid and deposition of DNA/PEI polyplexes. Int. J. Pharm. 2010, 384, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Nakano, K.; Egashira, K.; Masuda, S.; Funakoshi, K.; Zhao, G.; Kimura, S.; Matoba, T.; Sueishi, K.; Endo, Y.; Kawashima, Y.; et al. Formulation of nanoparticle-eluting stents by a cationic electrodeposition coating technology: Efficient nano-drug delivery via bioabsorbable polymeric nanoparticle-eluting stents in porcine coronary arteries. JACC Cardiovasc. Interv. 2009, 2, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Paul, A.; Shao, W.; Shum-Tim, D.; Prakash, S. The attenuation of restenosis following arterial gene transfer using carbon nanotube coated stent incorporating TAT/DNA(Ang1+Vegf) nanoparticles. Biomaterials 2012, 33, 7655–7664. [Google Scholar] [CrossRef] [PubMed]
- Kastrup, J. Gene therapy and angiogenesis in patients with coronary artery disease. Expert Rev. Cardiovasc. Ther. 2010, 8, 1127–1138. [Google Scholar] [CrossRef] [PubMed]
- Kastrup, J. Stem cells therapy for cardiovascular repair in ischemic heart disease: How to predict and secure optimal outcome? EPMA J. 2011, 2, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Janssens, S.; Dubois, C.; Bogaert, J.; Theunissen, K.; Deroose, C.; Desmet, W.; Kalantzi, M.; Herbots, L.; Sinnaeve, P.; Dens, J.; et al. Autologous bone marrow-derived stem-cell transfer in patients with ST-segment elevation myocardial infarction: Double-blind, randomised controlled trial. Lancet 2006, 367, 113–121. [Google Scholar] [CrossRef]
- Lunde, K.; Solheim, S.; Aakhus, S.; Arnesen, H.; Abdelnoor, M.; Egeland, T.; Endresen, K.; Ilebekk, A.; Mangschau, A.; Fjeld, J.G.; et al. Intracoronary injection of mononuclear bone marrow cells in acute myocardial infarction. N. Engl. J. Med. 2006, 355, 1199–1209. [Google Scholar] [CrossRef] [PubMed]
- Perin, E.C.; Dohmann, H.F.; Borojevic, R.; Silva, S.A.; Sousa, A.L.; Mesquita, C.T.; Rossi, M.I.; Carvalho, A.C.; Dutra, H.S.; Dohmann, H.J.; et al. Transendocardial, autologous bone marrow cell transplantation for severe, chronic ischemic heart failure. Circulation 2003, 107, 2294–2302. [Google Scholar] [CrossRef] [PubMed]
- Schachinger, V.; Erbs, S.; Elsasser, A.; Haberbosch, W.; Hambrecht, R.; Holschermann, H.; Yu, J.; Corti, R.; Mathey, D.G.; Hamm, C.W.; et al. Intracoronary bone marrow-derived progenitor cells in acute myocardial infarction. N. Engl. J. Med. 2006, 355, 1210–1221. [Google Scholar] [CrossRef] [PubMed]
- Gunaseeli, I.; Doss, M.X.; Antzelevitch, C.; Hescheler, J.; Sachinidis, A. Induced pluripotent stem cells as a model for accelerated patient- and disease-specific drug discovery. Curr. Med. Chem. 2010, 17, 759–766. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Piuzzi, N.S.; Chahla, J.; Jiandong, H.; Chughtai, M.; LaPrade, R.F.; Mont, M.A.; Muschler, G.F.; Pascual-Garrido, C. Analysis of cell therapies used in clinical trials for the treatment of osteonecrosis of the femoral head: A systematic review of the literature. J. Arthroplast. 2017, 32, 2612–2618. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.; Matsubara, Y.; Masuda, Y.; Togashi, K.; Ohno, T.; Tamura, T.; Toyoshima, Y.; Sugimachi, K.; Toyoda, M.; Marc, H.; et al. Characterization of adipose tissue-derived cells isolated with the Celution system. Cytotherapy 2008, 10, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Li, J.Y.; Christophersen, N.S.; Hall, V.; Soulet, D.; Brundin, P. Critical issues of clinical human embryonic stem cell therapy for brain repair. Trends Neurosci. 2008, 31, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Beltrami, A.P.; Barlucchi, L.; Torella, D.; Baker, M.; Limana, F.; Chimenti, S.; Kasahara, H.; Rota, M.; Musso, E.; Urbanek, K.; et al. Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell 2003, 114, 763–776. [Google Scholar] [CrossRef]
