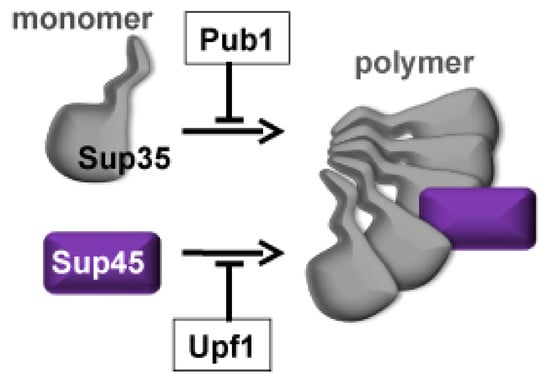
The Pub1 and Upf1 Proteins Act in Concert to Protect Yeast from Toxicity of the [PSI+] Prion
Abstract
1. Introduction
2. Results
2.1. Simultaneous Deletion of PUB1 and UPF1 in the Presence of [PSI+] Can Be Synthetic Lethal
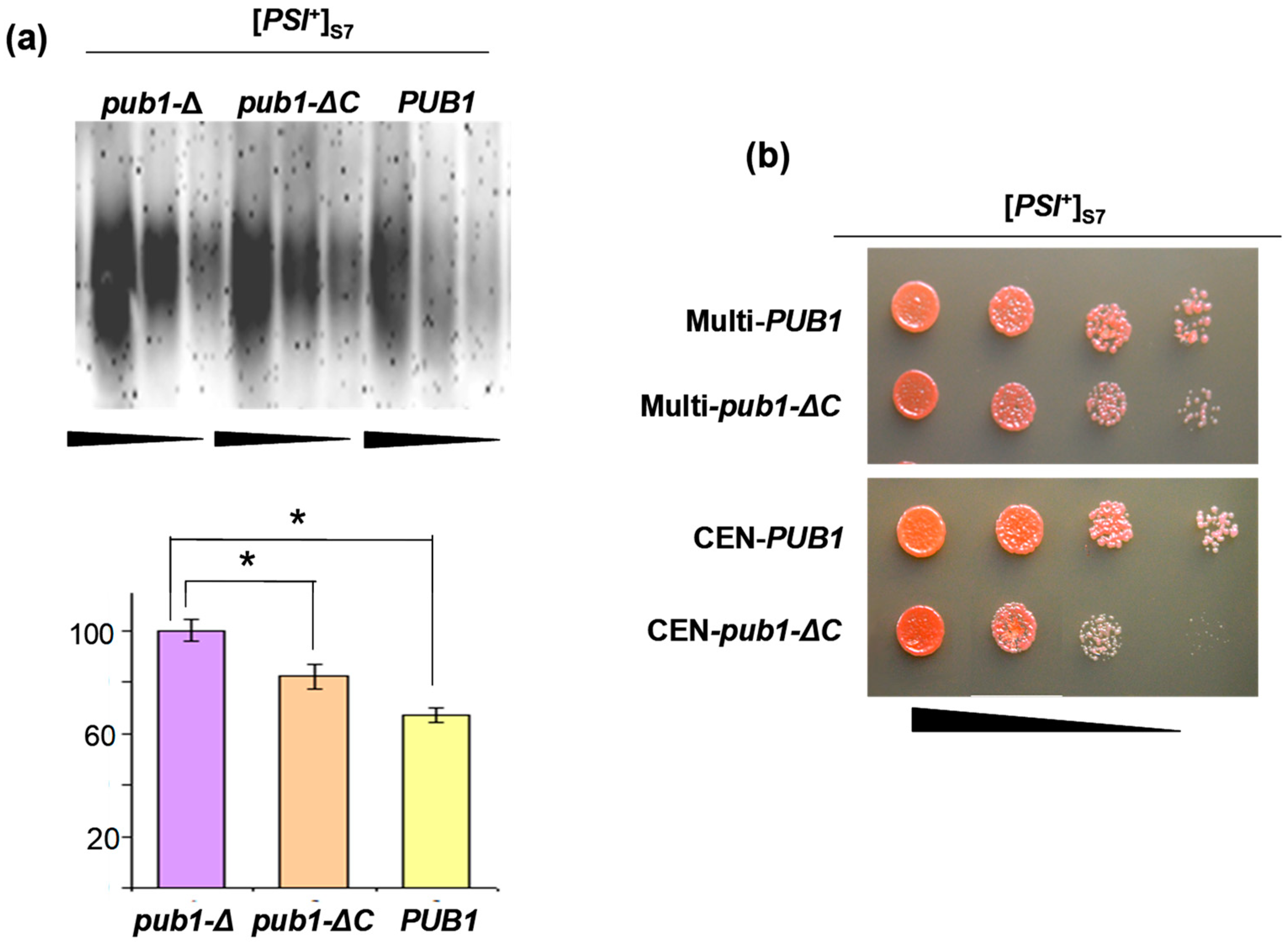
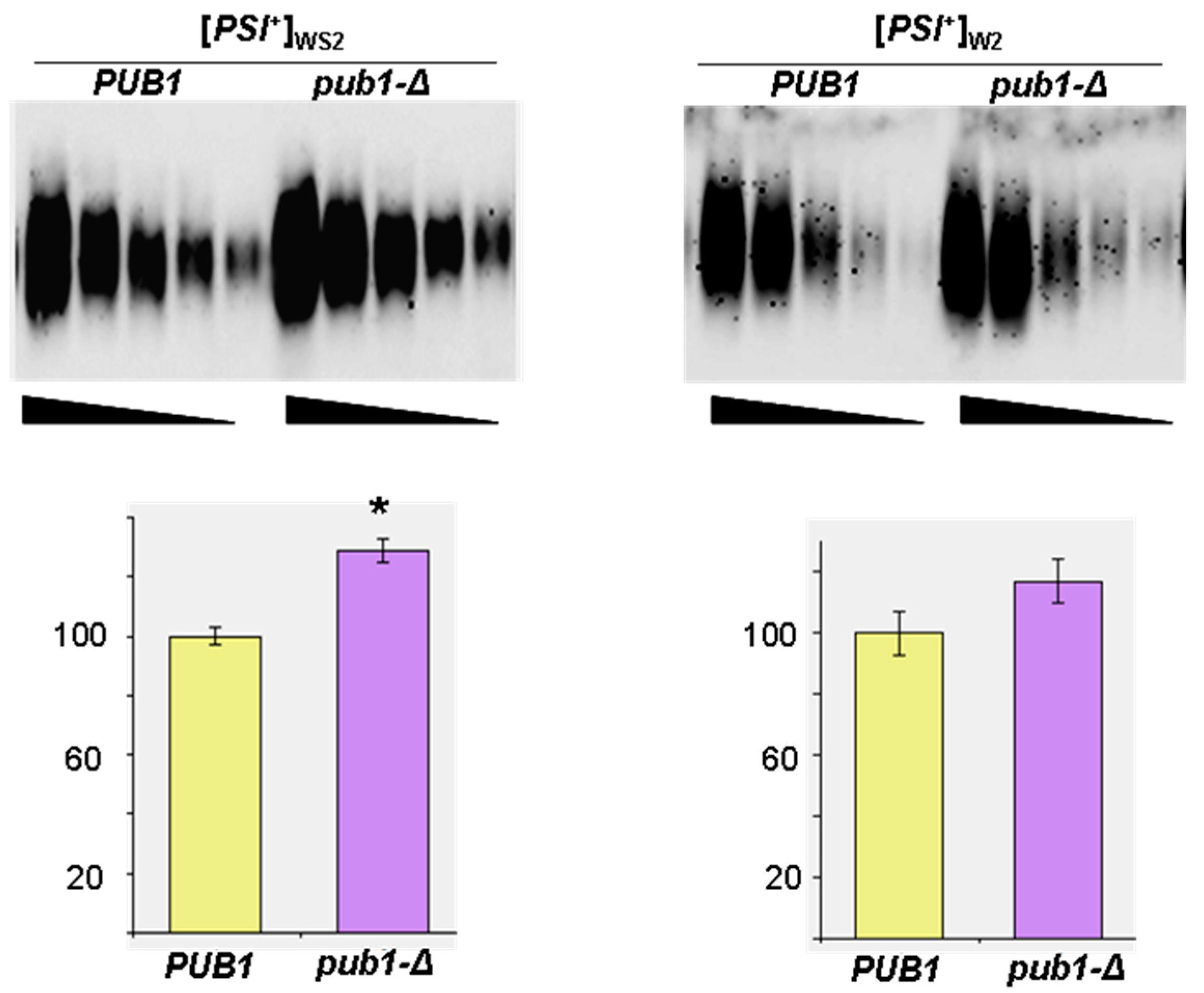
2.2. The Lack of Pub1 But Not of Upf1 Increases Prion Polymerization of Sup35
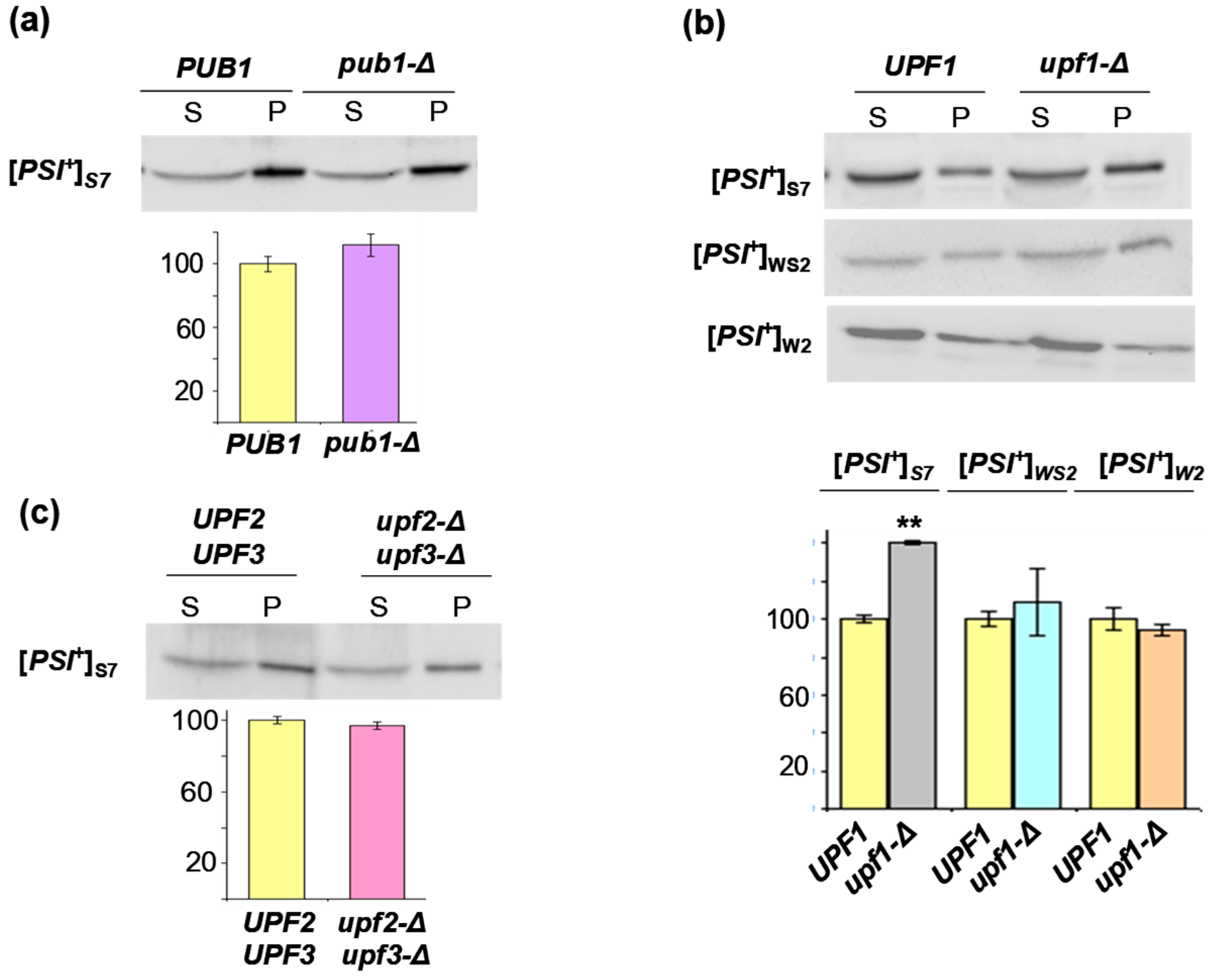
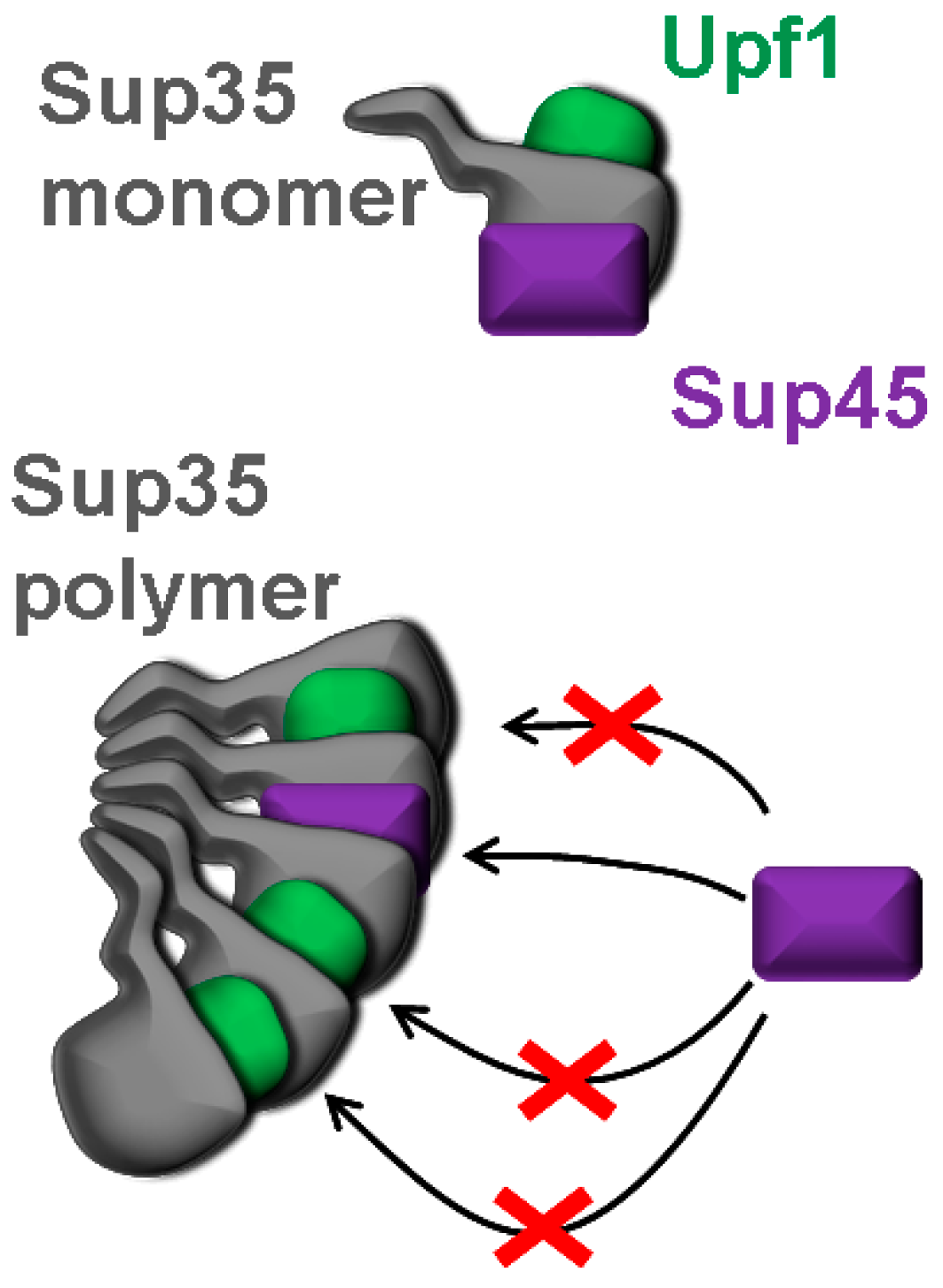
2.3. The Lack of Upf1, But Not of Pub1, Upf2 or Upf3 Increases Sequestration of Sup45 into Sup35 Prion Polymers
3. Discussion
4. Materials and Methods
4.1. Yeast Strains and Growth Conditions
4.2. Plasmids and Nucleic Acid Manipulation
4.3. Electrophoresis and Blotting
4.4. Preparation and Fractionation of Yeast Cell Lysates
4.5. Determination of the Efficiency of Nonsense Codon Readthrough
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chiti, F.; Dobson, C.M. Protein misfolding, functional amyloid and human disease. Annu. Rev. Biochem. 2006, 75, 333–366. [Google Scholar] [CrossRef] [PubMed]
- Nizhnikov, A.A.; Antonets, K.S.; Inge-Vechtomov, S.G. Amyloids: From pathogenesis to function. Biochemistry 2015, 80, 1127–1144. [Google Scholar] [CrossRef] [PubMed]
- Shkundina, I.S.; Ter-Avanesyan, M.D. Prions. Biochemistry 2007, 72, 1519–1536. [Google Scholar] [CrossRef] [PubMed]
- Wickner, R.B. Yeast and fungal prions. Cold Spring Harb. Perspect. Biol. 2016, 8, a023531. [Google Scholar] [CrossRef] [PubMed]
- Paushkin, S.V.; Kushnirov, V.V.; Smirnov, V.N.; Ter-Avanesyan, M.D. Propagation of the yeast prion-like [psi+] determinant is mediated by oligomerization of the SUP35-encoded polypeptide chain release factor. EMBO J. 1996, 15, 3127–3134. [Google Scholar] [CrossRef] [PubMed]
- Patino, M.M.; Liu, J.J.; Glover, J.R.; Lindquist, S. Support for the prion hypothesis for inheritance of a phenotypic trait in yeast. Science 1996, 273, 622–626. [Google Scholar] [CrossRef] [PubMed]
