Oncomirs Expression Profiling in Uterine Leiomyosarcoma Cells
Abstract
:1. Introduction
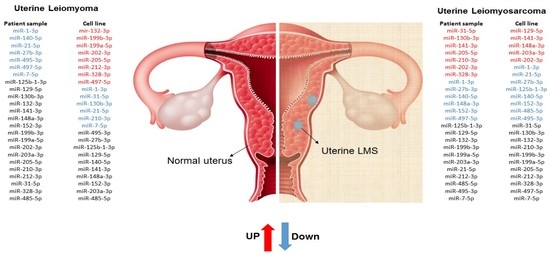
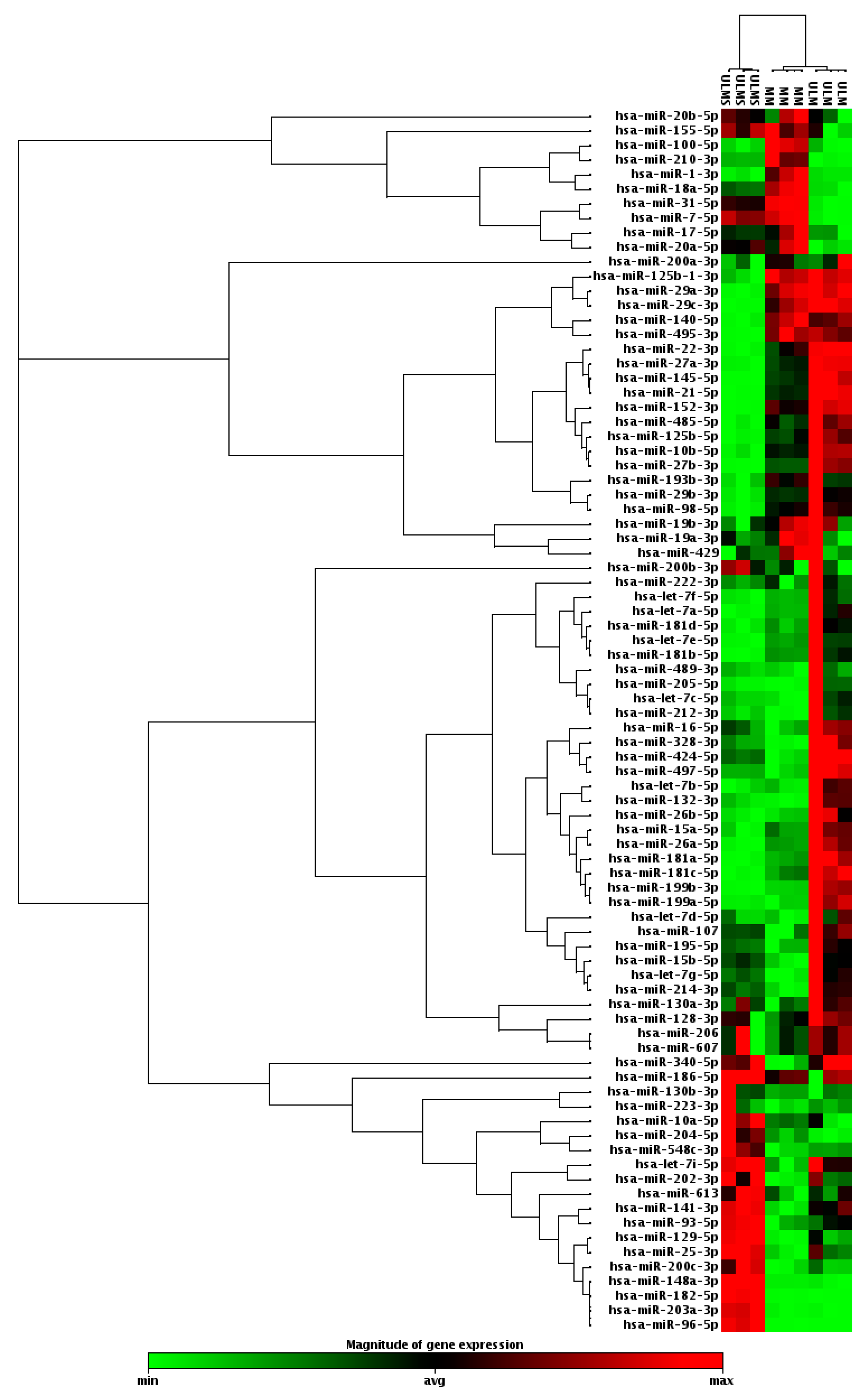
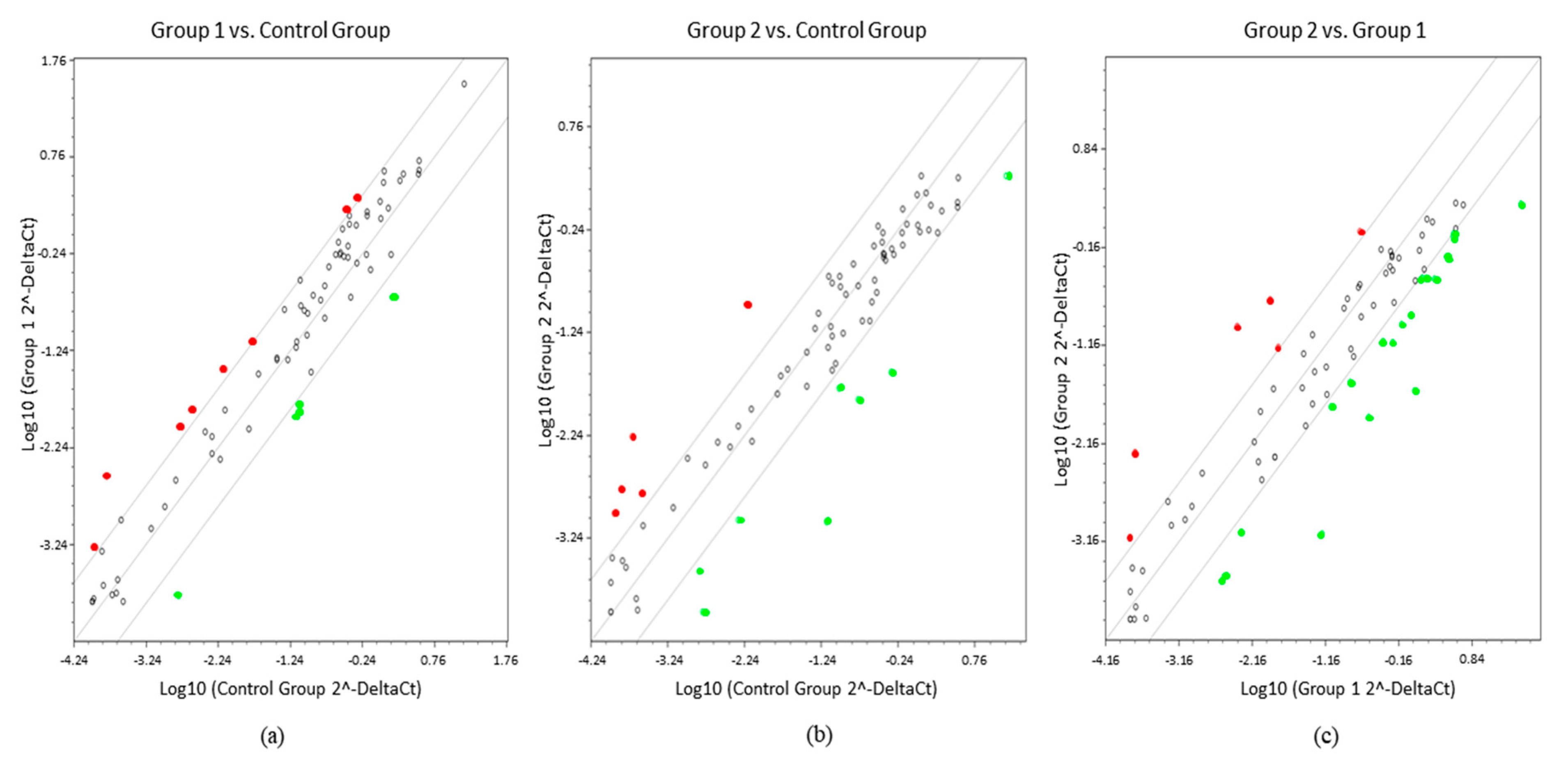
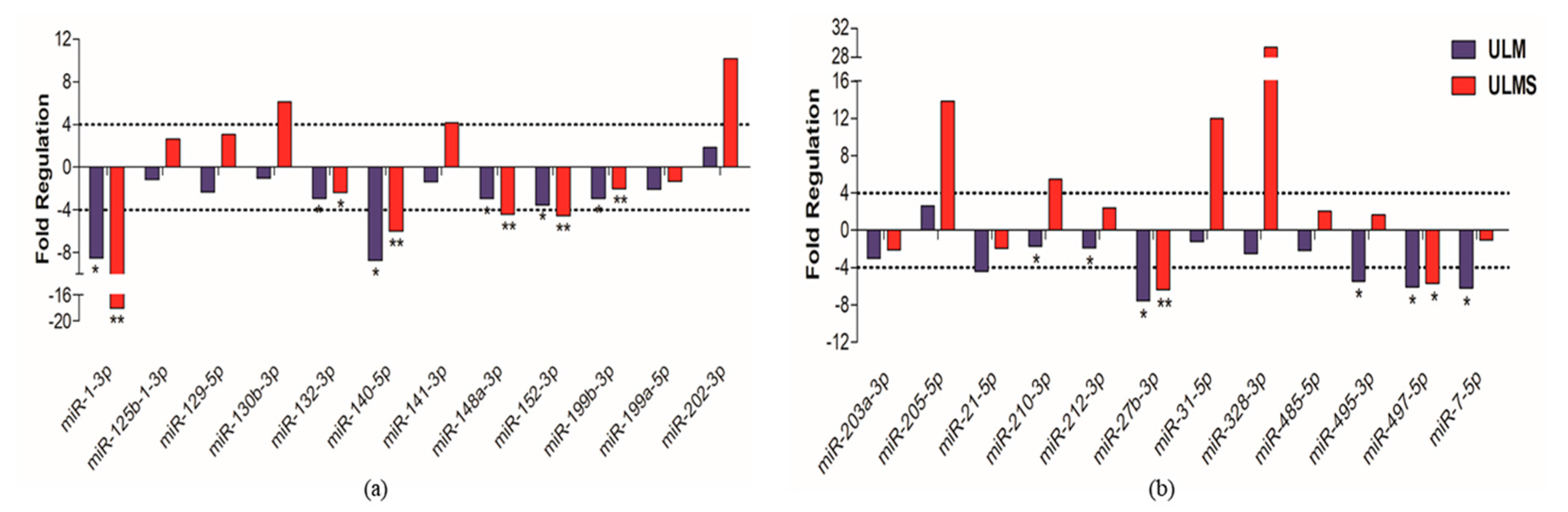
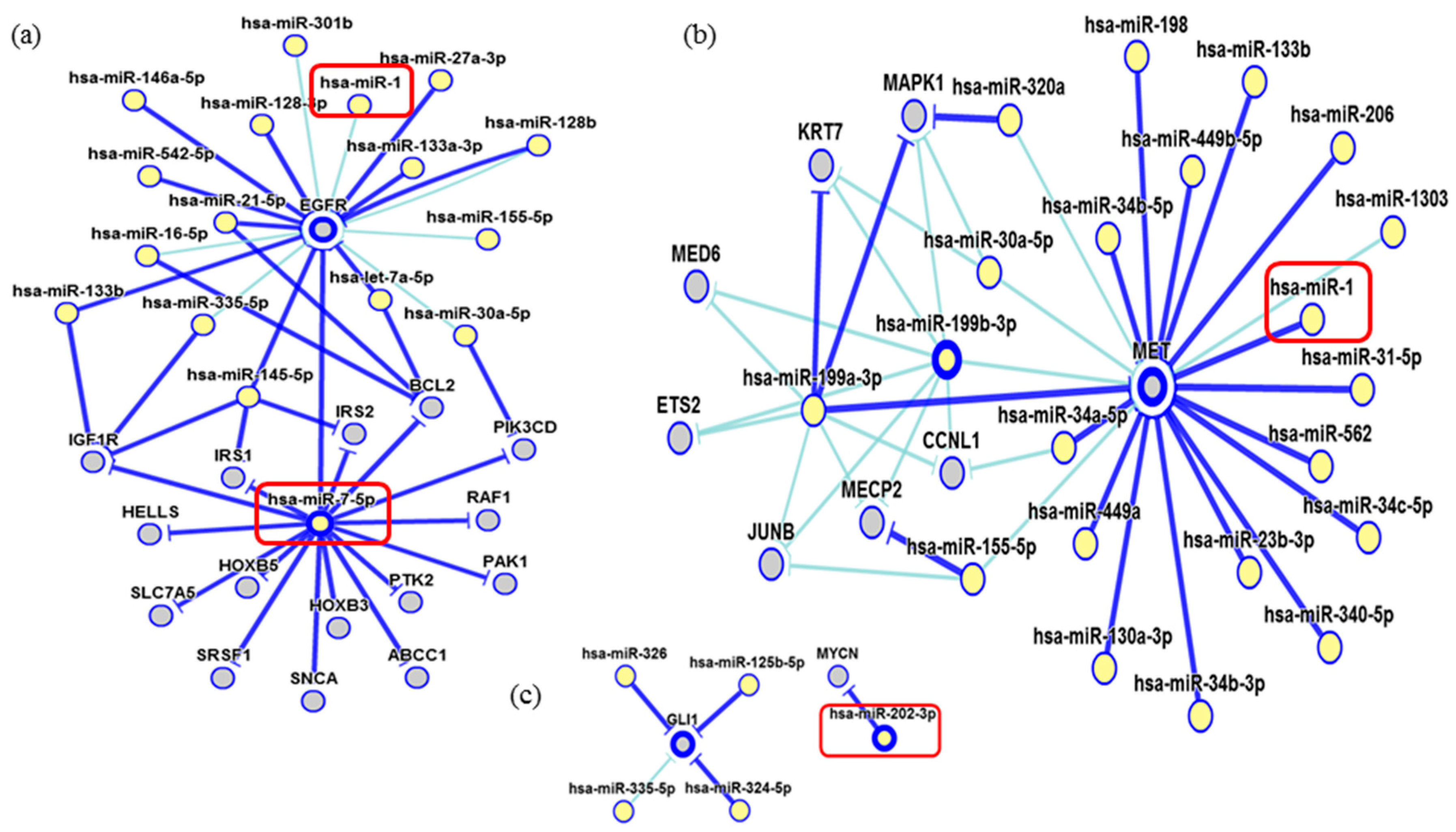
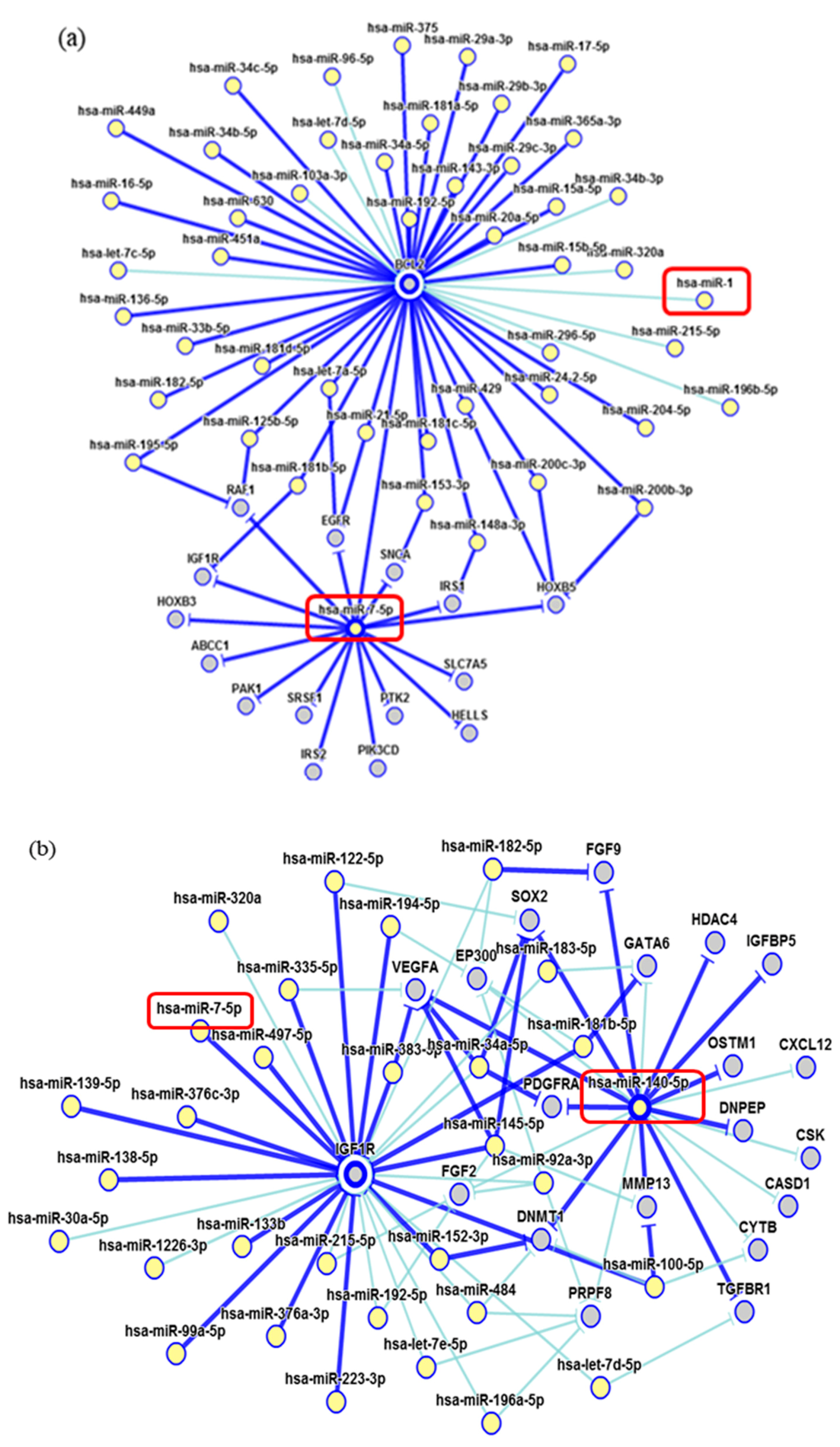
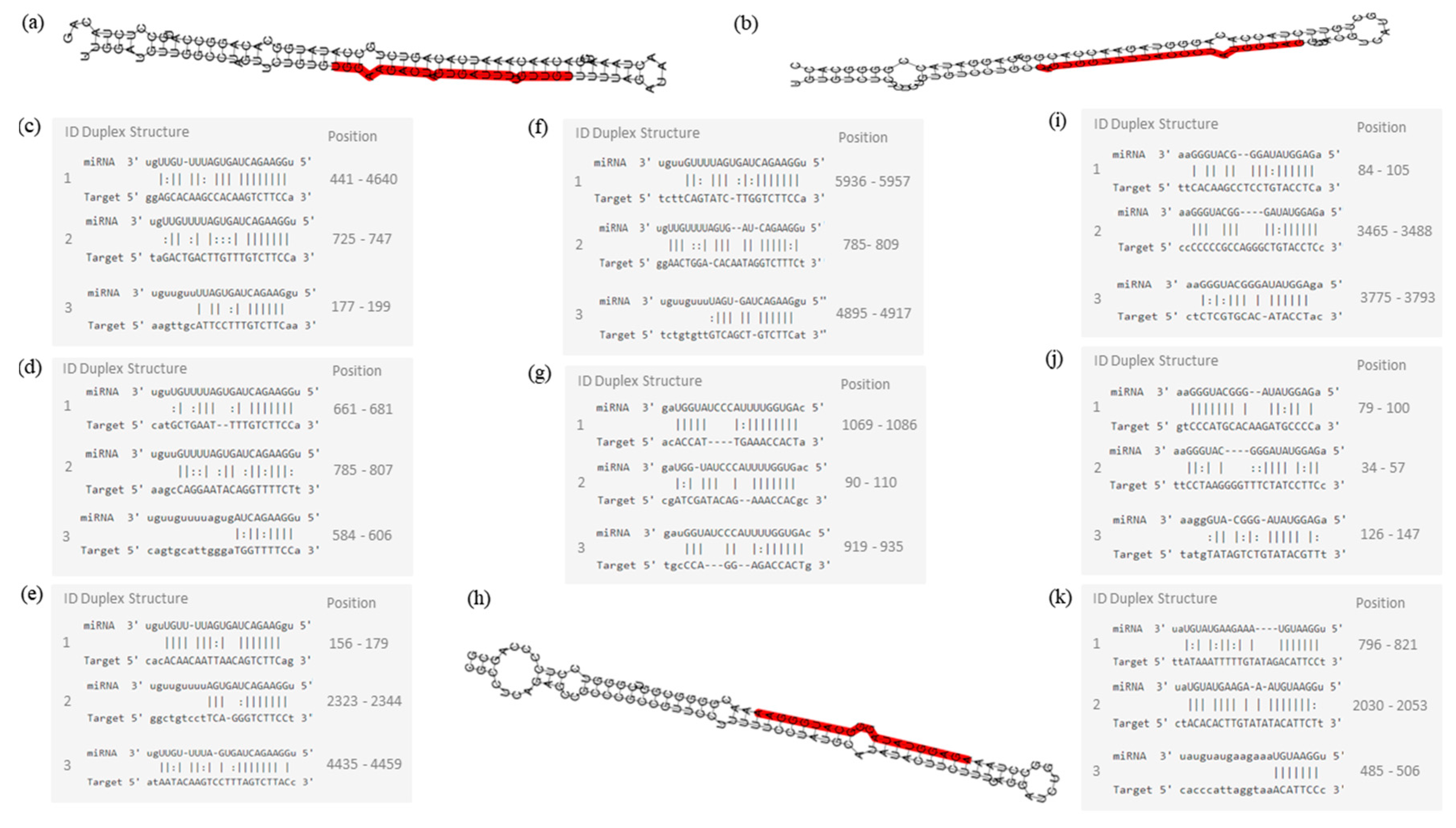
2. Results
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Human Samples
4.3. MicroRNA and RNA Isolation
4.4. microRNA qRT-PCR
4.5. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Georgieva, B.; Milev, I.; Minkov, I.; Dimitrova, I.; Bradford, A.P.; Baev, V. Characterization of the uterine leiomyoma microRNAome by deep sequencing. Genomics 2012, 99, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Bell, E.; Taylor, M.A. Functional roles for exosomal microRNAs in the tumour microenvironment. Comput. Struct. Biotechnol. J. 2017, 15, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Kowalewska, M.; Bakula-Zalewska, E.; Chechlinska, M.; Goryca, K.; Nasierowska-Guttmejer, A.; Danska-Bidzinska, A.; Bidzinski, M. microRNAs in uterine sarcomas and mixed epithelial-mesenchymal uterine tumors: A preliminary report. Tumour Biol. 2013, 34, 2153–2160. [Google Scholar] [CrossRef] [PubMed]
