Childhood-Onset Schizophrenia: Insights from Induced Pluripotent Stem Cells
Abstract
1. Introduction
2. The Neurodevelopmental Hypothesis of COS
3. The Genetic Architecture of AOS and COS
4. iPSCs Provide Unique Access to Early Neurodevelopment in AOS and COS
5. Tracing Early Neurodevelopment in Patients with COS
5.1. Role of the 22q11.2 Microdeletion as Risk Factor for AOS and COS
5.2. Role of the 15q11.2 Microdeletion as Risk Factor for AOS and COS
5.3. Role of the 2p16.3 Microdeletion as Risk Factor for AOS and COS
5.4. Role of the 16p11.2 Microdeletion as Risk Factor for AOS and COS
5.5. COS with or without CNVs
6. Future Perspectives and Challenges
6.1. High Resolution Karyotypes
6.2. Cellular Heterogeneity
6.3. Polygenic Disorders and the Environment
6.4. Organoids—From Structure to Function?
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization Mental Disorders Fact Sheet Schizophrenia 2016. Available online: http://www.who.int/mediacentre/factsheets/fs397/en/ (accessed on 18 September 2018).
- Olfson, M.; Gerhard, T.; Huang, C.; Crystal, S.; Stroup, T.S. Premature Mortality among Adults with Schizophrenia in the United States. JAMA Psychiatry 2015, 72, 1172–1181. [Google Scholar] [CrossRef]
- Thornicroft, G. Physical health disparities and mental illness: The scandal of premature mortality. Br. J. Psychiatry 2011, 199, 441–442. [Google Scholar] [CrossRef] [PubMed]
- McKenna, K.; Gordon, C.T.; Lenane, M.; Kaysen, D.; Fahey, K.; Rapoport, J.L. Looking for childhood-onset schizophrenia: The first 71 cases screened. J. Am. Acad. Child Adolesc. Psychiatry 1994, 33, 636–644. [Google Scholar] [CrossRef] [PubMed]
- Weinberger, D.R.; Harrison, P.J. (Eds.) Schizophrenia, 3rd ed.; Wiley-Blackwell: Chichester, UK; Hoboken, NJ, USA, 2011; ISBN 978-1-4051-7697-2. [Google Scholar]
- Ahn, K.; Gotay, N.; Andersen, T.M.; Anvari, A.A.; Gochman, P.; Lee, Y.; Sanders, S.; Guha, S.; Darvasi, A.; Glessner, J.T.; et al. High rate of disease-related copy number variations in childhood onset schizophrenia. Mol. Psychiatry 2014, 19, 568–572. [Google Scholar] [CrossRef]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131, 861–872. [Google Scholar] [CrossRef] [PubMed]
- Clemmensen, L.; Vernal, D.L.; Steinhausen, H.-C. A systematic review of the long-term outcome of early onset schizophrenia. BMC Psychiatry 2012, 12, 150. [Google Scholar] [CrossRef] [PubMed]
- Shorter, E.; Wachtel, L.E. Childhood catatonia, autism and psychosis past and present: Is there an “iron triangle”? Acta Psychiatr. Scand. 2013, 128, 21–33. [Google Scholar] [CrossRef]
- Mueser, K.T.; Jeste, D.V. Clinical Handbook of Schizophrenia; Guilford Press: New York, NJ, USA, 2008; ISBN 978-1-60623-045-9. [Google Scholar]
- NHS Choices. Schizophrenia—Symptoms. 2018. Available online: https://www.nhs.uk/conditions/schizophrenia/symptoms/ (accessed on 18 September 2018).
- Kinros, J.; Reichenberg, A.; Frangou, S. The neurodevelopmental theory of schizophrenia: Evidence from studies of early onset cases. Isr. J. Psychiatry Relat. Sci. 2010, 47, 110–117. [Google Scholar]
- Hollis, C. Adult outcomes of child- and adolescent-onset schizophrenia: Diagnostic stability and predictive validity. Am. J. Psychiatry 2000, 157, 1652–1659. [Google Scholar] [CrossRef]
- Remschmidt, H.; Martin, M.; Fleischhaker, C.; Theisen, F.M.; Hennighausen, K.; Gutenbrunner, C.; Schulz, E. Forty-two-years later: The outcome of childhood-onset schizophrenia. J. Neural Transm. 2007, 114, 505–512. [Google Scholar] [CrossRef]
- Eggers, C.; Bunk, D. The long-term course of childhood-onset schizophrenia: A 42-year followup. Schizophr. Bull. 1997, 23, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, L.K.; Giedd, J.N.; Castellanos, F.X.; Vaituzis, A.C.; Hamburger, S.D.; Kumra, S.; Lenane, M.C.; Rapoport, J.L. Progressive reduction of temporal lobe structures in childhood-onset schizophrenia. Am. J. Psychiatry 1998, 155, 678–685. [Google Scholar] [CrossRef] [PubMed]
- Rabinowitz, J.; Levine, S.Z.; Häfner, H. A population based elaboration of the role of age of onset on the course of schizophrenia. Schizophr. Res. 2006, 88, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Gornick, M.C.; Addington, A.M.; Sporn, A.; Gogtay, N.; Greenstein, D.; Lenane, M.; Gochman, P.; Ordonez, A.; Balkissoon, R.; Vakkalanka, R.; et al. Dysbindin (DTNBP1, 6p22.3) is associated with childhood-onset psychosis and endophenotypes measured by the Premorbid Adjustment Scale (PAS). J. Autism Dev. Disord. 2005, 35, 831–838. [Google Scholar] [CrossRef] [PubMed]
- Vourdas, A.; Pipe, R.; Corrigall, R.; Frangou, S. Increased developmental deviance and premorbid dysfunction in early onset schizophrenia. Schizophr. Res. 2003, 62, 13–22. [Google Scholar] [CrossRef]
- Hippius, H. The history of clozapine. Psychopharmacology 1989, 99, S3–S5. [Google Scholar] [CrossRef] [PubMed]
- Kasoff, L.I.; Ahn, K.; Gochman, P.; Broadnax, D.D.; Rapoport, J.L. Strong Treatment Response and High Maintenance Rates of Clozapine in Childhood-Onset Schizophrenia. J. Child Adolesc. Psychopharmacol. 2016, 26, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Frazier, J.A.; Giedd, J.N.; Hamburger, S.D.; Albus, K.E.; Kaysen, D.; Vaituzis, A.C.; Rajapakse, J.C.; Lenane, M.C.; McKenna, K.; Jacobsen, L.K.; et al. Brain anatomic magnetic resonance imaging in childhood-onset schizophrenia. Arch. Gen. Psychiatry 1996, 53, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Sporn, A.L.; Greenstein, D.K.; Gogtay, N.; Jeffries, N.O.; Lenane, M.; Gochman, P.; Clasen, L.S.; Blumenthal, J.; Giedd, J.N.; Rapoport, J.L. Progressive brain volume loss during adolescence in childhood-onset schizophrenia. Am. J. Psychiatry 2003, 160, 2181–2189. [Google Scholar] [CrossRef] [PubMed]
- Giedd, J.N.; Jeffries, N.O.; Blumenthal, J.; Castellanos, F.X.; Vaituzis, A.C.; Fernandez, T.; Hamburger, S.D.; Liu, H.; Nelson, J.; Bedwell, J.; et al. Childhood-onset schizophrenia: Progressive brain changes during adolescence. Biol. Psychiatry 1999, 46, 892–898. [Google Scholar] [CrossRef]