- Ellison, G.M.; Waring, C.D.; Vicinanza, C.; Torella, D. Physiological cardiac remodelling in response to endurance exercise training: Cellular and molecular mechanisms. Heart 2012, 98, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.S.K.; Shueh, Y.Z.; Liu, Y.W.; Hsieh, P.C.H. Harnessing endogenous intra- and extra-cardiac stem cells for cardiac regeneration—Hope or hype? Drug Discov. Today Ther. Strateg. 2009, 6, 127–133. [Google Scholar] [CrossRef]
- Barile, L.; Messina, E.; Giacomello, A.; Marban, E. Endogenous cardiac stem cells. Prog. Cardiovasc. Dis. 2007, 50, 31–48. [Google Scholar] [CrossRef] [PubMed]
- Leri, A. Human cardiac stem cells: The heart of a truth. Circulation 2009, 120, 2515–2518. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.R.; Barile, L.; Messina, E.; Marban, E. Stem cells in the heart: What’s the buzz all about?—Part 1: Preclinical considerations. Heart Rhythm 2008, 5, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Hatzistergos, K.E.; Quevedo, H.; Oskouei, B.N.; Hu, Q.; Feigenbaum, G.S.; Margitich, I.S.; Mazhari, R.; Boyle, A.J.; Zambrano, J.P.; Rodriguez, J.E.; et al. Bone marrow mesenchymal stem cells stimulate cardiac stem cell proliferation and differentiation. Circ. Res. 2010, 107, 913–922. [Google Scholar] [CrossRef] [PubMed]
- Ferreira-Martins, J.; Ogorek, B.; Cappetta, D.; Matsuda, A.; Signore, S.; D’Amario, D.; Kostyla, J.; Steadman, E.; Ide-Iwata, N.; Sanada, F.; et al. Cardiomyogenesis in the developing heart is regulated by c-kit-positive cardiac stem cells. Circ. Res. 2012, 110, 701–715. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.; Sussman, M.A. Rejuvenating the senescent heart. Curr. Opin. Cardiol. 2015, 30, 235–239. [Google Scholar] [CrossRef] [PubMed]
- Stamm, C.; Westphal, B.; Kleine, H.; Petzsch, M.; Kittner, C.; Klinge, H.; Schümichen, C.; Nienaber, C.; Freund, M.; Steinhoff, P.G. Autologous bone-marrow stem-cell transplantation for myocardial regeneration. Lancet 2003, 361, 45–46. [Google Scholar] [CrossRef]
- Yousef, M.; Schannwell, C.M.; Kostering, M.; Zeus, T.; Brehm, M.; Strauer, B.E. The BALANCE Study: Clinical benefit and long-term outcome after intracoronary autologous bone marrow cell transplantation in patients with acute myocardial infarction. J. Am. Coll. Cardiol. 2009, 53, 2262–2269. [Google Scholar] [CrossRef] [PubMed]
- Assmus, B.; Schachinger, V.; Teupe, C.; Britten, M.; Lehmann, R.; Dobert, N.; Grunwald, F.; Aicher, A.; Urbich, C.; Martin, H.; et al. Transplantation of progenitor cells and regeneration enhancement in acute myocardial infarction (TOPCARE-AMI). Circulation 2002, 106, 3009–3017. [Google Scholar] [CrossRef] [PubMed]
- Schachinger, V.; Assmus, B.; Britten, M.B.; Honold, J.; Lehmann, R.; Teupe, C.; Abolmaali, N.D.; Vogl, T.J.; Hofmann, W.K.; Martin, H.; et al. Transplantation of progenitor cells and regeneration enhancement in acute myocardial infarction: Final one-year results of the TOPCARE-AMI Trial. J. Am. Coll. Cardiol. 2004, 44, 1690–1699. [Google Scholar] [CrossRef] [PubMed]