- Kryndushkin, D.S.; Alexandrov, I.M.; Ter-Avanesyan, M.D.; Kushnirov, V.V. Yeast [PSI+] prion aggregates are formed by small Sup35 polymers fragmented by Hsp104. J. Biol. Chem. 2003, 278, 49636–49643. [Google Scholar] [CrossRef] [PubMed]
- Derkatch, I.L.; Chernoff, Y.O.; Kushnirov, V.V.; Inge-Vechtomov, S.G.; Liebman, S.W. Genesis and variability of [PSI+] prion factors in Saccharomyces cerevisiae. Genetics 1996, 144, 1375–1386. [Google Scholar] [PubMed]
- Kochneva-Pervukhova, N.V.; Chechenova, M.B.; Valouev, I.A.; Kushnirov, V.V.; Smirnov, V.N.; Ter-Avanesyan, M.D. [PSI+] prion generation in yeast: Characterization of the ‘strain’ difference. Yeast 2001, 18, 489–497. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Chien, P.; Naber, N.; Cooke, R.; Weissman, J.S. Conformational variations in an infectious protein determine prion strain differences. Nature 2004, 428, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, R.; Lindquist, S.L. Structural insights into a yeast prion illuminate nucleation and strain diversity. Nature 2005, 435, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Reidy, M.; Masison, D.C. Modulation and elimination of yeast prions by protein chaperones and co-chaperones. Prion 2011, 5, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Ganusova, E.E.; Ozolins, L.N.; Bhagat, S.; Newnam, G.P.; Wegrzyn, R.D.; Sherman, M.Y.; Chernoff, Y.O. Modulation of prion formation, aggregation, and toxicity by the actin cytoskeleton in yeast. Mol. Cell. Biol. 2006, 26, 617–629. [Google Scholar] [CrossRef] [PubMed]
- Derkatch, I.L.; Bradley, M.E.; Liebman, S.W. Overexpression of the SUP45 gene encoding a Sup35p-binding protein inhibits the induction of the de novo appearance of the [PSI+] prion. Proc. Natl. Acad. Sci. USA 1998, 95, 2400–2405. [Google Scholar] [CrossRef] [PubMed]
- Dagkesamanskaya, A.R.; Ter-Avanesyan, M.D. Interaction of the yeast omnipotent suppressors SUP1(SUP45) and SUP2(SUP35) with non-Mendelian factors. Genetics 1991, 128, 513–520. [Google Scholar] [PubMed]
