Non-Invasive Detection of Extracellular Matrix Metalloproteinase Inducer EMMPRIN, a New Therapeutic Target against Atherosclerosis, Inhibited by Endothelial Nitric Oxide
Abstract
1. Introduction
2. Results
2.1. Human Endarterectomy Samples Express Extracellular Matrix (ECM) Degrading MMP-9, MMP-13 and EMMPRIN
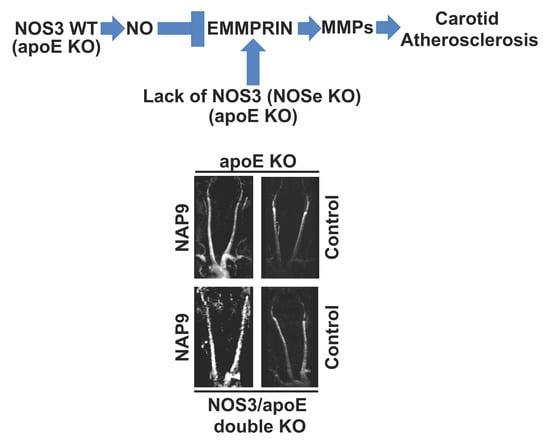
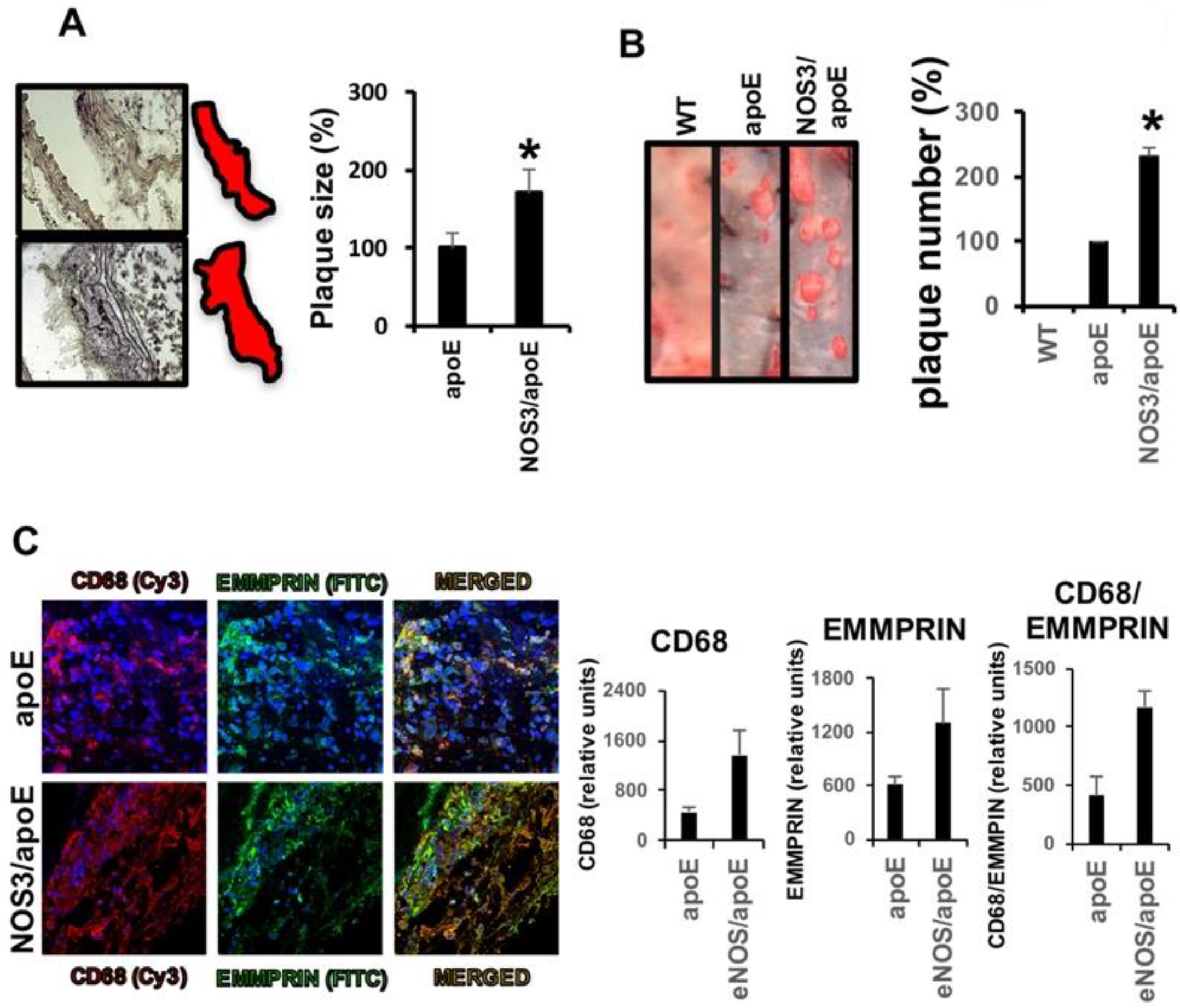
2.2. Lack of NOS3 Increases Atherosclerotic Lesions and Inflammatory Macrophages in apoE Null Mice
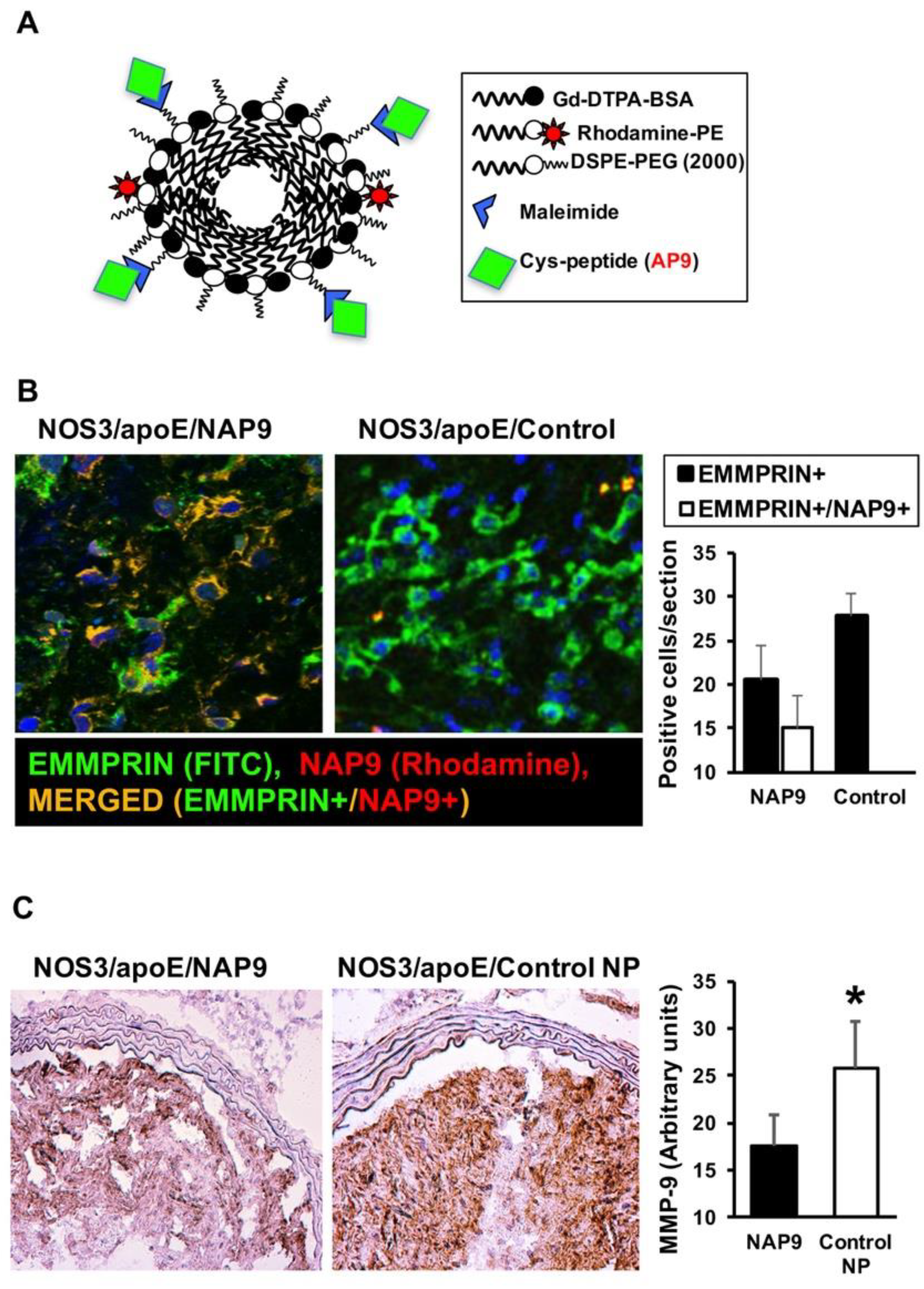
2.3. NAP9 Targets Extracellular Matrix Metalloproteinase Inducer (EMMPRIN) in Carotid Atherosclerotic Plaques
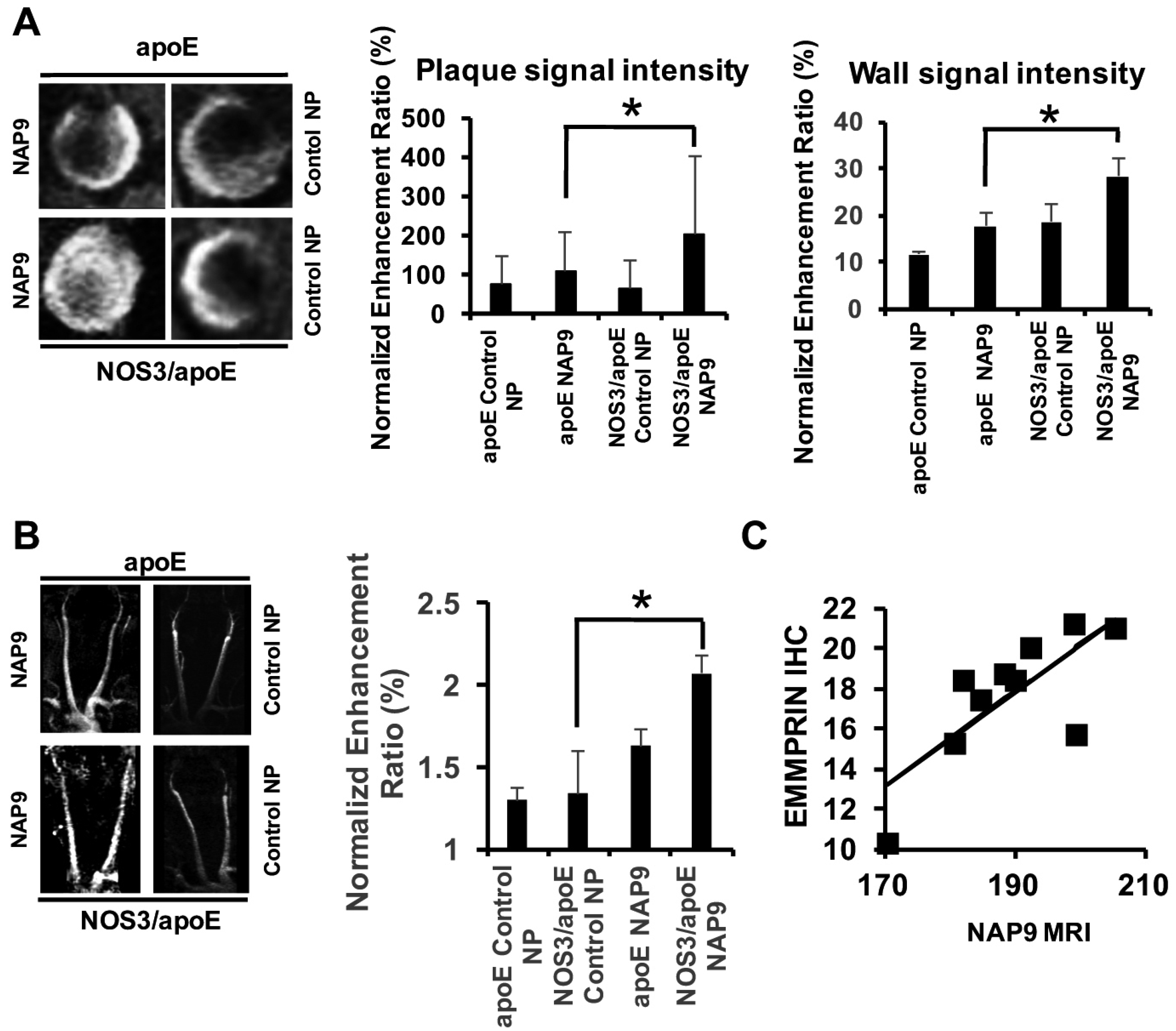
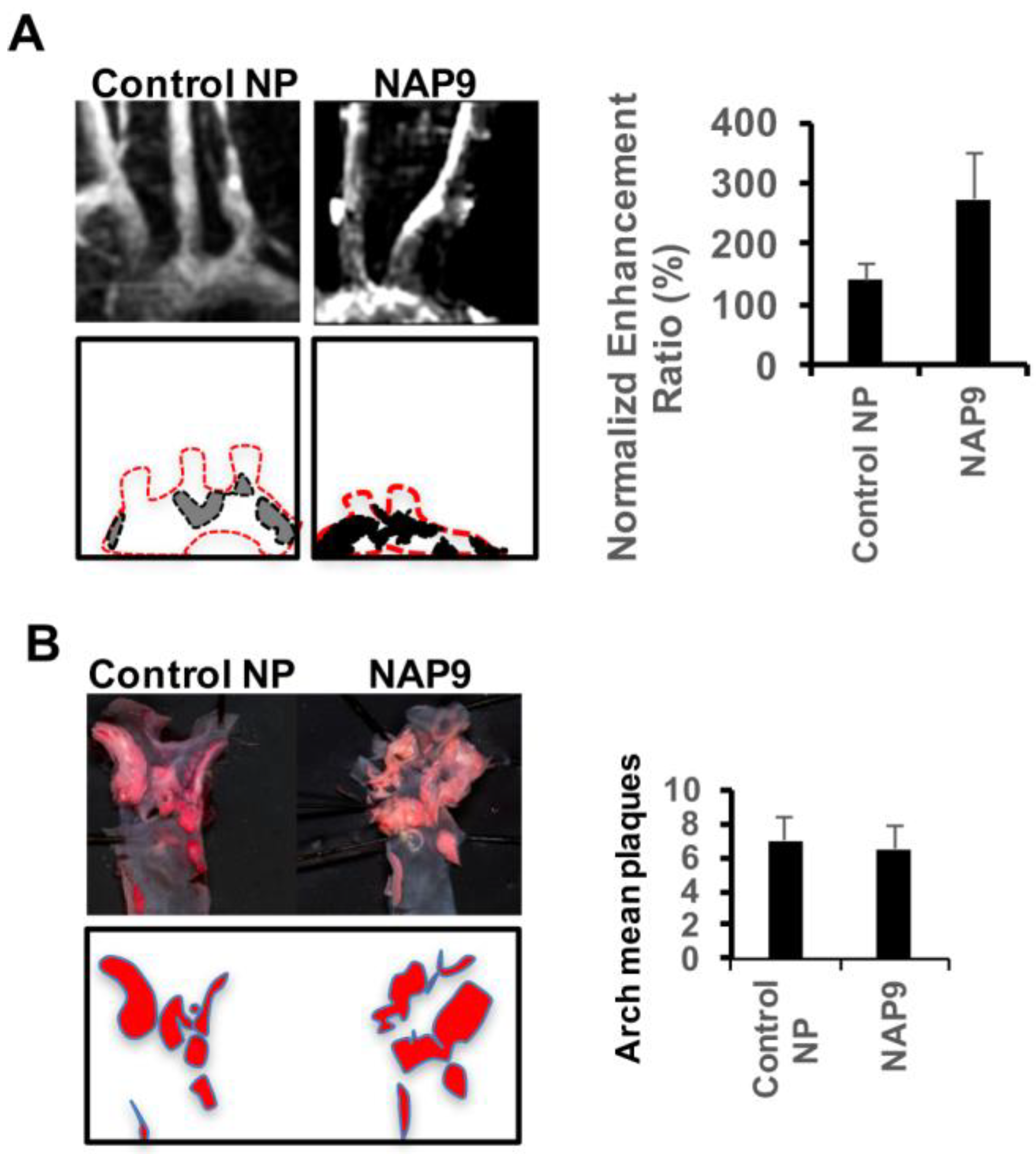
2.4. Non-Invasive Nanoparticle Detection of EMMPRIN by Magnetic Resonance Imaging (MRI)
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Human Arterial Specimens
4.3. Animals and Diet
4.4. Blood Lipids and Glucose
4.5. Blood Pressure
4.6. Quantitation of Lesion
4.7. Confocal Microscopy
4.8. Peptide and Nanoprobe Composition
4.9. Magnetic Resonance
4.10. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Tarín, C.; Gomez, M.; Calvo, E.; López, J.A.; Zaragoza, C. Endothelial nitric oxide deficiency reduces MMP-13-mediated cleavage of ICAM-1 in vascular endothelium: A role in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Kuhlencordt, P.J.; Gyurko, R.; Han, F.; Scherrer-Crosbie, M.; Aretz, T.H.; Hajjar, R.; Picard, M.H.; Huang, P.L. Accelerated