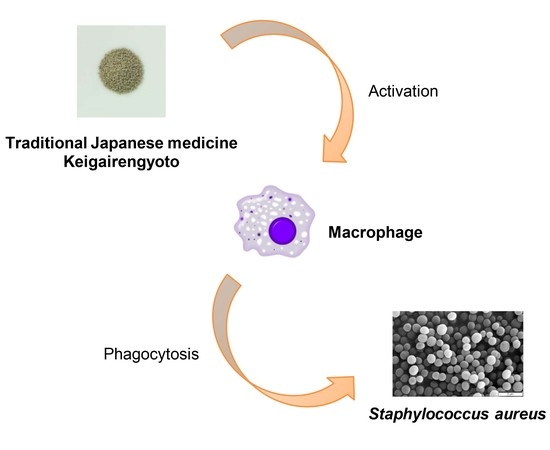
Pharmacokinetic Study of Bioactive Flavonoids in the Traditional Japanese Medicine Keigairengyoto Exerting Antibacterial Effects against Staphylococcus aureus
Abstract
:1. Introduction
2. Results
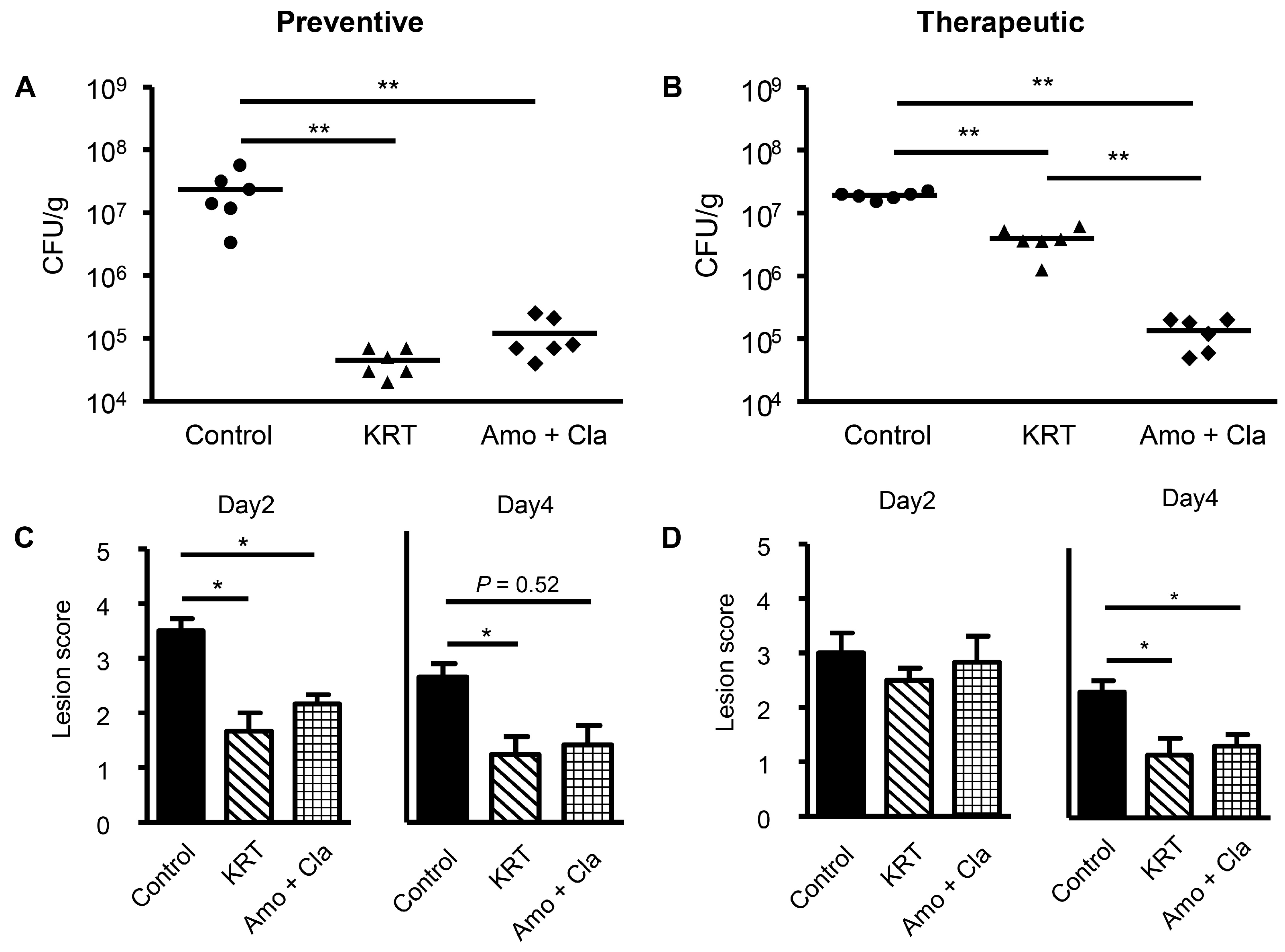
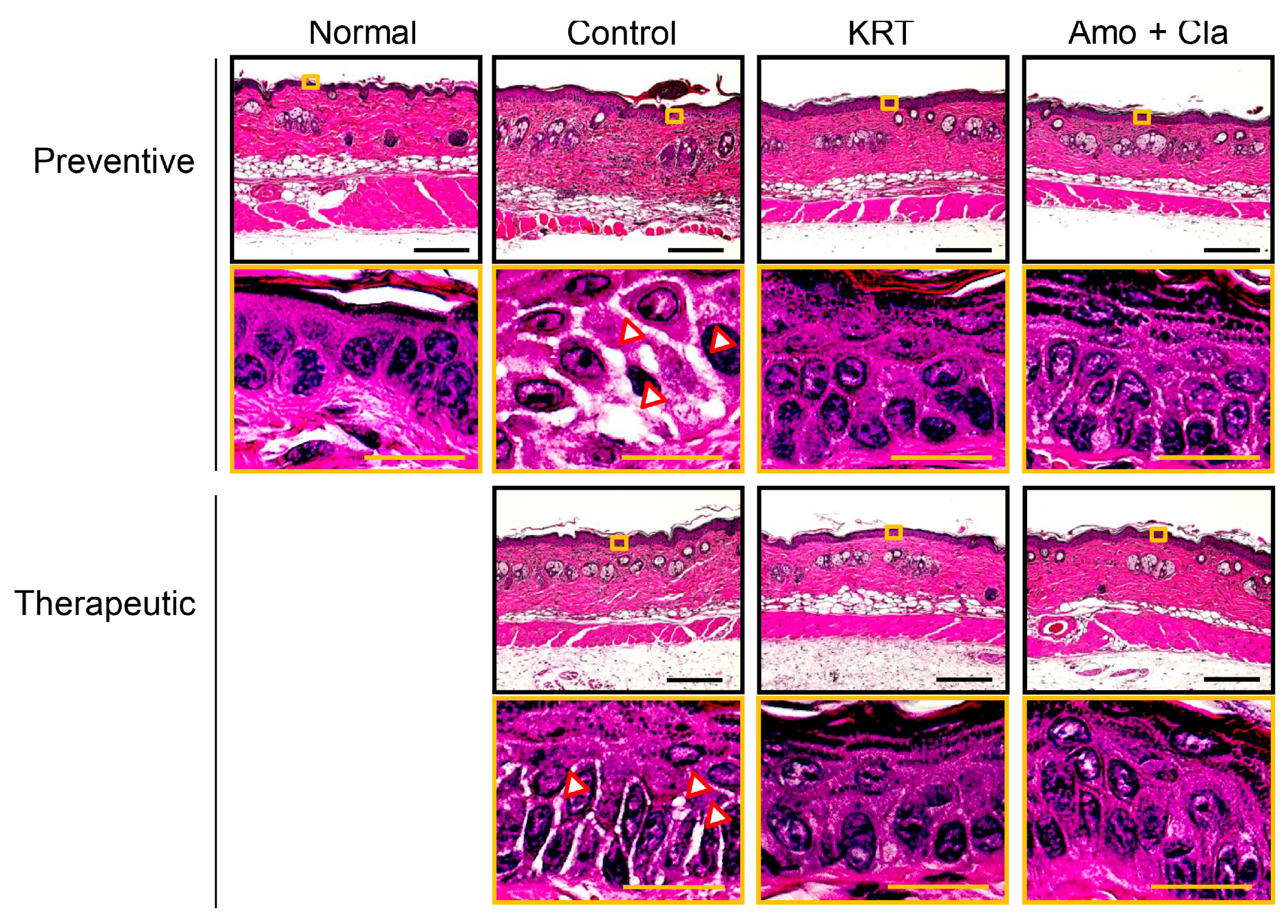
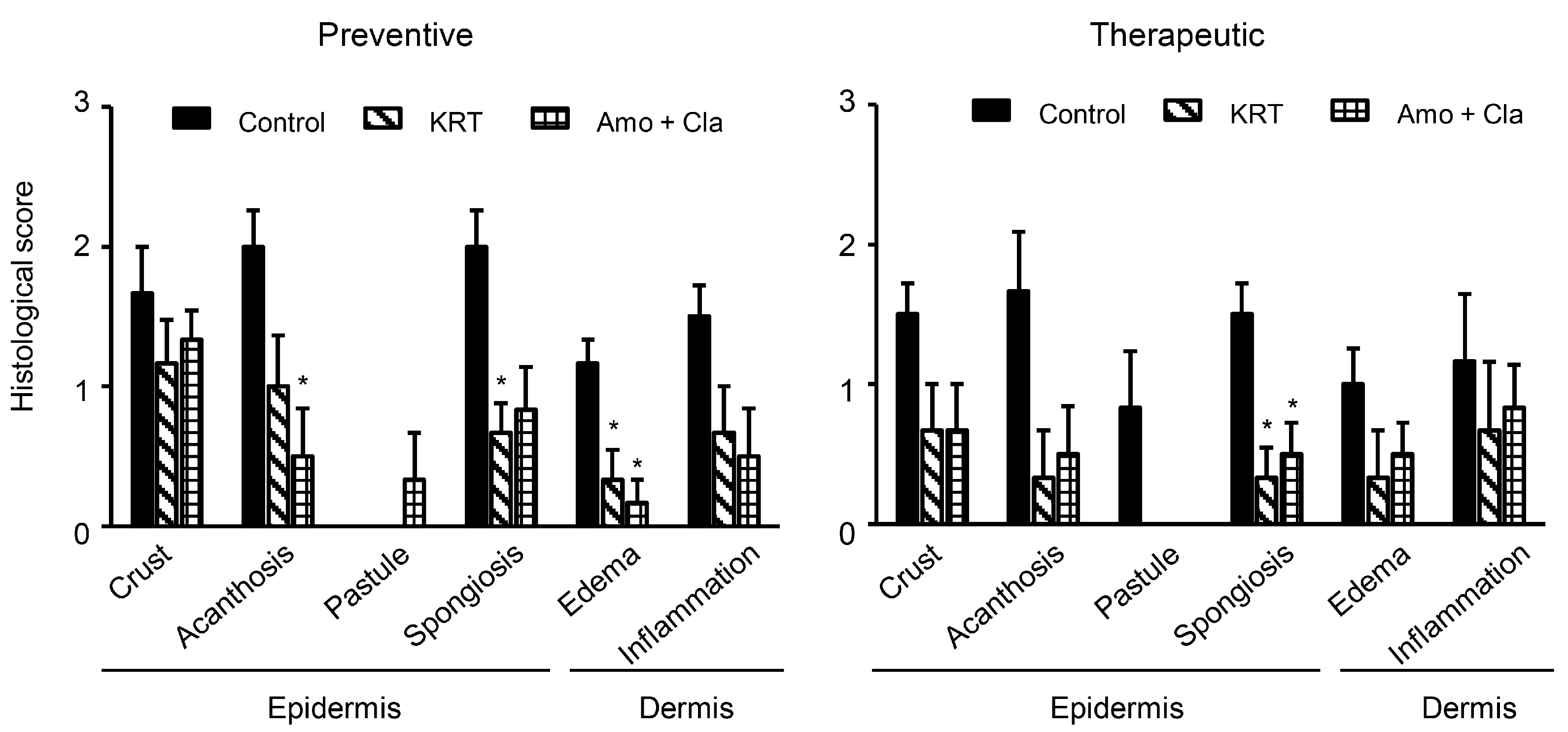
2.1. KRT Exhibited Antibacterial Effects in Cutaneous Infection
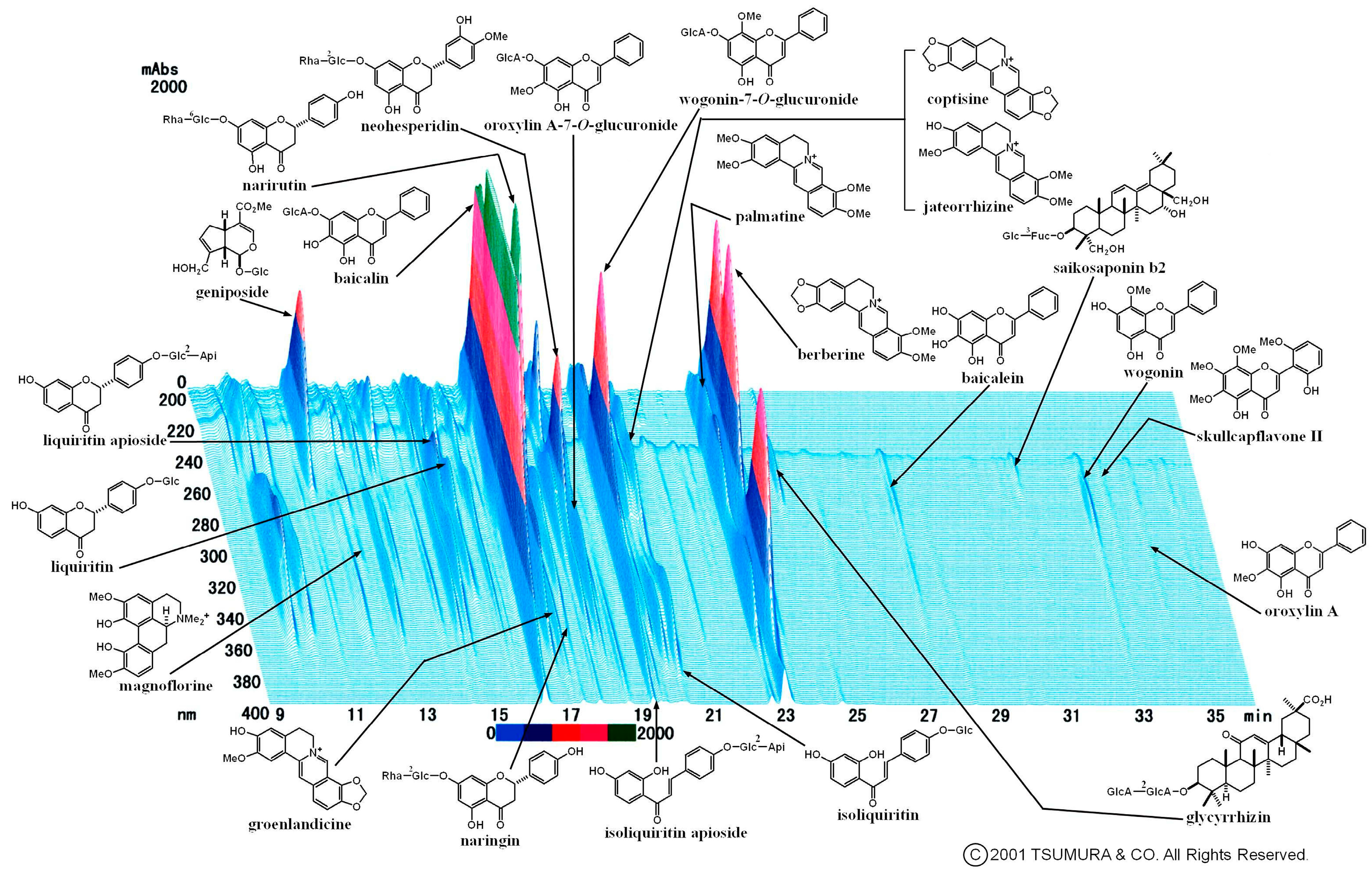
2.2. Multiple Flavonoids and Glucuronide Metabolites Were Identified in Plasma of KRT-Administered Rats
2.3. Active Flavonoids of KRT Augmented Phagocytosis by Macrophages
2.4. Macrophages Converted Flavonoid Glucuronide Baicalin to Aglycone Baicalein
3. Discussion
3.1. Antibacterial Activity of KRT
3.2. Pharmacokinetics of Multiple Flavonoids of KRT
3.3. Baicalin/Baicalein Is the Main Flavonoid Contributing to the Antibacterial Activity of KRT and Macrophages Are Involved in Deconjugation and Activation to Aglycone Baicalein
3.4. Comparison with Our Previous Papers Showing Genistein 7-O-glucuronide as a Key Flavonoid for Bacterial Clearance
3.5. Possible Utility of KRT as a Complement to Antibiotics
4. Materials and Methods
4.1. Test Drugs
4.2. Animals
4.3. Superficial Skin Infection Model
4.4. Pharmacokinetic Analysis of KRT-Derived Flavonoids and Glucuronide Metabolites by Liquid Chromatography–Mass Spectrometry with Tandem Mass Spectrometry
4.5. Macrophage Culture Assay
4.6. Conversion Assay of Baicalin to Baicalein
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| KRT | Keigairengyoto |
| 3D-HPLC | Three-dimensional high-performance liquid chromatograph |
| CFU | Colony forming units |
| BQL | Below the quantification limit (BQL) |
| Cmax | Maximum concentration |
| tmax | Time to maximum concentration |
| t1/2 | Apparent elimination half-life |
| AUC0–last | Area under the plasma concentration-time curve from time zero to the last observation |
| MFI | Mean fluorescence intensity |
| LC-MS/MS | Liquid chromatography–mass spectrometry with tandem mass spectrometry |
| FBS | Fetal bovine serum |
References
- Grassi, D.; Desideri, G.; Croce, G.; Tiberti, S.; Aggio, A.; Ferri, C. Flavonoids, vascular function and cardiovascular protection. Curr. Pharm. Des. 2009, 15, 1072–1084. [Google Scholar] [CrossRef] [PubMed]
