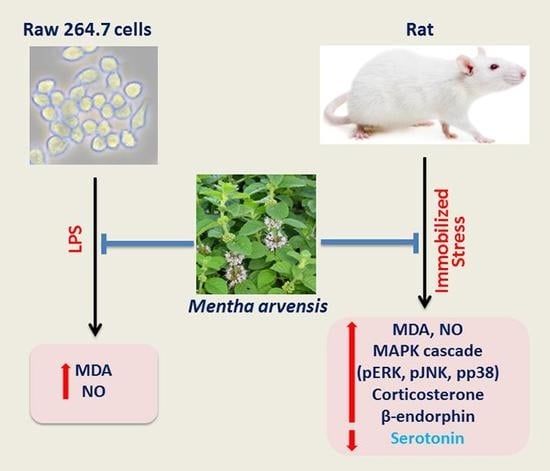
The Anti-Stress Effect of Mentha arvensis in Immobilized Rats
Abstract
:1. Introduction
2. Results
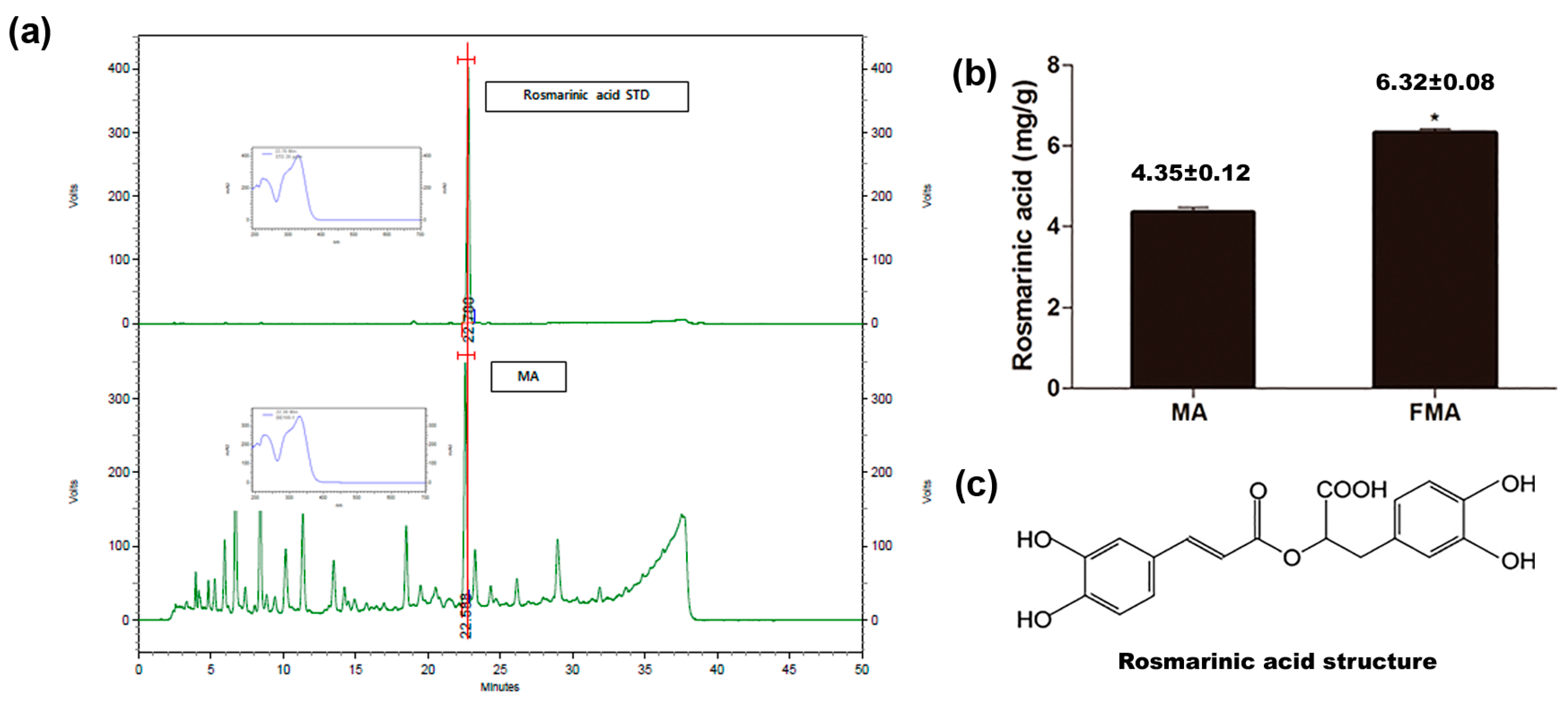
2.1. Evaluation of Active Compound of MA and Content in MA and FMA
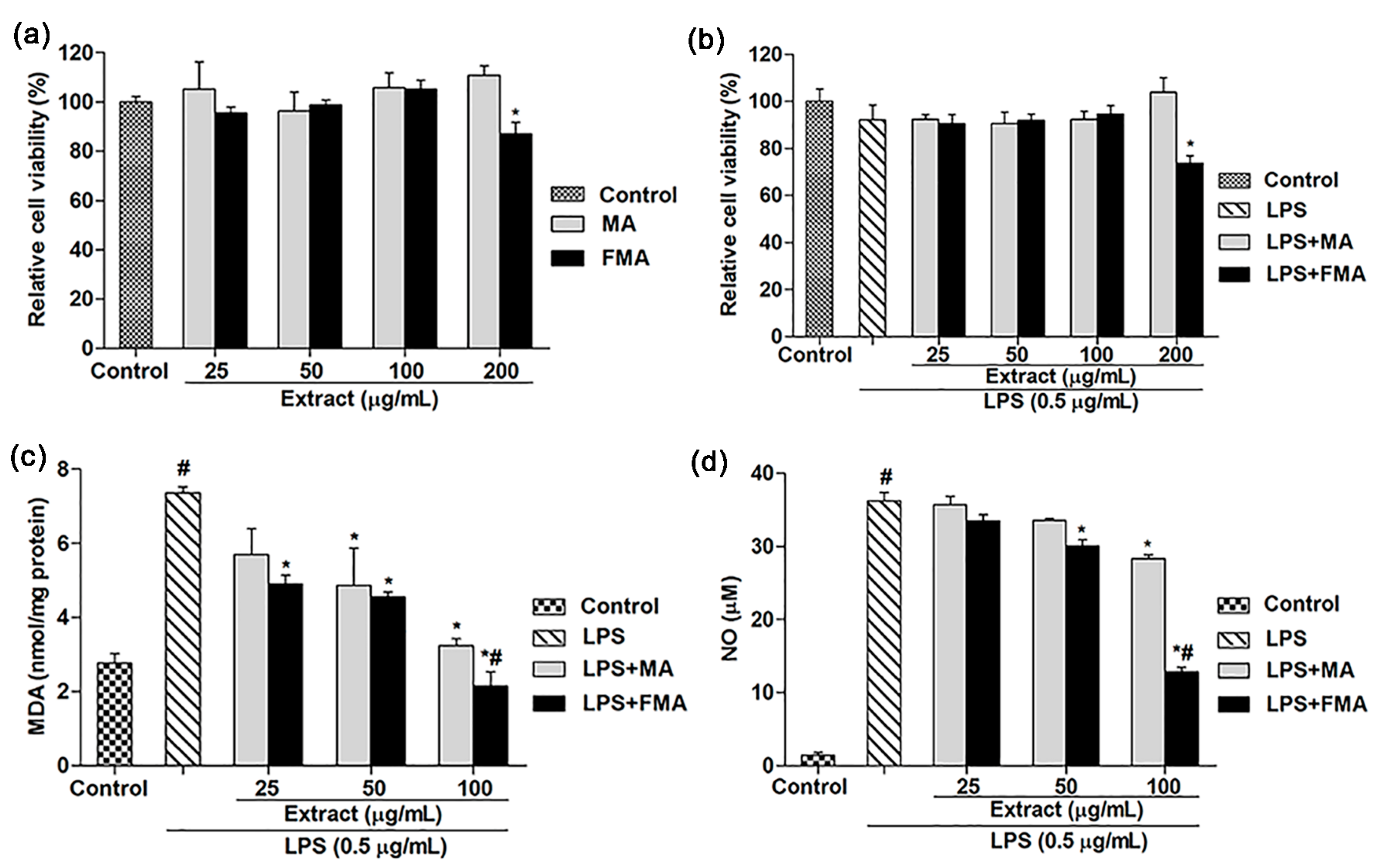
2.2. Effects of MA and FMA Extract on Viability of RAW 264.7 Cells
2.3. Effect of MA and FMA on MDA and NO Release in RAW 264.7 Cells
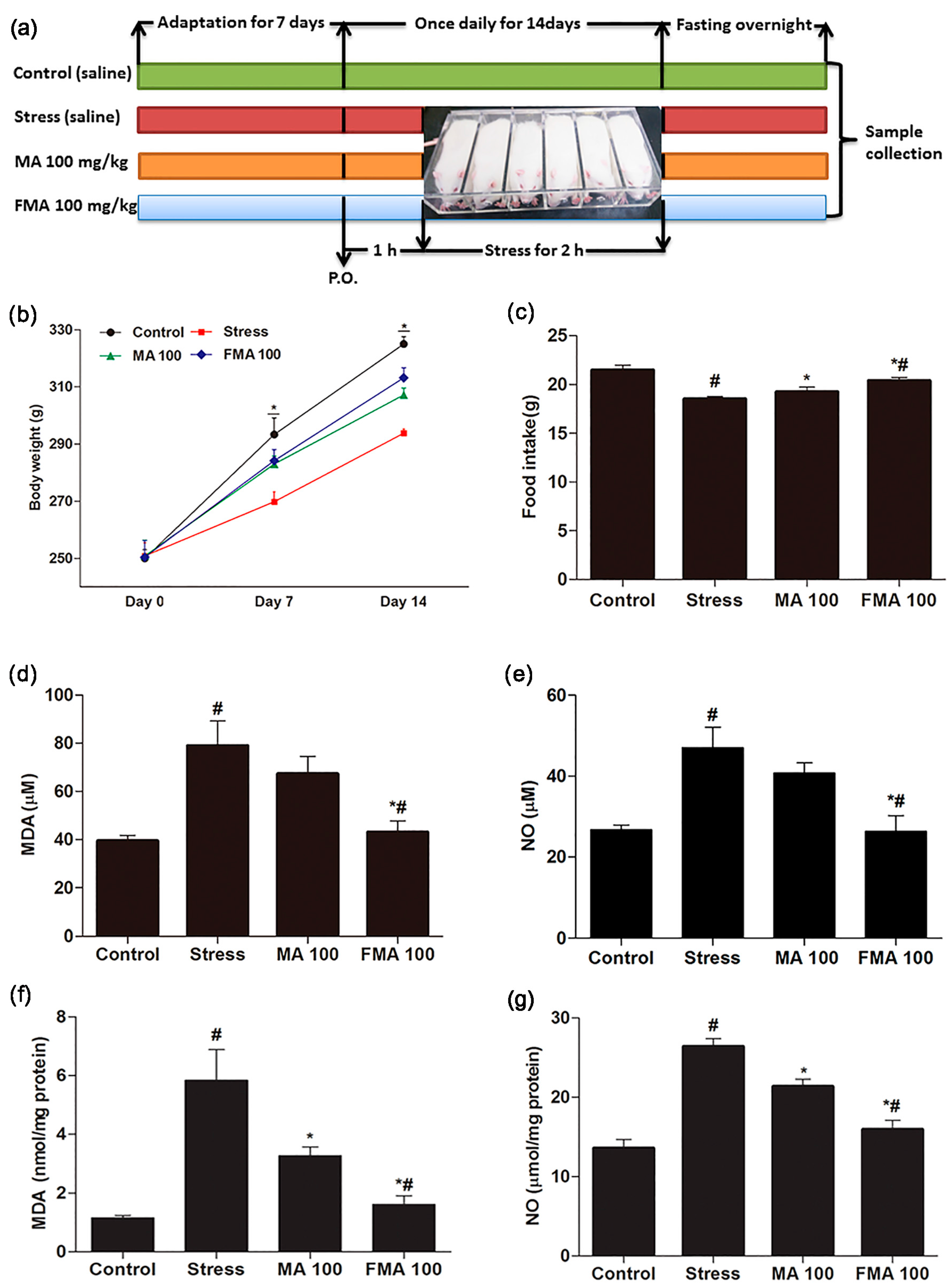
2.4. Effect of MA and FMA on Body Weight and Food Intake
2.5. Effect of MA and FMA on MDA and NO Production in Immobilized Rats Serum and Hippocampus
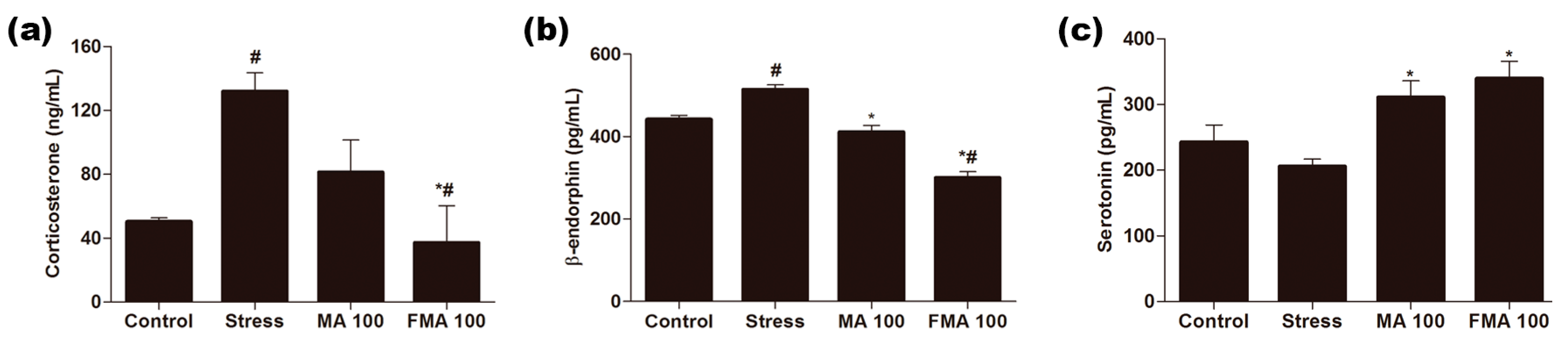
2.6. Effect of MA and FMA on Serum Corticosterone, β-Endorphin, and Serotonin in Immobilized Rats
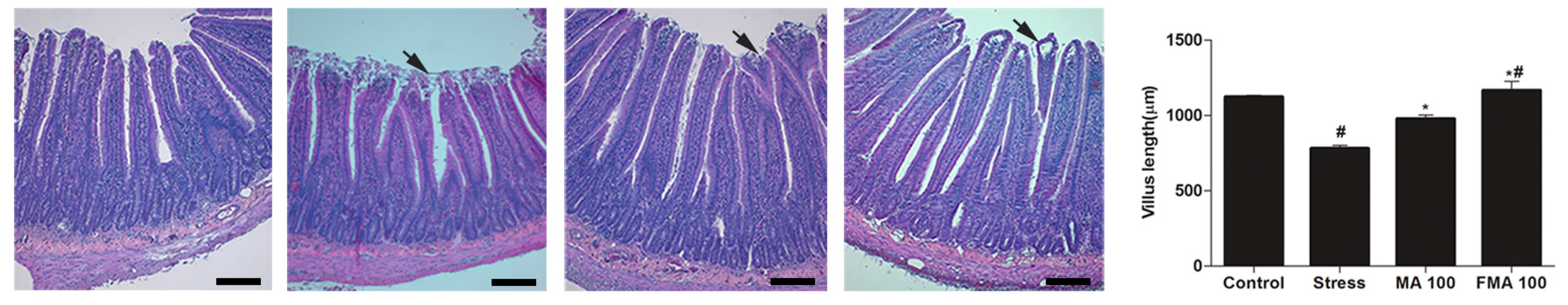
2.7. Effect of MA and FMA on Histology of the Duodenum
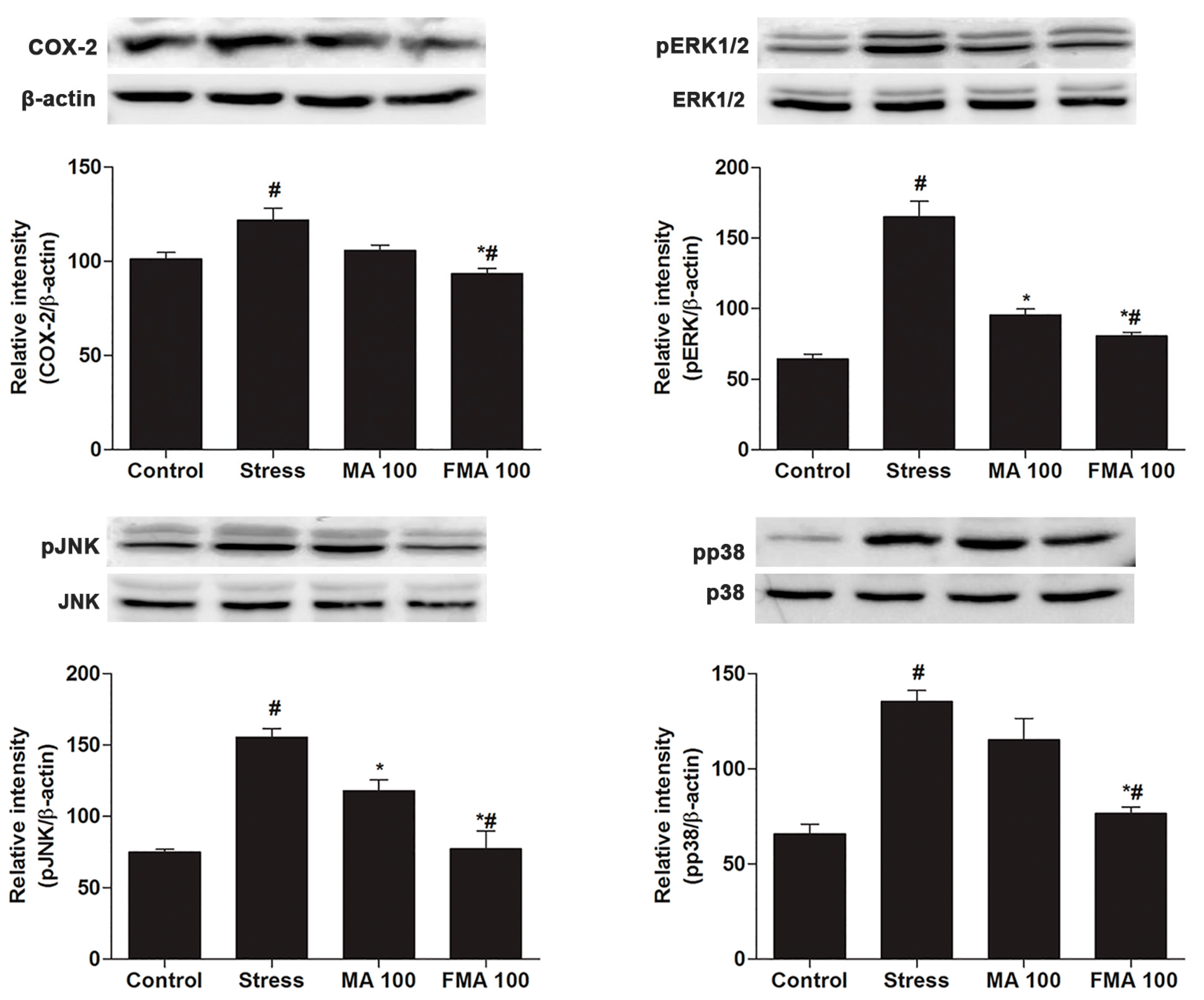
2.8. Effect of MA and FMA on MAPK/COX-2 Signaling Pathways
3. Discussion
4. Materials and Methods
4.1. Analytical Reagents and Chemicals
4.2. Preparation of Mentha Arvensis var Extract
4.3. HPLC Analysis and Measurement of the Content of the Rosmarinic Acid in MA and FMA Extract
4.4. Cell Culture and Assessment of Cell Viability
4.5. Measurement of MDA and NO Content in RAW 264.7 Cells
4.6. Animal Management and Experimental Design
4.7. Measurement of Body Weight and Food Intake
4.8. Measurement of MDA and NO Content in Rat Serum and Hippocampus
4.9. ELISA Assay for Corticosterone, β-endorphin, and Serotonin
4.10. Histological Analysis
4.11. Western Blot Analysis
4.12. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sheikh, N.; Ahmad, A.; Siripurapu, K.B.; Kuchibhotla, V.K.; Singh, S.; Palit, G. Effect of Bacopa monniera on stress induced changes in plasma corticosterone and brain monoamines in rats. J. Ethnopharmacol. 2007, 111, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Ramadan, K.S.; Alshamrani, S.A. Effects of Salvadora persica Extract on the Hematological and Biochemical Alterations against Immobilization-Induced Rats. Scientifica 2015, 2015, 253195. [Google Scholar] [CrossRef] [PubMed]
- Imperato, A.; Angelucci, L.; Casolini, P.; Zocchi, A.; Puglisi-Allegra, S. Repeated stressful experiences differently affect limbic dopamine release during and following stress. Brain Res. 1992, 577, 194–199. [Google Scholar] [CrossRef]
