Negative Air Ions and Their Effects on Human Health and Air Quality Improvement
Abstract
1. Introduction
2. Systematic Review of Literatures
3. Negative Air Ions and Their Generation
3.1. Radiant or Cosmic Rays in the Atmosphere
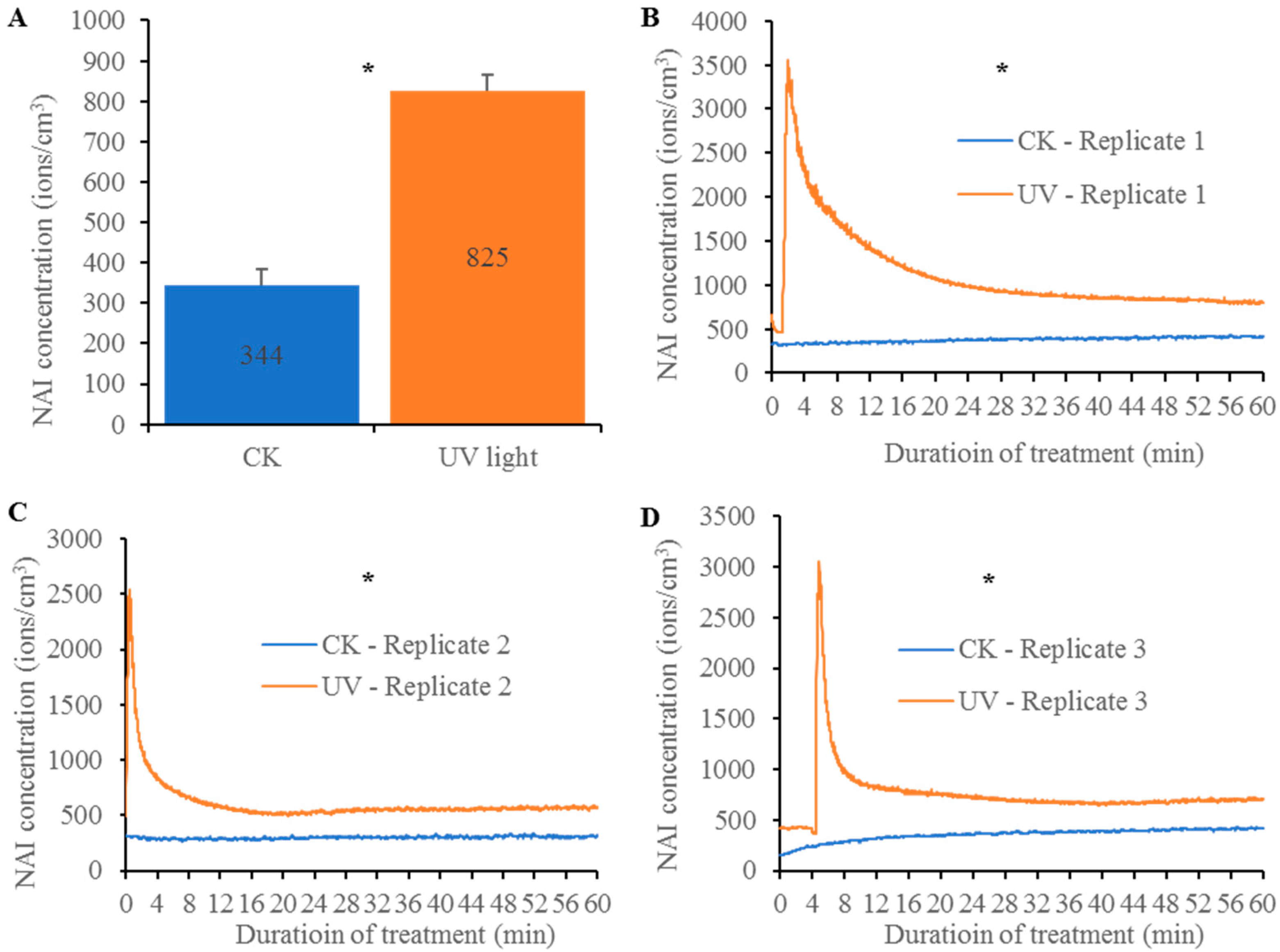
3.2. Sunlight Including Ultraviolet
3.3. Natural and Artificial Corona Discharge Including Thunderstorms and Lightning
3.4. The Shearing Forces of Water (Lenard Effect)
3.5. Plant-Based NAI Release under Normal Growth Conditions and by Pulse Electric Stimulation
3.6. The Fate of Generated NAIs
4. Plants as a Source for Generating NAIs
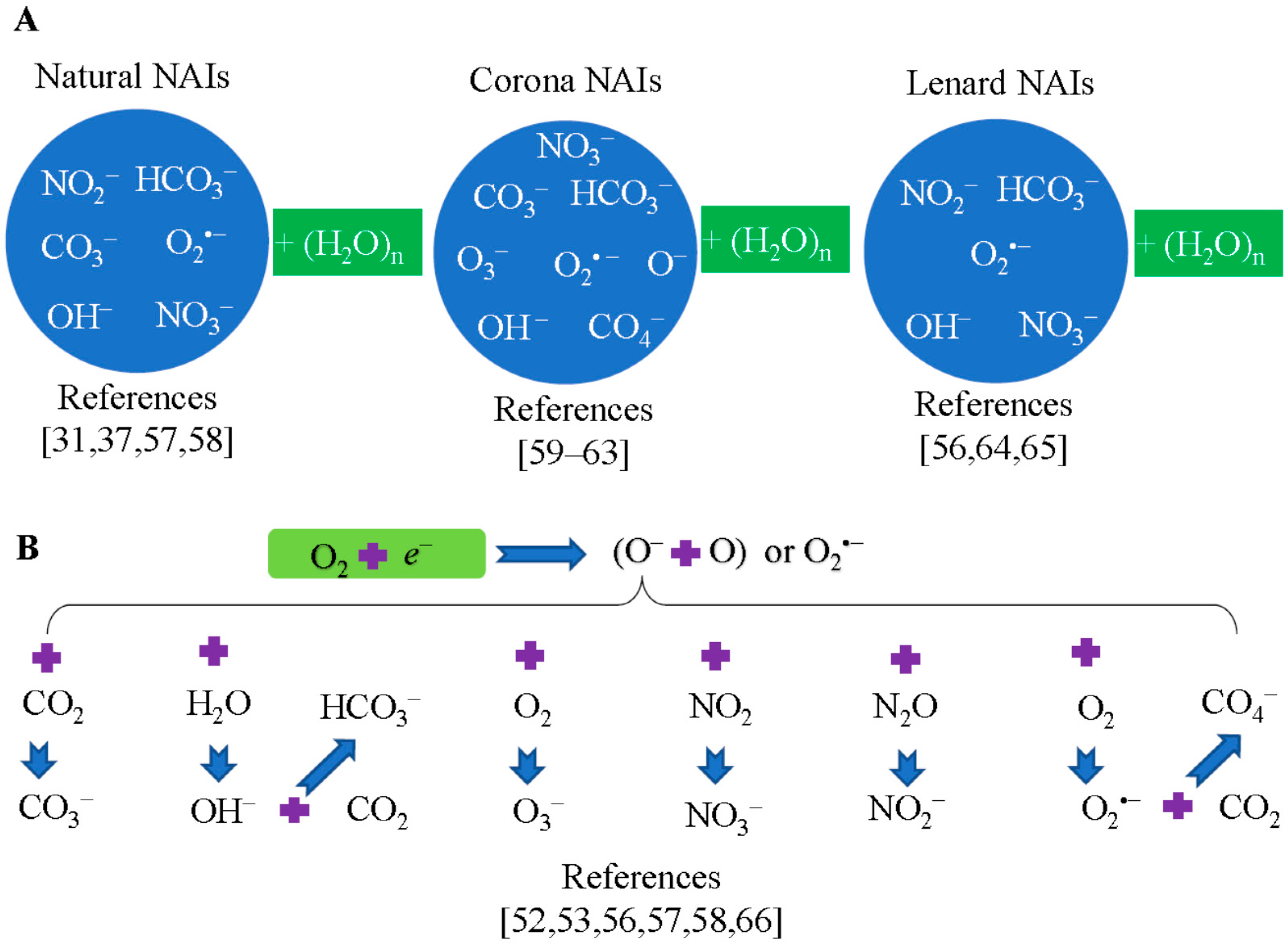
5. Composition of Negative Air Ions
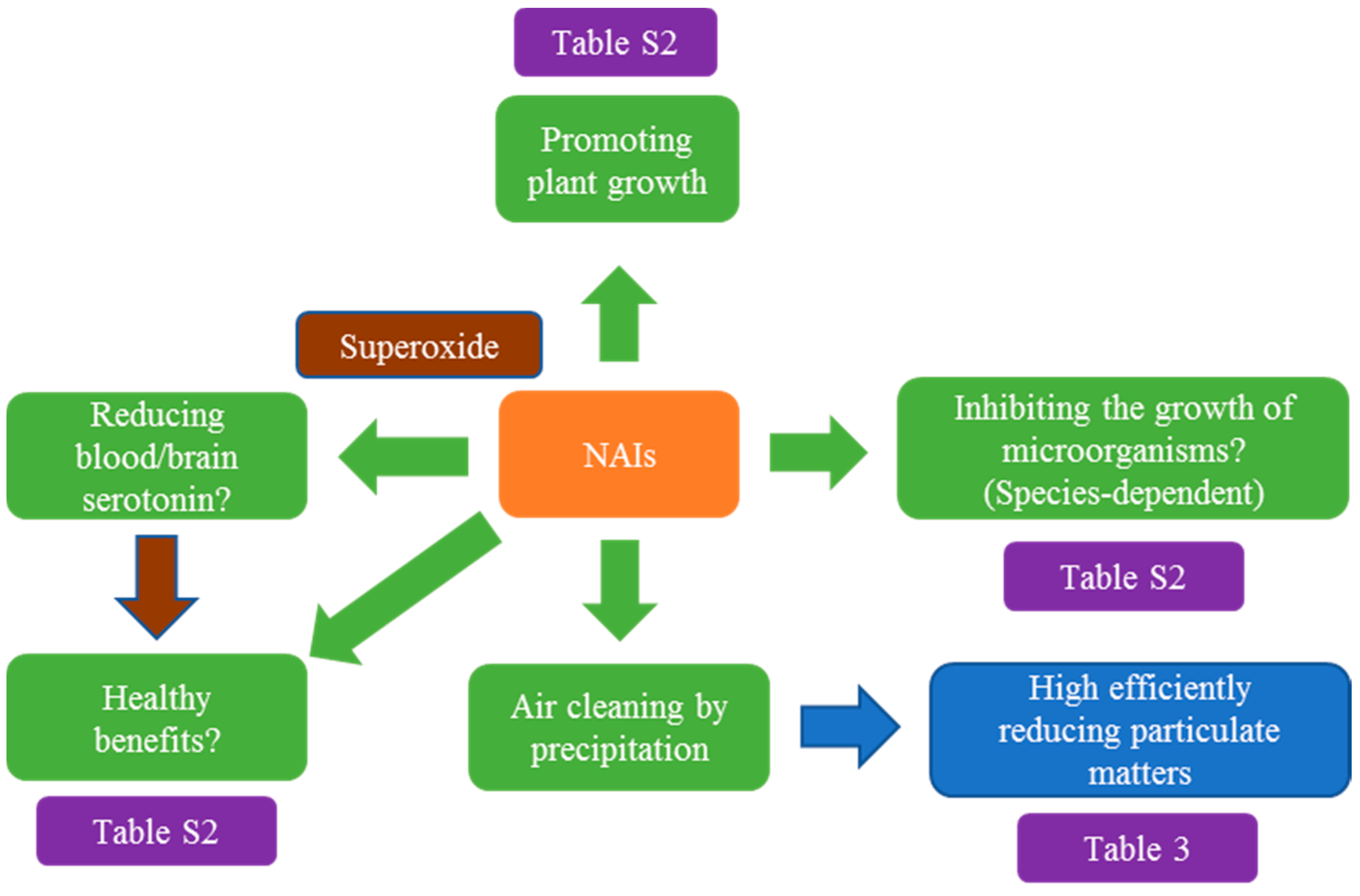
6. Biological Effects of Negative Air Ions on Human/Animal Health and Microorganism Growth as well as Plant Development
7. Superoxide Involvement in the Biological Effects of Negative Air Ions
8. The Release of Negative Air Ions as an Efficient Way to Remove Fine Particulates
9. Prospects and Future Works on Plant-Based NAI Generator
10. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| NAI | Negative air ion |
| PM | Particulate matter |
| PEF | Pulsed electric field |
| UV | Ultraviolet |
| SA | Salicylic acid |
| MV | Methyl viologen |
| KV | Kilovoltage |
| RANIs | Releasing amounts of negative ions |
| PVC | Polyvinyl chloride |
References
- Krueger, A.P.; Reed, E.J. Biological impact of small air ions. Science 1976, 193, 1209–1213. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, N.; Arshavskaya, T.V. Is atmospheric super oxide Vitally necessary? Accelerated death of animals in a quasi-neutral electric atmosphere. Z. Naturforsch C. 1997, 52, 396–404. [Google Scholar] [CrossRef] [PubMed]
- Laza, V. The environment and gaseous ions. Cent. Eur. J. Occup. Environ. Med. 2000, 6, 3–10. [Google Scholar]
- Hoppel, W.A.; Anderson, R.V.; Willet, J.C. Atmospheric Electricity in the Planetary Boundary Layer, The Earth’s Electrical Environment; NAS Press: Washington, DC, USA, 1986; pp. 195–205. [Google Scholar]
- Harrison, R.G.; Carslaw, K.S. Ion-aerosol-cloud processes in the lower atmosphere. Rev. Geophys. 2003, 41. [Google Scholar] [CrossRef]
- Chalmers, J.A. Atmospheric Electricity, 2nd ed.; Pergamon Press: Oxford, UK; New York, NY, USA, 1967. [Google Scholar]
- Ermakov, V.I.; Bazilevskaya, G.A.; Potrevsky, P.E.; Stozhkov, Y.I. Ion balance equation in the atmosphere. J. Geophys. Res. 1997, 102, 23413–23419. [Google Scholar] [CrossRef]
- Yates, A.; Gray, F.B.; Misiaszek, J.I.; Wolman, W. Air ions: Past problems and future directions. Environ Int. 1986, 12, 99–108. [Google Scholar] [CrossRef]
- Borra, J.P.; Roos, R.A.; Renard, D.; Lazar, H.; Goldman, A.; Goldman, M. Electrical and chemical consequences of point discharges in a forest during a mist and a thunderstorm. J. Phys. D: Appl. Phys. 1997, 30, 84–93. [Google Scholar] [CrossRef]
- Aubrecht, L.; Koller, J.; Stanek, Z. Onset voltages of atmospheric corona discharges on plants. Czech. J. Phys. 2000, 50, 313–318. [Google Scholar] [CrossRef]
- Aubrecht, L.; Stanek, Z.; Koller, J. Corona discharge on coniferous trees—Spruce and pine. Europhys. Lett. 2001, 53, 304–390. [Google Scholar] [CrossRef]
- Altamimi, G.; Illias, H.A.; Mokhtar, N.; Mokhlis, H.; Bakar, A.H.A. Corona discharges under various types of electrodes. In Proceedings of the 2014 IEEE International Conference on Power and Energy (PECon), Kuching Sarawak, Malaysia, 1–3 December 2014; pp. 5–8. [Google Scholar] [CrossRef]
- Ogar, V.N.; Bendor, S.A.; James, A.E. Analysis of corona effect on transmission line. Am. J. Eng. Res. 2017, 6, 75–87. [Google Scholar]
- Berendt, A.; Budnarowska, M.; Mizeraczyk, J. DC negative corona discharge characteristics in air flowing transversely and longitudinally through a needle-plate electrode gap. J. Electrostat. 2018, 92, 24–30. [Google Scholar] [CrossRef]
- Sharma, S.; Goel, K.; Gupta, A.; Kumar, H. Corona effects on eHv AC transmission lines. Int. J. Sci. Res. Eng. Technol. 2012, 1, 160–164. [Google Scholar]
- Manabe, Y.; Shimazaki, T. Formation mechanism of surface corona on dielectric plates under negative impulse voltage in atmospheric air. IEEE Trans. Dielectr. Electr. Insul. 2004, 11, 631–637. [Google Scholar] [CrossRef]
- Murr, L.E. Plant growth response in a simulated electric field-environment. Nature 1963, 200, 490–491. [Google Scholar] [CrossRef]
- Murr, L.E. Biophysics of plant growth in an electrostatic field. Nature 1965, 206, 467–470. [Google Scholar] [CrossRef]
- Bachman, C.H.; Hademenos, D.G. Ozone and air ions accompanying biological applications of electric fields. J. Atmos. Terr. Phys. 1971, 33, 497–505. [Google Scholar] [CrossRef]
- Krueger, A.P.; Strubbe, A.E.; Yost, M.G.; Reed, E.J. Electric fields, small air ions and biological effects. Int. J. Biometeorol. 1978, 22, 202–212. [Google Scholar] [CrossRef]