- Farazi, T.A.; Hoell, J.I.; Morozov, P.; Tuschl, T. MicroRNA cancer regulation. Adv. Exp. Med. Biol. 2013, 774, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Danielson, L.S.; Menendez, S.; Attolini, C.S.-O.; Guijarro, M.V.; Bisogna, M.; Wei, J.; Socci, N.D.; Levine, D.A.; Michor, F.; Hernando, E. A differentiation-based microRNA signature identifies leiomyosarcoma as a mesenchymal stem cell-related malignancy. Am. J. Pathol. 2010, 177, 908–917. [Google Scholar] [CrossRef] [PubMed]
- Mittal, K.R.; Chen, F.; Wei, J.J.; Rijhvani, K.; Kurvathi, R.; Streck, D.; Dermody, J.; Toruner, G.A. Molecular and immunohistochemical evidence for the origin of uterine leiomyosarcomas from associated leiomyoma and symplastic leiomyoma-like areas. Mod. Pathol. 2009, 22, 1303–1311. [Google Scholar] [CrossRef] [PubMed]
- De Almeida, T.G.; da Cunha, I.W.; Maciel, G.A.R.; Baracat, E.C.; Carvalho, K.C. Clinical and molecular features of uterine sarcomas. Med. Express 2014, 1, 291–297. [Google Scholar] [CrossRef]
