DNA Methylation as a Biomarker of Treatment Response Variability in Serious Mental Illnesses: A Systematic Review Focused on Bipolar Disorder, Schizophrenia, and Major Depressive Disorder
Abstract
1. Introduction
1.1. Pharmacogenetics Studies
1.2. Epigenetic Mechanisms
1.3. Variability of Response to Treatments
2. DNA Methylation Patterns in SCZ, BD and MDD
3. Treatment-Induced DNA Methylation Modifications in Animal Models
4. DNA Methylation Modifications and Responses to Treatment in Human Studies
4.1. Cross-Sectional Studies
- Schizophrenia: There is evidence that antipsychotics induce the alteration of the methylation status of several genes, including those coding for proteins and pathways targeted by these drugs. Very few human studies have investigated the influence of epigenetic mechanisms on the response to antipsychotics, but the DNA methylation status of candidate genes have been found to be differentially modulated by treatment in patients with SCZ according to their response phenotypes (Table 2). In a cohort of 177 SCZ patients, Melas and colleagues reported that patients treated with haloperidol (n = 16) displayed a significantly higher level of global DNA methylation in blood [49], as compared to other antipsychotic drugs. Interestingly though, correlations between the DNA methylation levels and the response status of SCZ patients have been found in several studies. However, a tendency to reverse hypermethylation at the DTNBP1 (Dysbindin) gene promoter in post-mortem brain samples of schizophrenic patients was found with antipsychotic treatments [79].
- Bipolar disorder: The decreased DNA methylation of the DTNBP1 gene promoter with antipsychotic drug treatment found in the post-mortem brain samples of patients with SCZ was not seen in post-mortem brains from patients with BD, probably due to small number of patients using classic antipsychotics [79] (Table 2). A lithium-induced decrease of global DNA methylation was found in lymphoblast cell lines derived from 14 lithium-responder BD patients, as compared to 16 healthy controls [80]. Recently, Houtepen and colleagues investigated the effects of antipsychotics (olanzapine and quetiapine) and mood stabilizers (lithium, VPA, carbamazepine) on genome-wide DNA methylation in blood samples from 172 patients with BD. After adjustment for drugs effects on blood cell types, composition-only VPA and quetiapine modified the DNA methylation status significantly [81]. In a study of global DNA methylation in the leukocytes of BD patients, no differences were found compared to the healthy control. However a significantly lower DNA methylation level was observed in patients on lithium monotherapy, compared to controls or BD patients treated with a combination of lithium + VPA [82]. In this study, the DNA methylation level could not be correlated with the lithium response as assessed with the Alda scale. However, since global DNA methylation studies provide an imprecise picture of the effect of a given drug, gene-specific effects have been investigated. A decrease of DNA methylation at the promoter I of BDNF was observed in the Peripheral Blood Mononuclear Cells (PBMC) of BD patients with antidepressant therapy, compared to no antidepressant therapy [50]. Similarly, patients treated with lithium or VPA displayed a decrease of DNA methylation levels, as compared to other medications [50,83]. A trend, yet not significant, for a decreased DNA methylation level at the BDNF promoter I was found in another study with a slightly different design [84].
- Major depressive disorder: A large study explored the DNA methylation of the BDNF gene in patients with MDD (n = 207), BD (n = 59), and controls (n = 278). They reported an increased methylation of BDNF gene in patients with MDD compared to those with BD and controls. Somewhat surprisingly, they also found that the increased methylation of BDNF is associated with antidepressant therapy, but not with the clinical features of MDD [85]. Although very informative, these studies did not discriminate between the antidepressant classes. The only available study on therapeutic response to the SSRI, paroxetine, reported an association with the methylation level of the PPFIA4 (Protein Tyrosine Phosphatase, Receptor Type, F Polypeptide, Interacting Protein, Alpha 4) and HS3ST1 (heparin sulfate-glucosamine 3-sulfotransferase 1) genes in MDD responders, compared to the worst responders (n = 10 per group) [86].
4.2. Longitudinal Studies
- Schizophrenia: DNA methylation change at the 13th CpG site of HTR1A is associated with negative symptoms in patients with SCZ after 10 weeks of treatment with antipsychotic drugs (n = 82) [87] (Table 3). Likewise, clozapine-induced DNA methylation changes in the CREB-binding protein (CREBBP) gene are inversely correlated with the percentage of Positive and Negative Syndrome Scale (PANSS) changes in treatment-resistant SCZ patients (n = 21) [88]. A recent study in Chinese Han schizophrenic patients investigated not only genes that were involved in the dopaminergic and serotoninergic pathways, but also in the metabolism and transport of risperidone. They found no significant CpG sites in HTR2A, ABCB1, and DRD2 gene promoters associated with responses, while differentially methylated CpG of the drug-metabolizing enzymes CYP3A4 and CYP2D6 genes promoter regions were associated with a response to risperidone [89]. Furthermore, a whole-genome study of DNA methylation modifications before and after treatment with antipsychotics found gender-specific differences in the methylation profiles of patients with SCZ. Significant differences were observed in the male patient group in complete remission [90]. In this study methylation levels of six genes (APIS3, C16orf59, KCNK15, LOC146336, MGC16384 and XRN2) and nine genes (C16orf70, CST3, DDRGK1, FA2H, FLJ30058, MFSD2B, RFX4, UBE2J1 and ZNF311) were respectively identified as good markers of treatment-induced effects, and good predictive markers of treatment response [90].
- Major depressive disorder: Several of the drugs used to treat an MDD target, a serotonin transporter, an association between its DNA methylation levels before treatment and impaired treatment response after 12 weeks of antidepressant therapy in patients with MDD (n = 108) have been reported [91] (Table 3). Another study comparing the methylation levels before and after six weeks of antidepressant therapy showed that an increased DNA methylation at the third CpG site of SLC6A4 was associated with better therapeutic response in patients with MDD [92]. The results of Okada and colleagues were confirmed in a naturalistic study of MDD patients (n = 94) treated with escitalopram [93]. They found that higher methylation at the SLC6A4 gene was associated with better treatment response after six weeks of treatment (Table 3). However, the response status to escitalopram was found not associated with the DNA methylation level of another gene MAO-A (mono amino oxidase A) in 61 MDD patients [94]. Recently, hypomethylation at two CpGs sites (HTR1A CpG 668 and HTR1B CpG 1401) was found to significantly differ in remitter and non-remitter Chinese Han patients with MDD (n = 85) with escitalopram treatment [95]. Very promising results have been obtained in MDD patients treated with escitalopram (n = 80) or with the tricyclic antidepressant nortriptyline (n = 33) [96]. In this study, higher DNA methylation level at the fourth CpG island of the interleukin-11 (IL-11) gene before treatment was associated with a better response to escitalopram, while hypomethylation at the same site was associated with a better nortriptyline response (Table 3). These results suggest that DNA methylation levels before treatment could be a predictor of the best suited antidepressant for an individual.
