Eradicating Suicide at Its Roots: Preclinical Bases and Clinical Evidence of the Efficacy of Ketamine in the Treatment of Suicidal Behaviors
Abstract
1. The Roots of Suicide: Current Advances in Neurobiology of Depression and Suicidal Behaviors
1.1. A Global Phenomenon
1.2. The Process of Suicidal Behavior
1.3. Integrated Theories of Suicide
2. Cognitive Bases of Suicidal Behaviors: The Role of Glutamate Neurotransmission
2.1. Cognitive Dysfunctions in Suicide Attempters
2.2. Glutamate Neurotransmission Dysregulation as the Foundation of Suicidal Cognitive Biases
3. Eradicating the Roots of Suicide: Neurobiological Bases of Anti-Suicidal Effects of Ketamine
3.1. Preclinical Evidence of Ketamine Antidepressant Effects
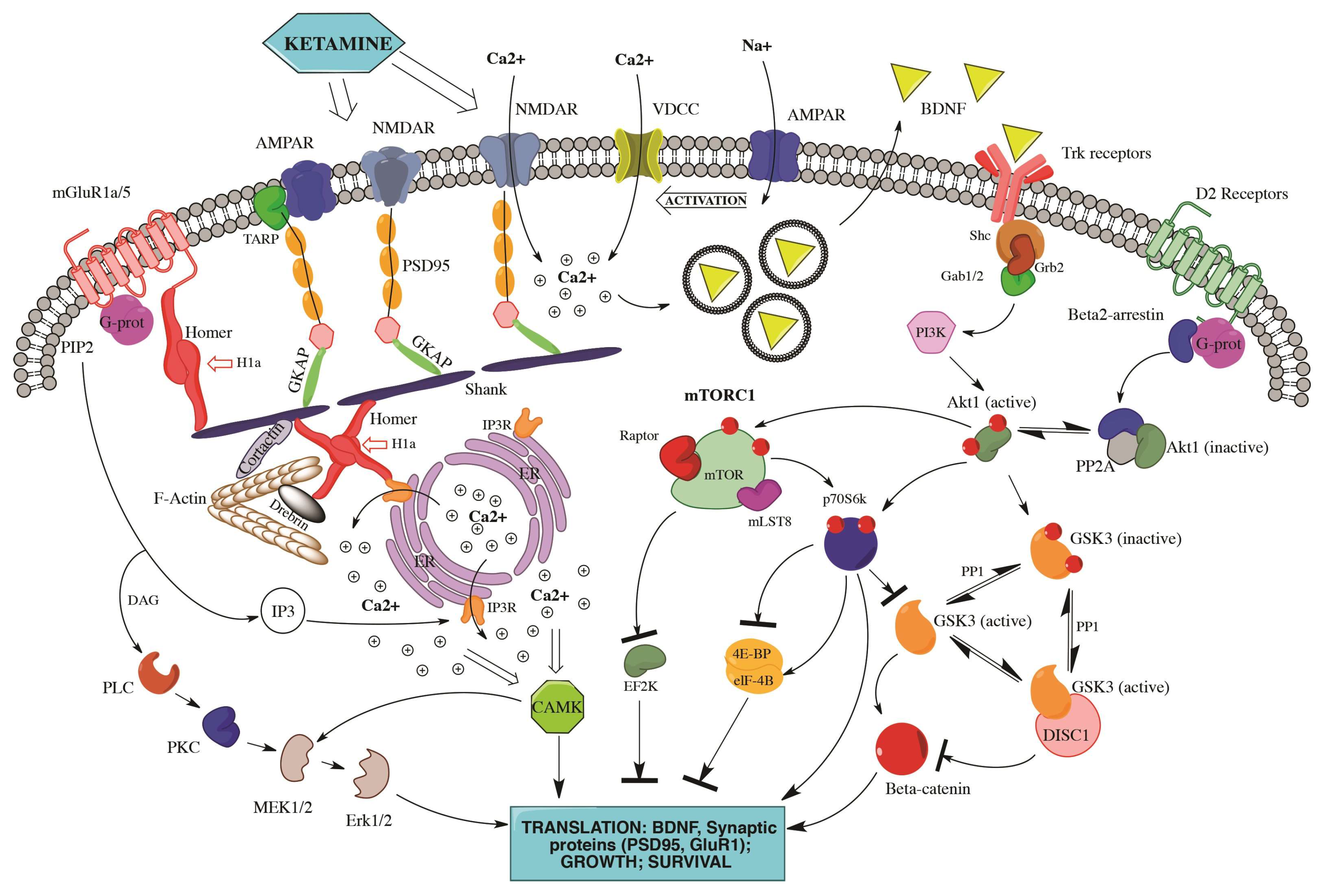
3.2. mTOR and BDNF: The Molecular Cores of Ketamine Antidepressant and Anti-Suicidal Action
3.3. Comparing Anti-Suicidal Treatment Neurobiology: Ketamine vs. Classical Antidepressants, Lithium and Clozapine
4. Eradicating the Roots of Suicide: Clinical Randomized Studies of Anti-Suicidal Effects of Ketamine
5. Eradicating the Roots of Suicide with Ketamine Treatment: Conclusions
Author Contributions
Conflicts of Interest
References
- World Health Organization (WHO). Suicide; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- World Health Organization (WHO). Suicide Data; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Arsenault-Lapierre, G.; Kim, C.; Turecki, G. Psychiatric diagnoses in 3275 suicides: A meta-analysis. BMC Psychiatry 2004, 4, 37. [Google Scholar] [CrossRef] [PubMed]
- Hegerl, U. Prevention of suicidal behavior. Dialogues Clin. Neurosci. 2016, 18, 183–190. [Google Scholar] [PubMed]
- Pompili, M.; Gonda, X.; Serafini, G.; Innamorati, M.; Sher, L.; Amore, M.; Rihmer, Z.; Girardi, P. Epidemiology of suicide in bipolar disorders: A systematic review of the literature. Bipolar Disord. 2013, 15, 457–490. [Google Scholar] [CrossRef] [PubMed]
- Wasserman, D.; Wasserman, C. Oxford Textbook of Suicidology and Suicide Prevention: A Global Perspective; OUP Oxford: Oxford, UK, 2009; 872p. [Google Scholar]
- Bertolote, J.M.; Fleischmann, A.; De Leo, D.; Wasserman, D. Psychiatric Diagnoses and Suicide: Revisiting the Evidence. Crisis 2004, 25, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Fountoulakis, K.N.; Kawohl, W.; Theodorakis, P.N.; Kerkhof, A.J.F.M.; Navickas, A.; Höschl, C.; Lecic-Tosevski, D.; Sorel, E.; Rancans, E.; Palova, E.; et al. Relationship of suicide rates to economic variables in Europe: 2000–2011. Br. J. Psychiatry 2014, 205, 486–496. [Google Scholar] [CrossRef] [PubMed]
- De Berardis, D.; Martinotti, G.; Di Giannantonio, M. Editorial: Understanding the Complex Phenomenon of Suicide: From Research to Clinical Practice. Front. Psychiatry 2018, 9, 61. [Google Scholar] [CrossRef] [PubMed]
- De Berardis, D.; Fornaro, M.; Orsolini, L.; Valchera, A.; Carano, A.; Vellante, F.; Perna, G.; Serafini, G.; Gonda, X.; Pompili, M.; et al. Alexithymia and Suicide Risk in Psychiatric Disorders: A Mini-Review. Front. Psychiatry 2017, 8, 148. [Google Scholar] [CrossRef] [PubMed]
- Pompili, M.; Innamorati, M.; Vichi, M.; Masocco, M.; Vanacore, N.; Lester, D.; Serafini, G.; Dominici, G.; Girardi, P.; De Leo, D.; et al. Suicide prevention among youths. Systematic review of available evidence-based interventions and implications for Italy. Minerva Pediatr. 2010, 62, 507–535. [Google Scholar] [PubMed]
- Serafini, G.; Muzio, C.; Piccinini, G.; Flouri, E.; Ferrigno, G.; Pompili, M.; Girardi, P.; Amore, M. Life adversities and suicidal behavior in young individuals: A systematic review. Eur. Child Adolesc. Psychiatry 2015, 24, 1423–1446. [Google Scholar] [CrossRef] [PubMed]
- Orsolini, L.; Valchera, A.; Vecchiotti, R.; Tomasetti, C.; Iasevoli, F.; Fornaro, M.; De Berardis, D.; Perna, G.; Pompili, M.; Bellantuono, C. Suicide during Perinatal Period: Epidemiology, Risk Factors, and Clinical Correlates. Front. Psychiatry 2016, 7, 138. [Google Scholar] [CrossRef] [PubMed]
- Large, M.; Kaneson, M.; Myles, N.; Myles, H.; Gunaratne, P.; Ryan, C. Meta-Analysis of Longitudinal Cohort Studies of Suicide Risk Assessment among Psychiatric Patients: Heterogeneity in Results and Lack of Improvement over Time. PLoS ONE 2016, 11, e0156322. [Google Scholar] [CrossRef] [PubMed]
- Schrijvers, D.L.; Bollen, J.; Sabbe, B.G.C. The gender paradox in suicidal behavior and its impact on the suicidal process. J. Affect. Disord. 2012, 138, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Zalsman, G.; Hawton, K.; Wasserman, D.; van Heeringen, K.; Arensman, E.; Sarchiapone, M.; Carli, V.; Hoschl, C.; Barzilay, R.; Balazs, J.; et al. Suicide prevention strategies revisited: 10-year systematic review. Lancet Psychiatry 2016, 3, 646–659. [Google Scholar] [CrossRef]