- Abeler, V.M.; Røyne, O.; Thoresen, S.; Danielsen, H.E.; Nesland, J.M.; Kristensen, G.B. Uterine sarcomas in Norway. A histopathological and prognostic survey of a total population from 1970 to 2000 including 419 patients. Histopathology 2009, 54, 355–364. [Google Scholar] [CrossRef] [PubMed]
- D’Angelo, E.; Spagnoli, L.G.; Prat, J. Comparative clinicopathologic and immunohistochemical analysis of uterine sarcomas diagnosed using the World Health Organization classification system. Hum. Pathol. 2009, 40, 1571–1585. [Google Scholar] [CrossRef] [PubMed]
- Chuang, T.-D.; Ho, M.; Khorram, O. The regulatory function of miR-200c on inflammatory and cell-cycle associated genes in SK-LMS-1, a leiomyosarcoma cell line. Reprod. Sci. 2015, 22, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, H. The biology of uterine sarcomas: A review and update. Mol. Clin. Oncol. 2013, 599–609. [Google Scholar] [CrossRef] [PubMed]
- Guled, M.; Pazzaglia, L.; Borze, I.; Mosakhani, N.; Novello, C.; Benassi, M.S.; Knuutila, S. Differentiating soft tissue leiomyosarcoma and undifferentiated pleomorphic sarcoma: A miRNA analysis. Genes Chromosomes Cancer 2014, 53, 693–702. [Google Scholar] [CrossRef] [PubMed]
