miRNAmotif—A Tool for the Prediction of Pre-miRNA–Protein Interactions
Abstract
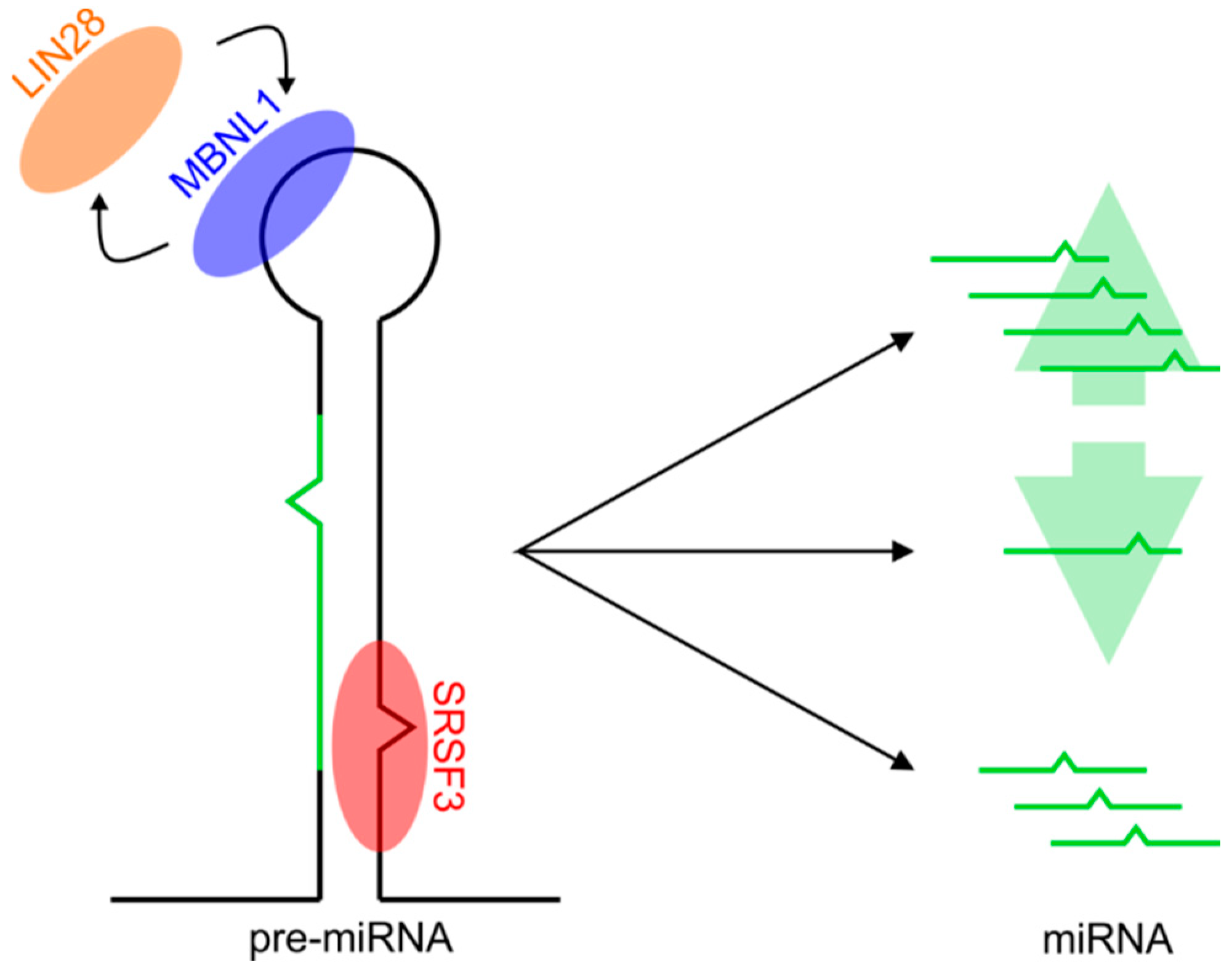
1. Introduction
2. Results
2.1. Searches for Known Motifs within Pre-miRNA Sequences
2.1.1. Human miRNAs with the Lin28 Motif
2.1.2. Human miRNAs Containing Highly Similar Motifs
2.2. Predicting Protein-Binding Motifs within Pre-miRNA Sequences
2.2.1. Common Motifs in Pre-miRNAs Interacting with DEAD-Box Helicases
3. Discussion
4. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| miRNA | microRNA |
| DDX | DEAD box proteins |
| RS | Arginine/serine-rich domain |
| RRM | RNA recognition motif |
| RBP | RNA-binding protein |
| IUPAC | International Union of Pure and Applied Chemistry |
| CLIP | Crosslinking immunoprecipitation |
| CLI | Command line interface |
References
- Ameres, S.L.; Zamore, P.D. Diversifying microRNA sequence and function. Nat. Rev. Mol. Cell Biol. 2013, 14, 475–488. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P. Metazoan MicroRNAs. Cell 2018, 173, 20–51. [Google Scholar] [CrossRef] [PubMed]
- Svoronos, A.A.; Engelman, D.M.; Slack, F.J. OncomiR or Tumor Suppressor? The Duplicity of MicroRNAs in Cancer. Cancer Res. 2016, 76, 3666–3670. [Google Scholar] [CrossRef] [PubMed]
- Garo, L.P.; Murugaiyan, G. Contribution of MicroRNAs to autoimmune diseases. Cell. Mol. Life Sci. 2016, 73, 2041–2051. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Liu, L.; Lü, L.; Zou, Q. Prediction of potential disease-associated microRNAs using structural perturbation method. Bioinforma. Oxf. Engl. 2018, 34, 2425–2432. [Google Scholar] [CrossRef]
- Tang, W.; Wan, S.; Yang, Z.; Teschendorff, A.E.; Zou, Q. Tumor origin detection with tissue-specific miRNA and DNA methylation markers. Bioinforma. Oxf. Engl. 2018, 34, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Zou, Q.; Mao, Y.; Hu, L.; Wu, Y.; Ji, Z. miRClassify: An advanced web server for miRNA family classification and annotation. Comput. Biol. Med. 2014, 45, 157–160. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, Z. Computational Biology in microRNA. Wiley Interdiscip. Rev. RNA 2015, 6, 435–452. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Wang, J.; Li, M.; Lan, W.; Wu, F.; Pan, Y. miRTRS: A Recommendation Algorithm for Predicting miRNA Targets. IEEE/ACM Trans. Comput. Biol. Bioinform. 2018. [Google Scholar] [CrossRef] [PubMed]