- Madsen, T.; Karstoft, K.-I.; Secher, R.G.; Austin, S.F.; Nordentoft, M. Trajectories of suicidal ideation in patients with first-episode psychosis: Secondary analysis of data from the OPUS trial. Lancet Psychiatry 2016, 3, 443–450. [Google Scholar] [CrossRef]
- Kasckow, J.; Youk, A.; Anderson, S.J.; Dew, M.A.; Butters, M.A.; Marron, M.M.; Begley, A.E.; Szanto, K.; Dombrovski, A.Y.; Mulsant, B.H.; et al. Trajectories of suicidal ideation in depressed older adults undergoing antidepressant treatment. J. Psychiatr. Res. 2016, 73, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Köhler-Forsberg, O.; Madsen, T.; Behrendt-Møller, I.; Sylvia, L.; Bowden, C.L.; Gao, K.; Bobo, W.V.; Trivedi, M.H.; Calabrese, J.R.; Thase, M.; et al. Trajectories of suicidal ideation over 6 months among 482 outpatients with bipolar disorder. J. Affect. Disord. 2017, 223, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Czyz, E.K.; King, C.A. Longitudinal Trajectories of Suicidal Ideation and Subsequent Suicide Attempts Among Adolescent Inpatients. J. Clin. Child Adolesc. Psychol. 2015, 44, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Bostwick, J.M.; Pabbati, C.; Geske, J.R.; McKean, A.J. Suicide Attempt as a Risk Factor for Completed Suicide: Even More Lethal Than We Knew. Am. J. Psychiatry 2016, 173, 1094–1100. [Google Scholar] [CrossRef] [PubMed]
- Turecki, G.; Ota, V.K.; Belangero, S.I.; Jackowski, A.; Kaufman, J. Early life adversity, genomic plasticity, and psychopathology. Lancet Psychiatry 2014, 1, 461–466. [Google Scholar] [CrossRef]
- Oquendo, M.A.; Sullivan, G.M.; Sudol, K.; Baca-Garcia, E.; Stanley, B.H.; Sublette, M.E.; Mann, J.J. Toward a Biosignature for Suicide. Am. J. Psychiatry 2014, 171, 1259–1277. [Google Scholar] [CrossRef] [PubMed]
- De Berardis, D.; Conti, C.M.; Serroni, N.; Moschetta, F.S.; Carano, A.; Salerno, R.M.; Cavuto, M.; Farina, B.; Alessandrini, M.; Janiri, L.; et al. The role of cholesterol levels in mood disorders and suicide. J. Biol. Regul. Homeost Agents 2009, 23, 133–140. [Google Scholar] [PubMed]
- Hariri, A.R.; Mattay, V.S.; Tessitore, A.; Kolachana, B.; Fera, F.; Goldman, D.; Egan, M.F.; Weinberger, D.R. Serotonin transporter genetic variation and the response of the human amygdala. Science 2002, 297, 400–403. [Google Scholar] [CrossRef] [PubMed]
- Plomin, R.; Owen, M.J.; McGuffin, P.; Sabol, S.Z.; Greenberg, B.D.; Petri, S.; Benjamin, J.; Müller, C.R.; Hamer, D.H.; Murphy, D.L. The genetic basis of complex human behaviors. Science 1994, 264, 1733–1739. [Google Scholar] [CrossRef] [PubMed]
- Bennett, A.; Lesch, K.; Heils, A.; Long, J.C.; Lorenz, J.G.; Shoaf, S.E.; Champoux, M.; Suomi, S.J.; Linnoila, M.V.; Higley, J.D. Early experience and serotonin transporter gene variation interact to influence primate CNS function. Mol. Psychiatry 2007, 7, 118–122. [Google Scholar] [CrossRef]
- Caspi, A.; Sugden, K.; Moffitt, T.E.; Taylor, A.; Craig, I.W.; Harrington, H.; McClay, J.; Mill, J.; Martin, J.; Braithwaite, A.; et al. Influence of Life Stress on Depression: Moderation by a Polymorphism in the 5-HTT Gene. Science 2003, 301, 386–389. [Google Scholar] [CrossRef] [PubMed]
- Sokolowski, M.; Ben-Efraim, Y.J.; Wasserman, J.; Wasserman, D. Glutamatergic GRIN2B and polyaminergic ODC1 genes in suicide attempts: Associations and gene–environment interactions with childhood/adolescent physical assault. Mol. Psychiatry 2013, 18, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Brodsky, B.S. Early Childhood Environment and Genetic Interactions: The Diathesis for Suicidal Behavior. Curr. Psychiatry Rep. 2016, 18, 86. [Google Scholar] [CrossRef] [PubMed]
- Jollant, F.; Lawrence, N.L.; Olié, E.; Guillaume, S.; Courtet, P. The suicidal mind and brain: A review of neuropsychological and neuroimaging studies. World J. Boil. Psychiatry 2011, 12, 319–339. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.N.; Tao, R.; Li, C.; Gao, Y.; Gondré-Lewis, M.C.; Lipska, B.K.; Shin, J.H.; Xie, B.; Ye, T.; Weinberger, D.R.; et al. GAD2 Alternative Transcripts in the Human Prefrontal Cortex, and in Schizophrenia and Affective Disorders. PLoS ONE 2016, 11, e0148558. [Google Scholar] [CrossRef] [PubMed]
- Joiner, T.E. Why People Die By Suicide 1 Why People Die By Suicide: Further Development and Tests of the Interpersonal-Psychological Theory of Suicidal Behavior. Available online: http://portal.idc.ac.il/en/symposium/hspsp/2011/documents/cjoiner11.pdf (accessed on 2 September 2018).