- Garcia, N.; Bozzini, N.; Baiocchi, G.; da Cunha, I.W.; Maciel, G.A.; Soares, J.M.; Soares, F.A.; Baracat, E.C.; Carvalho, K.C. May Sonic Hedgehog proteins be markers for malignancy in uterine smooth muscle tumors? Hum. Pathol. 2016, 50, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Stewart, E.A. Differentiating Uterine Leiomyomas (Fibroids) from Uterine Sarcomas—UpToDate. Available online: https://www.uptodate.com/contents/differentiating-uterine-leiomyomas-fibroids-from-uterine-sarcomas (accessed on 6 September 2017).
- Deng, X.; Hou, C.; Liang, Z.; Wang, H.; Zhu, L.; Xu, H. miR-202 suppresses cell proliferation by targeting FOXR2 in Endometrial adenocarcinoma. Dis. Markers 2017, 2017, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Huang, Z.; Guo, W.; Ni, S.; Xiao, X.; Wang, L.; Huang, D.; Tan, C.; Xu, Q.; Zha, R.; et al. MicroRNA-202-3p inhibits cell proliferation by targeting ADP-ribosylation factor-like 5A in human colorectal carcinoma. Clin. Cancer Res. 2014, 20, 1146–1157. [Google Scholar] [CrossRef] [PubMed]