atherosclerosis, aortic aneurysm formation, and ischemic heart disease in apolipoprotein E/Endothelial nitric oxide synthase double-knockout mice. Circulation 2001, 104, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Herman, A.G.; Moncada, S. Therapeutic potential of nitric oxide donors in the prevention and treatment of atherosclerosis. Eur. Heart J. 2005, 26, 1945–1955. [Google Scholar] [CrossRef] [PubMed]
- Lizarbe, T.R.; Tarín, C.; Gómez, M.; Lavin, B.; Aracil, E.; Orte, L.M.; Zaragoza, C. Nitric oxide induces the progression of abdominal aortic aneurysms through the matrix metalloproteinase inducer EMMPRIN. Am. J. Pathol. 2009, 175, 1421–1430. [Google Scholar] [CrossRef] [PubMed]
- Tarin, C.; Lavin, B.; Gomez, M.; Saura, M.; Diez-Juan, A.; Zaragoza, C. The extracellular matrix metalloproteinase inducer EMMPRIN is a target of nitric oxide in myocardial ischemia/reperfusion. Free Radic. Biol. Med. 2011, 51, 387–395. [Google Scholar] [CrossRef] [PubMed]
- von Ungern-Sternberg, S.N.; Zernecke, A.; Seizer, P. Extracellular Matrix Metalloproteinase Inducer EMMPRIN (CD147) in Cardiovascular Disease. Int. J. Mol. Sci. 2018, 19, 507. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Hemler, M.E. Caveolin-1 regulates matrix metalloproteinases-1 induction and CD147/EMMPRIN cell surface clustering. J. Biol. Chem. 2004, 279, 11112–11118. [Google Scholar] [CrossRef] [PubMed]