- Andrés-León, E.; Rojas, A.M. miARma-Seq, a comprehensive pipeline for the simultaneous study and integration of miRNA and mRNA expression data. Methods San Diego Calif. 2018. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Shi, J.; Gao, Y.; Cui, C.; Zhang, S.; Li, J.; Zhou, Y.; Cui, Q. HMDD v3.0: A database for experimentally supported human microRNA-disease associations. Nucleic Acids Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, C.-C.; Yin, J.; You, Z.-H. Novel Human miRNA-Disease Association Inference Based on Random Forest. Mol. Ther. Nucleic Acids 2018, 13, 568–579. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Cui, Z.; Zan, X. Identifying cancer-related microRNAs based on subpathways. IET Syst. Biol. 2018, 12, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Stroynowska-Czerwinska, A.; Fiszer, A.; Krzyzosiak, W.J. The panorama of miRNA-mediated mechanisms in mammalian cells. Cell. Mol. Life Sci. CMLS 2014, 71, 2253–2270. [Google Scholar] [CrossRef] [PubMed]
- Ha, M.; Kim, V.N. Regulation of microRNA biogenesis. Nat. Rev. Mol. Cell Biol. 2014, 15, 509–524. [Google Scholar] [CrossRef]
- Creugny, A.; Fender, A.; Pfeffer, S. Regulation of primary microRNA processing. FEBS Lett. 2018, 592, 1980–1996. [Google Scholar] [CrossRef] [PubMed]
- Bayoumi, A.S.; Sayed, A.; Broskova, Z.; Teoh, J.-P.; Wilson, J.; Su, H.; Tang, Y.-L.; Kim, I. Crosstalk between Long Noncoding RNAs and MicroRNAs in Health and Disease. Int. J. Mol. Sci. 2016, 17, 356. [Google Scholar] [CrossRef] [PubMed]
- Witkos, T.M.; Krzyzosiak, W.J.; Fiszer, A.; Koscianska, E. A potential role of extended simple sequence repeats in competing endogenous RNA crosstalk. RNA Biol. 2018, 1–11. [Google Scholar] [CrossRef]
- Ratnadiwakara, M.; Mohenska, M.; Änkö, M.-L. Splicing factors as regulators of miRNA biogenesis - links to human disease. Semin. Cell Dev. Biol. 2018, 79, 113–122. [Google Scholar] [CrossRef]
- Gebert, L.F.R.; MacRae, I.J. Regulation of microRNA function in animals. Nat. Rev. Mol. Cell Biol. 2018. [Google Scholar] [CrossRef]
- Treiber, T.; Treiber, N.; Meister, G. Regulation of microRNA biogenesis and its crosstalk with other cellular pathways. Nat. Rev. Mol. Cell Biol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Krol, J.; Loedige, I.; Filipowicz, W. The widespread regulation of microRNA biogenesis, function and decay. Nat. Rev. Genet. 2010, 11, 597–610. [Google Scholar] [CrossRef] [PubMed]
- Michlewski, G.; Caceres, J.F. Post-transcriptional control of miRNA biogenesis. RNA 2018, rna.068692.118. [Google Scholar] [CrossRef] [PubMed]
- Connerty, P.; Ahadi, A.; Hutvagner, G. RNA Binding Proteins in the miRNA Pathway. Int. J. Mol. Sci. 2016, 17, 31. [Google Scholar] [CrossRef] [PubMed]