- Kiktev, D.; Inge-Vechtomov, S.; Zhouravleva, G. Prion-dependent lethality of sup45 mutants in Saccharomyces cerevisiae. Prion 2007, 1, 136–143. [Google Scholar] [CrossRef] [PubMed]
- True, H.L.; Lindquist, S.L. A yeast prion provides a mechanism for genetic variation and phenotypic diversity. Nature 2000, 407, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Tyedmers, J.; Madariaga, M.L.; Lindquist, S. Prion switching in response to environmental stress. PLoS Biol. 2008, 6, e294. [Google Scholar] [CrossRef] [PubMed]
- Wickner, R.B.; Shewmaker, F.P.; Bateman, D.A.; Edskes, H.K.; Gorkovskiy, A.; Dayani, Y.; Bezsonov, E.E. Yeast prions: Structure, biology, and prion-handling systems. Microbiol. Mol. Biol. Rev. 2015, 79, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Mcglinchey, R.P.; Kryndushkin, D.; Wickner, R.B. Suicidal [PSI+] is a lethal yeast prion. Proc. Natl. Acad. Sci. USA 2011, 108, 5337–5341. [Google Scholar] [CrossRef] [PubMed]
- Wickner, R.B.; Bezsonov, E.E.; Son, M.; Ducatez, M.; Dewilde, M.; Edskes, H.K. Anti–prion systems in yeast and inositol polyphosphates. Biochemistry 2018, 57, 1285–1292. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, A.; Hosoda, N.; Tanaka, A.; Newnam, G.P.; Chernoff, Y.O.; Hoshino, S.I. Proteolysis suppresses spontaneous prion generation in yeast. J. Biol. Chem. 2017, 292, 20113–20124. [Google Scholar] [CrossRef] [PubMed]
- Urakov, V.N.; Mitkevich, O.V.; Safenkova, I.V.; Ter-Avanesyan, M.D. Ribosome-bound Pub1 modulates stop codon decoding during translation termination in yeast. FEBS J. 2017, 284, 1914–1930. [Google Scholar] [CrossRef] [PubMed]
- Czaplinski, K.; Ruiz-Echevarria, M.J.; Paushkin, S.V.; Han, X.; Weng, Y.; Perlick, H.A.; Dietz, H.C.; Ter-Avanesyan, M.D.; Peltz, S.W. The surveillance complex interacts with the translation release factors to enhance termination and degrade aberrant mRNAs. Genes Dev. 1998, 12, 1665–1677. [Google Scholar] [CrossRef] [PubMed]