- Gogtay, N.; Sporn, A.; Clasen, L.S.; Nugent, T.F.; Greenstein, D.; Nicolson, R.; Giedd, J.N.; Lenane, M.; Gochman, P.; Evans, A.; et al. Comparison of progressive cortical gray matter loss in childhood-onset schizophrenia with that in childhood-onset atypical psychoses. Arch. Gen. Psychiatry 2004, 61, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Gogtay, N.; Greenstein, D.; Lenane, M.; Clasen, L.; Sharp, W.; Gochman, P.; Butler, P.; Evans, A.; Rapoport, J. Cortical brain development in nonpsychotic siblings of patients with childhood-onset schizophrenia. Arch. Gen. Psychiatry 2007, 64, 772–780. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Greenstein, D.; Lerch, J.; Shaw, P.; Clasen, L.; Giedd, J.; Gochman, P.; Rapoport, J.; Gogtay, N. Childhood onset schizophrenia: Cortical brain abnormalities as young adults. J. Child Psychol. Psychiatry 2006, 47, 1003–1012. [Google Scholar] [CrossRef] [PubMed]
- Mattai, A.A.; Weisinger, B.; Greenstein, D.; Stidd, R.; Clasen, L.; Miller, R.; Tossell, J.W.; Rapoport, J.L.; Gogtay, N. Normalization of cortical gray matter deficits in nonpsychotic siblings of patients with childhood-onset schizophrenia. J. Am. Acad. Child Adolesc. Psychiatry 2011, 50, 697–704. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Alexander-Bloch, A.F.; Gogtay, N.; Meunier, D.; Birn, R.; Clasen, L.; Lalonde, F.; Lenroot, R.; Giedd, J.; Bullmore, E.T. Disrupted modularity and local connectivity of brain functional networks in childhood-onset schizophrenia. Front. Syst. Neurosci. 2010, 4, 147. [Google Scholar] [CrossRef] [PubMed]
- Alexander-Bloch, A.F.; Vértes, P.E.; Stidd, R.; Lalonde, F.; Clasen, L.; Rapoport, J.; Giedd, J.; Bullmore, E.T.; Gogtay, N. The anatomical distance of functional connections predicts brain network topology in health and schizophrenia. Cereb. Cortex 2013, 23, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Shi, F.; Yap, P.-T.; Gao, W.; Lin, W.; Gilmore, J.H.; Shen, D. Altered structural connectivity in neonates at genetic risk for schizophrenia: A combined study using morphological and white matter networks. Neuroimage 2012, 62, 1622–1633. [Google Scholar] [CrossRef]
- Jakob, H.; Beckmann, H. Prenatal developmental disturbances in the limbic allocortex in schizophrenics. J. Neural Transm. 1986, 65, 303–326. [Google Scholar] [CrossRef]
- Kovelman, J.A.; Scheibel, A.B. A neurohistological correlate of schizophrenia. Biol. Psychiatry 1984, 19, 1601–1621. [Google Scholar]
- Akbarian, S.; Bunney, W.E.; Potkin, S.G.; Wigal, S.B.; Hagman, J.O.; Sandman, C.A.; Jones, E.G. Altered distribution of nicotinamide-adenine dinucleotide phosphate-diaphorase cells in frontal lobe of schizophrenics implies disturbances of cortical development. Arch. Gen. Psychiatry 1993, 50, 169–177. [Google Scholar] [CrossRef]
- Bakhshi, K.; Chance, S.A. The neuropathology of schizophrenia: A selective review of past studies and emerging themes in brain structure and cytoarchitecture. Neuroscience 2015, 303, 82–102. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, P.; Yip, B.H.; Björk, C.; Pawitan, Y.; Cannon, T.D.; Sullivan, P.F.; Hultman, C.M. Common genetic determinants of schizophrenia and bipolar disorder in Swedish families: A population-based study. Lancet 2009, 373, 234–239. [Google Scholar] [CrossRef]
- Wray, N.R.; Gottesman, I.I. Using summary data from the danish national registers to estimate heritabilities for schizophrenia, bipolar disorder, and major depressive disorder. Front. Genet. 2012, 3, 118. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, P.F.; Kendler, K.S.; Neale, M.C. Schizophrenia as a complex trait: Evidence from a meta-analysis of twin studies. Arch. Gen. Psychiatry 2003, 60, 1187–1192. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, P.F.; Daly, M.J.; O’Donovan, M. Genetic architectures of psychiatric disorders: The emerging picture and its implications. Nat. Rev. Genet. 2012, 13, 537–551. [Google Scholar] [CrossRef] [PubMed]
- Kallmann, F.J.; Roth, B. Genetic aspects of preadolescent schizophrenia. Am. J. Psychiatry 1956, 112, 599–606. [Google Scholar] [CrossRef]
- Asarnow, R.F.; Forsyth, J.K. Genetics of childhood-onset schizophrenia. Child Adolesc. Psychiatr. Clin. N. Am. 2013, 22, 675–687. [Google Scholar] [CrossRef][Green Version]
- Purcell, S.M.; Moran, J.L.; Fromer, M.; Ruderfer, D.; Solovieff, N.; Roussos, P.; O’Dushlaine, C.; Chambert, K.; Bergen, S.E.; Kähler, A.; et al. A polygenic burden of rare disruptive mutations in schizophrenia. Nature 2014, 506, 185–190. [Google Scholar] [CrossRef]
- Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature 2014, 511, 421–427. [Google Scholar] [CrossRef]
- Pardiñas, A.F.; Holmans, P.; Pocklington, A.J.; Escott-Price, V.; Ripke, S.; Carrera, N.; Legge, S.E.; Bishop, S.; Cameron, D.; Hamshere, M.L.; et al. Common schizophrenia alleles are enriched in mutation-intolerant genes and in regions under strong background selection. Nat. Genet. 2018, 50, 381–389. [Google Scholar] [CrossRef]
- Schork, A.J.; Won, H.; Appadurai, V.; Nudel, R.; Gandal, M.; Delaneau, O.; Hougaard, D.; Baekved-Hansen, M.; Bybjerg-Grauholm, J.; Pedersen, M.G. A genome-wide association study for shared risk across major psychiatric disorders in a nation-wide birth cohort implicates fetal neurodevelopment as a key mediator. bioRxiv 2017. [Google Scholar] [CrossRef]
- Freedman, M.L.; Monteiro, A.N.A.; Gayther, S.A.; Coetzee, G.A.; Risch, A.; Plass, C.; Casey, G.; De Biasi, M.; Carlson, C.; Duggan, D.; et al. Principles for the post-GWAS functional characterization of cancer risk loci. Nat. Genet. 2011, 43, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Forrest, M.P.; Zhang, H.; Moy, W.; McGowan, H.; Leites, C.; Dionisio, L.E.; Xu, Z.; Shi, J.; Sanders, A.R.; Greenleaf, W.J.; et al. Open Chromatin Profiling in hiPSC-Derived Neurons Prioritizes Functional Noncoding Psychiatric Risk Variants and Highlights Neurodevelopmental Loci. Cell Stem Cell 2017, 21, 305–318.e8. [Google Scholar] [CrossRef]
- Kirov, G. CNVs in neuropsychiatric disorders. Hum. Mol. Genet. 2015, 24, R45–R49. [Google Scholar] [CrossRef] [PubMed]