- Castilla-Llorente, V.; Nicastro, G.; Ramos, A. Terminal loop-mediated regulation of miRNA biogenesis: Selectivity and mechanisms. Biochem. Soc. Trans. 2013, 41, 861–865. [Google Scholar] [CrossRef] [PubMed]
- Burd, C.G.; Dreyfuss, G. RNA binding specificity of hnRNP A1: Significance of hnRNP A1 high-affinity binding sites in pre-mRNA splicing. EMBO J. 1994, 13, 1197–1204. [Google Scholar] [CrossRef]
- Myer, V.E.; Fan, X.C.; Steitz, J.A. Identification of HuR as a protein implicated in AUUUA-mediated mRNA decay. EMBO J. 1997, 16, 2130–2139. [Google Scholar] [CrossRef] [PubMed]
- Nicastro, G.; García-Mayoral, M.F.; Hollingworth, D.; Kelly, G.; Martin, S.R.; Briata, P.; Gherzi, R.; Ramos, A. Noncanonical G recognition mediates KSRP regulation of let-7 biogenesis. Nat. Struct. Mol. Biol. 2012, 19, 1282–1286. [Google Scholar] [CrossRef]
- Nam, Y.; Chen, C.; Gregory, R.I.; Chou, J.J.; Sliz, P. Molecular basis for interaction of let-7 microRNAs with Lin28. Cell 2011, 147, 1080–1091. [Google Scholar] [CrossRef]
- Goers, E.S.; Purcell, J.; Voelker, R.B.; Gates, D.P.; Berglund, J.A. MBNL1 binds GC motifs embedded in pyrimidines to regulate alternative splicing. Nucleic Acids Res. 2010, 38, 2467–2484. [Google Scholar] [CrossRef]
- Suzuki, H.I.; Arase, M.; Matsuyama, H.; Choi, Y.L.; Ueno, T.; Mano, H.; Sugimoto, K.; Miyazono, K. MCPIP1 ribonuclease antagonizes dicer and terminates microRNA biogenesis through precursor microRNA degradation. Mol. Cell 2011, 44, 424–436. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.A.; Jo, M.H.; Choi, Y.-G.; Park, J.; Kwon, S.C.; Hohng, S.; Kim, V.N.; Woo, J.-S. Functional Anatomy of the Human Microprocessor. Cell 2015, 161, 1374–1387. [Google Scholar] [CrossRef] [PubMed]
- Treiber, T.; Treiber, N.; Plessmann, U.; Harlander, S.; Daiß, J.-L.; Eichner, N.; Lehmann, G.; Schall, K.; Urlaub, H.; Meister, G. A Compendium of RNA-Binding Proteins that Regulate MicroRNA Biogenesis. Mol. Cell 2017, 66, 270–284.e13. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Liu, Y.; Wan, G.; Choi, H.J.; Zhao, L.; Ivan, C.; He, X.; Sood, A.K.; Zhang, X.; Lu, X. The RNA-binding protein DDX1 promotes primary microRNA maturation and inhibits ovarian tumor progression. Cell Rep. 2014, 8, 1447–1460. [Google Scholar] [CrossRef] [PubMed]
- Moy, R.H.; Cole, B.S.; Yasunaga, A.; Gold, B.; Shankarling, G.; Varble, A.; Molleston, J.M.; TenOever, B.R.; Lynch, K.W.; Cherry, S. Stem-loop recognition by DDX17 facilitates miRNA processing and antiviral defense. Cell 2014, 158, 764–777. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zubovic, L.; Yang, F.; Godin, K.; Pavelitz, T.; Castellanos, J.; Macchi, P.; Varani, G. Rbfox proteins regulate microRNA biogenesis by sequence-specific binding to their precursors and target downstream Dicer. Nucleic Acids Res. 2016, 44, 4381–4395. [Google Scholar] [CrossRef] [PubMed]