- Paushkin, S.V.; Kushnirov, V.V.; Smirnov, V.N.; Ter-Avanesyan, M.D. Interaction between yeast Sup45p (eRF1) and Sup35p (eRF3) polypeptide chain release factors: Implications for prion-dependent regulation. Mol. Cell. Biol. 1997, 17, 2798–2805. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Broun, A.H.; Jacobson, A. Upf1p, Nmd2p, and Upf3p are interacting components of the yeast nonsense-mediated mRNA decay pathway. Mol. Cell. Biol. 1997, 17, 1580–1594. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Czaplinski, K.; Rao, Y.; Peltz, S.W. The role of Upf proteins in modulating the translation readthrough of nonsense-containing transcripts. EMBO J. 2001, 20, 880–890. [Google Scholar] [CrossRef] [PubMed]
- Salas-Marco, J.; Bedwell, D.M. GTP hydrolysis by eRF3 facilitates stop codon decoding during eukaryotic translation termination. Mol. Cell. Biol. 2004, 24, 7769–7778. [Google Scholar] [CrossRef] [PubMed]
- Derkatch, I.L.; Bradley, M.E.; Hong, J.Y.; Liebman, S.W. Prions affect the appearance of other prions: The story of [PIN+]. Cell 2001, 106, 171–182. [Google Scholar] [CrossRef]
- Sondheimer, N.; Lindquist, S. Rnq1: An epigenetic modifier of protein function in yeast. Mol. Cell 2000, 5, 163–172. [Google Scholar] [CrossRef]
- Eaglestone, S.S.; Cox, B.S.; Tuite, M.F. Translation termination efficiency can be regulated in Saccharomyces cerevisiae by environmental stress through a prion-mediated mechanism. EMBO J. 1999, 18, 1974–1981. [Google Scholar] [CrossRef] [PubMed]
- Urakov, V.N.; Vishnevskaya, A.B.; Alexandrov, I.M.; Kushnirov, V.V.; Smirnov, V.N.; Ter-Avanesyan, M.D. Interdependence of amyloid formation in yeast: Implications for polyglutamine disorders and biological functions. Prion 2010, 4, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Derkatch, I.L.; Uptain, S.M.; Patino, M.M.; Lindquist, S.; Liebman, S.W. The yeast non-Mendelian factor [ETA+] is a variant of [PSI+], a prion-like form of release factor eRF3. EMBO J. 1999, 18, 1182–1191. [Google Scholar] [CrossRef] [PubMed]