- Chopin, E.; Kerkhof, A.J.F.M.; Arensman, E. Psychological dimensions of attempted suicide: Theories and data. In Suicidal Behaviour: Theories and Research Findings; Hogrefe and Huber: Göttingen, Germany, 2004; pp. 41–60. [Google Scholar]
- Wenzel, A.; Brown, G.K.; Beck, A.T. Cognitive Therapy for Suicidal Patients: Scientific and Clinical Applications; American Psychological Association: Washington, DC, USA, 2009; 377p. [Google Scholar]
- Richard-Devantoy, S.; Turecki, G.; Jollant, F. Neurobiology of Elderly Suicide. Arch. Suicide Res. 2016, 20, 291–313. [Google Scholar] [CrossRef] [PubMed]
- Keilp, J.G.; Gorlyn, M.; Russell, M.; Oquendo, M.A.; Burke, A.K.; Harkavy-Friedman, J.; Mann, J.J. Neuropsychological function and suicidal behavior: Attention control, memory and executive dysfunction in suicide attempt. Psychol. Med. 2013, 43, 539–551. [Google Scholar] [CrossRef] [PubMed]
- McGirr, A.; Renaud, J.; Bureau, A.; Seguin, M.; Lesage, A.; Turecki, G. Impulsive-aggressive behaviours and completed suicide across the life cycle: A predisposition for younger age of suicide. Psychol. Med. 2008, 38, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Dougherty, D.M.; Bjork, J.M.; Moeller, F.G.; Harper, R.A.; Marsh, D.M.; Mathias, C.W.; Swann, A.C. Familial Transmission of Continuous Performance Test Behavior: Attentional and Impulsive Response Characteristics. J. Gen. Psychol. 2003, 130, 5–21. [Google Scholar] [CrossRef] [PubMed]
- Malloy-Diniz, L.F.; Neves, F.S.; Abrantes, S.S.C.; Fuentes, D.; Corrêa, H. Suicide behavior and neuropsychological assessment of type I bipolar patients. J. Affect. Disord. 2009, 112, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-Y.; Jiang, N.-Z.; Cheung, E.F.C.; Sun, H.-W.; Chan, R.C.K. Role of depression severity and impulsivity in the relationship between hopelessness and suicidal ideation in patients with major depressive disorder. J. Affect. Disord. 2015, 183, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Soloff, P.; White, R.; Diwadkar, V.A. Impulsivity, aggression and brain structure in high and low lethality suicide attempters with borderline personality disorder. Psychiatry Res. Neuroimaging 2014, 222, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Millan, M.J.; Agid, Y.; Brüne, M.; Bullmore, E.T.; Carter, C.S.; Clayton, N.S.; Connor, R.; Davis, S.; Deakin, B.; DeRubeis, R.J.; et al. Cognitive dysfunction in psychiatric disorders: Characteristics, causes and the quest for improved therapy. Nat. Rev. Drug Discov. 2012, 11, 141–168. [Google Scholar] [CrossRef] [PubMed]
- Anand, A.; Li, Y.; Wang, Y.; Wu, J.; Gao, S.; Bukhari, L.; Mathews, V.P.; Kalnin, A.; Lowe, M.J. Activity and Connectivity of Brain Mood Regulating Circuit in Depression: A Functional Magnetic Resonance Study. Boil. Psychiatry 2005, 57, 1079–1088. [Google Scholar] [CrossRef] [PubMed]
- Koenigs, M.; Grafman, J. The functional neuroanatomy of depression: Distinct roles for ventromedial and dorsolateral prefrontal cortex. Behav. Brain Res. 2009, 201, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Hasler, G.; van der Veen, J.W.; Tumonis, T.; Meyers, N.; Shen, J.; Drevets, W.C. Reduced Prefrontal Glutamate/Glutamine and γ-Aminobutyric Acid Levels in Major Depression Determined Using Proton Magnetic Resonance Spectroscopy. Arch. Gen. Psychiatry 2007, 64, 193. [Google Scholar] [CrossRef] [PubMed]
- Feyissa, A.M.; Chandran, A.; Stockmeier, C.A.; Karolewicz, B. Reduced levels of NR2A and NR2B subunits of NMDA receptor and PSD-95 in the prefrontal cortex in major depression. Prog. Neuro-Psychopharmacol. Boil. Psychiatry 2009, 33, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Moghaddam, B. Stress Preferentially Increases Extraneuronal Levels of Excitatory Amino Acids in the Prefrontal Cortex: Comparison to Hippocampus and Basal Ganglia. J. Neurochem. 1993, 60, 1650–1657. [Google Scholar] [CrossRef] [PubMed]
- Yuen, E.Y.; Liu, W.; Karatsoreos, I.N.; Feng, J.; McEwen, B.S.; Yan, Z. Acute stress enhances glutamatergic transmission in prefrontal cortex and facilitates working memory. Proc. Natl. Acad. Sci. USA 2009, 106, 14075–14079. [Google Scholar] [CrossRef] [PubMed]
- Dalton, G.L.; Ma, L.M.; Phillips, A.G.; Floresco, S.B. Blockade of NMDA GluN2B receptors selectively impairs behavioral flexibility but not initial discrimination learning. Psychopharmacology 2011, 216, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Yuen, E.Y.; Wei, J.; Liu, W.; Zhong, P.; Li, X.; Yan, Z. Repeated Stress Causes Cognitive Impairment by Suppressing Glutamate Receptor Expression and Function in Prefrontal Cortex. Neuron 2012, 73, 962–977. [Google Scholar] [CrossRef] [PubMed]
- Jett, J.D.; Bulin, S.E.; Hatherall, L.C.; McCartney, C.M.; Morilak, D.A. Deficits in cognitive flexibility induced by chronic unpredictable stress are associated with impaired glutamate neurotransmission in the rat medial prefrontal cortex. Neuroscience 2017, 346, 284–297. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, H.-G.; Tausch, A.; Wagner, R.; Steiner, J.; Seeleke, P.; Walter, M.; Dobrowolny, H.; Bogerts, B. Disruption of glutamate-glutamine-GABA cycle significantly impacts on suicidal behaviour: Survey of the literature and own findings on glutamine synthetase. CNS Neurol. Disord. Drug Targets 2013, 12, 900–913. [Google Scholar] [CrossRef] [PubMed]
- Gos, T.; Günther, K.; Bielau, H.; Dobrowolny, H.; Mawrin, C.; Trübner, K.; Brisch, R.; Steiner, J.; Bernstein, H.-G.; Jankowski, Z.; et al. Suicide and depression in the quantitative analysis of glutamic acid decarboxylase-Immunoreactive neuropil. J. Affect. Disord. 2009, 113, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Naaijen, J.; Lythgoe, D.J.; Zwiers, M.P.; Hartman, C.A.; Hoekstra, P.J.; Buitelaar, J.K.; Aarts, E. Anterior cingulate cortex glutamate and its association with striatal functioning during cognitive control. Eur. Neuropsychopharmacol. 2018, 28, 381–391. [Google Scholar] [CrossRef] [PubMed]
- Khokhar, J.Y.; Henricks, A.M.; Sullivan, E.D.K.; Green, A.I. Unique Effects of Clozapine: A Pharmacological Perspective. Adv. Pharmacol. 2018, 82, 137–162. [Google Scholar] [PubMed]
- Krakowski, M.I.; Czobor, P. Depression and Impulsivity as Pathways to Violence: Implications for Antiaggressive Treatment. Schizophr. Bull. 2014, 40, 886–894. [Google Scholar] [CrossRef] [PubMed]