- Davis, B.N.; Hilyard, A.C.; Nguyen, P.H.; Lagna, G.; Hata, A. Smad proteins bind a conserved RNA sequence to promote microRNA maturation by Drosha. Mol. Cell 2010, 39, 373–384. [Google Scholar] [CrossRef]
- Kim, K.; Nguyen, T.D.; Li, S.; Nguyen, T.A. SRSF3 recruits DROSHA to the basal junction of primary microRNAs. RNA N. Y. 2018, 24, 892–898. [Google Scholar] [CrossRef]
- Stork, C.; Zheng, S. Genome-Wide Profiling of RNA-Protein Interactions Using CLIP-Seq. Methods Mol. Biol. 2016, 1421, 137–151. [Google Scholar]
- Yang, Y.-C.T.; Di, C.; Hu, B.; Zhou, M.; Liu, Y.; Song, N.; Li, Y.; Umetsu, J.; Lu, Z.J. CLIPdb: A CLIP-seq database for protein-RNA interactions. BMC Genom. 2015, 16, 51. [Google Scholar] [CrossRef]
- Króliczewski, J.; Sobolewska, A.; Lejnowski, D.; Collawn, J.F.; Bartoszewski, R. microRNA single polynucleotide polymorphism influences on microRNA biogenesis and mRNA target specificity. Gene 2018, 640, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Moszyńska, A.; Gebert, M.; Collawn, J.F.; Bartoszewski, R. SNPs in microRNA target sites and their potential role in human disease. Open Biol. 2017, 7, 170019. [Google Scholar] [CrossRef] [PubMed]
- Roden, C.; Gaillard, J.; Kanoria, S.; Rennie, W.; Barish, S.; Cheng, J.; Pan, W.; Liu, J.; Cotsapas, C.; Ding, Y.; et al. Novel determinants of mammalian primary microRNA processing revealed by systematic evaluation of hairpin-containing transcripts and human genetic variation. Genome Res. 2017, 27, 374–384. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, N.; Cordiner, R.A.; Young, R.S.; Hug, N.; Macias, S.; Cáceres, J.F. Genetic variation and RNA structure regulate microRNA biogenesis. Nat. Commun. 2017, 8, 15114. [Google Scholar] [CrossRef] [PubMed]
- Starega-Roslan, J.; Galka-Marciniak, P.; Krzyzosiak, W.J. Nucleotide sequence of miRNA precursor contributes to cleavage site selection by Dicer. Nucleic Acids Res. 2015, 43, 10939–10951. [Google Scholar] [CrossRef] [PubMed]
- Galka-Marciniak, P.; Olejniczak, M.; Starega-Roslan, J.; Szczesniak, M.W.; Makalowska, I.; Krzyzosiak, W.J. siRNA release from pri-miRNA scaffolds is controlled by the sequence and structure of RNA. Biochim. Biophys. Acta 2016, 1859, 639–649. [Google Scholar] [CrossRef] [PubMed]
- Nowak, J.S.; Hobor, F.; Downie Ruiz Velasco, A.; Choudhury, N.R.; Heikel, G.; Kerr, A.; Ramos, A.; Michlewski, G. Lin28a uses distinct mechanisms of binding to RNA and affects miRNA levels positively and negatively. RNA 2017, 23, 317–332. [Google Scholar] [CrossRef] [PubMed]