- Grundt, A.; Grundt, C.; Gorbey, S.; Thomas, M.A.; Lemmer, B. Strain-dependent differences of restraint stress-induced hypertension in WKY and SHR. Physiol. Behav. 2009, 97, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Shukla, R.K.; Gupta, R.; Srivastava, P.; Dhuriya, Y.K.; Singh, A.; Chandravanshi, L.P.; Kumar, A.; Siddiqui, M.H.; Parmar, D.; Pant, A.B.; et al. Brain cholinergic alterations in rats subjected to repeated immobilization or forced swim stress on lambda-cyhalothrin exposure. Neurochem. Int. 2016, 93, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Patki, G.; Solanki, N.; Atrooz, F.; Allam, F.; Salim, S. Depression, anxiety-like behavior and memory impairment are associated with increased oxidative stress and inflammation in a rat model of social stress. Brain Res. 2013, 1539, 73–86. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, K.; Dhabhar, F.S. Stress-induced enhancement of leukocyte trafficking into sites of surgery or immune activation. Proc. Natl. Acad. Sci. USA 2005, 102, 5808–5813. [Google Scholar] [CrossRef] [PubMed]
- Bailey, M.T.; Dowd, S.E.; Galley, J.D.; Hufnagle, A.R.; Allen, R.G.; Lyte, M. Exposure to a social stressor alters the structure of the intestinal microbiota: Implications for stressor-induced immunomodulation. Brain Behav. Immun. 2011, 25, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Free radicals, antioxidants, and human disease: Curiosity, cause, or consequence? Lancet 1994, 344, 721–724. [Google Scholar] [CrossRef]
- Madrigal, J.L.; Moro, M.A.; Lizasoain, I.; Lorenzo, P.; Castrillo, A.; Bosca, L.; Leza, J.C. Inducible nitric oxide synthase expression in brain cortex after acute restraint stress is regulated by nuclear factor κB-mediated mechanisms. J. Neurochem. 2001, 76, 532–538. [Google Scholar] [CrossRef] [PubMed]
- Sasaguri, K.; Kikuchi, M.; Hori, N.; Yuyama, N.; Onozuka, M.; Sato, S. Suppression of stress immobilization-induced phosphorylation of ERK 1/2 by biting in the rat hypothalamic paraventricular nucleus. Neurosci. Lett. 2005, 383, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Hebert, M.A.; Serova, L.I.; Sabban, E.L. Single and repeated immobilization stress differentially trigger induction and phosphorylation of several transcription factors and mitogen-activated protein kinases in the rat locus coeruleus. J. Neurochem. 2005, 95, 484–498. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Rasheed, N.; Chand, K.; Maurya, R.; Banu, N.; Palit, G. Restraint stress-induced central monoaminergic & oxidative changes in rats & their prevention by novel Ocimum sanctum compounds. Indian J. Med. Res. 2012, 135, 548–554. [Google Scholar] [PubMed]