5. Materials and Methods
6. Conclusions
Funding
Conflicts of Interest
Abbreviations
| SMI | Severe mental illnesses |
| SCZ | Schizophrenia |
| BD | Bipolar disorder |
| MDD | Major depressive disorder |
References
- World Health Organization. The 10th Revision of the International Classification of Diseases and Related Health Problems (ICD-10); WHO: Geneva, Switzerland, 1992. [Google Scholar]
- Ferrari, A.J.; Saha, S.; McGrath, J.J.; Norman, R.; Baxter, A.J.; Vos, T.; Whiteford, H.A. Health states for schizophrenia and bipolar disorder within the Global Burden of Disease 2010 Study. Popul. Health Metr. 2012, 10. [Google Scholar] [CrossRef] [PubMed]
- Reddy, M.S. Depression: The Disorder and the Burden. Indian J. Psychol. Med. 2010, 32, 1–2. [Google Scholar] [CrossRef] [PubMed]
- McGrath, J.; Saha, S.; Chant, D.; Welham, J. Schizophrenia: A concise overview of incidence, prevalence, and mortality. Epidemiol. Rev. 2008, 30, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Merikangas, K.R.; Jin, R.; He, J.-P.; Kessler, R.C.; Lee, S.; Sampson, N.A.; Viana, M.C.; Andrade, L.H.; Hu, C.; Karam, E.G.; et al. Prevalence and Correlates of Bipolar Spectrum Disorder in the World Mental Health Survey Initiative. Arch. Gen. Psychiatry 2011, 68, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.J.; Lopez, A.D. Global mortality, disability, and the contribution of risk factors: Global Burden of Disease Study. Lancet 1997, 349, 1436–1442. [Google Scholar] [CrossRef]
- Kessler, R.C.; Bromet, E.J. The epidemiology of depression across cultures. Annu. Rev. Public Health 2013, 34, 119–138. [Google Scholar] [CrossRef] [PubMed]
- Kuswanto, C.N.; Sum, M.Y.; Sim, K. Neurocognitive Functioning in Schizophrenia and Bipolar Disorder: Clarifying Concepts of Diagnostic Dichotomy vs. Continuum. Front. Psychiatry 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.R.; Cherian, J.; Gohil, K.; Atkinson, D. Schizophrenia: Overview and Treatment Options. Pharm. Ther. 2014, 39, 638–645. [Google Scholar]
- Serafini, G.; Pompili, M.; Borgwardt, S.; Houenou, J.; Geoffroy, P.A.; Jardri, R.; Girardi, P.; Amore, M. Brain changes in early-onset bipolar and unipolar depressive disorders: A systematic review in children and adolescents. Eur. Child Adolesc. Psychiatry 2014, 23, 1023–1041. [Google Scholar] [CrossRef] [PubMed]
- Chesney, E.; Goodwin, G.M.; Fazel, S. Risks of all-cause and suicide mortality in mental disorders: A meta-review. World Psychiatry 2014, 13, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Need, A.C.; Goldstein, D.B. Schizophrenia genetics comes of age. Neuron 2014, 83, 760–763. [Google Scholar] [CrossRef] [PubMed]
- Harrison, P.J. Recent genetic findings in schizophrenia and their therapeutic relevance. J. Psychopharmacol. 2015, 29, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Piletz, J.E.; Zhang, X.; Ranade, R.; Liu, C. Database of genetic studies of bipolar disorder. Psychiatr. Genet. 2011, 21, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Serretti, A.; Mandelli, L. The genetics of bipolar disorder: Genome “hot regions,” genes, new potential candidates and future directions. Mol. Psychiatry 2008, 13, 742–771. [Google Scholar] [CrossRef] [PubMed]
- López-León, S.; Janssens, A.C.; González-Zuloeta Ladd, A.M.; Del-Favero, J.; Claes, S.J.; Oostra, B.A.; van Duijn, C.M. Meta-analyses of genetic studies on major depressive disorder. Mol. Psychiatry 2008, 13, 772–785. [Google Scholar] [CrossRef] [PubMed]
- Gatt, J.M.; Burton, K.L.O.; Williams, L.M.; Schofield, P.R. Specific and common genes implicated across major mental disorders: A review of meta-analysis studies. J. Psychiatr. Res. 2015, 60, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Avramopoulos, D. Recent Advances in the Genetics of Schizophrenia. Mol. Neuropsychiatry 2018, 4, 35–51. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, M.; Saito, T.; Kondo, K.; Iwata, N. Genome-wide association studies of bipolar disorder: A systematic review of recent findings and their clinical implications. Psychiatry Clin. Neurosci. 2018, 72, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Shadrina, M.; Bondarenko, E.A.; Slominsky, P.A. Genetics Factors in Major Depression Disease. Front. Psychiatry 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Lin, E.; Tsai, S.-J. Genome-wide microarray analysis of gene expression profiling in major depression and antidepressant therapy. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2016, 64, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Mamdani, F.; Martin, M.V.; Lencz, T.; Rollins, B.; Robinson, D.G.; Moon, E.A.; Malhotra, A.K.; Vawter, M.P. Coding and noncoding gene expression biomarkers in mood disorders and schizophrenia. Dis. Markers 2013, 35, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Molteni, R.; Calabrese, F.; Racagni, G.; Fumagalli, F.; Riva, M.A. Antipsychotic drug actions on gene modulation and signaling mechanisms. Pharmacol. Ther. 2009, 124, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Bellivier, F.; Marie-Claire, C. Lithium response variability: New avenues and hypotheses. In The Science and Practice of Lithium Therapy; Malhi, G.S., Masson, M., Bellivier, F., Eds.; Springer: Basel, Switzerland, 2017; pp. 157–178. [Google Scholar]