- Hart, F.X.; Schottenfeld, R.S. Evaporation and plant damage in electric fields. Int. J. Biometeor. 1979, 23, 63–68. [Google Scholar] [CrossRef]
- Hart, F.X.; Mudano, M.J.; Atchley, A.A. Plant damage produced by the passage of low level direct current. II. Further observations and possible mechanisms. Int. J. Biometeor. 1981, 25, 151–159. [Google Scholar] [CrossRef]
- Lenard, P. Über die Electrizität der Wasserfälle. Ann. Phys. 1892, 46, 584–636. [Google Scholar] [CrossRef]
- Iwama, H. Negative air ions created by water shearing improve erythrocyte deformability and aerobic metabolism. Indoor Air 2004, 14, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Kosenko, E.A.; Kaminsky, Y.G.; Stavrovskaya, I.G.; Sirota, T.V.; Kondrashova, M.N. The stimulatory effect of negative air ions and hydrogen peroxide on the activity of superoxide dismutase. FEBS Lett. 1997, 410, 309–312. [Google Scholar] [CrossRef]
- Nemeryuk, G.E. Migration of salts into the atmosphere during transpiration. Sov. Plant Physiol. (Engl. Transl.) 1970, 17, 560–566. [Google Scholar]
- Wang, J.; Li, S.H. Changes in negative air ions concentration under different light intensities and development of a model to relate light intensity to directional change. J. Environ. Manag. 2009, 90, 2746–2754. [Google Scholar] [CrossRef] [PubMed]
- Tikhonov, V.P.; Tsvetkov, V.D.; Litvinova, E.G.; Sirota, T.V.; Kondrashova, M.N. Generation of negative air ions by plants upon pulsed stimulation applied to soil. J. Plant Physiol. 2004, 51, 414–419. [Google Scholar] [CrossRef]
- Zhu, M.; Zhang, J.; You, Q.; Banuelos, G.S.; Yu, Z.; Li, M.; Bazai, N.A.; Tahir, M. Bio-generation of negative air ions by grass upon electrical stimulation applied to lawn. Fresen. Environ. Bull. 2016, 25, 2071–2078. [Google Scholar]
- Wu, R.; Zheng, J.; Sun, Y.; Wang, Q.; Deng, C.; Ye, D. Research on generation of negative air ions by plants and stomatal characteristics under pulsed electrical field stimulation. Int. J. Agric. Biol. 2017, 19, 1235–1245. [Google Scholar] [CrossRef]
- Guerra, L. A new instrument for the measurement of air ions. Int. J. Biometeorol. 1988, 32, 11–16. [Google Scholar] [CrossRef]
- Iwama, H.; Ohmizo, H.; Furuta, S.; Ohmori, S.; Watanabe, K.; Kaneko, T.; Tsutsumi, K. Inspired superoxide anions attenuate blood lactate concentrations in postoperative patients. Crit. Care Med. 2002, 30, 1246–1249. [Google Scholar] [CrossRef] [PubMed]
- Yamada, R.; Yanoma, S.; Akaike, M.; Tsuburaya, A.; Sugimasa, Y.; Takemiya, S.; Motohashi, H.; Rino, Y.; Takanashi, Y.; Imada, T. Water-generated negative air ions activate NK cell and inhibit carcinogenesis in mice. Cancer Lett. 2006, 239, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.C.; Lee, G.W.M.; Cheng, P.; Yang, S.; Yu, K.P. Effect of wall surface materials on deposition of particles with the aid of negative air ions. Aerosol Sci. 2006, 37, 616–630. [Google Scholar] [CrossRef]
- Bailey, W.H.; Williams, A.L.; Leonhard, M.J. Exposure of laboratory animals to small air ions: A systematic review of biological and behavioral studies. BioMed. Eng. online 2018, 17, 72. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, I.; Ben-Hur, E. Superoxide involvement in negative air ion effects. Nature 1980, 288, 739–740. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, N. Reactive oxygen species as essential components of ambient air. Biochemistry 2002, 67, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Pino, O.; Ragione, F.L. There’s something in the air: Empirical evidence for the effects of negative air ions (NAI) on psychophysiological state and performance. Res. Psychol. Behav Sci. 2013, 1, 48–53. [Google Scholar]
- Scarpeci, T.E.; Zanor, M.I.; Carrillo, N.; Mueller-Roeber, B.; Valle, E.M. Generation of superoxide anion in chloroplasts of Arabidopsis thaliana during active photosynthesis: A focus on rapidly induced genes. Plant Mol. Biol. 2008, 66, 361–378. [Google Scholar] [CrossRef] [PubMed]
- Ananyev, G.M.; Renger, G.; Wacker, U.; Klimov, V.V. Photoreduction of superoxide radicals and the superoxide dismutase activity of Photosystem II. The possible involvement of cytochrome b559. Photosynth. Res. 1994, 41, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Cleland, R.E.; Grace, S.C. Voltammetric detection of superoxide production by photosystem II. FEBS Lett. 1999, 457, 348–352. [Google Scholar] [CrossRef]
- Pospíšil, P.; Šnyrychova, I.; Kruk, J.; Strzałka, K.; Nauš, J. Evidence that cytochrome b559 is involved in superoxide production in photosystem II: Effect of synthetic short-chain plastoquinone in a cytochrome b559 tobacco mutant. Biochem. J. 2006, 397, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Allan, A.; Fluhr, R. Two distinct sources of elicited reactive oxygen species in tobacco epidermal cells. Plant Cell 1997, 9, 1559–1577. [Google Scholar] [CrossRef] [PubMed]