- Serafini, G.; Adavastro, G.; Canepa, G.; Capobianco, L.; Conigliaro, C.; Pittaluga, F.; Murri, M.B.; Valchera, A.; De Berardis, D.; Pompili, M.; et al. Abnormalities in Kynurenine Pathway Metabolism in Treatment-Resistant Depression and Suicidality: A Systematic Review. CNS Neurol. Disord. Drug Targets 2017, 16, 440–453. [Google Scholar] [CrossRef] [PubMed]
- Javitt, D.C.; Duncan, L.; Balla, A.; Sershen, H. Inhibition of System A-mediated glycine transport in cortical synaptosomes by therapeutic concentrations of clozapine: Implications for mechanisms of action. Mol. Psychiatry 2005, 10, 275–287. [Google Scholar] [CrossRef] [PubMed]
- Preti, A. Animal model and neurobiology of suicide. Prog. Neuropsychopharmacol. Biol. Psychiatry 2011, 35, 818–830. [Google Scholar] [CrossRef] [PubMed]
- Gould, T.D.; Georgiou, P.; Brenner, L.A.; Brundin, L.; Can, A.; Courtet, P.; Donaldson, Z.R.; Dwivedi, Y.; Guillaume, S.; Gottesman, I.I.; et al. Animal models to improve our understanding and treatment of suicidal behavior. Transl. Psychiatry 2017, 7, e1092. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.J.; Azzaro, A.J.; Zaldivar, S.B.; Palmer, S.; Lee, H.S. Properties of the optical isomers and metabolites of ketamine on the high affinity transport and catabolism of monoamines. Neuropharmacology 1981, 20, 391–396. [Google Scholar] [CrossRef]
- Smith, D.J.; Bouchal, R.L.; deSanctis, C.A.; Monroe, P.J.; Amedro, J.B.; Perrotti, J.M.; Crisp, T. Properties of the interaction between ketamine and opiate binding sites in vivo and in vitro. Neuropharmacology 1987, 26, 1253–1260. [Google Scholar] [CrossRef]
- Vollenweider, F.X.; Leenders, K.L.; Oye, I.; Hell, D.; Angst, J. Differential psychopathology and patterns of cerebral glucose utilisation produced by (S)- and (R)-ketamine in healthy volunteers using positron emission tomography (PET). Eur. Neuropsychopharmacol. J. Eur. Coll. Neuropsychopharmacol. 1997, 7, 25–38. [Google Scholar] [CrossRef]
- Trullas, R.; Skolnick, P. Functional antagonists at the NMDA receptor complex exhibit antidepressant actions. Eur. J. Pharmacol. 1990, 185, 1–10. [Google Scholar] [CrossRef]
- Yilmaz, A.; Schulz, D.; Aksoy, A.; Canbeyli, R. Prolonged effect of an anesthetic dose of ketamine on behavioral despair. Pharmacol. Biochem. Behav. 2002, 71, 341–344. [Google Scholar] [CrossRef]
- Garcia, L.S.B.; Comim, C.M.; Valvassori, S.S.; Réus, G.Z.; Barbosa, L.M.; Andreazza, A.C.; Stertz, L.; Fries, G.R.; Gavioli, E.C.; Kapczinski, F.; et al. Acute administration of ketamine induces antidepressant-like effects in the forced swimming test and increases BDNF levels in the rat hippocampus. Prog. Neuro-Psychopharmacol. Boil. Psychiatry 2008, 32, 140–144. [Google Scholar] [CrossRef] [PubMed]
- Maeng, S.; Zarate, C.A.; Du, J.; Schloesser, R.J.; McCammon, J.; Chen, G.; Manji, H.K. Cellular Mechanisms Underlying the Antidepressant Effects of Ketamine: Role of α-Amino-3-Hydroxy-5-Methylisoxazole-4-Propionic Acid Receptors. Boil. Psychiatry 2008, 63, 349–352. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Yu, H.-Y.; Shen, X.-F.; Gao, Z.-Q.; Yang, C.; Yang, J.-J.; Zhang, G.-F. The rapid antidepressant effect of ketamine in rats is associated with down-regulation of pro-inflammatory cytokines in the hippocampus. Upsala J. Med. Sci. 2015, 120, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Réus, G.Z.; Carlessi, A.S.; Titus, S.E.; Abelaira, H.M.; Ignácio, Z.M.; da Luz, J.R.; Matias, B.I.; Bruchchen, L.; Florentino, D.; Vieira, A.; et al. A single dose of S-ketamine induces long-term antidepressant effects and decreases oxidative stress in adulthood rats following maternal deprivation. Dev. Neurobiol. 2015, 75, 1268–1281. [Google Scholar] [CrossRef] [PubMed]
- Réus, G.Z.; Nacif, M.P.; Abelaira, H.M.; Tomaz, D.B.; dos Santos, M.A.B.; Carlessi, A.S.; da Luz, J.R.; Gonçalves, R.C.; Vuolo, F.; Dal-Pizzol, F.; et al. Ketamine ameliorates depressive-like behaviors and immune alterations in adult rats following maternal deprivation. Neurosci. Lett. 2015, 584, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Koike, H.; Iijima, M.; Chaki, S. Effects of ketamine and LY341495 on the depressive-like behavior of repeated corticosterone-injected rats. Pharmacol. Biochem. Behav. 2013, 107, 20–23. [Google Scholar] [CrossRef] [PubMed]
- Fuchikami, M.; Thomas, A.; Liu, R.; Wohleb, E.S.; Land, B.B.; DiLeone, R.J.; Aghajanian, G.K.; Duman, R.S. Optogenetic stimulation of infralimbic PFC reproduces ketamine’s rapid and sustained antidepressant actions. Proc. Natl. Acad. Sci. USA 2015, 112, 8106–8111. [Google Scholar] [CrossRef] [PubMed]
- Grimm, O.; Gass, N.; Weber-Fahr, W.; Sartorius, A.; Schenker, E.; Spedding, M.; Risterucci, C.; Schweiger, J.I.; Böhringer, A.; Zang, Z.; et al. Acute ketamine challenge increases resting state prefrontal-hippocampal connectivity in both humans and rats. Psychopharmacology 2015, 232, 4231–4241. [Google Scholar] [CrossRef] [PubMed]
- Gass, N.; Schwarz, A.J.; Sartorius, A.; Schenker, E.; Risterucci, C.; Spedding, M.; Zheng, L.; Meyer-Lindenberg, A.; Weber-Fahr, W. Sub-Anesthetic Ketamine Modulates Intrinsic BOLD Connectivity Within the Hippocampal-Prefrontal Circuit in the Rat. Neuropsychopharmacology 2014, 39, 895–906. [Google Scholar] [CrossRef] [PubMed]
- Hay, N.; Sonenberg, N. Upstream and downstream of mTOR. Genes Dev. 2004, 18, 1926–1945. [Google Scholar] [CrossRef] [PubMed]
- Fingar, D.C.; Blenis, J. Target of rapamycin (TOR): An integrator of nutrient and growth factor signals and coordinator of cell growth and cell cycle progression. Oncogene 2004, 23, 3151–3171. [Google Scholar] [CrossRef] [PubMed]
- Inoki, K.; Ouyang, H.; Zhu, T.; Lindvall, C.; Wang, Y.; Zhang, X.; Yang, Q.; Bennett, C.; Harada, Y.; Stankunas, K.; et al. TSC2 Integrates Wnt and Energy Signals via a Coordinated Phosphorylation by AMPK and GSK3 to Regulate Cell Growth. Cell 2006, 126, 955–968. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.-H.; Sarbassov, D.D.; Ali, S.M.; King, J.E.; Latek, R.R.; Erdjument-Bromage, H.; Tempst, P.; Sabatini, D.M. mTOR Interacts with Raptor to Form a Nutrient-Sensitive Complex that Signals to the Cell Growth Machinery. Cell 2002, 110, 163–175. [Google Scholar] [CrossRef]