- Nicolle, E.; Souard, F.; Faure, P.; Boumendjel, A. Flavonoids as promising lead compounds in type 2 diabetes mellitus: Molecules of interest and structure-activity relationship. Curr. Med. Chem. 2011, 18, 2661–2672. [Google Scholar] [CrossRef] [PubMed]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Surh, Y.J. Cancer chemoprevention with dietary phytochemicals. Nat. Rev. Cancer 2003, 3, 768–780. [Google Scholar] [CrossRef] [PubMed]
- Russo, G.I.; di Mauro, M.; Regis, F.; Reale, G.; Campisi, D.; Marranzano, M.; lo Giudice, A.; Solinas, T.; Madonia, M.; Cimino, S.; et al. Association between dietary phytoestrogens intakes and prostate cancer risk in Sicily. Aging Male 2017, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T. Flavonoids for allergic diseases: Present evidence and future perspective. Curr. Pharm. Des. 2014, 20, 879–885. [Google Scholar] [CrossRef] [PubMed]
- Emanuele, E.; Bertona, M.; Pareja-Galeano, H.; Fiuza-Luces, C.; Morales, J.S.; Sanchis-Gomar, F.; Lucia, A. Baicalin supplementation reduces serum biomarkers of skeletal muscle wasting and may protect against lean body mass reduction in cancer patients: Results from a pilot open-label study. Neuro Endocrinol. Lett. 2016, 37, 213–216. [Google Scholar] [PubMed]
- Dinda, B.; Dinda, S.; DasSharma, S.; Banik, R.; Chakraborty, A.; Dinda, M. Therapeutic potentials of baicalin and its aglycone, baicalein against inflammatory disorders. Eur. J. Med. Chem. 2017, 131, 68–80. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Wang, Y.; Ma, W.; Hu, Z.; Zhao, P. Genistein improves thyroid function in Hashimoto’s thyroiditis patients through regulating Th1 cytokines. Immunobiology 2017, 222, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Irrera, N.; Pizzino, G.; D’Anna, R.; Vaccaro, M.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Dietary Management of Skin Health: The Role of Genistein. Nutrients 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Yamada, H.; Tateishi, M.; Harada, K.; Ohashi, T.; Shimizu, T.; Atsumi, T.; Komagata, Y.; Iijima, H.; Komiyama, K.; Watanabe, H.; et al. A randomized clinical study of tea catechin inhalation effects on methicillin-resistant Staphylococcus aureus in disabled elderly patients. J. Am. Med. Dir. Assoc. 