- Seizer, P.; Ochmann, C.; Schönberger, T.; Zach, S.; Rose, M.; Borst, O.; Klingel, K.; Kandolf, R.; MacDonald, H.R.; Nowak, R.A.; et al. Disrupting the EMMPRIN (CD147)-cyclophilin A interaction reduces infarct size and preserves systolic function after myocardial ischemia and reperfusion. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 1377–1386. [Google Scholar] [CrossRef] [PubMed]
- Seizer, P.; Ungern-Sternberg, S.N.; Schönberger, T.; Borst, O.; Münzer, P.; Schmidt, E.M.; Mack, A.F.; Heinzmann, D.; Chatterjee, M.; Langer, H.; et al. Extracellular cyclophilin A activates platelets via EMMPRIN (CD147) and PI3K/Akt signaling, which promotes platelet adhesion and thrombus formation in vitro and in vivo. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Hou, X.; Yang, Y.; Liu, H.; Guo, R.; Yang, Z.; Yang, L. The feedback loop of “EMMPRIN/NF-κB” worsens atherosclerotic plaque via suppressing autophagy in macrophage. J. Mol. Cell. Cardiol. 2018, 114, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Seizer, P.; Schönberger, T.; Schött, M.; Lang, M.R.; Langer, H.F.; Bigalke, B.; Krämer, B.F.; Borst, O.; Daub, K.; Heidenreich, O.; et al. EMMPRIN and its ligand cyclophilin A regulate MT1-MMP, MMP-9 and M-CSF during foam cell formation. Atherosclerosis 2010, 209, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Yang, L.X.; Guo, R.; Shi, Y.; Hou, X.; Yang, Z.; Zhou, X.; Liu, H. Atorvastatin attenuates plaque vulnerability by downregulation of EMMPRIN expression via COX-2/PGE2 pathway. Exp. Ther. Med. 2017, 13, 835–844. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Yang, L.X.; Guo, R.W.; Zhu, G.F.; Shi, Y.K.; Wang, X.M.; Qi, F.; Guo, C.M.; Ye, J.S.; Yang, Z.H.; et al. Functional blockage of EMMPRIN ameliorates atherosclerosis in apolipoprotein E-deficient mice. Int. J. Cardiol. 2013, 168, 3248–3253. [Google Scholar] [CrossRef] [PubMed]
- Schulz, C.; von Brühl, M.L.; Barocke, V.; Cullen, P.; Mayer, K.; Okrojek, R.; Steinhart, A.; Ahmad, Z.; Kremmer, E.; Nieswandt, B.; et al. EMMPRIN (CD147/basigin) mediates platelet-monocyte interactions in vivo and augments monocyte recruitment to the vascular wall. J. Thromb. Haemost. 2011, 9, 1007–1019. [Google Scholar] [CrossRef] [PubMed]
- Cuadrado, I.; Piedras, M.J.; Herruzo, I.; Turpin Mdel, C.; Castejón, B.; Reventun, P.; Martin, A.; Saura, M.; Zamorano, J.L.; Zaragoza, C. EMMPRIN-Targeted Magnetic Nanoparticles for In Vivo Visualization and Regression of Acute Myocardial Infarction. Theranostics 2016, 6, 545–557. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, R.; Díez, J.; Sanmartín, M.; Saura, M.; Zamorano, J.L.; Zaragoza, C. Nanotechnology Applied to Preserve Extracelular Matrix as Teranostic Tool in Acute Myocardial Infarction. Rev. Esp. Cardiol. (Engl. Ed.) 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Kirk, P.; Wilson, M.C.; Heddle, C.; Brown, M.H.; Barclay, A.N.; Halestrap, A.P. CD147 is tightly associated with lactate transporters MCT1 and MCT4 and facilitates their cell surface expression. EMBO J. 2000, 19, 3896–3904. [Google Scholar] [CrossRef] [PubMed]