- Sarbassov, D.D.; Ali, S.M.; Sabatini, D.M. Growing roles for the mTOR pathway. Curr. Opin. Cell Boil. 2005, 17, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Wang, C.; Xue, Z.; Li, C.; Zhang, J.; Zhao, X.; Liu, A.; Wang, Q.; Zhou, W. PI3K/AKT/mTOR Signaling-Mediated Neuropeptide VGF in the Hippocampus of Mice Is Involved in the Rapid Onset Antidepressant-Like Effects of GLYX-13. Int. J. Neuropsychopharmacol. 2015, 18. [Google Scholar] [CrossRef] [PubMed]
- Bockaert, J.; Marin, P. mTOR in Brain Physiology and Pathologies. Physiol. Rev. 2015, 95, 1157–1187. [Google Scholar] [CrossRef] [PubMed]
- Niculescu, A.B.; Levey, D.F.; Phalen, P.L.; Le-Niculescu, H.; Dainton, H.D.; Jain, N.; Belanger, E.; James, A.; George, S.; Weber, H.; et al. Understanding and predicting suicidality using a combined genomic and clinical risk assessment approach. Mol. Psychiatry 2015, 20, 1266–1285. [Google Scholar] [CrossRef] [PubMed]
- Flory, J.D.; Donohue, D.; Muhie, S.; Yang, R.; Miller, S.A.; Hammamieh, R.; Ryberg, K.; Yehuda, R. Gene expression associated with suicide attempts in US veterans. Transl. Psychiatry 2017, 7, e1226. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Lee, B.; Liu, R.-J.; Banasr, M.; Dwyer, J.M.; Iwata, M.; Li, X.-Y.; Aghajanian, G.; Duman, R.S. mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science 2010, 329, 959–964. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Liu, R.-J.; Dwyer, J.M.; Banasr, M.; Lee, B.; Son, H.; Li, X.-Y.; Aghajanian, G.; Duman, R.S. Glutamate N-methyl-D-aspartate Receptor Antagonists Rapidly Reverse Behavioral and Synaptic Deficits Caused by Chronic Stress Exposure. Boil. Psychiatry 2011, 69, 754–761. [Google Scholar] [CrossRef] [PubMed]
- Miller, O.H.; Yang, L.; Wang, C.-C.; Hargroder, E.A.; Zhang, Y.; Delpire, E.; Hall, B.J. GluN2B-containing NMDA receptors regulate depression-like behavior and are critical for the rapid antidepressant actions of ketamine. eLife 2014, 3, e03581. [Google Scholar] [CrossRef] [PubMed]
- liu, Z.; Guo, H.; Cao, X.; Cheng, C.; Yang, C.; Xu, C.; Zhang, A.; Sun, N.; Li, X.; Zhang, K. A combined study of GSK3β polymorphisms and brain network topological metrics in major depressive disorder. Psychiatry Res. Neuroimaging 2014, 223, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Shen, N.; Jin, H.; Liu, D.; Miao, X.; Zhu, L.-Q. GSK-3β Polymorphism Discriminates Bipolar Disorder and Schizophrenia: A Systematic Meta-Analysis. Mol. Neurobiol. 2013, 48, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Beurel, E.; Song, L.; Jope, R.S. Inhibition of glycogen synthase kinase-3 is necessary for the rapid antidepressant effect of ketamine in mice. Mol. Psychiatry 2011, 16, 1068–1070. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.-J.; Fuchikami, M.; Dwyer, J.M.; Lepack, A.E.; Duman, R.S.; Aghajanian, G.K. GSK-3 inhibition potentiates the synaptogenic and antidepressant-like effects of subthreshold doses of ketamine. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2013, 38, 2268–2277. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.-T.; Scheuing, L.; Liu, G.; Liao, H.-M.; Linares, G.R.; Lin, D.; Chuang, D.-M. The mood stabilizer lithium potentiates the antidepressant-like effects and ameliorates oxidative stress induced by acute ketamine in a mouse model of stress. Int. J. Neuropsychopharmacol. 2014, 18, pyu102. [Google Scholar] [CrossRef] [PubMed]
- Autry, A.E.; Adachi, M.; Nosyreva, E.; Na, E.S.; Los, M.F.; Cheng, P.-F.; Kavalali, E.T.; Monteggia, L.M. NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses. Nature 2011, 475, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Lepack, A.E.; Fuchikami, M.; Dwyer, J.M.; Banasr, M.; Duman, R.S. BDNF release is required for the behavioral actions of ketamine. Int. J. Neuropsychopharmacol. 2014, 18, pyu033. [Google Scholar] [CrossRef] [PubMed]
- Kavalali, E.T.; Monteggia, L.M. Synaptic Mechanisms Underlying Rapid Antidepressant Action of Ketamine. Am. J. Psychiatry 2012, 169, 1150–1156. [Google Scholar] [CrossRef] [PubMed]
- Monteggia, L.M.; Zarate, C. Antidepressant actions of ketamine: From molecular mechanisms to clinical practice. Curr. Opin. Neurobiol. 2015, 30, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Ignácio, Z.M.; Réus, G.Z.; Arent, C.O.; Abelaira, H.M.; Pitcher, M.R.; Quevedo, J. New perspectives on the involvement of mTOR in depression as well as in the action of antidepressant drugs. Br. J. Clin. Pharmacol. 2016, 82, 1280–1290. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.-J.; Lee, F.S.; Li, X.-Y.; Bambico, F.; Duman, R.S.; Aghajanian, G.K. Brain-derived neurotrophic factor Val66Met allele impairs basal and ketamine-stimulated synaptogenesis in prefrontal cortex. Boil. Psychiatry 2012, 71, 996–1005. [Google Scholar] [CrossRef] [PubMed]
- Petryshen, T.L.; Sabeti, P.C.; Aldinger, K.A.; Fry, B.; Fan, J.B.; Schaffner, S.F.; Waggoner, S.G.; Tahl, A.R.; Sklar, P. Population genetic study of the brain-derived neurotrophic factor (BDNF) gene. Mol. Psychiatry 2010, 15, 810–815. [Google Scholar] [CrossRef] [PubMed]
- Filakovic, P.; Eric, A.P. Pharmacotherapy of suicidal behaviour in major depression, schizophrenia and bipolar disorder. Coll. Antropol. 2013, 37, 1039–1044. [Google Scholar] [PubMed]
- Yerevanian, B.I.; Choi, Y.M. Impact of psychotropic drugs on suicide and suicidal behaviors. Bipolar Disord. 2013, 15, 594–621. [Google Scholar] [CrossRef] [PubMed]
- Yerevanian, B.I.; Koek, R.J.; Feusner, J.D.; Hwang, S.; Mintz, J. Antidepressants and suicidal behaviour in unipolar depression. Acta Psychiatr. Scand. 2004, 110, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Barbui, C.; Esposito, E.; Cipriani, A. Selective serotonin reuptake inhibitors and risk of suicide: A systematic review of observational studies. CMAJ 2009, 180, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Umetsu, R.; Abe, J.; Ueda, N.; Kato, Y.; Matsui, T.; Nakayama, Y.; Kinosada, Y.; Nakamura, M. Association between Selective Serotonin Reuptake Inhibitor Therapy and Suicidality: Analysis of U.S. Food and Drug Administration Adverse Event Reporting System Data. Biol. Pharm. Bull. 2015, 38, 1689–1699. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, L.; Moncrieff, J. The psychoactive effects of antidepressants and their association with suicidality. Curr. Drug Saf. 2011, 6, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Pawlak, J.; Dmitrzak-Weglarz, M.; Maciukiewicz, M.; Kapelski, P.; Czerski, P.; Leszczynska-Rodziewicz, A.; Zaremba, D.; Hauser, J. Personality traits as an endophenotype in genetic studies on suicidality in bipolar disorder. Acta Neuropsychiatr. 