- Marshall, C.R.; Howrigan, D.P.; Merico, D.; Thiruvahindrapuram, B.; Wu, W.; Greer, D.S.; Antaki, D.; Shetty, A.; Holmans, P.A.; Pinto, D.; et al. Contribution of copy number variants to schizophrenia from a genome-wide study of 41,321 subjects. Nat. Genet. 2017, 49, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Walsh, T.; McClellan, J.M.; McCarthy, S.E.; Addington, A.M.; Pierce, S.B.; Cooper, G.M.; Nord, A.S.; Kusenda, M.; Malhotra, D.; Bhandari, A.; et al. Rare structural variants disrupt multiple genes in neurodevelopmental pathways in schizophrenia. Science 2008, 320, 539–543. [Google Scholar] [CrossRef]
- Addington, A.M.; Rapoport, J.L. The genetics of childhood-onset schizophrenia: When madness strikes the prepubescent. Curr. Psychiatry Rep. 2009, 11, 156–161. [Google Scholar] [CrossRef]
- Boyle, E.A.; Li, Y.I.; Pritchard, J.K. An Expanded View of Complex Traits: From Polygenic to Omnigenic. Cell 2017, 169, 1177–1186. [Google Scholar] [CrossRef]
- Marín, O. Developmental timing and critical windows for the treatment of psychiatric disorders. Nat. Med. 2016, 22, 1229–1238. [Google Scholar] [CrossRef]
- Kandel, E.R.; Schwartz, J.H.; Jessel, T.M.; Siegelbaum, S.A.; Hudspeth, A.J.; Mack, S. (Eds.) Principles of Neural Science, 15th ed.; McGraw-Hill Medical: New York, NY, USA; Lisbon, Portugal; London, UK, 2013; ISBN 978-0-07-139011-8. [Google Scholar]
- Molyneaux, B.J.; Arlotta, P.; Menezes, J.R.L.; Macklis, J.D. Neuronal subtype specification in the cerebral cortex. Nat. Rev. Neurosci. 2007, 8, 427–437. [Google Scholar] [CrossRef]
- Taverna, E.; Götz, M.; Huttner, W.B. The cell biology of neurogenesis: Toward an understanding of the development and evolution of the neocortex. Annu. Rev. Cell Dev. Biol. 2014, 30, 465–502. [Google Scholar] [CrossRef] [PubMed]
- Lewis, D.A.; González-Burgos, G. Neuroplasticity of neocortical circuits in schizophrenia. Neuropsychopharmacology 2008, 33, 141–165. [Google Scholar] [CrossRef] [PubMed]
- Rakic, P.; Bourgeois, J.P.; Eckenhoff, M.F.; Zecevic, N.; Goldman-Rakic, P.S. Concurrent overproduction of synapses in diverse regions of the primate cerebral cortex. Science 1986, 232, 232–235. [Google Scholar] [CrossRef] [PubMed]
- Turksen, K.; Nagy, A. (Eds.) Induced Pluripotent Stem (iPS) Cells: Methods and Protocols; Methods in Molecular Biology; Humana Press: New York, NY, USA, 2016; ISBN 978-1-4939-3055-5. [Google Scholar]
- Verma, P.J.; Sumer, H. (Eds.) Cell Reprogramming: Methods and Protocols; Methods in Molecular Biology; Humana Press: New York, NY, USA, 2015; ISBN 978-1-4939-2847-7. [Google Scholar]
- Brennand, K.; Savas, J.N.; Kim, Y.; Tran, N.; Simone, A.; Hashimoto-Torii, K.; Beaumont, K.G.; Kim, H.J.; Topol, A.; Ladran, I.; et al. Phenotypic differences in hiPSC NPCs derived from patients with schizophrenia. Mol. Psychiatry 2015, 20, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Burke, E.E.; Chenoweth, J.G.; Shin, J.H.; Collado-Torres, L.; Kim, S.K.; Micali, N.; Wang, Y.; Straub, R.E.; Hoeppner, D.J.; Chen, H.-Y. Dissecting transcriptomic signatures of neuronal differentiation and maturation using iPSCs. bioRxiv 2018. [Google Scholar] [CrossRef]
- Mariani, J.; Simonini, M.V.; Palejev, D.; Tomasini, L.; Coppola, G.; Szekely, A.M.; Horvath, T.L.; Vaccarino, F.M. Modeling human cortical development in vitro using induced pluripotent stem cells. Proc. Natl. Acad. Sci. USA 2012, 109, 12770–12775. [Google Scholar] [CrossRef] [PubMed]
- Nicholas, C.R.; Chen, J.; Tang, Y.; Southwell, D.G.; Chalmers, N.; Vogt, D.; Arnold, C.M.; Chen, Y.-J.J.; Stanley, E.G.; Elefanty, A.G.; et al. Functional maturation of hPSC-derived forebrain interneurons requires an extended timeline and mimics human neural development. Cell Stem Cell 2013, 12, 573–586. [Google Scholar] [CrossRef]
- Paşca, A.M.; Sloan, S.A.; Clarke, L.E.; Tian, Y.; Makinson, C.D.; Huber, N.; Kim, C.H.; Park, J.-Y.; O’Rourke, N.A.; Nguyen, K.D.; et al. Functional cortical neurons and astrocytes from human pluripotent stem cells in 3D culture. Nat. Methods 2015, 12, 671–678. [Google Scholar] [CrossRef] [PubMed]
- Stein, J.L.; de la Torre-Ubieta, L.; Tian, Y.; Parikshak, N.N.; Hernández, I.A.; Marchetto, M.C.; Baker, D.K.; Lu, D.; Hinman, C.R.; Lowe, J.K.; et al. A quantitative framework to evaluate modeling of cortical development by neural stem cells. Neuron 2014, 83, 69–86. [Google Scholar] [CrossRef] [PubMed]
- Brennand, K.J.; Simone, A.; Jou, J.; Gelboin-Burkhart, C.; Tran, N.; Sangar, S.; Li, Y.; Mu, Y.; Chen, G.; Yu, D.; et al. Modelling schizophrenia using human induced pluripotent stem cells. Nature 2011, 473, 221–225. [Google Scholar] [CrossRef]
- Ahmad, R.; Sportelli, V.; Ziller, M.; Spengler, D.; Hoffmann, A. Tracing Early Neurodevelopment in Schizophrenia with Induced Pluripotent Stem Cells. Cells 2018, 7, 140. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.; Sportelli, V.; Ziller, M.; Spengler, D. From the Psychiatrist’s Couch to Induced Pluripotent Stem Cells: Bipolar Disease in a Dish. Int. J. Mol. Sci. 2018, 19, 770. [Google Scholar] [CrossRef] [PubMed]
- Karayiorgou, M.; Simon, T.J.; Gogos, J.A. 22q11.2 microdeletions: Linking DNA structural variation to brain dysfunction and schizophrenia. Nat. Rev. Neurosci. 2010, 11, 402–416. [Google Scholar] [CrossRef] [PubMed]
- Karayiorgou, M.; Morris, M.A.; Morrow, B.; Shprintzen, R.J.; Goldberg, R.; Borrow, J.; Gos, A.; Nestadt, G.; Wolyniec, P.S.; Lasseter, V.K. Schizophrenia susceptibility associated with interstitial deletions of chromosome 22q11. Proc. Natl. Acad. Sci. USA 1995, 92, 7612–7616. [Google Scholar] [CrossRef]
- Bassett, A.S.; Chow, E.W.C. Schizophrenia and 22q11.2 deletion syndrome. Curr. Psychiatry Rep. 2008, 10, 148–157. [Google Scholar] [CrossRef]
- Pedrosa, E.; Sandler, V.; Shah, A.; Carroll, R.; Chang, C.; Rockowitz, S.; Guo, X.; Zheng, D.; Lachman, H.M. Development of patient-specific neurons in schizophrenia using induced pluripotent stem cells. J. Neurogenet. 2011, 25, 88–103. [Google Scholar] [CrossRef]