- Pushkarsky, T.; Yurchenko, V.; Vanpouille, C.; Brichacek, B.; Vaisman, I.; Hatakeyama, S.; Nakayama, K.I.; Sherry, B.; Bukrinsky, M.I. Cell surface expression of CD147/EMMPRIN is regulated by cyclophilin 60. J. Biol. Chem. 2005, 280, 27866–27871. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Zhou, H.; Walian, P.J.; Jap, B.K. CD147 is a regulatory subunit of the gamma-secretase complex in Alzheimer’s disease amyloid beta-peptide production. Proc. Natl. Acad. Sci. USA 2005, 102, 7499–7504. [Google Scholar] [CrossRef] [PubMed]
- Cuadrado, I.; Castejon, B.; Martin, A.M.; Saura, M.; Reventun-Torralba, P.; Zamorano, J.L.; Zaragoza, C. Nitric Oxide Induces Cardiac Protection by Preventing Extracellular Matrix Degradation through the Complex Caveolin-3/EMMPRIN in Cardiac Myocytes. PLoS ONE 2016, 11, e0162912. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Chang, S.B.; Hemler, M.E. Links between CD147 function, glycosylation, and caveolin-1. Mol. Biol. Cell 2004, 15, 4043–4050. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.; Yuan, W.; Liu, J.; He, Q.; Ding, S.; Pu, J.; He, B. Functional relevance of protein glycosylation to the pro-inflammatory effects of extracellular matrix metalloproteinase inducer (EMMPRIN) on monocytes/macrophages. PLoS ONE 2015, 10, e0117463. [Google Scholar] [CrossRef] [PubMed]