2006, 7, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Huo, X.K.; Wang, B.; Zheng, L.; Cong, H.J.; Xiang, T.; Wang, S.M.; Sun, C.P.; Wang, C.; Zhang, L.; Deng, S.; et al. Comparative pharmacokinetic study of baicalin and its metabolites after oral administration of baicalin and Chaiqin Qingning capsule in normal and febrile rats. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2017, 1059, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Holder, C.L.; Churchwell, M.I.; Doerge, D.R. Quantification of soy isoflavones, genistein and daidzein, and conjugates in rat blood using LC/ES-MS. J. Agric. Food Chem. 1999, 47, 3764–3770. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Matsubara, Y.; Mizuhara, Y.; Sekiguchi, K.; Koseki, J.; Tsuchiya, K.; Nishimura, H.; Watanabe, J.; Kaneko, A.; Maemura, K.; et al. Plasma Pharmacokinetics of Polyphenols in a Traditional Japanese Medicine, Jumihaidokuto, Which Suppresses Propionibacterium acnes-Induced Dermatitis in Rats. Molecules 2015, 20, 18031–18046. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, A.; Matsumoto, T.; Matsubara, Y.; Sekiguchi, K.; Koseki, J.; Yakabe, R.; Aoki, K.; Aiba, S.; Yamasaki, K. Glucuronides of phytoestrogen flavonoid enhance macrophage function via conversion to aglycones by β-glucuronidase in macrophages. Immun. Inflamm. Dis. 2017, 5, 265–279. [Google Scholar] [CrossRef] [PubMed]
- Galindo, P.; Rodriguez-Gomez, I.; Gonzalez-Manzano, S.; Duenas, M.; Jimenez, R.; Menendez, C.; Vargas, F.; Tamargo, J.; Santos-Buelga, C.; Perez-Vizcaino, F.; et al. Glucuronidated quercetin lowers blood pressure in spontaneously hypertensive rats via deconjugation. PLoS ONE 2012, 7, e32673. [Google Scholar] [CrossRef] [PubMed]
- Terao, J.; Murota, K.; Kawai, Y. Conjugated quercetin glucuronides as bioactive metabolites and precursors of aglycone in vivo. Food Funct. 2011, 2, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Koseki, J.; Kaneko, A.; Matsubara, Y.; Sekiguchi, K.; Ebihara, S.; Aiba, S.; Yamasaki, K. Keigairengyoto, a traditional Japanese medicine, promotes bacterial clearance by activating innate immune cells in mouse cutaneous infection models. Trends Immunother. 2017, 1, 35–49. [Google Scholar] [CrossRef]