- Son, M.; Wickner, R.B. Nonsense-mediated mRNA decay factors cure most [PSI+] prion variants. Proc. Natl. Acad. Sci. USA 2018, 2, E1184–E1193. [Google Scholar] [CrossRef] [PubMed]
- Chernova, T.A.; Wilkinson, K.D.; Chernoff, Y.O. Physiological and environmental control of yeast prions. FEMS Microbiol. Rev. 2014, 38, 326–344. [Google Scholar] [CrossRef] [PubMed]
- Barbitoff, Y.A.; Matveenko, A.G.; Moskalenko, S.E.; Zemlyanko, O.M.; Newnam, G.P.; Patel, A.; Chernova, T.A.; Chernoff, Y.O.; Zhouravleva, G.A. To CURe or not to CURe? Differential effects of the chaperone sorting factor Cur1 on yeast prions are mediated by the chaperone Sis1. Mol. Microbiol. 2017, 105, 242–257. [Google Scholar] [CrossRef] [PubMed]
- Keefer, K.M.; True, H.L. Prion-associated toxicity is rescued by elimination of cotranslational chaperones. PLoS Genet. 2016, 12, e1006431. [Google Scholar] [CrossRef] [PubMed]
- Kirkland, P.A.; Reidy, M.; Masison, D.C. Functions of yeast Hsp40 chaperone Sis1p dispensable for prion propagation but important for prion curing and protection from prion toxicity. Genetics 2011, 188, 565–577. [Google Scholar] [CrossRef] [PubMed]
- Valouev, I.A.; Kushnirov, V.V.; Ter-Avanesyan, M.D. Yeast polypeptide chain release factors eRF1 and eRF3 are involved in cytoskeleton organization and cell cycle regulation. Cell Motil. Cytoskelet. 2002, 52, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Urakov, V.N.; Valouev, I.A.; Kochneva-Pervukhova, N.V.; Packeiser, A.N.; Vishnevsky, A.Y.; Glebov, O.O.; Smirnov, V.N.; Ter-Avanesyan, M.D. N-terminal region of Saccharomyces cerevisiae eRF3 is essential for the functioning of the eRF1/eRF3 complex beyond translation termination. BMC Mol. Biol. 2006, 7, 34. [Google Scholar] [CrossRef] [PubMed]
- Bailleul, P.A.; Newnam, G.P.; Steenbergen, J.N.; Chernoff, Y.O. Genetic study of interactions between the cytoskeletal assembly protein Sla1. Genetics 1999, 153, 81–94. [Google Scholar] [PubMed]