- Verma, S.M.; Arora, H.; Dubey, R. Anti-inflammatory and sedative—Hypnotic activity of the methanolic extract of the leaves of mentha arvensis. Anc. Sci. Life 2003, 23, 95–99. [Google Scholar] [PubMed]
- Londonkar, R.L.; Poddar, P.V. Studies on activity of various extracts of Mentha arvensis Linn against drug induced gastric ulcer in mammals. World J. Gastrointest. Oncol. 2009, 1, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.T.; Kim, T.I.; Kim, Y.H.; Kang, J.S.; Min, B.S.; Son, J.K.; Woo, M.H. A comparative study of Mentha arvensis L. and Mentha haplocalyx Briq. by HPLC. Nat. Prod. Res. 2018, 32, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Salin, O.; Tormakangas, L.; Leinonen, M.; Saario, E.; Hagstrom, M.; Ketola, R.A.; Saikku, P.; Vuorela, H.; Vuorela, P.M. Corn mint (Mentha arvensis) extract diminishes acute Chlamydia pneumoniae infection in vitro and in vivo. J. Agric. Food Chem. 2011, 59, 12836–12842. [Google Scholar] [CrossRef] [PubMed]
- Adomako-Bonsu, A.G.; Chan, S.L.; Pratten, M.; Fry, J.R. Antioxidant activity of rosmarinic acid and its principal metabolites in chemical and cellular systems: Importance of physico-chemical characteristics. Toxicology 2017, 40, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Rocha, J.; Eduardo-Figueira, M.; Barateiro, A.; Fernandes, A.; Brites, D.; Bronze, R.; Duarte, C.M.; Serra, A.T.; Pinto, R.; Freitas, M.; et al. Anti-inflammatory effect of rosmarinic acid and an extract of Rosmarinus officinalis in rat models of local and systemic inflammation. Basic Clin. Pharmacol. Toxicol. 2015, 116, 398–413. [Google Scholar] [CrossRef] [PubMed]
- Park, M.J.; Bae, Y.S. Fermented Acanthopanax koreanum Root Extract Reduces UVB- and H2O2-Induced Senescence in Human Skin Fibroblast Cells. J. Microbiol. Biotechnol. 2016, 26, 1224–1233. [Google Scholar] [CrossRef] [PubMed]
- Rolle, R.; Satin, M. Basic requirements for the transfer of fermentation technologies to developing countries. Int. J. Food Microbiol. 2002, 75, 181–187. [Google Scholar] [CrossRef]
- Hong, K.J.; Lee, C.H.; Kim, S.W. Aspergillus oryzae GB-107 fermentation improves nutritional quality of food soybeans and feed soybean meals. J. Med. Food 2004, 7, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Oloyede, O.O.; James, S.; Ocheme, O.B.; Chinma, C.E.; Akpa, V.E. Effects of fermentation time on the functional and pasting properties of defatted Moringa oleifera seed flour. Food Sci. Nutr. 2016, 4, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligne, B.; Ganzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health