- Farhana, L.; Dawson, M.I.; Fontana, J.A. Down regulation of miR-202 modulates Mxd1 and Sin3A repressor complexes to induce apoptosis of pancreatic cancer cells. Cancer Biol. Ther. 2015, 16, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Conconi, D.; Chiappa, V.; Perego, P.; Redaelli, S.; Bovo, G.; Lavitrano, M.; Milani, R.; Dalprà, L.; Lissoni, A.A. Potential role of BCL2 in the recurrence of uterine smooth muscle tumors of uncertain malignant potential. Oncol. Rep. 2017, 37, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-S.; Ahn, C.; Kang, H.Y.; Jeung, E.-B. Effect of 2-methoxyestradiol on SK-LMS-1 uterine leiomyosarcoma cells. Oncol. Lett. 2017, 14, 103. [Google Scholar] [CrossRef] [PubMed]
- Bishop, E.A.; Lengyel, E.R.; Yamada, S.D.; Montag, A.; Temkin, S.M. The expression of hepatocyte growth factor (HGF) and c-Met in uterine serous carcinoma. Gynecol. Oncol. 2011, 121, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Korompelis, P.; Piperi, C.; Adamopoulos, C.; Dalagiorgou, G.; Korkolopoulou, P.; Sepsa, A.; Antsaklis, A.; Papavassiliou, A.G. Expression of vascular endothelial factor-A, gelatinases (MMP-2, MMP-9) and TIMP-1 in uterine leiomyomas. Clin. Chem. Lab. Med. 2015, 53, 1415–1424. [Google Scholar] [CrossRef] [PubMed]