- Belinsky, G.S.; Rich, M.T.; Sirois, C.L.; Short, S.M.; Pedrosa, E.; Lachman, H.M.; Antic, S.D. Patch-clamp recordings and calcium imaging followed by single-cell PCR reveal the developmental profile of 13 genes in iPSC-derived human neurons. Stem Cell Res. 2014, 12, 101–118. [Google Scholar] [CrossRef]
- Bundo, M.; Toyoshima, M.; Okada, Y.; Akamatsu, W.; Ueda, J.; Nemoto-Miyauchi, T.; Sunaga, F.; Toritsuka, M.; Ikawa, D.; Kakita, A.; et al. Increased l1 retrotransposition in the neuronal genome in schizophrenia. Neuron 2014, 81, 306–313. [Google Scholar] [CrossRef]
- Zhao, D.; Lin, M.; Chen, J.; Pedrosa, E.; Hrabovsky, A.; Fourcade, H.M.; Zheng, D.; Lachman, H.M. MicroRNA Profiling of Neurons Generated Using Induced Pluripotent Stem Cells Derived from Patients with Schizophrenia and Schizoaffective Disorder, and 22q11.2 Del. PLoS ONE 2015, 10, e0132387. [Google Scholar] [CrossRef]
- Lin, M.; Pedrosa, E.; Hrabovsky, A.; Chen, J.; Puliafito, B.R.; Gilbert, S.R.; Zheng, D.; Lachman, H.M. Integrative transcriptome network analysis of iPSC-derived neurons from schizophrenia and schizoaffective disorder patients with 22q11. 2 deletion. BMC Syst. Biol. 2016, 10, 105. [Google Scholar] [CrossRef]
- Toyoshima, M.; Akamatsu, W.; Okada, Y.; Ohnishi, T.; Balan, S.; Hisano, Y.; Iwayama, Y.; Toyota, T.; Matsumoto, T.; Itasaka, N.; et al. Analysis of induced pluripotent stem cells carrying 22q11.2 deletion. Transl. Psychiatry 2016, 6, e934. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.-J.; Nguyen, H.N.; Ursini, G.; Zhang, F.; Kim, N.-S.; Wen, Z.; Makri, G.; Nauen, D.; Shin, J.H.; Park, Y.; et al. Modeling a genetic risk for schizophrenia in iPSCs and mice reveals neural stem cell deficits associated with adherens junctions and polarity. Cell Stem Cell 2014, 15, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Das, D.K.; Tapias, V.; Chowdari, K.V.; Francis, L.; Zhi, Y.; Ghosh, A.; Surti, U.; Tischfield, J.; Sheldon, M.; Moore, J.C. Genetic and morphological features of human iPSC-derived neurons with chromosome 15q11. 2 (BP1-BP2) deletions. Mol. Neuropsychiatry 2015, 1, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Pak, C.; Danko, T.; Zhang, Y.; Aoto, J.; Anderson, G.; Maxeiner, S.; Yi, F.; Wernig, M.; Südhof, T.C. Human Neuropsychiatric Disease Modeling using Conditional Deletion Reveals Synaptic Transmission Defects Caused by Heterozygous Mutations in NRXN1. Cell Stem Cell 2015, 17, 316–328. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, A.; Yadav, S.; Dao, D.Q.; Wu, Z.-Y.; Hokanson, K.C.; Cahill, M.K.; Wiita, A.P.; Jan, Y.-N.; Ullian, E.M.; Weiss, L.A. Cellular Phenotypes in Human iPSC-Derived Neurons from a Genetic Model of Autism Spectrum Disorder. Cell Rep. 2017, 21, 2678–2687. [Google Scholar] [CrossRef] [PubMed]
- Topol, A.; Zhu, S.; Hartley, B.J.; English, J.; Hauberg, M.E.; Tran, N.; Rittenhouse, C.A.; Simone, A.; Ruderfer, D.M.; Johnson, J.; et al. Dysregulation of miRNA-9 in a Subset of Schizophrenia Patient-Derived Neural Progenitor Cells. Cell Rep. 2016, 15, 1024–1036. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Hartley, B.J.; Kurup, P.; Phillips, A.; Topol, A.; Xu, M.; Ononenyi, C.; Foscue, E.; Ho, S.-M.; Baguley, T.D.; et al. Inhibition of STEP61 ameliorates deficits in mouse and hiPSC-based schizophrenia models. Mol. Psychiatry 2018, 23, 271–281. [Google Scholar] [CrossRef]
- Hoffman, G.E.; Hartley, B.J.; Flaherty, E.; Ladran, I.; Gochman, P.; Ruderfer, D.M.; Stahl, E.A.; Rapoport, J.; Sklar, P.; Brennand, K.J. Transcriptional signatures of schizophrenia in hiPSC-derived NPCs and neurons are concordant with post-mortem adult brains. Nat. Commun. 2017, 8, 2225. [Google Scholar] [CrossRef]
- Jun, G.; Flickinger, M.; Hetrick, K.N.; Romm, J.M.; Doheny, K.F.; Abecasis, G.R.; Boehnke, M.; Kang, H.M. Detecting and estimating contamination of human DNA samples in sequencing and array-based genotype data. Am. J. Hum. Genet. 2012, 91, 839–848. [Google Scholar] [CrossRef]
- Muotri, A.R.; Gage, F.H. Generation of neuronal variability and complexity. Nature 2006, 441, 1087–1093. [Google Scholar] [CrossRef]
- Muotri, A.R.; Chu, V.T.; Marchetto, M.C.N.; Deng, W.; Moran, J.V.; Gage, F.H. Somatic mosaicism in neuronal precursor cells mediated by L1 retrotransposition. Nature 2005, 435, 903–910. [Google Scholar] [CrossRef]
- Coufal, N.G.; Garcia-Perez, J.L.; Peng, G.E.; Yeo, G.W.; Mu, Y.; Lovci, M.T.; Morell, M.; O’Shea, K.S.; Moran, J.V.; Gage, F.H. L1 retrotransposition in human neural progenitor cells. Nature 2009, 460, 1127–1131. [Google Scholar] [CrossRef] [PubMed]
- Muotri, A.R.; Marchetto, M.C.N.; Coufal, N.G.; Oefner, R.; Yeo, G.; Nakashima, K.; Gage, F.H. L1 retrotransposition in neurons is modulated by MeCP2. Nature 2010, 468, 443–446. [Google Scholar] [CrossRef] [PubMed]
- Rajman, M.; Schratt, G. MicroRNAs in neural development: From master regulators to fine-tuners. Development 2017, 144, 2310–2322. [Google Scholar] [CrossRef] [PubMed]
- Sun, A.X.; Crabtree, G.R.; Yoo, A.S. MicroRNAs: Regulators of neuronal fate. Curr. Opin. Cell Biol. 2013, 25, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 2009, 136, 215–233. [Google Scholar] [CrossRef]
- O’Connor, R.M.; Gururajan, A.; Dinan, T.G.; Kenny, P.J.; Cryan, J.F. All Roads Lead to the miRNome: miRNAs Have a Central Role in the Molecular Pathophysiology of Psychiatric Disorders. Trends Pharmacol. Sci. 2016, 37, 1029–1044. [Google Scholar] [CrossRef]
- Cross-Disorder Group of the Psychiatric Genomics Consortium Identification of risk loci with shared effects on five major psychiatric disorders: A genome-wide analysis. Lancet 2013, 381, 1371–1379. [CrossRef]
- Ripke, S.; O’Dushlaine, C.; Chambert, K.; Moran, J.L.; Kähler, A.K.; Akterin, S.; Bergen, S.E.; Collins, A.L.; Crowley, J.J.; Fromer, M.; et al. Genome-wide association analysis identifies 13 new risk loci for schizophrenia. Nat. Genet. 2013, 45, 1150–1159. [Google Scholar] [CrossRef]
- Kwon, E.; Wang, W.; Tsai, L.-H. Validation of schizophrenia-associated genes CSMD1, C10orf26, CACNA1C and TCF4 as miR-137 targets. Mol. Psychiatry 2013, 18, 11–12. [Google Scholar] [CrossRef]