- Ustianenko, D.; Chiu, H.-S.; Treiber, T.; Weyn-Vanhentenryck, S.M.; Treiber, N.; Meister, G.; Sumazin, P.; Zhang, C. LIN28 Selectively Modulates a Subclass of Let-7 MicroRNAs. Mol. Cell 2018, 71, 271–283.e5. [Google Scholar] [CrossRef] [PubMed]
- Warrander, F.; Faas, L.; Kovalevskiy, O.; Peters, D.; Coles, M.; Antson, A.A.; Genever, P.; Isaacs, H.V. lin28 proteins promote expression of 17∼92 family miRNAs during amphibian development. Dev. Dyn. 2016, 245, 34–46. [Google Scholar] [CrossRef]
- Berglund, L.; Björling, E.; Oksvold, P.; Fagerberg, L.; Asplund, A.; Szigyarto, C.A.-K.; Persson, A.; Ottosson, J.; Wernérus, H.; Nilsson, P.; et al. A genecentric Human Protein Atlas for expression profiles based on antibodies. Mol. Cell. Proteom. MCP 2008, 7, 2019–2027. [Google Scholar] [CrossRef]
- Panwar, B.; Omenn, G.S.; Guan, Y. miRmine: A database of human miRNA expression profiles. Bioinformatics 2017, 33, 1554–1560. [Google Scholar] [CrossRef] [PubMed]
- Nussbacher, J.K.; Yeo, G.W. Systematic Discovery of RNA Binding Proteins that Regulate MicroRNA Levels. Mol. Cell 2018, 69, 1005–1016.e7. [Google Scholar] [CrossRef] [PubMed]
- Sakaue, S.; Hirata, J.; Maeda, Y.; Kawakami, E.; Nii, T.; Kishikawa, T.; Ishigaki, K.; Terao, C.; Suzuki, K.; Akiyama, M.; et al. Integration of genetics and miRNA-target gene network identified disease biology implicated in tissue specificity. Nucleic Acids Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Piva, F.; Giulietti, M.; Burini, A.B.; Principato, G. SpliceAid 2: A database of human splicing factors expression data and RNA target motifs. Hum. Mutat. 2012, 33, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Kozomara, A.; Griffiths-Jones, S. miRBase: Annotating high confidence microRNAs using deep sequencing data. Nucleic Acids Res. 2014, 42, D68–D73. [Google Scholar] [CrossRef]
- Zuker, M. Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res. 2003, 31, 3406–3415. [Google Scholar] [CrossRef] [PubMed]
- Crooks, G.E.; Hon, G.; Chandonia, J.-M.; Brenner, S.E. WebLogo: A sequence logo generator. Genome Res. 2004, 14, 1188–1190. [Google Scholar] [CrossRef]
- Bailey, T.L. DREME: Motif discovery in transcription factor ChIP-seq data. Bioinforma. Oxf. Engl. 2011, 27, 1653–1659. [Google Scholar] [CrossRef]

| Protein | Motif | Protein Domain Type | Reference | |
|---|---|---|---|---|
| Terminal Loop | hnRNPA1 | UAGGGAW | RRM | [26] |
| HuR | AUUUUUAUUUU | RRM | [27] | |
| KSRP | GGGU | KH | [28] | |