- Fabbri, C.; Hosak, L.; Mössner, R.; Giegling, I.; Mandelli, L.; Bellivier, F.; Claes, S.; Collier, D.A.; Corrales, A.; Delisi, L.E.; et al. Consensus paper of the WFSBP Task Force on Genetics: Genetics, epigenetics and gene expression markers of major depressive disorder and antidepressant response. World J. Biol. Psychiatry 2017, 18, 5–28. [Google Scholar] [CrossRef] [PubMed]
- Bock, C. Epigenetic biomarker development. Epigenomics 2009, 1, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Chan, T.A.; Baylin, S.B. Epigenetic biomarkers. Curr. Top. Microbiol. Immunol. 2012, 355, 189–216. [Google Scholar] [CrossRef] [PubMed]
- Heerboth, S.; Lapinska, K.; Snyder, N.; Leary, M.; Rollinson, S.; Sarkar, S. Use of epigenetic drugs in disease: An overview. Genet. Epigenet. 2014, 6, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.D.; Le, T.; Fan, G. DNA methylation and its basic function. Neuropsychopharmacology 2013, 38, 23–38. [Google Scholar] [CrossRef] [PubMed]
- Afonso-Grunz, F.; Müller, S. Principles of miRNA-mRNA interactions: Beyond sequence complementarity. Cell. Mol. Life Sci. 2015, 72, 3127–3141. [Google Scholar] [CrossRef] [PubMed]
- Yeshurun, S.; Hannan, A.J. Transgenerational epigenetic influences of paternal environmental exposures on brain function and predisposition to psychiatric disorders. Mol. Psychiatry 2018. [Google Scholar] [CrossRef] [PubMed]
- Cholewa-Waclaw, J.; Bird, A.; von Schimmelmann, M.; Schaefer, A.; Yu, H.; Song, H.; Madabhushi, R.; Tsai, L.-H. The role of epigenetic mechanisms in the regulation of gene expression in the nervous system. J. Neurosci. 2016, 36, 11427–11434. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Chang, H.; Li, E.; Fan, G. Dynamic expression of de novo DNA methyltransferases DNMT3A and DNMT3B in the central nervous system. J. Neurosci. Res. 2005, 79, 734–746. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.U.; Ma, D.K.; Mo, H.; Ball, M.P.; Jang, M.-H.; Bonaguidi, M.A.; Balazer, J.A.; Eaves, H.L.; Xie, B.; Ford, E.; et al. Neuronal activity modifies the DNA methylation landscape in the adult brain. Nat. Neurosci. 2011, 14, 1345–1351. [Google Scholar] [CrossRef] [PubMed]
- Martinowich, K.; Hattori, D.; Wu, H.; Fouse, S.; He, F.; Hu, Y.; Fan, G.; Sun, Y.E. DNA methylation-related chromatin remodeling in activity-dependent BDNF gene regulation. Science 2003, 302, 890–893. [Google Scholar] [CrossRef] [PubMed]
- Poo, S.X.; Agius, M. Atypical anti-psychotics in adult bipolar disorder: Current evidence and updates in the NICE guidelines. Psychiatr. Danub. 2014, 26, 322–329. [Google Scholar] [PubMed]
- Patkar, A.A.; Pae, C.-U. Atypical antipsychotic augmentation strategies in the context of guideline-based care for the treatment of major depressive disorder. CNS Drugs 2013, 27, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Richtand, N.M.; Welge, J.A.; Logue, A.D.; Keck, P.E.; Strakowski, S.M.; McNamara, R.K. Role of serotonin and dopamine receptor binding in antipsychotic efficacy. Prog. Brain Res. 2008, 172, 155–175. [Google Scholar] [CrossRef] [PubMed]
- Simonsen, E.; Friis, S.; Opjordsmoen, S.; Mortensen, E.L.; Haahr, U.; Melle, I.; Joa, I.; Johannessen, J.O.; Larsen, T.K.; Røssberg, J.I.; et al. Early identification of non-remission in first-episode psychosis in a two-year outcome study. Acta Psychiatr. Scand. 2010, 122, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, G.M.; Haddad, P.M.; Ferrier, I.N.; Aronson, J.K.; Barnes, T.; Cipriani, A.; Coghill, D.R.; Fazel, S.; Geddes, J.R.; Grunze, H.; et al. Evidence-based guidelines for treating bipolar disorder: Revised third edition recommendations from the British Association for Psychopharmacology. J. Psychopharmacol. 2016, 30, 495–553. [Google Scholar] [CrossRef] [PubMed]
- Baldessarini, R.J.; Tondo, L. Does lithium treatment still work? Evidence of stable responses over three decades. Arch. Gen. Psychiatry 2000, 57, 187–190. [Google Scholar] [CrossRef] [PubMed]
- Garnham, J.; Munro, A.; Slaney, C.; Macdougall, M.; Passmore, M.; Duffy, A.; O’Donovan, C.; Teehan, A.; Alda, M. Prophylactic treatment response in bipolar disorder: Results of a naturalistic observation study. J. Affect. Disord. 2007, 104, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Rybakowski, J.K.; Chlopocka-Wozniak, M.; Suwalska, A. The prophylactic effect of long-term lithium administration in bipolar patients entering treatment in the 1970s and 1980s. Bipolar Disord. 2001, 3, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, E.M.; Aubry, J.-M. Lithium: Updated human knowledge using an evidence-based approach: Part III: Clinical safety. CNS Drugs 2009, 23, 397–418. [Google Scholar] [CrossRef] [PubMed]
- Crisafulli, C.; Fabbri, C.; Porcelli, S.; Drago, A.; Spina, E.; De Ronchi, D.; Serretti, A. Pharmacogenetics of antidepressants. Front. Pharmacol. 2011, 2. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, M.H.; Fava, M.; Wisniewski, S.R.; Thase, M.E.; Quitkin, F.; Warden, D.; Ritz, L.; Nierenberg, A.A.; Lebowitz, B.D.; Biggs, M.M.; et al. Medication augmentation after the failure of SSRIs for depression. N. Engl. J. Med. 2006, 354, 1243–1252. [Google Scholar] [CrossRef] [PubMed]