- Lett, T.A.; Chakravarty, M.M.; Chakavarty, M.M.; Felsky, D.; Brandl, E.J.; Tiwari, A.K.; Gonçalves, V.F.; Rajji, T.K.; Daskalakis, Z.J.; Meltzer, H.Y.; et al. The genome-wide supported microRNA-137 variant predicts phenotypic heterogeneity within schizophrenia. Mol. Psychiatry 2013, 18, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Petri, R.; Malmevik, J.; Fasching, L.; Åkerblom, M.; Jakobsson, J. miRNAs in brain development. Exp. Cell Res. 2014, 321, 84–89. [Google Scholar] [CrossRef]
- Forstner, A.J.; Degenhardt, F.; Schratt, G.; Nöthen, M.M. MicroRNAs as the cause of schizophrenia in 22q11.2 deletion carriers, and possible implications for idiopathic disease: A mini-review. Front. Mol. Neurosci. 2013, 6, 47. [Google Scholar] [CrossRef] [PubMed]
- Takumi, T.; Tamada, K. CNV biology in neurodevelopmental disorders. Curr. Opin. Neurobiol. 2018, 48, 183–192. [Google Scholar] [CrossRef]
- Cox, D.M.; Butler, M.G. The 15q11.2 BP1-BP2 microdeletion syndrome: A review. Int. J. Mol. Sci. 2015, 16, 4068–4082. [Google Scholar] [CrossRef] [PubMed]
- Stefansson, H.; Meyer-Lindenberg, A.; Steinberg, S.; Magnusdottir, B.; Morgen, K.; Arnarsdottir, S.; Bjornsdottir, G.; Walters, G.B.; Jonsdottir, G.A.; Doyle, O.M.; et al. CNVs conferring risk of autism or schizophrenia affect cognition in controls. Nature 2014, 505, 361–366. [Google Scholar] [CrossRef] [PubMed]
- De Wolf, V.; Brison, N.; Devriendt, K.; Peeters, H. Genetic counseling for susceptibility loci and neurodevelopmental disorders: The del15q11.2 as an example. Am. J. Med. Genet. A 2013, 161A, 2846–2854. [Google Scholar] [CrossRef]
- Abekhoukh, S.; Bardoni, B. CYFIP family proteins between autism and intellectual disability: Links with Fragile X syndrome. Front. Cell. Neurosci. 2014, 8, 81. [Google Scholar] [CrossRef]
- Oguro-Ando, A.; Rosensweig, C.; Herman, E.; Nishimura, Y.; Werling, D.; Bill, B.R.; Berg, J.M.; Gao, F.; Coppola, G.; Abrahams, B.S.; et al. Increased CYFIP1 dosage alters cellular and dendritic morphology and dysregulates mTOR. Mol. Psychiatry 2015, 20, 1069–1078. [Google Scholar] [CrossRef]
- Pathania, M.; Davenport, E.C.; Muir, J.; Sheehan, D.F.; López-Doménech, G.; Kittler, J.T. The autism and schizophrenia associated gene CYFIP1 is critical for the maintenance of dendritic complexity and the stabilization of mature spines. Transl. Psychiatry 2014, 4, e374. [Google Scholar] [CrossRef]
- Forrest, M.P.; Parnell, E.; Penzes, P. Dendritic structural plasticity and neuropsychiatric disease. Nat. Rev. Neurosci. 2018, 19, 215–234. [Google Scholar] [CrossRef] [PubMed]
- Dimitrion, P.; Zhi, Y.; Clayton, D.; Apodaca, G.L.; Wilcox, M.R.; Johnson, J.W.; Nimgaonkar, V.; D’Aiuto, L. Low-Density Neuronal Cultures from Human Induced Pluripotent Stem Cells. Mol. Neuropsychiatry 2017, 3, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Hoeffding, L.K.E.; Hansen, T.; Ingason, A.; Doung, L.; Thygesen, J.H.; Møller, R.S.; Tommerup, N.; Kirov, G.; Rujescu, D.; Larsen, L.A. Sequence analysis of 17 NRXN1 deletions. Am. J. Med. Genet. B 2014, 165, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Südhof, T.C. Synaptic Neurexin Complexes: A Molecular Code for the Logic of Neural Circuits. Cell 2017, 171, 745–769. [Google Scholar] [CrossRef] [PubMed]
- Rees, E.; Walters, J.T.R.; Georgieva, L.; Isles, A.R.; Chambert, K.D.; Richards, A.L.; Mahoney-Davies, G.; Legge, S.E.; Moran, J.L.; McCarroll, S.A.; et al. Analysis of copy number variations at 15 schizophrenia-associated loci. Br. J. Psychiatry 2014, 204, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, A.; Apud, J.A.; Zhang, F.; Decot, H.; Weinberger, D.R.; Law, A.J. Identification of candidate single-nucleotide polymorphisms in NRXN1 related to antipsychotic treatment response in patients with schizophrenia. Neuropsychopharmacology 2014, 39, 2170–2178. [Google Scholar] [CrossRef] [PubMed]
- Dabell, M.P.; Rosenfeld, J.A.; Bader, P.; Escobar, L.F.; El-Khechen, D.; Vallee, S.E.; Dinulos, M.B.P.; Curry, C.; Fisher, J.; Tervo, R.; et al. Investigation of NRXN1 deletions: Clinical and molecular characterization. Am. J. Med. Genet. A 2013, 161A, 717–731. [Google Scholar] [CrossRef]
- Etherton, M.R.; Blaiss, C.A.; Powell, C.M.; Südhof, T.C. Mouse neurexin-1α deletion causes correlated electrophysiological and behavioral changes consistent with cognitive impairments. Proc. Natl. Acad. Sci. USA 2009, 106, 17998. [Google Scholar] [CrossRef]
- Deshpande, A.; Weiss, L.A. Recurrent reciprocal copy number variants: Roles and rules in neurodevelopmental disorders. Dev. Neurobiol. 2018, 78, 519–530. [Google Scholar] [CrossRef]
- Qureshi, A.Y.; Mueller, S.; Snyder, A.Z.; Mukherjee, P.; Berman, J.I.; Roberts, T.P.L.; Nagarajan, S.S.; Spiro, J.E.; Chung, W.K.; Sherr, E.H.; et al. Opposing brain differences in 16p11.2 deletion and duplication carriers. J. Neurosci. 2014, 34, 11199–11211. [Google Scholar] [CrossRef]
- Lin, G.N.; Corominas, R.; Lemmens, I.; Yang, X.; Tavernier, J.; Hill, D.E.; Vidal, M.; Sebat, J.; Iakoucheva, L.M. Spatiotemporal 16p11.2 protein network implicates cortical late mid-fetal brain development and KCTD13-Cul3-RhoA pathway in psychiatric diseases. Neuron 2015, 85, 742–754. [Google Scholar] [CrossRef] [PubMed]
- Golzio, C.; Willer, J.; Talkowski, M.E.; Oh, E.C.; Taniguchi, Y.; Jacquemont, S.; Reymond, A.; Sun, M.; Sawa, A.; Gusella, J.F.; et al. KCTD13 is a major driver of mirrored neuroanatomical phenotypes of the 16p11.2 copy number variant. Nature 2012, 485, 363–367. [Google Scholar] [CrossRef] [PubMed]