- Gkioka, E.; Msaouel, P.; Philippou, A.; Vlaghogiannis, N.I.; Vogkou, C.T.; Margiolis, A.; Koutsilieris, M. The role of insulin-like growth factor-1 signaling pathways in uterine leiomyoma. In Vivo 2015, 29, 637–649. [Google Scholar] [PubMed]
- Ciarmela, P.; Islam, M.S.; Reis, F.M.; Gray, P.C.; Bloise, E.; Petraglia, F.; Vale, W.; Castellucci, M. Growth factors and myometrium: Biological effects in uterine fibroid and possible clinical implications. Hum. Reprod. Update 2011, 17, 772–790. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.Q.; Cao, B.R.; Zhang, H.; Luo, L.P.; Ren, Y.; Hu, T.; Chen, C.M. The lack of Raf-1 kinase feedback regulation enhances antiapoptosis in cancer cells. Oncogene 2017, 36, 2014–2022. [Google Scholar] [CrossRef] [PubMed]
- Berindan-Neagoe, I.; Monroig, P.C.; Pasculli, B.; Calin, G.A. MicroRNAome genome: A treasure for cancer diagnosis and therapy. CA Cancer J. Clin. 2014, 64, 311–336. [Google Scholar] [CrossRef] [PubMed]
- Mendell, J.T.; Olson, E.N. MicroRNAs in stress signaling and human disease. Cell 2013, 148, 1172–1187. [Google Scholar] [CrossRef] [PubMed]