- Dergalev, A.A.; Ivannikov, R.I.; Ter-Avanesyan, M.D.; Kushnirov, V.V. Yeast Sup35 prion structure: Three parts, two types, many variants. PLoS Genet. 2018, in press. [Google Scholar]
- Cui, Y.; Hagan, K.W.; Zhang, S.; Peltz, S.W. Identification and characterization of genes that are required for the accelerated degradation of mRNAs containing a premature translational termination codon. Genes Dev. 1995, 9, 423–436. [Google Scholar] [CrossRef] [PubMed]
- Kushnirov, V.V.; Alexandrov, I.M.; Mitkevich, O.V.; Shkundina, I.S.; Ter-Avanesyan, M.D. Purification and analysis of prion and amyloid aggregates. Methods 2006, 39, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Valouev, I.A.; Urakov, V.N.; Kochneva-Pervukhova, N.V.; Smirnov, V.N.; Ter-Avanesyan, M.D. Translation termination factors function outside of translation: Yeast eRF1 interacts with myosin light chain, Mlc1p, to effect cytokinesis. Mol. Microbiol. 2004, 53, 687–696. [Google Scholar] [CrossRef] [PubMed]
- Gietz, R.D.; Sugino, A. New yeast-Escherichia coli shuttle vectors constructed with in vitro mutagenized yeast genes lacking six-base pair restriction sites. Gene 1998, 74, 527–534. [Google Scholar] [CrossRef]
- Valouev, I.A.; Fominov, G.V.; Sokolova, E.E.; Smirnov, V.N.; Ter-Avanesyan, M.D. Elongation factor eEF1B modulates functions of the release factors eRF1 and eRF3 and the efficiency of translation termination in yeast. BMC Mol. Biol. 2009, 10, 60. [Google Scholar] [CrossRef] [PubMed]
- Urakov, V.N.; Valouev, I.A.; Lewitin, E.I.; Paushkin, S.V.; Kosorukov, V.S.; Kushnirov, V.V.; Smirnov, V.N.; Ter-Avanesyan, M.D. Itt1p, a novel protein inhibiting translation termination in Saccharomyces cerevisiae. BMC Mol. Biol. 2001, 2, 9. [Google Scholar] [CrossRef]
- Weng, Y.; Czaplinski, K.; Peltz, S.W. Genetic and biochemical characterization of mutations in the ATPase and helicase regions of the Upf1 protein. Mol. Cell Biol. 1996, 16, 5477–5490. [Google Scholar] [CrossRef] [PubMed]
- Sikorski, R.S.; Hieter, P. A system of shuttle vectors and yeast host strains designed for efficient manipulation of DNA in Saccharomyces cerevisiae. Genetics 1989, 122, 19–27. [Google Scholar] [PubMed]