- Bolwell, G.P.; Davies, D.R.; Gerrish, C.; Auh, C.-K.; Murphy, T.M. Comparative biochemistry of the oxidative burst produced by rose and french bean cells reveals two distinct mechanisms. Plant Physiol. 1998, 116, 1379–1385. [Google Scholar] [CrossRef] [PubMed]
- Minibayeva, F.; Beckett, R.P. High rates of extracellular superoxide production in bryophytes and lichens, and an oxidative burst in response to rehydration following desiccation. New Phytol. 2001, 152, 333–343. [Google Scholar] [CrossRef]
- Asada, K.; Takahashi, M. Production and Scavenging of Active Oxygens in Photosynthesis. In Photoinhibition; Kyle, D.J., Osmond, C.B., Arntzen, D.J., Eds.; Elsevier/North Holland Biomedical Press: Amsterdam, The Netherlands, 1987; pp. 227–280. [Google Scholar]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine; Oxford University Press: Oxford, UK, 1989. [Google Scholar]
- Kawano, T.; Sahashi, N.; Takahashi, K.; Uozumi, N.; Muto, S. Salicylic acid induces extracellular superoxide generation followed by an increase in cytosolic calcium ion in tobacco suspension culture: The earliest events in salicylic acid signal transduction. Plant Cell Physiol. 1998, 39, 721–730. [Google Scholar] [CrossRef]
- Park, J.; Gu, Y.; Lee, Y.; Yang, Z.; Lee, Y. Phosphatidic acid induces leaf cell death in Arabidopsis by activating the Rho-related small G protein GTPase-mediated pathway of reactive oxygen species generation. Plant Physiol. 2004, 134, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Zulfugarov, I.S.; Tovuu, A.; Eu, Y.J.; Dogsom, B.; Poudyal, R.S.; Nath, K.; Hall, M.; Banerjee, M.; Yoon, U.C.; Moon, Y.H.; et al. Production of superoxide from Photosystem II in a rice (Oryza sativa L.) mutant lacking PsbS. BMC Plant Biol. 2014, 14, 242. [Google Scholar] [CrossRef] [PubMed]
- Skromulis, A.; Noviks, G. Atmospheric light air ion concentrations and related meteorologic factors in Rezekne city, Latvia. J. Environ. Biol. 2012, 33, 455–462. [Google Scholar] [PubMed]
- Gravendeel, B.; de Hoog, F.J. Clustered negative ions in atmospheric negative corona discharges in the Trichel regime. J. Phys. B: Atom. Mol. Phys. 1999, 20, 6337–6361. [Google Scholar] [CrossRef]
- Luts, A.; Parts, T. Evolution of negative small air ions at two different temperatures. J. Atmos. Sol-Terr. Phy. 2002, 64, 763–774. [Google Scholar] [CrossRef]
- Dillard, J.G. Negative ion mass spectrometry. Chem. Rev. 1973, 73, 589–643. [Google Scholar] [CrossRef]
- Skalny, J.D.; Orszagh, J.; Mason, N.J.; John, R.; Yolanda, A.G.; Whitmore, T.D. Mass spectrometric study of negative ions extracted from point to plane negative corona discharge in ambient air at atmospheric pressure. Int. J. Mass Spectrom. 2008, 272, 12–21. [Google Scholar] [CrossRef]
- Parts, T.E.; Luts, A.; Laakso, L.; Hirsikko, A.; Gronholm, T.; Kulmala, M. Chemical composition of waterfall-induced air ions: Spectrometry vs. simulations. Boreal Environ. Res. 2007, 12, 409–420. [Google Scholar]
- Cabane, M.; Playe, P. Mass spectra of negative ions in air-like gas mixtures at atmospheric pressure. J. Aerosol Sci. 1980, 11, 475–482. [Google Scholar] [CrossRef]
- Lin, H.F.; Lin, J.M. Generation and determination of negative air ions. J. Anal. Test. 2017, 1, 6. [Google Scholar] [CrossRef]
- Goldman, M.; Goldman, A.; Sigmond, R.S. The corona discharge, its properties and specific uses. Pure Appl. Chem. 1985, 57, 1353–1362. [Google Scholar] [CrossRef]
- Sauers, I.; Harman, G. A mass spectrometric study of positive and negative ion formation in an sF6 corona. Part II: Influence of water and sF6 neutral by-products. J. Phys. D: Appl. Phys. 1992, 25, 774–782. [Google Scholar] [CrossRef]
- Stano, M.; Safonov, E.; Kucera, M.; Matejcik, S. Ion mobility spectrometry study of negative corona discharge in oxygen/nitrogen mixtures. Chemicke Listy. 2008, 102, s1414–s1417. [Google Scholar]
- Manninen, H.E.; Franchin, A.; Schobesberger, S.; Hirsikko, A.; Hakala, J.; Skromulis, A.; Kangasluoma, J.; Ehn, M.; Junninen, H.; Mirme, A.; et al. Characterization of corona-generated ions used in a Neutral cluster and Air Ion Spectrometer (NAIS). Atmos. Meas. Tech. 2011, 4, 2767–2776. [Google Scholar] [CrossRef]
- Sekimoto, K.; Takayama, M. Collision-induced dissociation analysis of negative atmospheric ion adducts in atmospheric pressure corona discharge ionization mass spectrometry. J. Am. Soc. Mass Spectrom. 2013, 24, 780–788. [Google Scholar] [CrossRef] [PubMed]
- Laakso, L.; Hirsikko, A.; Grönholm, T.; Kulmala, M.; Luts, A.; Parts, T.E. Waterfalls as sources of small charged aerosol particles. Atmos. Chem. Phys. 2006, 6, 9297–9314. [Google Scholar] [CrossRef]
- Kolarz, P.; Gaisberger, M.; Madl, P.; Hofmann, W.; Ritter, M.; Hartl, A. Characterization of ions at alpine waterfalls. atmos. Chem. Phys. 2012, 12, 3687–3697. [Google Scholar] [CrossRef]