2017, 29, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Hofer, P.; Schosser, A.; Calati, R.; Serretti, A.; Massat, I.; Kocabas, N.A.; Konstantinidis, A.; Mendlewicz, J.; Souery, D.; Zohar, J.; et al. The impact of serotonin receptor 1A and 2A gene polymorphisms and interactions on suicide attempt and suicide risk in depressed patients with insufficient response to treatment--a European multicentre study. Int. Clin. Psychopharmacol. 2016, 31, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Fanelli, G.; Serretti, A. The influence of the serotonin transporter gene 5-HTTLPR polymorphism on suicidal behaviors: A meta-analysis. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 88, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Jacob, D.; Baldessarini, R.; Conwell, Y.; Fawcett, J.; Horton, L.; Meltzer, H.; Pfeffer, C.; Simon, R. Practice guideline for the assessment and treatment of patients with suicidal behaviors. Am. J. Psychiatry 2003, 160, 1–60. [Google Scholar]
- Tondo, L.; Baldessarini, R.J. Antisuicidal Effects in Mood Disorders: Are They Unique to Lithium? Pharmacopsychiatry 2018, 51, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Toffol, E.; Hatonen, T.; Tanskanen, A.; Lonnqvist, J.; Wahlbeck, K.; Joffe, G.; Tiihonen, J.; Haukka, J.; Partonen, T. Lithium is associated with decrease in all-cause and suicide mortality in high-risk bipolar patients: A nationwide registry-based prospective cohort study. J. Affect. Disord. 2015, 183, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Halcomb, M.E.; Gould, T.D.; Grahame, N.J. Lithium, but not valproate, reduces impulsive choice in the delay-discounting task in mice. Neuropsychopharmacology 2013, 38, 1937–1944. [Google Scholar] [CrossRef] [PubMed]
- Ohmura, Y.; Tsutsui-Kimura, I.; Kumamoto, H.; Minami, M.; Izumi, T.; Yamaguchi, T.; Yoshida, T.; Yoshioka, M. Lithium, but not valproic acid or carbamazepine, suppresses impulsive-like action in rats. Psychopharmacology 2012, 219, 421–432. [Google Scholar] [CrossRef] [PubMed]
- Beurel, E.; Jope, R.S. Inflammation and lithium: Clues to mechanisms contributing to suicide-linked traits. Transl. Psychiatry 2014, 4, e488. [Google Scholar] [CrossRef] [PubMed]
- Malhi, G.S.; Outhred, T. Therapeutic Mechanisms of Lithium in Bipolar Disorder: Recent Advances and Current Understanding. CNS Drugs 2016, 30, 931–949. [Google Scholar] [CrossRef] [PubMed]
- Can, A.; Schulze, T.G.; Gould, T.D. Molecular actions and clinical pharmacogenetics of lithium therapy. Pharmacol. Biochem. Behav. 2014, 123, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, J.M.; Zhang, X.; Rodriguiz, R.M.; Sotnikova, T.D.; Cools, M.J.; Wetsel, W.C.; Gainetdinov, R.R.; Caron, M.G. Role of GSK3 beta in behavioral abnormalities induced by serotonin deficiency. Proc. Natl. Acad. Sci. USA 2008, 105, 1333–1338. [Google Scholar] [CrossRef] [PubMed]
- Pawlak, J.; Dmitrzak-Weglarz, M.; Maciukiewicz, M.; Wilkosc, M.; Leszczynska-Rodziewicz, A.; Zaremba, D.; Kapelski, P.; Hauser, J. Suicidal behavior in the context of disrupted rhythmicity in bipolar disorder--data from an association study of suicide attempts with clock genes. Psychiatry Res. 2015, 226, 517–520. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Maneen, M.J.; Stahl, S.M. Building a better antipsychotic: Receptor targets for the treatment of multiple symptom dimensions of schizophrenia. Neurotherapeutics 2009, 6, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.R.; Bai, Y.Y.; Ruan, C.S.; Zhou, H.F.; Liu, D.; Wang, X.F.; Shen, L.J.; Zheng, H.Y.; Zhou, X.F. Enhanced aggressive behaviour in a mouse model of depression. Neurotox. Res. 2015, 27, 129–142. [Google Scholar] [CrossRef] [PubMed]
- Deslauriers, J.; Belleville, K.; Beaudet, N.; Sarret, P.; Grignon, S. A two-hit model of suicide-trait-related behaviors in the context of a schizophrenia-like phenotype: Distinct effects of lithium chloride and clozapine. Physiol. Behav. 2016, 156, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Celada, P.; Bortolozzi, A.; Artigas, F. Serotonin 5-HT1A receptors as targets for agents to treat psychiatric disorders: Rationale and current status of research. CNS Drugs 2013, 27, 703–716. [Google Scholar] [CrossRef] [PubMed]
- Maurice-Gelinas, C.; Deslauriers, J.; Monpays, C.; Sarret, P.; Grignon, S. The 5α-reductase inhibitor finasteride increases suicide-related aggressive behaviors and blocks clozapine-induced beneficial effects in an animal model of schizophrenia. Physiol. Behav. 2018, 191, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Kudoh, A.; Katagai, H.; Takazawa, T. Anesthesia with ketamine, propofol, and fentanyl decreases the frequency of postoperative psychosis emergence and confusion in schizophrenic patients. J. Clin. Anesth. 2002, 14, 107–110. [Google Scholar] [CrossRef]
- Zarate, C.A., Jr.; Brutsche, N.E.; Ibrahim, L.; Franco-Chaves, J.; Diazgranados, N.; Cravchik, A.; Selter, J.; Marquardt, C.A.; Liberty, V.; Luckenbaugh, D.A. Replication of ketamine’s antidepressant efficacy in bipolar depression: A randomized controlled add-on trial. Biol. Psychiatry 2012, 71, 939–946. [Google Scholar] [CrossRef] [PubMed]
- Andrade, C. Ketamine for Depression, 3: Does Chirality Matter? J. Clin. Psychiatry 2017, 78, e674–e677. [Google Scholar] [CrossRef] [PubMed]
- Canuso, C.M.; Singh, J.B.; Fedgchin, M.; Alphs, L.; Lane, R.; Lim, P.; Pinter, C.; Hough, D.; Sanacora, G.; Manji, H.; et al. Efficacy and Safety of Intranasal Esketamine for the Rapid Reduction of Symptoms of Depression and Suicidality in Patients at Imminent Risk for Suicide: Results of a Double-Blind, Randomized, Placebo-Controlled Study. Am. J. Psychiatry 2018. [Google Scholar] [CrossRef] [PubMed]