- Higaki, S.; Hasegawa, Y.; Morohashi, M.; Takayoshi, Y. The correlation of Kampo formulations and their ingredients on anti-bacterial activities against Propionibacterium acnes. J. Dermatol. 1995, 22, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Higaki, S.; Morimatsu, S.; Morohashi, M.; Yamagishi, T.; Hasegawa, Y. Susceptibility of Propionibacterium acnes, Staphylococcus aureus and Staphylococcus epidermidis to 10 Kampo formulations. J. Int. Med. Res. 1997, 25, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liu, T.; Wang, K.; Hou, C.; Cai, S.; Huang, Y.; Du, Z.; Huang, H.; Kong, J. Baicalein Inhibits Staphylococcus aureus Biofilm Formation and the Quorum Sensing System In Vitro. PLoS ONE 2016, 11, e0153468. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.X.; Schrader, K.K.; Khan, I.A.; Rimando, A.M. Activities of wogonin analogs and other flavones against Flavobacterium columnare. Chem. Biodivers. 2015, 12, 259–272. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, S.; Morita, T.; Endo, H.; Hamamoto, T.; Baba, M.; Joichi, Y.; Kaneko, S.; Okada, Y.; Okuyama, T.; Nishino, H.; et al. Isoliquiritigenin suppresses pulmonary metastasis of mouse renal cell carcinoma. Cancer Lett. 2002, 183, 23–30. [Google Scholar] [CrossRef]
- Lim, E.K.; Mitchell, P.J.; Brown, N.; Drummond, R.A.; Brown, G.D.; Kaye, P.M.; Bowles, D.J. Regiospecific methylation of a dietary flavonoid scaffold selectively enhances IL-1β production following Toll-like receptor 2 stimulation in THP-1 monocytes. J. Biol. Chem. 2013, 288, 21126–21135. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kim, S.L.; Lee, S.; Chung, M.J.; Park, Y.I. Immunostimulating activity of maysin isolated from corn silk in murine RAW 264.7 macrophages. BMB Rep. 2014, 47, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Fung, M.C.; Szeto, Y.Y.; Leung, K.N.; Wong-Leung, Y.L.; Mak, N.K. Effects of biochanin A on the growth and differentiation of myeloid leukemia WEHI-3B (JCS) cells. Life Sci. 1997, 61, 105–115. [Google Scholar] [CrossRef]
- Takahashi, T.; Kobori, M.; Shinmoto, H.; Tsushida, T. Structure-activity relationships of flavonoids and the induction of granulocytic- or monocytic-differentiation in HL60 human myeloid leukemia cells. Biosci. Biotechnol. Biochem. 1998, 62, 2199–2204. [Google Scholar] [CrossRef] [PubMed]