- Skalny, J.D.; Horvath, G.; Masona, N.L. Mass spectrometric analysis of small negative ions (e/m < 100) produced by trichel pulse negative corona discharge fed by ozonised air. J. Optoelectron. Adv. Mater. 2007, 9, 887–893. [Google Scholar]
- Shahin, M.M. Nature of charge carriers in negative coronas. Appl. Opt. 1969, 8, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Gardiner, P.S.; Moruzzi, J.L.; Craggs, J.D. Negative ions in Trichel corona in carbon dioxide and oxygen. J. Phys. D: Appl. Phys. 1978, 11, 237–244. [Google Scholar] [CrossRef]
- Gardiner, P.S.; Cragss, J.D. Negative ions in Trichel corona in air. J. Phys. D: Appl. Phys. 1977, 10, 1003–1009. [Google Scholar] [CrossRef]
- Sakata, S.; Okada, T. Effect of humidity on hydrated cluster-ion formation in a clean room corona discharge neutralizer. J. Aerosol. Sci. 1994, 25, 879–885. [Google Scholar] [CrossRef]
- Skalnya, J.D.; Mikoviny, T.; Matejcik, S.; Mason, N.J. An analysis of mass spectrometric study of negative ions extracted from negative corona discharge in air. Int. J. Mass Spectrom. 2004, 233, 317–324. [Google Scholar] [CrossRef]
- Hill, C.A.; Thomas, C.L. A pulsed corona discharge switchable high resolution ion mobility spectrometer-mass spectrometer. Analyst 2003, 128, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.K.; Bell, A.J. Reverse flow continuous corona discharge ionisation applied to ion mobility spectrometry. Int. J. Mass Spectrom. 2002, 218, L1–L6. [Google Scholar] [CrossRef]
- Nagato, K.; Matsui, Y.; Miyata, T.; Yamauchi, T. An analysis of the evolution of negative ions produced by a corona ionizer in air. Int. J. Mass Spectrom. 2006, 248, 142–147. [Google Scholar] [CrossRef]
- Charry, J.M.; Kavet, R. Air Ions: Physical and Biological Aspects; CRC Press: Boca Raton, FL, USA, 1987. [Google Scholar]
- Diamond, M.C.; Connor, J.R., Jr.; Orenberg, E.K.; Bissell, M.; Yost, M.; Krueger, A. Environmental influences on serotonin and cyclic nucleotides in rat cerebral cortex. Science 1980, 210, 652–654. [Google Scholar] [CrossRef] [PubMed]
- Ryushi, T.; Kita, I.; Sakurai, T.; Yasumatsu, M.; Isokawa, M.; Aihara, Y.; Hama, K. The effect of exposure to negative air ions on the recovery of physiological responses after moderate endurance exercise. Int. J. Biometeorol. 1998, 41, 132–136. [Google Scholar] [CrossRef] [PubMed]
- Wrona, M.Z.; Dryhurst, G. Oxidation of serotonin by superoxide radical: Implications to neurodegenerative brain disorders. Chem. Res. Toxicol. 1998, 11, 639–650. [Google Scholar] [CrossRef] [PubMed]
- Bailey, W.H.; Charry, J.M. Acute exposure of rats to air ions: Effects on the regional concentration and utilization of serotonin in brain. Bioelectromagnetics 1987, 8, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Lazzerini, F.T.; Orlando, M.T.; De Prá, W. Progress of negative air ions in health tourism environments applications. Bol. Soc. Esp. Hidrol. Méd. 2018, 33, 27–46. [Google Scholar] [CrossRef]
- Ju, K.; Kubo, T. Power spectral analysis of autonomic nervous activity in spontaneously hypertensive rats. Biomed. Sci. Instrum. 1997, 33, 338–343. [Google Scholar] [PubMed]
- Wiszniewski, A.; Suchanowski, A.; Wielgomas, B. Effects of Air-Ions on human circulatory indicators. Pol. J. Environ. Stud. 2014, 23, 521–531. [Google Scholar]
- Watanabe, I.; Noro, H.; Ohtsuka, Y.; Mano, Y.; Agishi, Y. Physical effects of negative air ions in a wet sauna. Int. J. Biometeorol. 1997, 40, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Bachman, C.H.; McDonald, R.D.; Lorenz, P.J. Some physiological effects of measured air ions. Int. J. Biometeorol. 1965, 9, 127–139. [Google Scholar] [CrossRef]
- McDonald, R.D.; Bachman, C.H.; Lorenz, P.J. Some physiological effects of air ion treatment without ion inhalation. Int. J Biometeorol. 1965, 9, 141–147. [Google Scholar] [CrossRef]
- Hawkins, L.H.; Barker, T. Air ions and human performance. Ergonomics 1978, 21, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Bowers, B.; Flory, R.; Ametepe, J.; Staley, L.; Patrick, A.; Carrington, H. Controlled trial evaluation of exposure duration to negative air ions for the treatment of seasonal affective disorder. Psychiatry Res. 2018, 259, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Goel, N.; Terman, M.; Terman, J.S.; Macchi, M.M.; Stewart, J.W. Controlled trial of bright light and negative air ions for chronic depression. Psychol. Med. 2005, 35, 945–955. [Google Scholar] [CrossRef] [PubMed]
- Hedge, A.; Collis, M.D. Do negative air ions affect human mood and performance? Ann. Occup. Hyg. 1987, 31, 285–290. [Google Scholar] [PubMed]
- Reilly, T.; Stevenson, I.C. An investigation of the effects of negative air ions on responses to submaximal exercise at different times of day. J. Hum. Ergol. 1993, 22, 1–9. [Google Scholar]
- Alexander, D.D.; Bailey, W.H.; Perez, V.; Mitchell, M.E.; Su, S. Air ions and respiratory function outcomes: A comprehensive review. J. Negat. Results Biomed. 2013, 12, 14. [Google Scholar] [CrossRef] [PubMed]