benefits of fermented foods: Microbiota and beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Boye, J.; Wijesinha-Bettoni, R.; Burlingame, B. Protein quality evaluation twenty years after the introduction of the protein digestibility corrected amino acid score method. Br. J. Nutr. 2012, 108 (Suppl. S2), S183–S211. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [PubMed]
- Divyashri, G.; Krishna, G.; Muralidhara; Prapulla, S.G. Probiotic attributes, antioxidant, anti-inflammatory and neuromodulatory effects of Enterococcus faecium CFR 3003: In vitro and in vivo evidence. J. Med. Microbiol. 2015, 64, 1527–1540. [Google Scholar] [CrossRef] [PubMed]
- Polak-Berecka, M.; Wasko, A.; Szwajgier, D.; Chomaz, A. Bifidogenic and antioxidant activity of exopolysaccharides produced by Lactobacillus rhamnosus E/N cultivated on different carbon sources. Pol. J. Microbiol. 2013, 62, 181–188. [Google Scholar] [PubMed]
- Xing, J.; Wang, G.; Zhang, Q.; Liu, X.; Gu, Z.; Zhang, H.; Chen, Y.Q.; Chen, W. Determining antioxidant activities of lactobacilli cell-free supernatants by cellular antioxidant assay: A comparison with traditional methods. PLoS ONE 2015, 10, e0119058. [Google Scholar] [CrossRef] [PubMed]
- Jaggi, A.S.; Bhatia, N.; Kumar, N.; Singh, N.; Anand, P.; Dhawan, R. A review on animal models for screening potential anti-stress agents. Neurol. Sci. 2011, 32, 993–1005. [Google Scholar] [CrossRef] [PubMed]
- Kiecolt-Glaser, J.K.; Preacher, K.J.; MacCallum, R.C.; Atkinson, C.; Malarkey, W.B.; Glaser, R. Chronic stress and age-related increases in the proinflammatory cytokine IL-6. Proc. Natl. Acad. Sci. USA 2003, 100, 9090–9095. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.F.; Karioti, A.; Rohr, D.; Bilia, A.R.; Efferth, T. Production of rosmarinic acid and salvianolic acid B from callus culture of Salvia miltiorrhiza with cytotoxicity towards acute lymphoblastic leukemia cells. Food Chem. 2016, 201, 292–297. [Google Scholar] [CrossRef] [PubMed]
- Corral-Lugo, A.; Daddaoua, A.; Ortega, A.; Espinosa-Urgel, M.; Krell, T. Rosmarinic acid is a homoserine lactone mimic produced by plants that activates a bacterial quorum-sensing regulator. Sci. Signal. 2016, 9. [Google Scholar] [CrossRef] [PubMed]
- Fiore, R., 2nd; Miller, R.; Coffman, S.M. Mycobacterium mucogenicum infection following a cosmetic procedure with poly-l-lactic acid. J. Drugs Dermatol. 2013, 12, 353–357. [Google Scholar] [PubMed]