- Schachinger, V.; Erbs, S.; Elsasser, A.; Haberbosch, W.; Hambrecht, R.; Holschermann, H.; Yu, J.; Corti, R.; Mathey, D.G.; Hamm, C.W.; et al. Improved clinical outcome after intracoronary administration of bone-marrow-derived progenitor cells in acute myocardial infarction: Final 1-year results of the REPAIR-AMI trial. Eur. Heart J. 2006, 27, 2775–2783. [Google Scholar] [CrossRef] [PubMed]
- Martin-Rendon, E.; Brunskill, S.J.; Hyde, C.J.; Stanworth, S.J.; Mathur, A.; Watt, S.M. Autologous bone marrow stem cells to treat acute myocardial infarction: A systematic review. Eur. Heart J. 2008, 29, 1807–1818. [Google Scholar] [CrossRef] [PubMed]
- Beeres, S.L.; Bax, J.J.; Kaandorp, T.A.; Zeppenfeld, K.; Lamb, H.J.; Dibbets-Schneider, P.; Stokkel, M.P.; Fibbe, W.E.; de Roos, A.; van der Wall, E.E.; et al. Usefulness of intramyocardial injection of autologous bone marrow-derived mononuclear cells in patients with severe angina pectoris and stress-induced myocardial ischemia. Am. J. Cardiol. 2006, 97, 1326–1331. [Google Scholar] [CrossRef] [PubMed]
- Beeres, S.L.; Lamb, H.J.; Roes, S.D.; Holman, E.R.; Kaandorp, T.A.; Fibbe, W.E.; de Roos, A.; van der Wall, E.E.; Schalij, M.J.; Bax, J.J.; et al. Effect of intramyocardial bone marrow cell injection on diastolic function in patients with chronic myocardial ischemia. J. Magn. Reson. Imaging 2008, 27, 992–997. [Google Scholar] [CrossRef] [PubMed]
- Tse, H.F.; Thambar, S.; Kwong, Y.L.; Rowlings, P.; Bellamy, G.; McCrohon, J.; Thomas, P.; Bastian, B.; Chan, J.K.; Lo, G.; et al. Prospective randomized trial of direct endomyocardial implantation of bone marrow cells for treatment of severe coronary artery diseases (PROTECT-CAD trial). Eur. Heart J. 2007, 28, 2998–3005. [Google Scholar] [CrossRef] [PubMed]
- Veltman, C.E.; Soliman, O.I.; Geleijnse, M.L.; Vletter, W.B.; Smits, P.C.; ten Cate, F.J.; Jordaens, L.J.; Balk, A.H.; Serruys, P.W.; Boersma, E.; et al. Four-year follow-up of treatment with intramyocardial skeletal myoblasts injection in patients with ischaemic cardiomyopathy. Eur. Heart J. 2008, 29, 1386–1396. [Google Scholar] [CrossRef] [PubMed]
- Hare, J.M.; Fishman, J.E.; Gerstenblith, G.; DiFede Velazquez, D.L.; Zambrano, J.P.; Suncion, V.Y.; Tracy, M.; Ghersin, E.; Johnston, P.V.; Brinker, J.A.; et al. Comparison of allogeneic vs autologous bone marrow-derived mesenchymal stem cells delivered by transendocardial injection in patients with ischemic cardiomyopathy: The POSEIDON randomized trial. JAMA 2012, 308, 2369–2379. [Google Scholar] [CrossRef] [PubMed]
- Hare, J.M.; Traverse, J.H.; Henry, T.D.; Dib, N.; Strumpf, R.K.; Schulman, S.P.; Gerstenblith, G.; DeMaria, A.N.; Denktas, A.E.; Gammon, R.S.; et al. A randomized, double-blind, placebo-controlled, dose-escalation study of intravenous adult human mesenchymal stem cells (prochymal) after acute myocardial infarction. J. Am. Coll. Cardiol. 2009, 54, 2277–2286. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Lee, S.H.; Youn, Y.J.; Ahn, M.S.; Kim, J.Y.; Yoo, B.S.; Yoon, J.; Kwon, W.; Hong, I.S.; Lee, K.; et al. A randomized, open-label, multicenter trial for the safety and efficacy of adult mesenchymal stem cells after acute myocardial infarction. J. Korean Med. Sci. 2014, 29, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Mushtaq, M.; DiFede, D.L.; Golpanian, S.; Khan, A.; Gomes, S.A.; Mendizabal, A.; Heldman, A.W.; Hare, J.M. Rationale and design of the Percutaneous Stem Cell Injection Delivery Effects on Neomyogenesis in Dilated Cardiomyopathy (the POSEIDON-DCM study): A phase I/II, randomized pilot study of the comparative safety and efficacy of transendocardial injection of autologous mesenchymal stem cell vs. allogeneic mesenchymal stem cells in patients with non-ischemic dilated cardiomyopathy. J. Cardiovasc. Transl. Res. 2014, 7, 769–780. [Google Scholar] [CrossRef] [PubMed]
- Bolli, R.; Chugh, A.R.; D’Amario, D.; Loughran, J.H.; Stoddard, M.F.; Ikram, S.; Beache, G.M.; Wagner, S.G.; Leri, A.; Hosoda, T.; et al. Cardiac stem cells in patients with ischaemic cardiomyopathy (SCIPIO): Initial results of a randomised phase 1 trial. Lancet 2011, 378, 1847–1857. [Google Scholar] [CrossRef]
- Chugh, A.R.; Beache, G.M.; Loughran, J.H.; Mewton, N.; Elmore, J.B.; Kajstura, J.; Pappas, P.; Tatooles, A.; Stoddard, M.F.; Lima, J.A.; et al. Administration of cardiac stem cells in patients with ischemic cardiomyopathy: The SCIPIO trial: Surgical aspects and interim analysis of myocardial function and viability by magnetic resonance. Circulation 2012, 126, S54–S64. [Google Scholar] [CrossRef] [PubMed]
- Makkar, R.R.; Smith, R.R.; Cheng, K.; Malliaras, K.; Thomson, L.E.; Berman, D.; Czer, L.S.; Marban, L.; Mendizabal, A.; Johnston, P.V.; et al. Intracoronary cardiosphere-derived cells for heart regeneration after myocardial infarction (CADUCEUS): A prospective, randomised phase 1 trial. Lancet 2012, 379, 895–904. [Google Scholar] [CrossRef]
- Malliaras, K.; Makkar, R.R.; Smith, R.R.; Cheng, K.; Wu, E.; Bonow, R.O.; Marban, L.; Mendizabal, A.; Cingolani, E.; Johnston, P.V.; et al. Intracoronary cardiosphere-derived cells after myocardial infarction: Evidence of therapeutic regeneration in the final 1-year results of the CADUCEUS trial (CArdiosphere-Derived aUtologous stem CElls to reverse ventricUlar dySfunction). J. Am. Coll. Cardiol. 2014, 63, 110–122. [Google Scholar] [CrossRef] [PubMed]
- Quijada, P.; Sussman, M.A. Making it stick: Chasing the optimal stem cells for cardiac regeneration. Expert Rev. Cardiovasc. Ther. 2014, 12, 1275–1288. [Google Scholar] [CrossRef] [PubMed]
- Dixit, P.; Katare, R. Challenges in identifying the best source of stem cells for cardiac regeneration therapy. Stem Cell Res. Ther. 2015, 6, 26. [Google Scholar] [CrossRef] [PubMed]
- Leite, C.F.; Almeida, T.R.; Lopes, C.S.; Dias da Silva, V.J. Multipotent stem cells of the heart-do they have therapeutic promise? Front. Physiol. 2015, 6, 123. [Google Scholar] [CrossRef] [PubMed]
- Byrne, N.; Velasco Forte, M.; Tandon, A.; Valverde, I.; Hussain, T. A systematic review of image segmentation methodology, used in the additive manufacture of patient-specific 3D printed models of the cardiovascular system. JRSM Cardiovasc. Dis. 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Kurup, H.K.; Samuel, B.P.; Vettukattil, J.J. Hybrid 3D printing: A game-changer in personalized cardiac medicine? Expert Rev. Cardiovasc. Ther. 2015, 13, 1281–1284. [Google Scholar] [CrossRef] [PubMed]
- Schmauss, D.; Haeberle, S.; Hagl, C.; Sodian, R. Three-dimensional printing in cardiac surgery and interventional cardiology: A single-centre experience. Eur. J. Cardiothorac. Surg. 2015, 47, 1044–1052. [Google Scholar] [CrossRef] [PubMed]