- Karmon, A.E.; Cardozo, E.R.; Rueda, B.R.; Styer, A.K. MicroRNAs in the development and pathobiology of uterine leiomyomata: Does evidence support future strategies for clinical intervention? Hum. Reprod. Update 2014, 20, 670–687. [Google Scholar] [CrossRef] [PubMed]
- Genorm. Available online: https://genorm.cmgg.be/ (accessed on 18 September 2017).
- miScript miRNA PCR Array Data Analysis. Available online: http://pcrdataanalysis.sabiosciences.com/mirna/arrayanalysis.php (accessed on 18 September 2017).
- miRTarBase. Available online: http://mirtarbase.mbc.nctu.edu.tw/php/index.php (accessed on 13 August 2017).



| Variable | Features | N (%) |
|---|---|---|
| Menopause | YES | 8 (50%) |
| NO | 6 (37.5%) | |
| Clinical FIGO stage | Tumor restricted to the uterus | 8 (50%) |
| Pelvic extension | 2 (12.5%) | |
| Extra-pelvic extension | 2 (12.5%) | |
| Visceral metastasis | 4 (25%) | |
| Symptoms | Pain | 3 (19%) |
| Bleeding | 6 (37.5%) | |
| Discharge | 0 (0%) | |
| Pelvic mass | 1 (6%) | |
| No symptoms | 2 (12.5%) | |
| Relapse | YES | 3 (18.75%) |
| NO | 6 (37.5%) | |
| Persistence of disease | 3 (18.75%) | |
| Metastasis | YES | 4 (25%) |
| NO | 6 (37.5%) | |
| Death | YES | 11 (69%) |