- Grunebaum, M.F.; Galfalvy, H.C.; Choo, T.H.; Keilp, J.G.; Moitra, V.K.; Parris, M.S.; Marver, J.E.; Burke, A.K.; Milak, M.S.; Sublette, M.E.; et al. Ketamine for Rapid Reduction of Suicidal Thoughts in Major Depression: A Midazolam-Controlled Randomized Clinical Trial. Am. J. Psychiatry 2018, 175, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Grunebaum, M.F.; Ellis, S.P.; Keilp, J.G.; Moitra, V.K.; Cooper, T.B.; Marver, J.E.; Burke, A.K.; Milak, M.S.; Sublette, M.E.; Oquendo, M.A.; et al. Ketamine versus midazolam in bipolar depression with suicidal thoughts: A pilot midazolam-controlled randomized clinical trial. Bipolar Disord. 2017, 19, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Yang, H.; Sun, Y.; Zhang, J.; Li, G.; Zheng, Y.; Liu, Y. Ketamine rapidly relieves acute suicidal ideation in cancer patients: A randomized controlled clinical trial. Oncotarget 2017, 8, 2356–2360. [Google Scholar] [CrossRef] [PubMed]
- Burger, J.; Capobianco, M.; Lovern, R.; Boche, B.; Ross, E.; Darracq, M.A.; McLay, R. A Double-Blinded, Randomized, Placebo-Controlled Sub-Dissociative Dose Ketamine Pilot Study in the Treatment of Acute Depression and Suicidality in a Military Emergency Department Setting. Mil. Med. 2016, 181, 1195–1199. [Google Scholar] [CrossRef] [PubMed]
- Murrough, J.W.; Soleimani, L.; DeWilde, K.E.; Collins, K.A.; Lapidus, K.A.; Iacoviello, B.M.; Lener, M.; Kautz, M.; Kim, J.; Stern, J.B.; et al. Ketamine for rapid reduction of suicidal ideation: A randomized controlled trial. Psychol. Med. 2015, 45, 3571–3580. [Google Scholar] [CrossRef] [PubMed]
- Price, R.B.; Iosifescu, D.V.; Murrough, J.W.; Chang, L.C.; Al Jurdi, R.K.; Iqbal, S.Z.; Soleimani, L.; Charney, D.S.; Foulkes, A.L.; Mathew, S.J. Effects of ketamine on explicit and implicit suicidal cognition: A randomized controlled trial in treatment-resistant depression. Depress Anxiety 2014, 31, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Kudoh, A.; Takahira, Y.; Katagai, H.; Takazawa, T. Small-dose ketamine improves the postoperative state of depressed patients. Anesth. Analg. 2002, 95, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.D.; Xiang, Y.T.; Fang, J.X.; Zu, S.; Sha, S.; Shi, H.; Ungvari, G.S.; Correll, C.U.; Chiu, H.F.; Xue, Y.; et al. Single i.v. ketamine augmentation of newly initiated escitalopram for major depression: Results from a randomized, placebo-controlled 4-week study. Psychol. Med. 2016, 46, 623–635. [Google Scholar] [CrossRef] [PubMed]
- Andrade, C. Ketamine for Depression, 6: Effects on Suicidal Ideation and Possible Use as Crisis Intervention in Patients at Suicide Risk. J. Clin. Psychiatry 2018, 79. [Google Scholar] [CrossRef] [PubMed]
- Andrade, C. Ketamine for Depression, 2: Diagnostic and Contextual Indications. J. Clin. Psychiatry 2017, 78, e555–e558. [Google Scholar] [CrossRef] [PubMed]
- Nemeroff, C.B. Ketamine: Quo Vadis? Am. J. Psychiatry 2018, 175, 297–299. [Google Scholar] [CrossRef] [PubMed]
- Schak, K.M.; Vande Voort, J.L.; Johnson, E.K.; Kung, S.; Leung, J.G.; Rasmussen, K.G.; Palmer, B.A.; Frye, M.A. Potential Risks of Poorly Monitored Ketamine Use in Depression Treatment. Am. J. Psychiatry 2016, 173, 215–218. [Google Scholar] [CrossRef] [PubMed]
- Sanacora, G.; Frye, M.A.; McDonald, W.; Mathew, S.J.; Turner, M.S.; Schatzberg, A.F.; Summergrad, P.; Nemeroff, C.B. American Psychiatric Association (APA) Council of Research Task Force on Novel Biomarkers and Treatments. A Consensus Statement on the Use of Ketamine in the Treatment of Mood Disorders. JAMA Psychiatry 2017, 74, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Sanacora, G.; Heimer, H.; Hartman, D.; Mathew, S.J.; Frye, M.; Nemeroff, C.; Robinson Beale, R. Balancing the Promise and Risks of Ketamine Treatment for Mood Disorders. Neuropsychopharmacology 2017, 42, 1179–1181. [Google Scholar] [CrossRef] [PubMed]
- Tomasetti, C.; Iasevoli, F.; Buonaguro, E.F.; De Berardis, D.; Fornaro, M.; Fiengo, A.L.; Martinotti, G.; Orsolini, L.; Valchera, A.; Di Giannantonio, M.; et al. Treating the Synapse in Major Psychiatric Disorders: The Role of Postsynaptic Density Network in Dopamine-Glutamate Interplay and Psychopharmacologic Drugs Molecular Actions. Int. J. Mol. Sci. 2017, 18, 135. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Verwer, R.W.; van Wamelen, D.J.; Qi, X.R.; Gao, S.F.; Lucassen, P.J.; Swaab, D.F. Prefrontal changes in the glutamate-glutamine cycle and neuronal/glial glutamate transporters in depression with and without suicide. J. Psychiatr. Res. 2016, 82, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Andrade, C. Ketamine for Depression, 1: Clinical Summary of Issues Related to Efficacy, Adverse Effects, and Mechanism of Action. J. Clin. Psychiatry 2017, 78, e415–e419. [Google Scholar] [CrossRef] [PubMed]
- Duman, R.S. Ketamine and rapid-acting antidepressants: A new era in the battle against depression and suicide. F1000Res 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Zarate, C.A., Jr.; Machado-Vieira, R. GSK-3: A key regulatory target for ketamine’s rapid antidepressant effects mediated by enhanced AMPA to NMDA throughput. Bipolar Disord. 2016, 18, 702–705. [Google Scholar] [CrossRef] [PubMed]
- Le Nedelec, M.; Glue, P.; Winter, H.; Goulton, C.; Broughton, L.; Medlicott, N. Acute low-dose ketamine produces a rapid and robust increase in plasma BDNF without altering brain BDNF concentrations. Drug Deliv. Transl. Res. 2018, 8, 780–786. [Google Scholar] [CrossRef] [PubMed]
- Abelaira, H.M.; Reus, G.Z.; Ignacio, Z.M.; Dos Santos, M.A.; de Moura, A.B.; Matos, D.; Demo, J.P.; da Silva, J.B.; Michels, M.; Abatti, M.; et al. Effects of ketamine administration on mTOR and reticulum stress signaling pathways in the brain after the infusion of rapamycin into prefrontal cortex. J. Psychiatr. Res. 2017, 87, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Wang, N.; Yang, C.; Li, X.M.; Zhou, Z.Q.; Yang, J.J. Ketamine-induced antidepressant effects are associated with AMPA receptors-mediated upregulation of mTOR and BDNF in rat hippocampus and prefrontal cortex. Eur. Psychiatry 2014, 29, 419–423. [Google Scholar] [CrossRef] [PubMed]
| Authors | Year | Study Sample | Study Design | Comparator | Number of Subject | Study Aims | Ketamine Dose | Main Findings |
|---|---|---|---|---|---|---|---|---|
| Canuso et al. | 2018 | Subjects with MDD and suicidal ideation | Randomized, placebo-controlled trial | Intranasal Placebo | 68 | To compared the efficacy of standard-of-care treatment plus intranasal esketamine or placebo for rapid reduction of symptoms of MDD, including suicidality | Intranasal esketamine (84 mg) twice weekly for 4 weeks | Intranasal esketamine compared with placebo, given in addition to comprehensive standard-of-care treatment, may result in significantly rapid improvement in depressive symptoms, including suicidal ideation |