- Bartel, T.; Rivard, A.; Jimenez, A.; Mestres, C.A.; Muller, S. Medical three-dimensional printing opens up new opportunities in cardiology and cardiac surgery. Eur. Heart J. 2017. [Google Scholar] [CrossRef] [PubMed]
- Valverde, I. Three-dimensional printed cardiac models: Applications in the field of medical education, cardiovascular surgery, and structural heart interventions. Rev. Esp. Cardiol. 2017, 70, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Vukicevic, M.; Mosadegh, B.; Min, J.K.; Little, S.H. Cardiac 3D printing and its future directions. JACC Cardiovasc. Imaging 2017, 10, 171–184. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liu, J.; Zheng, X.; Rong, X.; Zheng, X.; Peng, H.; Silber-Li, Z.; Li, M.; Liu, L. Three-dimensional virtual surgery models for percutaneous coronary intervention (PCI) optimization strategies. Sci. Rep. 2015, 5, 10945. [Google Scholar] [CrossRef] [PubMed]
- Javan, R.; Herrin, D.; Tangestanipoor, A. Understanding spatially complex segmental and branch anatomy using 3D printing: Liver, lung, prostate, coronary arteries, and circle of willis. Acad. Radiol. 2016, 23, 1183–1189. [Google Scholar] [CrossRef] [PubMed]
- Kolli, K.K.; Min, J.K.; Ha, S.; Soohoo, H.; Xiong, G. Effect of varying hemodynamic and vascular conditions on fractional flow reserve: An in vitro study. J. Am. Heart Assoc. 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Gaetani, R.; Doevendans, P.A.; Metz, C.H.; Alblas, J.; Messina, E.; Giacomello, A.; Sluijter, J.P. Cardiac tissue engineering using tissue printing technology and human cardiac progenitor cells. Biomaterials 2012, 33, 1782–1790. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Kupfer, M.E.; Jung, J.P.; Yang, L.; Zhang, P.; Da Sie, Y.; Tran, Q.; Ajeti, V.; Freeman, B.T.; Fast, V.G.; et al. Myocardial tissue engineering with cells derived from human-induced pluripotent stem cells and a native-like, high-resolution, 3-dimensionally printed scaffold. Circ. Res. 2017, 120, 1318–1325. [Google Scholar] [CrossRef] [PubMed]
- McMahon, S.R.; Brummel-Ziedins, K.; Schneider, D.J. Novel oral anticoagulants in the management of coronary artery disease. Coron. Artery Dis. 2016, 27, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Oldgren, J.; Budaj, A.; Granger, C.B.; Khder, Y.; Roberts, J.; Siegbahn, A.; Tijssen, J.G.; van de Werf, F.; Wallentin, L.; Investigators, R.-D. Dabigatran vs. placebo in patients with acute coronary syndromes on dual antiplatelet therapy: A randomized, double-blind, phase II trial. Eur. Heart J. 2011, 32, 2781–2789. [Google Scholar] [CrossRef] [PubMed]
- Abifadel, M.; Varret, M.; Rabes, J.P.; Allard, D.; Ouguerram, K.; Devillers, M.; Cruaud, C.; Benjannet, S.; Wickham, L.; Erlich, D.; et al. Mutations in PCSK9 cause autosomal dominant hypercholesterolemia. Nat. Genet. 2003, 34, 154–156. [Google Scholar] [CrossRef] [PubMed]
- Della Pepa, G.; Bozzetto, L.; Annuzzi, G.; Rivellese, A.A. Alirocumab for the treatment of hypercholesterolaemia. Expert Rev. Clin. Pharmacol. 2017, 10, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Graham, M.J.; Lemonidis, K.M.; Whipple, C.P.; Subramaniam, A.; Monia, B.P.; Crooke, S.T.; Crooke, R.M. Antisense inhibition of proprotein convertase subtilisin/kexin type 9 reduces serum LDL in hyperlipidemic mice. J. Lipid Res. 2007, 48, 763–767. [Google Scholar] [CrossRef] [PubMed]