- Kawaii, S.; Tomono, Y.; Katase, E.; Ogawa, K.; Yano, M. Effect of citrus flavonoids on HL-60 cell differentiation. Anticancer Res. 1999, 19, 1261–1269. [Google Scholar] [PubMed]
- Bispo da Silva, A.; Cerqueira Coelho, P.L.; Alves Oliveira Amparo, J.; Alves de Almeida Carneiro, M.M.; Pereira Borges, J.M.; Dos Santos Souza, C.; Dias Costa, M.F.; Mecha, M.; Guaza Rodriguez, C.; Amaral da Silva, V.D.; et al. The flavonoid rutin modulates microglial/macrophage activation to a CD150/CD206 M2 phenotype. Chem. Biol. Interact. 2017, 274, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Cai, W.; Zhou, Y.; Liu, Y.; Wu, X.; Li, Y.; Lu, J.; Qiao, Y. Profiling and identification of the metabolites of baicalin and study on their tissue distribution in rats by ultra-high-performance liquid chromatography with linear ion trap-Orbitrap mass spectrometer. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2015, 985, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Walsh, K.R.; Haak, S.J.; Bohn, T.; Tian, Q.; Schwartz, S.J.; Failla, M.L. Isoflavonoid glucosides are deconjugated and absorbed in the small intestine of human subjects with ileostomies. Am. J. Clin. Nutr. 2007, 85, 1050–1056. [Google Scholar] [PubMed]
- Proteggente, A.R.; Basu-Modak, S.; Kuhnle, G.; Gordon, M.J.; Youdim, K.; Tyrrell, R.; Rice-Evans, C.A. Hesperetin glucuronide, a photoprotective agent arising from flavonoid metabolism in human skin fibroblasts. Photochem. Photobiol. 2003, 78, 256–261. [Google Scholar] [CrossRef]
- Zhao, J.; Agarwal, R. Tissue distribution of silibinin, the major active constituent of silymarin, in mice and its association with enhancement of phase II enzymes: Implications in cancer chemoprevention. Carcinogenesis 1999, 20, 2101–2108. [Google Scholar] [CrossRef] [PubMed]
- Suganuma, M.; Okabe, S.; Oniyama, M.; Tada, Y.; Ito, H.; Fujiki, H. Wide distribution of [3H](−)-epigallocatechin gallate, a cancer preventive tea polyphenol, in mouse tissue. Carcinogenesis 1998, 19, 1771–1776. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Sun, J.; Wang, Y.; He, W.; Wang, L.; Zheng, Y.; Wu, J.; Zhang, Y.; Jiang, X. Antimycobacterial and Anti-inflammatory Mechanisms of Baicalin via Induced Autophagy in Macrophages Infected with Mycobacterium tuberculosis. Front. Microbiol. 2017, 8, 2142. [Google Scholar] [CrossRef] [PubMed]