- Cheung, S.; Tai, J. Anti-proliferative and antioxidant properties of rosemary Rosmarinus officinalis. Oncol. Rep. 2007, 17, 1525–1531. [Google Scholar] [CrossRef] [PubMed]
- Zafir, A.; Banu, N. Induction of oxidative stress by restraint stress and corticosterone treatments in rats. Indian J. Biochem. Biophys. 2009, 46, 53–58. [Google Scholar] [PubMed]
- Lee, C.Y.; Cheng, H.M.; Sim, S.M. Mulberry leaves protect rat tissues from immobilization stress-induced inflammation. BioFactors 2007, 31, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Marazza, J.A.; Nazareno, M.A.; de Giori, G.S.; Garro, M.S. Enhancement of the antioxidant capacity of soymilk by fermentation with Lactobacillus rhamnosus. J. Funct. Foods 2012, 4, 594–601. [Google Scholar] [CrossRef]
- Mousavi, Z.E.; Mousavi, S.M.; Razavi, S.H.; Hadinejad, M.; Emam-Djomeh, Z.; Mirzapour, M. Effect of Fermentation of Pomegranate Juice by Lactobacillus plantarum and Lactobacillus acidophilus on the Antioxidant Activity and Metabolism of Sugars, Organic Acids and Phenolic Compounds. Food Biotechnol. 2013, 27, 1–13. [Google Scholar] [CrossRef]
- Tamang, J.P.; Shin, D.H.; Jung, S.J.; Chae, S.W. Functional Properties of Microorganisms in Fermented Foods. Front. Microbiol. 2016, 7, 578. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, B.; Anandan, R.; Lakshmanan, P.T. Studies on the protective effects of betaine against oxidative damage during experimentally induced restraint stress in Wistar albino rats. Cell Stress Chaperones 2011, 16, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Marti, O.; Marti, J.; Armario, A. Effects of chronic stress on food intake in rats: Influence of stressor intensity and duration of daily exposure. Physiol. Behav. 1994, 55, 747–753. [Google Scholar] [CrossRef]
- Samarghandian, S.; Farkhondeh, T.; Samini, F.; Borji, A. Protective Effects of Carvacrol against Oxidative Stress Induced by Chronic Stress in Rat’s Brain, Liver, and Kidney. Biochem. Res. Int. 2016, 2016, 2645237. [Google Scholar] [CrossRef] [PubMed]
- Shakhar, G.; Blumenfeld, B. Glucocorticoid involvement in suppression of NK activity following surgery in rats. J. Neuroimmunol. 2003, 138, 83–91. [Google Scholar] [CrossRef]
- Cruz, F.C.; Marin, M.T.; Leao, R.M.; Planeta, C.S. Behavioral and neuroendocrine effects of the exposure to chronic restraint or variable stress in early adolescent rats. Int. J. Dev. Neurosci. 2012, 30, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Ottenweller, J.E.; Servatius, R.J.; Tapp, W.N.; Drastal, S.D.; Bergen, M.T.; Natelson, B.H. A chronic stress state in rats: Effects of repeated stress on basal corticosterone and behavior. Physiol. Behav. 1992, 51, 689–698. [Google Scholar] [CrossRef]