- Stein, E.A.; Mellis, S.; Yancopoulos, G.D.; Stahl, N.; Logan, D.; Smith, W.B.; Lisbon, E.; Gutierrez, M.; Webb, C.; Wu, R.; et al. Effect of a monoclonal antibody to PCSK9 on LDL cholesterol. N. Engl. J. Med. 2012, 366, 1108–1118. [Google Scholar] [CrossRef] [PubMed]
- Stein, E.A.; Gipe, D.; Bergeron, J.; Gaudet, D.; Weiss, R.; Dufour, R.; Wu, R.; Pordy, R. Effect of a monoclonal antibody to PCSK9, REGN727/SAR236553, to reduce low-density lipoprotein cholesterol in patients with heterozygous familial hypercholesterolaemia on stable statin dose with or without ezetimibe therapy: A phase 2 randomised controlled trial. Lancet 2012, 380, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Roth, E.M.; McKenney, J.M.; Hanotin, C.; Asset, G.; Stein, E.A. Atorvastatin with or without an antibody to PCSK9 in primary hypercholesterolemia. N. Engl. J. Med. 2012, 367, 1891–1900. [Google Scholar] [CrossRef] [PubMed]
- Bays, H.; Gaudet, D.; Weiss, R.; Ruiz, J.L.; Watts, G.F.; Gouni-Berthold, I.; Robinson, J.; Zhao, J.; Hanotin, C.; Donahue, S. Alirocumab as add-on to atorvastatin versus other lipid treatment strategies: ODYSSEY OPTIONS I randomized trial. J. Clin. Endocrinol. Metab. 2015, 100, 3140–3148. [Google Scholar] [CrossRef] [PubMed]
- Cannon, C.P.; Cariou, B.; Blom, D.; McKenney, J.M.; Lorenzato, C.; Pordy, R.; Chaudhari, U.; Colhoun, H.M.; Investigators, O.C.I. Efficacy and safety of alirocumab in high cardiovascular risk patients with inadequately controlled hypercholesterolaemia on maximally tolerated doses of statins: The ODYSSEY COMBO II randomized controlled trial. Eur. Heart J. 2015, 36, 1186–1194. [Google Scholar] [CrossRef] [PubMed]
- Farnier, M.; Jones, P.; Severance, R.; Averna, M.; Steinhagen-Thiessen, E.; Colhoun, H.M.; Du, Y.; Hanotin, C.; Donahue, S. Efficacy and safety of adding alirocumab to rosuvastatin versus adding ezetimibe or doubling the rosuvastatin dose in high cardiovascular-risk patients: The ODYSSEY OPTIONS II randomized trial. Atherosclerosis 2016, 244, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Ginsberg, H.N.; Rader, D.J.; Raal, F.J.; Guyton, J.R.; Baccara-Dinet, M.T.; Lorenzato, C.; Pordy, R.; Stroes, E. Efficacy and safety of alirocumab in patients with heterozygous familial hypercholesterolemia and LDL-C of 160 mg/dL or higher. Cardiovasc. Drugs Ther. 2016, 30, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Kastelein, J.J.; Ginsberg, H.N.; Langslet, G.; Hovingh, G.K.; Ceska, R.; Dufour, R.; Blom, D.; Civeira, F.; Krempf, M.; Lorenzato, C.; et al. ODYSSEY FH I and FH II: 78 week results with alirocumab treatment in 735 patients with heterozygous familial hypercholesterolaemia. Eur. Heart J. 2015, 36, 2996–3003. [Google Scholar] [CrossRef] [PubMed]
- Kereiakes, D.J.; Robinson, J.G.; Cannon, C.P.; Lorenzato, C.; Pordy, R.; Chaudhari, U.; Colhoun, H.M. Efficacy and safety of the proprotein convertase subtilisin/kexin type 9 inhibitor alirocumab among high cardiovascular risk patients on maximally tolerated statin therapy: The ODYSSEY COMBO I study. Am. Heart J. 2015, 169, 906–915. [Google Scholar] [CrossRef] [PubMed]