- Ter-Avanesyan, M.D.; Kushnirov, V.V.; Dagkesamanskaya, A.R.; Didichenko, S.A.; Chernoff, Y.O.; Inge-Vechtomov, S.G.; Smirnov, V.N. Deletion analysis of the SUP35 gene of the yeast Saccharomyces cerevisiae reveals two non-overlapping functional regions in the encoded protein. Mol. Microbiol. 1993, 7, 683–692. [Google Scholar] [CrossRef] [PubMed]
- Keeling, K.M.; Lanier, J.; Du, M.; Salas-Marko, J.; Gao, L.; Kaenjak-Angeletti, A.; Bedwell, D.M. Leaky termination at premature stop codons antagonizes nonsense-mediated mRNA decay in S. cerevisiae. RNA 2004, 10, 691–703. [Google Scholar] [CrossRef] [PubMed]
- McNabb, D.S.; Reed, R.; Marciniak, R.A. Dual luciferase assay system for rapid assessment of gene expression in Saccharomyces cerevisiae. Eukaryot. Cell 2005, 4, 1539–1549. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.L.; Dinman, J.D. Systematic analysis of bicistronic reporter assay data. Nucleic Acids Res. 2004, 32, e160. [Google Scholar] [CrossRef] [PubMed]
| Plasmid | Rescue Plasmid Loss (%) | Suppression of Synthetic Lethality |
|---|---|---|
| Multi-UPF1 | 36 | + |
| CEN-UPF1 | 52 | + |
| *Multi-PUB1 | 86 | + |
| *CEN-PUB1 | 87 | + |
| *Multi-pub1-∆C | 36 | + |
| *CEN- pub1-∆C | 50 | + |
| Multi-SUP45 | 84 | + |
| CEN-SUP45 | 53 | + |
| Multi-sup35-C | 38 | + |
| CEN- sup35-C | 0 | − |
| Empty vector | 0 | − |
| [PSI+] Variant | % Readthrough |
|---|---|
| [PSI+]S7 | 6.1 ± 0.4 |
| [PSI+]WS2 | 2.1 ± 0.1 |
| [PSI+]W2 | 1.5 ± 0.2 |
| Plasmids | Characteristics | Source |
|---|---|---|
| YEplac181 | Multicopy LEU2 plasmid | [46] |
| YEplac181-PUB1 | Multicopy LEU2 plasmid harboring the PUB1 gene | This work |
| YEplac181-SUP35C | Multicopy LEU2 plasmid encoding Sup35C | [47] |
| Yeplac181-UPF1 | Multicopy LEU2 plasmid harboring the UPF1 gene | This work |
| YEplac195 | Multicopy URA3 plasmid | [46] |
| YEplac195-PUB1 | Multicopy URA3 plasmid harboring the PUB1 gene | [32] |