| NO | 3 (19%) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Almeida, B.C.; Garcia, N.; Maffazioli, G.; Gonzalez dos Anjos, L.; Chada Baracat, E.; Candido Carvalho, K. Oncomirs Expression Profiling in Uterine Leiomyosarcoma Cells. Int. J. Mol. Sci. 2018, 19, 52. https://doi.org/10.3390/ijms19010052
De Almeida BC, Garcia N, Maffazioli G, Gonzalez dos Anjos L, Chada Baracat E, Candido Carvalho K. Oncomirs Expression Profiling in Uterine Leiomyosarcoma Cells. International Journal of Molecular Sciences. 2018; 19(1):52. https://doi.org/10.3390/ijms19010052
Chicago/Turabian StyleDe Almeida, Bruna Cristine, Natalia Garcia, Giovana Maffazioli, Laura Gonzalez dos Anjos, Edmund Chada Baracat, and Katia Candido Carvalho. 2018. "Oncomirs Expression Profiling in Uterine Leiomyosarcoma Cells" International Journal of Molecular Sciences 19, no. 1: 52. https://doi.org/10.3390/ijms19010052
APA StyleDe Almeida, B. C., Garcia, N., Maffazioli, G., Gonzalez dos Anjos, L., Chada Baracat, E., & Candido Carvalho, K. (2018). Oncomirs Expression Profiling in Uterine Leiomyosarcoma Cells. International Journal of Molecular Sciences, 19(1), 52. https://doi.org/10.3390/ijms19010052