- Voss, D.M.; Spina, R.; Carter, D.L.; Lim, K.S.; Jeffery, C.J.; Bar, E.E. Disruption of the monocarboxylate transporter-4-basigin interaction inhibits the hypoxic response, proliferation, and tumor progression. Sci. Rep. 2017, 27, 4292. [Google Scholar] [CrossRef] [PubMed]
- Sangwung, P.; Greco, T.M.; Wang, Y.; Ischiropoulos, H.; Sessa, W.C.; Iwakiri, Y. Proteomic identification of S-nitrosylated Golgi proteins, new insights into endothelial cell regulation by eNOS-derived NO. PLoS ONE 2012, 7, e31564. [Google Scholar] [CrossRef] [PubMed]
- Lian, C.; Guo, Y.; Zhang, J.; Chen, X.; Peng, C. Targeting CD147 is a Novel Strategy for Antitumor Therapy. Curr. Pharm. Des. 2017, 23, 4410–4421. [Google Scholar] [CrossRef] [PubMed]
- Seizer, P.; Geisler, T.; Bigalke, B.; Schneider, M.; Klingel, K.; Kandolf, R.; Stellos, K.; Schreieck, J.; Gawaz, M.; May, A.E. EMMPRIN and its ligand cyclophilin A as novel diagnostic markers in inflammatory cardiomyopathy. Int. J. Cardiol. 2013, 163, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Marsden, C.J.; Eckersley, S.; Hebditch, M.; Kvist, A.J.; Milner, R.; Mitchell, D.; Warwicker, J.; Marley, A.E. The Use of Antibodies in Small-Molecule Drug Discovery. J. Biomol. Screen. 2014, 19, 829–838. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Wang, F. Development of RGD-based radiotracers for tumor imaging and therapy: Translating from bench to bedside. Curr. Mol. Med. 2013, 13, 1487–1505. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Mi, L.; Xu, J.; Yu, J.; Wang, X.; Jiang, J.; Xing, J.; Shang, P.; Qian, A.; Li, Y.; et al. Function of HAb18G/CD147 in invasion of host cells by severe acute respiratory syndrome coronavirus. J. Infect. Dis. 2005, 191, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhu, P.; Jiang, J.L.; Zhang, Q.; Wu, Z.B.; Yao, X.Y.; Tang, H.; Lu, N.; Yang, Y.; Chen, Z.N. Involvement of CD147 in overexpression of MMP-2 and MMP-9 and enhancement of invasive potential of PMA-differentiated THP-1. BMC Cell Biol. 2005, 6, 25. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Lu, N.; Zhou, J.; Chen, Z.N.; Zhu, P. Cyclophilin A up-regulates MMP-9 expression and adhesion of monocytes/macrophages via CD147 signalling pathway in rheumatoid arthritis. Rheumatology 2008, 47, 1299–1310. [Google Scholar] [CrossRef] [PubMed]

| Parameters | apoE | NOS3/apoE |
|---|---|---|
| Systolic BP (mmHg) | 122.1 ± 7.33 | 141 ± 5.03 (P: 0.003) |
| Diastolic BP (mmHg) | 82.1 ± 6.33 | 106 ± 9.08 (P: 0.003) |
| Blood glucose (mg/dL) | 115.6 ± 10.40 | 121.3 ± 13.86 |
| Total Cholesterol (mg/dL) | 811.1 ± 132 | 821.6 ± 128.82 |
| Triglicerides (mg/dL) | 174.7 ± 20.56 | 171.5 ± 22.07 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramirez-Carracedo, R.; Tesoro, L.; Hernandez, I.; Diez-Mata, J.; Filice, M.; Toro, R.; Rodriguez-Piñero, M.; Zamorano, J.L.; Saura, M.; Zaragoza, C. Non-Invasive Detection of Extracellular Matrix Metalloproteinase Inducer EMMPRIN, a New Therapeutic Target against Atherosclerosis, Inhibited by Endothelial Nitric Oxide. Int. J. Mol. Sci. 2018, 19, 3248. https://doi.org/10.3390/ijms19103248