- Li, C.G.; Yan, L.; Mai, F.Y.; Shi, Z.J.; Xu, L.H.; Jing, Y.Y.; Zha, Q.B.; Ouyang, D.Y.; He, X.H. Baicalin Inhibits NOD-Like Receptor Family, Pyrin Containing Domain 3 Inflammasome Activation in Murine Macrophages by Augmenting Protein Kinase A Signaling. Front. Immunol. 2017, 8, 1409. [Google Scholar] [CrossRef] [PubMed]
- Khaksa, G.; Zolfaghari, M.E.; Dehpour, A.R.; Samadian, T. Anti-inflammatory and anti-nociceptive activity of disodium glycyrrhetinic acid hemiphthalate. Planta Med. 1996, 62, 326–328. [Google Scholar] [CrossRef] [PubMed]
- Funakushi, N.; Yamaguchi, T.; Jiang, J.; Imamura, S.; Kuhara, T.; Suto, H.; Ueki, R.; Kase, Y.; Kobayashi, H.; Ogawa, H.; et al. Ameliorating effect of Yokukansan on the development of atopic dermatitis-like lesions and scratching behavior in socially isolated NC/Nga mice. Arch. Dermatol. Res. 2011, 303, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Mitsuhata, H.; Yuzurihara, M.; Kase, Y. Antiallodynic effect of herbal medicine yokukansan on peripheral neuropathy in rats with chronic constriction injury. Evid.-Based Complement. Alternat. Med. 2012, 2012, 953459. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.; Branham, W.S.; Ng, H.W.; Moland, C.L.; Dial, S.L.; Fang, H.; Perkins, R.; Sheehan, D.; Tong, W. Human sex hormone-binding globulin binding affinities of 125 structurally diverse chemicals and comparison with their binding to androgen receptor, estrogen receptor, and α-fetoprotein. Toxicol. Sci 2015, 143, 333–348. [Google Scholar] [CrossRef] [PubMed]
- Branham, W.S.; Dial, S.L.; Moland, C.L.; Hass, B.S.; Blair, R.M.; Fang, H.; Shi, L.; Tong, W.; Perkins, R.G.; Sheehan, D.M. Phytoestrogens and mycoestrogens bind to the rat uterine estrogen receptor. J. Nutr. 2002, 132, 658–664. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, J.; Hong, D.Y.; Chen, L.; Zhang, Y.Y.; Xu, Y.N.; Pan, D.; Fu, L.Y.; Tao, L.; Luo, H.; et al. Baicalein has protective effects on the 17β-estradiol-induced transformation of breast epithelial cells. Oncotarget 2017, 8, 10470–10484. [Google Scholar] [PubMed]