| Lin28 | GGAG | CSD&CCHC-ZnF | [29] | |
| MBNL1 | YGCY | CCCH-ZnF | [30] | |
| MCPIP1 | UGC | CCCH-ZnF | [31] | |
| DGCR8 | UGU | Rhed | [32] | |
| MATR3 | AUCUU | nd | [33] | |
| ZC3H7 | SMTANY | nd | ||
| YBX1 | CAUC | nd | ||
| TRIM71 | UAUAA | nd | ||
| PTBP1/3 | UUUUUCCNUCUUU | nd | ||
| Stem | DDX1 | AA | nd | [34] |
| DDX17 | VCAUCH | DEAD box | [35] | |
| Rbfox | GCAUG | RRM | [36] | |
| SMAD | CAGAC | MH1 | [37] | |
| CELF1/2 | UGUNNNNNNNUGU | nd | [33] | |
| ZC3H10 | GCAGCGC | nd | ||
| SRSF3 | CNNC | RS/RRM | [38] |
| pre-miRNA | |
|---|---|
| hsa-let-7a-1 | hsa-miR-204 |
| hsa-let-7a-2 | hsa-miR-30e |
| hsa-let-7c | hsa-miR-320a |
| hsa-let-7d | hsa-miR-324 |
| hsa-let-7e | hsa-miR-363 |
| hsa-let-7f-1 | hsa-miR-3655 |
| hsa-let-7f-2 | hsa-miR-378i |
| hsa-let-7g | hsa-miR-4286 |
| hsa-let-7i | hsa-miR-4784 |
| hsa-miR-107 | hsa-miR-5006 |
| hsa-miR-1236 | hsa-miR-5579 |
| hsa-miR-132 | hsa-miR-5690 |
| hsa-miR-1323 | hsa-miR-629 |
| hsa-miR-139 | hsa-miR-6775 |
| hsa-miR-142 | hsa-miR-6777 |
| hsa-miR-143 | hsa-miR-6834 |
| hsa-miR-149 | hsa-miR-7154 |
| hsa-miR-152 | hsa-miR-7161 |
| hsa-miR-200b | hsa-miR-7847 |
| hsa-miR-200c | hsa-miR-7975 |
| pre-miRNA | |
|---|---|
| hsa-miR-82 | hsa-miR-410 |
| hsa-miR-96 | hsa-miR-429 |
| hsa-miR-101 | hsa-miR-449a |
| hsa-miR-129 | hsa-miR-487b |
| hsa-miR-138 | hsa-miR-490 |
| hsa-miR-141 | hsa-miR-495 |
| hsa-miR-146b | hsa-miR-499 |
| hsa-miR-155 | hsa-miR-518e |
| hsa-miR-200a | hsa-miR-524 |
| hsa-miR-200b | hsa-miR-539 |
| hsa-miR-200c | hsa-miR-542 |
| hsa-miR-376 | hsa-miR-590 |
| hsa-miR-376a | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urbanek-Trzeciak, M.O.; Jaworska, E.; Krzyzosiak, W.J. miRNAmotif—A Tool for the Prediction of Pre-miRNA–Protein Interactions. Int. J. Mol. Sci. 2018, 19, 4075. https://doi.org/10.3390/ijms19124075
Urbanek-Trzeciak MO, Jaworska E, Krzyzosiak WJ. miRNAmotif—A Tool for the Prediction of Pre-miRNA–Protein Interactions. International Journal of Molecular Sciences. 2018; 19(12):4075. https://doi.org/10.3390/ijms19124075
Chicago/Turabian StyleUrbanek-Trzeciak, Martyna O., Edyta Jaworska, and Wlodzimierz J. Krzyzosiak. 2018. "miRNAmotif—A Tool for the Prediction of Pre-miRNA–Protein Interactions" International Journal of Molecular Sciences 19, no. 12: 4075. https://doi.org/10.3390/ijms19124075
APA StyleUrbanek-Trzeciak, M. O., Jaworska, E., & Krzyzosiak, W. J. (2018). miRNAmotif—A Tool for the Prediction of Pre-miRNA–Protein Interactions. International Journal of Molecular Sciences, 19(12), 4075. https://doi.org/10.3390/ijms19124075