- Abdolmaleky, H.M.; Nohesara, S.; Ghadirivasfi, M.; Lambert, A.W.; Ahmadkhaniha, H.; Ozturk, S.; Wong, C.K.; Shafa, R.; Mostafavi, A.; Thiagalingam, S. DNA hypermethylation of serotonin transporter gene promoter in drug naïve patients with schizophrenia. Schizophr. Res. 2014, 152, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Schneider, I.; Kugel, H.; Redlich, R.; Grotegerd, D.; Bürger, C.; Bürkner, P.-C.; Opel, N.; Dohm, K.; Zaremba, D.; Meinert, S.; et al. Association of serotonin transporter gene AluJb methylation with major depression, amygdala responsiveness, 5-HTTLPR/rs25531 polymorphism, and stress. Neuropsychopharmacology 2018, 43, 1308–1316. [Google Scholar] [CrossRef] [PubMed]
- Melas, P.A.; Rogdaki, M.; Ösby, U.; Schalling, M.; Lavebratt, C.; Ekström, T.J. Epigenetic aberrations in leukocytes of patients with schizophrenia: Association of global DNA methylation with antipsychotic drug treatment and disease onset. FASEB J. 2012, 26, 2712–2718. [Google Scholar] [CrossRef] [PubMed]
- D’Addario, C.; Dell’Osso, B.; Palazzo, M.C.; Benatti, B.; Lietti, L.; Cattaneo, E.; Galimberti, D.; Fenoglio, C.; Cortini, F.; Scarpini, E.; et al. Selective DNA methylation of BDNF promoter in bipolar disorder: Differences among patients with BDI and BDII. Neuropsychopharmacology 2012, 37, 1647–1655. [Google Scholar] [CrossRef] [PubMed]
- Starnawska, A.; Demontis, D.; McQuillin, A.; O’Brien, N.L.; Staunstrup, N.H.; Mors, O.; Nielsen, A.L.; Børglum, A.D.; Nyegaard, M. Hypomethylation of FAM63B in bipolar disorder patients. Clin. Epigenetics 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, H.; Murata, Y.; Ikegame, T.; Sawamura, R.; Shimanaga, S.; Takeoka, Y.; Saito, T.; Ikeda, M.; Yoshikawa, A.; Nishimura, F.; et al. DNA methylation analyses of the candidate genes identified by a methylome-wide association study revealed common epigenetic alterations in schizophrenia and bipolar disorder. Psychiatry Clin. Neurosci. 2018, 72, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Castle, D.J.; Galletly, C.A.; Dark, F.; Humberstone, V.; Morgan, V.A.; Killackey, E.; Kulkarni, J.; McGorry, P.; Nielssen, O.; Tran, N.T.; et al. The 2016 Royal Australian and New Zealand College of Psychiatrists guidelines for the management of schizophrenia and related disorders. Med. J. Aust. 2017, 206, 501–505. [Google Scholar] [CrossRef] [PubMed]
- Falkai, P.; Wobrock, T.; Lieberman, J.; Glenthoj, B.; Gattaz, W.F.; Möller, H.-J. WFSBP Task Force on Treatment Guidelines for Schizophrenia. World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for biological treatment of schizophrenia, part 2: Long-term treatment of schizophrenia. World J. Biol. Psychiatry 2006, 7, 5–40. [Google Scholar] [CrossRef] [PubMed]
- Lehman, A.F.; Lieberman, J.A.; Dixon, L.B.; McGlashan, T.H.; Miller, A.L.; Perkins, D.O.; Kreyenbuhl, J. American Psychiatric Association; Steering Committee on Practice Guidelines. Practice guideline for the treatment of patients with schizophrenia, second edition. Am. J. Psychiatry 2004, 161, 1–56. [Google Scholar] [PubMed]
- Henssler, J.; Kurschus, M.; Franklin, J.; Bschor, T.; Baethge, C. Long-term acute-phase treatment with antidepressants, 8 weeks and beyond. J. Clin. Psychiatry 2018, 79, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Malhi, G.S.; Gessler, D.; Outhred, T. The use of lithium for the treatment of bipolar disorder: Recommendations from clinical practice guidelines. J. Affect. Disord. 2017, 217, 266–280. [Google Scholar] [CrossRef] [PubMed]
- Phiel, C.J.; Zhang, F.; Huang, E.Y.; Guenther, M.G.; Lazar, M.A.; Klein, P.S. Histone deacetylase is a direct target of valproic acid, a potent anticonvulsant, mood stabilizer, and teratogen. J. Biol. Chem. 2001, 276, 36734–36741. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Zheng, S.-D.; Huang, H.-L.; Yan, L.-C.; Yin, X.-F.; Xu, H.-N.; Zhang, K.-J.; Gui, J.-H.; Chu, L.; Liu, X.-Y. Lithium down-regulates histone deacetylase 1 (HDAC1) and induces degradation of mutant huntingtin. J. Biol. Chem. 2013, 288, 35500–35510. [Google Scholar] [CrossRef] [PubMed]
- Matrisciano, F.; Dong, E.; Gavin, D.P.; Nicoletti, F.; Guidotti, A. Activation of group II metabotropic glutamate receptors promotes DNA demethylation in the mouse brain. Mol. Pharmacol. 2011, 80, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Labermaier, C.; Masana, M.; Müller, M.B. Biomarkers predicting antidepressant treatment response: How can we advance the field? Dis. Markers 2013, 35, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Fond, G.; d’Albis, M.-A.; Jamain, S.; Tamouza, R.; Arango, C.; Fleischhacker, W.W.; Glenthøj, B.; Leweke, M.; Lewis, S.; McGuire, P.; et al. The promise of biological markers for treatment response in first-episode psychosis: A systematic review. Schizophr. Bull. 2015, 41, 559–573. [Google Scholar] [CrossRef] [PubMed]
- Melka, M.G.; Castellani, C.A.; Laufer, B.I.; Rajakumar, R.N.; O’Reilly, R.; Singh, S.M. Olanzapine induced DNA methylation changes support the dopamine hypothesis of psychosis. J. Mol. Psychiatry 2013, 1. [Google Scholar] [CrossRef] [PubMed]