- Harrison, P.J.; Weinberger, D.R. Schizophrenia genes, gene expression, and neuropathology: On the matter of their convergence. Mol. Psychiatry 2005, 10, 40–68. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Sun, G.; Li, S.; Shi, Y. A feedback regulatory loop involving microRNA-9 and nuclear receptor TLX in neural stem cell fate determination. Nat. Struct. Mol. Biol. 2009, 16, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Hauberg, M.E.; Roussos, P.; Grove, J.; Børglum, A.D.; Mattheisen, M. Schizophrenia Working Group of the Psychiatric Genomics Consortium Analyzing the Role of MicroRNAs in Schizophrenia in the Context of Common Genetic Risk Variants. JAMA Psychiatry 2016, 73, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Carty, N.C.; Xu, J.; Kurup, P.; Brouillette, J.; Goebel-Goody, S.M.; Austin, D.R.; Yuan, P.; Chen, G.; Correa, P.R.; Haroutunian, V.; et al. The tyrosine phosphatase STEP: Implications in schizophrenia and the molecular mechanism underlying antipsychotic medications. Transl. Psychiatry 2012, 2, e137. [Google Scholar] [CrossRef] [PubMed]
- Mei, L.; Xiong, W.-C. Neuregulin 1 in neural development, synaptic plasticity and schizophrenia. Nat. Rev. Neurosci. 2008, 9, 437–452. [Google Scholar] [CrossRef]
- Coyle, J.T. NMDA receptor and schizophrenia: A brief history. Schizophr. Bull. 2012, 38, 920–926. [Google Scholar] [CrossRef]
- Cross-Disorder Group of the Psychiatric Genomics Consortium; Lee, S.H.; Ripke, S.; Neale, B.M.; Faraone, S.V.; Purcell, S.M.; Perlis, R.H.; Mowry, B.J.; Thapar, A.; Goddard, M.E.; et al. Genetic relationship between five psychiatric disorders estimated from genome-wide SNPs. Nat. Genet. 2013, 45, 984–994. [Google Scholar] [CrossRef]
- Luhmann, H.J.; Fukuda, A.; Kilb, W. Control of cortical neuronal migration by glutamate and GABA. Front. Cell. Neurosci. 2015, 9, 4. [Google Scholar] [CrossRef]
- Schlaeger, T.M.; Daheron, L.; Brickler, T.R.; Entwisle, S.; Chan, K.; Cianci, A.; DeVine, A.; Ettenger, A.; Fitzgerald, K.; Godfrey, M.; et al. A comparison of non-integrating reprogramming methods. Nat. Biotechnol. 2015, 33, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Kwon, E.M.; Connelly, J.P.; Hansen, N.F.; Donovan, F.X.; Winkler, T.; Davis, B.W.; Alkadi, H.; Chandrasekharappa, S.C.; Dunbar, C.E.; Mullikin, J.C.; et al. iPSCs and fibroblast subclones from the same fibroblast population contain comparable levels of sequence variations. Proc. Natl. Acad. Sci. USA 2017, 114, 1964–1969. [Google Scholar] [CrossRef] [PubMed]
- Banovich, N.E.; Li, Y.I.; Raj, A.; Ward, M.C.; Greenside, P.; Calderon, D.; Tung, P.Y.; Burnett, J.E.; Myrthil, M.; Thomas, S.M.; et al. Impact of regulatory variation across human iPSCs and differentiated cells. Genome Res. 2018, 28, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Burrows, C.K.; Banovich, N.E.; Pavlovic, B.J.; Patterson, K.; Gallego Romero, I.; Pritchard, J.K.; Gilad, Y. Genetic Variation, Not Cell Type of Origin, Underlies the Majority of Identifiable Regulatory Differences in iPSCs. PLoS Genet. 2016, 12, e1005793. [Google Scholar] [CrossRef] [PubMed]
- Carcamo-Orive, I.; Hoffman, G.E.; Cundiff, P.; Beckmann, N.D.; D’Souza, S.L.; Knowles, J.W.; Patel, A.; Papatsenko, D.; Abbasi, F.; Reaven, G.M.; et al. Analysis of Transcriptional Variability in a Large Human iPSC Library Reveals Genetic and Non-genetic Determinants of Heterogeneity. Cell Stem Cell 2017, 20, 518–532.e9. [Google Scholar] [CrossRef] [PubMed]
- Kilpinen, H.; Goncalves, A.; Leha, A.; Afzal, V.; Alasoo, K.; Ashford, S.; Bala, S.; Bensaddek, D.; Casale, F.P.; Culley, O.J.; et al. Common genetic variation drives molecular heterogeneity in human iPSCs. Nature 2017, 546, 370–375. [Google Scholar] [CrossRef]
- Rouhani, F.; Kumasaka, N.; de Brito, M.C.; Bradley, A.; Vallier, L.; Gaffney, D. Genetic background drives transcriptional variation in human induced pluripotent stem cells. PLoS Genet. 2014, 10, e1004432. [Google Scholar] [CrossRef]
- Ziller, M.J.; Gu, H.; Müller, F.; Donaghey, J.; Tsai, L.T.-Y.; Kohlbacher, O.; De Jager, P.L.; Rosen, E.D.; Bennett, D.A.; Bernstein, B.E.; et al. Charting a dynamic DNA methylation landscape of the human genome. Nature 2013, 500, 477–481. [Google Scholar] [CrossRef]
- Kyttälä, A.; Moraghebi, R.; Valensisi, C.; Kettunen, J.; Andrus, C.; Pasumarthy, K.K.; Nakanishi, M.; Nishimura, K.; Ohtaka, M.; Weltner, J.; et al. Genetic Variability Overrides the Impact of Parental Cell Type and Determines iPSC Differentiation Potential. Stem Cell Rep. 2016, 6, 200–212. [Google Scholar] [CrossRef]
- Bock, C.; Kiskinis, E.; Verstappen, G.; Gu, H.; Boulting, G.; Smith, Z.D.; Ziller, M.; Croft, G.F.; Amoroso, M.W.; Oakley, D.H.; et al. Reference Maps of human ES and iPS cell variation enable high-throughput characterization of pluripotent cell lines. Cell 2011, 144, 439–452. [Google Scholar] [CrossRef]
- Hu, B.-Y.; Weick, J.P.; Yu, J.; Ma, L.-X.; Zhang, X.-Q.; Thomson, J.A.; Zhang, S.-C. Neural differentiation of human induced pluripotent stem cells follows developmental principles but with variable potency. Proc. Natl. Acad. Sci. USA 2010, 107, 4335–4340. [Google Scholar] [CrossRef] [PubMed]
- Osafune, K.; Caron, L.; Borowiak, M.; Martinez, R.J.; Fitz-Gerald, C.S.; Sato, Y.; Cowan, C.A.; Chien, K.R.; Melton, D.A. Marked differences in differentiation propensity among human embryonic stem cell lines. Nat. Biotechnol. 2008, 26, 313–315. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Xu, J.; Pang, Z.P.; Ge, W.; Kim, K.J.; Blanchi, B.; Chen, C.; Südhof, T.C.; Sun, Y.E. Integrative genomic and functional analyses reveal neuronal subtype differentiation bias in human embryonic stem cell lines. Proc. Natl. Acad. Sci. USA 2007, 104, 13821–13826. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.; Sportelli, V.; Ziller, M.; Spengler, D. Switch-Like Roles for Polycomb Proteins from Neurodevelopment to Neurodegeneration. Epigenomes 2017, 1, 21. [Google Scholar] [CrossRef]
- Schwartzentruber, J.; Foskolou, S.; Kilpinen, H.; Rodrigues, J.; Alasoo, K.; Knights, A.J.; Patel, M.; Goncalves, A.; Ferreira, R.; Benn, C.L.; et al. Molecular and functional variation in iPSC-derived sensory neurons. Nat. Genet. 2018, 50, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Nehme, R.; Zuccaro, E.; Ghosh, S.D.; Li, C.; Sherwood, J.L.; Pietilainen, O.; Barrett, L.E.; Limone, F.; Worringer, K.A.; Kommineni, S.; et al. Combining NGN2 Programming with Developmental Patterning Generates Human Excitatory Neurons with NMDAR-Mediated Synaptic Transmission. Cell Rep. 2018, 23, 2509–2523. [Google Scholar] [CrossRef] [PubMed]