| YEplac195-PUB1ΔC | Multicopy URA3 plasmid encoding Pub1ΔC | [23] |
| YEplac195-SUP45 | Multicopy URA3 plasmid harboring the SUP45 gene | [48] |
| YEplac112-UPF1 | Multicopy TRP1 plasmid harboring the UPF1 gene | [49] |
| pRS315 | Centromeric LEU2 plasmid | [50] |
| pRS315-SUP35C | Centromeric LEU2 plasmid encoding Sup35C | [40] |
| pRS315-SUP45 | Centromeric LEU2 plasmid harboring the SUP45 gene | [45] |
| pRS315-UPF1 | Centromeric LEU2 plasmid harboring the UPF1 gene | This work |
| pRS316 | Centromeric URA3 plasmid | [50] |
| pRS316- PUB1 | Centromeric URA3 plasmid harboring the PUB1 gene | This work |
| pRS316- PUB1ΔC | Centromeric URA3 plasmid encoding Pub1ΔC | This work |
| pEMBLyex4(ΔLEU2d)-3ATG | Multicopy URA3 plasmid encoding Sup35C | [51] |
| pPUB1::TRP1 | Plasmid encoding pub1::TRP1 disruption cassette | [23] |
| pKOM | Plasmid encoding upf1::URA3 disruption cassette | [43] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urakov, V.N.; Mitkevich, O.V.; Dergalev, A.A.; Ter-Avanesyan, M.D. The Pub1 and Upf1 Proteins Act in Concert to Protect Yeast from Toxicity of the [PSI+] Prion. Int. J. Mol. Sci. 2018, 19, 3663. https://doi.org/10.3390/ijms19113663
Urakov VN, Mitkevich OV, Dergalev AA, Ter-Avanesyan MD. The Pub1 and Upf1 Proteins Act in Concert to Protect Yeast from Toxicity of the [PSI+] Prion. International Journal of Molecular Sciences. 2018; 19(11):3663. https://doi.org/10.3390/ijms19113663
Chicago/Turabian StyleUrakov, Valery N., Olga V. Mitkevich, Alexander A. Dergalev, and Michael D. Ter-Avanesyan. 2018. "The Pub1 and Upf1 Proteins Act in Concert to Protect Yeast from Toxicity of the [PSI+] Prion" International Journal of Molecular Sciences 19, no. 11: 3663. https://doi.org/10.3390/ijms19113663
APA StyleUrakov, V. N., Mitkevich, O. V., Dergalev, A. A., & Ter-Avanesyan, M. D. (2018). The Pub1 and Upf1 Proteins Act in Concert to Protect Yeast from Toxicity of the [PSI+] Prion. International Journal of Molecular Sciences, 19(11), 3663. https://doi.org/10.3390/ijms19113663