- Melka, M.G.; Castellani, C.A.; Rajakumar, N.; O’Reilly, R.; Singh, S.M. Olanzapine-induced methylation alters cadherin gene families and associated pathways implicated in psychosis. BMC Neurosci. 2014, 15, 112. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, H.; Bundo, M.; Asai, T.; Sunaga, F.; Ueda, J.; Ishigooka, J.; Kasai, K.; Kato, T.; Iwamoto, K. Effects of quetiapine on DNA methylation in neuroblastoma cells. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2015, 56, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Shimabukuro, M.; Jinno, Y.; Fuke, C.; Okazaki, Y. Haloperidol treatment induces tissue- and sex-specific changes in DNA methylation: A control study using rats. Behav. Brain Funct. 2006, 2, 37. [Google Scholar] [CrossRef] [PubMed]
- Dong, E.; Nelson, M.; Grayson, D.R.; Costa, E.; Guidotti, A. Clozapine and sulpiride but not haloperidol or olanzapine activate brain DNA demethylation. Proc. Natl. Acad. Sci. USA 2008, 105, 13614–13619. [Google Scholar] [CrossRef] [PubMed]
- Tremolizzo, L.; Doueiri, M.-S.; Dong, E.; Grayson, D.R.; Davis, J.; Pinna, G.; Tueting, P.; Rodriguez-Menendez, V.; Costa, E.; Guidotti, A. Valproate corrects the schizophrenia-like epigenetic behavioral modifications induced by methionine in mice. Biol. Psychiatry 2005, 57, 500–509. [Google Scholar] [CrossRef] [PubMed]
- Carrard, A.; Salzmann, A.; Malafosse, A.; Karege, F. Increased DNA methylation status of the serotonin receptor 5HTR1A gene promoter in schizophrenia and bipolar disorder. J. Affect. Disord. 2011, 132, 450–453. [Google Scholar] [CrossRef] [PubMed]
- Cruceanu, C.; Alda, M.; Grof, P.; Rouleau, G.A.; Turecki, G. Synapsin II is involved in the molecular pathway of lithium treatment in bipolar disorder. PLoS ONE 2012, 7, e32680. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.P.; Chen, Y.; Kundakovic, M.; Costa, E.; Grayson, D.R. Histone deacetylase inhibitors decrease reelin promoter methylation in vitro. J. Neurochem. 2005, 93, 483–492. [Google Scholar] [CrossRef] [PubMed]
- Pandey, G.; Dwivedi, Y.; SridharaRao, J.; Ren, X.; Janicak, P.G.; Sharma, R. Protein kinase C and phospholipase C activity and expression of their specific isozymes is decreased and expression of MARCKs is increased in platelets of bipolar but not in unipolar patients. Neuropsychopharmacology 2002, 26, 216–228. [Google Scholar] [CrossRef]
- Dwivedi, T.; Zhang, H. Lithium-induced neuroprotection is associated with epigenetic modification of specific BDNF gene promoter and altered expression of apoptotic-regulatory proteins. Front. Neurosci. 2014, 8, 457. [Google Scholar] [CrossRef] [PubMed]
- Aizawa, S.; Yamamuro, Y. Valproate administration to mice increases hippocampal p21 expression by altering genomic DNA methylation. Neuroreport 2015, 26, 915–920. [Google Scholar] [CrossRef] [PubMed]
- Perisic, T.; Zimmermann, N.; Kirmeier, T.; Asmus, M.; Tuorto, F.; Uhr, M.; Holsboer, F.; Rein, T.; Zschocke, J. Valproate and amitriptyline exert common and divergent influences on global and gene promoter-specific chromatin modifications in rat primary astrocytes. Neuropsychopharmacology 2010, 35, 792–805. [Google Scholar] [CrossRef] [PubMed]
- Popkie, A.P.; Zeidner, L.C.; Albrecht, A.M.; D’Ippolito, A.; Eckardt, S.; Newsom, D.E.; Groden, J.; Doble, B.W.; Aronow, B.; McLaughlin, K.J.; et al. Phosphatidylinositol 3-kinase (PI3K) signaling via glycogen synthase kinase-3 (Gsk-3) regulates DNA methylation of imprinted loci. J. Biol. Chem. 2010, 285, 41337–41347. [Google Scholar] [CrossRef] [PubMed]
- Asai, T.; Bundo, M.; Sugawara, H.; Sunaga, F.; Ueda, J.; Tanaka, G.; Ishigooka, J.; Kasai, K.; Kato, T.; Iwamoto, K. Effect of mood stabilizers on DNA methylation in human neuroblastoma cells. Int. J. Neuropsychopharmacol. 2013, 16, 2285–2294. [Google Scholar] [CrossRef] [PubMed]
- Melas, P.A.; Rogdaki, M.; Lennartsson, A.; Björk, K.; Qi, H.; Witasp, A.; Werme, M.; Wegener, G.; Mathé, A.A.; Svenningsson, P.; et al. Antidepressant treatment is associated with epigenetic alterations in the promoter of P11 in a genetic model of depression. Int. J. Neuropsychopharmacol. 2012, 15, 669–679. [Google Scholar] [CrossRef] [PubMed]
- Abdolmaleky, H.M.; Pajouhanfar, S.; Faghankhani, M.; Joghataei, M.T.; Mostafavi, A.; Thiagalingam, S. Antipsychotic drugs attenuate aberrant DNA methylation of DTNBP1 (dysbindin) promoter in saliva and post-mortem brain of patients with schizophrenia and Psychotic bipolar disorder. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2015, 168, 687–696. [Google Scholar] [CrossRef] [PubMed]
- Huzayyin, A.A.; Andreazza, A.C.; Turecki, G.; Cruceanu, C.; Rouleau, G.A.; Alda, M.; Young, L.T. Decreased global methylation in patients with bipolar disorder who respond to lithium. Int. J. Neuropsychopharmacol. 2014, 17, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Houtepen, L.C.; van Bergen, A.H.; Vinkers, C.H.; Boks, M.P.M. DNA methylation signatures of mood stabilizers and antipsychotics in bipolar disorder. Epigenomics 2016, 8, 197–208. [Google Scholar] [CrossRef] [PubMed]
- Backlund, L.; Wei, Y.B.; Martinsson, L.; Melas, P.A.; Liu, J.J.; Mu, N.; Östenson, C.-G.; Ekström, T.J.; Schalling, M.; Lavebratt, C. Mood stabilizers and the influence on global leukocyte DNA methylation in bipolar disorder. Mol. Neuropsychiatry 2015, 1, 76–81. [Google Scholar] [CrossRef] [PubMed]