- Sagal, J.; Zhan, X.; Xu, J.; Tilghman, J.; Karuppagounder, S.S.; Chen, L.; Dawson, V.L.; Dawson, T.M.; Laterra, J.; Ying, M. Proneural transcription factor Atoh1 drives highly efficient differentiation of human pluripotent stem cells into dopaminergic neurons. Stem Cells Transl. Med. 2014, 3, 888–898. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Chanda, S.; Marro, S.; Ng, Y.-H.; Janas, J.A.; Haag, D.; Ang, C.E.; Tang, Y.; Flores, Q.; Mall, M.; et al. Generation of pure GABAergic neurons by transcription factor programming. Nat. Methods 2017, 14, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Pak, C.; Han, Y.; Ahlenius, H.; Zhang, Z.; Chanda, S.; Marro, S.; Patzke, C.; Acuna, C.; Covy, J.; et al. Rapid single-step induction of functional neurons from human pluripotent stem cells. Neuron 2013, 78, 785–798. [Google Scholar] [CrossRef] [PubMed]
- Birnbaum, K.D. Power in Numbers: Single-Cell RNA-Seq Strategies to Dissect Complex Tissues. Annu. Rev. Genet. 2018. [Google Scholar] [CrossRef] [PubMed]
- Hoekstra, S.D.; Stringer, S.; Heine, V.M.; Posthuma, D. Genetically-Informed Patient Selection for iPSC Studies of Complex Diseases May Aid in Reducing Cellular Heterogeneity. Front. Cell. Neurosci. 2017, 11, 164. [Google Scholar] [CrossRef] [PubMed]
- Guennewig, B.; Bitar, M.; Obiorah, I.; Hanks, J.; O’Brien, E.A.; Kaczorowski, D.C.; Hurd, Y.L.; Roussos, P.; Brennand, K.J.; Barry, G. THC exposure of human iPSC neurons impacts genes associated with neuropsychiatric disorders. Transl. Psychiatry 2018, 8, 89. [Google Scholar] [CrossRef] [PubMed]
- Ksir, C.; Hart, C.L. Cannabis and Psychosis: A Critical Overview of the Relationship. Curr. Psychiatry Rep. 2016, 18, 12. [Google Scholar] [CrossRef] [PubMed]
- Honey, C.J.; Kötter, R.; Breakspear, M.; Sporns, O. Network structure of cerebral cortex shapes functional connectivity on multiple time scales. Proc. Natl. Acad. Sci. USA 2007, 104, 10240–10245. [Google Scholar] [CrossRef] [PubMed]
- Arlotta, P. Organoids required! A new path to understanding human brain development and disease. Nat. Methods 2018, 15, 27. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.; Quadrato, G.; Arlotta, P. Studying the Brain in a Dish: 3D Cell Culture Models of Human Brain Development and Disease. Curr. Top. Dev. Biol. 2018, 129, 99–122. [Google Scholar] [CrossRef] [PubMed]
- Real, R.; Peter, M.; Trabalza, A.; Khan, S.; Smith, M.A.; Dopp, J.; Barnes, S.J.; Momoh, A.; Strano, A.; Volpi, E.; et al. In vivo modeling of human neuron dynamics and Down syndrome. Science 2018. [Google Scholar] [CrossRef]
- Wainger, B.J.; Kiskinis, E.; Mellin, C.; Wiskow, O.; Han, S.S.W.; Sandoe, J.; Perez, N.P.; Williams, L.A.; Lee, S.; Boulting, G.; et al. Intrinsic membrane hyperexcitability of amyotrophic lateral sclerosis patient-derived motor neurons. Cell Rep. 2014, 7, 1–11. [Google Scholar] [CrossRef]
- Gandal, M.J.; Leppa, V.; Won, H.; Parikshak, N.N.; Geschwind, D.H. The road to precision psychiatry: Translating genetics into disease mechanisms. Nat. Neurosci. 2016, 19, 1397–1407. [Google Scholar] [CrossRef]
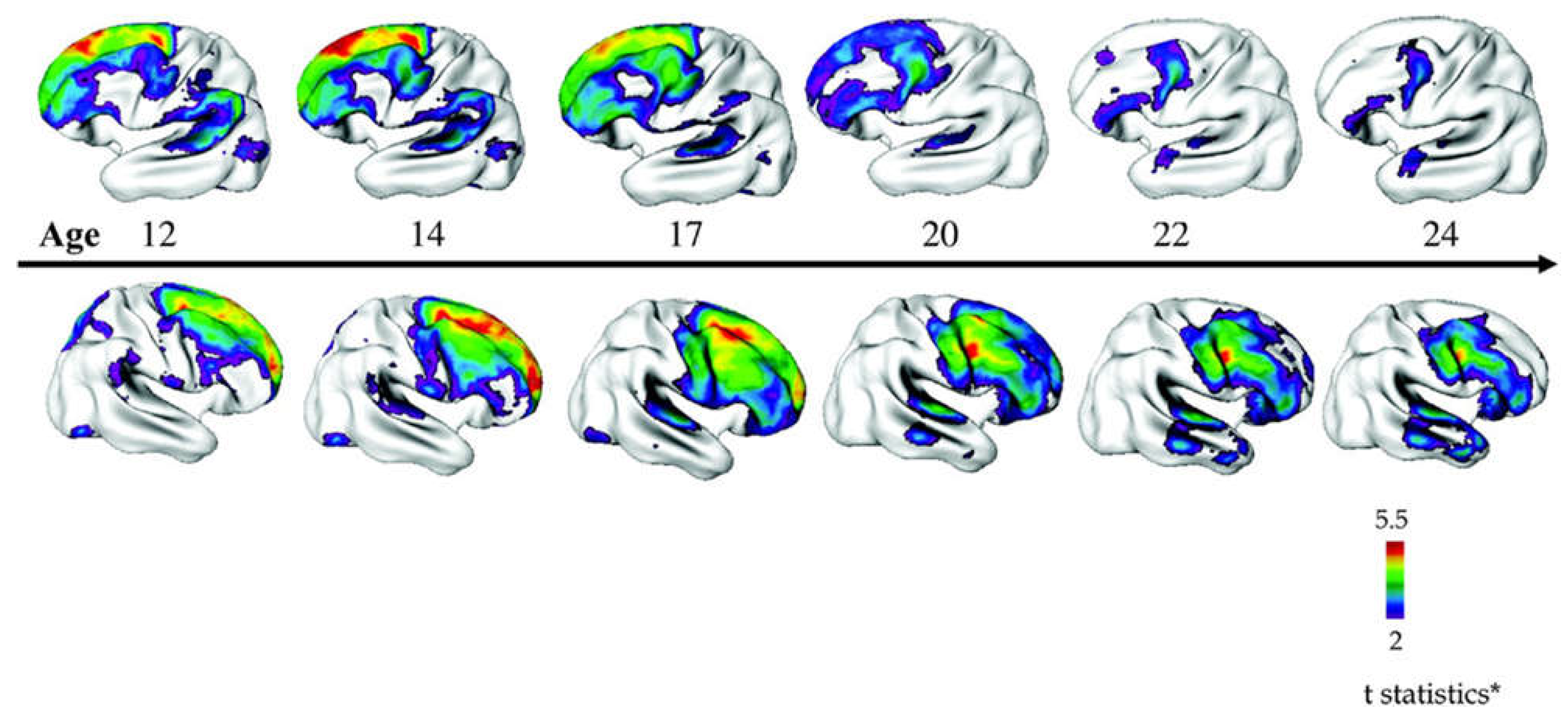
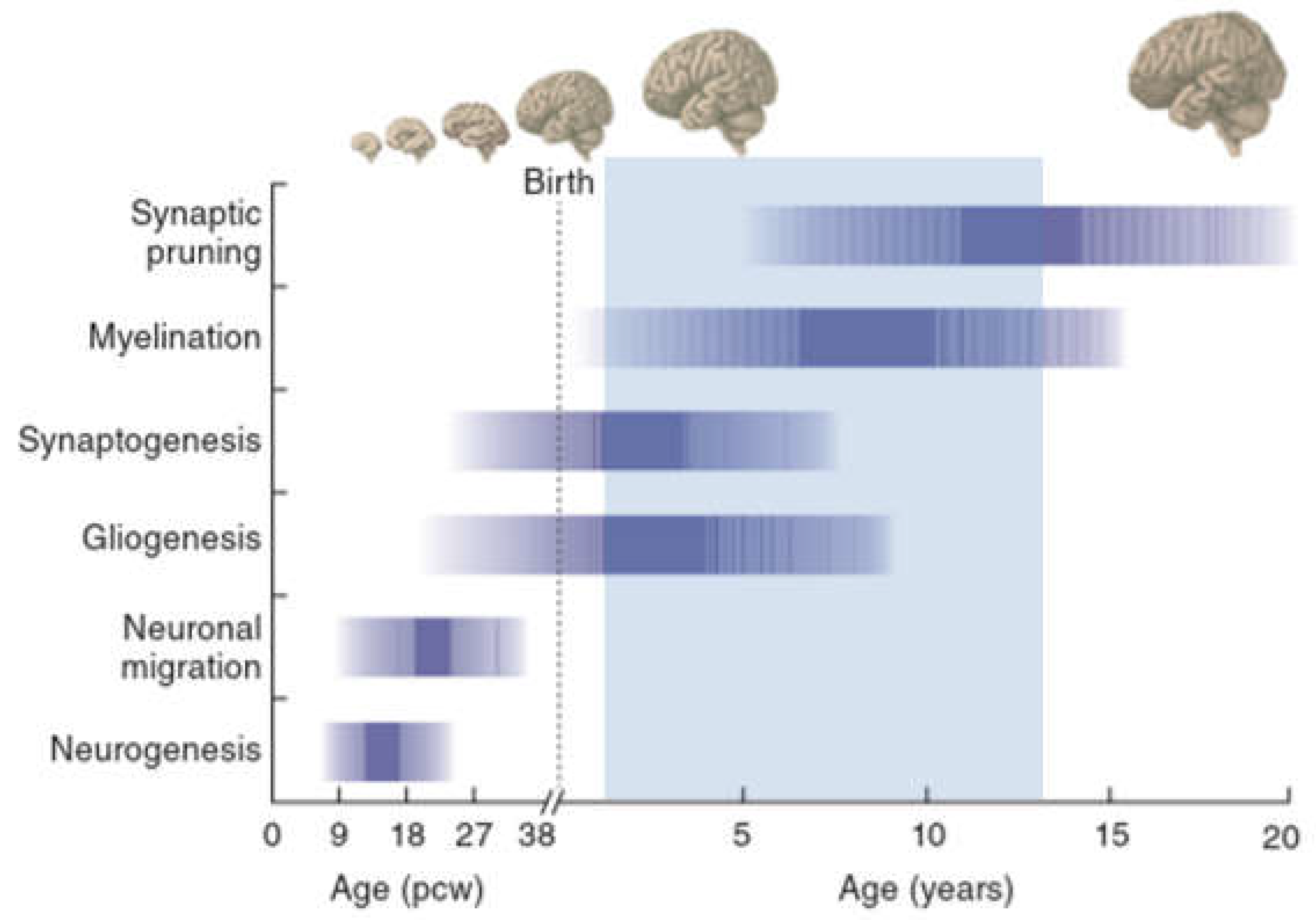
| Chr | Locus | Mechanism | CNV | Effect | OR (95% CI) | COS |
|---|---|---|---|---|---|---|
| 1 | 1q21.1 | NAHR | Loss + gain | Risk | 3.8 (2.1–6.9) | |
| 2 | 2p16.3 (NRXN1) | NHEJ | Loss | Risk | 14.4 (4.2–46.9) | + |
| 3 | 3q29 | NAHR | Loss | Risk | Infinite | + |
| 7 | 7p36.3 | NAHR | Loss + gain | Risk | 3.5 (1.3–9.0) | |
| 7 | 7q11.21 | NAHR | Loss + gain | Protective | 0.66 (0.52–0.84) | |
| 7 | 7q11.23 | NAHR | Gain | Risk | 16.1 (3.1–125.7) | |
| 8 | 8q22.2 | NHEJ | Loss | Risk | 14.5 (1.7–122.1) | |
| 9 | 9p24.3 | NHEJ | Loss + gain | Risk | 12.4 (1.6–98.1) | |
| 13 | 13q12.11 | NAHR | Gain | Protective | 0.36 (0.19–0.67) | |
| 15 | 15q11.2 | NAHR | Loss | Risk | 1.8 (1.2–2.6) | + |
| 15 | 15q13.3 | NAHR | Loss | Risk | 15.6 (3.7–66.5) | + |
| 16 | 16p11.2. proximal | NAHR | Gain | Risk | 9.4 (4.2–20.9) | |
| 16 | 16p11.2. distal | NAHR | Loss | Risk | 20.6 (2.6–162.2) | + |
| 22 | 22q11.21 | NAHR | Loss | Risk | 67.7 (9.3–492.8) | + |
| 22 | 22q11.21 | NAHR | Gain | Protective | 0.15 (0.04–0.52) | |
| X | Xq28 | NAHR | Gain | Protective | 0.35 (0.18–0.68) | |
| X | Xq28. distal | NAHR | Gain | Risk | 8.9 (2.0–39.9) |
| Ref | Source | Factors | Method | n | Authentication | Karyotype | Pluripotency |
|---|---|---|---|---|---|---|---|
| [73] | Fibroblast | OKSM | RV | - | - | G-B, F | ICC, EB |
| [74] | As in [73] | OKSM | RV | - | - | G-B, F | ICC, EB |
| [75] | Fibroblast | OKSM | RV | - | - | CGH | ICC, Tera, EB |
| [76] | Fibroblast | OKSML | Epi | add | - | G-B, F | ICC, EB |
| [77] | As in [76] | OKSML | Epi | add | - | G-B, F, micro | ICC, EB |
| [78] | As in [75] | OKSM | RV | add | - | CGH, Taq | ICC, Tera, EB |
| [79] | Fibroblast | OKSML | Epi or Sen | - | G-B, F | ICC, Tera | |
| [80] | Fibroblast | OKSM | Sen | add | - | G-B, CGH | ICC |
| [81] | hESC (H1) | na | na | - | - | na | na |
| [82] | Fibroblast | OKSML | Epi | na | CytoChip SNP | CGH, SNP | ICC |
| [83] | Fibroblast | OKSM | Sen | 2–3 | PsychChip SNP | G-B | FACS, PCR |
| [84] | As in [83] | OKSM | Sen | 2–3 | PsychChip SNP | G-B | FACS, PCR |
| [85] | As in [83] | OKSM | Sen | 2–3 | Verif-BamID [86] | G-B | FACS, PCR |
| Ref | Case/Control | Deletion | Model | Major Cell Type |
|---|---|---|---|---|
| [73] | AOS (n = 1) | 22q11.2 | iPSC | Forebrain glutamatergic neurons |
| AOS (n =1) COS (n = 1) Ctr (n = 2) | - - - | |||
| [74] | AOS (n = 1) [73] | 22q11.2 | iPSC | Early post-mitotic neurons |
| Ctr (n = 1) [73] | - | |||
| [75] | AOS (n = 2) | 22q11.2 | iPSC | Mixed early neuronal and glial cell types |
| Ctr (n = 2) | - | |||
| [76] | AOS (n = 1) | 22q11.2 | iPSC | Mixed early glutamatergic and GABAergic neurons |
| SAD (n = 3) | 22q11.2 | |||
| COS (n = 2) | - | |||
| Ctr (n = 6) | - | |||
| [77] | As in [76] | As in [76] | iPSC | As in [76] |
| + COS (n = 2) | - | |||
| + Ctr (n = 1) | - | |||
| [78] | As in [75] | As in [75] | iPSC | As in [75] |