- Roth, E.M.; Moriarty, P.M.; Bergeron, J.; Langslet, G.; Manvelian, G.; Zhao, J.; Baccara-Dinet, M.T.; Rader, D.J.; ODYSSEY CHOICE I investigators. A phase III randomized trial evaluating alirocumab 300 mg every 4 weeks as monotherapy or add-on to statin: ODYSSEY CHOICE I. Atherosclerosis 2016, 254, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Moriarty, P.M.; Thompson, P.D.; Cannon, C.P.; Guyton, J.R.; Bergeron, J.; Zieve, F.J.; Bruckert, E.; Jacobson, T.A.; Kopecky, S.L.; Baccara-Dinet, M.T.; et al. Efficacy and safety of alirocumab vs ezetimibe in statin-intolerant patients, with a statin rechallenge arm: The ODYSSEY ALTERNATIVE randomized trial. J. Clin. Lipidol. 2015, 9, 758–769. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.G.; Farnier, M.; Krempf, M.; Bergeron, J.; Luc, G.; Averna, M.; Stroes, E.S.; Langslet, G.; Raal, F.J.; El Shahawy, M.; et al. Efficacy and safety of alirocumab in reducing lipids and cardiovascular events. N. Engl. J. Med. 2015, 372, 1489–1499. [Google Scholar] [CrossRef] [PubMed]
- Roth, E.M.; Taskinen, M.R.; Ginsberg, H.N.; Kastelein, J.J.; Colhoun, H.M.; Robinson, J.G.; Merlet, L.; Pordy, R.; Baccara-Dinet, M.T. Monotherapy with the PCSK9 inhibitor alirocumab versus ezetimibe in patients with hypercholesterolemia: Results of a 24 week, double-blind, randomized Phase 3 trial. Int. J. Cardiol. 2014, 176, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Stroes, E.; Guyton, J.R.; Lepor, N.; Civeira, F.; Gaudet, D.; Watts, G.F.; Baccara-Dinet, M.T.; Lecorps, G.; Manvelian, G.; Farnier, M.; et al. Efficacy and safety of alirocumab 150 mg every 4 weeks in patients with hypercholesterolemia not on statin therapy: The ODYSSEY CHOICE II study. J. Am. Heart Assoc. 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Teramoto, T.; Kobayashi, M.; Tasaki, H.; Yagyu, H.; Higashikata, T.; Takagi, Y.; Uno, K.; Baccara-Dinet, M.T.; Nohara, A. Efficacy and safety of alirocumab in japanese patients with heterozygous familial hypercholesterolemia or at high cardiovascular risk with hypercholesterolemia not adequately controlled with statins-ODYSSEY JAPAN randomized controlled trial. Circ. J. 2016, 80, 1980–1987. [Google Scholar] [CrossRef] [PubMed]
- Ray, K.K.; Ginsberg, H.N.; Davidson, M.H.; Pordy, R.; Bessac, L.; Minini, P.; Eckel, R.H.; Cannon, C.P. Reductions in atherogenic lipids and major cardiovascular events: A pooled analysis of 10 ODYSSEY trials comparing alirocumab with control. Circulation 2016, 134, 1931–1943. [Google Scholar] [CrossRef] [PubMed]
- Sabatine, M.S.; Giugliano, R.P.; Keech, A.C.; Honarpour, N.; Wiviott, S.D.; Murphy, S.A.; Kuder, J.F.; Wang, H.; Liu, T.; Wasserman, S.M.; et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N. Engl. J. Med. 2017, 376, 1713–1722. [Google Scholar] [CrossRef] [PubMed]
- Pham, A.Q.; Patel, Y.; Gallagher, B. LCZ696 (angiotensin-neprilysin inhibition): The new kid on the heart failure block? J. Pharm. Pract. 2015, 28, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Seki, T.; Goto, K.; Kansui, Y.; Ohtsubo, T.; Matsumura, K.; Kitazono, T. Angiotensin II receptor-neprilysin inhibitor sacubitril/valsartan improves endothelial dysfunction in spontaneously hypertensive rats. J. Am. Heart Assoc. 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Vaduganathan, M.; Desai, A.S. Angiotensin-neprilysin inhibition as a paradigm for All? Curr. Cardiol. Rep. 2016, 18, 115. [Google Scholar] [CrossRef] [PubMed]