- D’Addario, C.; Palazzo, M.C.; Benatti, B.; Grancini, B.; Pucci, M.; di Francesco, A.; Camuri, G.; Galimberti, D.; Fenoglio, C.; Scarpini, E.; et al. Regulation of gene transcription in bipolar disorders: Role of DNA methylation in the relationship between prodynorphin and brain derived neurotrophic factor. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 82, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Dellosso, B.; Daddario, C.; Palazzo, M.C.; Benatti, B.; Camuri, G.; Galimberti, D.; Fenoglio, C.; Scarpini, E.; Di Francesco, A.; Maccarrone, M.; et al. Epigenetic modulation of BDNF gene: Differences in DNA methylation between unipolar and bipolar patients. J. Affect. Disord. 2014, 166, 330–333. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, L.; Scheibelreiter, J.; Hassler, M.R.; Schloegelhofer, M.; Schmoeger, M.; Ludwig, B.; Kasper, S.; Aschauer, H.; Egger, G.; Schosser, A. Brain-derived neurotrophic factor (BDNF)-epigenetic regulation in unipolar and bipolar affective disorder. J. Affect. Disord. 2014, 168, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, N.; Nonen, S.; Kato, M.; Wakeno, M.; Takekita, Y.; Kinoshita, T.; Kugawa, F. Therapeutic response to paroxetine in major depressive disorder predicted by DNA methylation. Neuropsychobiology 2017, 75, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Dalton, C.F.; Srisawat, U.; Zhang, Z.J.; Reynolds, G.P. Methylation at a transcription factor-binding site on the 5-HT1A receptor gene correlates with negative symptom treatment response in first episode schizophrenia. Int. J. Neuropsychopharmacol. 2014, 17, 645–649. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, M.; Numata, S.; Tajima, A.; Yamamori, H.; Yasuda, Y.; Fujimoto, M.; Watanabe, S.; Umehara, H.; Shimodera, S.; Nakazawa, T.; et al. Effect of clozapine on DNA methylation in peripheral leukocytes from patients with treatment-resistant schizophrenia. Int. J. Mol. Sci. 2017, 18, 632. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Li, M.; Song, C.; Xu, Q.; Huo, R.; Shen, L.; Xing, Q.; Cui, D.; Li, W.; Zhao, J.; et al. Combined study of genetic and epigenetic biomarker risperidone treatment efficacy in Chinese Han schizophrenia patients. Transl. Psychiatry 2017, 7, e1170. [Google Scholar] [CrossRef] [PubMed]
- Rukova, B.; Staneva, R.; Hadjidekova, S.; Stamenov, G.; Milanova, V.; Toncheva, D. Whole genome methylation analyses of schizophrenia patients before and after treatment. Biotechnol. Biotechnol. Equip. 2014, 28, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.-J.; Kim, J.-M.; Stewart, R.; Kim, S.-Y.; Bae, K.-Y.; Kim, S.-W.; Shin, I.-S.; Shin, M.-G.; Yoon, J.-S. Association of SLC6A4 methylation with early adversity, characteristics and outcomes in depression. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2013, 44, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Okada, S.; Morinobu, S.; Fuchikami, M.; Segawa, M.; Yokomaku, K.; Kataoka, T.; Okamoto, Y.; Yamawaki, S.; Inoue, T.; Kusumi, I.; et al. The potential of SLC6A4 gene methylation analysis for the diagnosis and treatment of major depression. J. Psychiatr. Res. 2014, 53, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Domschke, K.; Tidow, N.; Schwarte, K.; Deckert, J.; Lesch, K.-P.; Arolt, V.; Zwanzger, P.; Baune, B.T. Serotonin transporter gene hypomethylation predicts impaired antidepressant treatment response. Int. J. Neuropsychopharmacol. 2014, 17, 1167–1176. [Google Scholar] [CrossRef] [PubMed]
- Domschke, K.; Tidow, N.; Schwarte, K.; Ziegler, C.; Lesch, K.-P.; Deckert, J.; Arolt, V.; Zwanzger, P.; Baune, B.T. Pharmacoepigenetics of depression: No major influence of MAO-A DNA methylation on treatment response. J. Neural Transm. 2015, 122, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Lv, Q.; Mao, Y.; Zhang, C.; Bao, C.; Sun, H.; Chen, H.; Yi, Z.; Cai, W.; Fang, Y. HTR1A/1B DNA methylation may predict escitalopram treatment response in depressed Chinese Han patients. J. Affect. Disord. 2018, 228, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Powell, T.R.; Smith, R.G.; Hackinger, S.; Schalkwyk, L.C.; Uher, R.; McGuffin, P.; Mill, J.; Tansey, K.E. DNA methylation in interleukin-11 predicts clinical response to antidepressants in GENDEP. Transl. Psychiatry 2013, 3, e300. [Google Scholar] [CrossRef] [PubMed]
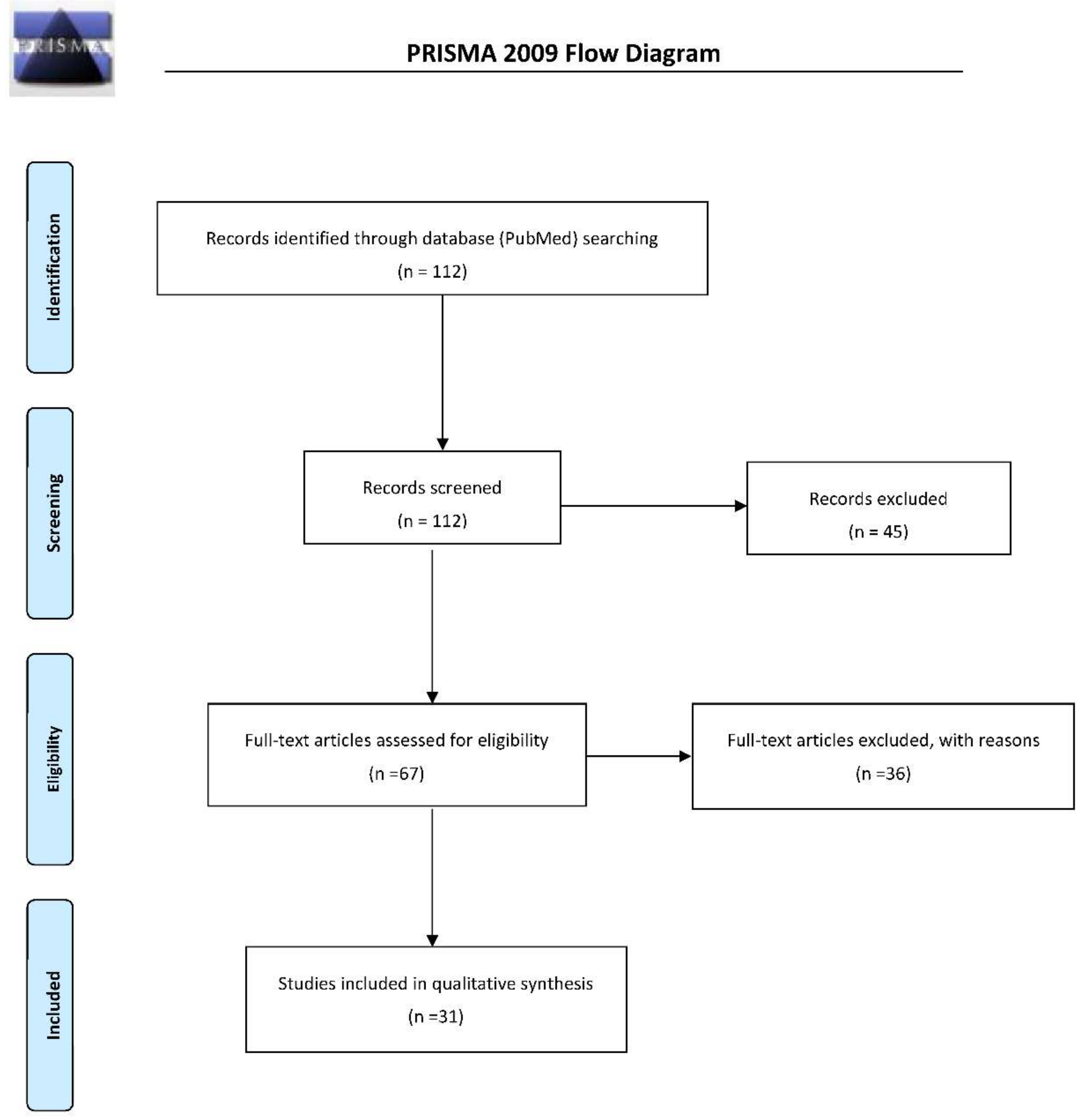
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef] [PubMed]
- Fries, G.R.; Quevedo, J.; Zeni, C.P.; Kazimi, I.F.; Zunta-Soares, G.; Spiker, D.E.; Bowden, C.L.; Walss-Bass, C.; Soares, J.C. Integrated transcriptome and methylome analysis in youth at high risk for bipolar disorder: A preliminary analysis. Transl. Psychiatry 2017, 7, e1059. [Google Scholar] [CrossRef] [PubMed]