| + Ctr (n = 1) | - | |||
| [79] | COS (n = 3) | 15q11.2 | iPSC | Rosette-derived cortical NPCs |
| Ctr (n = 5) | - | |||
| [80] | SAD (n = 1) | 15q11.2 | iPSC | Rosette derived neurons |
| Mother (n = 1) | 15q11.2 | |||
| Ctr (n = 1) | -- | |||
| [81] | Isogenic hESCs | Mutated heterogeneous | hESC | Induced glutamatergic neurons, mixed forebrain neurons |
| NRXN1 alleles | ||||
| [82] | ASD (n = 1) | 16p11.2 dup, de novo | iPSC | NPCs, dorsal forebrain neurons, up to 14 weeks maturated |
| NSD (n = 1) | 16p11.2 dup, de novo | |||
| NSD (n = 1) | 16p11.2 dup, inherited | |||
| Autism (n = 1) | 16p11.2 del, de novo | |||
| Autism (n = 1) | 16p11.2 del, unknown | |||
| Autism (n = 1) | 16p11.2 del, inherited | |||
| Ctr (n = 4) | - | |||
| [83] | COS-1 (n = 1) | 1p33 | iPSC | NPCs |
| COS-2 (n = 1) | 2p16.3 del (NRXN1) | |||
| COS-3 (n = 1) | 3p25.3 | |||
| COS-4 (n = 2) | 16p11.2 | |||
| COS-5 (n = 1) | 22q11.2 | |||
| COS-6 (n = 4) | - | |||
| Ctr (n = 10) | - | |||
| [84] | COS-1 to 4 (n = 5) | As in [83] | iPSC | NPCs, mixed glutamatergic and GABAergic forebrain neurons, Ngn2-induced excitatory neurons |
| COS-6 (n = 4) | As in [83] | |||
| Ctr (n = 8) | As in [83] | |||
| [85] | COS-1 to 5 (n = 6) | As in [83] | NPCs, mixed glutamatergic and GABAergic forebrain neurons | |
| COS-7 (n = 1) | 18q22.1 | |||
| COS-8 (n = 1) | 8q12.3, 22q11 | |||
| COS-9 (n = 1) | 15q11.2, 2p25.3 | |||
| COS-6 (n = 4) | As in [83] | |||
| COS-10 (n = 3) | - | |||
| Ctr (n = 10) | As in [83] | |||
| Ctr (n = 2) | - |
| Ref | Neural Induction | Patterning/Neural Progenitor Cells→Neural Cells |
|---|---|---|
| [73] | EB-/rosette formation | N2, WNT3A→N2, B27, BDNF, GDNF, IGF1, WNT3, cAMP |
| [74] | SB431542 + Dorsomorphin | N2, B27, bFGF→N2, B27, BDNF, GDNF |
| [75] | EB-formation + Noggin | FGF2, Shh or Wnt3a or BMP4→FGF2, EGF |
| [76] | EB-formation + Dorsomorphin | FGF2→N2, BDNF, GDNF, IGF1, WNT3, cAMP |
| [77] | As in [76] | As in [76] |
| [78] | As in [75] | As in [75] |
| [79] | SB431542 + CHIR99204 | N2, B27, Dorsomorphin, RA |
| [80] | Rosette formation, N2, bFGF | N2, BDNF |
| [81] | Ngn2-mediated iN | N2, B27, BDNF, NT3→mouse glia, Ara-C |
| [82] | EB-/rosette formation | StemCell Induction medium™→as above |
| [83] | SB431542 + LDN-193189 | N2, mTeSR™→BDNF, cAMP, AA→BrainPhys™ |
| [84] | SB431542 + LDN-193189 | N2, B27-RA, FGF2 |
| SB431542 + LDN-193189 | N2, B27-RA, FGF2→B27-RA, BDNF, GDNF, cAMP, Ara-C, astrocytes, N2 B27-RA | |
| [85] | Ngn2-mediated iN | BDNF, GDNF, cAMP, Ara-C, astrocytes |
| SB431542 + LDN-193189 | N2, B27-RA, FGF2→B27-RA, BDNF, GDNF, cAMP, Ara-C, astrocytes, N2 |
| Ref | Major Methods | Major Findings on COS and Associated CNVs |
|---|---|---|
| [73] | Microarray, WCPC | Delayed decline of pluripotency markers in AOS with 22q11.2 |
| [74] | WCPC, single cell Ca2+ imaging and PCR | Dysregulation of genes relevant to GABAergic, glutamatergic, and dopaminergic in electrical active neurons |
| [75] | Whole genome sequencing, postmortem brain | Increased L1 retrotransposition in postmortem brain from patients with AOS and iPSC derived neurons from AOS patients with 22q11.2 deletion |
| [76] | MicroRNA profiling | 32 miRNAs are upregulated in neurons with 22q11.2 microdeletion, miRNA deregulation is broadly shared across AOS, SAD, and COS |
| [77] | Paired-end mRNA sequencing | Perturbed neuronal MAPK signaling, differentially expressed genes from the 22q11.2 microdeletion act during critical periods of development |
| [78] | miRNA and mRNA arrays | Reduced neurosphere size, neural differentiation, neurite outgrowth, cellular migration, and expression of miR-17/92 cluster and miR-106a/b that inhibit p38a (MAPK14) expression, p38 inhibitors improve diminished neurogenic-to-gliogenic ratio |
| [79] | ICC/IHC, complementation and knock-down experiments | Defects in adherens junctions and apical polarity. Displacement of radial glia cells leads to cortical malformation during mouse development |
| [80] | ICC, IB | Lower expression of CYFIP1 and PSD-95, altered dendritic morphology |
| [81] | Gene editing, iNeurons, electrophysiology | Reduced spontaneous mEPSC frequency, but not amplitude, and decrease in evoked EPSC amplitude. Unaltered electrical properties of human neurons, synapse numbers, and dendritic arborization |
| [82] | Histomorphology, electro-physiology | 16p del- and 16p dup-derived NPCs show opposing differences in soma size and arborization, reduced excitability in 16p del-derived neurons, increased potassium current density in 16p dup-derived neurons, lower density of excitatory synapses in 16p del- and 16p dup-derived neurons associates with increased amplitude of mEPSCs |
| [83] | digital miRNA profiling | Downregulation of miR-9, a regulator of neurogenesis and of radial migration |
| [84] | IB, IHC, IP, knock-down | Increased STEP61 protein expression in forebrain neurons impairs NMDAR signaling |
| [85] | mRNA sequencing | Transcriptional signatures of NPCs and neurons show concordance with postmortem case/control brain samples from SCZ, BP, and ASD after adjusting for cell type composition |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hoffmann, A.; Ziller, M.; Spengler, D. Childhood-Onset Schizophrenia: Insights from Induced Pluripotent Stem Cells. Int. J. Mol. Sci. 2018, 19, 3829. https://doi.org/10.3390/ijms19123829
Hoffmann A, Ziller M, Spengler D. Childhood-Onset Schizophrenia: Insights from Induced Pluripotent Stem Cells. International Journal of Molecular Sciences. 2018; 19(12):3829. https://doi.org/10.3390/ijms19123829
Chicago/Turabian StyleHoffmann, Anke, Michael Ziller, and Dietmar Spengler. 2018. "Childhood-Onset Schizophrenia: Insights from Induced Pluripotent Stem Cells" International Journal of Molecular Sciences 19, no. 12: 3829. https://doi.org/10.3390/ijms19123829
APA StyleHoffmann, A., Ziller, M., & Spengler, D. (2018). Childhood-Onset Schizophrenia: Insights from Induced Pluripotent Stem Cells. International Journal of Molecular Sciences, 19(12), 3829. https://doi.org/10.3390/ijms19123829