- Samarghandian, S.; Samini, F.; Azimi-Nezhad, M.; Farkhondeh, T. Anti-oxidative effects of safranal on immobilization-induced oxidative damage in rat brain. Neurosci. Lett. 2017. [Google Scholar] [CrossRef] [PubMed]
- Kondo, S.; El Omri, A.; Han, J.; Isoda, H. Antidepressant-like effects of rosmarinic acid through mitogen-activated protein kinase phosphatase-1 and brain-derived neurotrophic factor modulation. J. Funct. Foods 2015, 14, 758–766. [Google Scholar] [CrossRef]
- Fontana, F.; Bernardi, P.; Pich, E.M.; Boschi, S.; De Iasio, R.; Spampinato, S.; Grossi, G. Opioid peptide modulation of circulatory and endocrine response to mental stress in humans. Peptides 1997, 18, 169–175. [Google Scholar] [CrossRef]
- Dalayeun, J.F.; Nores, J.M.; Bergal, S. Physiology of β-endorphins. A close-up view and a review of the literature. Biomed. Pharmacother. 1993, 47, 311–320. [Google Scholar] [CrossRef]
- Iwai, K.; Takahashi, T.; Nakahashi, T.; Nomura, K.; Atsumi, M.; Zeng, L.; Ishigami, K.; Kanda, T.; Yamaguchi, N.; Morimoto, S. Immobilization stress inhibits intimal fibromuscular proliferation in the process of arterial remodeling in rats. Hypertens. Res. 2008, 31, 977–986. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, N.; Shibasaki, T.; Wakabayashi, I.; Demura, H. Brain β-endorphin and other opioids are involved in restraint stress-induced stimulation of the hypothalamic-pituitary-adrenal axis, the sympathetic nervous system, and the adrenal medulla in the rat. Brain Res. 1997, 777, 140–146. [Google Scholar] [PubMed]
- Amat, N.; Hoxur, P.; Ming, D.; Matsidik, A.; Kijjoa, A.; Upur, H. Behavioral, neurochemical and neuroendocrine effects of abnormal savda munziq in the chronic stress mice. Evid. Based Complement. Altern. Med. 2012, 2012, 426757. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.L.; Licinio, J. From monoamines to genomic targets: A paradigm shift for drug discovery in depression. Nat. Rev. Drug Discov. 2004, 3, 136–151. [Google Scholar] [CrossRef] [PubMed]
- Haleem, D.J.; Haque, Z.; Ikram, H.; Haleem, M.A. Leptin and other Hormonal Responses to Different Stressors: Relationship with Stress-Induced Behavioral Deficits. Pak. Vet. J. 2014, 34, 504–507. [Google Scholar]