- Ghadirivasfi, M.; Nohesara, S.; Ahmadkhaniha, H.-R.; Eskandari, M.-R.; Mostafavi, S.; Thiagalingam, S.; Abdolmaleky, H.M. Hypomethylation of the serotonin receptor type-2A Gene (HTR2A) at T102C polymorphic site in DNA derived from the saliva of patients with schizophrenia and bipolar disorder. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2011, 156, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Fries, G.R.; Bauer, I.E.; Scaini, G.; Wu, M.-J.; Kazimi, I.F.; Valvassori, S.S.; Zunta-Soares, G.; Walss-Bass, C.; Soares, J.C.; Quevedo, J. Accelerated epigenetic aging and mitochondrial DNA copy number in bipolar disorder. Transl. Psychiatry 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Kidnapillai, S.; Bortolasci, C.C.; Udawela, M.; Panizzutti, B.; Spolding, B.; Connor, T.; Sanigorski, A.; Dean, O.M.; Crowley, T.; Jamain, S.; et al. The use of a gene expression signature and connectivity map to repurpose drugs for bipolar disorder. World J. Biol. Psychiatry 2018, 1–9. [Google Scholar] [CrossRef] [PubMed]
| Model/Tissue | Method | Main Findings | Reference |
|---|---|---|---|
| Leukocytes/brain/liver tissues of rat | High-performance liquid chromatography | • Decreased mC in the brain of haloperidol-treated female rats Increased mC in liver DNA in haloperidol-treated male rats | [66] |
| Frontal cortex from a mouse model receiving l-methionine | Bisulfite conversion + PCR + sequencing | • VPA corrects Reln promoter hypermethylation induced in this SCZ-like model | [68] |
| Brian/liver tissues of rats | MeDIP | • 19 days of treatment with olanzapine increases DMA methylation at several dopaminergic genes | [63] |
| Mouse prefrontal cortex and striatum | MeDIP ChIP followed by qPCR | • VPA, clozapine, sulpiride, VPA + clozapine, and VPA + sulpiride treatment induce DNA demethylation of GAD67 and Reln genes | [67] |
| Human neuroblastoma cell lines SK-N-SH | Infinium HumanMethylation27 BeadChip + bisulfite sequencing | • Quetiapine decreases DNA methylation of the CpG3 island of SLC6A4 | [65] |
| Brian/liver tissues of rats | MeDIP | • Olanzapine alters DNA methylation at several cadherin/procadherin promoter region | [64] |
| Cultured rat hippocampal neurons | Methylation-specific PCR | • 48 h exposition to 1 and 2 mM lithium: 0.6-fold decrease DNA methylation at the promoter IV of BDNF • 2-fold increase of mRNA | [73] |
| Mouse hippocampal cells | PCR methylation-sensitive restriction site analysis | • 30% decrease of DNA methylation at the distal CpG island of the Cdkn p21 gene | [74] |
| Rat primary astrocytes | luminometric methylation analysis (LUMA) | • Decrease of DNA methylation at the Glt-1 promoter | [75] |
| Mouse embryonic and neural stem cells | Bisulfite sequencing | • 10 and 20 mM of lithium induce a 66% decrease of DNA methylation at the Igf2/H19 differentially methylated domain (DMD) in embryonic stem cells • 5 mM of lithium induce a 33% decrease of DNA methylation at the Igf2/H19 DMD in neural stem cells | [76] |
| Human neuroblastoma cell lines SK-N-SH | Infinium HumanMethylation27 BeadChip | • Hypermethylation of 345 genes (lithium), 64 genes (VPA), and 64 genes (carbamazepine) • Hypomethylation of 138 genes (lithium), 36 genes (VPA), and 14 genes (carbamazepine) | [77] |
| Model/Tissue | Method | Main Findings | Reference |
|---|---|---|---|
| Leukocytes of SCZ patients (n = 177) and controls (n = 171) | luminometric methylation analysis (LUMA) | • Increased global methylation in patients treated with haloperidol compared to other treatments | [49] |
| Saliva samples of SCZ (n = 30), first-degree relatives of SCZ (n = 15), controls (n = 30)/postmortem brain samples of patients with SCZ (n = 35) and BD (n = 35) | Quantitative methylation specific PCR | • Increased DNA methylation of DTNBP1 promoter in the saliva of patients with SCZ compared to controls • Inverse correlation between DTNBP1 methylation and expression in post-mortem brains of SCZ patients • Trend to reduced DNA methylation of DTNBP1 by antipsychotics treatment | [79] |
| Postmortem brain samples of patients with BD (n = 35) and controls (n = 35) | Quantitative methylation specific PCR | • No significant difference of the DTNBP1 promoter region associated with antipsychotic treatment in patients with BD | [79] |
| Transformed lymphoblast cell lines from: lithium responders BD (n = 14), affected relatives (n = 14), unaffected relatives (n = 16), Healthy controls (n = 16) | ELISA | • Decreased DNA methylation in cell lines of BD patients, affected and unaffected relatives, compared to healthy controls • Lithium-induced decrease in global DNA methylation in BD patients (lithium responders) compared to controls | [80] |
| Whole blood of patients with BD (n = 172), Human | Infinium Human-Methylation450 BeadChip | • Quetiapine, VPA showed significant DNA methylation alterations patients with BD | [81] |
| Peripheral blood from BD patients on Li monotherapy (n = 29), Lithium + VPA (n = 11), Lithium + antipsychotics (n = 21), healthy controls (n = 26) | ELISA | • Hypomethylation of DNA in BD patients treated with lithium monotherapy vs. lithium + VPA or healthy controls • No significant relation between DNA methylation and lithium response | [82] |
| PBMC from BDI (n = 45), BDII (n = 49), and control subjects (n = 52) | Methylation-specific qPCR | • Decrease of DNA methylation at BDNF promoter I in patients with antidepressant therapy vs. controls • Decrease of DNA methylation at BDNF promoter I in patients with lithium therapy vs. other medications • Decrease of DNA methylation at BDNF promoter I in patients with VPA therapy vs. other medications | [50] |