- McMurray, J.J.; Packer, M.; Desai, A.S.; Gong, J.; Lefkowitz, M.P.; Rizkala, A.R.; Rouleau, J.L.; Shi, V.C.; Solomon, S.D.; Swedberg, K.; et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N. Engl. J. Med. 2014, 371, 993–1004. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.S.; McMurray, J.J.; Packer, M.; Swedberg, K.; Rouleau, J.L.; Chen, F.; Gong, J.; Rizkala, A.R.; Brahimi, A.; Claggett, B.; et al. Effect of the angiotensin-receptor-neprilysin inhibitor LCZ696 compared with enalapril on mode of death in heart failure patients. Eur. Heart J. 2015, 36, 1990–1997. [Google Scholar] [CrossRef] [PubMed]
- Jhund, P.S.; Fu, M.; Bayram, E.; Chen, C.H.; Negrusz-Kawecka, M.; Rosenthal, A.; Desai, A.S.; Lefkowitz, M.P.; Rizkala, A.R.; Rouleau, J.L.; et al. Efficacy and safety of LCZ696 (sacubitril-valsartan) according to age: Insights from PARADIGM-HF. Eur. Heart J. 2015, 36, 2576–2584. [Google Scholar] [CrossRef] [PubMed]
- Solomon, S.D.; Claggett, B.; Desai, A.S.; Packer, M.; Zile, M.; Swedberg, K.; Rouleau, J.L.; Shi, V.C.; Starling, R.C.; Kozan, O.; et al. Influence of ejection fraction on outcomes and efficacy of sacubitril/valsartan (LCZ696) in heart failure with reduced ejection fraction: The prospective comparison of ARNI with ACEI to determine impact on global mortality and morbidity in heart failure (PARADIGM-HF) trial. Circ. Heart Fail. 2016, 9, e002744. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, S.L.; Preiss, D.; Jhund, P.S.; Squire, I.; Cardoso, J.S.; Merkely, B.; Martinez, F.; Starling, R.C.; Desai, A.S.; Lefkowitz, M.P.; et al. Risk related to pre-diabetes mellitus and diabetes mellitus in heart failure with reduced ejection fraction: Insights from prospective comparison of ARNI with ACEI to determine impact on global mortality and morbidity in heart failure trial. Circ. Heart Fail. 2016, 9. [Google Scholar] [CrossRef] [PubMed]
| S. No. | Author’s Name | Results | Additional Comments |
|---|---|---|---|
| 1. | Dogan et al. [17] | They reported a patency rate of 100%. | TECAB was performed on hearts arrested intraoperatively. |
| 2. | Kappert et al. [18] | Reduced duration of surgery (down from 280 to 186 minutes); All of them had normal wound healing | TECAB was performed on a beating heart. 3 patients had to undergo re-exploration due to bleeding. |
| 3. | Mohr et al. [19] | Successful procedure in 22 patients (5 of them had to be converted to manual procedure); At discharge, patency was 100% and 95.4% at 3 months; In the TECAB group, success rate was 50%. | TECAB was performed on beating (n = 8) and arrested (n = 27) heart. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kandaswamy, E.; Zuo, L. Recent Advances in Treatment of Coronary Artery Disease: Role of Science and Technology. Int. J. Mol. Sci. 2018, 19, 424. https://doi.org/10.3390/ijms19020424
Kandaswamy E, Zuo L. Recent Advances in Treatment of Coronary Artery Disease: Role of Science and Technology. International Journal of Molecular Sciences. 2018; 19(2):424. https://doi.org/10.3390/ijms19020424
Chicago/Turabian StyleKandaswamy, Eswar, and Li Zuo. 2018. "Recent Advances in Treatment of Coronary Artery Disease: Role of Science and Technology" International Journal of Molecular Sciences 19, no. 2: 424. https://doi.org/10.3390/ijms19020424
APA StyleKandaswamy, E., & Zuo, L. (2018). Recent Advances in Treatment of Coronary Artery Disease: Role of Science and Technology. International Journal of Molecular Sciences, 19(2), 424. https://doi.org/10.3390/ijms19020424