- Jung, H.Y.; Yoo, D.Y.; Kim, W.; Nam, S.M.; Kim, J.W.; Choi, J.H.; Kwak, Y.G.; Yoon, Y.S.; Hwang, I.K. Valeriana officinalis root extract suppresses physical stress by electric shock and psychological stress by nociceptive stimulation-evoked responses by decreasing the ratio of monoamine neurotransmitters to their metabolites. BMC Complement. Altern. Med. 2014, 14, 476. [Google Scholar] [CrossRef] [PubMed]
- Soderholm, J.D.; Perdue, M.H. Stress and the gastrointestinal tract II. Stress and intestinal barrier function. Am. J. Physiol. Gastrointest. Liver Physiol. 2001, 280, G7–G13. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.; Yang, Y.; Kwak, Y.S.; Song, G.G.; Kim, M.Y.; Rhee, M.H.; Cho, J.Y. Ginsenoside Rc from Panax ginseng exerts anti-inflammatory activity by targeting TANK-binding kinase 1/interferon regulatory factor-3 and p38/ATF-2. J. Ginseng Res. 2017, 41, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Mushtaq, N.; Schmatz, R.; Ahmed, M.; Pereira, L.B.; da Costa, P.; Reichert, K.P.; Dalenogare, D.; Pelinson, L.P.; Vieira, J.M.; Stefanello, N.; et al. Protective effect of rosmarinic acid against oxidative stress biomarkers in liver and kidney of strepotozotocin-induced diabetic rats. J. Physiol. Biochem. 2015, 71, 743–751. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.R.; Hu, C.H.; Xia, M.S.; Zhan, X.A.; Wang, M.Q. Effects of dietary fructooligosaccharide on digestive enzyme activities, intestinal microflora and morphology of male broilers. Poult. Sci. 2003, 82, 1030–1036. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Cho, Y.J.; Kim, J.H.; Kim, Y.B.; Lee, K.J. Stress-induced alterations in mast cell numbers and proteinase-activated receptor-2 expression of the colon: Role of corticotrophin-releasing factor. J. Korean Med. Sci. 2010, 25, 1330–1335. [Google Scholar] [CrossRef] [PubMed]
- Limon, R.I.; Penas, E.; Torino, M.I.; Martinez-Villaluenga, C.; Duenas, M.; Frias, J. Fermentation enhances the content of bioactive compounds in kidney bean extracts. Food Chem. 2015, 172, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Radak, Z.; Suzuki, K.; Higuchi, M.; Balogh, L.; Boldogh, I.; Koltai, E. Physical exercise, reactive oxygen species and neuroprotection. Free Radic. Biol. Med. 2016, 98, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, S.; Unsicker, K. ERK and cell death: ERK1/2 in neuronal death. FEBS J. 2010, 277, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Madrigal, J.L.; Moro, M.A.; Lizasoain, I.; Lorenzo, P.; Fernandez, A.P.; Rodrigo, J.; Bosca, L.; Leza, J.C. Induction of cyclooxygenase-2 accounts for restraint stress-induced oxidative status in rat brain. Neuropsychopharmacology 2003, 28, 1579–1588. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.P.; Jin, G.J.; Xiong, Y.; Hu, P.F.; Bao, J.P.; Wu, L.D. Rosmarinic acid down-regulates NO and PGE2 expression via MAPK pathway in rat chondrocytes. J. Cell. Mol. Med. 2018, 22, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Akanda, M.R.; Park, B.Y. Involvement of MAPK/NF-κB signal transduction pathways: Camellia japonica mitigates inflammation and gastric ulcer. Biomed. Pharmacother. 2017, 95, 1139–1146. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.J.; Kim, K.Y.; Kang, P.; Lee, H.S.; Seol, G.H. Effects of Salvia sclarea on chronic immobilization stress induced endothelial dysfunction in rats. BMC Complement. Altern. Med. 2014, 14, 396. [Google Scholar] [CrossRef] [PubMed]
- Rubisz-Brzezinska, J.; Jonderko, G.; Zebracka, T.; Dyczek-Parys, E. Immunoglobulin E levels in selected dermatoses. Prz. Dermatol. 1977, 64, 17–22. [Google Scholar]
- Akanda, M.R.; Tae, H.J.; Kim, I.S.; Ahn, D.; Tian, W.; Islam, A.; Nam, H.H.; Choo, B.K.; Park, B.Y. Hepatoprotective Role of Hydrangea macrophylla against Sodium Arsenite-Induced Mitochondrial-Dependent Oxidative Stress via the Inhibition of MAPK/Caspase-3 Pathways. Int. J. Mol. Sci. 2017, 18, E1482. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, W.; Akanda, M.R.; Islam, A.; Yang, H.-D.; Lee, S.-C.; Lee, J.-H.; Kim, S.-K.; Choi, Y.-J.; Im, S.-Y.; Park, B.-Y. The Anti-Stress Effect of Mentha arvensis in Immobilized Rats. Int. J. Mol. Sci. 2018, 19, 355. https://doi.org/10.3390/ijms19020355
Tian W, Akanda MR, Islam A, Yang H-D, Lee S-C, Lee J-H, Kim S-K, Choi Y-J, Im S-Y, Park B-Y. The Anti-Stress Effect of Mentha arvensis in Immobilized Rats. International Journal of Molecular Sciences. 2018; 19(2):355. https://doi.org/10.3390/ijms19020355
Chicago/Turabian StyleTian, Weishun, Md Rashedunnabi Akanda, Anowarul Islam, Hae-Dong Yang, Sang-Cheon Lee, Jeong-Ho Lee, Sang-Ki Kim, Yu-Jin Choi, So-Yeon Im, and Byung-Yong Park. 2018. "The Anti-Stress Effect of Mentha arvensis in Immobilized Rats" International Journal of Molecular Sciences 19, no. 2: 355. https://doi.org/10.3390/ijms19020355
APA StyleTian, W., Akanda, M. R., Islam, A., Yang, H.-D., Lee, S.-C., Lee, J.-H., Kim, S.-K., Choi, Y.-J., Im, S.-Y., & Park, B.-Y. (2018). The Anti-Stress Effect of Mentha arvensis in Immobilized Rats. International Journal of Molecular Sciences, 19(2), 355. https://doi.org/10.3390/ijms19020355