| PBMC from BDI (n = 45), BDII (n = 45) | methylation specific qPCR | • Decrease of DNA methylation at PDYN promoter I in patients with lithium or VPA therapy (n = 25) vs. other medications | [83] |
| PBMC from BDI (n = 61), BDII (n = 50), MDD (n = 43) patients | methylation specific qPCR | • Not significant trend for a decrease of DNA methylation in patients treated with lithium and VPA | [84] |
| PBMC from MDD (n = 207), BD (n = 59) and controls (n = 278) | Methylation-specific quantitative PCR | • BDNF gene exon I promoter methylation increased in MDD compared to BD and controls • Increased BDNF DNA methylation in MDD patients associated with antidepressant therapy | [85] |
| Peripheral leukocytes from MDD patients (10 best responders and 10 worst responders to paroxetine) | Infinium Human-Methylation450 BeadChip | • Methylation levels of the CpG sites in PPFIA4 and HS3ST1 gene can discriminate between best and worst responders | [86] |
| Model/Tissue | Method | Main Findings | Reference |
|---|---|---|---|
| Peripheral blood of patients with SCZ (n = 82) | Bisulfite conversion + PCR + pyrosequencing | • Decreased DNA methylation at CpG13 of HTR1A associated with poorer response to antipsychotics | [87] |
| Peripheral blood from SCZ patients (n = 21) | Infinium Human-Methylation450 BeadChip | • Clozapine-induced DNA methylation changes in the CREBBP gene were significantly correlated with clinical improvements | [88] |
| Peripheral blood from SCZ patients; good responders (n = 88), poor responders (n = 54) | Methylation-specific PCR + mass spectrometry | • Seven CpGs at CYP3A4 and CYP2D6 genes were differentially methylated in good vs. poor responders to risperidone therapy | [89] |
| Peripheral blood from SCZ patients n = 20 (12 M/8 F) | MeDIP ChIP | • Before treatment: nine genes with DMR in male SCZ patients in complete remission after treatment (vs. matched control subjects) • After treatment: six genes with DMR in male SCZ patients in complete remission after treatment (vs. matched control subjects) • Before treatment: one gene (M1R181C) with DMR in female SCZ patients in complete remission after treatment (vs. matched control subjects) • After treatment: one gene (BCOR) with DMR in female SCZ patients in complete remission after treatment (vs. matched control subjects) | [90] |
| Leukocytes from patients with MDD (n = 108) | Bisulfite conversion followed by PCR | • Increased of SLC6A4 DNA methylation level associated with impaired treatment response to antidepressants | [91] |
| Whole blood from patients with MDD (n = 50 before treatment and n = 40 after 6 weeks of treatment) and controls (n = 50) | Methylation-specific PCR + mass spectrometry | • Methylation level of the third CpG site of SLC6A4 gene association with better therapeutic response to antidepressant therapy in patients MD | [92] |
| Whole blood from patients with MDD (n = 94), (Human) | Bisulfite conversion + PCR | • Increased DNA methylation of SLC6A4 gene associated with better treatment response to escitalopram | [93] |
| Whole blood from patients with MDD, (n = 61) (Human) | Bisulfite conversion + PCR | • No major influence of mono amino oxidase (MAO-A) gene methylation status on escitalopram response | [94] |
| Peripheral blood of patients with MDD (n = 85) | Bisulfite sequencing | • Significant association of 668 CpG sites of HTR1A and 1401 CpG sites of HTR1B gene methylation with treatment response to escitalopram | [95] |
| Peripheral blood samples of patients with MDD treated with escitalopram (n = 80) or nortriptyline (n = 33) | Bisulfite conversion + PCR | • Fourth CpG island hypomethylation of IL-11 gene associated with better response to nortriptyline • Hypermethylation of fourth CpG island of IL-11 gene associated with better response to escitalopram | [96] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goud Alladi, C.; Etain, B.; Bellivier, F.; Marie-Claire, C. DNA Methylation as a Biomarker of Treatment Response Variability in Serious Mental Illnesses: A Systematic Review Focused on Bipolar Disorder, Schizophrenia, and Major Depressive Disorder. Int. J. Mol. Sci. 2018, 19, 3026. https://doi.org/10.3390/ijms19103026
Goud Alladi C, Etain B, Bellivier F, Marie-Claire C. DNA Methylation as a Biomarker of Treatment Response Variability in Serious Mental Illnesses: A Systematic Review Focused on Bipolar Disorder, Schizophrenia, and Major Depressive Disorder. International Journal of Molecular Sciences. 2018; 19(10):3026. https://doi.org/10.3390/ijms19103026
Chicago/Turabian StyleGoud Alladi, Charanraj, Bruno Etain, Frank Bellivier, and Cynthia Marie-Claire. 2018. "DNA Methylation as a Biomarker of Treatment Response Variability in Serious Mental Illnesses: A Systematic Review Focused on Bipolar Disorder, Schizophrenia, and Major Depressive Disorder" International Journal of Molecular Sciences 19, no. 10: 3026. https://doi.org/10.3390/ijms19103026
APA StyleGoud Alladi, C., Etain, B., Bellivier, F., & Marie-Claire, C. (2018). DNA Methylation as a Biomarker of Treatment Response Variability in Serious Mental Illnesses: A Systematic Review Focused on Bipolar Disorder, Schizophrenia, and Major Depressive Disorder. International Journal of Molecular Sciences, 19(10), 3026. https://doi.org/10.3390/ijms19103026