- Ogungbe, A.S.; Akintoye, O.H.; Idowu, B.A. Effects of gaseous ions on the environment and human performance. Trends Appl. Sci. Res. 2011, 6, 130–133. [Google Scholar] [CrossRef]
- Perez, V.; Alexander, D.D.; Bailey, W.H. Air ions and mood outcomes: A review and meta-analysis. BMC Psychiatry 2013, 13, 29. [Google Scholar] [CrossRef] [PubMed]
- Voeikov, V.L. Reactive oxygen species--(ROS) pathogens or sources of vital energy? Part 1. ROS in normal and pathologic physiology of living systems. J. Altern. Complement. Med. 2006, 12, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Phillips, G.; Harris, G.J.; Jones, M.W. Effects of air ions on bacterial aerosols. Int. J. Biometeorol. 1964, 8, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, A.K.; Nirala, B.K.; Malik, A.; Singh, K. The effect of negative air ion exposure on Escherichia coli and Pseudomonas fluorescens. J. Environ. Sci. Health A Tox. Hazard Subst. Environ. Eng. 2008, 43, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Shargawi, J.M.; Theaker, E.D.; Drucker, D.B.; MacFarlane, T.; Duxbury, A.J. Sensitivity of Candida albicans to negative air ion streams. J. Appl. Microbiol. 1999, 87, 889–897. [Google Scholar] [CrossRef] [PubMed]
- Dobrynin, D.; Friedman, G.; Fridman, A.; Starikovskiy, A. Inactivation of bacteria using DC corona discharge: Role of ions and humidity. New J. Phys. 2011, 103033. [Google Scholar] [CrossRef] [PubMed]
- Noyce, J.O.; Hughes, J.F. Bactericidal effects of negative and positive ions generated in nitrogen on Escherichia coli. J. Electrostatics 2002, 54, 179–187. [Google Scholar] [CrossRef]
- Timoshkin, I.V.; Maclean, M.; Wilson, M.P.; Given, M.J.; MacGregor, S.J.; Wang, T.; Anderson, J.G. Bactericidal effect of corona discharges in atmospheric air. IEEE Trans. Plasma Sci. 2012, 40, 2322–2333. [Google Scholar] [CrossRef]
- Noyce, J.O.; Hughes, J.F. Bactericidal effects of negative and positive ions generated in nitrogen on starved Pseudomonas veronii. J. Electrostatics 2003, 57, 49–58. [Google Scholar] [CrossRef]
- Escombe, A.R.; Moore, D.A.; Gilman, R.H.; Navincopa, M.; Ticona, E.; Mitchell, B.; Noakes, C.; Martínez, C.; Sheen, P.; Ramirez, R.; et al. Upper-room ultraviolet light and negative air ionization to prevent tuberculosis transmission. PLoS Med. 2009, 6, e43. [Google Scholar] [CrossRef] [PubMed]
- Pratt, R.; Barnard, R.W. Some effects of ionized air on Penicillium notatum. J. Am. Pharm. Assoc. Sci. 1960, 49, 643–646. [Google Scholar] [CrossRef]
- Mitchell, B.W.; King, D.J. Effect of negative air ionization on airborne transmission of newcastle disease virus. Avian. Dis. 1994, 38, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Fett, W.F.; Mitchell, B.W. Effect of negative air ions on Escherichia coli ATCC 25922 inoculated onto mung bean seed and apple fruit. J. Food Prot. 2007, 70, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, L.A.; Gaunt, L.F.; Beggs, C.B.; Shepherd, S.J.; Sleigh, P.A.; Noakes, C.J.; Kerr, K.G. Bactericidal action of positive and negative ions in air. BMC Microbiol. 2007, 7, 32. [Google Scholar] [CrossRef] [PubMed]
- Krueger, A.P.; Kotaka, S.; Andriese, P.C. Some observations on the physiological effects of gaseous ions. Int. J. Biometeorol. 1962, 6, 33–48. [Google Scholar] [CrossRef]
- Krueger, A.P.; Kotaka, S.; Andriese, P.C. Studies on the Effects of Gaseous Ions on Plant Growth I. The influence of positive and negative air ions on the growth of Avena sativa. J. Gen Physiol. 1962, 45, 879–895. [Google Scholar] [CrossRef] [PubMed]
- Kotaka, S.; Krueger, A.P.; Andriese, P.C.; Nishizawa, K.; Ohuchi, T.; Takenobu, M.; Kozure, Y. Air ion effects on the oxygen consumption of barley seedlings. Nature 1965, 208, 1112–1113. [Google Scholar] [CrossRef] [PubMed]
- Elkiey, T.M.; Bhartendu, S.; Barthakur, N. Air ion effect on respiration and photosynthesis of barley and Antirrhinum majus. Int. J. Biometeorol. 1985, 29, 285–292. [Google Scholar] [CrossRef]
- Song, M.J.; Kang, T.H.; Han, C.S.; Oh, M.M. Air anions enhance lettuce growth in plant factories. Hortic. Environ. Biotechnol. 2014, 55, 293–298. [Google Scholar] [CrossRef]
- Song, M.J.; Jeon, Y.M.; Oh, M.M. Growth of and bacterial counts on several edible sprouts exposed to spray ionization. Protected Hort. Plant Fac. 2015, 24, 45–50. [Google Scholar] [CrossRef]
- Lee, S.R.; Kang, T.H.; Han, C.S.; Oh, M.M. Air anions improve growth and mineral content of kale in plant factories. Hortic. Environ. Biotechnol. 2015, 56, 462–471. [Google Scholar] [CrossRef]
- Barthakur, N.N.; Arnold, N.P. Growth and certain chemical constituents of tobacco plants exposed to air ions. Int. J. Biometeorol. 1988, 32, 78–80. [Google Scholar] [CrossRef]
- Sirota, T.V.; Safronova, V.G.; Amelina, A.G.; Mal’tseva, V.N.; Avkhacheeva, N.V.; Sofin, A.D.; Ianin, V.A.; Mubarakshina, E.K.; Romanova, L.K.; Novoselov, VI. Effect of negative air ions on respiratory organs and blood. Biofizika 2008, 53, 886–893. [Google Scholar] [CrossRef] [PubMed]