Ramirez-Carracedo R, Tesoro L, Hernandez I, Diez-Mata J, Filice M, Toro R, Rodriguez-Piñero M, Zamorano JL, Saura M, Zaragoza C. Non-Invasive Detection of Extracellular Matrix Metalloproteinase Inducer EMMPRIN, a New Therapeutic Target against Atherosclerosis, Inhibited by Endothelial Nitric Oxide. International Journal of Molecular Sciences. 2018; 19(10):3248. https://doi.org/10.3390/ijms19103248
Chicago/Turabian StyleRamirez-Carracedo, Rafael, Laura Tesoro, Ignacio Hernandez, Javier Diez-Mata, Marco Filice, Rocío Toro, Manuel Rodriguez-Piñero, Jose Luis Zamorano, Marta Saura, and Carlos Zaragoza. 2018. "Non-Invasive Detection of Extracellular Matrix Metalloproteinase Inducer EMMPRIN, a New Therapeutic Target against Atherosclerosis, Inhibited by Endothelial Nitric Oxide" International Journal of Molecular Sciences 19, no. 10: 3248. https://doi.org/10.3390/ijms19103248
APA StyleRamirez-Carracedo, R., Tesoro, L., Hernandez, I., Diez-Mata, J., Filice, M., Toro, R., Rodriguez-Piñero, M., Zamorano, J. L., Saura, M., & Zaragoza, C. (2018). Non-Invasive Detection of Extracellular Matrix Metalloproteinase Inducer EMMPRIN, a New Therapeutic Target against Atherosclerosis, Inhibited by Endothelial Nitric Oxide. International Journal of Molecular Sciences, 19(10), 3248. https://doi.org/10.3390/ijms19103248