| Grunebaum et al. | 2018 | Tested the acute effect of adjunctive subanesthetic intravenous ketamine on clinically significant suicidal ideation in patients with MDD | Randomized, controlled trial | Midazolam | 80 | To test the acute effect of adjunctive subanesthetic intravenous ketamine on clinically significant suicidal ideation in MDD | 0.5 mg/kg infused over 40 min | Adjunctive ketamine demonstrated a greater reduction in clinically significant suicidal ideation in depressed patients within 24 h compared with midazolam, partially independently of antidepressant effect |
| Grunebaum et al. | 2017 | Subjects with bipolar depression and suicidal ideation | Pilot, randomized, controlled trial | Midazolam | 16 | To evaluate feasibility and effects of a sub-anesthetic infusion dose of ketamine versus midazolam on suicidal ideation in bipolar depression | 0.5 mg/kg infused over 40 min | Suicidal thoughts were lower after ketamine than after midazolam at a trend level of significance |
| Fan et al. | 2017 | Cancer patients with suicidal ideation | Randomized, controlled trial | Midazolam | 37 | To evaluate the rapid antidepressant effects of single dose ketamine on suicidal ideation and overall depression level in patients with newly-diagnosed cancer | 0.5 mg/kg infused over 40 min | Antidepressant and anti-suicidal effects of ketamine were significantly seen as soon as 1 day following administration and typically lasted for at least 3 days |
| Burger et al. | 2016 | Individuals presenting with suicidal thinking in a military setting | Randomized, placebo-controlled trial | Normal saline (placebo) | 10 | To evaluate the potential benefits of ketamine vs. placebo administered to individuals presenting with suicidal thinking in a military setting | 0.5 mg/kg infused over 40 min | Two of three who received ketamine experienced dramatic decreases in suicidality and hopelessness within 40 min |
| Hu et al. | 2016 | Thirty outpatients with severe MDD | Randomized, placebo-controlled 4-week study combined with escitalopram 10 mg | Normal saline (placebo) | 30 | To evaluate the antidepressant and anti-suicidal effects of single i.v. ketamine combined with escitalopram initiation in MDD | 0.5 mg/kg infused over 40 min | Single-dose i.v. ketamine showed an attenuation of the antidepressant effects by the end of week 1. Suicidal ideation improved significantly, which was sustained for up to 10 days |
| Murrough et al. | 2016 | Subjects with mood and anxiety disorders spectrum | Randomized, controlled trial | Midazolam | 24 | To evaluate acute effect of i.v. ketamine on clinically significant suicidal ideation in patients with mood and anxiety disorders spectrum | 0.5 mg/kg infused over 40 min | A single adjunctive ketamine infusion was associated with a clinically significant reduction in suicidal ideation at 48 h that was greater than with the midazolam control infusion |
| Price et al. | 2014 | Symptomatic patients with treatment-resistant MDD | Randomized, controlled trial | Midazolam | 57 | To evaluate acute effect of i.v. ketamine vs midazolam on explicit and implicit suicidal cognition | 0.5 mg/kg infused over 40 min | i.v. ketamine showed rapid reductions in suicidal cognition over and above midazolam |
| Zarate et al. | 2012 | Subjects with DSM-IV bipolar I or II depression maintained on therapeutic levels of lithium or valproate | Double-blind, randomized, crossover, placebo-controlled study | Normal saline (placebo) | 15 | To evaluate acute effect of i.v. ketamine on clinically significant suicidal ideation in patients with bipolar depression | 0.5 mg/kg infused over 40 min | Within 40 min, depressive symptoms, as well as suicidal ideation, significantly improved in subjects receiving ketamine compared with placebo and this improvement remained significant through day 3 |
| Kudoh et al. | 2002 | Depressed patients who had undergone orthopedic surgery | Randomized study. Patients in Group A were induced with propofol, fentanyl, and ketamine and patients in Group B were induced with propofol and fentanyl | Propofol, fentanyl | 95 | To evaluate acute effect of i.v. ketamine on clinically significant suicidal ideation in depressed patients who had undergone orthopedic surgery | 0.5 mg/kg combined with propofol and fentanyl | Depressed mood, suicidal tendencies, somatic anxiety, and hypochondriasis significantly decreased in Group A as compared with Group B |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Berardis, D.; Fornaro, M.; Valchera, A.; Cavuto, M.; Perna, G.; Di Nicola, M.; Serafini, G.; Carano, A.; Pompili, M.; Vellante, F.; et al. Eradicating Suicide at Its Roots: Preclinical Bases and Clinical Evidence of the Efficacy of Ketamine in the Treatment of Suicidal Behaviors. Int. J. Mol. Sci. 2018, 19, 2888. https://doi.org/10.3390/ijms19102888
De Berardis D, Fornaro M, Valchera A, Cavuto M, Perna G, Di Nicola M, Serafini G, Carano A, Pompili M, Vellante F, et al. Eradicating Suicide at Its Roots: Preclinical Bases and Clinical Evidence of the Efficacy of Ketamine in the Treatment of Suicidal Behaviors. International Journal of Molecular Sciences. 2018; 19(10):2888. https://doi.org/10.3390/ijms19102888
Chicago/Turabian StyleDe Berardis, Domenico, Michele Fornaro, Alessandro Valchera, Marilde Cavuto, Giampaolo Perna, Marco Di Nicola, Gianluca Serafini, Alessandro Carano, Maurizio Pompili, Federica Vellante, and et al. 2018. "Eradicating Suicide at Its Roots: Preclinical Bases and Clinical Evidence of the Efficacy of Ketamine in the Treatment of Suicidal Behaviors" International Journal of Molecular Sciences 19, no. 10: 2888. https://doi.org/10.3390/ijms19102888
APA StyleDe Berardis, D., Fornaro, M., Valchera, A., Cavuto, M., Perna, G., Di Nicola, M., Serafini, G., Carano, A., Pompili, M., Vellante, F., Orsolini, L., Fiengo, A., Ventriglio, A., Yong-Ku, K., Martinotti, G., Di Giannantonio, M., & Tomasetti, C. (2018). Eradicating Suicide at Its Roots: Preclinical Bases and Clinical Evidence of the Efficacy of Ketamine in the Treatment of Suicidal Behaviors. International Journal of Molecular Sciences, 19(10), 2888. https://doi.org/10.3390/ijms19102888