- Wallner, P.; Kundi, M.; Panny, M.; Tappler, P.; Hutter, H.P. Exposure to air ions in indoor environments: Experimental study with healthy adults. Int. J. Environ. Res. Public Health. 2015, 12, 14301–14311. [Google Scholar] [CrossRef] [PubMed]
- Kellogg, E.W.; Yost, M.G.; Barthakur, N.; Kreuger, A.P. Superoxide involvement in the bactericidal effects of negative air ions on Staphylococcus albus. Nature 1979, 281, 400–401. [Google Scholar] [CrossRef] [PubMed]
- Gabbay, J.; Bergerson, O.; Levi, N.; Brenner, S.; Eli, I. Effect of ionization on microbial air pollution in the dental clinic. Environ. Res. 1990, 52, 99–106. [Google Scholar] [CrossRef]
- Seo, K.H.; Mitchell, B.W.; Holt, P.S.; Gast, R.K. Bactericidal effects of negative air ions on airborne and surface Salmonella enteritidis from an artificially generated aerosol. J. Food Prot. 2001, 64, 113–116. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Song, J.; Hildebrand, P.D.; Forney, C.F. Interaction of ozone and negative air ions to control micro-organisms. J. Appl. Microbiol. 2002, 93, 144–148. [Google Scholar] [CrossRef] [PubMed]
- Kerr, K.G.; Beggs, C.B.; Dean, S.G.; Thornton, J.; Donnelly, J.K.; Todd, N.J.; Sleigh, P.A.; Qureshi, A.; Taylor, C.C. Air ionisation and colonization / infection with methicillin-resistant Staphylococcus aureus and Acinetobacter species in an intensive care unit. Intensive Care Med. 2006, 32, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Kampmann, Y.; Klingshirn, A.; Kloft, K.; Kreyenschmidt, J. The application of ionizers in domestic refrigerators for reduction in airborne and surface bacteria. J. Appl. Microbiol. 2009, 107, 1789–1798. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, A.K.; Malik, A. Antimicrobial action of essential oil vapours and negative air ions against Pseudomonas fluorescens. Int. J. Food Microbiol. 2010, 143, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Yang, Y.; Huang, G.; Lai, A.C.K. Numerical and experimental study on airborne disinfection by negative ions in air duct flow. Build. Environ. 2018, 127, 204–210. [Google Scholar] [CrossRef]
- Alisa, P. Oxygen Toxicity and Free Radical Injury. In Mark’s Basic Medical Biochemistry: A Clinical Approach, 4th ed.; Marks, A.D., Liebermann, M.A., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; pp. 437–456. [Google Scholar]
- Hayyan, M.; Hashim, M.A.; AlNashef, I.M. Superoxide ion: Generation and chemical implications. Chem Rev. 2016, 116, 3029–3085. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, N.I.; Goldstein, R.N.; Merzlyak, M.N. Negative air ions as a source of superoxide. Int. J. Biometeorol. 1992, 36, 118–122. [Google Scholar] [CrossRef]
- Luts, A.; Salm, J. Chemical composition of small atmospheric ions near the ground. J. Geophys. Res. 1994, 99, 10781–10785. [Google Scholar] [CrossRef]
- Goldstein, N.; Lewin, T.; Kamensky, A.; Dubinin, V.; Baumann, S.; Konstantinova, O. Exogenous gaseous superoxide potentiates the antinociceptive effect of opioid analgesic agents. Inflamm. Res. 1996, 45, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Byczkowski, J.Z.; Gessner, T. Biological role of superoxide ion-radical. Int. J. Biochem. 1988, 20, 569–580. [Google Scholar] [CrossRef]
- Fridovich, I. Superoxide dismutases. An adaptation to a paramagnetic gas. J. Biol. Chem. 1989, 264, 7761–7764. [Google Scholar] [PubMed]
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; Amann, M.; Anderson, H.R.; Andrews, K.G.; Aryee, M.; et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990-2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef]
- Chow, J.C.; Watson, J.G.; Mauderly, J.L.; Costa, D.L.; Wyzga, R.E.; Vedal, S.; Hidy, G.M.; Altshuler, S.L.; Marrack, D.; Heuss, J.M.; et al. Health effects of fine particulate air pollution: Lines that connect. J. Air Waste Manage. Assoc. 2006, 56, 1368–1380. [Google Scholar] [CrossRef]
- Xie, Y.; Zhao, B.; Zhang, L.; Luo, R. Spatiotemporal variations of PM2.5 and PM10 concentrations between 31 Chinese cities and their relationships with SO2, NO2, CO and O3. Particuology 2015, 20, 141–149. [Google Scholar] [CrossRef]
- Xing, Y.F.; Xu, Y.H.; Shi, M.H.; Lian, Y.X. The impact of PM2.5 on the human respiratory system. J. Thorac. Dis. 2016, 8, E69–E74. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Mengersen, K.; Kimlin, M.; Zhou, M.; Tong, S.; Fang, L.; Wang, B.; Hu, W. Lung cancer and particulate pollution: A critical review of spatial and temporal analysis evidence. Environ. Res. 2018, 164, 585–596. [Google Scholar] [CrossRef] [PubMed]