- Jaillon, S.; Berthenet, K.; Garlanda, C. Sexual Dimorphism in Innate Immunity. Clin. Rev. Allergy Immunol. 2017, 46, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Mistry, R.D. Skin and soft tissue infections. Pediatr. Clin. N. Am. 2013, 60, 1063–1082. [Google Scholar] [CrossRef] [PubMed]
- Manichanh, C.; Reeder, J.; Gibert, P.; Varela, E.; Llopis, M.; Antolin, M.; Guigo, R.; Knight, R.; Guarner, F. Reshaping the gut microbiome with bacterial transplantation and antibiotic intake. Genome Res. 2010, 20, 1411–1419. [Google Scholar] [CrossRef] [PubMed]
- Willyard, C. The drug-resistant bacteria that pose the greatest health threats. Nature 2017, 543, 15. [Google Scholar] [CrossRef] [PubMed]
- Kugelberg, E.; Norstrom, T.; Petersen, T.K.; Duvold, T.; Andersson, D.I.; Hughes, D. Establishment of a superficial skin infection model in mice by using Staphylococcus aureus and Streptococcus pyogenes. Antimicrob. Agents Chemother. 2005, 49, 3435–3441. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Oscherwitz, J.; Cease, K.B.; Chan, S.M.; Munoz-Planillo, R.; Hasegawa, M.; Villaruz, A.E.; Cheung, G.Y.; McGavin, M.J.; Travers, J.B.; et al. Staphylococcus delta-toxin induces allergic skin disease by activating mast cells. Nature 2013, 503, 397–401. [Google Scholar] [CrossRef] [PubMed]
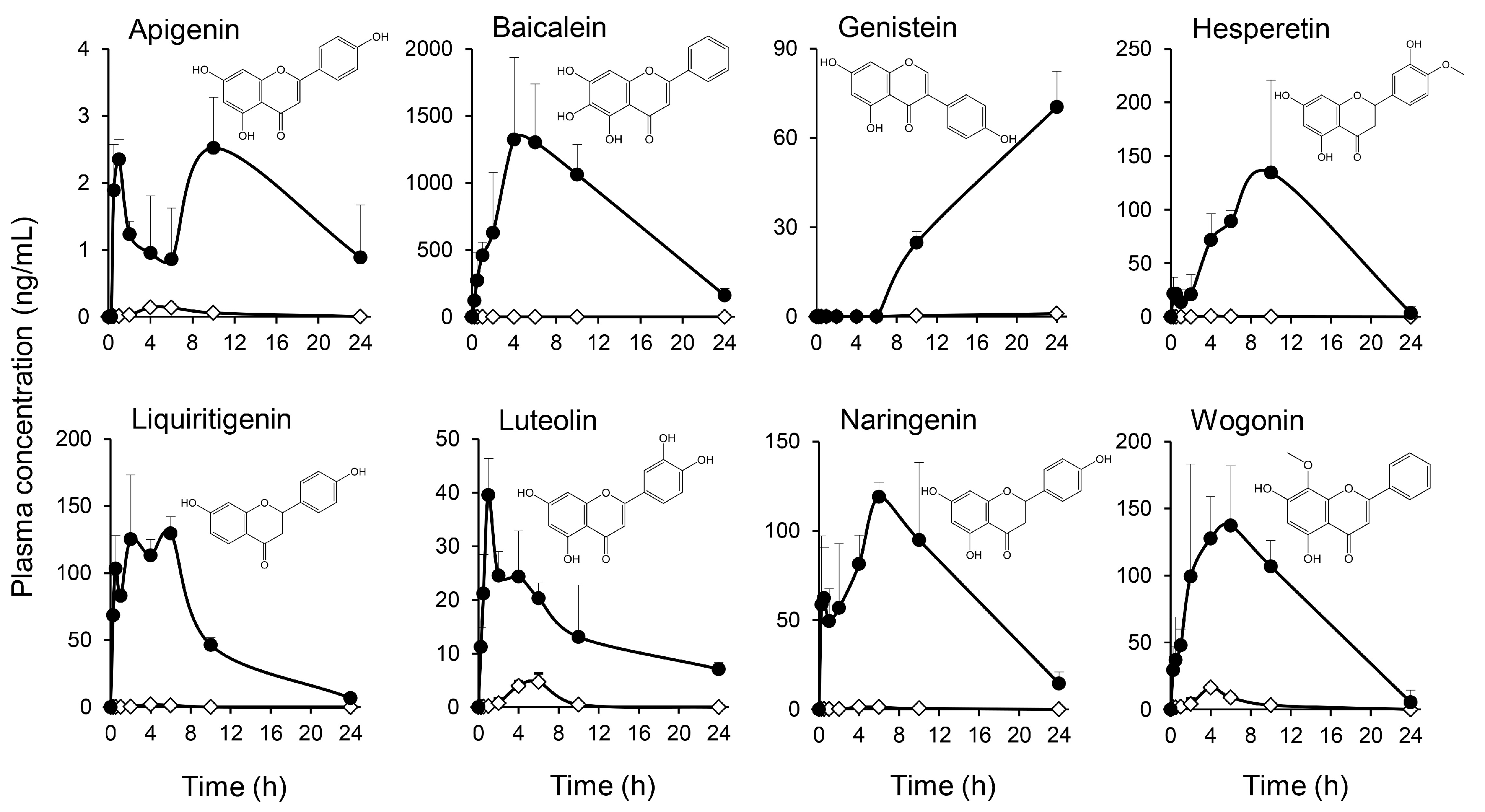
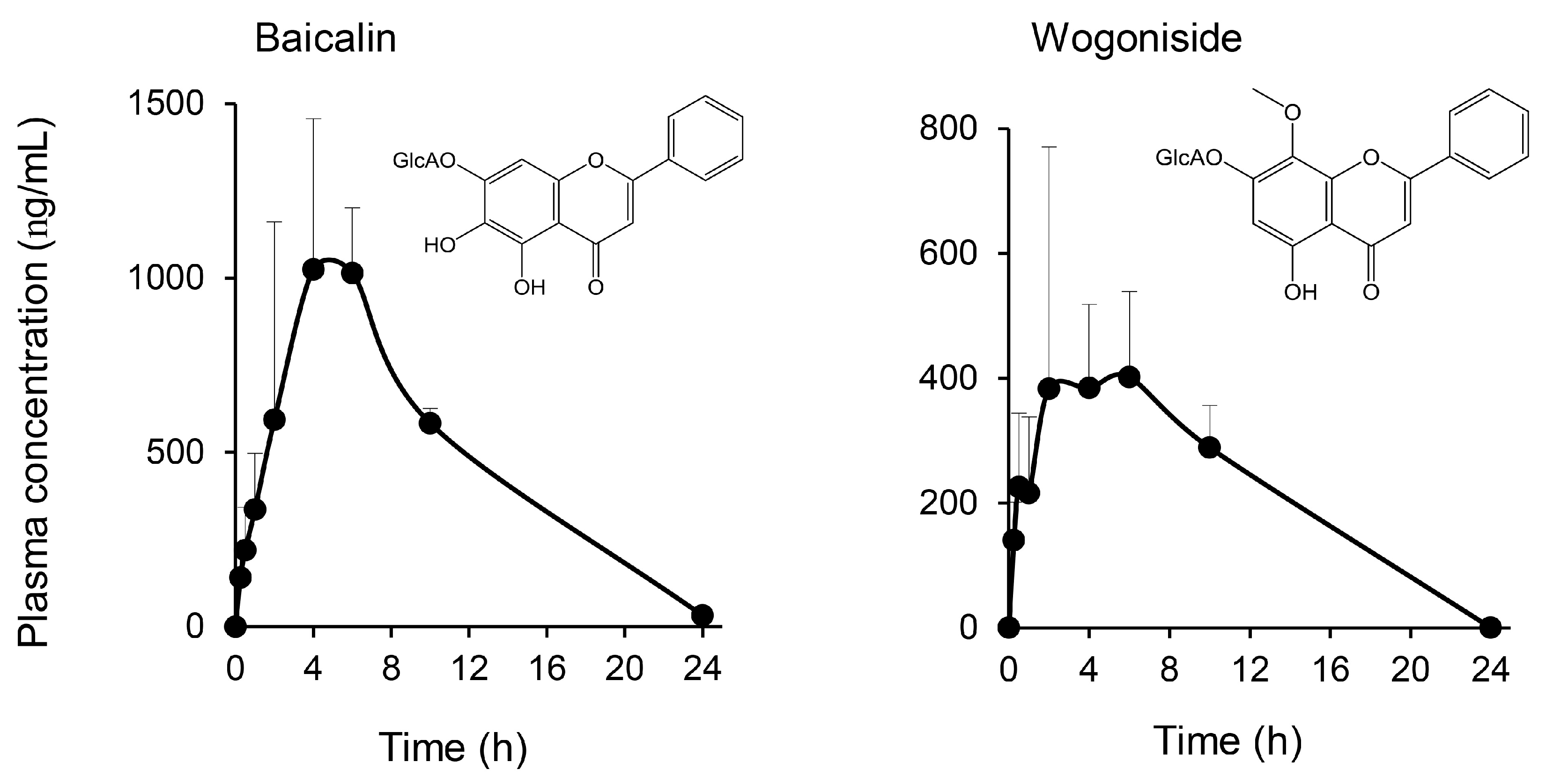

| Treatment | Compound | tmax (h) | Cmax (ng/mL) | AUC0–last (ng·h/mL) | t1/2 (h) |
|---|---|---|---|---|---|
| No treatment | Apigenin | 4.00 | 0.140 | 0.843 | - |
| Baicalein | - | BQL # | - | - | |
| Genistein | 24.0 | 0.998 | 9.65 | - | |
| Hesperetin | 4.00 | 0.644 | 2.83 | - | |
| Liquiritigenin | 4.00 | 2.11 | 8.68 | - | |
| Luteolin | 6.00 | 4.70 | 21.3 | - | |
| Naringenin | 4.00 | 1.34 | 9.53 | 3.01 | |
| Wogonin | 4.00 | 16.3 | 71.5 | - | |
| Treatment with β-glucuronidase | Apigenin | 10.0 | 2.52 | 35.7 | - |
| Baicalein | 4.00 | 1320 | 16,800 | 5.72 | |
| Genistein | 24.0 | 75.2 | 763 | - | |
| Hesperetin | 10.0 | 135 | 735 | - | |
| Liquiritigenin | 6.00 | 130 | 1270 | - | |
| Luteolin | 1.00 | 39.6 | 348 | 11.7 | |
| Naringenin | 6.00 | 119 | 1470 | - | |
| Wogonin | 6.00 | 146 | 1150 | - |