- Erqou, S.; Clougherty, J.E.; Olafiranye, O.; Magnani, J.W.; Aiyer, A.; Tripathy, S.; Kinnee, E.; Kip, K.E.; Reis, S.E. Particulate matter air pollution and racial differences in cardiovascular disease risk. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.U.; Yermakov, M.; Grinshpun, S.A. Removal of fine and ultrafine particles from indoor air environments by the unipolar ion emission. Atmos Environ. 2004, 38, 4815–4823. [Google Scholar] [CrossRef]
- Mayya, Y.S.; Sapra, B.K.; Khan, A.; Sunny, F. Aerosol removal by unipolar ionization in indoor environments. Aerosol Sci. 2004, 35, 923–941. [Google Scholar] [CrossRef]
- Grinshpun, S.A.; Mainelis, G.; Trunov, M.; Adhikari, A.; Reponen, T.; Willeke, K. Evaluation of ionic air purifiers for reducing aerosol exposure in confined indoor spaces. Indoor Air 2005, 15, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Berry, D.; Mainelis, G.; Fennell, D. Effect of an ionic air cleaner on indoor/outdoor particle ratios in a residential environment. Aerosol Sci. Technol. 2007, 41, 315–328. [Google Scholar] [CrossRef]
- Qiu, B.; Li, Q.; Hong, W.; Xing, G. Characterization of the key material for elimination of PM2.5 particles in the atmosphere. J. Spectrosc. 2015. [Google Scholar] [CrossRef]
- Tanaka, A.; Zhang, Y. Dust settling efficiency and electrostatic effect of a negative ionization system. J. Agr. Saf. Health 1996, 2, 39–47. [Google Scholar] [CrossRef]
- Grabarczyk, Z. Electiveness of indoor air cleaning with corona ionizers. J. Electrostat. 2001, 51, 278–283. [Google Scholar] [CrossRef]
- Niu, J.L.; Tung, T.C.W.; Burnett, J. Quantication of dust removal and ozone emission of ionizer air-cleaners by chamber testing. J. Electrostatics 2001, 51, 20–24. [Google Scholar] [CrossRef]
- Daniels, S.L. On the ionization of air for removal of noxious effluvia (Air ionization of indoor environments for control of volatile and particulate contaminants with nonthermal plasmas generated by dielectric-barrier discharge. IEEE T. Plasma Sci. 2002, 30, 1471–1481. [Google Scholar] [CrossRef]
- Sawant, V.S.; Meena, G.S.; Jadhav, D.B. Effect of negative air ions on fog and smoke. Aerosol Air Qual. Res. 2012, 12, 1007–1015. [Google Scholar] [CrossRef]
- Sawant, V.S. Control of respirable particles in indoor air with portable negative air ion generator. IOSR J. Environ. Sci. Toxicol. Food Technol. 2013, 3, 28–31. [Google Scholar] [CrossRef]
- Sawant, V.S. Removal of particulate matter by using negative electric discharge. Int. J. Eng. Innov. Technol. 2013, 2, 48–51. [Google Scholar]
- Černecký, J.; Valentová, K.; Pivarčiová, E.; Božek, P. Ionization impact on the air cleaning efficiency in the interior. Meas. Sci. Rev. 2015, 15, 156–166. [Google Scholar] [CrossRef]
- Pushpawela, B.; Jayaratne, R.; Nguy, A.; Morawska, L. Efficiency of ionizers in removing airborne particles in indoor environments. J. Electrostatics 2017, 90, 79–84. [Google Scholar] [CrossRef]
- Zhao, X.; Li, Y.; Hua, T.; Jiang, P.; Yin, X.; Yu, J.; Ding, B. Low-resistance dual-purpose air filter releasing negative ions and effectively capturing PM2.5. ACS Appl. Mater. Interfaces 2017, 9, 12054–12063. [Google Scholar] [CrossRef] [PubMed]
- Yu, K.P.; Lee, W.M.G.; Peng, C.J.; Chen, Y.C.; Shen, W.T. Effects of roughness, dielectric constant and electrical resistivity of wall on deposition of submicron particles driven by ionic air purifier. J. Environ. Chem. Engineer. 2017, 5, 3108–3114. [Google Scholar] [CrossRef]
- Sutto, H.C. Superoxide and ozone production by corona discharge. J. Chem. Soc. Faraday Trans. 1984, 80, 2301–2311. [Google Scholar] [CrossRef]
- Wu, C.C.; Lee, G.W.M. Oxidation of Volatile Organic Compounds by Negative Air Ions. Atmos. Environ. 2004, 38, 6287–6295. [Google Scholar] [CrossRef]
- Waring, M.S.; Siegel, J.A.; Corsi, R.L. Ultrafine particle removal and generation by portable air cleaners. Atmos. Environ. 2008, 42, 5003–5014. [Google Scholar] [CrossRef]
- Yu, K.P. Enhancement of the deposition of ultrafine secondary organic aerosols by the negative air ion and the effect of relative humidity. J. Air Waste Manage. 2012, 62, 1296–1304. [Google Scholar] [CrossRef]
| Treatment | Distance between Ion Counter and Plant | NAI Concentration (ions/cm3) | Reference |
|---|---|---|---|
| Soil pot without plants | 70 cm | 135 ± 70 # | [28] |
| Soil pot with plants | 70 cm | 133 ± 45 # | [28] |
| Soil pot without plants | ≤32.5 cm * | 35 | [30] |
| Soil pot with plants | |||
| M. adansonii var. laniata | ≤32.5 cm * | 36 | [30] |
| C. asiaticum var. sinicum | ≤32.5 cm * | 68 | [30] |
| O. bodinieri | ≤32.5 cm * | 56 | [30] |
| A. macrorrhizos | ≤32.5 cm * | 66 | [30] |
| A. densiflorus | ≤32.5 cm * | 55 | [30] |
| D. ensifolia | ≤32.5 cm * | 40 | [30] |
| N. tazetta var. chinensis | ≤32.5 cm * | 73 | [30] |
| H. fulva | ≤32.5 cm * | 55 | [30] |
| Z. carinata | ≤32.5 cm * | 72 | [30] |
| D. cochinchinensis | ≤32.5 cm * | 43 | [30] |
| Plant Species/Variety/Treatment | Distance between Ion Counter and Plant | Output Voltage | NAI Concentration (ions/cm3) | Reference |
|---|---|---|---|---|
| Soil pot without plants | ≤32.5 cm * | 25 kv * | 131 ± 4 | [30] |
| Soil pot without plants | 70 cm | 20 kv | 4000 ± 400 | [28] |
| Aloe arborescens | 70 cm | 25 kv | 280,000 ± 41,000 | [28] |
| Haworthia rasalata | 70 cm | 25 kv | 41,000 ± 8000 | [28] |
| Chlorophytum comosum | 70 cm | 25 kv | 95,000 ± 12,000 | [28] |
| Opuntia brunnescens | 70 cm | 25 kv | 53,000 ± 13,000 | [28] |
| Zephyranthes carinata | ≤32.5 cm * | 15 kv | 1,791,067 ± 27,243 | [30] |
| Zephyranthes carinata | ≤32.5 cm * | 20 kv | 3,593,489 ± 358,104 | [30] |
| Crinum asiaticum | ≤32.5 cm * | 15 kv | 201,000 ± 23,798 | [30] |
| Crinum asiaticum | ≤32.5 cm * | 20 kv | 59,475 ± 839 | [30] |
| Narcissus tazetta | ≤32.5 cm * | 15 kv | 162 ± 28 | [30] |
| Narcissus tazetta | ≤32.5 cm * | 20 kv | 315 ± 33 | [30] |
| Ophiopogon japonicus | Near the grass | 37.6 kv | 2,000,000 ± 300,000 | [29] |
| L. Muttiflorum | Near the grass | 37.3 kv | 500,000 ± 60,000 | [29] |
| Zoysia spp | Near the grass | 38 kv | 200,000 ± 30,000 | [29] |
| Poa pratensis | Near the grass | 38.6 kv | 200,000 ± 30,000 | [29] |
| Zephyranthes candida | Near the grass | 42.8 kv | 100,000 ± 23,000 | [29] |
| Apocynum venetum | Near the grass | 30.7 kv | 80,000 ± 9000 | [29] |
| Item | Description | Reference |
|---|---|---|
| PM removal efficiency | NAIs reduced respirable and inhalable dust counts by 46%. | [143] |
| PM concentration was reduced by up to two orders of magnitude after 2 h of treatment by NAI generator d in a 50 m3 unoccupied office room. | [144] | |
| From around 800 µg/m3 to 50 µg/m3 in a chamber (2 m × 2 m × 1.6 m) | [145] | |
| Ionizer is efficient in reducing fine PM | [146] | |
| More than 80% 0.1 or 1 mm particles were removed within 1 h | [138] | |
| The removal efficiency under high concentration of NAIs reached about 50% after 15 min and almost 100% after 1.5 h. | [140] | |
| In a glass chamber (60 cm × 30 cm × 40 cm), 93% to 97% of the particles from fog or smoke was removed by NAIs within 6 min. | [147] | |
| Around 95% of respirable particles from indoor air can be removed | [148] | |
| The particle removal efficiency increases with enhanced ion emission rate and the duration of emission. | [149] | |
| NAIs can be used to eliminate cigarette smoke | [150] | |
| Air ionizers are efficient even in a ventilated room with 132 m3 and can be used for removing ultrafine particles. | [151] | |
| Field tests conducted in Shanghai showed stable PM2.5 purification efficiency of 99.99% at high releasing amounts of negative ions (RANIs), in the event of haze. | [152] | |
| Factors affecting PM reduction | PM removal rate is related to particle concentrations, particle sizes, and the ventilation rate and a model is raised to compute the dependence. | [139] |
| The NAI could remove particles from the wood and polyvinyl chloride (PVC) wall surfaces substantially more effectively than from other wall materials such as wallpaper, stainless steel, and cement paint. | [34] | |
| PM reducing efficiency was related to the heights and distances from the source of NAIs. The highest efficiency of PM10 removal was achieved when the distance between smoking cigarettes and ionizer was 3 m and the air humidity was 39%. | [150] | |
| PM diameter significantly affects the deposition of particles and wall roughness is a key beneficial factor to particle deposition driven by ionizer | [153] | |
| Drawback | Ozone was produced by corona discharge of air | [154] |
| A total of 5 out of the 27 ionizers were found to emit ozone | [145] | |
| The high voltage used for ion generation produces Ozone above the threshold voltage of 16,000 volts in the tested system. | [155] | |
| The ozone levels were increased by at least 3 times after using a leading commercially available ionic air cleaner | [141] | |
| Portable air cleaners can both remove and generate pollutants indoors due to ozone release | [156] | |
| Many NAI generators emitted ozone | [157] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, S.-Y.; Ma, A.; Ramachandran, S. Negative Air Ions and Their Effects on Human Health and Air Quality Improvement. Int. J. Mol. Sci. 2018, 19, 2966. https://doi.org/10.3390/ijms19102966
Jiang S-Y, Ma A, Ramachandran S. Negative Air Ions and Their Effects on Human Health and Air Quality Improvement. International Journal of Molecular Sciences. 2018; 19(10):2966. https://doi.org/10.3390/ijms19102966
Chicago/Turabian StyleJiang, Shu-Ye, Ali Ma, and Srinivasan Ramachandran. 2018. "Negative Air Ions and Their Effects on Human Health and Air Quality Improvement" International Journal of Molecular Sciences 19, no. 10: 2966. https://doi.org/10.3390/ijms19102966
APA StyleJiang, S.-Y., Ma, A., & Ramachandran, S. (2018). Negative Air Ions and Their Effects on Human Health and Air Quality Improvement. International Journal of Molecular Sciences, 19(10), 2966. https://doi.org/10.3390/ijms19102966