| Experiment No. | IFN-γ | Test Sample | Concentration (μmol/L) | Phagocytosis MFI (Relative Change #) |
|---|---|---|---|---|
| Exp. 1 | − | No IFN-γ | - | 144 ± 6 |
| + | Control | - | 155 ± 4 | |
| + | apigenin | 10 | 507 ± 11 ** (3.3) | |
| + | baicalein | 10 | 530 ± 6 ** (3.4) | |
| + | genistein | 10 | 525 ± 22 ** (3.4) | |
| + | hesperetin | 10 | 193 ± 12 (1.2) | |
| + | liquiritigenin | 10 | 245 ± 4 ** (1.6) | |
| + | luteolin | 10 | 408 ± 22 ** (2.6) | |
| + | naringenin | 10 | 236 ± 10 ** (1.5) | |
| + | wogonin | 10 | 271 ± 16 ** (1.7) | |
| Exp. 2 | − | No IFN-γ | - | 162 ± 1 |
| + | Control | - | 226 ± 12 | |
| + | baicalein | 1 | 223 ± 10 (1.0) | |
| + | baicalein | 3 | 445 ± 7 ** (2.0) | |
| + | baicalein | 10 | 866 ± 25 ** (3.8) | |
| + | baicalin | 1 | 238 ± 9 (1.1) | |
| + | baicalin | 3 | 269 ± 10 (1.2) | |
| + | baicalin | 10 | 531 ± 19 ** (2.4) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matsumoto, T.; Kaneko, A.; Koseki, J.; Matsubara, Y.; Aiba, S.; Yamasaki, K. Pharmacokinetic Study of Bioactive Flavonoids in the Traditional Japanese Medicine Keigairengyoto Exerting Antibacterial Effects against Staphylococcus aureus. Int. J. Mol. Sci. 2018, 19, 328. https://doi.org/10.3390/ijms19020328
Matsumoto T, Kaneko A, Koseki J, Matsubara Y, Aiba S, Yamasaki K. Pharmacokinetic Study of Bioactive Flavonoids in the Traditional Japanese Medicine Keigairengyoto Exerting Antibacterial Effects against Staphylococcus aureus. International Journal of Molecular Sciences. 2018; 19(2):328. https://doi.org/10.3390/ijms19020328
Chicago/Turabian StyleMatsumoto, Takashi, Atsushi Kaneko, Junichi Koseki, Yosuke Matsubara, Setsuya Aiba, and Kenshi Yamasaki. 2018. "Pharmacokinetic Study of Bioactive Flavonoids in the Traditional Japanese Medicine Keigairengyoto Exerting Antibacterial Effects against Staphylococcus aureus" International Journal of Molecular Sciences 19, no. 2: 328. https://doi.org/10.3390/ijms19020328
APA StyleMatsumoto, T., Kaneko, A., Koseki, J., Matsubara, Y., Aiba, S., & Yamasaki, K. (2018). Pharmacokinetic Study of Bioactive Flavonoids in the Traditional Japanese Medicine Keigairengyoto Exerting Antibacterial Effects against Staphylococcus aureus. International Journal of Molecular Sciences, 19(2), 328. https://doi.org/10.3390/ijms19020328